Yield and Quality of Essential Oils in Hemp Varieties in Different Environments
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Environments, Trial Description and Characters Analyzed
2.3. Chemicals
2.4. Essential Oil Extraction Method
2.5. Apparatus, Sample Preparation, Identification and Quantification of Terpenes in EOs
2.6. Identification and Quantification of Phytocannabinoides in Inflorescences
2.7. Statistical analysis
3. Results and discussion
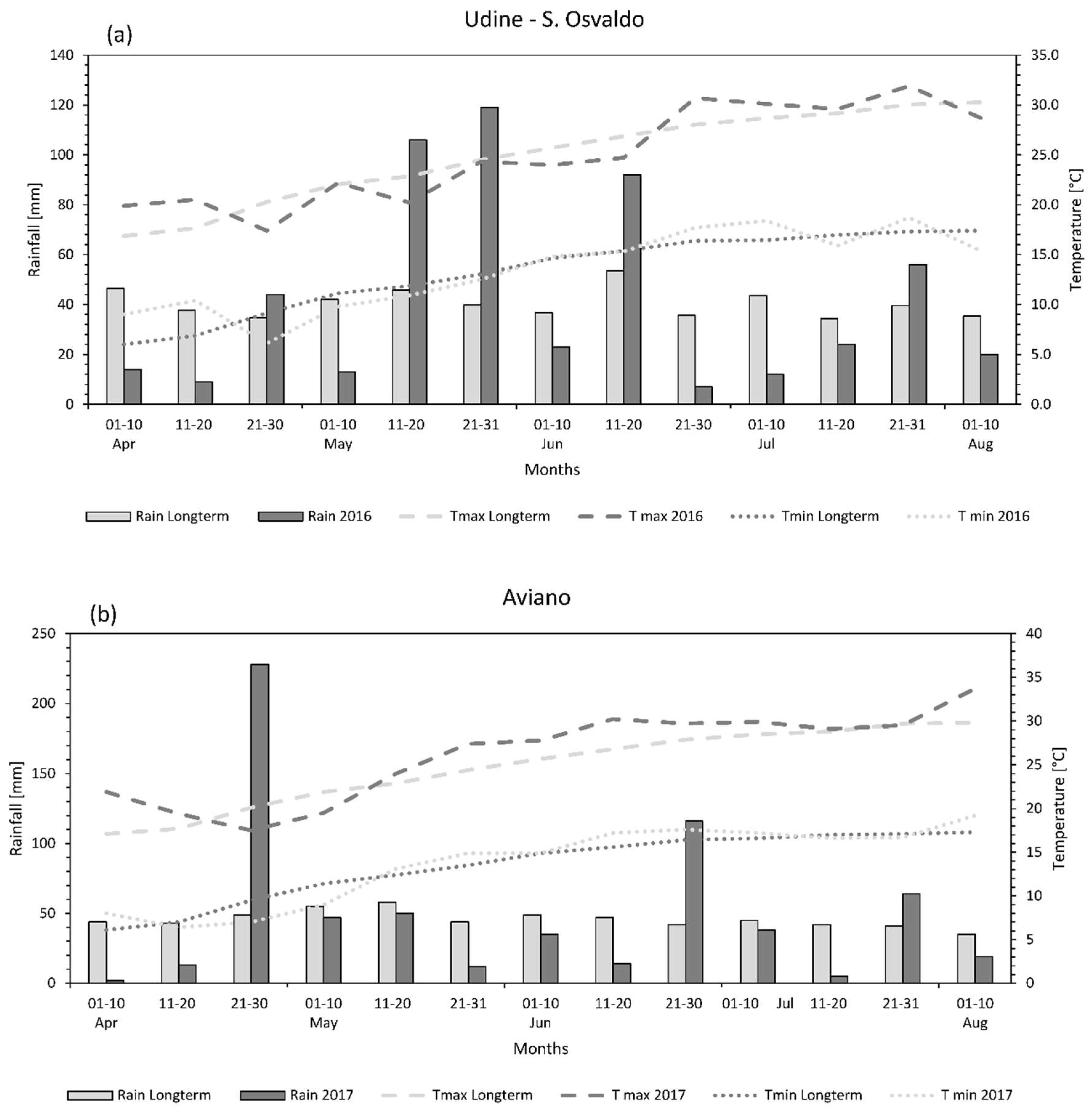
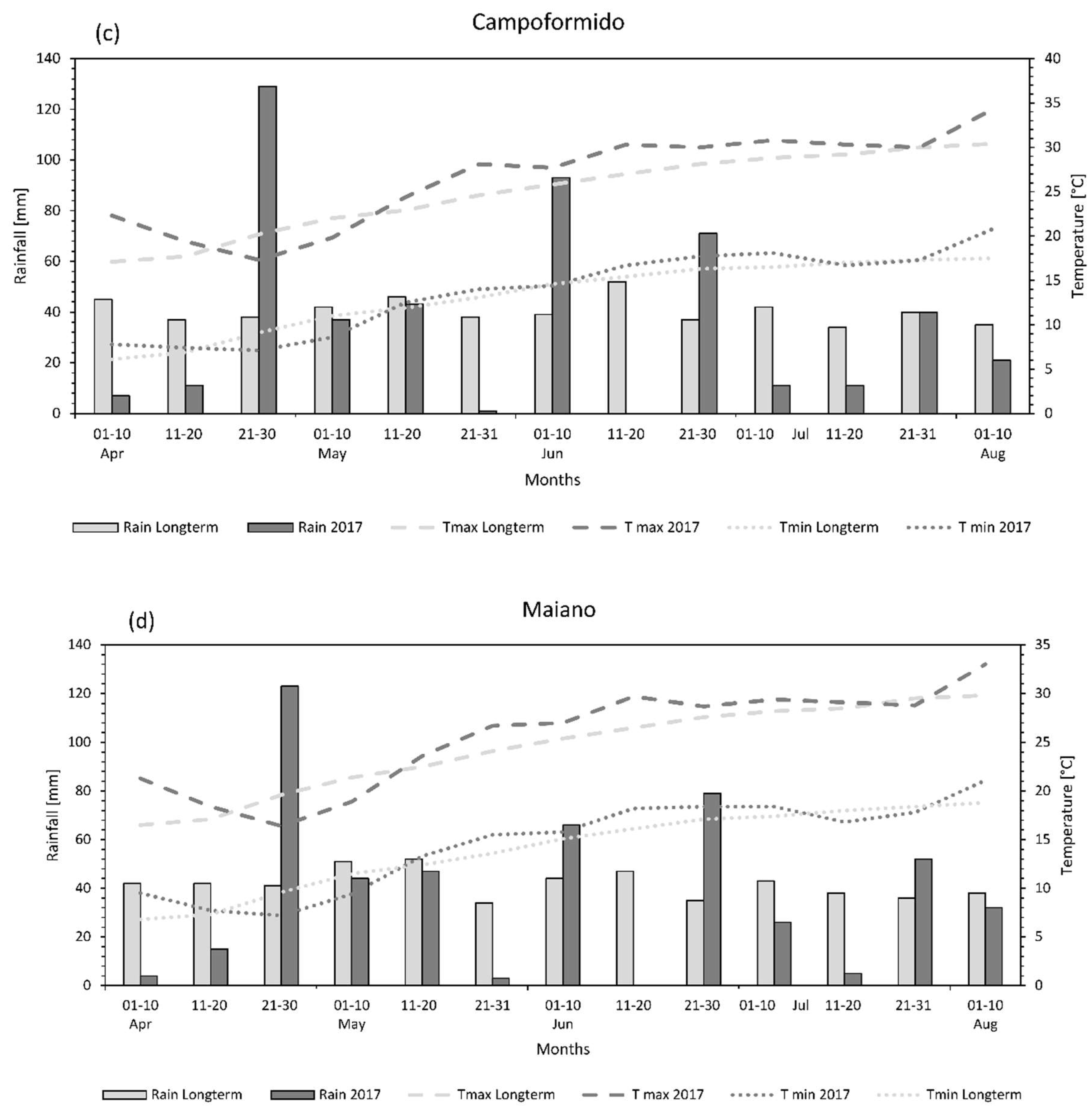
3.1. Weather Conditions
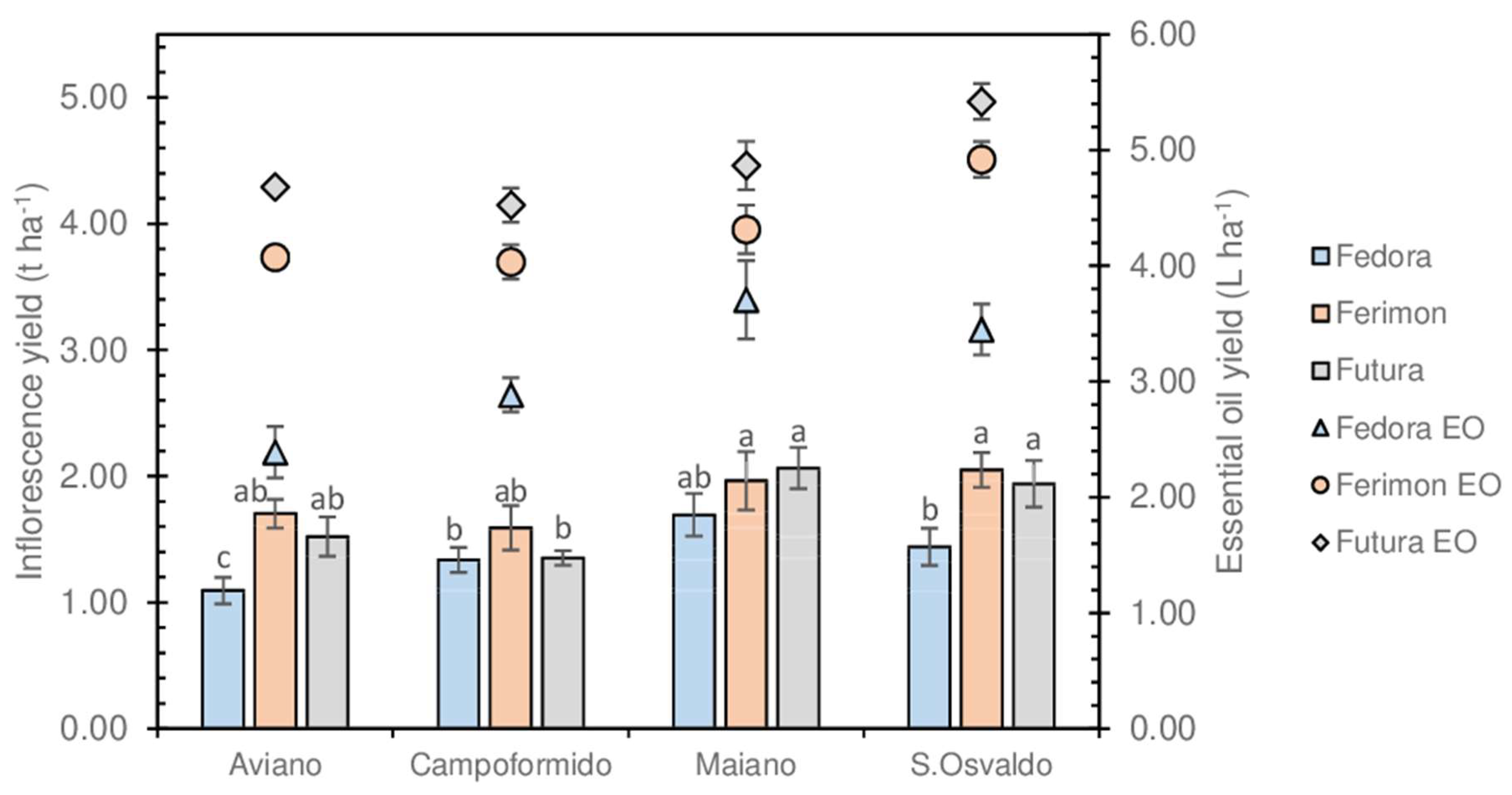
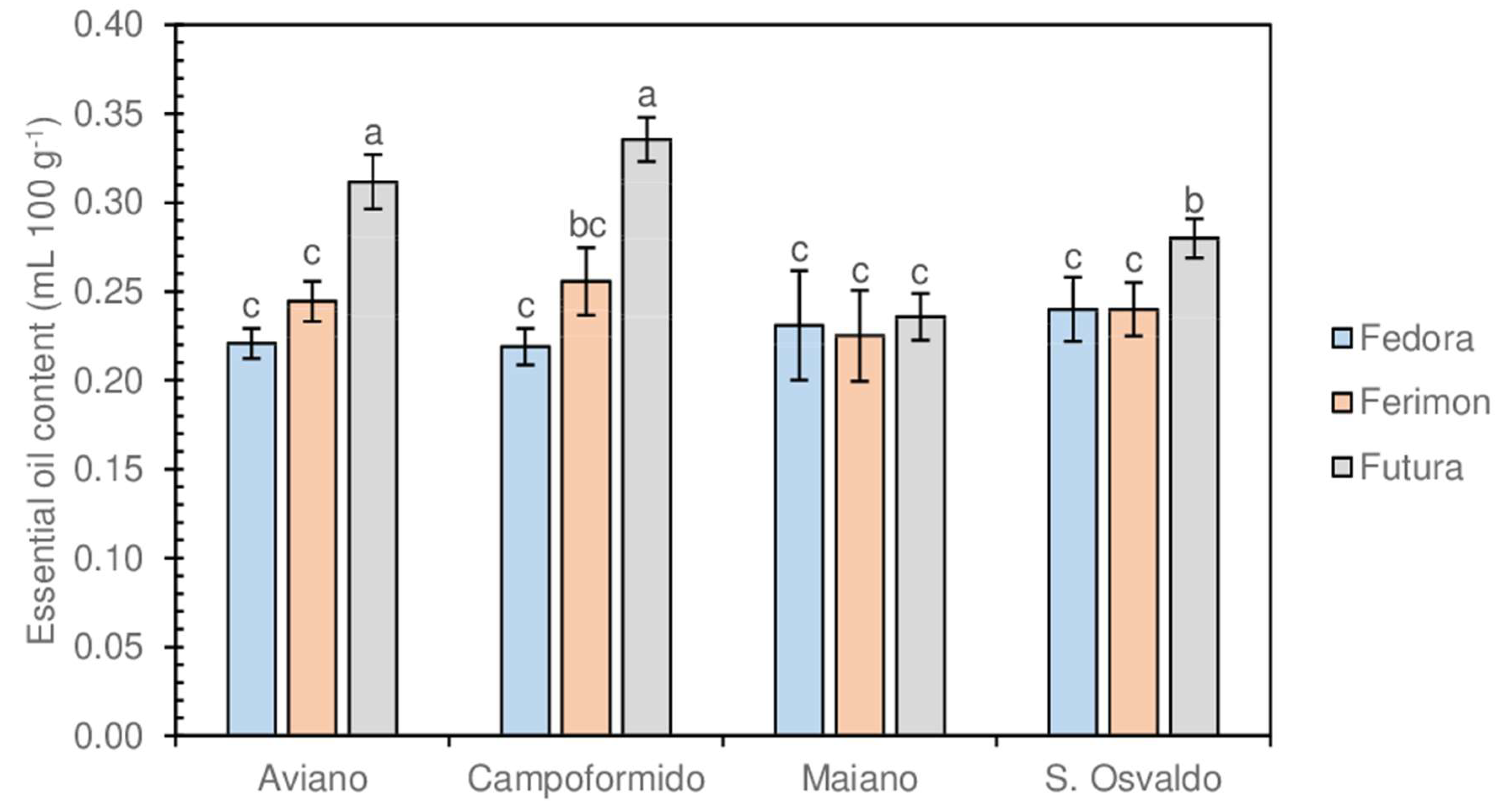
3.2. Inflorescences and EOs Yield
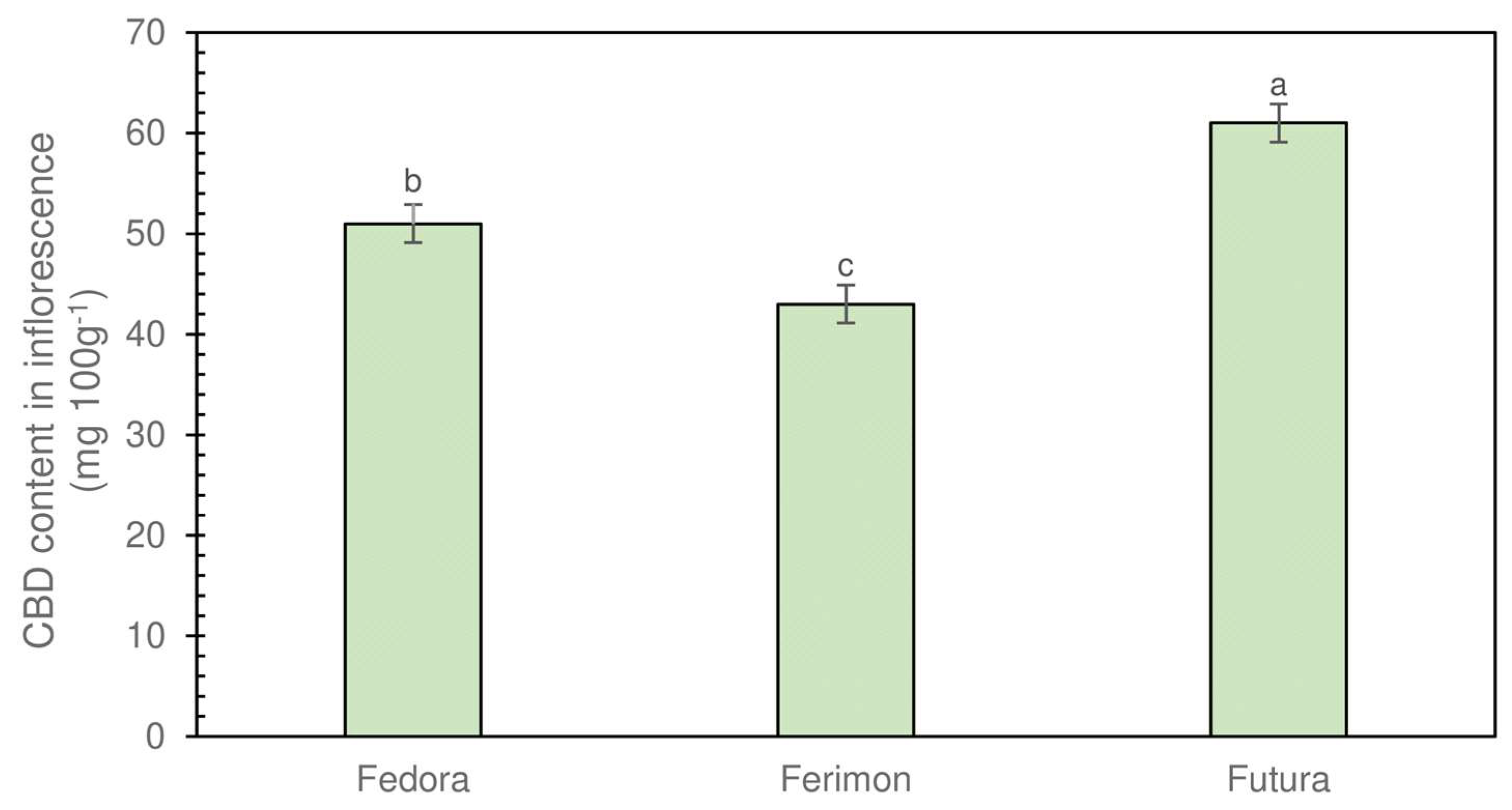
3.3. Phytocannabinoids Determination in Inflorescences and EOs
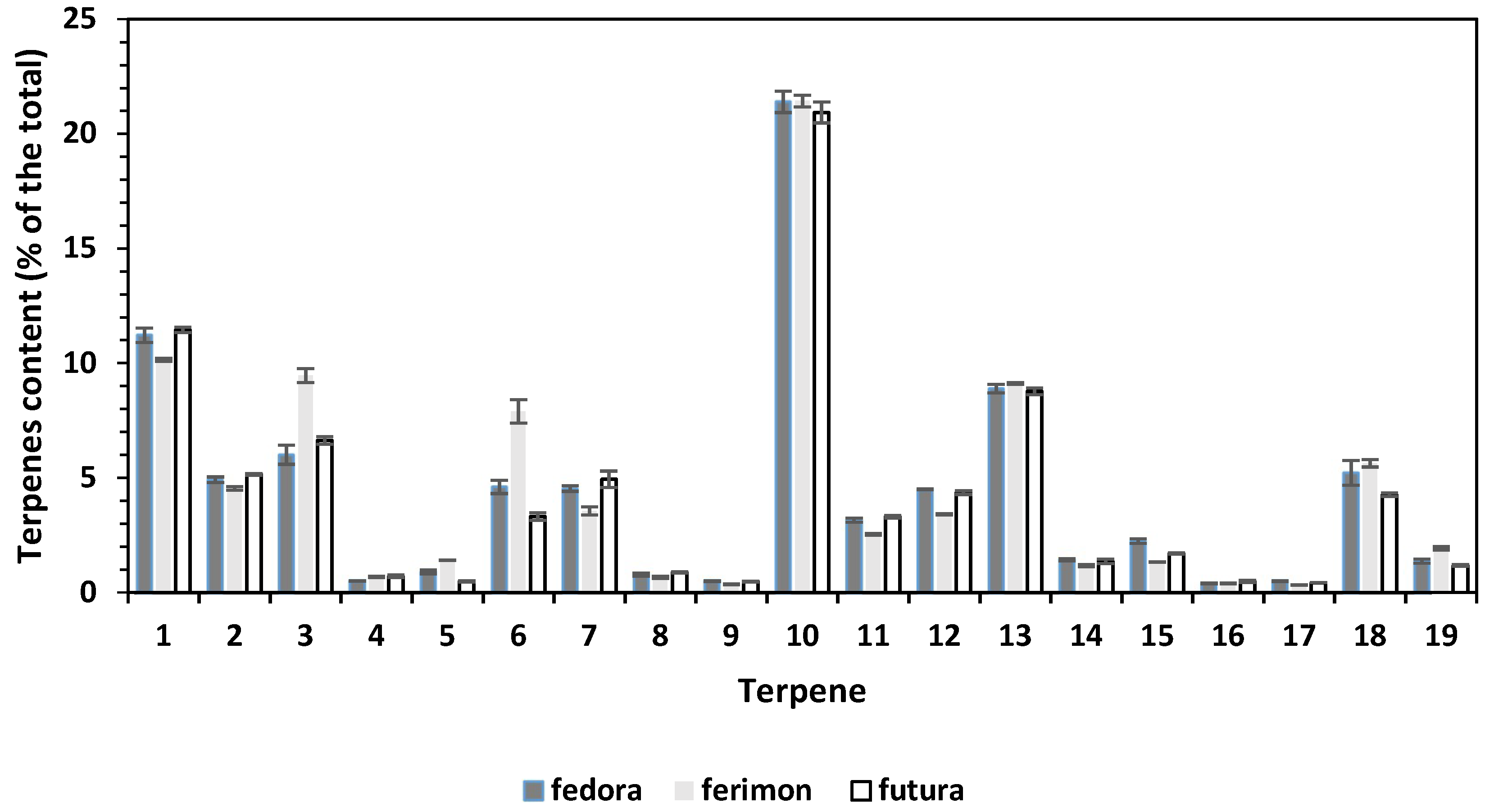
3.4. Characterization of EOs Composition
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Clarke, R.C.; Merlin, M.D. Cannabis Domestication, Breeding History, Present-day Genetic Diversity, and Future Prospects. CRC Crit. Rev. Plant Sci. 2016, 35, 293–327. [Google Scholar] [CrossRef]
- Calzolari, D.; Magagnini, G.; Lucini, L.; Grassi, G.; Appendino, G.B.; Amaducci, S. High added-value compounds from Cannabis threshing residues. Ind. Crops Prod. 2017, 108, 558–563. [Google Scholar] [CrossRef]
- Tang, K.; Struik, P.C.; Yin, X.; Calzolari, D.; Musio, S.; Thouminot, C.; Bjelková, M.; Stramkale, V.; Magagnini, G.; Amaducci, S. A comprehensive study of planting density and nitrogen fertilization effect on dual-purpose hemp (Cannabis sativa L.) cultivation. Ind. Crops Prod. 2017, 107, 427–438. [Google Scholar] [CrossRef]
- Welling, M.T.; Liu, L.; Shapter, T.; Raymond, C.A.; King, G.J. Characterisation of cannabinoid composition in a diverse Cannabis sativa L. germplasm collection. Euphytica 2017, 208, 463–475. [Google Scholar] [CrossRef]
- Hillig, K.W. A chemotaxonomic analysis of terpenoid variation in Cannabis. Biochem. Syst. Ecol. 2004, 32, 875–891. [Google Scholar] [CrossRef]
- Bakkali, F.; Averbeck, S.; Averbeck, D.; Idaomar, M. Biological effects of essential oils—A review. Food Chem. Toxicol. 2008, 46, 446–475. [Google Scholar] [CrossRef] [PubMed]
