Fatty Acid Composition of Sesame (Sesamum indicum L.) Seeds in Relation to Yield and Soil Chemical Properties on Continuously Monocropped Upland Fields Converted from Paddy Fields
Abstract
1. Introduction
2. Materials and Methods
2.1. Location and Site Description
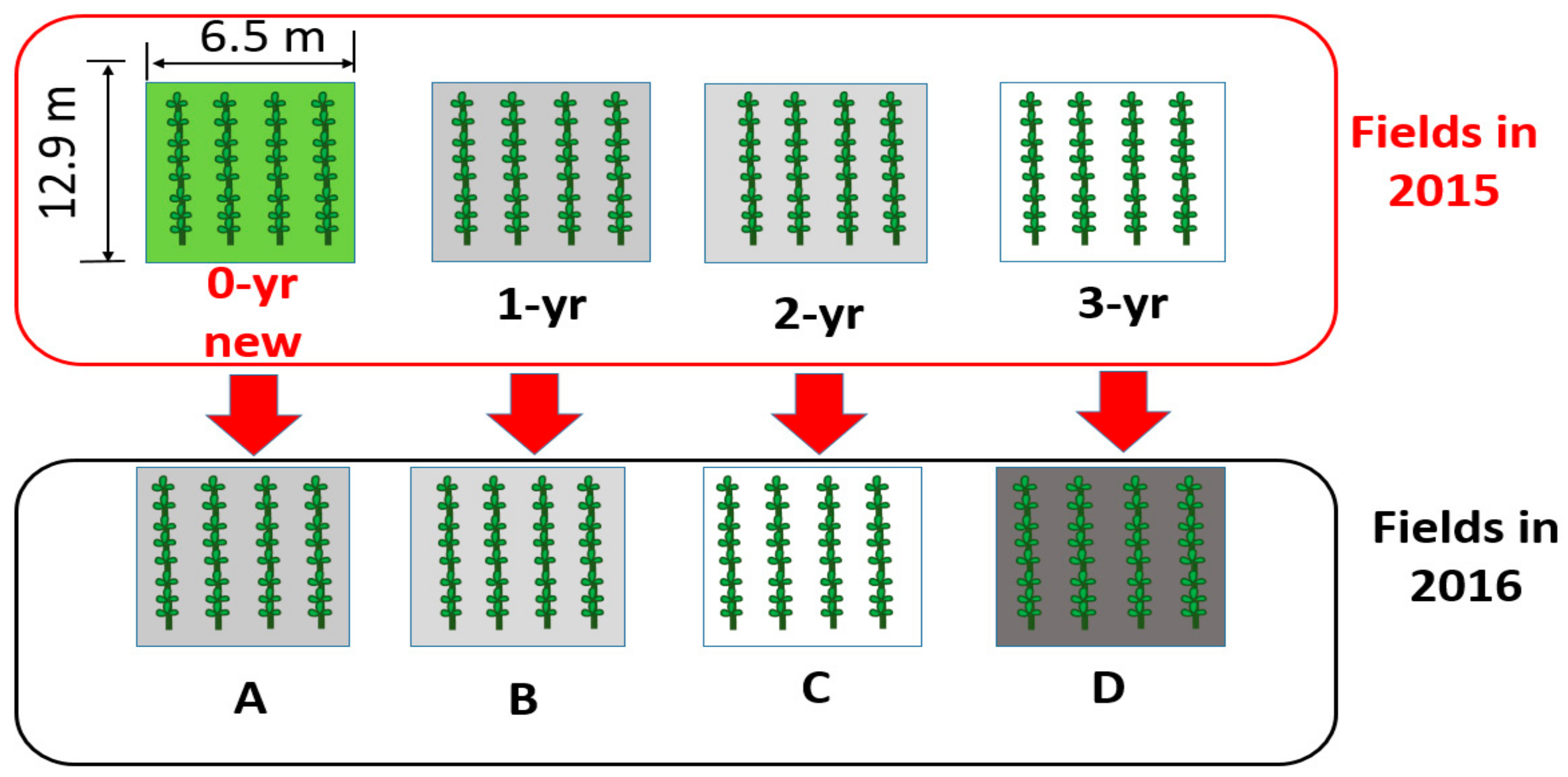
2.2. Field Experiment and Experimental Design
2.3. Determination of Seed Yield and 1000-Seed Weight
2.4. Determination of Fatty Acids Compositions in Sesame Seeds
2.5. Soil Sampling and Analysis
2.6. Data Analysis
3. Results
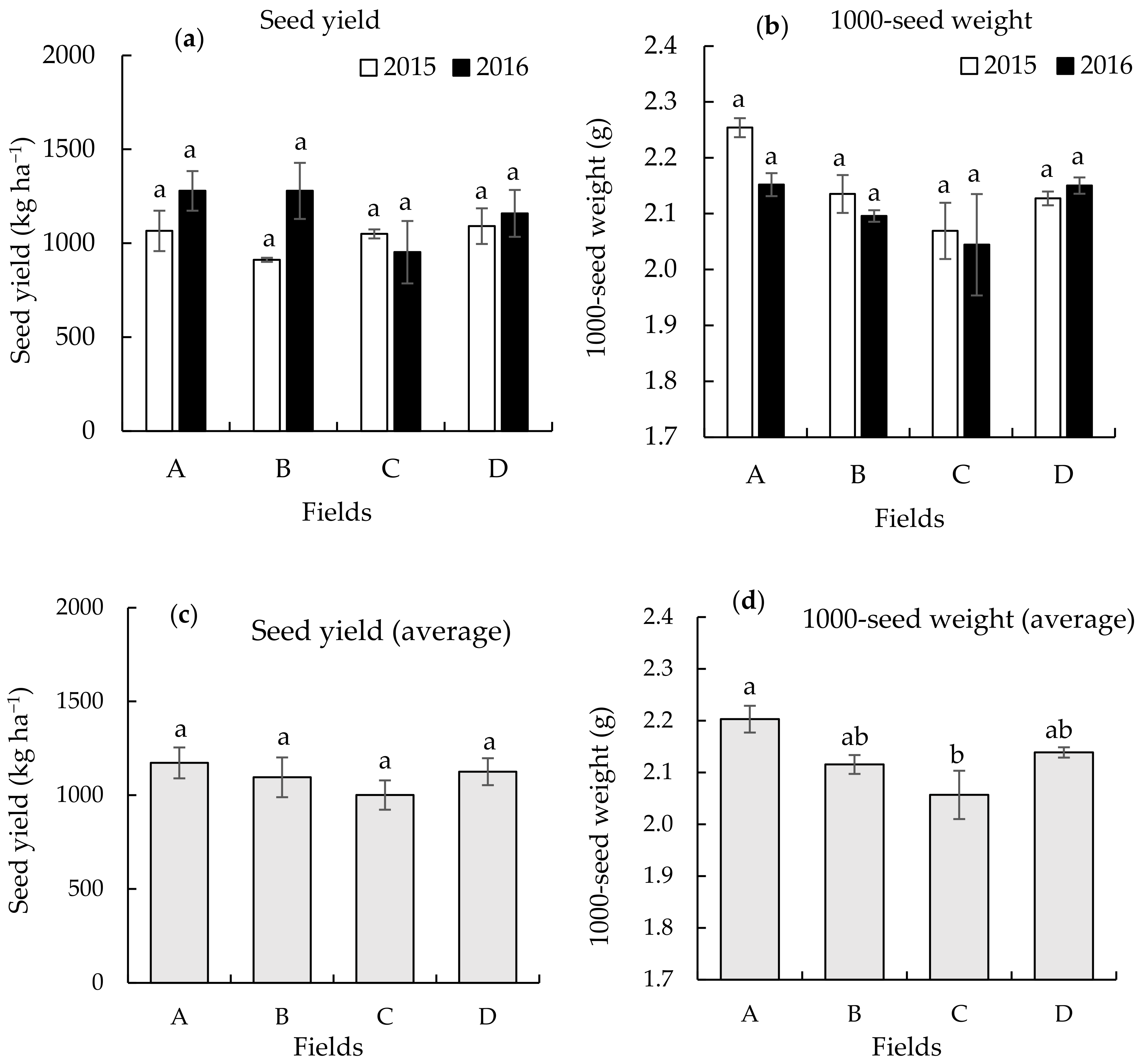
3.1. Seed Yield and 1000-Seed Weight on the Different Continuously Monocropped Fields in 2015 and 2016
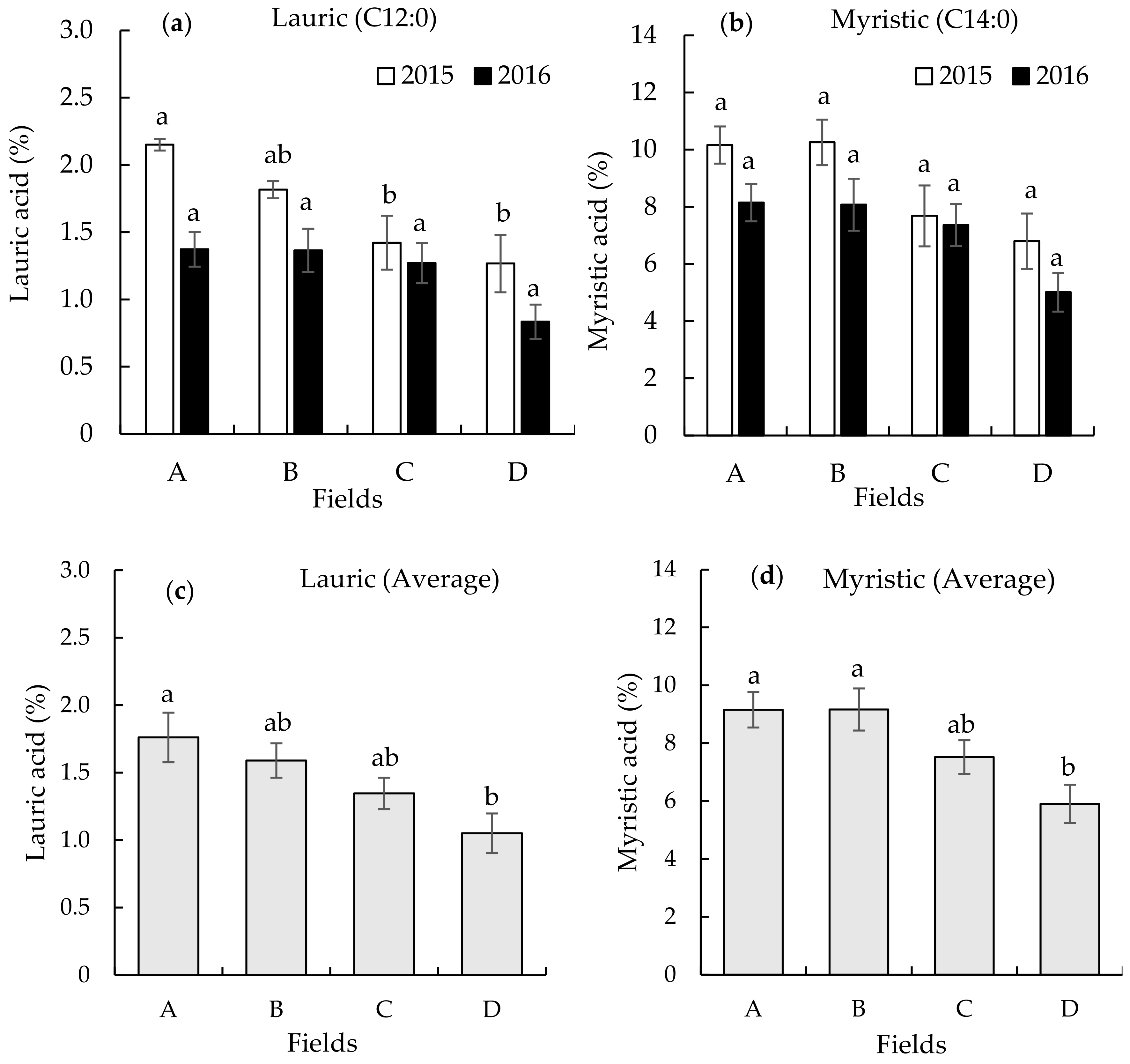
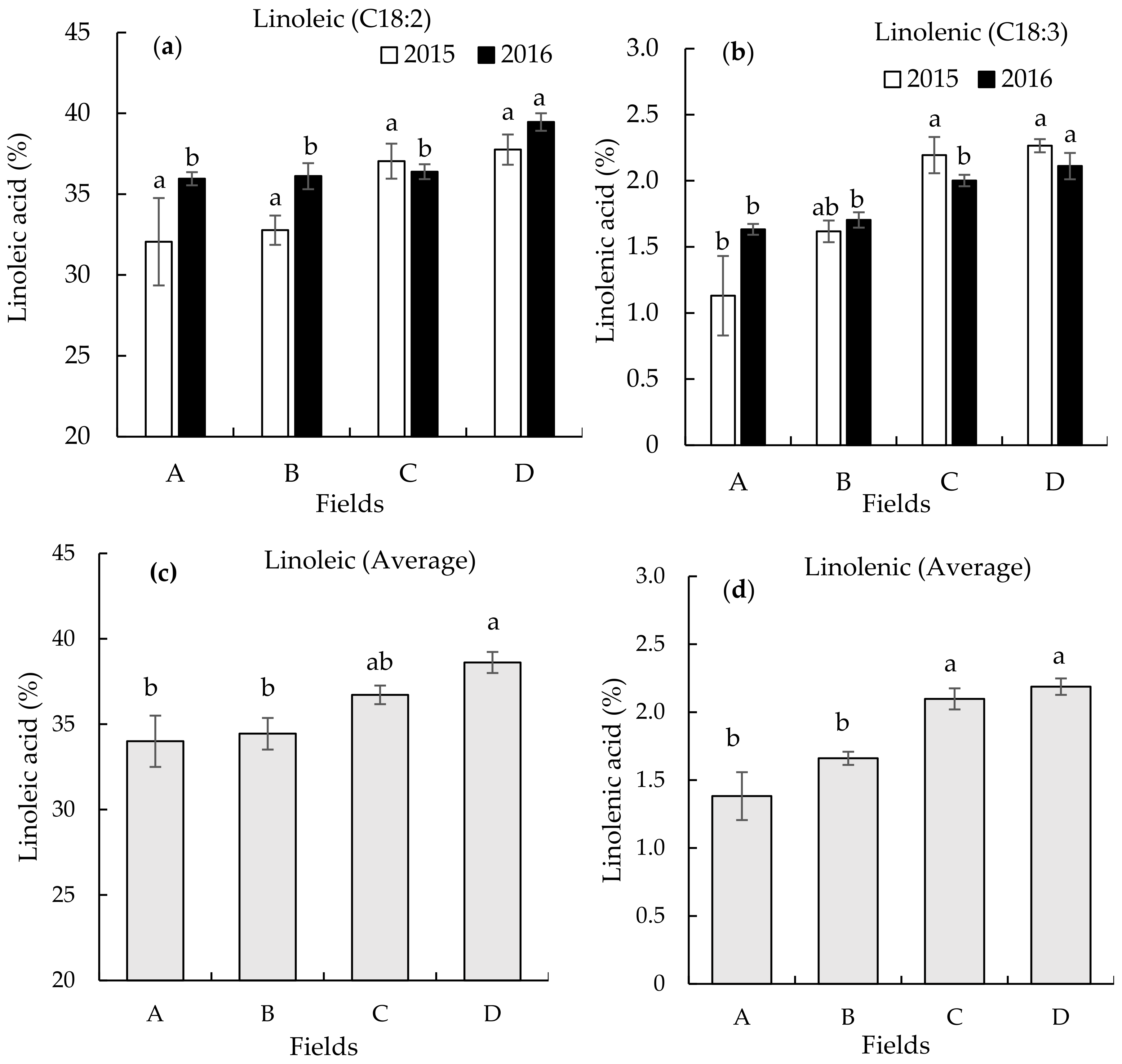
3.2. Seed Fatty Acid Composition on the Different Continuously Monocropped Fields in 2015 and 2016
3.3. Soil Chemical Properties on the Different Continuously Monocropped Fields in 2015 and 2016
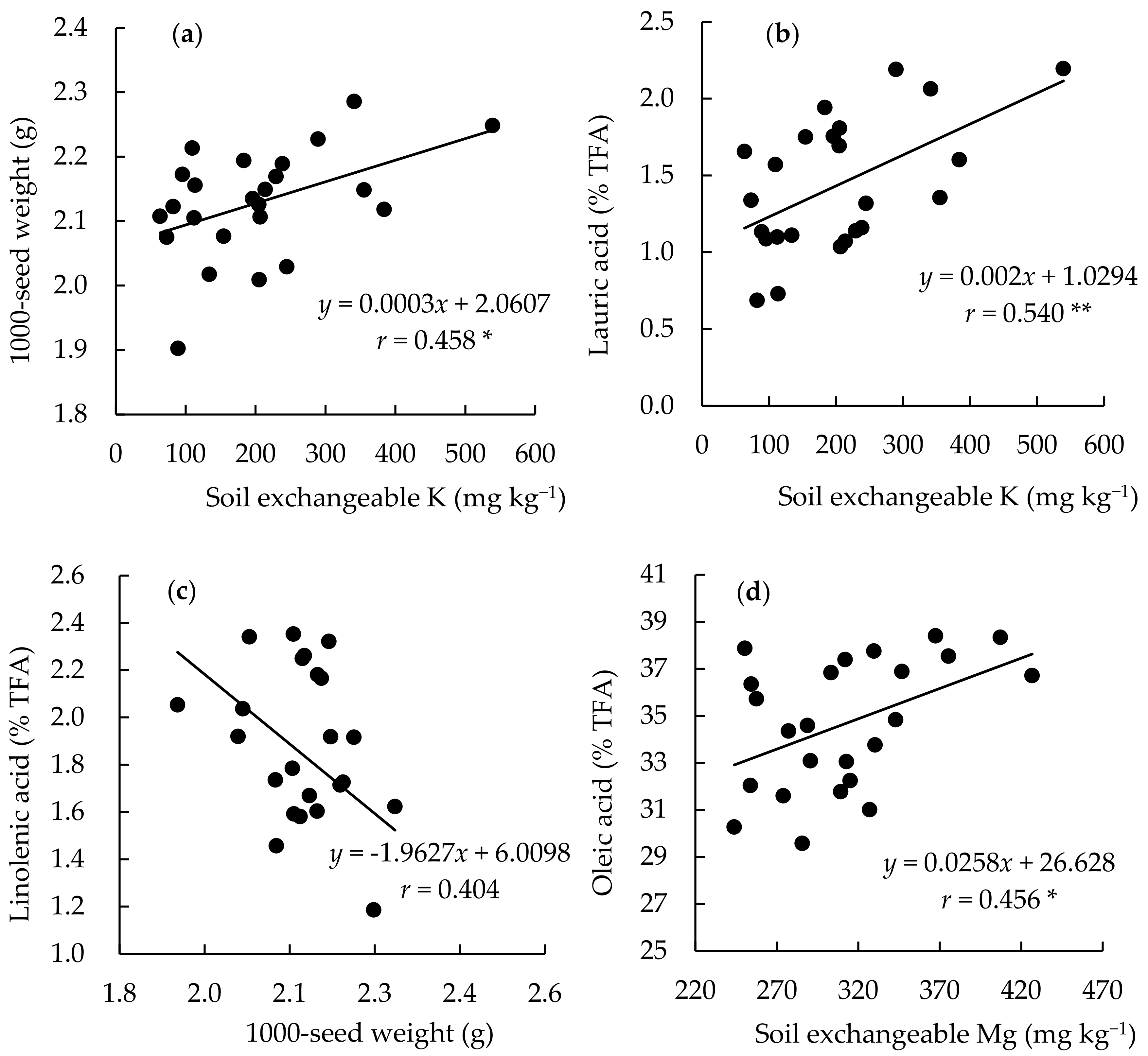
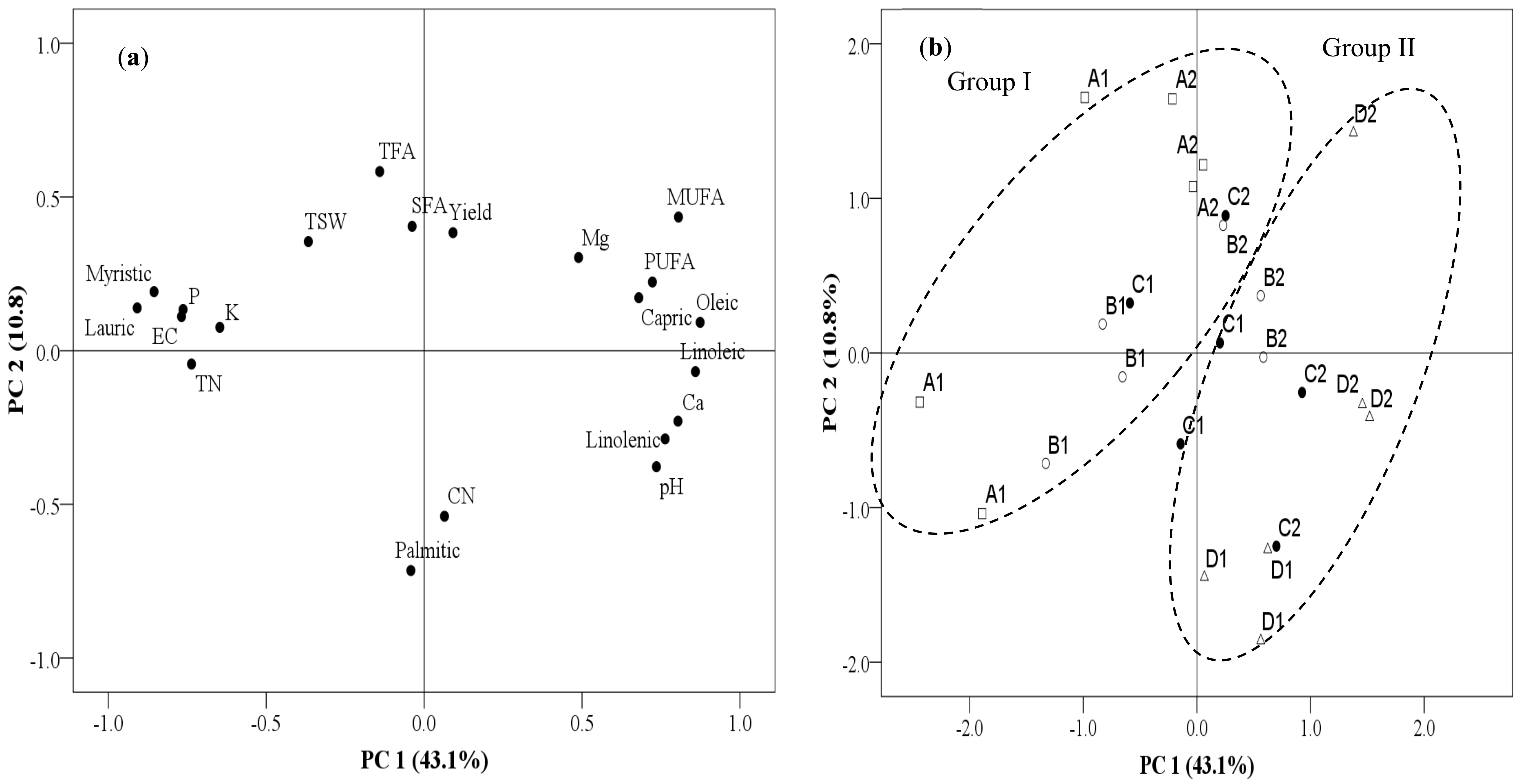
3.4. Relationship between Yield, Fatty Acid, and Soil Chemical Parameters among the Continuously Monocropped Fields
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ashri, A. Sesame. In Oil Crops of the World; Robbelen, G., Downey, R.K., Ashri, A., Eds.; McGraw–Hill: New York, NY, USA, 1989; pp. 375–387. [Google Scholar]
- Hwang, L.S. Sesame oil. In Bailey’s Industrial Oil and Fat Products, 6th ed.; Shahidi, F., Ed.; John Wiley and Sons Inc.: New York, NY, USA, 2005; pp. 538–577. ISBN 047167849X. [Google Scholar]
- Kaur, N.; Chugh, V.; Gupta, A.K. Essential fatty acids as functional components of foods—A review. J. Food Sci. Technol. 2014, 51, 2289–2303. [Google Scholar] [CrossRef] [PubMed]
- Goksu Erol, A.Y.; Bulbul, A.; Avci, G.; Ozdemir, M.; Akkaya, O. The Protective Effects of Omega-3 Fatty Acids and Sesame Oil on Cyclosporine—A Induced Liver Apoptosis. J. Acad. Res. Med. 2011, 1, 8–11. [Google Scholar] [CrossRef]
- Wacal, C.; Ogata, N.; Basalirwa, D.; Handa, T.; Sasagawa, D.; Acidri, R.; Ishigaki, T.; Kato, M.; Masunaga, T.; Yamamoto, S.; et al. Growth, seed yield, mineral nutrients and soil properties of sesame (Sesamum indicum L.) as influenced by biochar addition on upland field converted from paddy. Agronomy 2019, 9, 55. [Google Scholar] [CrossRef]
- Yasumoto, S.; Katsuta, M. Breeding a high-lignan-content sesame cultivar in the prospect of promoting metabolic functionality. Jpn. Agric. Res. Q. 2006, 40, 123–129. [Google Scholar] [CrossRef]
