Abstract
Nitrogen (N) fertilizers are needed to enhance maize (Zea mays L.) production. Maize plays a major role in the livestock industry, biofuels, and human nutrition. Globally, less than one-half of applied N is recovered by maize. Although the application of N fertilizer can improve maize yield, excess N application due to low knowledge of the mechanisms of nitrogen use efficiency (NUE) poses serious threats to environmental sustainability. Increased environmental consciousness and an ever-increasing human population necessitate improved N utilization strategies in maize production. Enhanced understanding of the relationship between maize growth and productivity and the dynamics of maize N recovery are of major significance. A better understanding of the metabolic and genetic control of N acquisition and remobilization during vegetative and reproductive phases are important to improve maize productivity and to avoid excessive use of N fertilizers. Synchronizing the N supply with maize N demand throughout the growing season is key to improving NUE and reducing N loss to the environment. This review examines the mechanisms of N use in maize to provide a basis for driving innovations to improve NUE and reduce risks of negative environmental impacts.
1. Nitrogen Application in Crop Production
Crop production is the principal cause of human modification of the global nitrogen (N) cycle [1,2]. Nitrogen is a crucial input in agriculture to sustain agricultural livelihoods and support the increasing human population [3]. From 1930 to 1960, N fertilizer use escalated from 1.3 to 10.2 million metric tons (MMt) [4]. During this time of increased N fertilizer use, there was a concomitant increase in N losses to the environment, leaching of nitrate into groundwater, aquatic eutrophication, emission of ammonia and nitrous oxide, and soil acidification [5,6,7]. The global demand for N fertilizers was 112 MMt in 2014 [8] and is predicted to reach 240 MMt by 2050 [9].
Since the 1960s, the global use of N fertilizers has grown nearly seven-fold [10,11], but the overall yield increase was only 2.4-fold [12,13]. Worldwide, more than 50% to 75% of applied N fertilizer is not taken up by crops [14,15,16], and recovery of applied N by maize (Zea mays L.) rarely exceeds 50% [17,18]. Nitrogen use efficiency (NUE) can be defined as the increase in grain yield per unit of applied N and is the product of absorption efficiency (increase in absorbed N per unit of applied N) and utilization efficiency (increase in grain yield per unit of absorbed N) [19,20].
Synthetic fertilizers are now the primary source of N applied to cropland, although organic N from livestock manure remains important [20]. The environmental cost of excess N application in Europe is estimated at 78 to 357 billion US dollars per year [21]. Nitrogen availability to plants is the difference between N supply and losses due to pathways such as leaching, runoff, ammonia volatilization, additional gaseous N losses, and immobilization, highlighting the multifaceted interaction among the soil, plant, and atmosphere in regulating N availability and uptake by plants [22]. Nitrogen is an essential nutrient for maize and a key determinant of grain yield, particularly through its role in photosynthesis and other biological processes such as absorption of water and minerals, vacuole storage, and xylem transport. However, N application is a great concern in maize production due to the negative effects of excess application on groundwater quality [23]. Mineral N fertilizers manufactured by the Haber–Bosh process can represent up to 50% of the operational cost in crop production, depending on the crop [24]. Comparatively, the overall cost of reduced N fertilizer requirements for grain crops due to crop rotation with legumes is just 4% to 71% of the cost of an equivalent amount of synthetic N fertilizer [25,26]. Legumes can also increase the yield of subsequent grain crops and have a delayed N release compared to synthetic fertilizers [27,28]. Therefore, it is environmentally beneficial to include legumes in cropping systems [20,26].
2. Nitrogen Use Efficiency in Maize
Cereal grains such as maize, rice (Oryza sativa L.), and wheat (Triticum aestivum L.) provide 60% to 94% of the world’s nourishment [20,29]. Among these, maize ranks second to wheat in global production, with 500 MMt produced annually on 130 million hectares [30]. Maize is used for livestock feed, biofuel, and human consumption [31,32], and global demand for maize is anticipated to increase by 16% by 2027, due primarily to a > 50% increase in consumption by livestock [33].
