Nematode Management in the Strawberry Fields of Southern Spain
Abstract
1. Introduction
- To determine the prevalence, abundance, and incidence of plant-parasitic nematodes in the strawberry fields in Southern Spain.
- To determine the host suitability to M. hapla of currently cropped strawberry cultivars.
- To establish plant damage and reproductive function models for M. hapla in strawberry.
- To compare the efficacies of various soil disinfestation methods against populations of M. hapla and P. penetrans.
2. Materials and Methods
2.1. Nematological Survey
2.1.1. A Poll on Perception of Nematode Caused Diseases on Strawberries
2.1.2. Nematological Field Survey of Strawberry Fields
2.2. Host Suitability of Strawberry Cultivars
2.2.1. Production of M. hapla Inoculum
2.2.2. Establishment of Pot Experiments
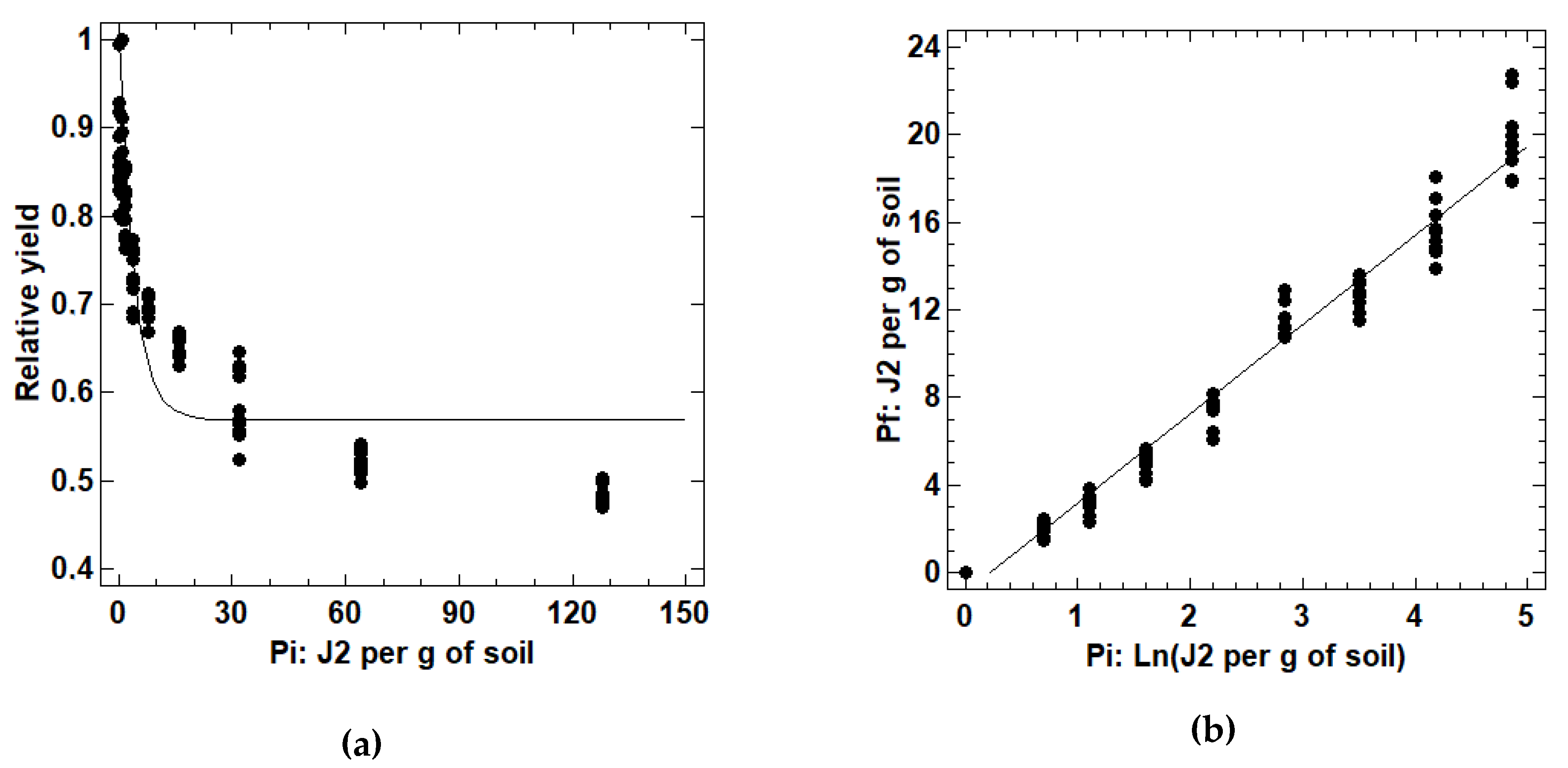
2.3. Estimation of Plant Damage and Reproductive Function Models for M. hapla in Strawberry
2.3.1. Production of M. hapla Inoculum
2.3.2. Establishment of Pot Experiments
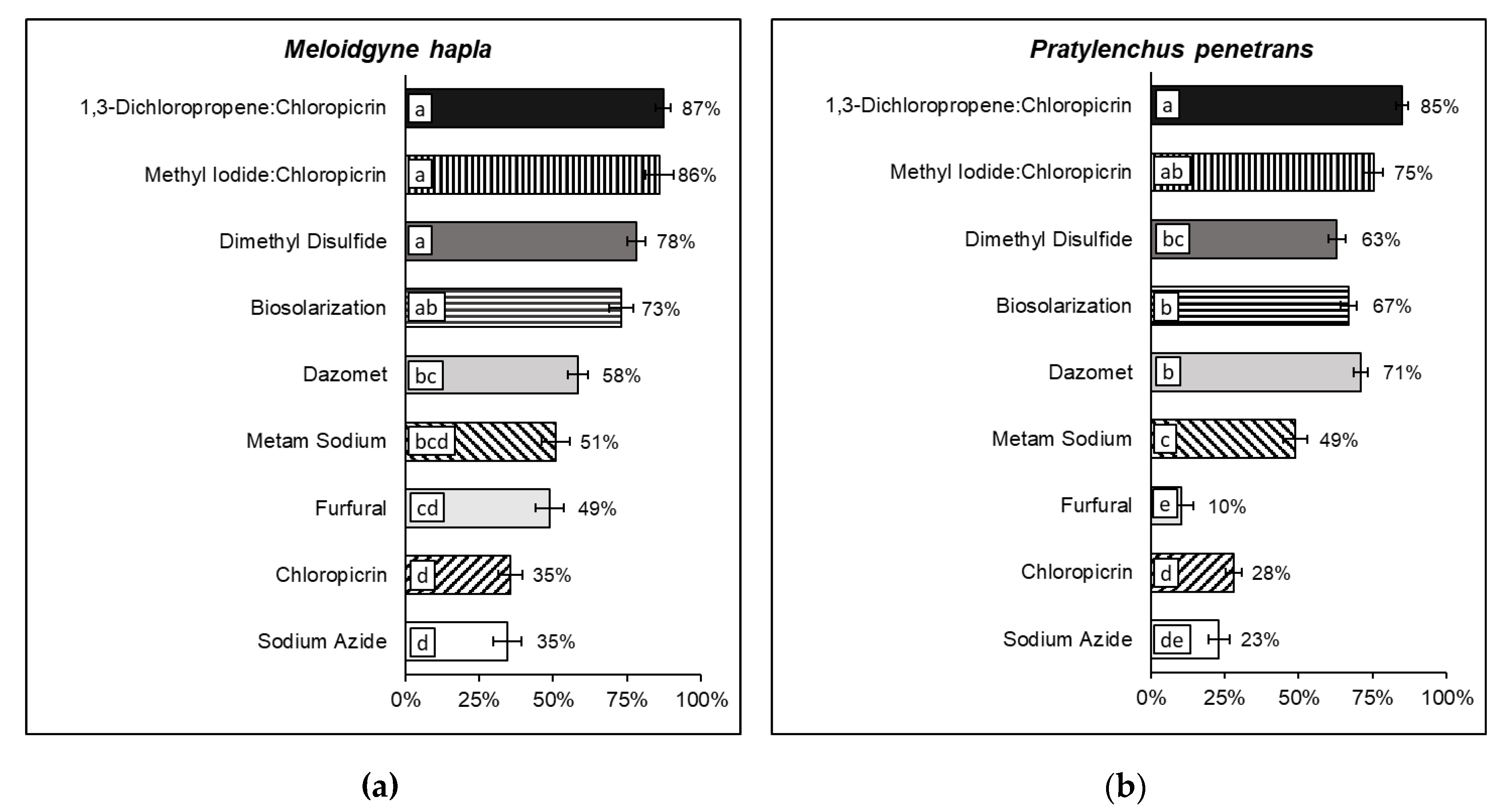
2.4. Soil Disinfection Efficacy in Field Trials
2.4.1. Experimental Fields and Strawberry Growing Conditions
2.4.2. Soil Disinfestation Treatments
2.4.3. Estimation of Soil Nematode Densities
2.5. Statistical Analyses
3. Results
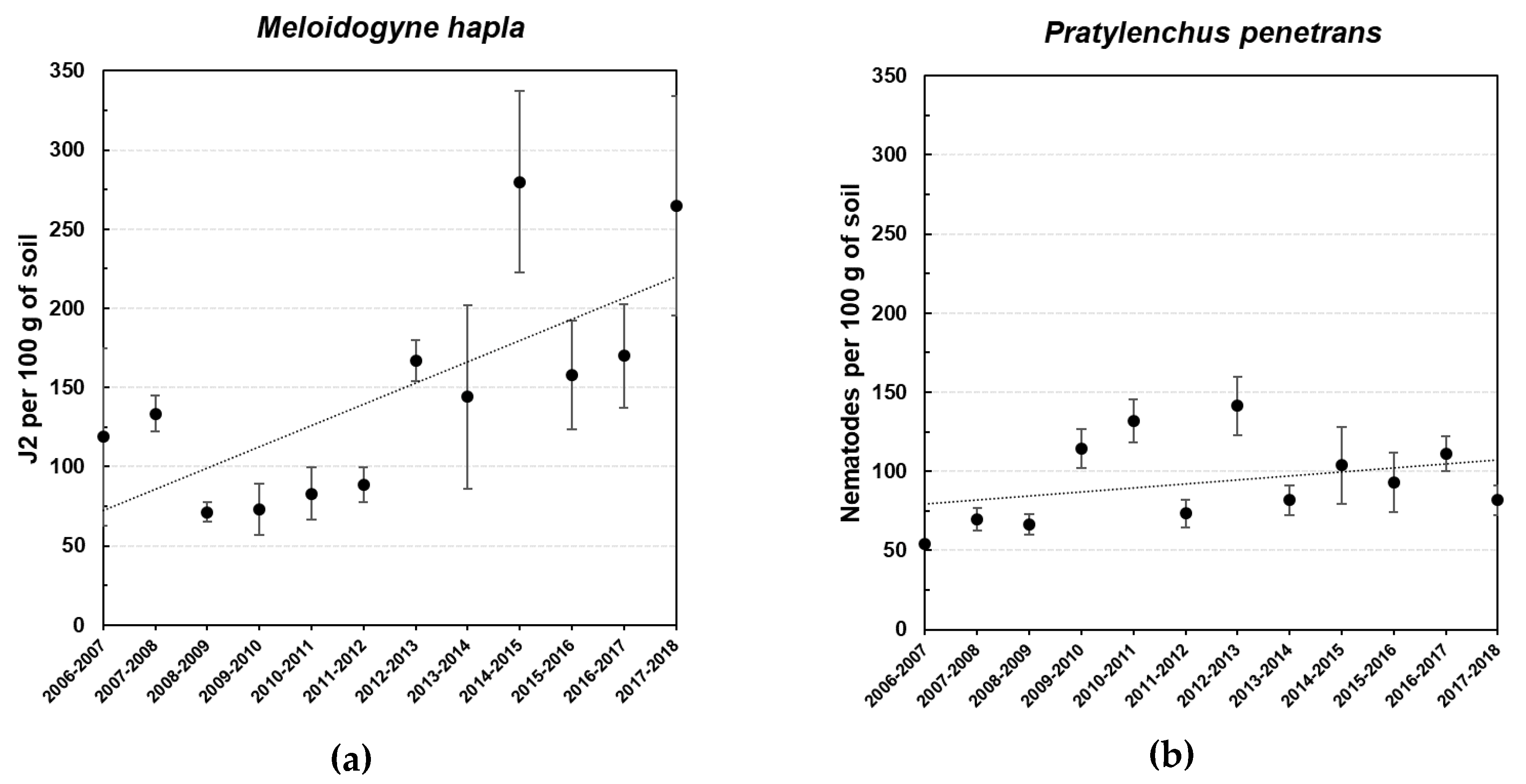
3.1. Nematological Survey
3.1.1. A Poll on Perception of Nematode Caused Diseases in Strawberries
3.1.2. Nematological Field Survey
3.2. Host Suitability of Strawberry Cultivars
3.3. Estimation of Plant Damage and Reproductive Function Models for M. hapla in Strawberry
3.4. Efficacy of Soil Disinfection in Field Trials
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Available online: http://www.fao.org/faostat (accessed on 16 January 2019).
- Ministerio de Agricultura, Pesca y Alimentación. Anuario de Estadística 2017. Available online: https://www.mapa.gob.es/es/estadistica/temas/publicaciones/anuario-de-estadistica/2017/ (accessed on 16 January 2019).