- Wacal, C.; Ogata, N.; Basalirwa, D.; Sasagawa, D.; Ishigaki, T.; Handa, T.; Kato, M.; Tenywa, M.M.; Masunaga, T.; Yamamoto, S.; et al. Imbalanced Soil Chemical Properties and Mineral Nutrition in Relation to Growth and Yield Decline of Sesame on Different Continuously Cropped Upland Fields Converted Paddy. Agronomy 2019, 9, 184. [Google Scholar] [CrossRef]
- Jørgensen, H.; Bach Knudsen, K.E.; Lauridsen, C. Influence of different cultivation methods on carbohydrate and lipid compositions and digestibility of energy of fruits and vegetables. J. Sci. Food Agric. 2012, 92, 2876–2882. [Google Scholar] [CrossRef]
- Patil, B.N.; Lakkineni, K.C.; Bhargava, S.C. Seed yield and yield contributing characters as influenced by N supply in rapeseed-mustared. J. Agron. Crop Sci. 1996, 177, 197–205. [Google Scholar] [CrossRef]
- Ali, S.; Jan, A.; Zhikuan, J.; Sohail, A.; Tie, C.; Ting, W.; Peng, Z. Growth and Fatty Acid Composition of Sesame ( Sesamum indicum L.) Genotypes as Influence by Planting Dates and Nitrogen Fertilization in Semiarid Region of Northwest, Pakistan 1. Russ. Agric. Sci. 2016, 42, 224–229. [Google Scholar] [CrossRef]
- Bellaloui, N.; Abbas, H.K.; Ebelhar, M.W.; Mengistu, A.; Mulvaney, M.J.; Accinelli, C.; Shier, W.T. Effect of Increased Nitrogen Application Rates and Environment on Protein, Oil, Fatty Acids, and Minerals in Sesame (Sesamum indicum) Seed Grown under Mississippi Delta Conditions. Food Nutr. Sci. 2018, 9, 1112–1135. [Google Scholar] [CrossRef]
- Aytac, Z.; Gulmezoglu, N.; Saglam, T.; Kulan, E.G.; Selengil, U.; Hosgun, H.L. Changes in N, K, and Fatty Acid Composition of Black Cumin Seeds Affected by Nitrogen Doses under Supplemental Potassium Application. J. Chem. 2017, 7, 3162062. [Google Scholar] [CrossRef]
- Hodges, S.C. Soil Fertility Basics; Soil Science Extension; North Carolina State University: Raleigh, NC, USA, 2010; pp. 1–75. [Google Scholar]
- Riedell, W.E.; Pikul, J.L.; Jaradat, A.A.; Schumacher, T.E. Crop Rotation and Nitrogen Input Effects on Soil Fertility, Maize Mineral Nutrition, Yield, and Seed Composition. Agron. J. 2009, 10, 870–879. [Google Scholar] [CrossRef]
- Bellaloui, N.; Bruns, H.A.; Gillen, A.M.; Abbas, H.K.; Zablotowicz, R.M.; Mengistu, A.; Paris, R.L. Soybean seed protein, oil, fatty acids, and mineral composition as influenced by soybean-corn rotation. Agric. Sci. 2010, 1, 102–109. [Google Scholar] [CrossRef][Green Version]
- Crookston, R.K.; Kurle, J.E.; Copeland, P.J.; Ford, J.H.; Lueschen, W.E. Rotational Cropping Sequence Affects Yield of Corn and Soybean. Agron. J. 1991, 83, 108. [Google Scholar] [CrossRef]
