Molecular Characterization and Expression Analysis of MYB Transcription Factors Involved in the Glucosinolate Pathway in Chinese Cabbage (Brassica rapa ssp. pekinensis)
Abstract
1. Introduction
2. Materials and Methods
2.1. Identification of BrMYB Genes
2.2. Sequence Analysis of BrMYB Genes
2.3. Growth Condition and Preparation of Plant Materials
2.4. Quantitative Real-Time PCR (qRT-PCR) Expression Analysis in B. Rapa
2.5. Extraction of Desulfo-Glucosinolates (DS-GSLs) and UPLC Analysis
2.6. Statistical Analysis
3. Results
3.1. Identification of the MYB Genes in Chinese Cabbage
3.2. Multiple Sequence Alignment, Phylogenetic, Motif and Physiochemical Property Analyses of BrMYBs
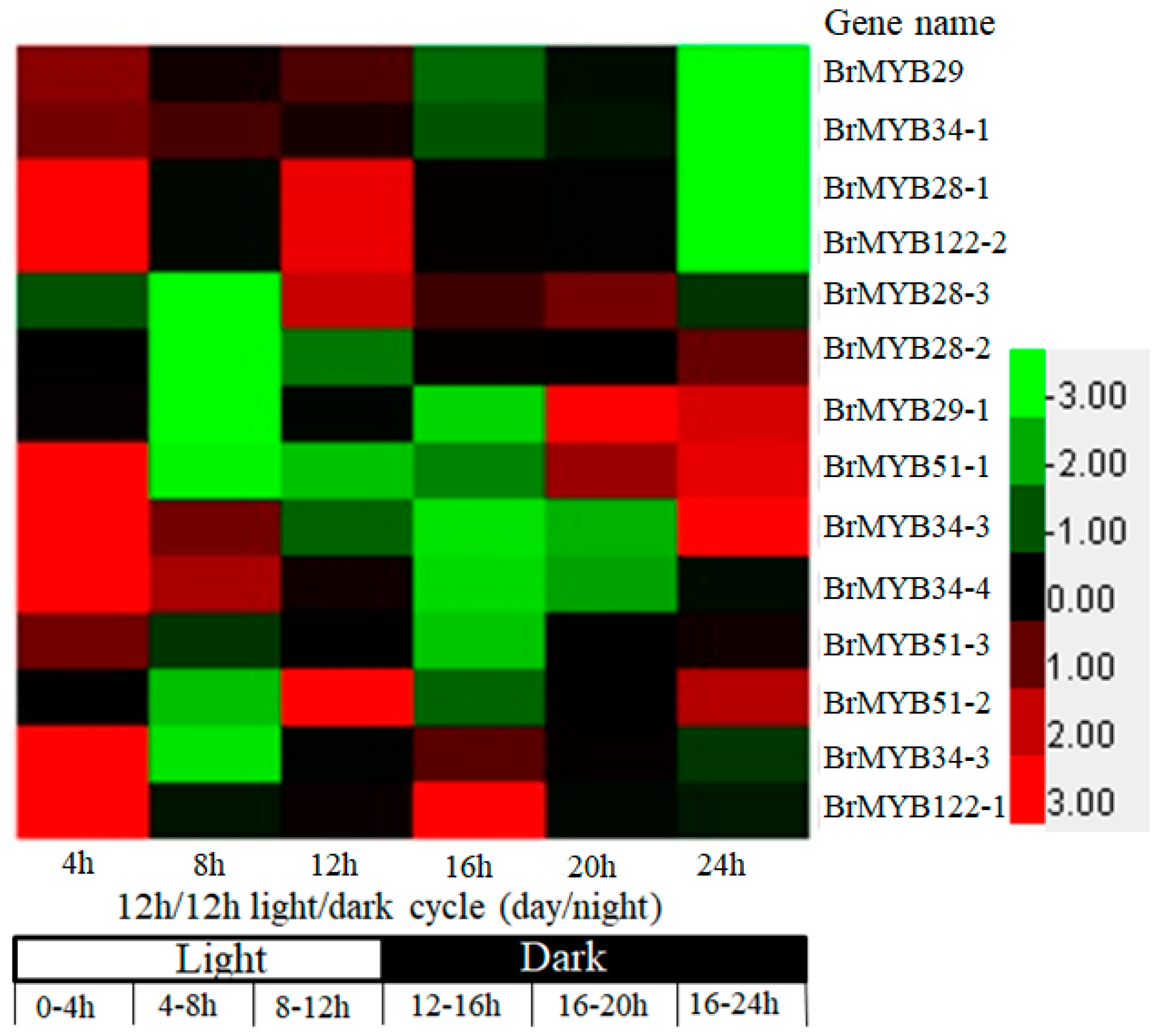
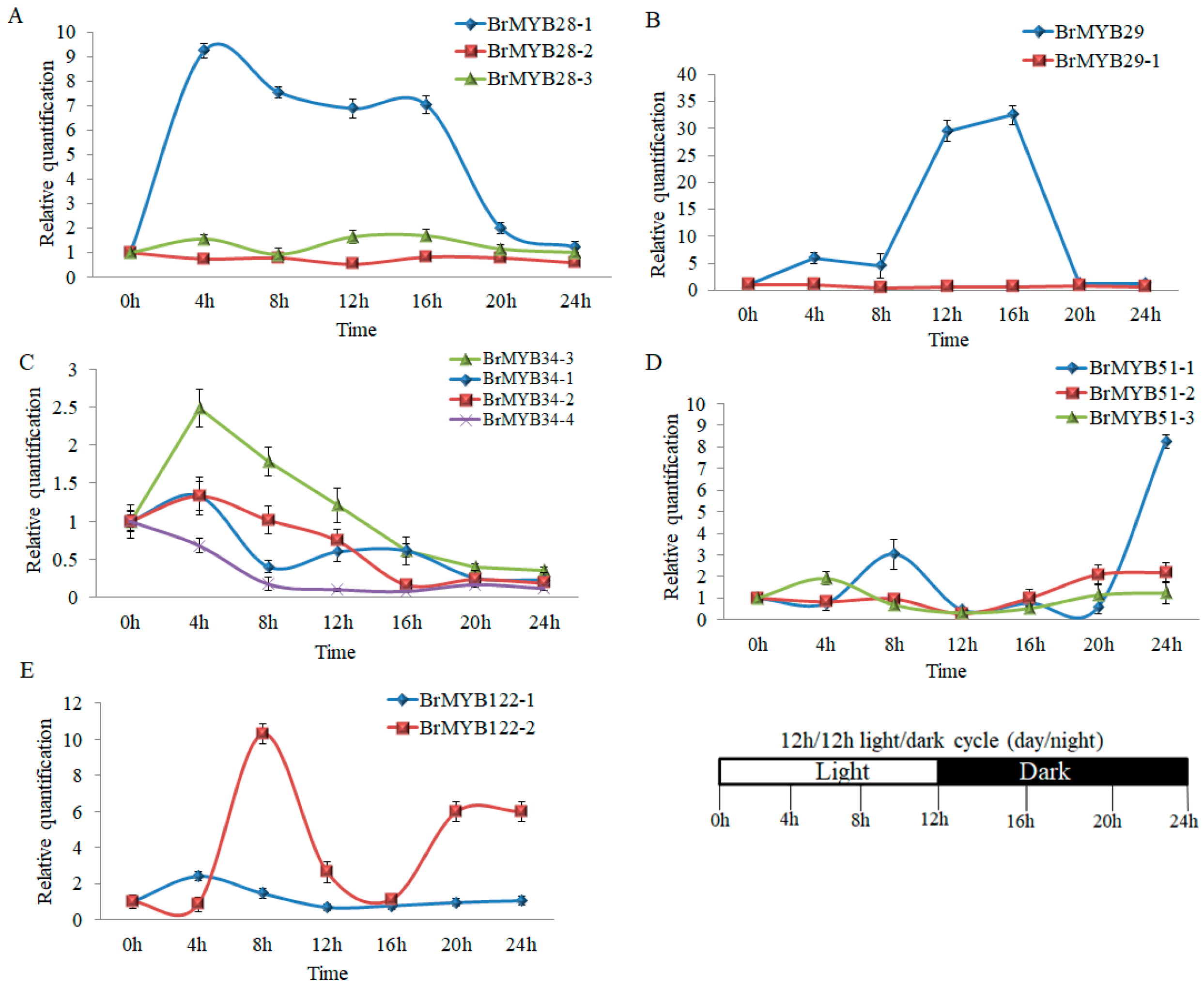
3.3. Expression Pattern of BrMYB Genes
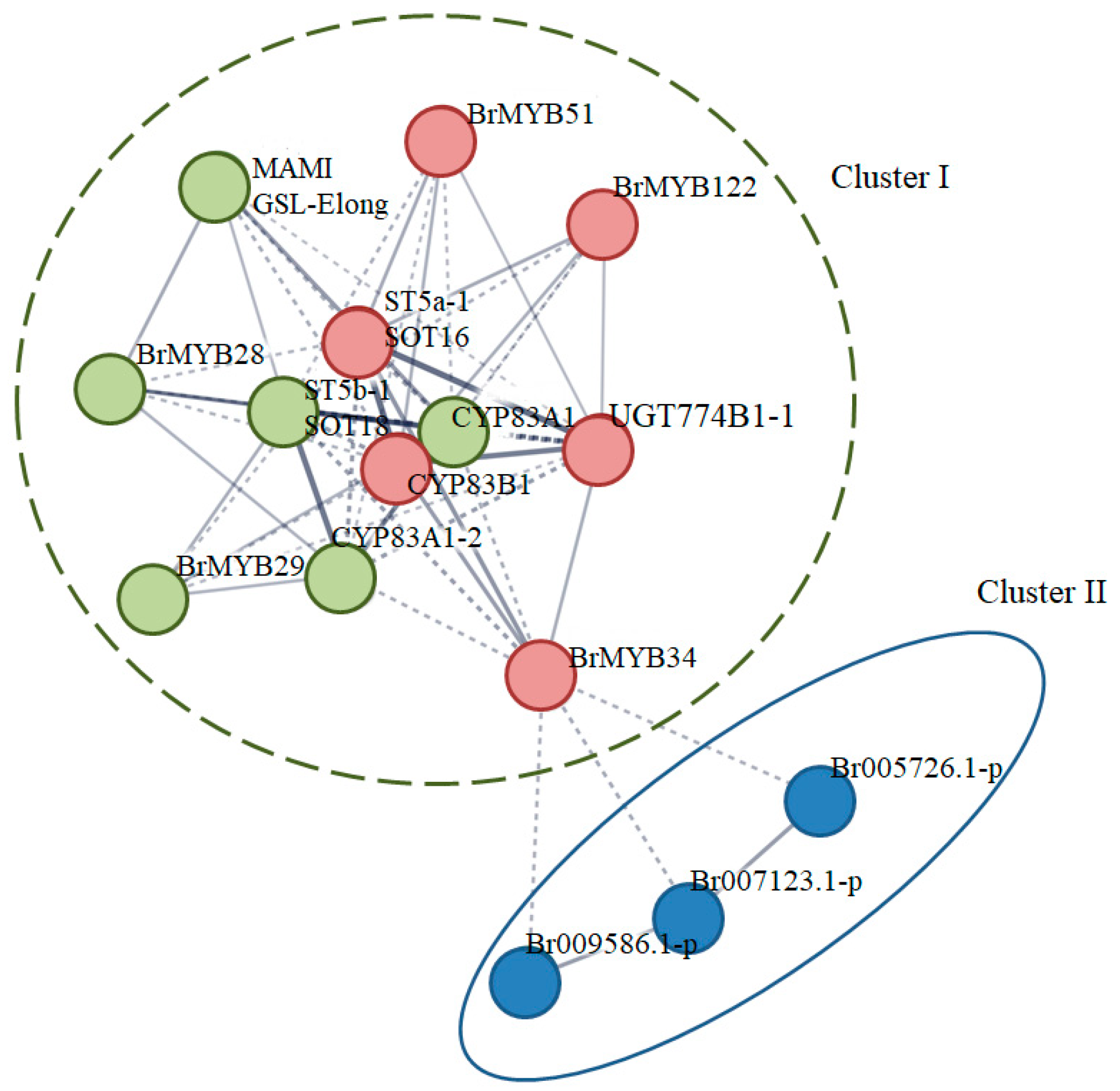
3.4. Functional Network Analysis of Candidate BrMYB in Glucosinolate Pathway
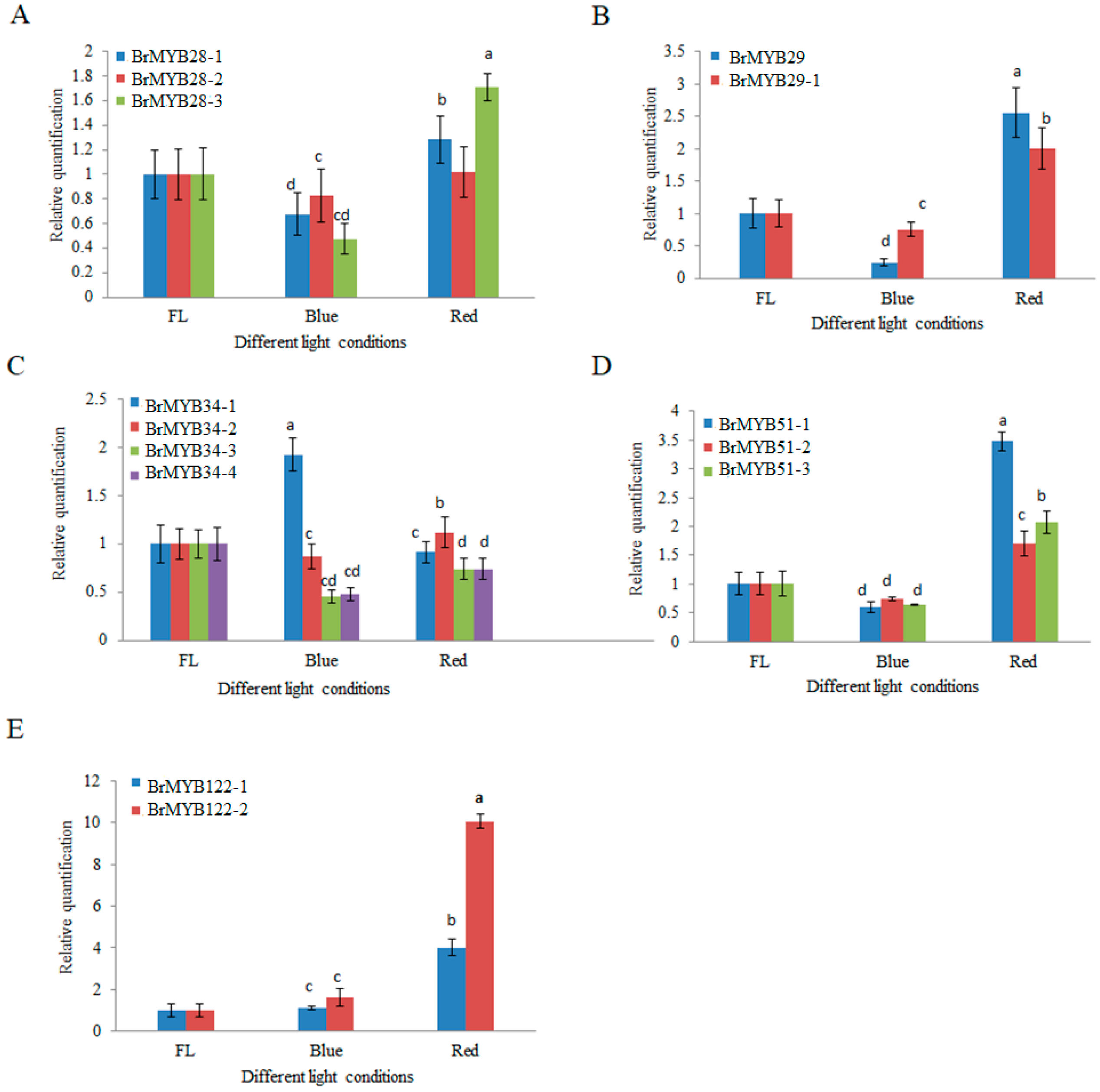
3.5. Expression Analysis of BrMYB Genes under Different Light Qualities
3.6. Production of Glucosinolates (GSLs) in Different Light Conditions
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Talalay, P.; Fahey, W. Phytochemicals from cruciferous plants protect against cancer by modulating carcinogen metabolism. J. Nutr. 2001, 131, 3027S–3033S. [Google Scholar] [CrossRef]
- Hayes, J.D.; Kelleher, M.O.; Eggleston, I.M. The cancer chemo-preventive actions of phytochemicals derived from GSLs. Eur. J. Nutr. 2008, 47, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.J.; Ishii, G. Glucosinolate profiles in the seeds, leaves and roots of rocket salad (Eruca sativa Mill.) and anti-oxidative activities of intact plant powder and purified 4-methoxyglucobrassicin. Soil Sci. Plant Nutr. 2006, 52, 394–400. [Google Scholar] [CrossRef]
