Deciphering the Symbiotic Plant Microbiome: Translating the Most Recent Discoveries on Rhizobia for the Improvement of Agricultural Practices in Metal-Contaminated and High Saline Lands
Abstract
:1. Introduction
2. The Need for Rhizobial Inoculants
3. Plant Growth Promoting Rhizobia in Saline and Harsh Soil
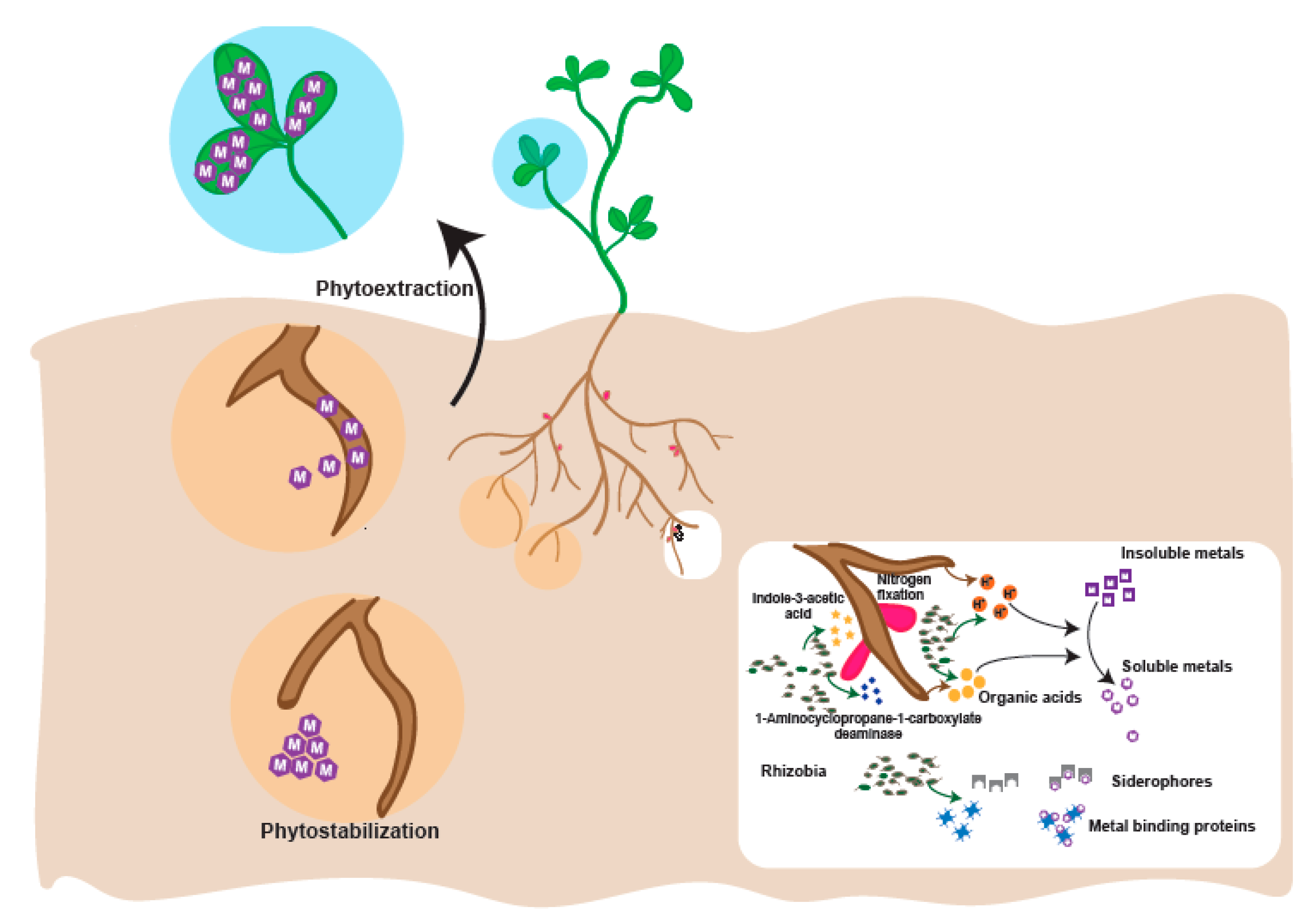
4. Plant Growth Promoting Rhizobia in Heavy Metal Contaminated Soil
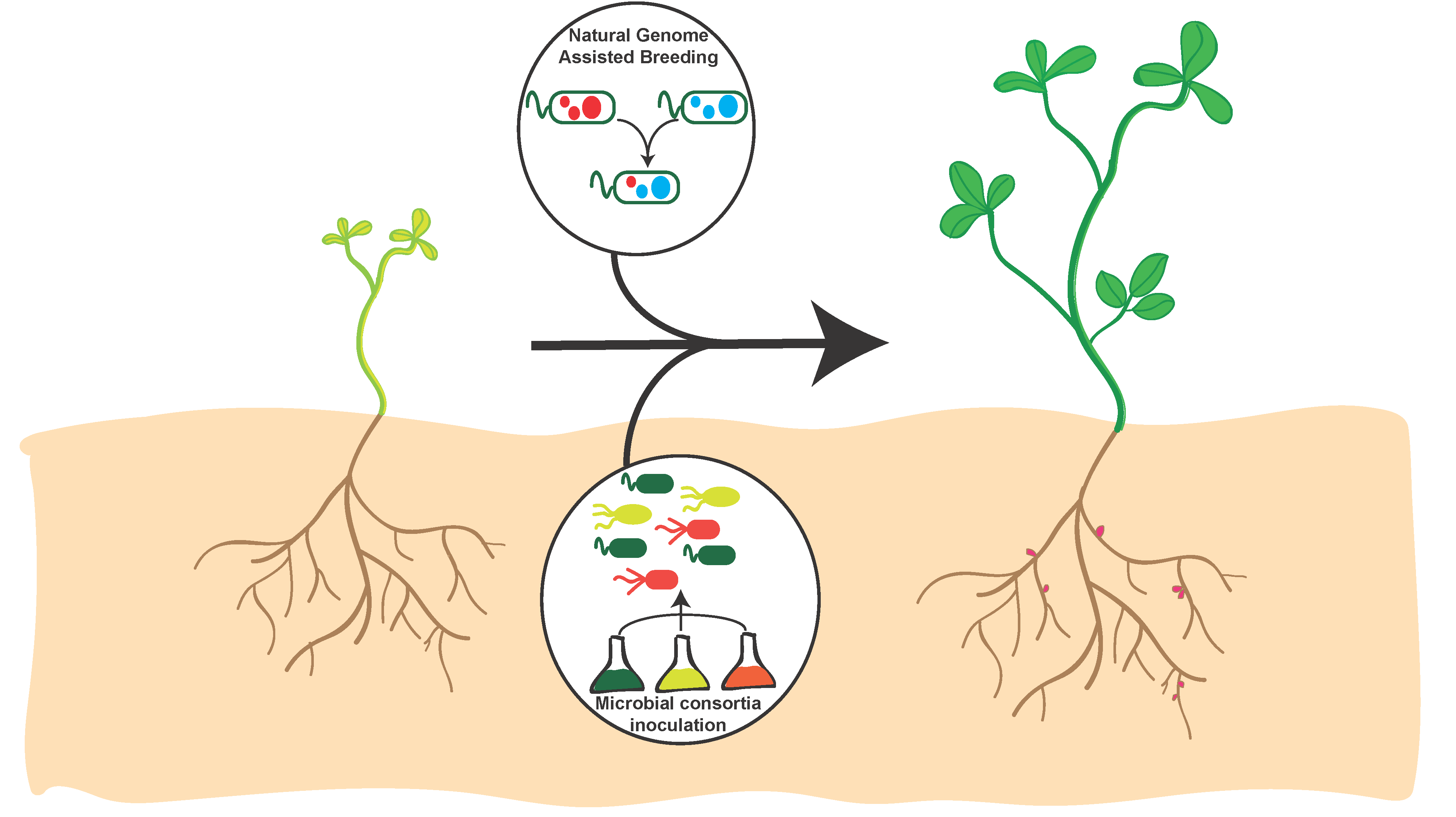
5. Development of Rhizobial Inoculants
6. Inoculant Combinations and Phenomic Strategies for Improving Crop Yield
7. Concluding Remarks
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Vandenkoornhuyse, P.; Quaiser, A.; Duhamel, M.; Le Van, A.; Dufresne, A. The importance of the microbiome of the plant holobiont. New Phytol. 2015, 206, 1196–1206. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Hamady, M.; Fraser-Liggett, C.M.; Knight, R.; Gordon, J.I. The human microbiome project: Exploring the microbial part of ourselves in a changing world. Nature 2007, 449, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Checcucci, A.; DiCenzo, G.C.; Bazzicalupo, M.; Mengoni, A. Trade, diplomacy, and warfare: The quest for elite rhizobia inoculant strains. Front. Microbiol. 2017, 8, 2207. [Google Scholar] [CrossRef] [PubMed]
- Hassani, M.A.; Durán, P.; Hacquard, S. Microbial interactions within the plant holobiont. Microbiome 2018, 6, 58. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Cañizares, C.; Jorrín, B.; Poole, P.S.; Tkacz, A. Understanding the holobiont: The interdependence of plants and their microbiome. Curr. Opin. Microbiol. 2017, 38, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Gans, J.; Wolinsky, M.; Dunbar, J. Computational improvements reveal great bacterial diversity and high metal toxicity in soil. Science 2005, 309, 1387–1390. [Google Scholar] [CrossRef] [PubMed]
- Schloss, P.D.; Handelsman, J. Toward a census of bacteria in soil. PLoS Comput. Biol. 2006, 2, e92. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, A.; Schmid, M.; van Tuinen, D.; Berg, G. Plant-driven selection of microbes. Plant Soil 2009, 321, 235–257. [Google Scholar] [CrossRef]
- Pini, F.; East, A.K.; Appia-Ayme, C.; Tomek, J.; Karunakaran, R.; Mendoza-Suárez, M.; Edwards, A.; Terpolilli, J.J.; Roworth, J.; Downie, J.A. Bacterial biosensors for in vivo spatiotemporal mapping of root secretion. Plant Physiol. 2017, 174, 1289–1306. [Google Scholar] [CrossRef] [PubMed]
- Edwards, J.A.; Santos-Medellín, C.M.; Liechty, Z.S.; Nguyen, B.; Lurie, E.; Eason, S.; Phillips, G.; Sundaresan, V. Compositional shifts in root-associated bacterial and archaeal microbiota track the plant life cycle in field-grown rice. PLoS Biol. 2018, 16, e2003862. [Google Scholar] [CrossRef] [PubMed]
- Thijs, S.; Sillen, W.; Rineau, F.; Weyens, N.; Vangronsveld, J. Towards an enhanced understanding of plant–microbiome interactions to improve phytoremediation: Engineering the metaorganism. Front. Microbiol. 2016, 7, 341. [Google Scholar] [CrossRef] [PubMed]
- Berendsen, R.L.; Pieterse, C.M.; Bakker, P.A. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012, 17, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Mueller, U.G.; Sachs, J.L. Engineering microbiomes to improve plant and animal health. Trends Microbiol. 2015, 23, 606–617. [Google Scholar] [CrossRef] [PubMed]
- Trivedi, P.; Schenk, P.M.; Wallenstein, M.D.; Singh, B.K. Tiny microbes, big yields: Enhancing food crop production with biological solutions. Microb. Biotechnol. 2017, 10, 999–1003. [Google Scholar] [CrossRef] [PubMed]
- Pini, F.; Galardini, M.; Bazzicalupo, M.; Mengoni, A. Plant-bacteria association and symbiosis: Are there common genomic traits in Alphaproteobacteria? Genes (Basel) 2011, 2, 1017–1032. [Google Scholar] [CrossRef]
- Brill, W.J. Genetics of N2-fixing organisms. In The Biology of Nitrogen Fixation; Quispel, A., Ed.; North-Hol land Pub. Co.: Amsterdam, The Netherlands, 1974. [Google Scholar]
- Chi, F.; Shen, S.-H.; Cheng, H.-P.; Jing, Y.-X.; Yanni, Y.G.; Dazzo, F.B. Ascending migration of endophytic rhizobia, from roots to leaves, inside rice plants and assessment of benefits to rice growth physiology. Appl. Environ. Microbiol. 2005, 71, 7271–7278. [Google Scholar] [CrossRef] [PubMed]
