Long-Term Monoculture Negatively Regulates Fungal Community Composition and Abundance of Tea Orchards
Abstract
1. Introduction
2. Materials and Methods
2.1. Site Description and Soil Sampling
2.2. Yield Determination
2.3. Measurement of Soil Physiochemical Properties
2.4. DNA Extraction and PCR Amplification
2.5. Statistical and Bioinformatics Analysis
3. Results
3.1. Yield of Tea Leaves in Different Age Monoculture Tea Fields
3.2. Soil Physicochemical Characteristics from Different Age Monoculture Tea Fields
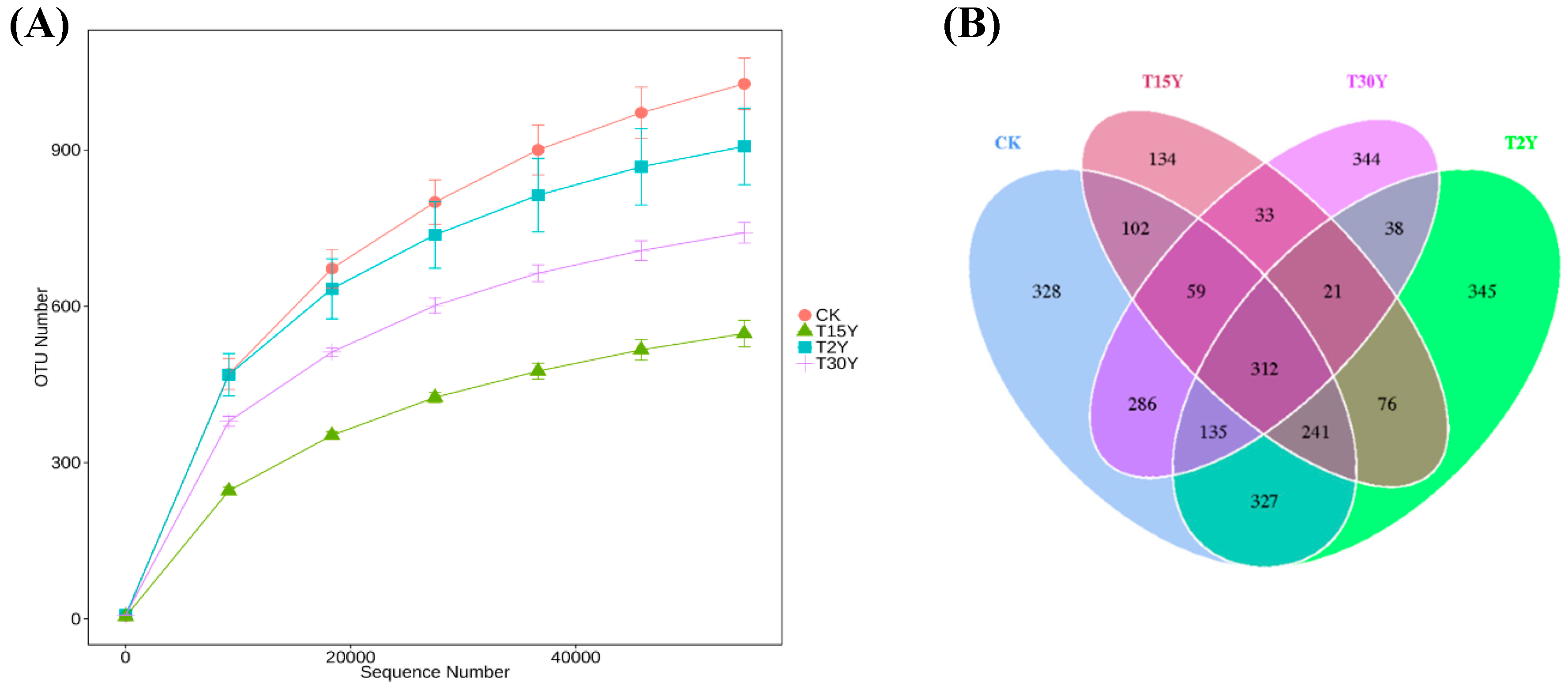
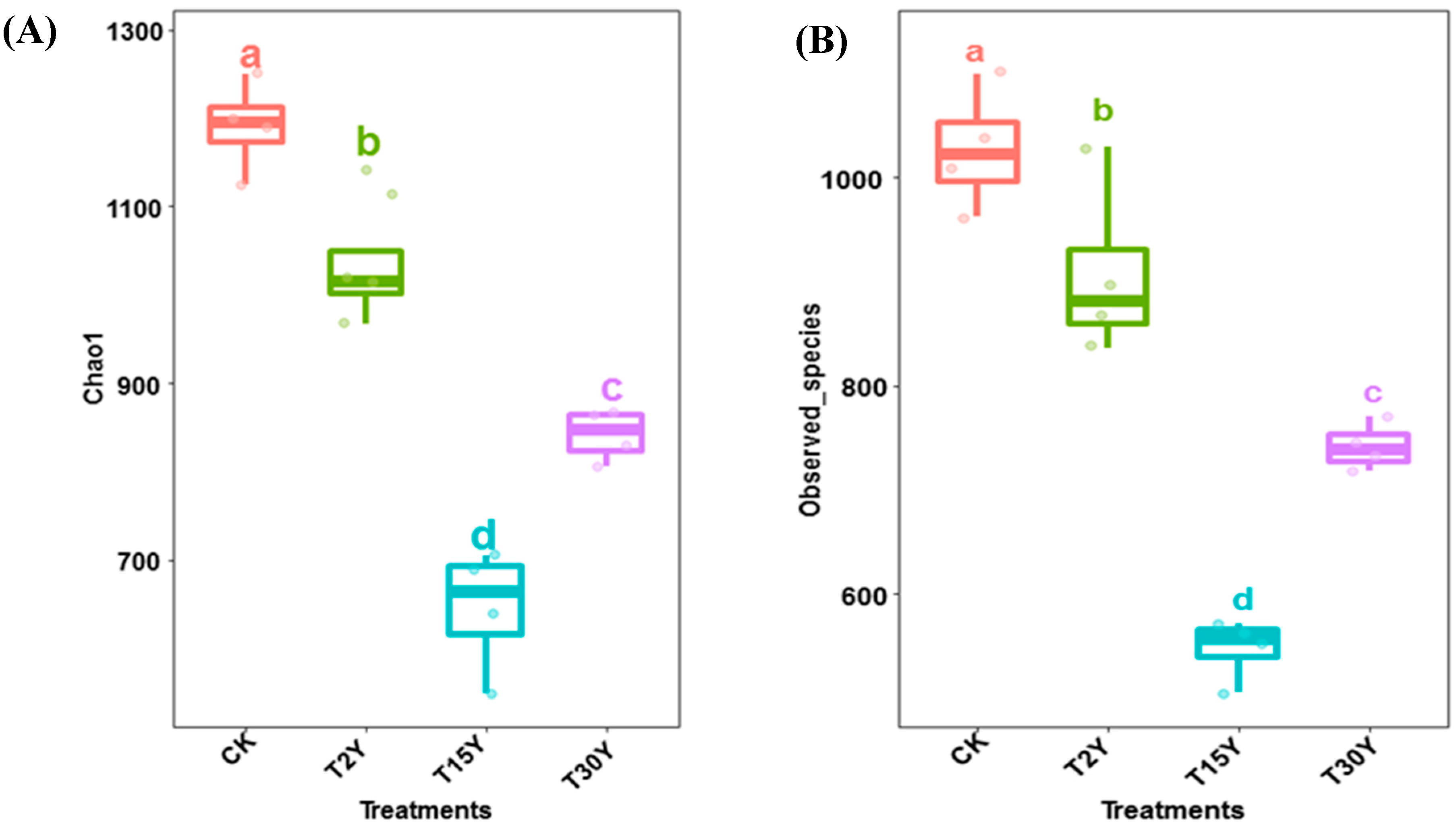
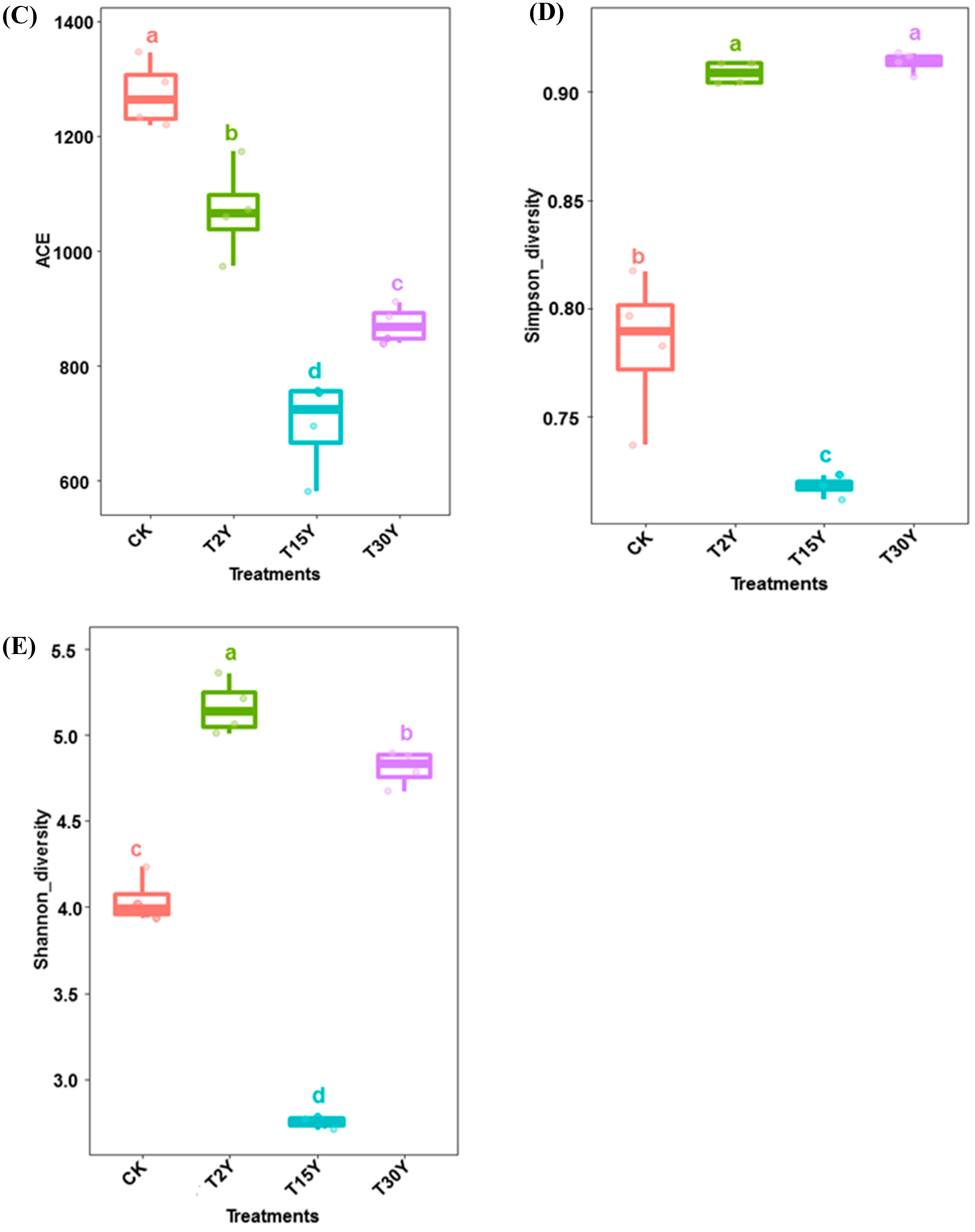
3.3. Fungal Alpha Diversity and Species Richness
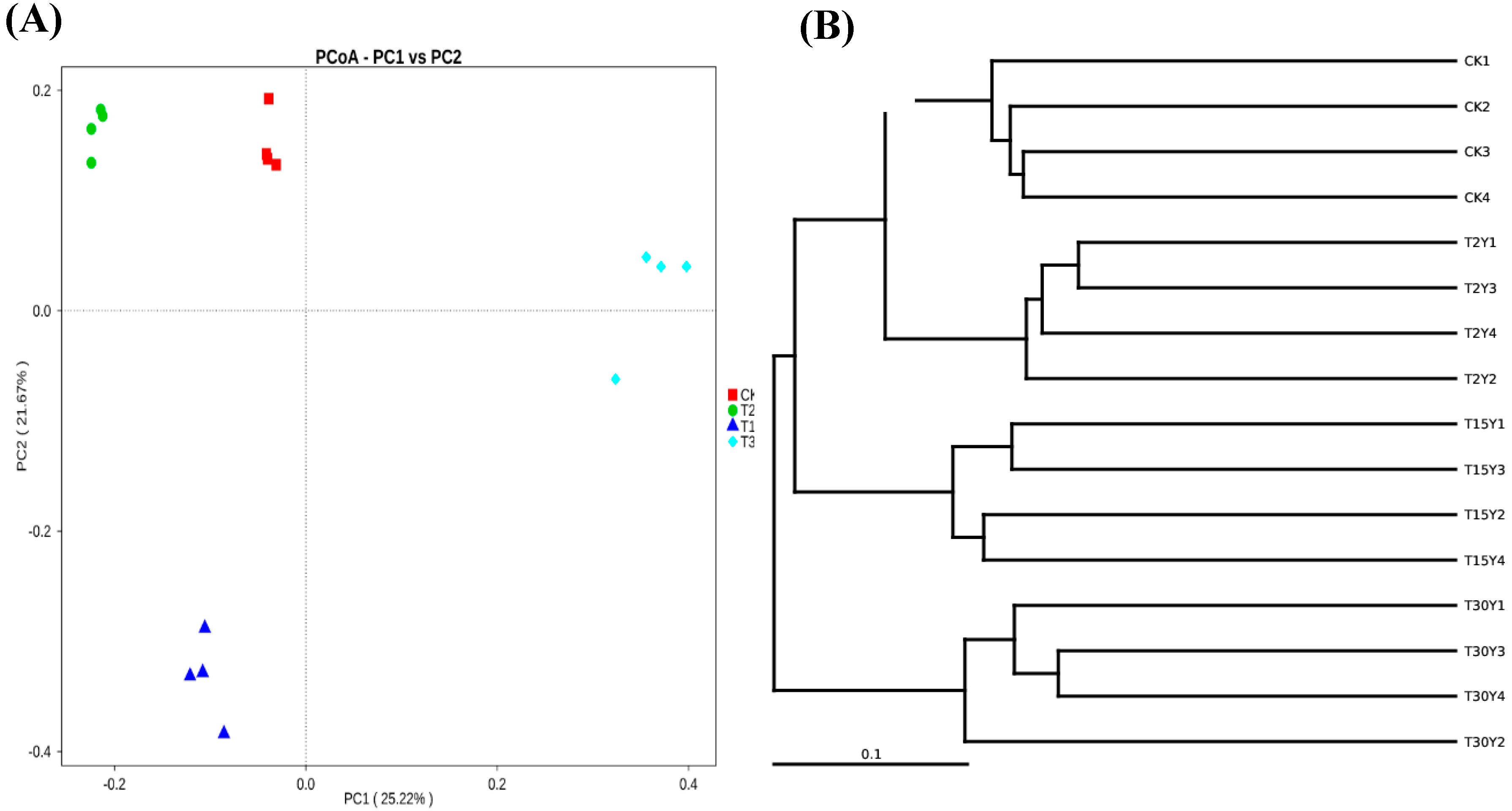
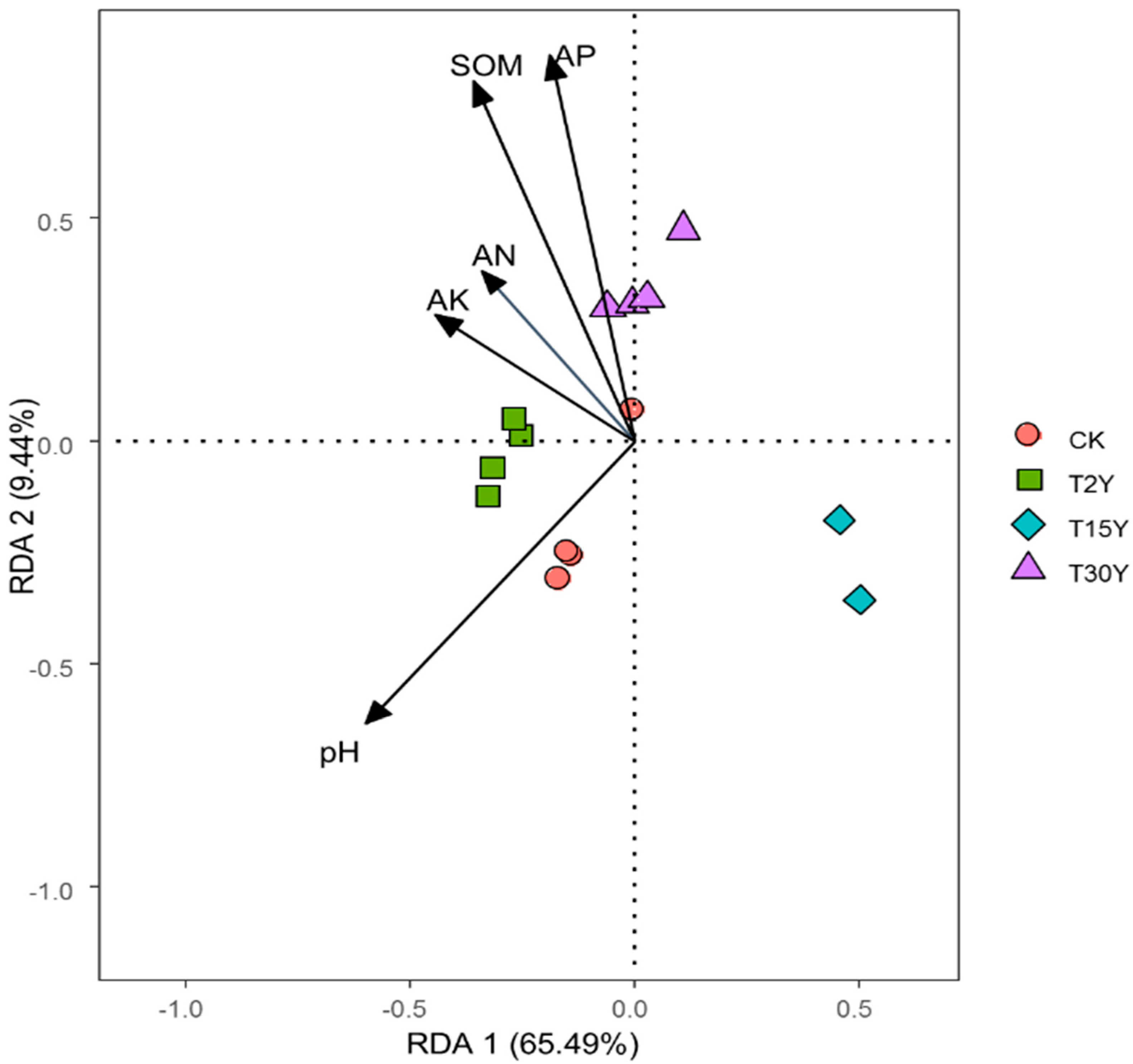
3.4. Fungal B-Diversity
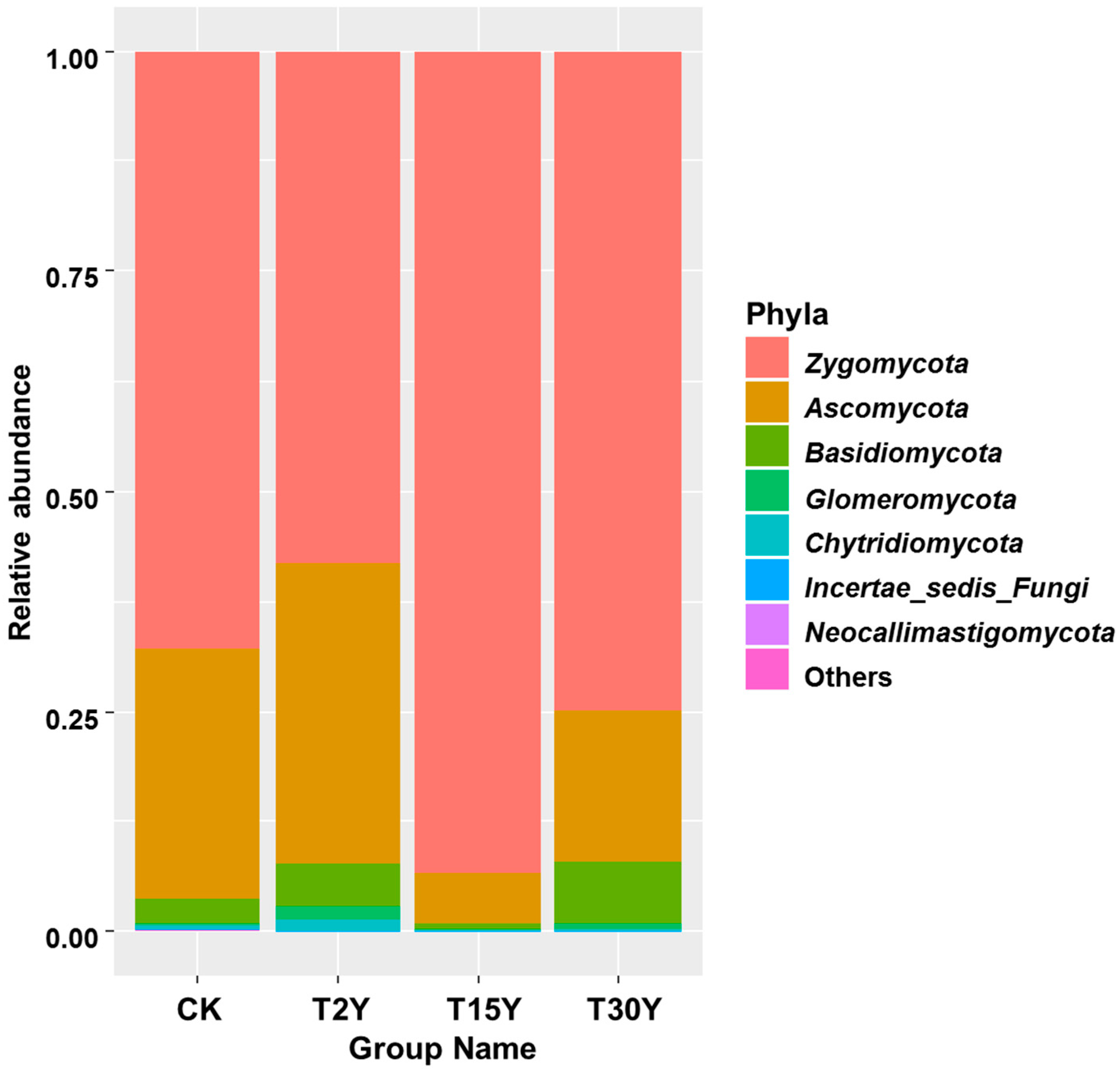
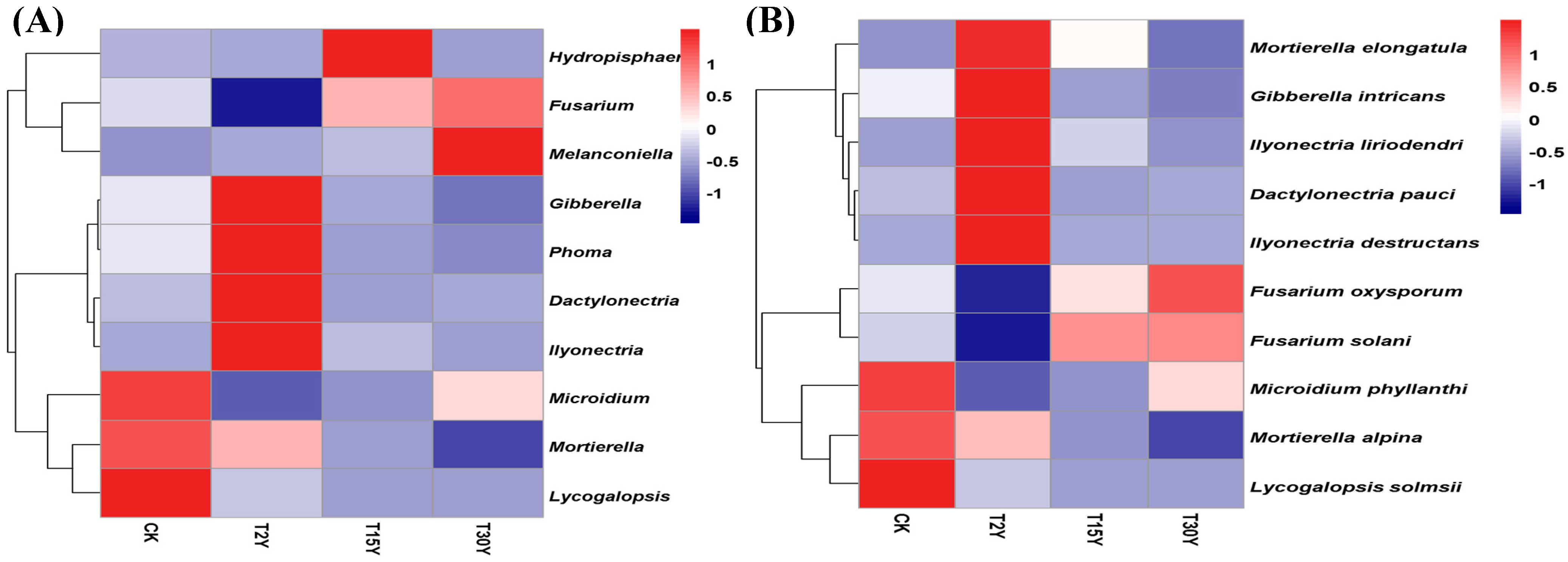
3.5. Relative Abundance of Fungal Communities From Different Age Monoculture Tea Fields
3.6. Effects of Soil Physiochemical Properties on Fungal Communities
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Han, W.; Kemmitt, S.J.; Brookes, P.C. Soil microbial biomass and activity in Chinese tea gardens of varying stand age and productivity. Soil Biol. Biochem. 2007, 39, 1468–1478. [Google Scholar] [CrossRef]
- Li, Y.; Jiang, Y.; Weng, B.; Lin, W. Variations of rhizosphere bacterial communities in tea (Camellia sinensis L.) continuous cropping soil by high-throughput pyrosequencing approach. J. Appl. Microbiol. 2016, 121, 787–799. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Wang, M.; Zhao, J.; Wang, Y.-H.; Tang, Q.; Khan, I.A. Yellow tea (Camellia sinensis L.), a promising Chinese tea: Processing, chemical constituents and health benefits. Food Res. Int. 2018, 107, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Arafat, Y.; Wei, X.; Jiang, Y.; Chen, T.; Saqib, H.S.A.; Lin, S.; Lin, W. Spatial Distribution Patterns of Root-Associated Bacterial Communities Mediated by Root Exudates in Different Aged Ratooning Tea Monoculture Systems. Int. J. Mol. Sci. 2017, 18, 1727. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Li, H.; Yang, C.; Wang, Y.; Xue, H.; Niu, Y. Rates of soil acidification in tea plantations and possible causes. Agric. Ecosyst. Environ. 2016, 233, 60–66. [Google Scholar] [CrossRef]