- Andre, C.M.; Hausman, J.-F.; Guerriero, G. Cannabis sativa: The plant of the thousand and one molecules. Front. Plant Sci. 2016, 7, 1–17. [Google Scholar] [CrossRef]
- Stahl, E.; Kunde, P. Die Leitsubstanzen der Haschisch-Suchhunde (Leading substances for hashish narcotic dogs). Kriminalistik 1973, 9, 385–388. [Google Scholar]
- Lanyon, V.S.; Turner, J.C.; Mahlberg, P.G. Quantitative analysis of cannabinoids in the secretory product from capitate-stalked glands of Cannabis sativa L. (Cannabaceae). Bot. Gaz. 1981, 142, 316–319. [Google Scholar] [CrossRef]
- Booth, J.K.; Page, J.E.; Bohlmann, J. Terpene synthases from Cannabis sativa. PLoS ONE 2017, 12, 1–20. [Google Scholar] [CrossRef]
- Meier, C.; Mediavilla, V. Factors influencing the yield and the quality of hemp (Cannabis sativa L.) essential oil. J. Int. Hemp Assoc. 1998, 5, 16–20. [Google Scholar]
- Fournier, G.; Paris, M.R.; Fourniat, M.C.; Quero, A.M. Activité bactériostatique d’huiles essentielles de Cannabis sativa L. [Bacteriostatic activity of Cannabis sativa L. essential oil.]. Ann. Pharm. Fr. 1978, 36, 603–606. [Google Scholar] [PubMed]
- Nissen, L.; Zatta, A.; Stefanini, I.; Grandi, S.; Sgorbati, B.; Biavati, B.; Monti, A. Characterization and antimicrobial activity of essential oils of industrial hemp varieties (Cannabis sativa L.). Fitoterapia 2010, 81, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Novak, J.; Zitterl-Eglseer, K.; Deans, S.G.; Franz, C.M. Essential oils of different cultivars of Cannabis sativa L. and their antimicrobial activity. Flavour Fragr. J. 2001, 16, 259–262. [Google Scholar] [CrossRef]
- Górski, R.; Sobieralski, K.; Siwulski, M. The effect of hemp essential oil on mortality Aulacorthum Solani Kalt. And Tetranychus Urticae Koch. Ecol. Chem. Eng. S 2016, 23, 505–511. [Google Scholar] [CrossRef]
- Verma, R.S.; Padalia, R.C.; Verma, S.K.; Chauhan, A.; Darokar, M.P. The essential oil of ‘bhang’ (Cannabis sativa L.) for non-narcotic applications. Curr. Sci. 2014, 107, 645–650. [Google Scholar]