- Vitousek, P.M.; Menge, D.N.L.; Reed, S.C.; Cleveland, C.C. Biological nitrogen fixation: Rates, patterns and ecological controls in terrestrial ecosystems. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20130119. [Google Scholar] [CrossRef]
- Checcucci, A.; Perrin, E.; Bazzicalupo, M.; Mengoni, A. Genomic diversity and evolution of rhizobia. In Microbial Diversity in the Genomic Era; Das, S., Dash, H.R., Eds.; Elsevier Science BV: Amsterdam, The Netherlands, 2019; pp. 37–46. [Google Scholar]
- Estrada-de Los Santos, P.; Palmer, M.; Steenkamp, E.T.; Maluk, M.; Beukes, C.; Hirsch, A.M.; James, E.K.; Venter, S.N. Trinickia dabaoshanensis sp. nov., a new name for a lost species. Arch. Microbiol. 2019, 1–4. [Google Scholar] [CrossRef]
- Estrada-de los Santos, P.; Palmer, M.; Chávez-Ramírez, B.; Beukes, C.; Steenkamp, E.; Briscoe, L.; Khan, N.; Maluk, M.; Lafos, M.; Humm, E.; et al. Whole genome analyses suggests that Burkholderia sensu lato contains two additional novel genera (Mycetohabitans gen. nov., and Trinickia gen. nov.): Implications for the Evolution of Diazotrophy and Nodulation in the Burkholderiaceae. Genes 2018, 9, 389. [Google Scholar] [CrossRef]
- Sprent, J.I.; Ardley, J.; James, E.K. Biogeography of nodulated legumes and their nitrogen-fixing symbionts. New Phytol. 2017, 215, 40–56. [Google Scholar] [CrossRef]
- Dwivedi, F.; Upadhyaya, K.L.; Galardini, A.; Bazzicalupo, M.; Biondi, M.; Hungria, E.G.; Kaschuk, M.; Blair, G.; Dwivedi, S.L.; Sahrawat, K.L.; et al. Advances in host plant and rhizobium genomics to enhance symbiotic nitrogen fixation in grain legumes. In Advances in Agronomy; Sparks, D.L., Ed.; Elsevier Science BV: Amsterdam, The Netherlands, 2015; Volume 129, pp. 1–116. [Google Scholar]
- Galloway, J.N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the nitrogen cycle: Recent trends, questions, and potential solutions. Science 2008, 320, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Alori, E.T.; Dare, M.O.; Babalola, O.O. Microbial inoculants for soil quality and plant health. In Sustainable Agriculture Reviews; Lichtfouse, E., Ed.; Springer: Cham, Switzerland, 2017; Volume 22, pp. 281–307. [Google Scholar]
- Carvajal-Muñoz, J.S.; Carmona-Garcia, C.E. Benefits and limitations of biofertilization in agricultural practices. Livest. Res. Rural Dev. 2012, 24, 1–8. [Google Scholar]
- Bhardwaj, D.; Ansari, M.W.; Sahoo, R.K.; Tuteja, N. Biofertilizers function as key player in sustainable agriculture by improving soil fertility, plant tolerance and crop productivity. Microb. Cell Fact. 2014, 13, 66. [Google Scholar] [CrossRef] [PubMed]
- Arora, N.K.; Verma, M.; Mishra, J. Rhizobial bioformulations: Past, present and future. In Rhizotrophs: Plant Growth Promotion To Bioremediation; Mehnaz, S., Ed.; Springer: Singapore, 2017; Volume 2, pp. 69–99. [Google Scholar]
- Tairo, E.V.; Ndakidemi, P.A. Possible benefits of rhizobial inoculation and phosphorus supplementation on nutrition, growth and economic sustainability in grain legumes. Am. J. Res. Commun 2013, 1, 532–556. [Google Scholar]
- Smil, V. Enriching the Earth: Fritz Haber, Carl Bosch and the Transformation of World Food Production; The MIT Press: Cambridge, MA, USA, 2001. [Google Scholar]
- Herridge, D.F.; Peoples, M.B.; Boddey, R.M. Global inputs of biological nitrogen fixation in agricultural systems. Plant Soil 2008, 311, 1–18. [Google Scholar] [CrossRef]
- Rajwar, A.; Sahgal, M.; Johri, B.N. Legume–rhizobia symbiosis and interactions in agroecosystems. In Plant Microbe Symbiosis: Fundamentals and Advances; Arora, N., Ed.; Springer: New Delhi, India, 2013; pp. 233–265. [Google Scholar]
- Deshwal, V.K.; Singh, S.B.; Kumar, P.; Chubey, A. Rhizobia unique plant growth promoting rhizobacteria: A review. Int. J. Life Sci. 2013, 2, 74–86. [Google Scholar]
- Xavier, I.J.; Holloway, G.; Leggett, M. Development of rhizobial inoculant formulations. Crop Manag. 2004, 3. [Google Scholar] [CrossRef]
- Akibode, C.S.; Maredia, M.K. Global and Regional Trends in Production, Trade and Consumption of Food Legume Crops. 2012. Available online: https://ispc.cgiar.org/sites/default/files/images/Legumetrendsv2.pdf (accessed on 30 April 2019).