- Zhao, Q.; Xiong, W.; Xing, Y.; Sun, Y.; Lin, X.; Dong, Y. Long-Term Coffee Monoculture Alters Soil Chemical Properties and Microbial Communities. Sci. Rep. 2018, 8, 6116. [Google Scholar] [CrossRef]
- Lin, W.; Wu, L.; Lin, S.; Zhang, A.; Zhou, M.; Lin, R.; Wang, H.; Chen, J.; Zhang, Z.; Lin, R. Metaproteomic analysis of ratoon sugarcane rhizospheric soil. BMC Microbiol. 2013, 13, 135. [Google Scholar] [CrossRef]
- Mazzola, M.; Manici, L.M. Apple Replant Disease: Role of Microbial Ecology in Cause and Control. Annu. Rev. Phytopathol. 2012, 50, 45–65. [Google Scholar] [CrossRef]
- Lu, L.; Yin, S.; Liu, X.; Zhang, W.; Gu, T.; Shen, Q.; Qiu, H. Fungal networks in yield-invigorating and -debilitating soils induced by prolonged potato monoculture. Soil Biol. Biochem. 2013, 65, 186–194. [Google Scholar] [CrossRef]
- Bai, L.; Cui, J.; Jie, W.; Cai, B. Analysis of the community compositions of rhizosphere fungi in soybeans continuous cropping fields. Microbiol. Res. 2015, 180, 49–56. [Google Scholar] [CrossRef]
- Gil, S.V.; Meriles, J.; Conforto, C.; Figoni, G.; Basanta, M.; Lovera, E.; March, G.J. Field assessment of soil biological and chemical quality in response to crop management practices. World J. Microbiol. Biotechnol. 2009, 25, 439–448. [Google Scholar]
- Li, X.-G.; Ding, C.-F.; Zhang, T.-L.; Wang, X.-X. Fungal pathogen accumulation at the expense of plant-beneficial fungi as a consequence of consecutive peanut monoculturing. Soil Biol. Biochem. 2014, 72, 11–18. [Google Scholar] [CrossRef]
- Lin, W.; Chen, T.; Zhou, M. New dimensions in agroecology. Zhongguo Shengtai Nongye Xuebao Chin. J. Eco-Agric. 2012, 20, 253–264. [Google Scholar] [CrossRef]
- Xiong, W.; Zhao, Q.; Zhao, J.; Xun, W.; Li, R.; Zhang, R.; Wu, H.; Shen, Q. Different continuous cropping spans significantly affect microbial community membership and structure in a vanilla-grown soil as revealed by deep pyrosequencing. Microb. Ecol. 2015, 70, 209–218. [Google Scholar] [CrossRef] [PubMed]