- Bedini, S.; Flamini, G.; Cosci, F.; Ascrizzi, R.; Benelli, G.; Conti, B. Cannabis sativa and Humulus lupulus essential oils as novel control tools against the invasive mosquito Aedes albopictus and fresh water snail Physella acuta. Ind. Crops Prod. 2016, 85, 318–323. [Google Scholar] [CrossRef]
- Mukhtar, T.; Kayani, M.Z.; Hussain, M.A. Nematicidal activities of Cannabis sativa L. and Zanthoxylum alatum Roxb. against Meloidogyne incognita. Ind. Crops Prod. 2013, 42, 447–453. [Google Scholar] [CrossRef]
- Wielgusz, K.; Heller, K.; Byczyńska, M. The assessment of fungistatic effect of hemp essential oil as seed dressing, depending on the composition. J. Res. Appl. Agric. Eng. 2012, 57, 183–187. [Google Scholar]
- Synowiec, A.; Rys, M.; Bocianowski, J.; Wielgusz, K.; Byczyñska, M.; Heller, K.; Kalemba, D. Phytotoxic effect of fiber hemp essential oil on germination of some weeds and crops. J. Essent. Oil Bear. Plants 2016, 19, 262–276. [Google Scholar] [CrossRef]
- McPartland, J.M. Cannabis as repellent and pesticide. J. Int. Hemp Assoc. 1997, 4, 87–92. [Google Scholar]
- Pate, D.W. Chemical ecology of Cannabis. J. Int. Hemp Assoc. 1994, 2, 32–37. [Google Scholar]
- Wanas, A.S.; Radwan, M.M.; Mehmedic, Z.; Jacob, M.; Khan, I.A.; Elsohly, M.A. Antifungal activity of the volatiles of high potency Cannabis sativa L. against Cryptococcus neoformans. Rec. Nat. Prod. 2016, 10, 214–220. [Google Scholar]
- Hazekamp, A.; Fischedick, J.T. Cannabis—From cultivar to chemovar: Towards a better definition of Cannabis potency. Drug Test. Anal. 2012, 4, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.A.; ElSohly, M.A. The volatile oil composition of fresh and air-dried buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Fischedick, J.T.; Hazekamp, A.; Erkelens, T.; Choi, Y.H.; Verpoorte, R. Metabolic fingerprinting of Cannabis sativa L., cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes. Phytochemistry 2010, 71, 2058–2073. [Google Scholar] [CrossRef] [PubMed]
- Bertoli, A.; Tozzi, S.; Pistelli, L.; Angelini, L.G. Fibre hemp inflorescences: from crop-residues to essential oil production. Ind. Crops Prod. 2010, 32, 329–337. [Google Scholar] [CrossRef]