- Pérez-Montaño, F.; Alías-Villegas, C.; Bellogín, R.A.; Del Cerro, P.; Espuny, M.R.; Jiménez-Guerrero, I.; López-Baena, F.J.; Ollero, F.J.; Cubo, T. Plant growth promotion in cereal and leguminous agricultural important plants: From microorganism capacities to crop production. Microbiol. Res. 2014, 169, 325–336. [Google Scholar] [CrossRef] [Green Version]
- Glick, B.R. Using soil bacteria to facilitate phytoremediation. Biotechnol. Adv. 2010, 28, 367–374. [Google Scholar] [CrossRef]
- Checcucci, A.; Azzarello, E.; Bazzicalupo, M.; Carlo, A.D.; Emiliani, G.; Mancuso, S.; Spini, G.; Viti, C.; Mengoni, A. Role and regulation of ACC deaminase gene in Sinorhizobium melilotr: Is it a symbiotic, rhizospheric or endophytic gene? Front. Genet. 2017, 8, 6. [Google Scholar] [CrossRef]
- Brígido, C.; Nascimento, F.X.; Duan, J.; Glick, B.R.; Oliveira, S. Expression of an exogenous 1-aminocyclopropane-1-carboxylate deaminase gene in Mesorhizobium spp. reduces the negative effects of salt stress in chickpea. FEMS Microbiol. Lett. 2013, 349, 46–53. [Google Scholar] [PubMed]
- Djekoun, A.; Planchon, C. Water status effect on dinitrogen fixation and photosynthesis in soybean. Agron. J. 1991, 83, 316–322. [Google Scholar] [CrossRef]
- Serraj, R.; Sinclair, T.R.; Purcell, L.C. Symbiotic N2 fixation response to drought. J. Exp. Bot. 1999, 50, 143–155. [Google Scholar] [CrossRef] [Green Version]
- Elsheik, E.A.E. Effects of salt on rhizobia and bradyrhizobia: A review. Ann. Appl. Biol. 1998, 132, 507–524. [Google Scholar] [CrossRef]
- Fagorzi, C.; Checcucci, A.; diCenzo, G.; Debiec-Andrzejewska, K.; Dziewit, L.; Pini, F.; Mengoni, A. Harnessing rhizobia to improve heavy-metal phytoremediation by legumes. Genes 2018, 9, 542. [Google Scholar] [CrossRef] [PubMed]
- Chibuike, G.U.; Obiora, S.C. Heavy metal polluted soils: Effect on plants and bioremediation methods. Appl. Environ. Soil Sci. 2014, 2014, 752708. [Google Scholar] [CrossRef]
- Lebrazi, S.; Fikri-Benbrahim, K. Rhizobium-legume symbioses: Heavy metal effects and principal approaches for bioremediation of contaminated soil. In Legumes for Soil Health and Sustainable Management; Meena, R., Das, A., Yadav, G., Lal, R., Eds.; Springer: Singapore, 2018; pp. 205–233. [Google Scholar]
- Mahar, A.; Wang, P.; Ali, A.; Awasthi, M.K.; Lahori, A.H.; Wang, Q.; Li, R.; Zhang, Z. Challenges and opportunities in the phytoremediation of heavy metals contaminated soils: A review. Ecotoxicol. Environ. Saf. 2016, 126, 111–121. [Google Scholar] [CrossRef] [PubMed]
- Bolan, N.S.; Park, J.H.; Robinson, B.; Naidu, R.; Huh, K.Y. Phytostabilization: A green approach to contaminant containment. Adv. Agron. 2011, 112, 145–204. [Google Scholar]
- Kong, Z.; Glick, B.R. The role of plant growth-promoting bacteria in metal phytoremediation. In Advances in Microbial Physiology; Poole, R.K., Ed.; Academic Press: Cambridge, MA, USA, 2017; Volume 71, pp. 97–132. [Google Scholar]
- Sturz, A.V.; Nowak, J. Endophytic communities of rhizobacteria and the strategies required to create yield enhancing associations with crops. Appl. Soil Ecol. 2000, 15, 183–190. [Google Scholar] [CrossRef]
- Theis, K.R.; Dheilly, N.M.; Klassen, J.L.; Brucker, R.M.; John, F.; Bosch, T.C.G.; Cryan, J.F.; Gilbert, S.F.; Goodnight, C.J.; Lloyd, E.A.; et al. Getting the hologenome concept right: An eco—Evolutionary framework for hosts and their microbiomes. Systems 2016, 29, 00028-16. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.H.; Panda, S.K. Alterations in root lipid peroxidation and antioxidative responses in two rice cultivars under NaCl-salinity stress. Acta Physiol. Plant. 2008, 30, 81. [Google Scholar] [CrossRef]
- Bouhmouch, I.; Souad-Mouhsine, B.; Brhada, F.; Aurag, J. Influence of host cultivars and Rhizobium species on the growth and symbiotic performance of Phaseolus vulgaris under salt stress. J. Plant Physiol. 2005, 162, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Zahran, H.H. Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol. Mol. Biol. Rev. 1999, 63, 968–989. [Google Scholar] [PubMed]
- Cordovilla, M.P.; Ligero, F.; Lluch, C. The effect of salinity on N fixation and assimilation in Vicia faba. J. Exp. Bot. 1994, 45, 1483–1488. [Google Scholar] [CrossRef]
- Abbaspoor, A.; Zabihi, H.R.; Movafegh, S.; Asl, M.H.A. The efficiency of plant growth promoting rhizobacteria (PGPR) on yield and yield components of two varieties of wheat in salinity condition. Am. Eurasian J. Sustain. Agric. 2009, 3, 824–828. [Google Scholar]
- Arora, N.K.; Tewari, S.; Singh, S.; Lal, N.; Maheshwari, D.K. PGPR for protection of plant health under saline conditions. In Bacteria in Agrobiology: Stress Management; Maheshwari, D.K., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 239–258. [Google Scholar]
- Vijayan, K.; Chakraborti, S.P.; Ercisli, S.; Ghosh, P.D. NaCl induced morpho-biochemical and anatomical changes in mulberry (Morus spp.). Plant Growth Regul. 2008, 56, 61. [Google Scholar] [CrossRef]
- Drew, M.C.; Hold, P.S.; Picchioni, G.A. Inhibition by NaCl of net CO2 fixation and yield of cucumber. J. Am. Soc. Hortic. Sci. 1990, 115, 472–477. [Google Scholar] [CrossRef]
- Cheeseman, J.M. Mechanisms of salinity tolerance in plants. Plant Physiol. 1988, 87, 57. [Google Scholar] [CrossRef]
- Gupta, B.; Huang, B. Mechanism of salinity tolerance in plants: Physiological, biochemical, and molecular characterization. Int. J. Genom. 2014, 2014, 701596. [Google Scholar] [CrossRef]
- Cordovilla, M.P.; Ocana, A.; Ligero, F.; Lluch, C. Growth stage response to salinity in symbiosis Vicia faba-Rhizobium leguminosarum bv. viciae. Plant Physiol. 1995, 14, 105–111. [Google Scholar]
- Fagg, C.W.; Stewart, J.L. The value of Acacia and Prosopis in arid and semi-arid environments. J. Arid Environ. 1994, 27, 3–25. [Google Scholar] [CrossRef]
- Zhang, X.; Harper, R.; Karsisto, M.; Lindström, K. Diversity of Rhizobium bacteria isolated from the root nodules of leguminous trees. Int. J. Syst. Evol. Microbiol. 1991, 41, 104–113. [Google Scholar] [CrossRef]
- Wahab, A.M.A.; Zahran, H.H. Effects of salt stress on nitrogenase activity and growth of four legumes. Biol. Plant. 1981, 23, 16. [Google Scholar] [CrossRef]
- Bernstein, L.; Francois, L.E.; Clark, R.A. Interactive effects of salinity and fertility on yields of grains and vegetables 1. Agron. J. 1974, 66, 412–421. [Google Scholar] [CrossRef]
- Maas, E.V.; Hoffman, G.J. Crop salt tolerance–current assessment. J. Irrig. Drain. Div. 1977, 103, 115–134. [Google Scholar]
- Delgado, M.J.; Ligero, F.; Lluch, C. Effects of salt stress on growth and nitrogen fixation by pea, faba-bean, common bean and soybean plants. Soil Biol. Biochem. 1994, 26, 371–376. [Google Scholar] [CrossRef]
- Sulieman, S.; Tran, L.-S. Asparagine: An amide of particular distinction in the regulation of symbiotic nitrogen fixation of legumes. Crit. Rev. Biotechnol. 2013, 33, 309–327. [Google Scholar] [CrossRef] [PubMed]
- Ballen, K.G.; Graham, P.H. The role of acid pH in symbiosis between plants and soil organisms. In Handbook of Plant Growth pH as the Master Variable; Rengel, Z., Ed.; Marcel Dekker: New York, NY, USA, 2002; pp. 383–404. [Google Scholar]
- Qureshi, M.I.; Muneer, S.; Bashir, H.; Ahmad, J.; Iqbal, M. Nodule physiology and proteomics of stressed legumes. In Advances in Botanical Research; Kader, J.C., Delseny, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2010; Volume 56, pp. 1–48. ISBN 0065-2296. [Google Scholar]
- Zahran, H.H. Conditions for successful Rhizobium-legume symbiosis in saline environments. Biol. Fertil. Soils 1991, 12, 73–80. [Google Scholar] [CrossRef]
- Tu, J.C. Effect of salinity on Rhizobium-root-hair interaction, nodulation and growth of soybean. Can. J. Plant Sci. 1981, 61, 231–239. [Google Scholar] [CrossRef]
- Zahran, H.H.; Sprent, J.I. Effects of sodium chloride and polyethylene glycol on root-hair infection and nodulation of Vicia faba L. plants by Rhizobium leguminosarum. Planta 1986, 167, 303–309. [Google Scholar] [CrossRef]
- Ikeda, J.; Kobayashi, M.; Takahashi, E. Salt stress increases the respiratory cost of nitrogen fixation. Soil Sci. Plant Nutr. 1992, 38, 51–56. [Google Scholar] [CrossRef]
- Walsh, K.B. Physiology of the legume nodule and its response to stress. Soil Biol. Biochem. 1995, 27, 637–655. [Google Scholar] [CrossRef]