- Padilla, G.; Cartea, M.E.; Velasco, P.; Haro, A.D.; Ordras, A. Variation of glucosinolates in vegetable crops of Brassica rapa. Phytochemistry. 2007, 68, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Sønderby, I.E.; Burow, M.; Rowe, H.C.; Kliebenstein, D.J.; Halkier, B.A. A complex interplay of three R2R3 MYB transcription factors determines the profile of aliphatic GSLs in Arabidopsis. Plant Physiol. 2010, 153, 348–363. [Google Scholar] [CrossRef]
- Del Carmen Martinez-Ballesta, M.; Moreno, D.A.; Carvajal, M. The physiological importance of glucosinolates on plant response to abiotic stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607–11625. [Google Scholar] [CrossRef]
- Klempnauer, K.H.; Gonda, T.J.; Bishop, J.M. Nucleotide sequence of the retroviral leukemia gene v-myb and its cellular progenitor c-myb: The architecture of a transduced oncogene. Cell 1982, 31, 453–463. [Google Scholar] [CrossRef]
- Lipsick, J.S. One billion years of Myb. Oncogene 1996, 13, 223–235. [Google Scholar]
- Tominaga, R.; Iwata, M.; Okada, K.; Wada, T. Functional analysis of the epidermal-specific MYB genes CAPRICE and WEREWOLF in Arabidopsis. Plant Cell 2007, 19, 2264–2277. [Google Scholar] [CrossRef]
- Abe, H.; Urao, T.; Ito, T.; Seki, M.; Shinozaki, K.; Yamaguchi-Shinozaki, K. Arabidopsis AtMYC2 (bHLH) and AtMYB2 (MYB) function as transcriptional activators in abscisic acid signaling. Plant Cell 2003, 15, 63–78. [Google Scholar] [CrossRef]
- Stracke, R.; Ishihara, H.; Huep, G.; Barsch, A.; Mehrtens, F.; Niehaus, K.; Weisshaar, B. Differential regulation of closely related R2R3-MYB transcription factors controls flavonol accumulation in different parts of the Arabidopsis thaliana seedling. Plant J. 2007, 50, 660–677. [Google Scholar] [CrossRef] [PubMed]
- Jung, C.; Seo, J.S.; Han, S.W.; Koo, Y.J.; Kim, C.H.; Song, S.I.; Nahm, B.H.; Choi, Y.D.; Cheong, J.J. Overexpression of AtMYB44 enhances stomatal closure to confer abiotic stress tolerance in transgenic Arabidopsis. Plant Physiol. 2008, 146, 623–635. [Google Scholar] [CrossRef] [PubMed]