- Harker, K.N.; O’Donovan, J.T.; Blackshaw, R.E.; Hall, L.M.; Willenborg, C.J.; Kutcher, H.R.; Gan, Y.; Lafond, G.P.; May, W.E.; Grant, C.A.; et al. Effect of agronomic inputs and crop rotation on biodiesel quality and fatty acid profiles of direct-seeded canola. Can. J. Plant Sci. 2013, 93, 577–588. [Google Scholar] [CrossRef]
- Stepien, A.; Wojtkowiak, K.; Pietrzak-Fiecko, R. Nutrient content, fat yield and fatty acid profile of winter rapeseed (Brassica napus L.) grown under different agricultural production systems. Chil. J. Agric. Res. 2017, 77, 266–272. [Google Scholar] [CrossRef]
- Hoffmann, A.; Bobinger, S.; Feifel, S.; Kurowski, C. Simple, Fast and Reliable Determination of Fat in Food According to the Caviezel Method Using Turnkey Fat Determination System; Gerstel: Linthicum, MD, USA, 2009. [Google Scholar]
- Truog, E. The determination of the readily available phosphorous of soils. Agron. J. 1930, 22, 874–882. [Google Scholar] [CrossRef]
- Kelley, K.W.; Long, J.H., Jr.; Todd, T.C. Long-term crop rotations affect soybean yield, seed weight, and soil chemical properties. Field Crops Res. 2003, 83, 41–50. [Google Scholar] [CrossRef]
- Zhong, S.; Mo, Y.; Guo, G.; Zeng, H.; Jin, Z. Effect of Continuous Cropping on Soil Chemical Properties and Crop Yield in Banana Plantation. J. Agric. Sci. Technol. 2014, 16, 239–250. [Google Scholar]
- Reeves, D.W. The role of soil organic matter in maintaining soil quality in continuous cropping systems. Soil Tillage Res. 1997, 43, 131–167. [Google Scholar] [CrossRef]
- Wyngaard, N.; Echeverria, H.E.; Sainz Rozas, H.R.; Divito, G.A. Fertilization and tillage effects on soil properties and maize yield in a Southern Pampas Argiudoll. Soil Tillage Res. 2012, 119, 22–30. [Google Scholar] [CrossRef]
- Huang, L.; Song, L.; Xia, X. Plant-Soil Feedbacks and Soil Sickness: From Mechanisms to Application in Agriculture. J. Chem. Ecol. 2013, 39, 232–242. [Google Scholar] [CrossRef] [PubMed]
- Bennett, A.J.; Bending, G.D.; Chandler, D.; Hilton, S.; Mills, P. Meeting the demand for crop production: The challenge of yield decline in crops grown in short rotations. Biol. Rev. 2012, 87, 52–71. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Sharma, M.K. Nutrient Deficiencies of Field Crops: Guide to Diagnosis and Management; CAB International: Wallingford, UK, 2013; p. 243. [Google Scholar]
- Mitchell, G.A.; Bingham, F.T.; Yermanos, D.M. Growth, mineral composition and seed characteristics of sesame as affected by nitrogen, phosphorus and potassium nutrition. Soil Sci. Am. Proc. 1974, 38, 925–931. [Google Scholar] [CrossRef]