- Baldini, M.; Ferfuia, C.; Piani, B.; Sepulcri, A.; Dorigo, G.; Zuliani, F.; Danuso, F.; Cattivello, C. The performance and potentiality of monoecious hemp (Cannabis sativa L.) cultivars as a multipurpose crop. Agronomy 2018, 8, 162. [Google Scholar] [CrossRef]
- Mediavilla, V.; Steinemann, S. Essential oil of Cannabis sativa L. strains. J. Int. Hemp Assoc. 1997, 4, 80–82. [Google Scholar]
- Thomas, T.G.; Sharma, S.K.; Prakash, A.; Sharma, B.R. Insecticidal properties of essential oil of Cannabis sativa Linn against mosquito larvae. Entomon 2000, 25, 21–24. [Google Scholar]
- European Commission Plant Variety Databases-European Commission. Available online: https://ec.europa.eu/food/plant/plant_propagation_material/plant_variety_catalogues_databases_en (accessed on 15 April 2019).
- Tang, K.; Struik, P.C.; Yin, X.; Thouminot, C.; Bjelková, M.; Stramkale, V.; Amaducci, S. Comparing hemp (Cannabis sativa L.) cultivars for dual-purpose production under contrasting environments. Ind. Crops Prod. 2016, 87, 33–44. [Google Scholar] [CrossRef]
- Mediavilla, V.; Jonquera, M.; Schmid-Slembrouck, I.; Soldati, A. Decimal code for growth stages of hemp (Cannabis sativa L.). J. Int. Hemp Assoc. 1998, 5, 68–74. [Google Scholar]
- Commissione Permanente per la Revisione e la Pubblicazione della Farmacopea Ufficiale Farmacopea Ufficiale della Repubblica Italiana; XII; Istituto poligrafico e zecca dello Stato: Roma, Italy, 2008.
- Pellegrini, M.; Marchei, E.; Pacifici, R.; Pichini, S. A rapid and simple procedure for the determination of cannabinoids in hemp food products by gas chromatography-mass spectrometry. J. Pharm. Biomed. Anal. 2005, 36, 939–946. [Google Scholar] [CrossRef] [PubMed]
- Pavel, M. GC-MS analysis of essential oil obtained from the species Thymus comosus Heuff. Ex Griseb. (Lamiaceae). Farmacia 2009, 57, 7. [Google Scholar]
- Giese, M.W.; Lewis, M.A.; Giese, L.; Smith, K.M. Method for the analysis of cannabinoids and terpenes in Cannabis. J. AOAC Int. 2015, 98, 1503–1522. [Google Scholar] [CrossRef] [PubMed]
- Heinl, S.; Lerch, O.; Erdmann, F. Automated GC–MS determination of Δ9-tetrahydrocannabinol, cannabinol and cannabidiol in hair. J. Anal. Toxicol. 2016, 40, 498–503. [Google Scholar] [CrossRef] [PubMed]