- Mun, J.H.; Kwon, S.J.; Yang, T.J.; Seol, Y.J.; Jin, M.; Kim, J.A.; Lim, M.H.; Kim, J.S.; Baek, S.; Choi, B.S.; et al. Genome-wide comparative analysis of the Brassica rapa gene space reveals genome shrinkage and differential loss of duplicated genes after whole genome triplication. Genome Biol. 2009, 10, R111. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, X.; Tang, H.; Tan, X.; Ficklin, S.P.; Feltus, F.A.; Paterson, A.H. Modes of gene duplication contribute differently to genetic novelty and redundancy but show parallels across divergent angiosperms. PLoS ONE 2011, 6, e28150. [Google Scholar] [CrossRef] [PubMed]
- Udvardi, M.K.; Kakar, K.; Wandrey, M.; Montanri, O.; Murray, J.; Andriankaja, A.; Zhang, J.Y.; Benedito, V.; Hofer, J.M.I.; Chueng, F.; et al. Legume transcription factors: Global regulators of plant development and response to the environment. Plant Physiol. 2007, 144, 538–549. [Google Scholar] [CrossRef]
- Stracke, R. The R2R3-MYB gene family in Arabidopsis thaliana. Curr. Opin. Plant Biol. 2001, 4, 447–456. [Google Scholar] [CrossRef]
- Wang, Z.; Tang, J.; Hu, R.; Wu, P.; Hou, X.L.; Song, X.M.; Xiong, A.S. Genome-wide analysis of the R2R3-MYB transcription factor genes in Chinese cabbage (Brassica rapa ssp. pekinensis) reveals their stress and hormone responsive patterns. BMC Genomics 2015, 16, 17. [Google Scholar] [CrossRef]
- Cheng, F.; Liu, S.; Wu, J.; Fang, L.; Sun, S.; Liu, B. BRAD, the genetics and genomics database for Brassica plants. BMC Plant Biol. 2011, 11, 136. [Google Scholar] [CrossRef]
- Punta, M.; Coggill, P.C.; Eberhardt, R.Y.; Mistry, J.; Tate, J.; Boursnell, C. The Pfam protein family’s database. Nucl. Acids Res. 2012, 40, D290–D301. [Google Scholar] [CrossRef]
- Thompson, J.D.; Gibson, T.J.; Plewniak, F.; Jeanmougin, F.; Higgins, D.G. The CLUSTAL_X Windows interface: Flexible strategies for multiple sequence alignment aided by quality analysis tools. Nucl. Acids Res. 1997, 25, 4876–4882. [Google Scholar] [CrossRef]