- Brown, D.J.F.; Dalmasso, A.; Trudgill, D.L. Nematode pests of soft fruits and vines. In Plant Parasitic Nematodes in Temperate Agriculture; Evans, K., Trudgill, D.L., Webster, J.M., Eds.; CAB International: Wallingford, UK, 1993; pp. 427–462. ISBN 978-0851988085. [Google Scholar]
- Bélair, G.; Khanizadeh, S. Distribution of plant-parasitic nematodes in strawberry and raspberry fields in Quebec. Phytoprotection 1994, 75, 101–107. [Google Scholar] [CrossRef][Green Version]
- Samaliev, H.Y.; Mohamedova, M. Plant-parasitic nematodes associated with strawberry (Fragaria ananassa Duch.) in Bulgaria. Bulg. J. Agric. Sci. 2011, 17, 730–735. [Google Scholar]
- Nyoike, T.W.; Mekete, T.; McSorley, R.; Weibelzahl-Karigi, E.; Liburd, O.E. Confirmation of Meloidogyne hapla on strawberry in Florida using molecular and morphological techniques. Nematropica 2012, 42, 253–259. [Google Scholar]
- Noling, J.W. Estimating strawberry yield and sting nematode impacts using counts of plant sizes and fruit stems. Proc. Fla. State Hort. Soc. 2011, 124, 197–201. [Google Scholar]
- Vega, J.M.; Páez, J.; López-Aranda, J.M.; Medina, J.J.; Miranda, L.; Montes, F. Nematodes on strawberries in Southern Spain. Effects on yield and control of Meloidogyne hapla. Nematology 2002, 4, 309. [Google Scholar]
- RAIF. Balance Fitosanitario. Provincia de Huelva. Fresa. Campaña 2016–2017; Red de Alerta e Información Fitosanitaria, Consejería de Agricultura, Pesca y Desarrollo Rural, Junta de Andalucía: Sevilla, Spain, 2017; pp. 1–7. ISBN 978-8484382422. [Google Scholar]
- Bascón, J.C.; Arcos, S.; Páez, J.I.; Avilés, M.; Robertson, L.; Vega, J.M.; Navas, A. Hemicycliophora spp. ¿Un nuevo problema en el cultivo de la fresa en Huelva? In Proceedings of the Resúmenes del XVI Congreso de la Sociedad Española de Fitopatología, Málaga, Spain, 17–21 September 2012; p. 336. [Google Scholar]
- Peña-Santiago, R.; Castillo, P.; Escuer, M.; Guerrero, P.; Talavera, M.; Vieira, P. Tylenchid Species (Nematoda, Tylenchida) Recorded in the Iberian Peninsula and the Balearic Islands: A Compendium; Universidad de Jaén: Jaén, Spain, 2004; pp. 1–127. ISBN 978-8484392422. [Google Scholar]
- Seinhorst, J.W. The relation between nematode density and damage to plants. Nematologica 1965, 11, 137–154. [Google Scholar] [CrossRef]
- Ehwaeti, M.E.; Phillips, M.S.; Trudgill, D.L. Dynamics of damage to tomato by Meloidogyne incognita. Fund. Appl. Nematol. 1998, 21, 627–635. [Google Scholar]
- Greco, N.; Di Vito, M. Population dynamics and damage levels. In Root Knot Nematodes; Perry, R.N., Moens, M., Starr, J.L., Eds.; CAB International: Wallingford, UK, 2009; pp. 246–274. ISBN 978-1845934927. [Google Scholar]
- Schomaker, C.H.; Been, T.H. Plant growth and population dynamics. In Plant Nematology; Perry, R.N., Moens, M., Eds.; CAB International: Wallingford, UK, 2006; pp. 275–301. ISBN 978-1845930561. [Google Scholar]
- López-Aranda, J.M.; Miranda, L.; Medina, J.J.; Soria, C.; de los Santos, B.; Romero, F.; Pérez-Jiménez, R.; Talavera, M.; Fennimore, S.A.; Santos, B.M. Methyl bromide alternatives for high tunnel strawberry production in southern Spain. HortTechnology 2009, 19, 187–192. [Google Scholar] [CrossRef]
- López-Aranda, J.M.; Domínguez, P.; Miranda, L.; de Los Santos, B.; Talavera, M.; Daugovish, O.; Soria, C.; Chamorro, M.; Medina, J.J. Fumigant use for strawberry production in Europe: The Current Landscape and Solutions. Int. J. Fruit Sci. 2016, 16, 1–15. [Google Scholar] [CrossRef]