- Manici, L.M.; Kelderer, M.; Franke-Whittle, I.; Rühmer, T.; Baab, G.; Nicoletti, F.; Caputo, F.; Topp, A.; Insam, H.; Naef, A. Relationship between root-endophytic microbial communities and replant disease in specialized apple growing areas in Europe. Appl. Soil Ecol. 2013, 72, 207–214. [Google Scholar] [CrossRef]
- Song, X.; Pan, Y.; Li, L.; Wu, X.; Wang, Y. Composition and diversity of rhizosphere fungal community in Coptis chinensis Franch. continuous cropping fields. PLoS ONE 2018, 13, e0193811. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Arafat, Y.; Letuma, P.; Ali, L.; Tayyab, M.; Waqas, M.; Li, Y.; Lin, W.; Lin, S.; Lin, W. Restoration of Long-Term Monoculture Degraded Tea Orchard by Green and Goat Manures Applications System. Sustainability 2019, 11, 1011. [Google Scholar] [CrossRef]
- Murphy, J.; Riley, J. A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta 1962, 27, 31–36. [Google Scholar] [CrossRef]
- Pansu, M.; Gautheyrou, J. Handbook of Soil Analysis: Mineralogical, Organic and Inorganic Methods; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2007. [Google Scholar]
- Sparks, D.; Page, A.; Helmke, P.; Loeppert, R.; Nelson, D.W.; Sommers, L.E. Total Carbon, Organic Carbon, and Organic Matter; Purdue University: West Lafayette, IN, USA, 1996; pp. 961–1010. [Google Scholar]
- Huang, Y.; Kuang, Z.; Wang, W.; Cao, L. Exploring potential bacterial and fungal biocontrol agents transmitted from seeds to sprouts of wheat. Biol. Control 2016, 98, 27–33. [Google Scholar] [CrossRef]
- Magoč, T.; Salzberg, S.L. FLASH: Fast length adjustment of short reads to improve genome assemblies. Bioinformatics 2011, 27, 2957–2963. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naïve Bayesian Classifier for Rapid Assignment of rRNA Sequences into the New Bacterial Taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- Keylock, C.J.; Keylock, C. Simpson diversity and the Shannon-Wiener index as special cases of a generalized entropy. Oikos 2005, 109, 203–207. [Google Scholar] [CrossRef]