- Kumar, S.; Stecher, G.; Tamura, K. MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016, 33, 1870–1874. [Google Scholar] [CrossRef] [PubMed]
- Bailey, T.L.; Williams, N.; Misleh, C.; Li, W.W. MEME: Discovering and analyzing DNA and protein sequence motifs. Nucl. Acids Res. 2006, 34, 369–373. [Google Scholar] [CrossRef] [PubMed]
- Moon, J.; Jeong, M.J.; Lee, S.I.; Lee, J.G.; Hwang, H.; Yu, J.; Kim, Y.R.; Park, S.W.; Kim, J.A. Effect of LED mixed light conditions on the glucosinolate pathway in Brassica rapa. J. Plant Biotechnol. 2015, 42, 245–256. [Google Scholar] [CrossRef]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2 (-Delta Delta C(T)) Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Bhandari, S.R.; Jo, J.S.; Lee, J.G. Comparison of Glucosinolate Profiles in Different Tissues of Nine Brassica Crops. Molecules 2015, 20, 15827–15841. [Google Scholar] [CrossRef]
- Kim, J.A.; Shim, D.; Kumari, S.; Jung, H.; Jung, K.H.; Jeong, H.; Kim, W.Y.; Lee, S.I.; Jeong, M.J. Transcriptome analysis of diurnal gene expression in Chinese cabbage. Genes 2019, 10, 130. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Franceschini, A.; Wyder, S.; Forslund, K.; Heller, D.; Huerta-Cepas, J.; Simonovic, M.; Roth, A.; Santos, A.; Tsafou, K.P.; et al. STRING v10: Protein–protein interaction networks, integrated over the tree of life. Nucl. Acids Res. 2015, 43, D447–D452. [Google Scholar] [CrossRef]
- Augustine, R.; Majee, M.; Gershenzon, J.; Bisht, N.C. Four genes encoding MYB28, a major transcriptional regulator of the aliphatic glucosinolate pathway, are differentially expressed in the allopolyploid Brassica juncea. J. Exp. Bot. 2013, 64, 4907–4921. [Google Scholar] [CrossRef]
- Ogata, K.; Kanei-Ishii, C.; Sasaki, M.; Hatanaka, H.; Nagadoi, A.; Enari, M.; Sarai, A. The cavity in the hydrophobic core of Myb DNA-binding domain is reserved for DNA recognition and trans-activation. Nat. Struct Biol. 1996, 3, 178–187. [Google Scholar] [CrossRef]
