1. Introduction
Spinal cord injury (SCI) can result in debilitating conditions that lead to the transient or long-term loss of motor and sensory function as well as impacting people’s physical and social activities and well-being [
1,
2]. Worldwide, the incidence of SCI has gradually increased in conjunction with the advancement of human activities. Among developed nations, the incidence rate ranges from 13.1 to 163.4 per million people compared to 13.0 to 220.0 per million people in non-developed countries [
1,
2,
3,
4]. The aetiology varies among different countries, ethnicities, ages and genders, and is not easily defined due to the variable nature of SCIs and disparity in diagnoses; therefore, the true incidence rate is not captured [
2,
3,
4]. SCIs are often divided into traumatic and non-traumatic origins, resulting from physical trauma to the spinal cord or due to non-external factors such as degenerative, infectious or inflammatory causes. The prevailing cause of SCI continues to be motor vehicle accidents, particularly in developed countries, followed by falls, which dominate in non-developed countries [
1,
2,
3]. This links to the bimodal nature of the age profile of patients with traumatic SCI, peaking at 15 and 29 years and again in older years, >65. The nature of the global aging population has meant that the incidence rate of non-traumatic- and fall-associated SCI is growing within the older generation [
2,
3,
4].
Local injury to the spinal cord damages neurons and associated glial cells, disrupts the vasculature and initiates the breakdown of the blood–spinal cord barrier (BSCB) [
5,
6,
7]. Haemorrhaging, oedema and ischaemia are hallmarks of primary injury and lead to a self-propagating progression into secondary injury, which exaggerates and remodels the injury site [
8,
9]. The extent of the primary injury often determines the severity of the SCI. The breakdown of the BSCB induces haemorrhaging, particularly in the grey matter, which disrupts the micro vasculature, leading to a severe reduction in blood flow to the lesion site and systemic hypotension [
10,
11]. This, consequently, leads to progressive tissue ischaemia which further contributes to cell death through mechanisms including loss of ATP, oxygen deprivation, excitotoxicity and ionic imbalance [
8]. In addition, subsequent reperfusion to the damaged site introduces additional oxygen free radicals and immune cells which contribute to the oxidative stress on endothelial cells, further enhancing BSCB breakdown [
8].
Oedema, as previously mentioned, is a key feature in the pathogenesis of SCI and starts to develop within minutes after injury [
7]. It further exacerbates the lesion site, progressing it into secondary injury, resulting in a more severe outcomes for the person [
12]. It occurs shortly following the primary injury, forming from the epicentre of the lesion site and developing out into the surrounding tissue, to form a fluid-filled cavity around 48 h post-injury [
13]. Although the exact biomechanical mechanisms which lead to its formation have yet to be discovered, studies have shown that inhibiting it can greatly improve the prognosis of the person, thereby indicating that the extent of oedema has a subsequent effect on neurological recovery. It is believed that oedema forms as a consequence of dysregulation of water transport regulators within the CNS, predominantly on astrocytes, (cytotoxic oedema) [
14] and the mechanical and biological disruption of the BSCB (vasogenic oedema) [
13].
Astrocytes are the key regulators of water transport within the CNS. They express aquaporin 4 (AQP4), the main water channel protein in the CNS, within their end-feet and processes as well as the membrane of ependymal cells and play a key role in oedema formation [
13,
15,
16]. Aquaporins are a family of transmembrane protein channels which conduct water transcellularly. To date, 13 members have been identified; however, only AQP1, AQP4 and AQP9 have been identified to play a role in oedema following SCI [
13,
16]. AQP1 is mostly expressed on the apical membrane of the choroid plexus epithelial cells, ependymal cells and astrocytes within the CNS, and facilitates the production of cerebrospinal fluid (CSF). AQP9, however, is localised to tanycytes, ependymal cells and astrocytes within the CNS, identified to play a role in energy metabolism [
17]. AQP4 is the main transporter of water within the spinal cord, and its dysregulation is pivotal to oedema formation. Normally, AQP4 is localised to astrocytes and regulates fast water transport, mediated by tightly controlled osmotic or hydrostatic pressures [
13].
