Application of Nano-Silicon Dioxide Improves Salt Stress Tolerance in Strawberry Plants
Abstract
:1. Introduction
2. Materials and Methods
2.1. Growth Conditions and Treatments
2.2. Phenotypic Measurements
2.3. Statistical Analysis
3. Results
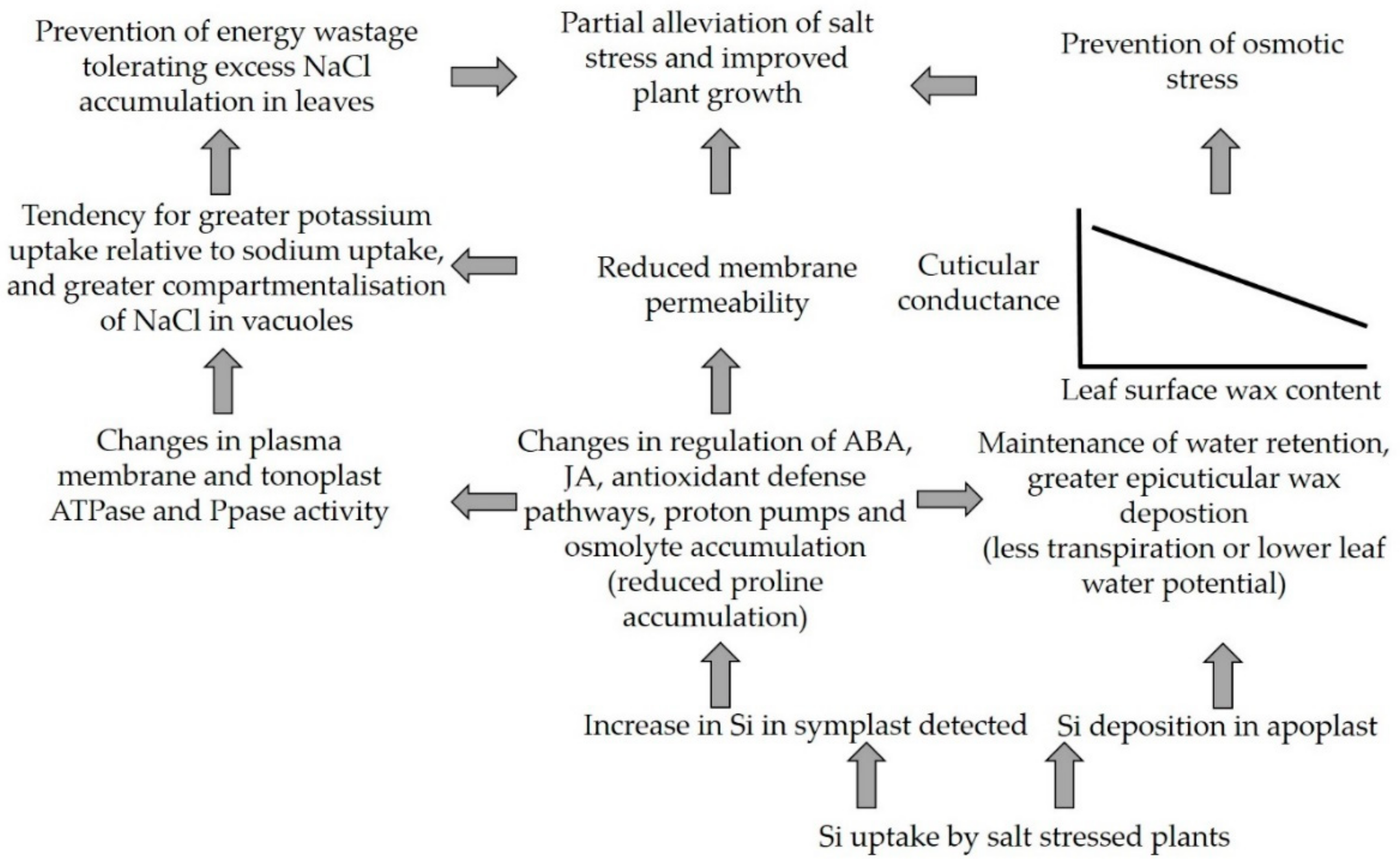
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gómez-del-Campo, M.; Baeza, P.; Ruiz, C.; Lissarrague, J.R. Water-stress induced physiological changes in leaves of four container-grown grapevine cultivars (Vitis vinifera L.). VITIS J. Grapevine Res. 2015, 43, 99. [Google Scholar]
- Azizinya, S.; Ghanadha, M.R.; Zali, A.A.; Samadi, B.Y.; Ahmadi, A. An evaluation of quantitative traits related to drought resistance in synthetic wheat genotypes in stress and non-stress conditions. Iran. J. Agric. Sci. 2005, 36, 281–293. [Google Scholar]
- Malakuti, M.J.; Keshavarz, P.; Karimian, N. Comprehensive Diagnosis and Optimal Fertilizer Recommendation for Sustainable Agriculture; Tarbiat Modares University Press: Tehran, Iran, 2008; p. 132. [Google Scholar]
- Sun, Y.; Niu, G.; Wallace, R.; Masabni, J.; Gu, M. Relative Salt Tolerance of Seven Strawberry Cultivars. Horticulturae 2015, 1, 27–43. [Google Scholar]
- Rousseau-Gueutin, M.; Lerceteau-Köhler, E.; Barrot, L.; Sargent, D.J.; Monfort, A.; Simpson, D.; Arús, P.; Guérin, G.; Denoyes-Rothan, B. Comparative genetic mapping between octoploid and diploid fragaria species reveals a high level of colinearity between their genomes and the essentially disomic behavior of the cultivated octoploid strawberry. Genetics 2008, 179, 2045–2060. [Google Scholar] [CrossRef] [PubMed]
- Flam-Shepherd, R.; Huynh, W.Q.; Coskun, D.; Hamam, A.M.; Britto, D.T.; Kronzucker, H.J. Membrane fluxes, bypass flows, and sodium stress in rice: The influence of silicon. J. Exp. Bot. 2018, 69, 1679–1692. [Google Scholar] [CrossRef]
- Bao-Shan, L.; Shao-Qi, D.; Chun-Hui, L.; Li-Jun, F.; Shu-Chun, Q.; Min, Y. Effect of TMS (nanostructured silicon dioxide) on growth of Changbai larch seedlings. J. For. 2004, 15, 138–140. [Google Scholar] [CrossRef]
- Alva, A.K.; Mattos, D.; Paramasivam, S.; Patil, B.; Dou, H.; Sajwan, K.S. Potassium Management for Optimizing Citrus Production and Quality. Int. J. Fruit Sci. 2006, 6, 3–43. [Google Scholar] [CrossRef]
- Gao, X.; Zou, C.; Wang, L.; Zhang, F. Silicon Decreases Transpiration Rate and Conductance from Stomata of Maize Plants. J. Plant Nutr. 2006, 29, 1637–1647. [Google Scholar] [CrossRef]