- Hungria, M.; Vargas, M.A.T. Environmental factors affecting N2 fixation in grain legumes in the tropics, with an emphasis on Brazil. Field Crop. Res. 2000, 65, 151–164. [Google Scholar] [CrossRef]
- Delgado, M.J.; Garrido, J.M.; Ligero, F.; Lluch, C. Nitrogen fixation and carbon metabolism by nodules and bacteroids of pea plants under sodium chloride stress. Physiol. Plant. 1993, 89, 824–829. [Google Scholar] [CrossRef]
- Zahran, H.H. Rhizobia from wild legumes: Diversity, taxonomy, ecology, nitrogen fixation and biotechnology. J. Biotechnol. 2001, 91, 143–153. [Google Scholar] [CrossRef]
- Lal, B.; Khanna, S. Selection of salt-tolerant Rhizobium isolates of Acacia nilotica. World J. Microbiol. Biotechnol. 1994, 10, 637–639. [Google Scholar] [CrossRef] [PubMed]
- Zahran, H.H.; Räsänen, L.A.; Karsisto, M.; Lindström, K. Alteration of lipopolysaccharide and protein profiles in SDS-PAGE of rhizobia by osmotic and heat stress. World J. Microbiol. Biotechnol. 1994, 10, 100–105. [Google Scholar] [CrossRef]
- Sakrouhi, I.; Belfquih, M.; Sbabou, L.; Moulin, P.; Bena, G.; Filali-Maltouf, A.; Le Quéré, A. Recovery of symbiotic nitrogen fixing acacia rhizobia from Merzouga desert sand dunes in South East Morocco–Identification of a probable new species of Ensifer adapted to stressed environments. Syst. Appl. Microbiol. 2016, 39, 122–131. [Google Scholar] [CrossRef]
- Gouffi, K.; Pica, N.; Pichereau, V.; Blanco, C. Disaccharides as a new class of nonaccumulated osmoprotectants for Sinorhizobium meliloti. Appl. Environ. Microbiol. 1999, 65, 1491–1500. [Google Scholar]
- Gouffi, K.; Blanco, C. Is the accumulation of osmoprotectant the unique mechanism involved in bacterial osmoprotection? Int. J. Food Microbiol. 2000, 55, 171–174. [Google Scholar] [CrossRef]
- Talibart, R.; Jebbar, M.; Gouesbet, G.; Himdi-Kabbab, S.; Wroblewski, H.; Blanco, C.; Bernard, T. Osmoadaptation in rhizobia: Ectoine-induced salt tolerance. J. Bacteriol. 1994, 176, 5210–5217. [Google Scholar] [CrossRef] [PubMed]
- Arora, N.K.; Singhal, V.; Maheshwari, D.K. Salinity-induced accumulation of poly-β-hydroxybutyrate in rhizobia indicating its role in cell protection. World J. Microbiol. Biotechnol. 2006, 22, 603–606. [Google Scholar] [CrossRef]
- Bree6dveld, M.W.; Zevenhuizen, L.P.T.M.; Zehnder, A.J.B. Osmotically-regulated trehalose accumulation and cyclic β-(1, 2)-glucan excretion by Rhizobium leguminosarum biovar trifolii TA-1. Arch. Microbiol. 1991, 156, 501–506. [Google Scholar] [CrossRef]
- Ghittoni, N.E.; Bueno, M.A. Changes in the cellular content of trehalose in four peanut rhizobia strains cultured under hypersalinity. Symbiosis 1996, 20, 117–127. [Google Scholar]
- Le Rudulier, D.; Bernard, T. Salt tolerance in Rhizobium: A possible role for betaines. FEMS Microbiol. Rev. 1986, 2, 67–72. [Google Scholar] [CrossRef]
- Smith, L.T.; Pocard, J.-A.; Bernard, T.; Le Rudulier, D. Osmotic control of glycine betaine biosynthesis and degradation in Rhizobium meliloti. J. Bacteriol. 1988, 170, 3142–3149. [Google Scholar] [CrossRef] [PubMed]
- Elsheikh, E.A.E.; Wood, M. Salt effects on survival and multiplication of chickpea and soybean rhizobia. Soil Biol. Biochem. 1990, 22, 343–347. [Google Scholar] [CrossRef]
- Fougere, F.; Le Rudulier, D. Uptake of glycine betaine and its analogues by bacteroids of Rhizobium meliloti. Microbiology 1990, 136, 157–163. [Google Scholar] [CrossRef]
- Hickey, E.W.; Hirshfield, I.N. Low-pH-induced effects on patterns of protein synthesis and on internal pH in Escherichia coli and Salmonella typhimurium. Appl. Environ. Microbiol. 1990, 56, 1038–1045. [Google Scholar]
- Shaharoona, B.; Arshad, M.; Zahir, Z.A.; Khalid, A. Performance of Pseudomonas spp. containing ACC-deaminase for improving growth and yield of maize (Zea mays L.) in the presence of nitrogenous fertilizer. Soil Biol. Biochem. 2006, 38, 2971–2975. [Google Scholar] [CrossRef]
- Diagne, N.; Swanson, E.; Pesce, C.; Fall, F.; Diouf, F.; Bakhoum, N.; Fall, D.; Faye, M.N.; Oshone, R.; Simpson, S. Permanent draft genome sequence of Rhizobium sp. strain LCM 4573, a salt-tolerant, nitrogen-fixing bacterium isolated from Senegalese soils. Genome Announc. 2017, 5, e00285-17. [Google Scholar] [CrossRef] [PubMed]
- Ormeño-Orrillo, E.; Gomes, D.F.; del Cerro, P.; Vasconcelos, A.T.R.; Canchaya, C.; Almeida, L.G.P.; Mercante, F.M.; Ollero, F.J.; Megías, M.; Hungria, M. Genome of Rhizobium leucaenae strains CFN 299 T and CPAO 29.8: Searching for genes related to a successful symbiotic performance under stressful conditions. BMC Genom. 2016, 17, 534. [Google Scholar]
- Molina-Sánchez, M.D.; López-Contreras, J.A.; Toro, N.; Fernández-López, M. Genomic characterization of Sinorhizobium meliloti AK21, a wild isolate from the Aral Sea region. Springerplus 2015, 4, 259. [Google Scholar] [CrossRef] [PubMed]
- Le Quéré, A.; Tak, N.; Gehlot, H.S.; Lavire, C.; Meyer, T.; Chapulliot, D.; Rathi, S.; Sakrouhi, I.; Rocha, G.; Rohmer, M. Genomic characterization of Ensifer aridi, a proposed new species of nitrogen-fixing rhizobium recovered from Asian, African and American deserts. BMC Genom. 2017, 18, 85. [Google Scholar]
- DiCenzo, G.C.; Zamani, M.; Checcucci, A.; Fondi, M.; Griffitts, J.S.; Finan, T.M.; Mengoni, A. Multidisciplinary approaches for studying Rhizobium–legume symbioses. Can. J. Microbiol. 2018, 65, 1–33. [Google Scholar] [CrossRef]
- Alexandre, A.N.A.; Laranjo, M.; Oliveira, S. Global transcriptional response to heat shock of the legume symbiont Mesorhizobium loti MAFF303099 comprises extensive gene downregulation. DNA Res. 2013, 21, 195–206. [Google Scholar] [CrossRef]
- Laranjo, M.; Alexandre, A.; Oliveira, S. Global transcriptional response to salt shock of the plant microsymbiont Mesorhizobium loti MAFF303099. Res. Microbiol. 2017, 168, 55–63. [Google Scholar] [CrossRef]
- Draghi, W.O.; Del Papa, M.F.; Hellweg, C.; Watt, S.A.; Watt, T.F.; Barsch, A.; Lozano, M.J.; Lagares Jr, A.; Salas, M.E.; López, J.L. A consolidated analysis of the physiologic and molecular responses induced under acid stress in the legume-symbiont model-soil bacterium Sinorhizobium meliloti. Sci. Rep. 2016, 6, 29278. [Google Scholar] [CrossRef]
- Irar, S.; González, E.M.; Arrese-Igor, C.; Marino, D. A proteomic approach reveals new actors of nodule response to drought in split-root grown pea plants. Physiol. Plant. 2014, 152, 634–645. [Google Scholar] [CrossRef]
- Larrainzar, E.; Wienkoop, S.; Scherling, C.; Kempa, S.; Ladrera, R.; Arrese-Igor, C.; Weckwerth, W.; González, E.M. Carbon metabolism and bacteroid functioning are involved in the regulation of nitrogen fixation in Medicago truncatula under drought and recovery. Mol. Plant-Microbe Interact. 2009, 22, 1565–1576. [Google Scholar] [CrossRef]
- Larrainzar, E.; Wienkoop, S. A proteomic view on the role of legume symbiotic interactions. Front. Plant Sci. 2017, 8, 1267. [Google Scholar] [CrossRef] [PubMed]
- Graham, P.H. Stress tolerance in Rhizobium and Bradyrhizobium, and nodulation under adverse soil conditions. Can. J. Microbiol. 1992, 38, 475–484. [Google Scholar] [CrossRef]
- Da-Silva, J.R.; Alexandre, A.; Brígido, C.; Oliveira, S. Can stress response genes be used to improve the symbiotic performance of rhizobia. AIMS Microbiol. 2017, 3, 365–382. [Google Scholar] [CrossRef] [PubMed]
- Suárez, R.; Wong, A.; Ramírez, M.; Barraza, A.; Orozco, M.C.; Cevallos, M.A.; Lara, M.; Hernández, G.; Iturriaga, G. Improvement of drought tolerance and grain yield in common bean by overexpressing trehalose-6-phosphate synthase in rhizobia. Mol. Plant-Microbe Interact. 2008, 21, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Valera, F.; Martin-Cuadrado, A.B.; Rodriguez-Brito, B.; Pašić, L.; Thingstad, T.F.; Rohwer, F.; Mira, A. Explaining microbial population genomics through phage predation. Nat. Rev. Microbiol. 2009, 7, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Straub, P.F.; Shearer, G.; Reynolds, P.H.S.; Sawyer, S.A.; Kohl, D.H. Effect of disabling bacteroid proline catabolism on the response of soybeans to repeated drought stress. J. Exp. Bot. 1997, 48, 1299–1307. [Google Scholar] [CrossRef] [Green Version]
- Boscari, A.; Van de Sype, G.; Le Rudulier, D.; Mandon, K. Overexpression of BetS, a Sinorhizobium meliloti high-affinity betaine transporter, in bacteroids from Medicago sativa nodules sustains nitrogen fixation during early salt stress adaptation. Mol. Plant-Microbe Interact. 2006, 19, 896–903. [Google Scholar] [CrossRef]