To date, no pharmacological interventions have been approved for SCI, and treatment relies heavily on palliative care. However, several potential therapies for oedema have been evaluated in preclinical animal models of SCI. The aim of this study is to systematically review the potential experimental therapies to treat oedema and determine the most potent treatment option for potential translational development.
2. Materials and Methods
2.1. Review Process
A systematic review of the literature was undertaken, following the guidelines set out by the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [
18]. The search was conducted by two independent reviewers (E.M. and Z.A.) and any discrepancies identified were resolved by consensus.
2.2. Literature Search
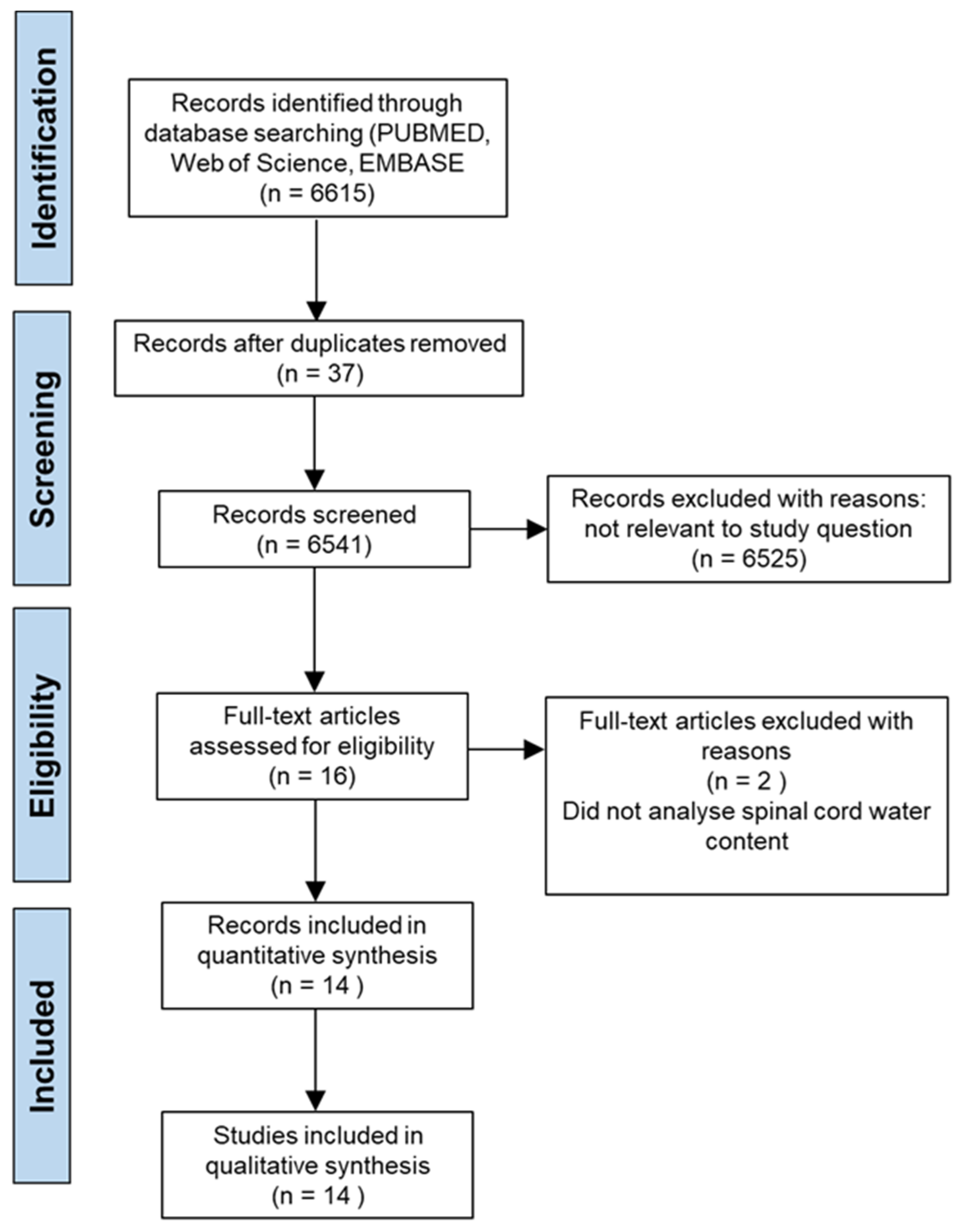
Published papers which analysed experimental treatments for oedema in SCI were identified following an extensive search of the electronic databases PubMed, Web of Science and Ovid Embase. The following common search string was formulated using the Boolean operators ‘(spinal cord injury) OR (oedema) AND (oedema) AND (rat) NOT (mouse) NOT (reviews)’, in order to encapsulate all the required papers for the study. The search was restricted to studies published from 2011 to 28 January 2021 and in the English language only, to limit the number of results, because most advances in SCI research have taken place within the last 10 years. After exporting to Excel, duplicates were removed, and the studies were screened and shortlisted based upon the on title and abstract, abiding to the outlined inclusion and exclusion criteria. The shortlisted studies were independently reviewed again and approved for full text reading, which produced the final selection of studies for subsequent analyses.
2.3. Inclusion and Exclusion Criteria
Studies which were deemed eligible for the systematic review met the following inclusion criteria: (1) studies published in English; (2) published in the last 10 years; (3) articles published in scientific journals; and (4) studies conducted on rats only. We excluded the following: (1) reviews and systematic review articles; (2) studies other than in rats; (3) clinical and in vitro studies; (4) non-English studies; and (5) no full-text articles available. The articles were first assessed based on title and abstract; then, once shortlisted, a full text read was performed by two independent reviewers (E.M. and Z.A.) using an unblinded standardised method to obtain the final selection.