- Panda, R.; Patra, S.K. Depletion and Contribution Pattern of Available Potassium in Indian Coastal Soils under Intensive Cropping and Fertilization. Int. J. Pure Appl. Biosci. 2017, 5, 1144–1152. [Google Scholar] [CrossRef]
- Tanaka, A.; Era, M.; Kawahara, T.; Kanyama, T.; Morita, H.; Co, S.S. Antimicrobial Activity of Fatty Acid Salts Against Microbial in Koji-Muro. MATEC Web Conf. 2016, 3002, 1–6. [Google Scholar] [CrossRef]
- McBain, J.W.; Sierichs, W.C. The solubility of sodium and potassium soaps and the phase diagrams of aqueous potassium soaps. J. Am. Oil Chem. Soc. 1948, 25, 221–225. [Google Scholar] [CrossRef]
- Aid, F. Plant Lipid Metabolism [Online First]. IntechOpen 2019. [Google Scholar] [CrossRef]
- Jadav, D.P.; Padamani, D.R.; Polara, K.B.; Parmar, K.B.; Babaria, N.B. Effect of different level of sulphur and potassium on growth, yield and yield attributes of sesame (Sesamum indicum L.). Asian J. Soil Sci. 2010, 5, 106–108. [Google Scholar]
- Thompson, A.E.; Dierig, D.A.; Knapp, S.J.; Kleiman, R. Variation in fatty acid content and seed weight in some lauric acid rich cuphea species. J. Am. Oil Chem. Soc. 1990, 67, 611–617. [Google Scholar] [CrossRef]
- Biglar, M. Profiling of major fatty acids in different raw and roasted sesame seeds cultivars. Afr. J. Biotechnol. 2012, 11, 6619–6623. [Google Scholar]
- Kurt, C. Variation in oil content and fatty acid composition of sesame accessions from different origins. Grasas Aceites 2018, 69, 1–9. [Google Scholar] [CrossRef]
- Were, B.A.; Onkware, A.O.; Gudu, S.; Welander, M.; Carlsson, A.S. Seed oil content and fatty acid composition in East African sesame (Sesamum indicum L.) accessions evaluated over 3 years. Field Crop. Res. 2006, 97, 254–260. [Google Scholar] [CrossRef]
- Mahmoud, T.S.M.; Mohamed, E.S.A.; El-Sharony, T.F. Influence of foliar application with potassium and magnesium on growth, yield and oil quality of “Koroneiki’’ olive trees. Am. J. Food Technol. 2017, 12, 209–220. [Google Scholar] [CrossRef][Green Version]
- Mondal, S. Efficiency of Sulphur source on sesame (Sesamum indicum L.) in red and lateritic soil of West Bengal. Int. J. Plant An. Envrion. Sci. 2016, 6, 65–70. [Google Scholar]
- Gerendás, J.; Führs, H. The significance of magnesium for crop quality. Plant Soil. 2013, 368, 101–128. [Google Scholar] [CrossRef]
- Gupta, R.; Singh, R. Fatty acid synthesis in leucoplasts isolated from developing seeds of Brassica campestris. J. Plant Biochem. Biotechnol. 1996, 5, 127–130. [Google Scholar] [CrossRef]
- Singh, R. Carbon and energy sources for fatty acid biosynthesis in non-photosynthetic plastids of higher plants. Proc. Indian Natl. Sci. Acad. B 1998, 64, 335–354. [Google Scholar]