- Gao, X.; Zou, C.; Wang, L.; Zhang, F. Silicon Improves Water Use Efficiency in Maize Plants. J. Plant Nutr. 2005, 27, 1457–1470. [Google Scholar] [CrossRef]
- Naranjo, E.M.; Andrades-Moreno, L.; Davy, A.J. Silicon alleviates deleterious effects of high salinity on the halophytic grass Spartina densiflora. Plant Physiol. Biochem. 2013, 63, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.; Kamran, A.; Asif, M.; Qadeer, U.; Ahmed, Z.I.; Goyal, A. Silicon priming: A potential source to impart abiotic stress tolerance in wheat: A review. Aust. J. Crop Sci. 2013, 7, 484. [Google Scholar]
- Shi, Y.; Wang, Y.; Flowers, T.J.; Gong, H. Silicon decreases chloride transport in rice (Oryza sativa L.) in saline conditions. J. Plant Physiol. 2013, 170, 847–853. [Google Scholar] [CrossRef]
- Agarie, S. Effect of silicon on growth, dry matter production and photosynthesis in rice plants. Crop Prod. Improv. Tech. Asia 1993, 225–234. [Google Scholar]
- Prasad, T.; Sudhakar, P.; Sreenivasulu, Y.; Latha, P.; Munaswamy, V.; Reddy, K.R.; Sreeprasad, T.S.; Sajanlal, P.R.; Pradeep, T. Effect of nanoscale zinc oxide particles on the germination, growth and yield of peanut. J. Plant Nutr. 2012, 35, 905–927. [Google Scholar] [CrossRef]
- Wang, L.-J.; Wang, Y.-H.; Li, M.; Fan, M.-S.; Zhang, F.-S.; Wu, X.-M.; Yang, W.-S.; Li, T.-J. Synthesis of ordered biosilica materials. Chin. J. Chem. 2002, 20, 107–110. [Google Scholar] [CrossRef]
- Samuels, A.L. The Effects of Silicon Supplementation on Cucumber Fruit: Changes in Surface Characteristics. Ann. Bot. 1993, 72, 433–440. [Google Scholar] [CrossRef]
- Jenks, M.A.; Ashworth, E.N. Plant epicuticular waxes: Function, production, and genetics. Hortic. Rev. 1999, 23, 1–68. [Google Scholar]
- González, A.; Ayerbe, L. Effect of terminal water stress on leaf epicuticular wax load, residual transpiration and grain yield in barley. Euphytica 2010, 172, 341–349. [Google Scholar] [CrossRef]
- Cameron, K.D.; Teece, M.A.; Smart, L.B. Increased accumulation of cuticular wax and expression of lipid transfer protein in response to periodic drying events in leaves of tree tobacco. Plant Physiol. 2006, 140, 176–183. [Google Scholar] [CrossRef]
- Abdi, S.; Abbaspur, N.; Avestan, S.; Barker, A.V. Physiological responses of two grapevine (Vitis vinifera L.) cultivars to CycocelTM treatment during drought. J. Hortic. Sci. Biotechnol. 2016, 91, 211–219. [Google Scholar] [CrossRef]
- Hasheminasab, H.; Assad, M.T.; Aliakbari, A.; Sahhafi, S.R. Evaluation of some physiological traits associated with improved drought tolerance in Iranian wheat. Annu. Biol. Res. 2012, 3, 1719–1725. [Google Scholar]