2.4. Risk of Publication Bias
The risk of bias for the selected studies was assessed using the SYRCLE’s risk of bias tool for animal studies, adapted from Cochranes’s risk of bias tool [
19].
2.5. Data Extraction
The required data for the study were extracted from the selected articles, obtained using narrative analysis and a meta-analysis, following a full text read. Using pre-designed tables, the basic study characteristics were obtained using a narrative analysis, which qualitatively extracted data from within the publication text. It included the author, the publication date of the study, the location of the experiment, the rat strain used in the animal model, the level of SCI, the type of SCI induced in the experiment, the therapeutic treatment and regime investigated, follow-up timeframe following SCI, and all outcome measures of the study. Secondly, the method the studies used to determine the level of oedema within the spinal cord was also reported. The percentage of spinal cord tissue water content from each experimental group, stated within each investigation, was also noted.
All studies included the percentage of water content in a sham, control and treatment groups. This information, however, was presented in 64% of the publications as a graph and not an exact report of the raw data they obtained, which meant a close estimate of the actual number had to be calculated. The data extracted from the graphs were checked and confirmed by an independent reviewer (Z.A.), and the data used within this systematic review were as accurate as possible. The relative percentage of oedema attenuated by each treatment was then calculated in comparison to the sham and control groups they had described; the standard deviation was also calculated. The processed data were then categorised based upon the follow-up time following the SCI within the experiments for future meta-analysis. Finally, the summary of the other outcomes of the included studies was also extracted.