- Cakmak, I.; Kirkby, E.A. Role of magnesium in carbon partitioning and alleviating photooxidative damage. Physiol. Plant. 2008, 133, 692–704. [Google Scholar] [CrossRef]
- Ali, A.; Ullah, S. Effect of nitrogen on achene protein, oil, fatty acid profile, and yield of sunflower hybrids. Chil. J. Agric. Res. 2012, 72, 564–567. [Google Scholar] [CrossRef]
- Shoja, T.; Majidian, M.; Rabiee, M. Effects of zinc, boron and sulfur on grain yield, activity of some antioxidant enzymes and fatty acid composition of rapeseed (Brassica napus L.). Acta Agric. Slov. 2018, 111, 73–84. [Google Scholar] [CrossRef][Green Version]
- Zheljazkov, V.D.; Vick, B.A.; Baldwin, B.S.; Buehring, N.; Astatkie, T.; Johnson, B. Oil Content and Saturated Fatty Acids in Sun flower as a Function of Planting Date, Nitrogen Rate, and Hybrid. Agron. J. 2009, 101, 1003–1011. [Google Scholar] [CrossRef]
- Bahrani, A.; Pourreza, J. Effect of alternate furrow irrigation and potassium fertilizer on seed yield, water use efficiency and fatty acids of rapeseed. Idesia (Arica) 2016, 34, 35–41. [Google Scholar] [CrossRef]
- Kaptan, M.A.; Koca, Y.O.; Canavar, Ö. Effect of N-P-K fertilization on mineral content and fatty acid compounds of corn seed. Adnan Menderes Üniversitesi Ziraat Fakültesi Derg. 2017, 14, 19–22. [Google Scholar] [CrossRef]
- Suzer, S. Effects of plant nutrition on canola (Brassica napus L.) growth. Trak. Univ. J. Nat. Sci. 2015, 16, 87–90. [Google Scholar]
- Rathke, G.W.; Christen, O.; Diepenbrock, W. Effects of nitrogen source and rate on productivity and quality of winter oilseed rape (Brassica napus L.) grown in different crop rotations. Filed Crop. Res. 2005, 94, 103–113. [Google Scholar] [CrossRef]
- Podkowinski, J.; Jelenska, J.; Sirikhachornkit, A.; Zuther, E.; Haselkorn, R.; Gornicki, P. Expression of Cytosolic and Plastid Acetyl-Coenzyme Carboxylase Genes in Young Wheat Plantsl[w]. Plant Physiol. 2003, 131, 763–772. [Google Scholar] [CrossRef]
- Xu, Z.; Li, J.; Guo, X.; Jin, S.; Zhang, X. Metabolic engineering of cottonseed oil biosynthesis pathway via RNA interference. Sci. Rep. 2016, 6, 1–14. [Google Scholar] [CrossRef]
- Wacal, C.; Ogata, N.; Sasagawa, D.; Handa, T.; Basalirwa, D.; Acidri, R.; Ishigaki, T.; Yamamoto, S.; Nishihara, E. Seed yield, crude protein and mineral nutrient contents of sesame during a two-year continuous cropping on upland field converted from a paddy. Filed Crop. Res. 2019, 240, 125–133. [Google Scholar] [CrossRef]