- Aizpurua-Olaizola, O.; Soydaner, U.; Öztürk, E.; Schibano, D.; Simsir, Y.; Navarro, P.; Etxebarria, N.; Usobiaga, A. Evolution of the cannabinoid and terpene content during the growth of Cannabis sativa plants from different chemotypes. J. Nat. Prod. 2016, 79, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Jin, D.; Jin, S.; Yu, Y.; Lee, C.; Chen, J. Classification of Cannabis cultivars marketed in Canada for medical purposes by quantification of cannabinoids and terpenes using HPLC-DAD and GC-MS. J. Anal. Bioanal. Tech. 2017, 8. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2018. [Google Scholar]
- De Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research; Universidad Nacional Agraria La Molina: Lima, Peru, 2019. [Google Scholar]
- Brighenti, V.; Pellati, F.; Steinbach, M.; Maran, D.; Benvenuti, S. Development of a new extraction technique and HPLC method for the analysis of non-psychoactive cannabinoids in fibre-type Cannabis sativa L. (hemp). J. Pharm. Biomed. Anal. 2017, 143, 228–236. [Google Scholar] [CrossRef]
- De Backer, B.; Debrus, B.; Lebrun, P.; Theunis, L.; Dubois, N.; Decock, L.; Verstraete, A.; Hubert, P.; Charlier, C. Innovative development and validation of an HPLC/DAD method for the qualitative and quantitative determination of major cannabinoids in Cannabis plant material. J. Chromatogr. B 2009, 877, 4115–4124. [Google Scholar] [CrossRef]
- Medical Subject Headings (MeSH). Available online: https://www.ncbi.nlm.nih.gov/mesh (accessed on 23 June 2017).
- Flavor Extract Manufacturers Association (FEMA). Available online: https://www.femaflavor.org/ (accessed on 17 June 2017).
- Hazardous Substances Data Bank (HSDB). Available online: https://toxnet.nlm.nih.gov/newtoxnet/hsdb.htm (accessed on 21 June 2017).
- Human Metabolome Database (HMDB). Available online: http://www.hmdb.ca/ (accessed on 10 April 2019).
- US EPA (United States Environmental Protection Agency). Chemical Data Reporting under the Toxic Substances Control Act. Available online: https://www.epa.gov/chemical-data-reporting (accessed on 15 April 2019).