2.6. Data Synthesis
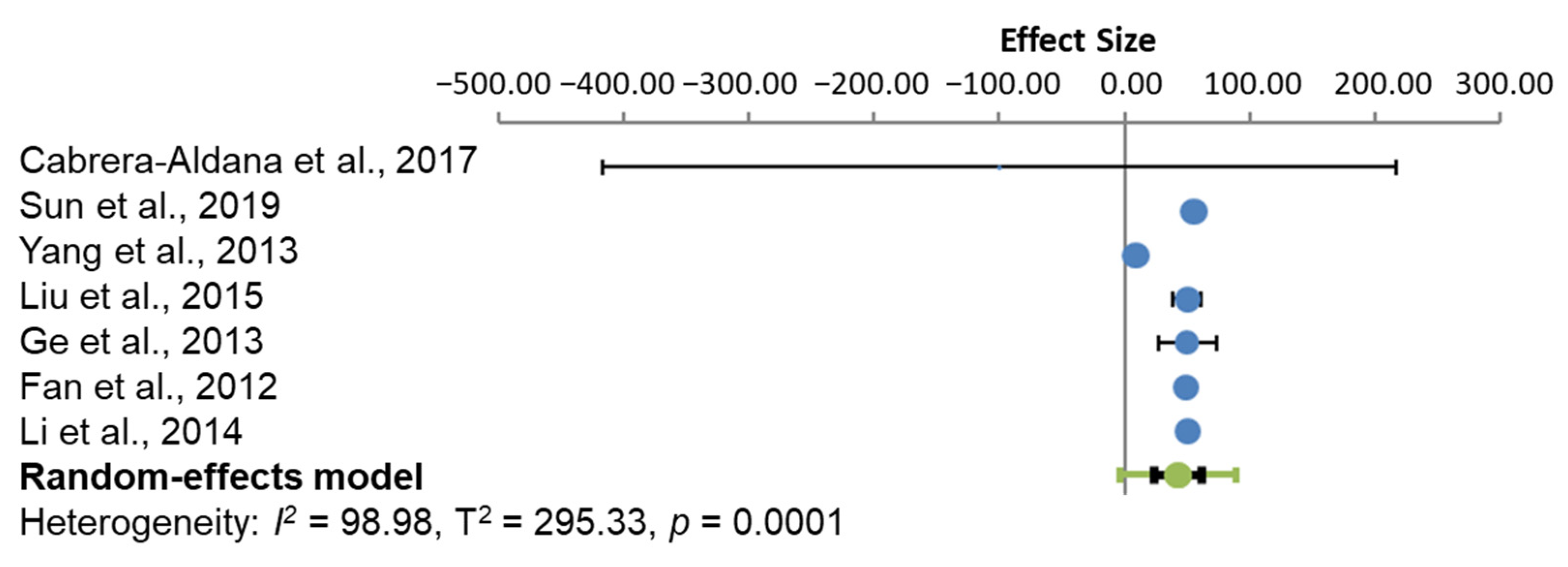
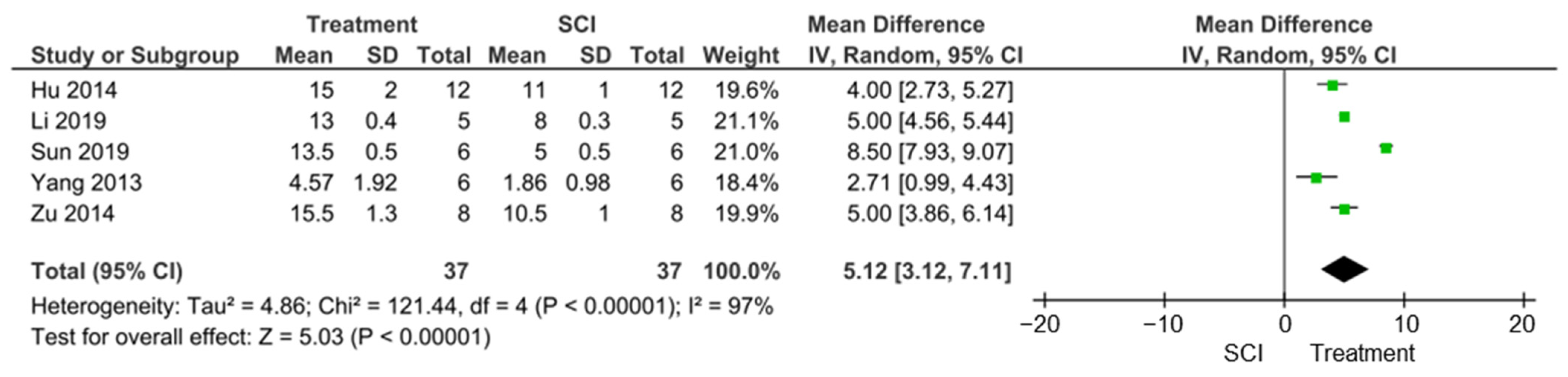
The data were then synthesised by performing a meta-analysis. The primary purpose of the meta-analysis was to produce a more precise estimate of the effect size of the treatments by combining the effect sizes from the included studies. It was performed on each subgroup analysis, 24 and 72 h post-treatment. We also performed a meta-analysis on the improvement of locomotor function after the various treatments.