- Nogales, J.; Campos, R.; BenAbdelkhalek, H.; Olivares, J.; Lluch, C.; Sanjuan, J. Rhizobium tropici genes involved in free-living salt tolerance are required for the establishment of efficient nitrogen-fixing symbiosis with Phaseolus vulgaris. Mol. Plant-Microbe Interact. 2002, 15, 225–232. [Google Scholar] [CrossRef]
- Vaishnav, A.; Varma, A.; Tuteja, N.; Choudhary, D.K. PGPR-mediated amelioration of crops under salt stress. In Plant-Microbe Interaction: An Approach to Sustainable Agriculture; Choudhary, D.K., Varma, A., Tuteja, N., Eds.; Springer: Singapore, 2016; pp. 205–226. [Google Scholar]
- Glick, B.R. Modulation of plant ethylene levels by the bacterial enzyme ACC deaminase. FEMS Microbiol. Lett. 2005, 251, 1–7. [Google Scholar] [CrossRef]
- Estevez, J.; Dardanelli, M.S.; Megías, M.; Rodríguez-Navarro, D.N. Symbiotic performance of common bean and soybean co-inoculated with rhizobia and Chryseobacterium balustinum Aur9 under moderate saline conditions. Symbiosis 2009, 49, 29–36. [Google Scholar] [CrossRef]
- Aamir, M.; Aslam, A.; Khan, M.Y.; Jamshaid, M.U.; Ahmad, M.; Asghar, H.N.; Zahir, Z.A. Co-inoculation with Rhizobium and plant growth promoting rhizobacteria (PGPR) for inducing salinity tolerance in mung bean under field condition of semi-arid climate. Asian J. Agric. Biol. 2013, 1, 17–22. [Google Scholar]
- Ahmad, M.; Zahir, Z.A.; Asghar, H.N.; Asghar, M. Inducing salt tolerance in mung bean through coinoculation with rhizobia and plant-growth-promoting rhizobacteria containing 1-aminocyclopropane-1-carboxylate deaminase. Can. J. Microbiol. 2011, 57, 578–589. [Google Scholar] [CrossRef] [PubMed]
- Etesami, H. Bacterial mediated alleviation of heavy metal stress and decreased accumulation of metals in plant tissues: Mechanisms and future prospects. Ecotoxicol. Environ. Saf. 2018, 147, 175–191. [Google Scholar] [CrossRef] [PubMed]
- Noll, M.R. Trace elements in terrestrial environments: Biogeochemistry, bioavailability, and risks of metals. J. Environ. Qual. 2003, 32, 374. [Google Scholar] [CrossRef]
- Ahemad, M.; Khan, M.S. Effect of fungicides on plant growth promoting activities of phosphate solubilizing Pseudomonas putida isolated from mustard (Brassica compestris) rhizosphere. Chemosphere 2012, 86, 945–950. [Google Scholar] [CrossRef] [PubMed]
- Pajuelo, E.; Rodríguez-Llorente, I.D.; Dary, M.; Palomares, A.J. Toxic effects of arsenic on Sinorhizobium-Medicago sativa symbiotic interaction. Environ. Pollut. 2008, 154, 203–211. [Google Scholar] [CrossRef] [PubMed]
- Nagajyoti, P.C.; Lee, K.D.; Sreekanth, T.V.M. Heavy metals, occurrence and toxicity for plants: A review. Environ. Chem. Lett. 2010, 8, 199–216. [Google Scholar] [CrossRef]
- Del Rio, M.; Font, R.; Almela, C.; Velez, D.; Montoro, R.; De Haro Bailon, A. Heavy metals and arsenicuptake by wild vegetation in the Guadiamar river area after the toxic spill of the Aznalcollar mine. J. Biotechnol. 2002, 98, 125–137. [Google Scholar] [CrossRef]
- Adejumo, S.A. Mechanisms of lead and chromium hyperaccumulation and tolerance in plant. Environtropica 2019, 15, 11–29. [Google Scholar]
- Ma, Y.; Oliveira, R.S.; Freitas, H.; Zhang, C. Biochemical and molecular mechanisms of plant-microbe-metal interactions: Relevance for phytoremediation. Front. Plant Sci. 2016, 7, 918. [Google Scholar] [CrossRef]
- Zubair, M.; Shakir, M.; Ali, Q.; Rani, N.; Fatima, N.; Farooq, S.; Shafiq, S.; Kanwal, N.; Ali, F.; Nasir, I.A. Rhizobacteria and phytoremediation of heavy metals. Environ. Technol. Rev. 2016, 5, 112–119. [Google Scholar] [CrossRef]
- Sarwar, N.; Imran, M.; Shaheen, M.R.; Ishaque, W.; Kamran, M.A.; Matloob, A.; Rehim, A.; Hussain, S. Phytoremediation strategies for soils contaminated with heavy metals: Modifications and future perspectives. Chemosphere 2017, 171, 710–721. [Google Scholar] [CrossRef] [PubMed]
- Erakhrumen, A.A. Phytoremediation: An environmentally sound technology for pollution prevention, control and remediation in developing countries. Educ. Res. Rev. 2007, 2, 151–156. [Google Scholar]
- Ghosh, M.; Singh, S.P. A review on phytoremediation of heavy metals and utilization of it’s by products. Asian J. Energy Env. 2005, 6, 18. [Google Scholar]
- He, Z.L.; Yang, X.E.; Stoffella, P.J. Trace elements in agroecosystems and impacts on the environment. J. Trace Elem. Med. Biol. 2005, 19, 125–140. [Google Scholar] [CrossRef]
- Seth, C.S. A review on mechanisms of plant tolerance and role of transgenic plants in environmental clean-up. Bot. Rev. 2012, 78, 32–62. [Google Scholar] [CrossRef]
- Ali, H.; Khan, E.; Sajad, M.A. Phytoremediation of heavy metals—Concepts and applications. Chemosphere 2013, 91, 869–881. [Google Scholar] [CrossRef]
- Glick, B.R. The enhancement of plant growth by free-living bacteria. Can. J. Microbiol. 1995, 41, 109–117. [Google Scholar] [CrossRef]
- Hao, X.; Taghavi, S.; Xie, P.; Orbach, M.J.; Alwathnani, H.A.; Rensing, C.; Wei, G. Phytoremediation of Heavy and Transition Metals Aided by LegumeRhizobia Symbiosis, Int. J. Phytoremediation 2014, 16, 179–202. [Google Scholar] [CrossRef]
- Rajkumar, M.; Sandhya, S.; Prasad, M.N.; Freitas, H. Perspectives of plant-associated microbes in heavy metal phytoremediation. Biotechnol. Adv. 2012, 30, 1562–1574. [Google Scholar] [CrossRef]
- Sessitsch, A.; Kuffner, M.; Kidd, P.; Vangronsveld, J.; Wenzel, W.W.; Fallmann, K.; Puschenreiter, M. The role of plant-associated bacteria in the mobilization and phytoextraction of trace elements in contaminated soils. Soil Biol. Biochem. 2013, 60, 182–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, R.K.; Archana, G. Cadmium minimization in food crops by cadmium resistant plant growth promoting rhizobacteria. Appl. Soil Ecol. 2016, 107, 66–78. [Google Scholar] [CrossRef]
- Ullah, A.; Heng, S.; Munis, M.F.H.; Fahad, S.; Yang, X. Phytoremediation of heavy metals assisted by plant growth promoting (PGP) bacteria: A review. Environ. Exp. Bot. 2015, 117, 28–40. [Google Scholar] [CrossRef]
- Wheaton, G.; Counts, J.; Mukherjee, A.; Kruh, J.; Kelly, R. The confluence of heavy metal biooxidation and heavy metal resistance: Implications for bioleaching by extreme thermoacidophiles. Minerals 2015, 5, 397–451. [Google Scholar] [CrossRef]
- Bruins, M.R.; Kapil, S.; Oehme, F.W. Microbial resistance to metals in the environment. Ecotoxicol. Environ. Saf. 2000, 45, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Atieno, M.; Lesueur, D. Opportunities for improved legume inoculants: Enhanced stress tolerance of rhizobia and benefits to agroecosystems. Symbiosis 2018, 77, 191–205. [Google Scholar] [CrossRef]
- Pal, A.; Paul, A.K. Microbial extracellular polymeric substances: Central elements in heavy metal bioremediation. Indian J. Microbiol. 2008, 48, 49–64. [Google Scholar] [CrossRef] [PubMed]
- Giller, K.E.; Witter, E.; McGrath, S.P. Heavy metals and soil microbes. Soil Biol. Biochem. 2009, 41, 2031–2037. [Google Scholar] [CrossRef]
- Martensson, A.; Witter, E. Influence of various soil amendments on nitrogen-fixing soil microorganisms in a long-term field experiment, with special reference to sewage sludge. Soil Biol. Biochem. 1990, 22, 977–982. [Google Scholar] [CrossRef]
- Obbard, J.P.; Jones, K.C. The effect of heavy metals on dinitrogen fixation by Rhizobium-white clover in a range of long-term sewage sludge amended and metal-contaminated soils. Environ. Pollut. 1993, 79, 105–112. [Google Scholar] [CrossRef]
- Gulz, P.A.; Gupta, S.-K.; Schulin, R. Arsenic accumulation of common plants from contaminated soils. Plant Soil 2005, 272, 337–347. [Google Scholar] [CrossRef]
- Fatnassi, I.C.; Chiboub, M.; Saadani, O.; Jebara, M.; Jebara, S.H. Phytostabilization of moderate copper contaminated soils using co-inoculation of Vicia faba with plant growth promoting bacteria. J. Basic Microbiol. 2015, 55, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Delgadillo, J.; Lafuente, A.; Doukkali, B.; Redondo-Gómez, S.; Mateos-Naranjo, E.; Caviedes, M.A.; Pajuelo, E.; Rodríguez-Llorente, I.D. Improving legume nodulation and Cu rhizostabilization using a genetically modified rhizobia. Environ. Technol. 2015, 36, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Pajuelo, E.; Pérez-Palacios, P.; Romero-Aguilar, A.; Delgadillo, J.; Doukkali, B.; Rodríguez-Llorente, I.D.; Caviedes, M.A. Improving legume–rhizobium symbiosis for copper phytostabilization through genetic manipulation of both symbionts. In Biological Nitrogen Fixation and Beneficial Plant-Microbe Interaction; González-Andrés, F., James, F., Eds.; Springer: Cham, Switzerland, 2016; pp. 183–193. [Google Scholar]