- Yang, R.-C.; Jana, S.; Clarke, J.M. Phenotypic Diversity and Associations of Some Potentially Drought-responsive Characters in Durum Wheat. Crop. Sci. 1991, 31, 1484–1491. [Google Scholar] [CrossRef]
- Sairam, R.K. Effect of moisture-stress on physiological activities of two contrasting wheat genotypes. Indian J. Exp. Biol. 1994, 32, 594. [Google Scholar]
- David, M. Osmotic adjustment capacity and cuticular transpiration in several wheat cultivars cultivated in Algeria. Rom. Agric. Res. 2009, 26, 29–33. [Google Scholar]
- Dong, B.; Shi, L.; Shi, C.; Qiao, Y.; Liu, M.; Zhang, Z. Grain yield and water use efficiency of two types of winter wheat cultivars under different water regimes. Agric. Water Manag. 2011, 99, 103–110. [Google Scholar] [CrossRef]
- Åström, H.; Metsovuori, E.; Saarinen, T.; Lundell, R.; Hänninen, H. Morphological characteristics and photosynthetic capacity of Fragaria vesca L. winter and summer leaves. Flora Morphol. Distrib. Funct. Ecol. Plants 2015, 215, 33–39. [Google Scholar] [CrossRef]
- Li, X.-W.; Jiang, J.; Zhang, L.-P.; Yu, Y.; Ye, Z.-W.; Wang, X.-M.; Zhou, J.-Y.; Chai, M.-L.; Zhang, H.-Q.; Arus, P.; et al. Identification of volatile and softening-related genes using digital gene expression profiles in melting peach. Tree Genet. Genomes 2015, 11. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I.D. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Avestan, S.; Naseri, L.; Barker, A.V. Evaluation of nanosilicon dioxide and chitosan on tissue culture of apple under agar-induced osmotic stress. J. Plant Nutr. 2017, 40, 2797–2807. [Google Scholar] [CrossRef]
- Chen, W.; Yao, X.; Cai, K.; Chen, J. Silicon alleviates drought stress of rice plants by improving plant water status, photosynthesis and mineral nutrient absorption. Biol. Trace Elem. Res. 2011, 142, 67–76. [Google Scholar] [CrossRef]
- Yin, L.; Wang, S.; Liu, P.; Wang, W.; Cao, D.; Deng, X.; Zhang, S. Silicon-mediated changes in polyamine and 1-aminocyclopropane-1-carboxylic acid are involved in silicon-induced drought resistance in Sorghum bicolor L. Plant Physiol. Biochem. 2014, 80, 268–277. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Chen, K.; Chen, G.; Wang, S.; Zhang, C. Effects of Silicon on Growth of Wheat Under Drought. J. Plant Nutr. 2003, 26, 1055–1063. [Google Scholar] [CrossRef]
- Hattori, T.; Sonobe, K.; Inanaga, S.; An, P.; Morita, S. Effects of Silicon on Photosynthesis of Young Cucumber Seedlings Under Osmotic Stress. J. Plant Nutr. 2008, 31, 1046–1058. [Google Scholar] [CrossRef]