- Yadav, N.; Yadav, R.; Goyal, A. Chemistry of terpenoids. Int. J. Pharm. Sci. Rev. Res. 2014, 27, 272–278. [Google Scholar]
- Bernhammer, A.-K.; Fischer, L.; Mentler, B.; Heinritzi, M.; Simon, M.; Hansel, A. Production of highly oxygenated organic molecules (HOMs) from trace contaminants during isoprene oxidation. Atmospheric Meas. Tech. 2018, 11, 4763–4773. [Google Scholar] [CrossRef]
- Kotan, R.; Kordali, S.; Cakir, A. Screening of antibacterial activities of twenty-one oxygenated monoterpenes. Z. Nat. C 2007, 62, 507–513. [Google Scholar] [CrossRef]
- Beer, A.; Sagorchev, P.; Uzunova, J.; Lukanov, J. Effects of 1.8-cineol (eucalyptol) on the activity of histamine H1 receptors. SciFed J. Plant Physiol. 2017, 1, 1. Available online: http://scifedpublishers.com/fulltext/effects-of-18-cineol-eucalyptol-on-the-activity-of-histamine-h1-receptors/21840 (accessed on 10 April 2019).
- National Center for Biotechnology Information (NCBI). Available online: https://www.ncbi.nlm.nih.gov/ (accessed on 10 April 2019).
- Fidyt, K.; Fiedorowicz, A.; Strządała, L.; Szumny, A. β-caryophyllene and β-caryophyllene oxide-natural compounds of anticancer and analgesic properties. Cancer Med. 2016, 5, 3007–3017. [Google Scholar] [CrossRef] [PubMed]
| Parameters | S. Osvaldo 2016 | Campoformido 2017 | Aviano 2017 | Maiano 2017 |
|---|---|---|---|---|
| Sand (> 0.05 < 2 mm) (%) | 43 | 26 | 29 | 43 |
| Loam (> 0.002 < 0.05 mm) (%) | 40 | 59 | 53 | 50 |
| Clay (< 0.002 mm) (%) | 17 | 15 | 18 | 7 |
| pH (KCl 1:2.5) | 7.35 | 6.4 | 7.1 | 7.2 |
| Total calcareous (%) | 5.5 | 1 | 37 | 56 |
| Active calcium carbonate (%) | 0.2 | n/a | 1.6 | 1.4 |
| Organic matter (%) | 1.8 | 2.2 | 5.8 | 4.5 |
| Total nitrogen (g kg−1) | 1.85 | 1.9 | 3.7 | 2.8 |
| C/N | 10 | 6.8 | 9.2 | 9.4 |
| Phosphorus available (mg kg−1) | 34 | 47 | 26 | 30 |
| Potassium exchangeable (ppm) | 164 | 135 | 195 | 98 |
| Environment | Previous Crop | Soil Tillage | Fertilization 1 | Sowing–Emergence Time | Irrigation |
|---|---|---|---|---|---|
| S. Osvaldo-2016 | Wheat | Ploughing at 30 cm and n.2 pre-sowing harrowing | Pre-sowing: 0-60-0 N-P-K + 80 N in post-emergence | 20th–29th April | n.3 of 20 mm each |
| Aviano-2017 | Corn | Ploughing at 30 cm and n.2 pre-sowing harrowing | Pre-sowing: 18-18-25.5 of N-P-K + 50 N in post- emergence | 13th–24th April | n.4 of 20 mm each |
| Campoformido-2017 | Corn | Ploughing at 30 cm and n.2 pre-sowing harrowing | Pre-sowing: 5 t ha−1 of manure | 10th–22th April | - |
| Maiano-2017 | Chicory | Ploughing at 30 cm and n.2 pre-sowing harrowing | Pre-sowing: 0-60-0 of N-P-K + 60 N in post-emergence | 11th–23th April | - |
| Source of Variation | Degrees of Freedom | EOs a Concentration in Inflorescence | Inflorescence Yield | EOs Yield |
|---|---|---|---|---|
| Cultivar (C) | 2 | 34.38 *** | 21.28 *** | 27.13 *** |
| Environment (E) | 3 | 7.68 ** | 12.68 ** | 0.82 ns |
| C × E | 6 | 5.03 * | 5.05 * | 4.84 * |
| LRI | Aviano | Campoformido | Maiano | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Ferimon | Fedora | Futura | Ferimon | Fedora | Futura | Ferimon | Fedora | Futura | |
| α-Pinene | 942 | 10.12 ± 1.01 | 11.34 ± 1.45 | 11.64 ± 1.42 | 10.25 ± 1.92 | 10.62 ± 1.44 | 11.53 ± 0.34 | 10.06 ± 1.19 | 11.84 ± 1.40 | 11.36 ± 0.69 |
| Camphene | 946 | 0.19 ± 0.00 | 0.25 ± 0.03 | 0.28 ± 0.03 | 0.22 ± 0.01 | 0.24 ± 0.01 | 0.30 ± 0.02 | 0.21 ± 0.10 | 0.23 ± 0.02 | 0.27 ± 0.02 |
| β-Pinene | 984 | 4.49 ± 0.39 | 4.96 ± 0.53 | 5.24 ± 0.50 | 4.67 ± 0.53 | 4.68 ± 0.40 | 5.14 ± 0.13 | 4.43 ± 0.45 | 5.17 ± 0.24 | 5.15 ± 0.38 |
| β-Myrcene | 992 | 9.91 ± 1.33 | 5.41 ± 0.79 | 7.17 ± 0.79 | 8.95 ± 1.42 | 5.77 ± 0.56 | 6.39 ± 0.34 | 10.01 ± 1.08 | 6.68 ± 0.58 | 6.61 ± 0.36 |
| α-Phellandrene | 996 | tr | 0.22 ± 0.01 | 0.28 ± 0.03 | tr | 0.24 ± 0.03 | 0.25 ± 0.00 | tr | 0.23 ± 0.02 | 0.25 ± 0.00 |
| δ 3-Carene | 998 | tr | 1.55 ± 0.22 | 0.59 ± 0.22 | tr | 1.58 ± 0.24 | 0.52 ± 0.19 | 0.22 ± 0.00 | 1.86 ± 0.14 | 0.45 ± 0.10 |