- Medina, J.J.; Miranda, L.; Domínguez, P.; Soria, C.; Pérez-Jiménez, R.M.; Zea, T.; Talavera, M.; Velasco, L.; Romero, F.; De los Santos, B.; et al. Comparison of different chemical and non-chemical alternatives to Methyl Bromide for strawberry in Huelva (Spain). J. Berry Res. 2012, 2, 113–121. [Google Scholar] [CrossRef]
- Watson, T.T.; Desaeger, J.A. Evaluation of non-fumigant chemical and biological nematicides for strawberry production in Florida. Crop Prot. 2019, 117, 100–107. [Google Scholar] [CrossRef]
- Domínguez, P.; Miranda, L.; Medina, J.J.; de los Santos, B.; Talavera, M.; Daugovish, O.; Soria, C.; Chamorro, M.; López-Aranda, J.M. Evaluation of non-fumigant alternative soil treatments for strawberry production in Huelva (Spain). Int. J. Fruit Sci. 2016, 16, 28–36. [Google Scholar] [CrossRef]
- Edwards, W.H.; Jones, R.K.; Schmitt, D.P. Host suitability and parasitism of selected strawberry cultivars by Meloidogyne hapla and M. incognita. Plant Dis. 1985, 69, 40–42. [Google Scholar] [CrossRef]
- Khanizadeh, S.; Belair, G.; Lareau, M.J. Relative susceptibility of five strawberry cultivars to Meloidogyne hapla under three soil water deficit levels. Phytoprotection 2012, 75, 133–137. [Google Scholar] [CrossRef][Green Version]
- Pinkerton, J.; Finn, C.E. Responses of strawberry species and cultivars to the root-lesion and northern root-knot nematodes. HortScience 2005, 40, 33–38. [Google Scholar]
- Dale, A.; Potter, J. Strawberry cultivars vary in their resistance to northern lesion nematode. J. Nematol. 1998, 30, 577–580. [Google Scholar] [PubMed]
- Verdejo-Lucas, S.; Talavera, M. Pathogenic potential. parasitic success and host efficiency of Meloidogyne incognita and M. javanica on cucurbitaceous plant genotypes. Eur. J. Plant Pathol. 2018. [Google Scholar] [CrossRef]
- SIOSE 2013. Base Cartográfica de Ocupación del Suelo en Andalucía. Available online: http://www.juntadeandalucia.es/medioambiente/mapwms/REDIAM_siose_2013 (accessed on 30 March 2017).
- Whitehead, A.G.; Hemming, J.R. A comparison of some quantitative methods of extracting small vermiform nematodes from soil. Ann. Appl. Biol. 1965, 55, 25–38. [Google Scholar] [CrossRef]
- Hartman, K.M.; Sasser, J.N. Identification of Meloidogyne species on the basis of differential host test and perineal-pattern morphology. In An Advanced Treatise on Meloidogyne. Biology and Control; Barker, K.R., Carter, C.C., Sasser, J.N., Eds.; North Carolina State University: Raleigh, NC, USA, 1985; Volume I, pp. 69–77. ISBN 978-0931901014. [Google Scholar]
- Esbenshade, P.R.; Triantaphyllou, A.C. Electrophoretic methods for the study of root-knot nematode enzymes. In An Advanced Treatise on Meloidogyne. Methodology; Barker, K.R., Carter, C.C., Sasser, J.N., Eds.; North Carolina State University: Raleigh, NC, USA, 1985; Volume II, pp. 115–123. ISBN 978-0931901022. [Google Scholar]
- Hussey, R.S.; Barker, K.R. A comparison of methods of collecting inocula of Meloidogyne spp., including a new technique. Plant Dis. Rep. 1973, 57, 1025–1028. [Google Scholar]
- Hussey, R.S.; Janssen, G.J.W. Root-knot nematode species. In Plant Resistance to Parasitic Nematodes; Starr, J.L., Cook, R., Bridge, J., Eds.; CAB International: Wallingford, UK, 2002; pp. 43–70. ISBN 978-0851994660. [Google Scholar]
- López-Aranda, J.M.; Medina, J.J.; Marsal, J.I.; Bartual, R. Strawberry production in Spain. In The Strawberry: A Book for Growers. Others; Childers, N.F., Ed.; IFAS Publ. University Florida: Gainesville, FL, USA, 2002; pp. 230–237. ISBN 978-0938378112. [Google Scholar]