- Kranz, H.; Stracke, R.; Weisshaar, B. c-MYB oncogene-like genes encoding three MYB repeats occur in all major plant lineages. Plant J. 2000, 21, 231–235. [Google Scholar] [CrossRef]
- Urzainqui, A.; Bevan, M.; Martin, C.; Smeekens, S.; Tonelli, C.; Paz-Ares, J.; Weisshaar, B. Towards functional characterization of the members of the R2R3-MYB gene family from Arabidopsis thaliana. Plant J. 1998, 16, 263–276. [Google Scholar]
- Jin, H.; Martin, C. Multi functionality and diversity within the plant MYB-gene family. Plant Mol. Biol. 1999, 41, 577–585. [Google Scholar] [CrossRef] [PubMed]
- Dubos, C.; Stracke, R.; Grotewold, E.; Weisshaar, B.; Martin, C.; Lepiniec, L. MYB transcription factors in Arabidopsis. Trends Plant Sci. 2010, 15, 573–581. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.; Wang, X.; Yu, J.; Wu, J.; Li, W.; Huang, J.; Dong, C.; Hua, W.; Liu, S. Comprehensive analysis of RNA-seq data reveals the complexity of the transcriptome in Brassica rapa. BMC Genom. 2013, 14, 689. [Google Scholar] [CrossRef]
- Kalsotra, A.; Cooper, T.A. Functional consequences of developmentally regulated alternative splicing. Nat. Rev. Genet. 2011, 12, 715–729. [Google Scholar] [CrossRef]
- Zhou, R.; Moshgabadi, N.; Adams, K.L. Extensive changes to alternative splicing patterns following allopolyploidy in natural and resynthesized polyploids. Proc. Natl. Acad. Sci USA 2011, 108, 16122–16127. [Google Scholar] [CrossRef]
- Adams, K.L. Evolution of duplicate gene expression in polyploidy and hybrid plants. J. Hered. 2007, 98, 136–141. [Google Scholar] [CrossRef]
- Seo, M.-S.; Jin, M.; Chun, J.-H.; Kim, S.-J.; Park, B.-S.; Shon, S.-H.; Kim, J.S. Functional analysis of three BrMYB28 transcription factors controlling the biosynthesis of glucosinolates in Brassica rapa. Plant Mol. Biol. 2016, 90, 503–516. [Google Scholar] [CrossRef]