| α-Terpinene | 1010 | - | tr | 0.25 ± 0.02 | - | tr | 0.22 ± 0.00 | - | tr | 0.23 ± 0.01 |
| o-Cymene | 1025 | tr | 0.21 ± 0.07 | - | - | 0.31 ± 0.11 | - | - | 0.32 ± 0.07 | tr |
| D-Limonene | 1035 | - | tr | - | tr | 2.55 ± 1.04 | - | - | 2.00 ± 1.15 | tr |
| D-Limonene +β-Phellandrene | 1035 | 2.45 ± 0.12 | 2.38 ± 0.33 | 2.72 ± 0.30 | 3.66 ± 0.26 | 2.63 ± 0.33 | 2.77 ± 0.15 | 3.90 ± 0.18 | 2.53 ± 0.32 | 2.78 ± 0.31 |
| β-Phellandrene | 1037 | tr | 0.48 ± 0.03 | 0.74 ± 0.26 | - | - | - | tr | 0.53 ± 0.31 | tr |
| 1,8 Cineol | 1040 | 0.88 ± 0.09 | 0.49 ± 0.04 | 0.64 ± 0.10 | 0.62 ± 0.04 | 0.49 ± 0.07 | 0.80 ± 0.06 | 0.71 ± 0.08 | 0.53 ± 0.04 | 0.70 ± 0.12 |
| trans-β-Ocimene | 1042 | 1.44 ± 0.28 | 0.76 ± 0.08 | 0.45 ± 0.05 | 1.40 ± 0.33 | 0.90 ± 0.31 | 0.43 ± 0.04 | 1.40 ± 0.10 | 1.08 ± 0.26 | 0.52 ± 0.05 |
| β-Ocimene | 1044 | 8.88 ± 2.01 | 4.93 ± 0.48 | 3.31 ± 0.32 | 6.94 ± 1.12 | 4.03 ± 1.47 | 2.92 ± 0.22 | 8.63 ± 0.48 | 4.89 ± 0.98 | 3.42 ± 0.31 |
| γ-Terpinene | 1064 | tr | tr | 0.24 ± 0.02 | tr | tr | 0.24 ± 0.01 | tr | tr | 0.24 ± 0.02 |
| cis-Sabinene hydrate | 1068 | - | - | - | - | tr | 0.18 ± 0.07 | - | - | tr |
| Terpinolene | 1079 | 3.45 ± 0.42 | 4.63 ± 0.49 | 5.82 ± 0.56 | 3.22 ± 0.83 | 4.29 ± 1.85 | 0.55 ± 0.11 | 3.81 ± 0.40 | 4.90 ± 0.67 | 4.83 ± 0.64 |
| Linalool | 1082 | - | tr | 0.21 ± 0.07 | tr | tr | 0.22 ± 0.08 | - | - | - |
| trans-(-)-Pinocarveol + 2-Pinen-10-ol | 1126 | tr | 0.22 ± 0.01 | tr | 0.24 ± 0.09 | 0.21 ± 0.01 | tr | tr | tr | |
| p-Cymen-8-ol | 1163 | - | - | - | - | 0.32 ± 0.12 | tr | - | 0.31 ± 0.09 | tr |
| Cosmen-2-ol | 1210 | tr | - | - | 0.32 ± 0.23 | tr | - | 0.27 ± 0.13 | - | tr |
| 2-Nonen-4-one, 2-methyl- | 1215 | - | tr | - | tr | - | - | - | - | tr |
| β-Curcumene | 1389 | tr | 0.23 ± 0.08 | tr | tr | 0.22 ± 0.01 | tr | tr | 0.24 ± 0.14 | tr |
| 7-epi-Sesquithujene | 1389 | tr | 0.21 ± 0.07 | tr | tr | tr | tr | tr | tr | tr |
| β-Elemene, (-) | 1387 | 0.25 ± 0.02 | tr | tr | tr | tr | tr | tr | tr | tr |
| Isocaryophyllene | 1406 | 0.67 ± 0.18 | 0.72 ± 0.13 | 0.81 ± 0.13 | 0.74 ± 0.13 | 0.89 ± 0.08 | 0.86 ± 0.11 | 0.56 ± 0.12 | 0.71 ± 0.11 | 0.92 ± 0.01 |
| cis-α-Bergamotene | 1416 | 0.56 ± 0.02 | 0.46 ± 0.04 | 0.47 ± 0.05 | 0.36 ± 0.03 | 0.52 ± 0.02 | 0.45 ± 0.02 | 0.34 ± 0.03 | 0.48 ± 0.04 | 0.46 ± 0.08 |
| Caryophyllene | 1420 | 22.00 ± 1.44 | 22.40 ± 1.48 | 20.52 ± 1.13 | 21.01 ± 0.98 | 20.72 ± 1.43 | 20.39 ± 1.31 | 21.88 ± 0.70 | 21.12 ± 1.18 | 21.43 ± 0.76 |
| trans-α-Bergamotene | 1434 | 2.55 ± 0.22 | 2.98 ± 0.20 | 3.36 ± 0.25 | 2.61 ± 0.24 | 3.28 ± 0.24 | 3.22 ± 0.10 | 2.48 ± 0.16 | 3.19 ± 0.21 | 3.35 ± 0.25 |
| α-Longipinene | 1444 | tr | 0.31 ± 0.11 | 0.26 ± 0.05 | 0.26 ± 0.18 | 0.27 ± 0.01 | 0.21 ± 0.00 | tr | 0.24 ± 0.01 | 0.32 ± 0.16 |
| cis-β-Farnesene | 1454 | 3.38 ± 0.15 | 4.46 ± 0.20 | 4.43 ± 0.27 | 3.47 ± 0.20 | 4.51 ± 0.21 | 4.20 ± 0.14 | 3.36 ± 0.29 | 4.51 ± 0.29 | 4.40 ± 0.27 |
| Humulene | 1457 | 9.20 ± 0.55 | 9.28 ± 0.29 | 8.62 ± 0.31 | 9.07 ± 0.16 | 8.67 ± 0.23 | 8.61 ± 0.37 | 9.18 ± 0.47 | 8.68 ± 0.16 | 8.88 ± 0.36 |
| Aromandendrene | 1462 | 1.12 ± 0.11 | 1.46 ± 0.10 | 1.29 ± 0.15 | 1.23 ± 0.27 | 1.46 ± 0.07 | 1.55 ± 0.10 | 1.07 ± 0.10 | 1.36 ± 0.15 | 1.20 ± 0.07 |
| β-Patchoulene | 1461 | tr | - | tr | - | tr | 0.34 ± 0.14 | - | tr | |
| γ-Gurjunene | 1461 | tr | 0.28 ± 0.05 | 0.39 ± 0.10 | 0.24 ± 0.17 | 0.30 ± 0.02 | 0.34 ± 0.15 | tr | 0.25 ± 0.04 | 0.32 ± 0.06 |
| α-Curcumene | 1473 | - | tr | - | tr | 0.33 ± 0.13 | tr | - | - | - |
| β-Selinene | 1489 | 2.12 ± 0.21 | 3.13 ± 1.12 | 2.19 ± 0.29 | 1.78 ± 0.26 | 3.25 ± 0.26 | 2.49 ± 0.09 | 1.76 ± 0.13 | 2.92 ± 0.27 | 2.43 ± 0.38 |
| Valencene | 1492 | tr | 1.17 ± 1.85 | 0.35 ± 0.06 | 0.23 ± 0.01 | 0.23 ± 0.00 | 0.44 ± 0.02 | tr | 0.23 ± 0.00 | 0.33 ± 0.05 |
| α-Selinene | 1496 | 1.25 ± 0.15 | 2.37 ± 0.21 | 1.55 ± 0.20 | 1.34 ± 0.21 | 2.30 ± 0.19 | 1.76 ± 0.06 | 1.31 ± 0.08 | 2.06 ± 0.07 | 1.70 ± 0.30 |