2.7. Statistical Analysis
Between-study heterogeneity was determined using the inconsistency index I2, which describes the range of possible heterogeneity by the confidence interval. A random-effects models was implemented because heterogeneity was assumed among studies, and it allows for inter-study variability. The effect size was presented as a forest plot, which displays the studies effect size within its corresponding 95% confidence interval. This was carried out using the latest Review Manager 5.4.1 software from Cochrane Informatics & Technology (London, UK), using the random-effects model.
4. Discussion
In this study, we systematically reviewed the published literature within the last 10 years which focused on experimental treatments to alleviate oedema after SCI. The search identified 6541 studies, which were eventually narrowed down to 14 studies after applying our inclusion/exclusion criteria. Following a full text read, the outcome characteristics, including the primary outcome—percentage oedema attenuation—as well as secondary outcomes were extracted and analysed. Studies were grouped into specific follow-up times, 24 h and 72 h, to enable subgroup and meta-analysis. The meta-analysis was conducted to calculate an accurate estimate for the effect size of each treatment. In light of this, the best intervention was the calmodulin (CaM) kinase inhibitor, trifluoperazine (TFP), which blocked the subcellular localisation of AQP4 [
20]. TFP significantly attenuated oedema by 108% at 7 days after SCI, reducing oedema to baseline sham uninjured levels. Other treatments failed to completely resolve oedema, although some did significantly attenuate oedema to various degrees.
Our study is novel and impactful because it analysed all of the proposed experimental treatments to reduce oedema after SCI in the last 10 years. As we discovered, there are various ways of reducing injury oedema; however, many of these rely on the suppression of AQP4 levels. In terms of deducing a direct correlation between the level of oedema attenuation and improvements in functional recovery, this was not possible, because none of the studies reported spinal cord water content at the time of functional improvements. However, the reduction in AQP4 levels produced a negative correlation with improvements in functional recovery. This is based only on three studies, and hence must be interpreted with caution because this is not enough evidence to draw definitive conclusions. Moreover, not all drugs targeted AQP4, and not all drugs suppressed AQP4 levels. For example, the most effective treatment for oedema showed that a temporary relocalisation of AQP4 was beneficial in producing significant long-term sensory and functional recovery [
20].
Spinal cord oedema is a hallmark of SCI and is a major factor in the propagation of secondary injury. It exacerbates primary injury by increasing the intrathecal pressure, causing further damage through the restriction of blood flow, haemorrhaging and BSCB disruption, and thus inciting further cellular necrosis [
5,
6,
7,
8,
9,
10,
11]. Although the exact mechanisms associated with the formation of oedema remain largely unknown, it is strongly associated with an increase in water transport through the AQP4 water channel protein [
13,
15,
16]. Both cytotoxic and vasogenic oedema contribute to the overall excess accumulation of fluid within the spinal cord. For example, ischaemia causes the depletion of ATP, which results in dysfunction of the Na
+ K
+ ATPase co-transporter 1 (NKCC1), altering ionic concertation gradients that are normally maintained between the astrocytes and ECM [
34]. Oedema occurs within hours of SCI, coincides with ischaemia, and is thought to peak at 7 days after injury [
12,
20]. Therefore, early treatment with drugs that suppress oedema might be beneficial in improving outcomes after SCI.