- Katiyar, A.; Smita, S.; Lenka, S.K.; Rajwanshi, R.; Chinnusamy, V.; Bansal, K.C. Genome-wide classification and expression analysis of MYB transcription factor families in rice and Arabidopsis. BMC Genom. 2012, 13, 544. [Google Scholar] [CrossRef]
- Gigolashvili, T.; Engqvist, M.; Yatusevich, R.; Muller, C.; Flugge, U.I. The transcription factor HIG1/MYB51 regulates indolic glucosinolate biosynthesis in Arabidopsis thaliana. Plant J. 2007, 50, 886–901. [Google Scholar] [CrossRef]
- Smith, H. Light quality, photoperception, and plant strategy. Annu. Rev. Plant Physiol. 1982, 33, 481–518. [Google Scholar] [CrossRef]
- Sang, J.P.; Minchinton, I.R.; Johnstone, P.K.; Truscott, R.J.W. Glucosinolate profiles in the seed, root and leaf tissue of cabbage, mustard, rapeseed radish and swede. Can. J. Plant Sci. 1984, 64, 77–93. [Google Scholar] [CrossRef]
- Rosa, E.A.S.; Rodrigues, P.M.F. The effect of light and temperature on glucosinolate concentration in the leaves and roots of cabbage seedlings. J. Sci. Food Agric. 1998, 78, 208–212. [Google Scholar] [CrossRef]
- Sofia, D.C.; Kevin, M.F. Sequential light programs shape kale (Brassica napus) sprout appearance and alter metabolic and nutrient content. Hortres 2014, 8. [Google Scholar] [CrossRef]
- Abe, K.; Kido, S.; Maeda, T.; Kami, D.; Matsuura, H.; Shimura, H.; Suzuki, T. Glucosinolate profiles in Cardamine fauriei and effect of light quality on glucosinolate concentration. Sci. Hort. 2015, 189, 12–16. [Google Scholar] [CrossRef]
- Huseby, S.; Koprivova, A.; Lee, B.-R.; Saha, S.; Mithen, R.; Wold, A.-B.; Bengtsson, G.B.; Kopriva, S. Diurnal and light regulation of sulphur assimilation and glucosinolate biosynthesis in Arabidopsis. J. Exp. Bot. 2013, 64, 1039–1048. [Google Scholar] [CrossRef]
- Kim, Y.B.; Li, X.; Kim, S.J.; Kim, H.H.; Lee, J.; Kim, H.; Park, S.U. MYB transcription factors regulate glucosinolate biosynthesis in different organs of Chinese cabbage (Brassica rapa ssp. pekinensis). Molecules 2013, 18, 8682–8695. [Google Scholar] [CrossRef]