- Lu, C.; Zhang, C.; Wen, J.; Wu, G.; Tao, M. Research of the effect of nanometer materials on germination and growth enhancement of Glycine max and its mechanism. Soybean Sci. 2001, 21, 168–171. [Google Scholar]
- Zheng, L.; Hong, F.; Lu, S.; Liu, C. Effect of Nano-TiO2 on Strength of Naturally Aged Seeds and Growth of Spinach. Boil. Trace Elem. Res. 2005, 104, 083–092. [Google Scholar] [CrossRef]
- Haghighi, M.; Pessarakli, M. Influence of silicon and nano-silicon on salinity tolerance of cherry tomatoes (Solanum lycopersicum L.) at early growth stage. Sci. Hortic. 2013, 161, 111–117. [Google Scholar] [CrossRef]
- Nayyar, H.; Walia, D. Water Stress Induced Proline Accumulation in Contrasting Wheat Genotypes as Affected by Calcium and Abscisic Acid. Boil. Plant. 2003, 46, 275–279. [Google Scholar] [CrossRef]
- Pei, Z.; Ming, D.F.; Liu, D.; Wan, G.L.; Geng, X.X.; Gong, H.J.; Zhou, W.J. Silicon improves the tolerance to water-deficit stress induced by polyethylene glycol in wheat (Triticum aestivum L.) seedlings. J. Plant Growth Regul. 2010, 29, 106–115. [Google Scholar] [CrossRef]
- Lee, S.K.; Sohn, E.Y.; Hamayun, M.; Yoon, J.Y.; Lee, I.J. Effect of silicon on growth and salinity stress of soybean plant grown under hydroponic system. Agrofor. Syst. 2010, 80, 333–340. [Google Scholar] [CrossRef]
- Jain, M.; Mathur, G.; Koul, S.; Sarin, N. Ameliorative effects of proline on salt stress-induced lipid peroxidation in cell lines of groundnut (Arachis hypogaea L.). Plant Cell Rep. 2001, 20, 463–468. [Google Scholar] [CrossRef]
- Moussa, H.R. Influence of exogenous application of silicon on physiological response of salt-stressed maize (Zea mays L.). Int. J. Agric. Biol. 2006, 8, 293–297. [Google Scholar]
- Surendar, K.K.S.K.K.; Devi, D.D.D.D.D.; Ravi, I.R.I.; Jeyakumar, P.J.P.; Kumar, S.R.K.S.R.; Velayudham, V.K. Impact of water deficit on epicuticular wax, proline and free amino acid content and yield of banana cultivars and hybrids. Plant Gene Trait 2013, 4. [Google Scholar] [CrossRef]
- Zhu, J.-K. Salt and Drought Stress Signal Transduction in Plants. Annu. Rev. Plant Boil. 2002, 53, 247–273. [Google Scholar] [CrossRef]
- Marafon, A.C.; Endres, L. Silicon: Fertilization and nutrition in higher plants. Rev. Cienc. Amazon. J. Agric. Environ. Sci. 2013. [Google Scholar] [CrossRef]
- Liang, Y.; Nikolic, M.; Bélanger, R.; Gong, H.; Song, A. Silicon in Agriculture: From Theory to Practice; Springer: New York, NY, USA, 2015; p. 235. [Google Scholar]