Ischaemia also initiates an increase in damage-associated molecular patterns (DAMPs), which elicit an immune response. The surge of immune cells and increase in inflammatory mediators at the lesion site develops a self-propagating amplification of a pro-inflammatory environment, furthering the exacerbation of secondary injury [
17]. In addition, the infiltration of immune cells and the production of MMPs and associated ECM molecules leads to the continual disruption of the BSCB [
35,
36], increasing the permeability of the surrounding capillaries, which are normally tightly regulated. This allows the extravasation of molecules and fluid into the extracellular space of the spinal cord parenchyma, creating vasogenic oedema. Although AQP4 is thought to play a key role in the development of cytotoxic oedema, it is also implicated in vasogenic oedema, but beneficially. AQP4, in later stages of SCI, aids the removal of fluid from the lesion site, clearing oedema, as well as maintaining physiological homeostasis following the chronic stage of SCI [
13,
37].
Inhibiting oedema soon after SCI has profound benefits to functional recovery. Reducing intrathecal pressure and the development of ischaemia limits the degree of secondary damage and necrosis, facilitating neuroregeneration and the recovery of sensorimotor activity [
12]. The downregulation of AQP4 effectively attenuates oedema, but the duality of AQP4 in SCI means that inhibition should be transient [
13,
20]. Limiting cytotoxic oedema while ensuring AQP4 protein levels are sustained in later stages for elimination of vasogenic oedema and maintaining water homeostasis is important [
20]. Although each study investigated a different therapeutic, mechanistically, nearly all reported the modulation of AQP4. Not only was downregulation or the subcellular relocalisation of AQP4 a hallmark of an effective oedema treatment, the degree of oedema attenuation determined the extent of functional recovery [
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32,
33]. To complement these findings, the majority of studies also measured locomotor activity, glial activity using GFAP immunoreactivity and visualised the lesion site using IHC or other imaging techniques in an attempt to understand the anti-oedemic action of the novel treatments as well as the complex pathophysiology behind the development of oedema. The therapies also came under three key modalities: selective AQP4 inhibition; reduced inflammation at the lesion site leading to the downregulation of AQP4; or surgically, with the aim of manually alleviating oedema. To the best of our knowledge, this is the most comprehensive systematic review of experimental treatments proposed in the last 10 years to reduce spinal cord oedema after SCI.
The most successful strategy to treat SCI-induced oedema was to acutely inhibit AQP4 subcellular localisation by a single injection immediately after SCI. Inhibiting CaM kinase, which is required to translocate AQP4 into the cell membrane, maintained normal AQP4 levels, but completely eliminated oedema by 7 days after SCI [
20]. This occurred by blocking AQP4 relocalisation to the membrane and stopping its normal function temporarily. TFP, a licensed antipsychotic drug used to treat schizophrenia and short-term severe anxiety, was administered to rats at equivalent doses to humans, and significantly attenuated oedema by 56% at 72 h and completely attenuated oedema by 7 days [
20]. The efficacy at 7 days was superior to any other treatment within this systematic review. In addition, acute inhibition of the subcellular localisation of AQP4 with TFP significantly improved electrophysiological, sensory and locomotor function in treated rats, as well as suppressing BSCB breakdown [
20]. Acute inhibition of AQP4 relocalisation was key to this success, because the sustained downregulation of AQP4 using a short hairpin RNA against AQP4, although effective at reducing spinal cord water content in the early phase, caused the experiment to be halted as water content reached high levels by 4 weeks after SCI [
20]. This reiterates the biphasic nature of AQP4 after SCI; hence, inhibitors to temporarily inhibit AQP4 function should be developed.
Although TFP is reported to work by blocking postsynaptic mesolimbic dopaminergic D1 and D2 receptors in the brain, it is also known to be an antagonist of neuron-specific vesicular protein calcycon, alpha-A1A adrenergic receptor and an inhibitor of calmodulin. We showed that calmodulin kinase (CaM) directly binds to AQP4 and that this binding is inhibited by TFP, suggesting a direct role of TFP in lowering spinal cord oedema. However, kinase inhibitors are often non-specific; hence, there may be other off-target effects of TFP which we have not considered. For example, Midostaurin, a small-molecule inhibitor of multiple kinases such as PKC, caused a significant number of kinomic changes in a cervical SCI model [
38]. Nonetheless, we showed that the inhibition of D2 and PKC increased spinal cord water content, demonstrating that the direct and specific inhibition of CaM and PKA is responsible for attenuating SCI-induced oedema after TFP treatment [
20].