- Schneider-Orelli, O. Entomologisches Praktikum; Verlag Sauerländer: Aarau, Germany, 1947; pp. 1–149. [Google Scholar]
- Talavera, M.; Sayadi, S.; Chirosa-Ríos, M.; Salmerón, T.; Flor-Peregrín, E.; Verdejo-Lucas, S. Perception of the impact of root-knot nematode-induced diseases in horticultural protected crops of south-eastern Spain. Nematology 2012, 14, 517–527. [Google Scholar] [CrossRef]
- Andreu, M.; Salmerón, T.; Martínez, V.; Tobar, A. Nematodos fitoparásitos de los cultivos hortícolas de Trigueros (Huelva). Bol San. Veg. Plagas 1986, 12, 319–322. [Google Scholar]
- Horst, R.K. Nematodes. In Westcott’s Plant Disease Handbook; Horst, R.K., Ed.; Springer: Dordrecht, The Netherlands, 2013; pp. 251–268. ISBN 978-9400721401. [Google Scholar]
- Lamondia, J.A.; Martin, S.B. The influence of Pratylenchus penetrans and temperature on black root rot of strawberry by binucleate Rhizoctonia spp. Plant Dis. 1989, 73, 107–110. [Google Scholar] [CrossRef]
- Avilés, M.; Castillo, S.; Bascón, J.; Zea-Bonilla, T.; Martín-Sánchez, P.M.; Pérez-Jiménez, R.M. First report of Macrophomina phaseolina causing crown and root rot of strawberry in Spain. Plant Pathol. 2008, 57, 382. [Google Scholar] [CrossRef]
- Avilés, M.; Bascón, J.; Gallardo, M.A.; Orta, M.S.; Borrero, C. New foci of strawberry Fusarium wilt in Huelva (Spain) and susceptibility of the most commonly used cultivars. Sci. Hortic. 2017, 226, 85–90. [Google Scholar] [CrossRef]
- Inserra, R.N.; O’bannon, J.H.; Di Vito, M.; Ferris, H. Response of two alfalfa cultivars to Meloidogyne hapla. J. Nematol. 1983, 15, 644–645. [Google Scholar]
- Gugino, B.K.; Abawi, G.S.; Ludwig, J.W. Damage and management of Meloidogyne hapla using oxamyl on carrot in New York. J. Nematol. 2006, 38, 483–490. [Google Scholar]
- Watanabe, T.; Masumura, H.; Kioka, Y.; Noguchi, K.; Min, Y.Y.; Murakami, R.; Toyota, K. Development of a direct quantitative detection method for Meloidogyne incognita and M. hapla in andosol and analysis of relationship between the initial population of Meloidogyne spp. and yield of eggplant in an andosol. Nematol. Res. 2013, 43, 21–29. [Google Scholar] [CrossRef]
- Brodie, B.B.; Evans, K.; Franco, J. Nematode parasites of potatoes. In Plant Parasitic Nematodes in Temperate Agriculture; Evans, K., Trudgill, D.L., Webster, J.M., Eds.; CAB International: Wallingford, UK, 1993; pp. 87–132. ISBN 978-0851988085. [Google Scholar]
- Potter, J.W.; Olthof, T.H.A. Yield losses in fall-maturing vegetables relative to population densities of Pratylenchus penetrans and Meloidogyne hapla. Phytopathology 1974, 64, 1072–1075. [Google Scholar] [CrossRef]
- Viaene, N.; Abawi, G.S. Damage Threshold of Meloidogyne hapla to Lettuce in Organic Soil. J. Nematol. 1996, 28, 537–545. [Google Scholar]
- Potter, J.W.; Olthof, T.H.A. Effects of population densities Meloidogyne hapla on growth and yield of tomato. J. Nematol. 1977, 9, 296–300. [Google Scholar]
- Aalders, L.T.; Minchin, R.; Hill, R.A.; Braithwaite, M.; Bell, N.L.; Stewart, A. Development of a tomato/root knot nematode bioassay to screen beneficial microbes. N. Z. Plant Prot. 2009, 62, 28–33. [Google Scholar]
- Lahtinen, A.E.; Trudgill, D.L.; Thlikkala, K. Threshold temperature and minimum time requirements for the complete life cycle of Meloidogyne hapla from northern Europe. Nematologica 1988, 34, 443–451. [Google Scholar]