| α-Farnesene | 1503 | 0.45 ± 0.06 | 0.83 ± 0.13 | 0.51 ± 0.10 | 0.38 ± 0.02 | 0.73 ± 0.10 | 0.57 ± 0.03 | 0.38 ± 0.03 | 0.68 ± 0.18 | 0.58 ± 0.05 |
| β-Bisabolene | 1506 | 0.48 ± 0.15 | 0.71 ± 0.13 | 0.78 ± 0.13 | 0.58 ± 0.04 | 0.68 ± 0.14 | 0.74 ± 0.16 | 0.50 ± 0.02 | 0.64 ± 0.08 | 0.72 ± 0.10 |
| β-Elemene, (-)- | 1506 | tr | - | - | tr | tr | - | - | - | - |
| γ-Muurolene | 1506 | tr | 0.25 ± 0.09 | 0.24 ± 0.02 | 0.20 ± 0.14 | 0.22 ± 0.01 | 0.19 ± 0.01 | - | tr | tr |
| y-Gurjunene | 1506 | 0.15 ± 0.01 | 0.24 ± 0.08 | 0.34 ± 0.07 | 0.24 ± 0.01 | 0.21 ± 0.01 | 0.37 ± 0.08 | tr | 0.20 ± 0.11 | 0.32 ± 0.14 |
| γ-Cadinene | 1506 | - | 0.19 ± 0.06 | 0.27 ± 0.04 | - | tr | 0.17 ± 0.03 | - | tr | tr |
| Selina -3,7(11)-diene | 1507 | 0.43 ± 0.11 | 0.36 ± 0.07 | 0.46 ± 0.13 | 0.41 ± 0.19 | 0.42 ± 0.10 | 0.54 ± 0.13 | 0.36 ± 0.07 | 0.37 ± 0.03 | 0.41 ± 0.06 |
| β-Sesquiphellandrene | 1516 | 0.29 ± 0.03 | 0.50 ± 0.09 | 0.43 ± 0.07 | 0.33 ± 0.01 | 0.49 ± 0.08 | 0.41 ± 0.03 | 0.29 ± 0.01 | 0.46 ± 0.03 | 0.41 ± 0.03 |
| (±)-β-Acoradiene | 1518 | tr | 0.20 ± 0.07 | 0.24 ± 0.02 | 0.20 ± 0.14 | 0.23 ± 0.00 | 0.25 ± 0.01 | tr | tr | 0.23 ± 0.00 |
| δ-Cadinene | 1519 | 0.52 ± 0.25 | 0.47 ± 0.05 | 0.76 ± 0.10 | 0.49 ± 0.03 | 0.42 ± 0.07 | 0.88 ± 0.04 | 0.44 ± 0.07 | 0.37 ± 0.07 | 0.78 ± 0.06 |
| Nerolidol | 1560 | - | tr | tr | - | tr | - | - | - | tr |
| Germacrene B | 1570 | 0.88 ± 0.18 | 1.10 ± 0.15 | 0.92 ± 0.14 | 1.03 ± 0.33 | 0.82 ± 0.27 | 0.91 ± 0.07 | 0.96 ± 0.17 | 0.75 ± 0.07 | 1.03 ± 0.07 |
| Caryophyllene oxide | 1584 | 5.48 ± 0.88 | 4.29 ± 0.35 | 4.04 ± 0.65 | 5.95 ± 1.31 | 6.07 ± 1.91 | 4.33 ± 0.14 | 5.44 ± 0.60 | 5.03 ± 0.84 | 4.25 ± 0.34 |
| trans-Sesquisabinene hydrate | 1580 | tr | 0.21 ± 0.07 | tr | - | tr | - | - | tr | - |
| (-)-Globulol | 1590 | tr | 0.27 ± 0.06 | 0.30 ± 0.03 | 0.34 ± 0.24 | 0.27 ± 0.04 | 0.32 ± 0.02 | tr | 0.22 ± 0.00 | 0.26 ± 0.03 |
| Humulene epoxide 2 | 1592 | 1.88 ± 0.08 | 1.20 ± 0.17 | 1.10 ± 0.22 | 2.05 ± 0.61 | 1.45 ± 0.68 | 1.19 ± 0.06 | 1.81 ± 0.21 | 1.36 ± 0.19 | 1.17 ± 0.12 |
| Aromadendrene oxide-(2) | 1594 | - | tr | - | tr | 0.93 ± 1.01 | tr | - | tr | - |
| β-Guaiene | 1599 | 0.41 ± 0.20 | 0.29 ± 0.07 | 0.43 ± 0.10 | 0.35 ± 0.10 | 0.35 ± 0.03 | 0.50 ± 0.05 | 0.30 ± 0.04 | 0.24 ± 0.14 | 0.45 ± 0.03 |
| Isovalencenol | 1640 | 0.33 ± 0.05 | 0.36 ± 0.13 | 0.30 ± 0.05 | 0.35 ± 0.24 | 0.26 ± 0.03 | 0.36 ± 0.04 | 0.24 ± 0.12 | tr | 0.30 ± 0.03 |
| Isoaromadendrene epoxide | 1645 | 0.44 ± 0.11 | 0.35 ± 0.11 | 0.34 ± 0.06 | 0.48 ± 0.20 | 0.35 ± 0.06 | 0.37 ± 0.04 | 0.34 ± 0.09 | 0.23 ± 0.00 | 0.38 ± 0.04 |
| Allohimachalol | 1652 | - | 0.19 ± 0.06 | tr | - | - | - | - | - | - |
| α-Bisabolol | 1646 | - | - | 0.22 ± 0.07 | - | 0.25 ± 0.02 | 0.12 ± 0.01 | - | tr | 0.29 ± 0.02 |
| 7-epi-cis-sesquisabinene hydrate | 1660 | tr | 0.39 ± 0.14 | 0.30 ± 0.04 | 0.22 ± 0.15 | 0.21 ± 0.01 | 0.12 ± 0.03 | tr | tr | 0.39 ± 0.19 |
| Cannabidiol | 2431 | 0.25 ± 0.05 | 0.85 ± 0.15 | 0.37 ± 0.03 | tr | 0.22 ± 0.01 | 0.37 ± 0.04 | 0.27 ± 0.08 | tr | 0.97 ± 0.12 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vuerich, M.; Ferfuia, C.; Zuliani, F.; Piani, B.; Sepulcri, A.; Baldini, M. Yield and Quality of Essential Oils in Hemp Varieties in Different Environments. Agronomy 2019, 9, 356. https://doi.org/10.3390/agronomy9070356
Vuerich M, Ferfuia C, Zuliani F, Piani B, Sepulcri A, Baldini M. Yield and Quality of Essential Oils in Hemp Varieties in Different Environments. Agronomy. 2019; 9(7):356. https://doi.org/10.3390/agronomy9070356
Chicago/Turabian StyleVuerich, Marco, Claudio Ferfuia, Fabio Zuliani, Barbara Piani, Angela Sepulcri, and Mario Baldini. 2019. "Yield and Quality of Essential Oils in Hemp Varieties in Different Environments" Agronomy 9, no. 7: 356. https://doi.org/10.3390/agronomy9070356
APA StyleVuerich, M., Ferfuia, C., Zuliani, F., Piani, B., Sepulcri, A., & Baldini, M. (2019). Yield and Quality of Essential Oils in Hemp Varieties in Different Environments. Agronomy, 9(7), 356. https://doi.org/10.3390/agronomy9070356