Other treatments such as TGN-020 are thought to bind to different residues of AQP4 and selectively inhibit AQP4 [
39]. However, their ability to modulate oedema is weak, reducing oedema by 44% at best [
21,
30]. Bumetanide, a diuretic used in renal failure and oedema, blocked NKCC1 and reduced AQP4 expression, correlating with a 33% reduction in oedema after 48 h, and when used in combination with TGN-020, caused attenuation of oedema by nearly 89% at 72 h after SCI [
30,
39]. Melatonin, a methoxyindole derivative produced from the pineal gland, acts a synchroniser for circadian rhythm, predominantly during the dark phase. It is also known for its antioxidant properties, which can be neuroprotective, predominantly during secondary injury [
35]. Melatonin showed limited effects in reducing oedema at 12 h after SCI, peaking to a 50% reduction in oedema after 24 h, and thereafter decreasing to 33% by 42 h. All of these studies reported significant reductions in AQP4 and GFAP expression after treatment, suggesting that the suppression of AQP4 may reduce glial activity after SCI. In addition, melatonin has a plethora of reported effects within the spinal cord, such as reducing oxidative stress, inflammation, NOS, regulating BSCB repair, inhibiting apoptosis, regulating MMPs, as well as affecting other tissues [
35].
Along with pharmacological treatments, surgical interventions which mainly decompress the extradural elements in SCI can limit local ischaemia and promote neurological recovery [
9,
11]. For example, myelotomy removes haemorrhagic and necrotic tissue from the lesion site by opening the dura and pia maters of the spinal cord. Although easily performed in rat models, its application within 24 h of SCI makes it a dangerous procedure which can potentially lead to further complications. The limited reduction in oedema one week following SCI indicates the limited therapeutic benefit of myelotomy [
25]. A downregulation of AQP4 and AQP9 expression and an improvement in locomotor function was also detected, indicating that the reduction in oedema was correlated to the reduction in AQP4. Although the study presented beneficial findings, the procedure is too risky for the limited efficacy it delivers, and further studies are required before myelotomy can be considered an option for SCI.
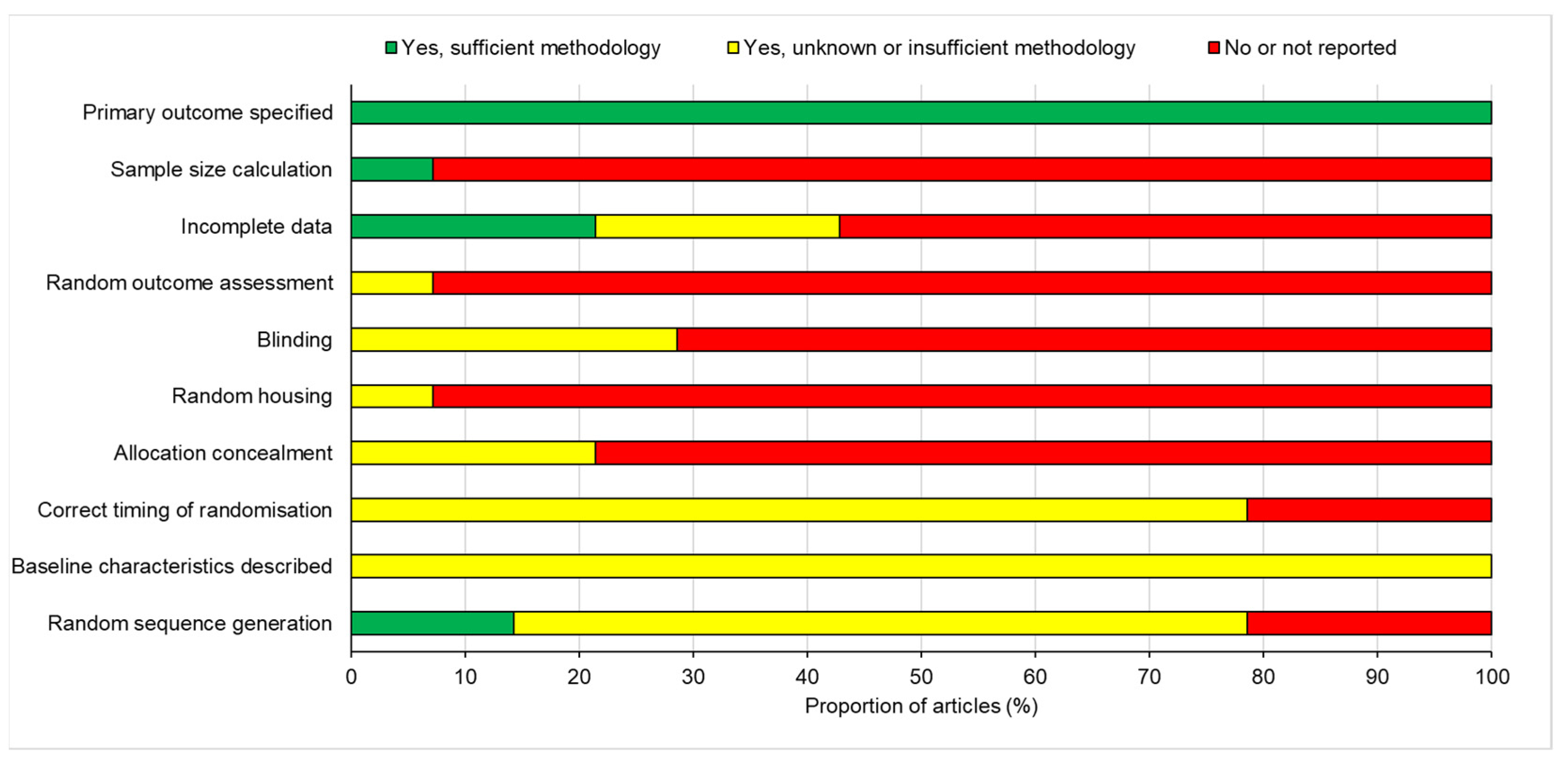