- Giné, A.; López-Gómez, M.; Vela, M.D.; Ornat, C.; Talavera, M.; Verdejo-Lucas, S.; Sorribas, F.J. Thermal requirements and population dynamics of root-knot nematodes on cucumber and yield losses under protected cultivation. Plant Pathol. 2014, 63, 1446–1453. [Google Scholar] [CrossRef]
- Vela, M.D.; Giné, A.; López-Gómez, M.; Sorribas, F.J.; Ornat, C.; Verdejo-Lucas, S.; Talavera, M. Thermal time requirements of root-knot nematodes on zucchini-squash and population dynamics with associated yield losses on spring and autumn cropping cycles. Eur. J. Plant Pathol. 2014, 140, 481–490. [Google Scholar] [CrossRef]
- López-Gómez, M.; Giné, A.; Vela, M.D.; Ornat, C.; Sorribas, F.J.; Talavera, M.; Verdejo-Lucas, S. Damage functions and thermal requirements of Meloidogyne javanica and Meloidogyne incognita on watermelon. Ann. Appl. Biol. 2014, 165, 466–473. [Google Scholar] [CrossRef]
- Santos, B.M.; Gilreath, J.P.; Motis, T. Managing nutsedge and stunt nematode in pepper with reduced Methyl Bromide plus chloropicrin rates under Virtually Impermeable Films. HortTechnology 2005, 15, 596–599. [Google Scholar] [CrossRef]
- Cabrera, J.A.; Hanson, B.D.; Gerik, J.S.; Gao, S.; Qin, R.; Wang, D. Pre-plant soil fumigation with reduced rates under low permeability films for nursery production, orchard and vineyard replanting. Crop. Prot. 2015, 75, 34–39. [Google Scholar] [CrossRef]
- Gómez-Tenorio, M.A.; Zanón, M.J.; de Cara, M.; Lupión, B.; Tello, J.C. Efficacy of dimethyl disulfide (DMDS) against Meloidogyne sp. and three formae speciales of Fusarium oxysporum under controlled conditions. Crop Prot. 2015, 78, 263–269. [Google Scholar] [CrossRef]
- Cabrera, J.A.; Wang, D.; Gerik, J.S.; Gan, J. Spot drip application of dimethyl disulfide as a post-plant treatment for the control of plant parasitic nematodes and soilborne pathogens in grape production. Manag. Sci. 2014, 70, 115–1157. [Google Scholar] [CrossRef]
- Mao, L.; Yan, D.; Wang, Q.; Li, Y.; Ouyang, C.; Liu, P.; Shen, J.; Guo, M.; Cao, A. Evaluation of the Combination of Dimethyl Disulfide and Dazomet as an Efficient Methyl Bromide Alternative for Cucumber Production in China. J. Agric. Food Chem. 2014, 62, 4864–4869. [Google Scholar] [CrossRef]
- Desaeger, J.A.; Seebold, K.W.; Csinos, A.S. Effect of application timing and method on efficacy and phytotoxicity of 1,3-D, chloropicrin and metam-sodium combinations in squash plasticulture. Pest Manag. Sci. 2008, 64, 230–238. [Google Scholar] [CrossRef]
- Schneider, S.M.; Ajwa, H.A.; Trout, T.J.; Gao, S. Nematode control from shank- and drip-applied fumigant alternatives to methyl bromide. HortScience 2008, 43, 1826–1832. [Google Scholar] [CrossRef]
- Chamorro, M.; Domínguez, P.; Medina, J.J.; Miranda, L.; Soria, C.; Romero, F.; López-Aranda, J.M.; Daugovish, O.; Mertely, J.; De los Santos, B. Assessment of chemical and biosolarization treatments for the control of Macrophomina phaseolina in strawberries. Sci. Hortic. 2015, 192, 361–368. [Google Scholar] [CrossRef]
- Benlioǧlu, S.; Boz, Ö.; Yildiz, A.; Kaşkavalci, G.; Benlioǧlu, K. Alternative soil solarization treatments for the control of soil-borne diseases and weeds of strawberry in the western Anatolia of Turkey. J. Phytopathol. 2005, 153, 423–430. [Google Scholar] [CrossRef]
| Soil Treatment | Dosage (kg/ha) | Application | Plastic Mulch | n |
|---|---|---|---|---|
| Untreated Control | – | – | PE | 36 |
| 1,3-dichloropropene:chloropicrin (61:33) | 300–400 | Shank/Drip | PE/VIF | 33 |