- Lee, M.K.; Arasu, M.V.; Park, S.; Byeon, D.H.; Chung, S.O.; Park, S.U.; Lim, Y.P.; Kim, S.J. LED lights enhance metabolites and antioxidants in Chinese cabbage and kale. Braz. Arch. Bio. Technol. 2016, 59, e16150546. [Google Scholar] [CrossRef]
- Kuo, T.C.Y.; Chen, C.H.; Chen, S.H.; Lu, I.H.; Chu, M.J.; Huang, L.C.; Lin, C.Y.; Chen, C.Y.; Lo, H.F.; Jeng, S.T.; et al. The effect of red light and far-red light conditions on secondary metabolism in Agarwood. BMC Plant Biol. 2015, 15, 139. [Google Scholar] [CrossRef]
| Gene ID | Gene Name | Chr | Length (aa) | Length (bp) | pI/Mw | No. of MYB Domain | A. thaliana Gene ID & Name | % Identity with A. thaliana |
|---|---|---|---|---|---|---|---|---|
| Bra012961 | BrMYB28-1 | A03 | 354 | 1065 | 5.69/39870.09 | 2 | AT5G61420 (AtMYB28) | 84.1–85.4 |
| Bra035929 | BrMYB28-2 | A09 | 357 | 1074 | 5.59/40509.60 | 2 | ||
| Bra029311 | BrMYB28-3 | A02 | 372 | 1119 | 6.08/42063.90 | 2 | ||
| Bra005949 | BrMYB29 | A03 | 330 | 993 | 4.78/37140.91 | 2 | AT5G07690 (AtMYB29) | 83.2–87.0 |
| Bra009245 | BrMYB29-1 | A10 | 88 | 267 | 9.30/10148.72 | 1 | ||
| Bra013000 | BrMYB34-1 | A03 | 316 | 951 | 4.99/34916.87 | 2 | AT5G60890 (AtMYB34) | 82.1–94.3 |
| Bra035954 | BrMYB34-2 | A09 | 302 | 909 | 5.45/33544.80 | 2 | ||
| Bra029350 | BrMYB34-3 | A02 | 309 | 930 | 5.38/34468.68 | 2 | ||
| Bra029349 | BrMYB34-4 | A02 | 280 | 843 | 5.12/31179.99 | 2 | ||
| Bra025666 | BrMYB51-1 | A06 | 341 | 1026 | 5.96/38407.18 | 2 | AT1G18570 (AtMYB51) | 81.0–89.6 |
| Bra031035 | BrMYB51-2 | A09 | 320 | 963 | 5.60/36371.6 | 2 | ||
| Bra016553 | BrMYB51-3 | A08 | 333 | 1002 | 5.12/37303.49 | 2 | ||
| Bra015939 | BrMYB122-1 | A07 | 326 | 981 | 5.77/6555.59 | 2 | AT1G74080 (AtMYB122) | 82.4 |
| Bra008131 | BrMYB122-2 | A02 | 334 | 1005 | 5.76/7635.81 | 2 |
| Glucosinolate | FL | B | R |
|---|---|---|---|