- Chao, A.; Lee, S.-M. Estimating the Number of Classes via Sample Coverage. J. Am. Stat. Assoc. 1992, 87, 210–217. [Google Scholar] [CrossRef]
- Chao, A. Nonparametric estimation of the number of classes in a population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Dutta, R. Impact of age and management factors on tea yield and modelling the influence of leaf area index on yield variations. Sci. Asia 2011, 37, 83–87. [Google Scholar] [CrossRef]
- Dutta, R.; Smaling, E.M.A.; Bhagat, R.M.; Tolpekin, V. Analysis of factors that determine tea productivity in northeastern india: A combined statistical and modelling approach. Exp. Agric. 2012, 48, 64–84. [Google Scholar] [CrossRef]
- Wang, H.; Xu, R.-K.; Wang, N.; Li, X.-H. Soil acidification of alfisols as influenced by tea cultivation in Eastern China. Pedosphere 2010, 20, 799–806. [Google Scholar] [CrossRef]
- Ladha, J.; Bijay-Singh; Ladha, J.K.; Khind, C.S.; Gupta, R.K.; Meelu, O.P.; Pasuquin, E. Long-term effects of organic inputs on yield and soil fertility in the rice–wheat rotation. Soil Sci. Soc. Am. J. 2004, 68, 845–853. [Google Scholar]
- Hara, Y. Tea catechins and their applications as supplements and pharmaceutics. Pharmacol. Res. 2011, 64, 100–104. [Google Scholar] [CrossRef]
- Cao, P.; Liu, C.; Li, D. Effects of different autotoxins on antioxidant enzymes and chemical compounds in tea (Camellia sinensis L.) Kuntze. Afr. J. Biotechnol. 2011, 10, 7480–7486. [Google Scholar]
- Xue, D.; Yao, H.; Huang, C. Microbial Biomass, N Mineralization and Nitrification, Enzyme Activities, and Microbial Community Diversity in Tea Orchard Soils. Plant Soil 2006, 288, 319–331. [Google Scholar] [CrossRef]
- Zhao, J.; Wu, X.; Nie, C.; Wu, T.; Dai, W.; Liu, H.; Yang, R. Analysis of unculturable bacterial communities in tea orchard soils based on nested PCR-DGGE. World J. Microbiol. Biotechnol. 2012, 28, 1967–1979. [Google Scholar] [CrossRef] [PubMed]