| Chloropicrin | 300–400 | Shank | PE/VIF | 12 |
| Dazomet | 300–500 | Broadcast | PE | 18 |
| Dimethyl-disulphide | 400–600 | Shank/Drip | PE/VIF | 21 |
| Furfural | 600 | Drip | PE/VIF | 9 |
| Metam-sodium | 153 | Drip | PE | 9 |
| Methyl iodide:chloropicrin (33:67) | 150–300 | Shank | VIF | 9 |
| Sodium-azide | 125–160 | Drip | PE/VIF | 9 |
| Biosolarization with chicken manure | 20,000–25,000 | Broadcast | PE | 12 |
| Soil Treatment | Dosage (kg/ha) | Application | Plastic Mulch | n |
|---|---|---|---|---|
| Untreated Control | - | - | PE | 36 |
| 1,3-dichloropropene:chloropicrin (61:33) | 300–400 | Shank/Drip | PE/VIF | 36 |
| Chloropicrin | 300–400 | Shank | PE/VIF | 21 |
| Dazomet | 300–500 | Broadcast | PE/VIF | 27 |
| Dimethyl-disulphide | 400–600 | Shank | PE/VIF | 18 |
| Furfural | 600 | Drip | PE/VIF | 9 |
| Metam-sodium | 153 | Shank | PE | 9 |
| Methyl iodide:chloropicrin (33:67) | 150–300 | Shank | VIF | 15 |
| Sodium-azide | 125–160 | Drip | PE/VIF | 9 |
| Biosolarization with chicken manure | 20,000–25,000 | Broadcast | PE | 21 |
| Prevalence (%) | Abundance | ||
|---|---|---|---|
| Meloidogyne hapla | 71 | 452 | (20–2560) |
| Meloidogyne incognita | 8 | 122 | (13–480) |
| Meloidogyne javanica | 6 | 320 | (33–890) |
| Meloidogyne sp. | 6 | 8 | (8–11) |
| Pratylenchus penetrans | 20 | 27 | (3–94) |
| Hemicycliophora spp. | 20 | 143 | (5–1200) |
| Ditylenchus dipsaci | 6 | 9 | (5–20) |
| Cultivar | Yield Loss (%) | Gall Index | Pf/Pi | |
|---|---|---|---|---|
| Calderon | 8.0 ± 0.1 a | 0.7 ± 0.5 a | 2.1 ± 0.3 | bcd |
| Calinda | 13.9 ± 0.1 a | 1.3 ± 0.4 a | 4.2 ± 0.3 | a |
| Candonga | 14.9 ± 0.1 a | 0.3 ± 0.4 a | 2.1 ± 0.3 | bcd |
| Charlene | 9.8 ± 0.1 a | 0.7 ± 0.4 a | 3.3 ± 0.2 | ab |
| Flaminia | 8.8 ± 0.1 a | 1.5 ± 0.6 a | 2.1 ± 0.4 | bcd |
| Flavia | 8.7 ± 0.1 a | 2.0 ± 0.5 a | 3.2 ± 0.3 | abc |
| Fortuna | 11.0 ± 0.1 a | 0.3 ± 0.3 a | 2.3 ± 0.2 | bcd |
| Marisol | 11.6 ± 0.1 a | 0.5 ± 0.3 a | 1.4 ± 0.2 | d |
| Marquis | 12.7 ± 0.1 a | 1.2 ± 0.3 a | 1.8 ± 0.2 | cd |
| Melissa | 8.0 ± 0.1 a | 0.8 ± 0.4 a | 1.9 ± 0.3 | cd |
| Palmeritas | 15.4 ± 0.1 a | 1.0 ± 0.5 a | 2.3 ± 0.3 | bcd |
| Petaluma | 11.1 ± 0.1 a | 0.7 ± 0.5 a | 2.1 ± 0.3 | bcd |
| Primoris | 12.2 ± 0.1 a | 0.3 ± 0.3 a | 2.5 ± 0.2 | bcd |
| Rabida | 19.2 ± 0.1 a | 1.0 ± 0.3 a | 2.1 ± 0.2 | bcd |
| Rociera | 15.4 ± 0.1 a | 1.0 ± 0.2 a | 1.5 ± 0.2 | d |
| Sabrina | 8.3 ± 0.1 a | 0.6 ± 0.4 a | 2.5 ± 0.2 | bcd |
| Savana | 10.0 ± 0.1 a | 0.5 ± 0.6 a | 2.2 ± 0.4 | bcd |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Talavera, M.; Miranda, L.; Gómez-Mora, J.A.; Vela, M.D.; Verdejo-Lucas, S. Nematode Management in the Strawberry Fields of Southern Spain. Agronomy 2019, 9, 252. https://doi.org/10.3390/agronomy9050252
Talavera M, Miranda L, Gómez-Mora JA, Vela MD, Verdejo-Lucas S. Nematode Management in the Strawberry Fields of Southern Spain. Agronomy. 2019; 9(5):252. https://doi.org/10.3390/agronomy9050252
Chicago/Turabian StyleTalavera, Miguel, Luis Miranda, José Antonio Gómez-Mora, María Dolores Vela, and Soledad Verdejo-Lucas. 2019. "Nematode Management in the Strawberry Fields of Southern Spain" Agronomy 9, no. 5: 252. https://doi.org/10.3390/agronomy9050252
APA StyleTalavera, M., Miranda, L., Gómez-Mora, J. A., Vela, M. D., & Verdejo-Lucas, S. (2019). Nematode Management in the Strawberry Fields of Southern Spain. Agronomy, 9(5), 252. https://doi.org/10.3390/agronomy9050252