| Aliphatic glucosinolates | |||
| Progoitrin | 4.40 ± 0.80 zb | 2.65 ± 0.10 cy | 5.22 ± 0.62 a |
| Epiprogoitrin | 0.14 ± 0.01 a | 0.08 ± 0.0 c | 0.14 ± 0.02 a |
| Sinigrin | 0.00 ± 0.00 c | 0.12 ± 0.01 b | 0.13 ± 0.04 a |
| Glucoraphanin | 0.02 ± 0.00 a | 0.00 ± 0.00 c | 0.01 ± 0.00 b |
| Gluconapin | 13.22 ± 0.38 b | 10.28 ± 0.86 c | 14.26 ± 0.28 a |
| Glucobrassicanapin | 8.11 ± 0.32 b | 5.37 ± 0.51 c | 8.22 ± 0.97 a |
| Indolic glucosinolates | |||
| Glucobrassicin | 1.34 ± 0.12 c | 1.62 ± 0.40 b | 3.24 ± 0.65 a |
| Total GSL | 27.22 ± 0.91 b | 20.87 ± 1.05 c | 31.21 ± 1.85 a |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumari, S.; Jo, J.S.; Choi, H.S.; Lee, J.G.; Lee, S.I.; Jeong, M.-J.; Kim, J.A. Molecular Characterization and Expression Analysis of MYB Transcription Factors Involved in the Glucosinolate Pathway in Chinese Cabbage (Brassica rapa ssp. pekinensis). Agronomy 2019, 9, 807. https://doi.org/10.3390/agronomy9120807
Kumari S, Jo JS, Choi HS, Lee JG, Lee SI, Jeong M-J, Kim JA. Molecular Characterization and Expression Analysis of MYB Transcription Factors Involved in the Glucosinolate Pathway in Chinese Cabbage (Brassica rapa ssp. pekinensis). Agronomy. 2019; 9(12):807. https://doi.org/10.3390/agronomy9120807
Chicago/Turabian StyleKumari, Shipra, Jung Su Jo, Hyo Seon Choi, Jun Gu Lee, Soo In Lee, Mi-Jeong Jeong, and Jin A Kim. 2019. "Molecular Characterization and Expression Analysis of MYB Transcription Factors Involved in the Glucosinolate Pathway in Chinese Cabbage (Brassica rapa ssp. pekinensis)" Agronomy 9, no. 12: 807. https://doi.org/10.3390/agronomy9120807
APA StyleKumari, S., Jo, J. S., Choi, H. S., Lee, J. G., Lee, S. I., Jeong, M.-J., & Kim, J. A. (2019). Molecular Characterization and Expression Analysis of MYB Transcription Factors Involved in the Glucosinolate Pathway in Chinese Cabbage (Brassica rapa ssp. pekinensis). Agronomy, 9(12), 807. https://doi.org/10.3390/agronomy9120807