- Xiong, W.; Li, Z.; Liu, H.; Xue, C.; Zhang, R.; Wu, H.; Li, R.; Shen, Q. The Effect of Long-Term Continuous Cropping of Black Pepper on Soil Bacterial Communities as Determined by 454 Pyrosequencing. PLoS ONE 2015, 10, e0136946. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Zhang, J.; Gu, T.; Zhang, W.; Shen, Q.; Yin, S.; Qiu, H. Microbial Community Diversities and Taxa Abundances in Soils along a Seven-Year Gradient of Potato Monoculture Using High Throughput Pyrosequencing Approach. PLoS ONE 2014, 9, e86610. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Li, R.; Zhang, F.; Fan, J.; Tang, C.; Rengel, Z. Crop yields, soil fertility and phosphorus fractions in response to long-term fertilization under the rice monoculture system on a calcareous soil. Field Crops Res. 2004, 86, 225–238. [Google Scholar] [CrossRef]
- Tayyab, M.; Islam, W.; Lee, C.G.; Pang, Z.; Khalil, F.; Lin, S.; Lin, W.; Zhang, H. Short-Term Effects of Different Organic Amendments on Soil Fungal Composition. Sustainability 2019, 11, 198. [Google Scholar] [CrossRef]
- Bai, L.; Sun, H.; Zhang, X.; Cai, B. Next-generation sequencing of root fungal communities in continuous cropping soybean. Chil. J. Agric. Res. 2018, 78, 528–538. [Google Scholar] [CrossRef]
- Tan, Y.; Cui, Y.; Li, H.; Kuang, A.; Li, X.; Wei, Y.; Ji, X. Rhizospheric soil and root endogenous fungal diversity and composition in response to continuous Panax notoginseng cropping practices. Microbiol. Res. 2017, 194, 10–19. [Google Scholar] [CrossRef]
- Singh, B.K.; Quince, C.; Macdonald, C.A.; Khachane, A.; Thomas, N.; Abu Al-Soud, W.; Sørensen, S.J.; He, Z.; White, D.; Sinclair, A.; et al. Loss of microbial diversity in soils is coincident with reductions in some specialized functions. Environ. Microbiol. 2014, 16, 2408–2420. [Google Scholar] [CrossRef]
- Van Der Heijden, M.G.A.; Bardgett, R.D.; Van Straalen, N.M. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar] [CrossRef]