- Drewniak, L.; Sklodowska, A. Arsenic-transforming microbes and their role in biomining processes. Environ. Sci. Pollut. Res. 2013, 20, 7728–7739. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pinter, M.I.F.; Salomon, M.V.; Berli, F.; Gil, R.; Bottini, R.; Piccoli, P. Plant growth promoting rhizobacteria alleviate stress by AsIII in grapevine. Agric. Ecosyst. Environ. 2018, 267, 100–108. [Google Scholar] [CrossRef]
- Bianucci, E.; Godoy, A.; Furlan, A.; Peralta, J.M.; Hernández, L.E.; Carpena-Ruiz, R.O.; Castro, S. Arsenic toxicity in soybean alleviated by a symbiotic species of Bradyrhizobium. Symbiosis 2018, 74, 167–176. [Google Scholar] [CrossRef]
- Fatnassi, I.C.; Chiboub, M.; Saadani, O.; Jebara, M.; Jebara, S.H. Impact of dual inoculation with Rhizobium and PGPR on growth and antioxidant status of Vicia faba L. under copper stress. C. R. Biol. 2015, 338, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Dary, M.; Chamber-Pérez, M.A.; Palomares, A.J.; Pajuelo, E. “In situ” phytostabilisation of heavy metal polluted soils using Lupinus luteus inoculated with metal resistant plant-growth promoting rhizobacteria. J. Hazard. Mater. 2010, 177, 323–330. [Google Scholar] [CrossRef]
- Kamaludeen, S.P.B.; Ramasamy, K. Rhizoremediation of metals: Harnessing microbial communities. Indian J. Microbiol. 2008, 48, 80–88. [Google Scholar] [CrossRef]
- Singh, N.K.; Rai, U.N.; Tewari, A.; Singh, M. Metal accumulation and growth response in Vigna radiata L. inoculated with chromate tolerant rhizobacteria and grown on tannery sludge amended soil. Bull. Environ. Contam. Toxicol. 2010, 84, 118–124. [Google Scholar] [CrossRef]
- Armendariz, A.L.; Talano, M.A.; Nicotra, M.F.O.; Escudero, L.; Breser, M.L.; Porporatto, C.; Agostini, E. Impact of double inoculation with Bradyrhizobium japonicum E109 and Azospirillum brasilense Az39 on soybean plants grown under arsenic stress. Plant Physiol. Biochem. 2019, 138, 26–35. [Google Scholar] [CrossRef]
- Spaepen, S.; Vanderleyden, J.; Remans, R. Indole-3-acetic acid in microbial and microorganism-plant signaling. FEMS Microbiol. Rev. 2007, 31, 425–448. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, P.; Dubey, R.S. Lead toxicity in plants. Braz. J. Plant Physiol. 2005, 17, 35–52. [Google Scholar] [CrossRef] [Green Version]
- Di Gregorio, S.; Barbafieri, M.; Lampis, S.; Sanangelantoni, A.M.; Tassi, E.; Vallini, G. Combined application of Triton X-100 and Sinorhizobium sp. Pb002 inoculum for the improvement of lead phytoextraction by Brassica juncea in EDTA amended soil. Chemosphere 2006, 63, 293–299. [Google Scholar] [CrossRef] [PubMed]
- Zhao, F.J.; Lombi, E.; McGrath, S.P. Assessing the potential for zinc and cadmium phytoremediation with the hyperaccumulator Thlaspi caerulescens. Plant Soil 2003, 249, 37–43. [Google Scholar] [CrossRef]
- Seregin, I.V.; Kozhevnikova, A.D. Physiological role of nickel and its toxic effects on higher plants. Russ. J. Plant Physiol. 2006, 53, 257–277. [Google Scholar] [CrossRef]
- Wani, P.A.; Khan, M.S.; Zaidi, A. Effect of metal-tolerant plant growth-promoting Rhizobium on the performance of pea grown in metal-amended soil. Arch. Environ. Contam. Toxicol. 2008, 55, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Wani, P.A.; Khan, M.S.; Zaidi, A. Effect of metal tolerant plant growth promoting Bradyrhizobium sp. (vigna) on growth, symbiosis, seed yield and metal uptake by greengram plants. Chemosphere 2007, 70, 36–45. [Google Scholar] [CrossRef]
- Hasanuzzaman, M.; Prasad, M.N.V.; Fujita, M. Cadmium Toxicity and Tolerance in Plants: From Physiology to Remediation; Academic Press: Cambridge, MA, USA, 2018; ISBN 0128148659. [Google Scholar]
- Yu, R.; Tang, Y.; Liu, C.; Du, X.; Miao, C.; Shi, G. Comparative transcriptomic analysis reveals the roles of ROS scavenging genes in response to cadmium in two pak choi cultivars. Sci. Rep. 2017, 7, 9217. [Google Scholar] [CrossRef]
- Guo, J.; Chi, J. Effect of Cd-tolerant plant growth-promoting Rhizobium on plant growth and Cd uptake by Lolium multiflorum Lam. and Glycine max (L.) Merr. in Cd-contaminated soil. Plant Soil 2014, 375, 205–214. [Google Scholar] [CrossRef]
- Porter, S.S.; Chang, P.L.; Conow, C.A.; Dunham, J.P.; Friesen, M.L. Association mapping reveals novel serpentine adaptation gene clusters in a population of symbiotic Mesorhizobium. ISME J. 2017, 11, 248–262. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Jiao, S.; Gao, E.; Song, X.; Li, Z.; Hao, X.; Rensing, C.; Wei, G.; Lu, C.M. Transcriptome response to heavy metals in Sinorhizobium meliloti CCNWSX0020 reveals new metal eesistance determinants that also promote bioremediation by Medicago lupulina in metal-contaminated soil. Appl. Environ. Microbiol. 2017, 83, 1244–1261. [Google Scholar] [CrossRef] [PubMed]
- Hao, X.; Xie, P.; Zhu, Y.-G.; Taghavi, S.; Wei, G.; Rensing, C. Copper tolerance mechanisms of Mesorhizobium amorphae and its role in aiding phytostabilization by Robinia pseudoacacia in copper contaminated soil. Environ. Sci. Technol. 2015, 49, 2328–2340. [Google Scholar] [CrossRef] [PubMed]
- Hussain, M.B.; Zahir, Z.A.; Asghar, H.N.; Asgher, M. Can catalase and exopolysaccharides producing rhizobia ameliorate drought stress in wheat? Int. J. Agric. Biol. 2014, 16, 3–13. [Google Scholar]
- Baldani, V.L.D.; Alvarez, M.A.B.; Baldani, J.I.; Döbereiner, J. Establishment of inoculated Azospirillum spp. in the rhizosphere and in roots of field grown wheat and sorghum. Plant Soil 1986, 90, 35–46. [Google Scholar] [CrossRef]
- Fasciglione, G.; Casanovas, E.M.; Quillehauquy, V.; Yommi, A.K.; Goñi, M.G.; Roura, S.I.; Barassi, C.A. Azospirillum inoculation effects on growth, product quality and storage life of lettuce plants grown under salt stress. Sci. Hortic. Amst. 2015, 195, 154–162. [Google Scholar] [CrossRef]
- Staudinger, C.; Mehmeti-Tershani, V.; Gil-Quintana, E.; Gonzalez, E.M.; Hofhansl, F.; Bachmann, G.; Wienkoop, S. Evidence for a rhizobia-induced drought stress response strategy in Medicago truncatula. J. Proteom. 2016, 136, 202–213. [Google Scholar] [CrossRef]
- Bordeleau, L.M.; Antoun, H.; Lachance, R.-A. Effets des souches de Rhizobium meliloti et des coupes successives de la luzerne (Medicago sativa) sur la fixation symbiotique d’azote. Can. J. Plant Sci. 1977, 57, 433–439. [Google Scholar] [CrossRef]
- Bertrand, A.; Dhont, C.; Bipfubusa, M.; Chalifour, F.-P.; Drouin, P.; Beauchamp, C.J. Improving salt stress responses of the symbiosis in alfalfa using salt-tolerant cultivar and rhizobial strain. Appl. Soil Ecol. 2015, 87, 108–117. [Google Scholar] [CrossRef]
- Bromfield, E.S.P.; Wheatcroft, R.; Barran, L.R. Medium for direct isolation of Rhizobium meliloti from soils. Soil Biol. Biochem. 1994, 26, 423–428. [Google Scholar] [CrossRef]
- Noel, K.D.; Vandenbosch, K.A.; Kulpaca, B. Mutations in Rhizobium phaseoli that lead to arrested development of infection threads. J. Bacteriol. 1986, 168, 1392–1401. [Google Scholar] [CrossRef] [Green Version]
- Smith, R.S. Legume inoculant formulation and application. Can. J. Microbiol. 1992, 38, 485–492. [Google Scholar] [CrossRef]
- Nobbe, F.; Hiltner, L. Inoculation of the soil for cultivating leguminous plants. US Pat. 1896, 570, 813. [Google Scholar]
- Brockwell, J. Inoculation methods for field experimenters and farmers. In Nitrogen Fixation in Legumes; Vincent, J.M., Ed.; Academic Press: New York, NY, USA, 1982; pp. 211–227. [Google Scholar]
- Dommergues, Y.R.; Diem, H.G.; Divies, C. Polyacrylamide-entrapped Rhizobium as an inoculant for legumes. Appl. Environ. Microbiol. 1979, 37, 779–781. [Google Scholar] [PubMed]
- Jung, G.; Mugnier, J.; Diem, H.G.; Dommergues, Y.R. Polymer-entrapped Rhizobium as an inoculant for legumes. Plant Soil 1982, 65, 219–231. [Google Scholar] [CrossRef]
- Paau, A.S.; Graham, L.L.; Bennett, M. Progress in formulation research for PGPR and biocontrol inoculants. Bull. OILB SROP 1991, 14, 8. Available online: http://agris.fao.org/agris-search/search.do?recordID=FR9202869 (accessed on 30 April 2019).