- Romero-Aranda, M.R.; Jurado, O.; Cuartero, J. Silicon alleviates the deleterious salt effect on tomato plant growth by improving plant water status. J. Plant Physiol. 2006, 163, 847–855. [Google Scholar] [CrossRef]
- Tuna, A.L.; Kaya, C.; Higgs, D.; Murillo-Amador, B.; Aydemir, S.; Girgin, A.R. Silicon improves salinity tolerance in wheat plants. Environ. Exp. Bot. 2008, 62, 10–16. [Google Scholar] [CrossRef]
- Tahir, M.A.; Aziz, T.; Farooq, M.; Sarwar, G. Silicon-induced changes in growth, ionic composition, water relations, chlorophyll contents and membrane permeability in two salt-stressed wheat genotypes. Arch. Agron. Soil Sci. 2012, 58, 247–256. [Google Scholar] [CrossRef]
- Zuccarini, P. Effects of silicon on photosynthesis, water relations and nutrient uptake of Phaseolus vulgaris under NaCl stress. Boil. Plant. 2008, 52, 157–160. [Google Scholar] [CrossRef]
- Savant, N.K.; Korndörfer, G.H.; Datnoff, L.E.; Snyder, G.H. Silicon nutrition and sugarcane production: A review 1. J. Plant Nutr. 1999, 22, 1853–1903. [Google Scholar] [CrossRef]
- Yaghubi, K.; Ghaderi, N.; Vafaee, Y.; Javadi, T. Potassium silicate alleviates deleterious effects of salinity on two strawberry cultivars grown under soilless pot culture. Sci. Hortic. 2016, 213, 87–95. [Google Scholar] [CrossRef]
- Blum, A.; Shpiler, L.; Golan, G.; Mayer, J. Yield stability and canopy temperature of wheat genotypes under drought-stress. Field Crop Res. 1989, 22, 289–296. [Google Scholar] [CrossRef]
- Ndiso, J.; Chemining’Wa, G.; Olubayo, F.; Saha, H. Effect of drought stress on canopy temperature, growth and yield performance of cowpea varieties. Int. J. Plant Soil Sci. 2016, 9, 1–12. [Google Scholar] [CrossRef]
- Shepherd, T.; Griffiths, D.W. The effects of stress on plant cuticular waxes. New Phytol. 2006, 171, 469–499. [Google Scholar] [CrossRef] [PubMed]
- Mohammed, S. The Role of Leaf Epicuticular Wax an Improved Adaptation to Moisture Deficit Environments in Wheat. Ph.D. Thesis, Texas A & M University, College Station, TX, USA, May 2014. [Google Scholar]
- Keller, C.; Rizwan, M.; Davidian, J.C.; Pokrovsky, O.S.; Bovet, N.; Chaurand, P.; Meunier, J.D. Effect of silicon on wheat seedlings (Triticum turgidum L.) grown in hydroponics and exposed to 0 to 30 µM Cu. Planta 2015, 241, 847–860. [Google Scholar] [CrossRef]