- Ngure, J.W.; Cheng, C.; Yang, S.; Lou, Q.; Li, J.; Qian, C.; Chen, J.; Chen, J. Cultivar and seasonal effects on seed oil content and fatty acid composition of cucumber as a potential industrial crop. J. Am. Soc. Hort. Sci. 2015, 140, 362–373. [Google Scholar] [CrossRef]
- Izquierdo, N.G.; Aguirrezábal, L.A.N.; Andrade, F.H.; Geroudet, C.; Valentinuz, O.; Pereyra Iraola, M. Intercepted solar radiation affects oil fatty acid composition in crop species. Filed Crop. Res. 2009, 114, 66–74. [Google Scholar] [CrossRef]
- Garcés, R.; Sarmiento, C.; Mancha, M. Temperature regulation of oleate desaturase in sunflower (Helianthus annuus L.) seeds. Planta 1992, 186, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Kabbaj, A.; Vervoort, V.; Abbot, A.; Tersac, M.; Berville, A. Expression of stearate oleate and linoleate desaturase genes in sunflower with normal and high oleic acid contents. Helia 1996, 19, 1–7. [Google Scholar]
- Alpaslan, M.; Boydak, E.; Hayta, M.; Gerçek, S.; Simsek, M. Effect of row space and irrigation on seed composition of Turkish sesame (Sesamum indicum L.). J. Am. Oil Chem. Soc. 2001, 78, 933–935. [Google Scholar] [CrossRef]
- Uzun, B.; Arslan, C.; Furat, S. Variation in fatty acid compositions, oil content and oil yield in a germplasm collection of sesame (Sesamum indicum L.). J. Am. Oil Chem. Soc. 2008, 85, 1135–1142. [Google Scholar] [CrossRef]
| Exchangeable Cations | ||||||||
|---|---|---|---|---|---|---|---|---|
| Field | pH (H2O) | EC (dS m−1) | TN (g kg−1) | C/N ratio | P (mg kg−1) | K (mg kg−1) | Ca (mg kg−1) | Mg (mg kg−1) |
| A | 5.39 | 0.05 | 2.32 | 9.66 | 29.0 | 248 | 918 | 108 |
| B | 5.44 | 0.04 | 2.94 | 8.11 | 68.4 | 112 | 1220 | 297 |
| C | 5.73 | 0.04 | 2.90 | 7.84 | 76.2 | 113 | 1632 | 307 |
| D | 6.01 | 0.04 | 2.86 | 7.74 | 46.4 | 101 | 1904 | 353 |
| Year | Daily/Monthly | June | July | August | September | Average Seasonal |
|---|---|---|---|---|---|---|
| 2015 | Mean daily temperature °C | 21 | 25.0 | 25.6 | 21.0 | 23.2 |
| Mean monthly rainfall (mm) | 132 | 102.5 | 123.0 | 171.5 | 132.3 | |
| 2016 | Mean daily temperature °C | 22 | 25.7 | 26.4 | 22.9 | 24.3 |
| Mean monthly rainfall (mm) | 135 | 69.5 | 126.5 | 330.0 | 165.3 |
| SFA | MUFA | PUFA | Capric (C10:0) | Palmitic (C16:0) | Oleic (C18:1) | |||
|---|---|---|---|---|---|---|---|---|
| Year | Fields | TFA (%) | % Weight of Total Fatty Acid (TFA) | |||||
| 2015 | A | 51.4 ± 3.51 a | 10.7 ± 0.62 ab | 16.2 ± 1.34 a | 17.5 ± 2.19 a | 0.29 ± 0.04 a | 6.84 ± 0.31 a | 30.8 ± 0.64 a |
| B | 54.7 ± 0.77 a | 11.4 ± 0.67 a | 17.6 ± 0.33 a | 19.0 ± 0.33 a | 0.34 ± 0.08 a | 7.50 ± 0.30 a | 31.9 ± 0.83 a | |
| C | 52.7 ± 0.95 a | 9.20 ± 0.76 ab | 17.6 ± 0.27 a | 20.6 ± 0.32 a | 0.43 ± 0.03 a | 6.94 ± 0.15 a | 33.0 ± 0.79 a | |