The risk of bias analysis identified significant problems with most of the included studies, ranging from a lack of blinding, inadequate randomisation strategies, and incomplete reporting of excluded animals and prior sample size calculation. The biggest issue was failing to report specific experimental details within the manuscript that relate to risk of bias. Specific guidance on standardised techniques for animal experiments, produced by ARRIVE (Animal Research Reporting of In Vivo Experiments) [
40], can help mitigate against a high risk of bias if adhered to, and are recommended for future use in in vivo experiments of this nature. Despite the relatively high tendency of bias, the results obtained are still deemed reliable and enabled significant comparisons and conclusions to be drawn.
Limitations
One of the main limitations of this study was the variation in time points of the analysis of oedema. However, we were able to group this into subgroups for analysis to within 24 h, 72 h and 7 days after SCI. Another limitation was that there were a variety of locomotor tests used to assess the recovery of function and at time-points ranging from 5 days to 6 weeks. This makes it difficult to compare studies and the potential of reduced oedema and AQP4 levels and their contribution to improvements in locomotor function. Although the majority of animal studies included in this systematic review assessed a thoracic SCI lesion model, 50% of human SCIs affect the cervical region, with C5 being the most common region affected [
8]. Key anatomical differences exist between cervical versus thoracic regions of the spinal cord that will affect the severity of injury and the level of oedema experienced after injury. For example, cervical spinal cords are highly vascularised, more permeable blood–spinal cord barrier, exhibit considerable spontaneous recovery and injury interrupts sympathetic innervation to the major immune organs such as the spleen [
41]. All these differences will affect lesion development and the attenuation of oedema by the treatments reviewed in this study; hence, further studies are required. Another significant limitation is the potential high risk of bias in some studies, due primarily to a lack of clear reporting. This can easily be mitigated by use of the ARRIVE guidelines in reporting animal experiments.