- Sahebi, M.; Hanafi, M.M.; Akmar, A.S.N.; Rafii, M.Y.; Azizi, P.; Tengoua, F.F.; Azwa, J.N.M.; Shabanimofrad, M. Importance of Silicon and Mechanisms of Biosilica Formation in Plants. BioMed Int. 2015, 2015, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Prychid, C.J.; Rudall, P.J.; Gregory, M. Systematics and Biology of Silica Bodies in Monocotyledons. Bot. Rev. 2003, 69, 377–440. [Google Scholar] [CrossRef]
- Fawe, A.; Menzies, J.G.; Chérif, M.; Bélanger, R.R. Silicon and disease resistance in dicotyledons. In Studies Plant Science; Elsevier: Amsterdam, The Netherlands, 2001; Volume 8, pp. 159–169. [Google Scholar]
- Pozza, E.A.; Pozza, A.A.A.; Botelho, D.M.D.S. Silicon in plant disease control. Rev. Ceres 2015, 62, 323–331. [Google Scholar] [CrossRef]
- Zhang, W.; Xie, Z.; Lang, D.; Cui, J.; Zhang, X. Beneficial effects of silicon on abiotic stress tolerance in legumes. J. Plant Nutr. 2017, 40, 2224–2236. [Google Scholar] [CrossRef]
- Agarie, S.; Uchida, H.; Agata, W.; Kubota, F.; Kaufman, P.B. Effects of Silicon on Transpiration and Leaf Conductance in Rice Plants (Oryza sativa L.). Plant Prod. Sci. 1998, 1, 89–95. [Google Scholar] [CrossRef]

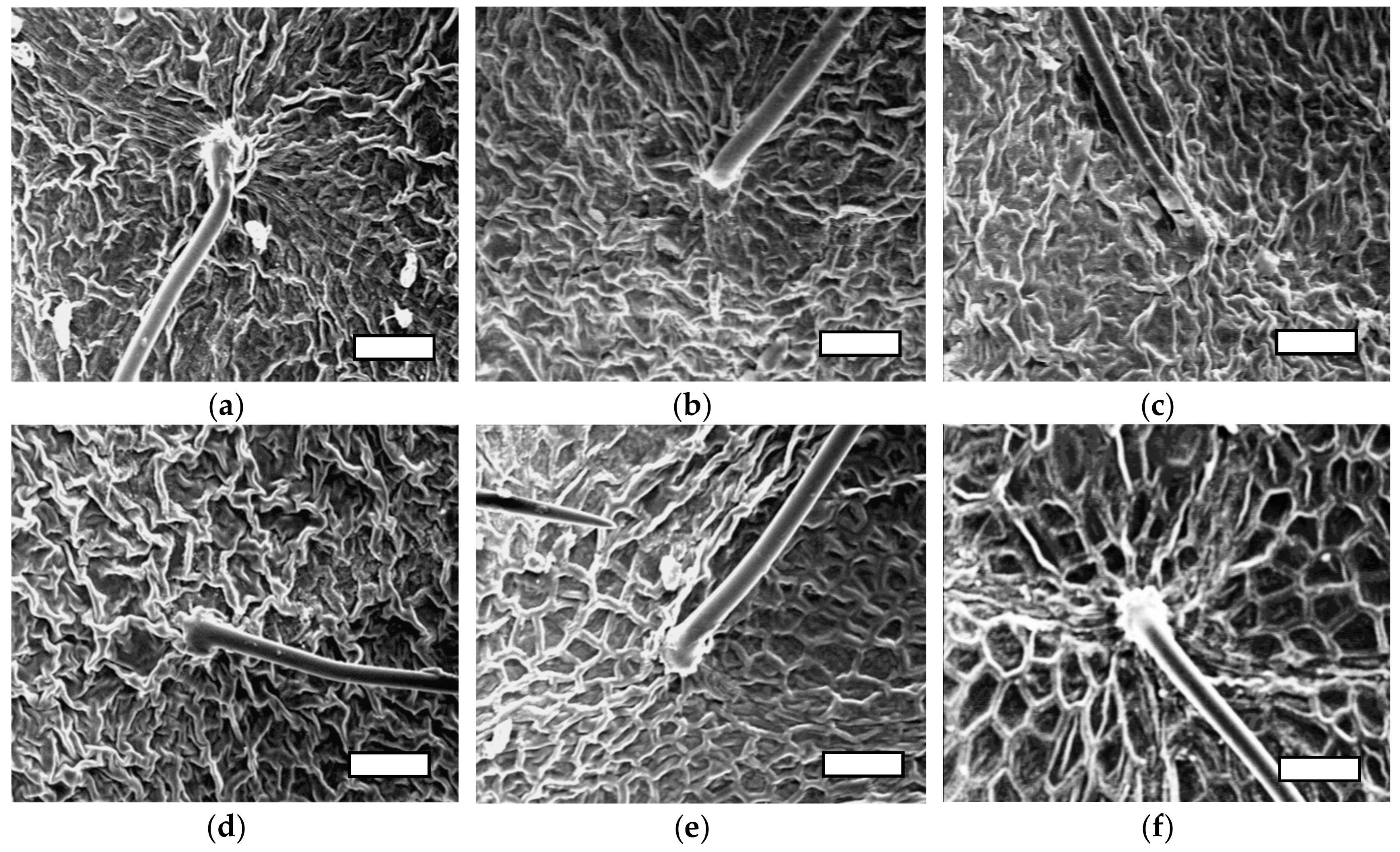

| Salinity (mM) | nSiO2 mg L−1 before BBCH: 61 | nSiO2 mg L−1 after BBCH: 61 | Treatments | |
|---|---|---|---|---|
| 0 mM (Control—no NaCl) | 0 | 0 (Control—no NaCl, no SiO2) | S1 | 0 mM NaCl + 0 mg L−1 nSiO2 |
| 50 | S2 | 0 mM NaCl + 0.50 mg L−1 SiO2 | ||
| 50 | 0 | S3 | 0 mM NaCl + 50. 0 mg L−1 SiO2 | |
| 50 | S4 | 0 mM NaCl + 50.50 mg L−1 SiO2 | ||
| 100 | 0 | S5 | 0 mM NaCl + 100.0 mg L−1 SiO2 | |
| 50 | S6 | 0 mM NaCl + 100.50 mg L−1 SiO2 | ||
| 25 mM | 0 | 0 (Control—no SiO2) | S1 | 25 mM NaCl + 0 mg L−1 nSio2 |
| 50 | S2 | 25 mM NaCl + 0.50 mg L−1 SiO2 | ||
| 50 | 0 | S3 | 25 mM NaCl + 50.0 mg L−1 SiO2 | |
| 50 | S4 | 25 mM NaCl + 50.50 mg L−1 SiO2 | ||
| 100 | 0 | S5 | 25 mM NaCl + 100.0 mg L−1 SiO2 | |
| 50 | S6 | 25 mM NaCl + 100.50 mg L−1 SiO2 | ||
| 50 mM | 0 | 0 (Control—no SiO2) | S1 | 50 mM NaCl + 0 mg L−1 nSio2 |
| 50 | S2 | 50 mM NaCl + 0.50 mg L−1 SiO2 | ||
| 50 | 0 | S3 | 50 mM NaCl + 50.0 mg L−1 SiO2 | |
| 50 | S4 | 50 mM NaCl + 50.50 mg L−1 SiO2 | ||
| 100 | 0 | S5 | 50 mM NaCl+ 100.0 mg L−1 SiO2 | |
| 50 | S6 | 50 mM NaCl+ 100.50 mg L−1 SiO2 | ||
| Root Fresh Weight (g) | Root Dry Weight (g) | Shoot Fresh Weight (g) | Shoot Dry Weight (g) | Shoot/Root | Root Volume (cm3 per plant) | |
|---|---|---|---|---|---|---|
| Salinity (mM) | ||||||
| 0 | 52.1 a | 10.99 a | 51.94 a | 16.16 a | 0.996 a | 51.00 a |
| 25 | 43.2 b | 8.44 b | 36.76 b | 12.23 b | 0.850 a | 41.88 b |
| 50 | 36.7 c | 7.11 c | 18.14 c | 5.58 c | 0.494 b | 37.50 b |
| Nano-silicon Dioxide (mg L−1) | ||||||