- Smith, R.S. Inoculant formulations and applications to meet changing needs. In Nitrogen Fixation: Fundamentals and Applications; Tikhonovich, I.A., Provorov, N.A., Romanov, V.I., Newton, W.E., Eds.; Springer: Dordrecht, The Netherlands, 1995; Volume 27, pp. 653–657. [Google Scholar]
- Singleton, P.; Keyser, H.; Sande, E. Development and evaluation of liquid inoculants. In Proceedings of the Inoculants and Nitrogen Fixation of Legumes in Vietnam, Hanoi, Vietnam, 17–18 February 2001; pp. 52–66. [Google Scholar]
- Rohr, T. Rheological Study of the Mixture Carboxymethylcellulose/Starch and Its Use As a Vehicle for the Bacterial Inoculation. Ph.D. Thesis, Rural Federal University of Rio de Janeiro, Rio De Janeiro, Brazil, 2007. [Google Scholar]
- Manikandan, R.; Saravanakumar, D.; Rajendran, L.; Raguchander, T.; Samiyappan, R. Standardization of liquid formulation of Pseudomonas fluorescens Pf1 for its efficacy against Fusarium wilt of tomato. Biol. Control. 2010, 54, 83–89. [Google Scholar] [CrossRef]
- Ferreira, E.M.; Castro, I.V. e Residues of the cork industry as carriers for the production of legumes inoculants. Silva. Lusit. 2005, 13, 159–167. [Google Scholar]
- Kaljeet, S.; Keyeo, F.; Amir, H. Temperature on survivability of rhizobial inoculant. Asian J. Plant Sci. 2011, 10, 331–337. [Google Scholar]
- Bashan, Y.; de-Bashan, L.E.; Prabhu, S.R.; Hernandez, J.-P. Advances in plant growth-promoting bacterial inoculant technology: Formulations and practical perspectives (1998–2013). Plant Soil 2014, 378, 1–33. [Google Scholar] [CrossRef]
- Albareda, M.; Rodríguez-Navarro, D.N.; Camacho, M.; Temprano, F.J. Alternatives to peat as a carrier for rhizobia inoculants: Solid and liquid formulations. Soil Biol. Biochem. 2008, 40, 2771–2779. [Google Scholar] [CrossRef]
- Rivera, D.; Obando, M.; Barbosa, H.; Rojas Tapias, D.; Bonilla Buitrago, R. Evaluation of polymers for the liquid rhizobial formulation and their influence in the Rhizobium-Cowpea interaction. Univ. Sci. 2014, 19, 265–275. [Google Scholar] [CrossRef]
- Brahmaprakash, G.P.; Sahu, P.K. Biofertilizers for sustainability. J. Indian Inst. Sci. 2012, 92, 37–62. [Google Scholar]
- Vandergheynst, J.; Scher, H.; Guo, H.-Y.; Schultz, D. Water-in-oil emulsions that improve the storage and delivery of the biolarvacide Lagenidium giganteum. BioControl 2007, 52, 207–229. [Google Scholar] [CrossRef]
- Nápoles, M.C.; Carrió, E.G.; Montero, F.; Ferreira, A.; Rossi, A. Role of Bradyrhizobium japonicum induced by genistein on soybean stressed by water deficit. Spanish J. Agric. Res. 2009, 7, 665–671. [Google Scholar] [CrossRef]
- Tank, N.; Saraf, M. Salinity-resistant plant growth promoting rhizobacteria ameliorates sodium chloride stress on tomato plants. J. Plant Interact. 2010, 5, 51–58. [Google Scholar] [CrossRef]
- Kudoyarova, G.R.; Arkhipova, T.N.; Melent’ev, A.I. Role of bacterial phytohormones in plant growth regulation and their development. In Bacterial Metabolites in Sustainable Agroecosystem; Maeshwari, D., Ed.; Springer: Cham, Switzerland, 2015; pp. 69–86. [Google Scholar]
- Giller, K.E.; Murwira, M.S.; Dhliwayo, D.K.C.; Mafongoya, P.L.; Mpepereki, S. Soyabeans and sustainable agriculture in southern Africa. Int. J. Agric. Sustain. 2011, 9, 50–58. [Google Scholar] [CrossRef]
- Lupwayi, N.Z.; Clayton, G.W.; Rice, W.A. Rhizobial inoculants for legume crops. J. Crop. Improv. 2006, 15, 289–321. [Google Scholar] [CrossRef]
- Thilakarathna, M.S.; Raizada, M.N. A meta-analysis of the effectiveness of diverse rhizobia inoculants on soybean traits under field conditions. Soil Biol. Biochem. 2017, 105, 177–196. [Google Scholar] [CrossRef]
- Friesen, M.L. Widespread fitness alignment in the legume—Rhizobium symbiosis. New Phytol. 2012, 194, 1096–1111. [Google Scholar] [CrossRef] [PubMed]
- Checcucci, A.; Azzarello, E.; Bazzicalupo, M.; Galardini, M.; Lagomarsino, A.; Mancuso, S.; Marti, L.; Marzano, M.C.; Mocali, S.; Squartini, A.; et al. Mixed nodule infection in Sinorhizobium meliloti–Medicago sativa symbiosis suggest the presence of cheating behavior. Front. Plant Sci. 2016, 7, 835. [Google Scholar] [CrossRef] [PubMed]
- West, S.A.; Kiers, E.T.; Simms, E.L.; Denison, R.F. Sanctions and mutualism stability: Why do rhizobia fix nitrogen? Proc. R. Soc. B Biol. Sci. 2002, 269, 685–694. [Google Scholar] [CrossRef] [PubMed]
- Westhoek, A.; Field, E.; Rehling, F.; Mulley, G.; Webb, I.; Poole, P.S.; Turnbull, L.A. Policing the legume-Rhizobium symbiosis: A critical test of partner choice. Sci. Rep. 2017, 7, 1419. [Google Scholar] [CrossRef]
- Kiers, E.T.; Denison, R.F. Sanctions, cooperation, and the stability of plant-rhizosphere mutualisms. Annu. Rev. Ecol. Evol. Syst. 2008, 39, 215–236. [Google Scholar] [CrossRef]
- MacLean, A.M.; Finan, T.M.; Sadowsky, M.J. Genomes of the symbiotic nitrogen-fixing bacteria of legumes. Plant Physiol. 2007, 144, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Checcucci, A.; DiCenzo, G.C.; Ghini, V.; Bazzicalupo, M.; Beker, A.; Decorosi, F.; Dohlemann, J.; Fagorzi, C.; Finan, T.M.; Fondi, M.; et al. Creation and Characterization of a Genomically Hybrid Strain in the Nitrogen-Fixing Symbiotic Bacterium Sinorhizobium meliloti. ACS Synth. Biol. 2018, 7, 296483. [Google Scholar] [CrossRef]
- Brewin, N.J.; Wood, E.A.; Young, J.P.W. Contribution of the symbiotic plasmid to the competitiveness of Rhizobium leguminosarum. Microbiology 1983, 129, 2973–2977. [Google Scholar] [CrossRef]
- Brewin, N.J.; DeJong, T.M.; Phillips, D.A.; Johnston, A.W.B. Co-transfer of determinants for hydrogenase activity and nodulation ability in Rhizobium leguminosarum. Nature 1980, 288, 77–79. [Google Scholar] [CrossRef]
- DeJong, T.M.; Brewin, N.J.; Phillips, D.A. Effects of plasmid content in Rhizobium leguminosarum on pea nodule activity and plant growth. Microbiology 1981, 124, 1–7. [Google Scholar] [CrossRef]
- DiCenzo, G.C.; Checcucci, A.; Bazzicalupo, M.; Mengoni, A.; Viti, C.; Dziewit, L.; Finan, T.M.; Galardini, M.; Fondi, M. Metabolic modelling reveals the specialization of secondary replicons for niche adaptation in Sinorhizobium meliloti. Nat. Commun. 2016, 7, 12219. [Google Scholar] [CrossRef]
- Checcucci, A.; Bazzicalupo, M.; Mengoni, A. Exploiting Nitrogen-Fixing Rhizobial Symbionts Genetic Resources for Improving Phytoremediation of Contaminated Soils. In Enhancing Cleanup of Environmental Pollutants; Anjum, N., Tuteja, N., Eds.; Spinger: Cham, Switzerland, 2017; Volume 1, pp. 275–288. [Google Scholar]