| D | 50.5 ± 0.71 a | 8.08 ± 0.63 b | 17.8 ± 0.43 a | 20.2 ± 0.22 a | 0.45 ± 0.01 a | 7.24 ± 0.16 a | 34.7 ± 1.34 a | |
| ANOVA (p-values) | ns | * | ns | ns | ns | ns | ns | |
| 2016 | A | 53.1 ± 0.72 a | 9.19 ± 0.43 a | 19.5 ± 0.20 a | 19.9 ± 0.13 ab | 0.44 ± 0.01 a | 6.55 ± 0.13 a | 36.5 ± 0.89 a |
| B | 51.1 ± 1.01 a | 8.82 ± 0.62 a | 18.8 ± 0.11 a | 19.2 ± 0.13 b | 0.41 ± 0.01 a | 6.58 ± 0.12 a | 36.4 ± 0.90 a | |
| C | 51.5 ± 1.31 a | 8.79 ± 0.44 a | 18.9 ± 0.43 a | 19.8 ± 0.58 ab | 0.42 ± 0.01 a | 6.86 ± 0.15 a | 36.2 ± 1.23 a | |
| D | 51.4 ± 0.59 a | 13.2 ± 6.25 a | 19.8 ± 0.26 a | 21.3 ± 0.31 a | 0.42 ± 0.03 a | 7.10 ± 0.17 a | 37.8 ± 0.55 a | |
| ANOVA (p-values) | ns | ns | ns | * | ns | ns | ns | |
| Average | A | 52.2 ± 1.65 a | 9.92 ± 0.47 a | 17.9 ± 0.96 a | 18.7 ± 1.12 a | 0.36 ± 0.04 a | 6.69 ± 0.16 a | 33.7 ± 1.37 a |
| B | 52.9 ± 0.98 a | 10.1 ± 0.71 a | 18.2 ± 0.32 a | 19.1 ± 0.17 a | 0.38 ± 0.04 a | 7.04 ± 0.25 a | 34.1 ± 1.16 a | |
| C | 52.1 ± 0.77 a | 9.00 ± 0.40 a | 18.3 ± 0.37 a | 20.2 ± 0.34 a | 0.42 ± 0.01 a | 6.90 ± 0.10 a | 34.6 ± 0.98 a | |
| D | 51.0 ± 0.45 a | 10.7 ± 3.04 a | 18.8 ± 0.50 a | 20.7 ± 0.31 a | 0.43 ± 0.01 a | 7.17 ± 0.11 a | 36.3 ± 0.95 a | |
| Source of variation | ||||||||
| Year | ns | ns | *** | ns | ns | * | *** | |
| Field | ns | ns | ns | ns | ns | ns | ns | |
| Year × Field | ns | ns | ns | ns | ns | ns | ns | |
| Exchangeable Cations | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Year | Fields | pH (H2O) | EC (dS m−1) | TN g kg−1 | C/N Ratio | P (mg kg−1) | K (mg kg−1) | Ca (mg kg−1) | Mg (mg kg−1) |
| 2015 | A | 5.26 ± 0.06 c | 0.15 ± 0.00 a | 2.56 ± 0.00 a | 10.5 ± 0.21 b | 58.4 ± 4.43 a | 389.9 ± 76.1 a | 1191.8 ± 12.6 b | 307.3 ± 12.0 a |
| B | 5.39 ± 0.04 bc | 0.10 ± 0.01 b | 2.46 ± 0.02 b | 10.3 ± 0.24 b | 43.5 ± 2.78 b | 177.9 ± 12.3 b | 1182.6 ± 82.5 b | 283.2 ± 20.9 a | |
| C | 5.66 ± 0.06 b | 0.11 ± 0.01 ab | 2.62 ± 0.02 a | 9.59 ± 0.23 b | 47.6 ± 2.32 ab | 226.4 ± 11.5 ab | 1492.4 ± 42.6 a | 288.0 ± 12.4 a | |
| D | 6.14 ± 0.07 a | 0.07 ± 0.00 b | 2.11 ± 0.02 c | 11.9 ± 0.21 a | 35.5 ± 1.76 b | 208.4 ± 2.76 b | 1711.7 ± 49.6 a | 255.2 ± 1.17 a | |
| ANOVA (p-values) | *** | ** | *** | *** | ** | * | *** | ns | |
| 2016 | A | 5.46 ± 0.05 b | 0.10 ± 0.00 a | 2.12 ± 0.07 a | 9.81 ± 0.03 b | 53.3 ± 3.94 a | 325.9 ± 44.5 a | 1174.3 ± 84.5 c | 298.9 ± 26.8 b |
| B | 5.64 ± 0.20 ab | 0.07 ± 0.01 b | 1.98 ± 0.03 a | 10.3 ± 0.06 a | 24.2 ± 2.78 b | 82.9 ± 14.9 b | 1518.2 ± 119 bc | 321.7 ±17.2 ab | |
| C | 5.80 ± 0.07 ab | 0.06 ± 0.01 b | 2.13 ± 0.06 a | 10.1 ± 0.09 ab | 29.7 ± 4.97 b | 110.9 ± 13.0 b | 1726.7 ± 83.2 ab | 339.2 ±18.9 ab | |