| S1 | 36.72 c | 6.95 c | 28.54 c | 9.22 c | 0.777 a | 35.88 b |
| S2 | 44.86 abc | 9.04 ab | 33.97 b | 10.7 bc | 0.757 a | 41.77 ab |
| S3 | 50.92 a | 10.35 a | 38.33 b | 11.50 ab | 0.752 a | 49.77 a |
| S4 | 41.41 bc | 8.01 bc | 35.92 b | 11.25 abc | 0.867 a | 41.11 ab |
| S5 | 46.34 abc | 9.13 ab | 35.74 c | 12.24 ab | 0.771 a | 43.77 ab |
| S6 | 47.53 ab | 9.92 a | 41.18 a | 13.04 a | 0.866 a | 48.44 a |
| Analysis of Variance | ||||||
| Salinity | ** | ** | ** | ** | ** | ** |
| nSiO2 | ** | * | * | ** | ns | ns |
| Salinity × nSiO2 | ns | ns | ns | ns | ns | ns |
| S1 | S6 | T Value | Pr > F | Pr > [t] | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | Std Dev | Std Err | Mean | Std Dev | Std Err | Pooled (Equal) | Satterthwaite (Unequal) | |||
| Fresh weight | 12.98 | 0.849 | 0.49 | 21.32 | 3.89 | 2.24 | −3.63 | 0.0909 | 0.0222 * | 0.059 ns |
| Dry weight | 4.28 | 1.017 | 0587 | 6.55 | 0.606 | 0.35 | −3.31 | 0.524 | 0.0297 * | 0.0402 * |
| Root fresh weight | 30.42 | 4.79 | 2.76 | 34.55 | 7.72 | 4.45 | −0.79 | 0.475 | 0.475 ns | 0.483 ns |
| Root dry weight | 4.92 | 0.70 | 0.407 | 8.29 | 2.314 | 1.33 | −2.41 | 0.17 | 0.0733 ns | 0.117 ns |
| Root volume | 28.33 | 7.63 | 4.40 | 40.00 | 5.00 | 2.88 | −2.21 | 0.60 | 0.091 ns | 0.102 ns |
| Shoot/root | 0.431 | 0.0449 | 0.0259 | 0.637 | 0.183 | 0.106 | −1.89 | 0.112 | 0.131 ns | 0.185 ns |
| Membrane stability index (MSI) | 64.22 | 10.31 | 5.95 | 80.00 | 2.68 | 1.55 | −2.57 | 0.127 | 0.062ns | 0.109 ns |
| Proline | 13.42 | 0.549 | 0.316 | 8.19 | 0.641 | 0.370 | 10.72 | 0.844 | 0.0004 ** | 0.0005 ** |
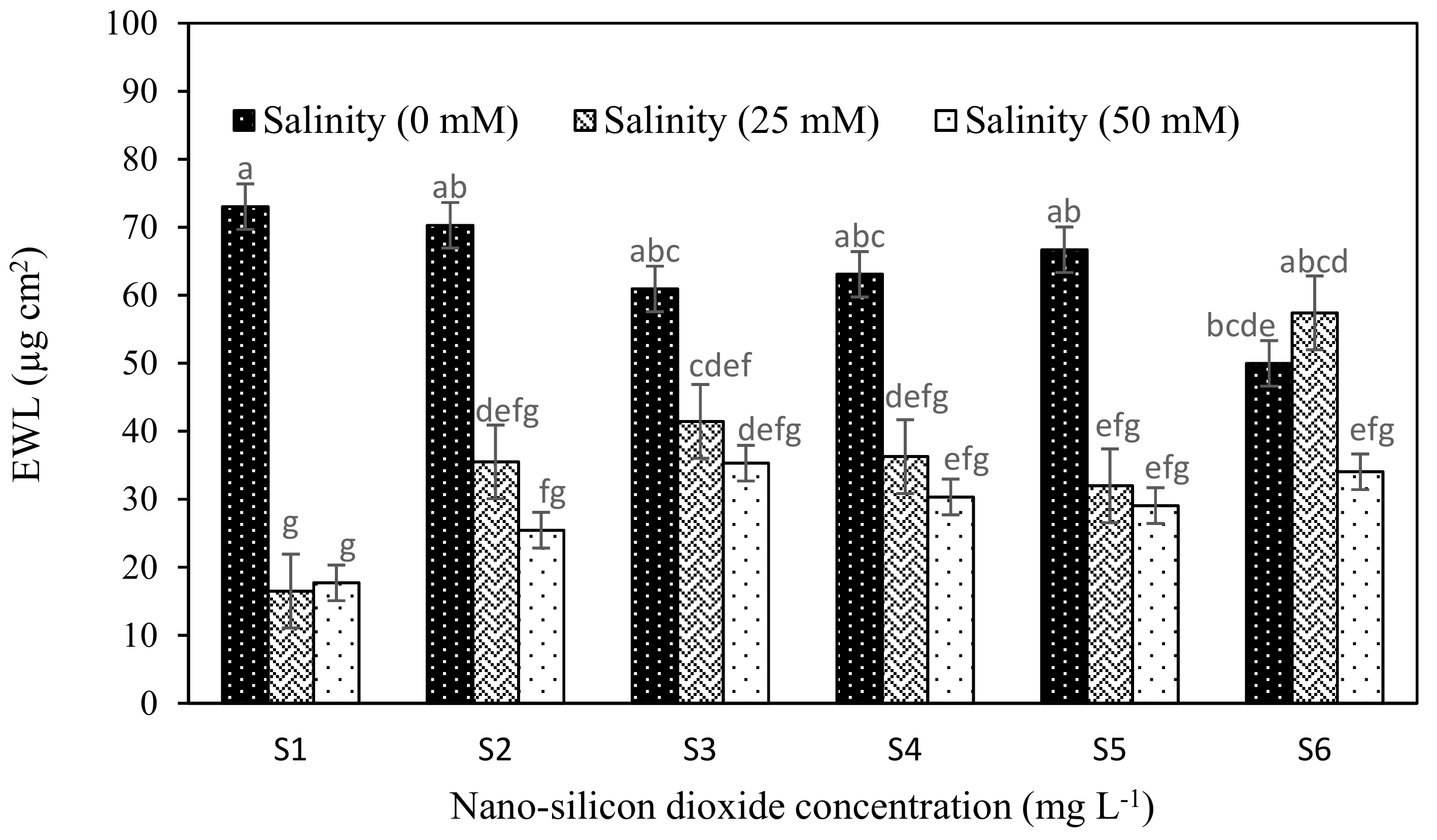
| Epicuticular wax layer (EWL) | 17.06 | 5.65 | 3.266 | 34.03 | 8.29 | 4.78 | −2.93 | 0.635 | 0.043 * | 0.050 * |
| Fruit Yield (g) | RWC (%) | RWP (%) | RWL (%) | MSI (%) | CT (g H2O/g Dry Weight) | Canopy Temperature (°C) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cloudy Day | Sunny Day | |||||||||
| Salinity (mM) | ||||||||||
| 0 | 198.06 a | 85.1 a | 0.91 a | 0.156 a | 83.9 a | 0.587 a | 3.91 a | 2.72 a | ||
| 25 | 149.40 b | 81.79 a | 0.87 ab | 0.154 a | 79.3 a | 0.832 a | 3.57 a | 2.04 a | ||
| 50 | 77.39 c | 67.37 b | 0.86 b | 0.168 a | 75.5 b | 0.908 a | 2.18 b | 0.10 b | ||
| Nano-silicon dioxide (mg L−1) | ||||||||||
| S1 | 124.05 c | 76.71 a | 0.861 a | 0.107 c | 74.2 bc | 1.111 a | 2.33 c | 0.713 c | ||
| S2 | 142.33 abc | 75.99 a | 0.901 a | 0.161 ab | 79.1 abc | 0.593 a | 3.15 b | 0.861 c | ||
| S3 | 151.92 ab | 75.26 a | 0.877 a | 0.161 ab | 79.6 abc | 0.788 a | 3.20 b | 1.14 c | ||