- Bradáčová, K.; Florea, A.S.; Bar-Tal, A.; Minz, D.; Yermiyahu, U.; Shawahna, R.; Kraut-Cohen, J.; Zolti, A.; Erel, R.; Dietel, K. Microbial consortia versus single-strain inoculants: An advantage in PGPM-assisted tomato production? Agronomy 2019, 9, 105. [Google Scholar] [CrossRef]
- Bashan, Y. Inoculants of plant growth-promoting bacteria for use in agriculture. Biotechnol. Adv. 1998, 16, 729–770. [Google Scholar] [CrossRef]
- Nadeem, S.M.; Naveed, M.; Zahir, Z.A.; Asghar, H.N. Plant–microbe interactions for sustainable agriculture: Fundamentals and recent advances. In Plant Microbe Symbiosis: Fundamentals and Advances; Arora, N., Ed.; Springer: New Dehli, India, 2013; pp. 51–103. [Google Scholar]
- Sekar, J.; Raj, R.; Prabavathy, V.R. Microbial consortial products for sustainable agriculture: Commercialization and regulatory issues in india. In Agriculturally Importantmicroorganisms; Sarma, B., Keswani, C., Eds.; Springer: Singapore, 2016; pp. 107–132. [Google Scholar]
- Abd-Alla, M.H.; El-Enany, A.-W.E.; Nafady, N.A.; Khalaf, D.M.; Morsy, F.M. Synergistic interaction of Rhizobium leguminosarum bv. viciae and arbuscular mycorrhizal fungi as a plant growth promoting biofertilizers for faba bean (Vicia faba L.) in alkaline soil. Microbiol. Res. 2014, 169, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Tajini, F.; Trabelsi, M.; Drevon, J.-J. Co-inoculation with Glomus intraradices and Rhizobium tropici CIAT899 increases P use efficiency for N 2 fixation in the common bean (Phaseolus vulgaris L.) under P deficiency in hydroaeroponic culture. Symbiosis 2011, 53, 123. [Google Scholar] [CrossRef]
- Zhu, R.-F.; Tang, F.; Liu, J.; Liu, F.-Q.; Deng, X.-Y.; Chen, J.-S. Co-inoculation of arbuscular mycorrhizae and nitrogen fixing bacteria enhance alfalfa yield under saline conditions. Pakistan J. Bot. 2016, 48, 763–769. [Google Scholar]
- Meng, L.; Zhang, A.; Wang, F.; Han, X.; Wang, D.; Li, S. Arbuscular mycorrhizal fungi and Rhizobium facilitate nitrogen uptake and transfer in soybean/maize intercropping system. Front. Plant Sci. 2015, 6, 339. [Google Scholar] [CrossRef] [PubMed]
| Strain | Isolation Conditions | Crop | Effect/Action Mechanism | Reference | |
|---|---|---|---|---|---|
| Site | Metal Contamination | ||||
| Bradyrhizobium diazoefficiensUSDA110 | Ref. strain | As | Soybean | Limits metalloid translocation and accumulation in edible parts of the legume | [151] |
| Bradyrhizobium sp. Per 3.61 | Nodules of soybeans | As | |||
| A. brasilense Az39 | As | Soybean | Enhances growth of the plant and phytostabilization of As when co-inoculated | [156] | |
| B. japonicum E109 | As | ||||
| Sinorhizobium sp. Pb002 | Rhizospere of Brassica juncea | Cd | Brassica juncea | Increases plant survival and lead uptake | [159] |
| Bradyrhizobium sp. (vigna) RM8 | Nodules of greengram | Ni, Zn | Greengram | Increases the number of nodules on the plant, as well as IAA and siderophore production | [163] |
| Rhizobium sp. RP5 | |||||
| Bradyrhizobium sp. | Nodules of Glycine max | Cd | Glycine max | Increases mineral nutrient uptake (Fe) and reduces cadmium accumulation in the plant | [166] |
| Rhizobium leguminosarum (LR-30), Mesorhizobium ciceri (CR.30 and CR-39) and Rhizobium phaseoli (MR-2) | Lens culinaris L., Vigna radiata L., Cicer aretinum L. | Wheat (Triticum aestivum) | Improves plant growth, biomass, and drought stress through production of catalase, IAA, and exopolysaccharides under PEG-6000 simulated drought conditions | [170] | |
| Strain | Isolation conditions | Crop | Effect/Mechanism of action | Reference | |
| Azospirillum Sp245 | Surface-sterilized wheat roots of (Brazil) [171] | Lattuce (L.sativa L.) | Promotes aerial biomass, higher ascorbic acid content accompanied by a lower oxidation rate, better overall visual quality due to higher chlorophyll content, hue, Chroma, L and lower browning intensity | [172] | |
| Sinorhizobium medicae WSM419 | Nodules of Medicago murex | Medicago truncatula | Delays stress-induced leaf senescence and abscission and nutrient acquisition during drought stress | [173] | |
| Sinorhizobium meliloti A2 strains | Commercial strain, Eastern Canada [174] | Medicago sativa cv Apica, Medicago sativa cv Halo | Increases shoot/root ratio, shoot water content, and the concentrations of starch and pinitol in nodules | [175] | |
| Sinorhizobium meliloti Rm1521 | Ottawa vicinity [176] | ||||
| Rhizobium etli CE3 overexpressing trehalose-6-phosphate synthase gene | CFN42 derivate [177], original isolate from P. vulgaris nodule | P. vulgaris var. Negro Jamapa | Enhances drought tolerance due to upregulation of genes involved in stress tolerance, carbon and nitrogen metabolism by trehalose | [107] | |
| Co-inoculation | Crop | Effect/Mechanism of action | Reference | ||
| Rhizobia | PGPRs | ||||
| Rhizobium phaseoli M6; M9. phaseoli M6 and M9 and PGPR | Pseudomonas syringae, Mk1; Pseudomonas fluorescens Mk20; Pseudomonas fluorescens biot. G Mk25 | Vigna radiata L. | Decreases damaging effect of salinity stress on mung bean growth | [116] | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bellabarba, A.; Fagorzi, C.; diCenzo, G.C.; Pini, F.; Viti, C.; Checcucci, A. Deciphering the Symbiotic Plant Microbiome: Translating the Most Recent Discoveries on Rhizobia for the Improvement of Agricultural Practices in Metal-Contaminated and High Saline Lands. Agronomy 2019, 9, 529. https://doi.org/10.3390/agronomy9090529
Bellabarba A, Fagorzi C, diCenzo GC, Pini F, Viti C, Checcucci A. Deciphering the Symbiotic Plant Microbiome: Translating the Most Recent Discoveries on Rhizobia for the Improvement of Agricultural Practices in Metal-Contaminated and High Saline Lands. Agronomy. 2019; 9(9):529. https://doi.org/10.3390/agronomy9090529
Chicago/Turabian StyleBellabarba, Agnese, Camilla Fagorzi, George C. diCenzo, Francesco Pini, Carlo Viti, and Alice Checcucci. 2019. "Deciphering the Symbiotic Plant Microbiome: Translating the Most Recent Discoveries on Rhizobia for the Improvement of Agricultural Practices in Metal-Contaminated and High Saline Lands" Agronomy 9, no. 9: 529. https://doi.org/10.3390/agronomy9090529
APA StyleBellabarba, A., Fagorzi, C., diCenzo, G. C., Pini, F., Viti, C., & Checcucci, A. (2019). Deciphering the Symbiotic Plant Microbiome: Translating the Most Recent Discoveries on Rhizobia for the Improvement of Agricultural Practices in Metal-Contaminated and High Saline Lands. Agronomy, 9(9), 529. https://doi.org/10.3390/agronomy9090529