| D | 6.10 ± 0.06 a | 0.07 ± 0.00 ab | 2.00 ± 0.02 a | 10.3 ± 0.12 a | 17.2 ± 2.87 b | 97.0 ± 9.10 b | 2100.1 ± 36.4 a | 400.4 ± 17.4 a | |
| ANOVA (p-values) | * | ** | ns | ** | *** | *** | *** | * | |
| Average | A | 5.36 ± 0.06 c | 0.12 ± 0.01 a | 2.34 ± 0.10 a | 10.2 ± 0.19 b | 55.8 ± 2.88 a | 357.9 ± 41.9 a | 1183.1 ± 38.4 c | 303.1 ± 13.3 a |
| B | 5.51 ± 0.11 bc | 0.09 ± 0.01 ab | 2.22 ± 0.11 a | 10.3 ± 0.11 ab | 33.8 ± 4.67 a | 130.4 ± 22.9 b | 1350.4 ± 99.0 bc | 302.5 ± 14.9 a | |
| C | 5.73 ± 0.05 b | 0.09 ± 0.01 ab | 2.37 ± 0.11 a | 9.85 ± 0.16 b | 38.7 ± 4.70 a | 168.7 ± 27.0 b | 1609.5 ± 67.0 ab | 313.6 ± 15.3 a | |
| D | 6.12 ± 0.04 a | 0.08 ±0.00 b | 2.06 ± 0.03 a | 11.1 ± 0.38 a | 26.3 ± 4.36 a | 152.7 ± 25.3 b | 1905.9 ± 91.1 a | 327.8 ± 33.4 a | |
| Source of variation | |||||||||
| Year | * | ** | *** | ** | *** | ** | *** | *** | |
| Field | *** | ** | *** | ** | *** | *** | *** | ns | |
| Year × Field | ns | * | *** | *** | ns | ns | ns | ** | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wacal, C.; Ogata, N.; Basalirwa, D.; Sasagawa, D.; Kato, M.; Handa, T.; Masunaga, T.; Yamamoto, S.; Nishihara, E. Fatty Acid Composition of Sesame (Sesamum indicum L.) Seeds in Relation to Yield and Soil Chemical Properties on Continuously Monocropped Upland Fields Converted from Paddy Fields. Agronomy 2019, 9, 801. https://doi.org/10.3390/agronomy9120801
Wacal C, Ogata N, Basalirwa D, Sasagawa D, Kato M, Handa T, Masunaga T, Yamamoto S, Nishihara E. Fatty Acid Composition of Sesame (Sesamum indicum L.) Seeds in Relation to Yield and Soil Chemical Properties on Continuously Monocropped Upland Fields Converted from Paddy Fields. Agronomy. 2019; 9(12):801. https://doi.org/10.3390/agronomy9120801
Chicago/Turabian StyleWacal, Cosmas, Naoki Ogata, Daniel Basalirwa, Daisuke Sasagawa, Masako Kato, Takuo Handa, Tsugiyuki Masunaga, Sadahiro Yamamoto, and Eiji Nishihara. 2019. "Fatty Acid Composition of Sesame (Sesamum indicum L.) Seeds in Relation to Yield and Soil Chemical Properties on Continuously Monocropped Upland Fields Converted from Paddy Fields" Agronomy 9, no. 12: 801. https://doi.org/10.3390/agronomy9120801
APA StyleWacal, C., Ogata, N., Basalirwa, D., Sasagawa, D., Kato, M., Handa, T., Masunaga, T., Yamamoto, S., & Nishihara, E. (2019). Fatty Acid Composition of Sesame (Sesamum indicum L.) Seeds in Relation to Yield and Soil Chemical Properties on Continuously Monocropped Upland Fields Converted from Paddy Fields. Agronomy, 9(12), 801. https://doi.org/10.3390/agronomy9120801