| S4 | 140.79 bc | 78.33 a | 0.883 a | 0.202 a | 84.64 a | 0.753 a | 3.15 b | 1.46 bc | ||
| S5 | 129.34 c | 80.91 a | 0.875 a | 0.158 b | 71.9 c | 0.843 a | 3.48 ab | 3.06 a | ||
| S6 | 161.26 a | 81.32 a | 0.911 a | 0.168 ab | 82.2 ab | 0.564 a | 4.02 a | 2.48 ab | ||
| Analysis of Variance | ||||||||||
| Salinity | ** | ** | ns | ns | ** | ns | ** | ** | ||
| Nano-silicon dioxide | ** | ns | ns | ** | * | ns | ** | ** | ||
| Salinity × Naon-silicon dioxide | ns | ns | ns | ns | ns | ns | ns | ns | ||
| EWL (µg cm2) | Proline (µmol g−1) | Chl a (mg g−1 Fresh Weight) | Chl b (mg g−1 Fresh Weight) | Total Chl (mg g−1 Fresh Weight) | Carotenoids (mg g−1 Fresh Weight) | |
|---|---|---|---|---|---|---|
| Salinity (mM) | ||||||
| 0 | 63.43 a | 5.83 c | 7.78 a | 2.75 a | 10.53 a | 2.86 b |
| 25 | 36.52 b | 6.68 b | 7.41 b | 2.88 a | 10.30 a | 3.24 a |
| 50 | 28.54 b | 10.53 a | 5.96 c | 2.38 b | 8.35 b | 2.63 c |
| Nano-Silicon Dioxide (mg L−1) | ||||||
| S1 | 35.53 b | 8.36 a | 6.48 c | 2.38 c | 8.86 c | 2.66 c |
| S2 | 43.74 ab | 7.06 bcd | 6.87 bc | 2.46 c | 9.34 bc | 2.69 c |
| S3 | 45.89 ab | 7.69 abc | 6.68 c | 2.39 c | 9.08 c | 2.82 bc |
| S4 | 43.22 ab | 7.91 ab | 7.57 a | 3.11 a | 10.68 a | 3.02 ab |
| S5 | 42.57 ab | 6.03 d | 7.53 a | 2.96 a | 10.50 a | 3.23 a |
| S6 | 47.12 a | 6.61 cd | 7.18 ab | 2.71 b | 9.89 b | 3.03 ab |
| Analysis of Variance | ||||||
| Salinity | ** | ** | ** | ** | ** | ** |
| Nano-silicon dioxide | ns | ** | ** | ** | ** | ** |
| Salininty × Nano-silicon dioxide | * | * | ** | ** | ** | ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Avestan, S.; Ghasemnezhad, M.; Esfahani, M.; Byrt, C.S. Application of Nano-Silicon Dioxide Improves Salt Stress Tolerance in Strawberry Plants. Agronomy 2019, 9, 246. https://doi.org/10.3390/agronomy9050246
Avestan S, Ghasemnezhad M, Esfahani M, Byrt CS. Application of Nano-Silicon Dioxide Improves Salt Stress Tolerance in Strawberry Plants. Agronomy. 2019; 9(5):246. https://doi.org/10.3390/agronomy9050246
Chicago/Turabian StyleAvestan, Saber, Mahmood Ghasemnezhad, Masoud Esfahani, and Caitlin S. Byrt. 2019. "Application of Nano-Silicon Dioxide Improves Salt Stress Tolerance in Strawberry Plants" Agronomy 9, no. 5: 246. https://doi.org/10.3390/agronomy9050246
APA StyleAvestan, S., Ghasemnezhad, M., Esfahani, M., & Byrt, C. S. (2019). Application of Nano-Silicon Dioxide Improves Salt Stress Tolerance in Strawberry Plants. Agronomy, 9(5), 246. https://doi.org/10.3390/agronomy9050246