- Maron, P.-A.; Sarr, A.; Kaisermann, A.; Lévêque, J.; Mathieu, O.; Guigue, J.; Karimi, B.; Bernard, L.; Dequiedt, S.; Terrat, S.; et al. High Microbial Diversity Promotes Soil Ecosystem Functioning. Appl. Environ. Microbiol. 2018, 84, e02738-17. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Tang, C.; Chen, Z. The role of plant residues in pH change of acid soils differing in initial pH. Soil Biol. Biochem. 2006, 38, 709–719. [Google Scholar] [CrossRef]
- Phong, N.H.; Pongnak, W.; Soytong, K. Antifungal activities of Chaetomium spp. against Fusarium wilt of tea. Plant Prot. Sci. 2015, 52, 10–17. [Google Scholar] [CrossRef]
- Sinniah, G.D.; Munasinghe, C.E.; Mahadevan, N.; Jayasinghe, S.K.; Kulatunga, D.C.M. Recent incidence of collar canker and dieback of tea (Camellia sinensis) caused by Fusarium solani species complex in Sri Lanka. Australas. Plant Dis. Notes 2017, 12, 41. [Google Scholar] [CrossRef]
- Hartmann, M.; Frey, B.; Mayer, J.; Mäder, P.; Widmer, F. Distinct soil microbial diversity under long-term organic and conventional farming. ISME J. 2015, 9, 1177. [Google Scholar] [CrossRef]
- LeComte, C.; Alabouvette, C.; Edel-Hermann, V.; Robert, F.; Steinberg, C. Biological control of ornamental plant diseases caused by Fusarium oxysporum: A review. Boil. Control 2016, 101, 17–30. [Google Scholar] [CrossRef]
- Shen, Z.; Penton, C.R.; Lv, N.; Xue, C.; Yuan, X.; Ruan, Y.; Li, R.; Shen, Q. Banana fusarium wilt disease incidence is influenced by shifts of soil microbial communities under different monoculture spans. Microb. Ecol. 2018, 75, 739–750. [Google Scholar] [CrossRef]
- Wani, Z.A.; Kumar, A.; Sultan, P.; Bindu, K.; Riyaz-Ul-Hassan, S.; Ashraf, N. Mortierella alpina CS10E4, an oleaginous fungal endophyte of Crocus sativus L. enhances apocarotenoid biosynthesis and stress tolerance in the host plant. Sci. Rep. 2017, 7, 8598. [Google Scholar] [CrossRef]
- Tam, L.T.T.; Hoai, H.T.; Thao, L.P.; Huong, N.T.; Khue, N.M. First report of Microidium phyllanthi causing powdery mildew on chamber bitter in Vietnam. New Dis. Rep. 2015, 32, 32. [Google Scholar] [CrossRef]
- Salch, Y.P.; Beremand, M.N. Gibberella pulicaris transformants: State of transforming DNA during asexual and sexual growth. Curr. Genet. 1993, 23, 343–350. [Google Scholar] [CrossRef]
- Liu, J.; Yao, Q.; Li, Y.; Zhang, W.; Mi, G.; Chen, X.; Yu, Z.; Wang, G. Continuous cropping of soybean alters the bulk and rhizospheric soil fungal communities in a Mollisol of Northeast China. Land Degrad. Dev. 2019. [Google Scholar] [CrossRef]
- Li, W.-H.; Liu, Q.-Z. Changes in fungal community and diversity in strawberry rhizosphere soil after 12 years in the greenhouse. J. Integr. Agric. 2019, 18, 677–687. [Google Scholar] [CrossRef]
| (Yield per unit) | ||
|---|---|---|
| Treatment | (Fresh weight) | (Dry weight) |
| -------kg·ha−1----- | ||
| T2Y | 1436.37 ± 5.54 a | 408.33 ± 2.03 a |
| T15Y | 1395.00 ± 1.73 b | 382.67 ± 1.75 b |
| T30Y | 1371.67 ± 5.55 c | 345.67 ± 2.03 c |
| Samples | pH | SOM (g kg−1) | AN (mg kg−1) | AK (mg kg−1) | AP (mg kg−1) |
|---|---|---|---|---|---|
| CK | 5.07 ± 0.06 a | 12.20 ± 0.37 b | 13.09 ± 0.10 c | 132.79 ± 2.69 a | 85.72 ± 0.52 c |
| T2Y | 4.23 ± 0.06 b | 13.45 ± 0.32 a | 14.69 ± 0.10 a | 130.23 ± 0.87 a | 91.19 ± 0.42 a |
| T15Y | 3.58 ± 0.28 c | 12.39 ± 0.26 b | 13.74 ± 0.10 b | 128.48 ± 0.53 a | 88.04 ± 0.80 b |
| T30Y | 3.32 ± 0.08 c | 13.74 ± 0.05 a | 13.95 ± 0.09 b | 133.24 ± 0.59 a | 92.40 ± 0.71 a |
| Fungal abundant phyla | pH | SOM | AN | AK | AP |
| Zygomycota | −0.50 * | −0.32 | −0.34 | −0.36 | −0.17 |
| Ascomycota | 0.61 * | 0.15 | 0.24 | 0.33 | −0.00 |
| Basidiomycota | −0.26 | 0.79 ** | 0.39 | 0.45 | 0.75 ** |
| Glomeromycota | 0.08 | 0.60 * | 0.7 ** | −0.03 | 0.54 * |
| Chytridiomycota | 0.37 | 0.32 | 0.60 * | −0.11 | 0.24 |
| Incertae_sedis_Fungi | −0.49 * | 0.47 | 0.04 | 0.33 | 0.49 |
| Neocallimastigomycota | 0.61 * | −0.10 | −0.103 | −0.00 | −0.26 |
| Fungal abundant genera | |||||
| Mortierella | 0.94 ** | −0.41 | −0.23 | 0.05 | −0.58 * |
| Microidium | 0.36 | −0.11 | −0.49 | 0.26 | −0.22 |
| Fusarium | −0.45 | −0.08 | −0.33 | 0.16 | −0.06 |
| Dactylonectria | 0.21 | 0.35 | 0.77 ** | −0.16 | 0.34 |
| Gibberella | 0.37 | 0.17 | 0.62 ** | −0.15 | 0.13 |
| Ilyonectria | 0.14 | 0.35 | 0.80 ** | −0.27 | 0.36 |
| Lycogalopsis | 0.63 ** | −0.26 | −0.43 | 0.09 | −0.45 |
| Melanconiella | −0.64 ** | 0.53 * | 0.12 | 0.31 | 0.51 * |
| Phoma | 0.35 | 0.21 | 0.64 ** | −0.12 | 0.18 |
| Hydropisphaera | −0.34 | −0.45 | −0.14 | −0.60 * | −0.31 |
| Fungal abundant species | |||||
| Mortierella alpina | 0.94 ** | −0.40 | −0.24 | 0.09 | −0.58 * |
| Microidium phyllanthi | 0.36 | −0.11 | −0.49 | 0.26 | −0.22 |
| Dactylonectria pauciseptata | 0.21 | 0.35 | 0.77 ** | −0.16 | 0.34 |
| Fusarium oxysporum | −0.39 | −0.00 | −0.25 | 0.22 | −0.00 |
| Gibberella intricans | 0.38 | 0.18 | 0.62 ** | −0.13 | 0.14 |
| Fusarium solani | −0.52 * | −0.12 | −0.36 | 0.10 | −0.08 |
| Mortierella elongatula | 0.08 | 0.20 | 0.75 ** | −0.43 | 0.22 |
| Ilyonectria liriodendri | 0.12 | 0.33 | 0.80 ** | −0.31 | 0.34 |
| Lycogalopsis solmsii | 0.63 ** | −0.26 | −0.43 | 0.09 | −0.45 |
| Ilyonectria destructans | 0.16 | 0.37 | 0.80 ** | −0.22 | 0.37 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arafat, Y.; Tayyab, M.; Khan, M.U.; Chen, T.; Amjad, H.; Awais, S.; Lin, X.; Lin, W.; Lin, S. Long-Term Monoculture Negatively Regulates Fungal Community Composition and Abundance of Tea Orchards. Agronomy 2019, 9, 466. https://doi.org/10.3390/agronomy9080466
Arafat Y, Tayyab M, Khan MU, Chen T, Amjad H, Awais S, Lin X, Lin W, Lin S. Long-Term Monoculture Negatively Regulates Fungal Community Composition and Abundance of Tea Orchards. Agronomy. 2019; 9(8):466. https://doi.org/10.3390/agronomy9080466
Chicago/Turabian StyleArafat, Yasir, Muhammad Tayyab, Muhammad Umar Khan, Ting Chen, Hira Amjad, Saadia Awais, Xiangmin Lin, Wenxiong Lin, and Sheng Lin. 2019. "Long-Term Monoculture Negatively Regulates Fungal Community Composition and Abundance of Tea Orchards" Agronomy 9, no. 8: 466. https://doi.org/10.3390/agronomy9080466
APA StyleArafat, Y., Tayyab, M., Khan, M. U., Chen, T., Amjad, H., Awais, S., Lin, X., Lin, W., & Lin, S. (2019). Long-Term Monoculture Negatively Regulates Fungal Community Composition and Abundance of Tea Orchards. Agronomy, 9(8), 466. https://doi.org/10.3390/agronomy9080466