Nitrogen accumulation in aboveground maize biomass typically increases as the N level in the soil increases [34], and aboveground biomass can be reduced if the soil N supply is insufficient for crop demand [35]. Aboveground biomass production also influences maize N uptake potential [36]. Nitrogen accumulated in maize biomass is partitioned into grain and Stover, with luxury N uptake occurring when N supply exceeds the minimum requirements for maximum grain yield [37,38]. Several factors can affect N uptake by maize, of which soil moisture, temperature, structure, and bulk density are most important [39,40]. Thus, improvement in aboveground biomass production requires adequate N supply and uptake by maize.
Increases in maize grain yield are associated with increases in aboveground biomass [41,42,43], and grain yield improvements are associated with increased harvest index [41,44]. Harvest index is the ratio of grain to total aboveground biomass (grain, cob, and Stover) per unit area at physiological maturity [45,46]. Harvest index of maize produced in favorable growing environments is about 50% [46]. High levels of harvest index in maize typically occur with intermediate values of total aboveground biomass relative to grain and decreases when individual plants are small (as with high plant density) or large (as with low plant density) [47]. Harvest index can increase with heterosis if the increase in kernel number is proportionately greater than that in total aboveground dry matter accumulation during the grain-filling period [48,49]. Kernel number is related to dry matter accumulation [50] and partitioning during the periods of anthesis and kernel establishment [51,52].
Nitrogen use efficiency has two components: (i) the efficiency of crop uptake of applied N and (ii) the transformation efficiency of total aboveground crop N uptake to grain yield [53]. Nitrogen use efficiency in maize is multifaceted and rests on N availability in the soil and rhizosphere, and the utilization ability of the crop [22,54]. This is complicated by the many soil and environmental factors that control the rates and products of N mineralization [55]. The use of soil N by maize involves numerous steps, including uptake, translocation, and remobilization (Figure 1). This occurs during two phases: (i) the N assimilation phase and (ii) the remobilization phase [22,38,56]. During the N assimilation and remobilization phases, young roots and leaves are sink organs that efficiently absorb and assimilate inorganic N for amino acids and protein synthesis.
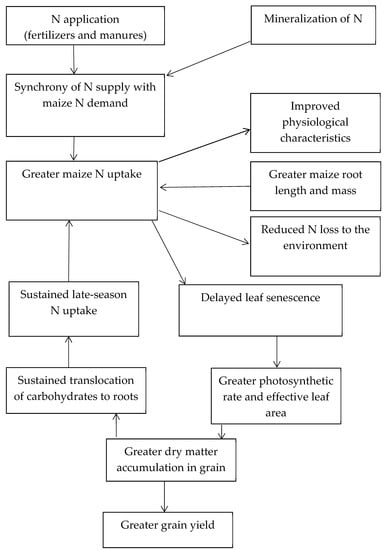
Figure 1.
Diagram of nitrogen (N) use in maize.
The components of NUE include recovery efficiency of applied nitrogen (RE), physiological efficiency of applied nitrogen (PE), and agronomic efficiency of applied nitrogen (AE) [38]. The recovery efficiency of applied N reflects the efficiency of aboveground N uptake per unit of N applied and is closely associated with assimilation capacity and decreases at greater N supply [57]. Physiological efficiency is the efficiency by which N in aboveground plant tissues is converted to grain, and AE is grain yield per unit of N applied [58,59]. High NUE in maize is a combination of high N uptake and high N utilization efficiency [60]. With a high level of N application, genetic differences in maize grain yield are mainly due to variation in N uptake [53]. Since RE in maize is commonly < 50% [17,18], there is a great opportunity to enhance maize RE and yield and reduce environmental impact [61,62].
3. Nitrogen Assimilation and Utilization by Maize
Nitrate and ammonium are the forms of inorganic N most greatly absorbed by the roots of maize [63]. In warm and moist conditions, ammonium is oxidized to nitrate in soils almost as rapidly as it is formed [64]. Thus, nitrate is usually the dominant form of plant-available N in soils [65]. Assimilated nitrate is stored in cell vacuoles, where it serves as a reservoir for N remobilization when N uptake declines [22]. This pool of nitrate is the major source of N that contributes to grain filling. The absorption of nitrate is a two-step process that involves a collective set of enzymes, namely nitrate reductase and nitrite reductase, which catalyze and activate the reduction of nitrate to ammonium. The reduction of nitrate to ammonium is the most limiting phase in N assimilation [66]. Nitrate reductase increases leaf nitrate quantity at initial growth stages, and therefore can be used as a selection criterion for maize genotypes with superior N absorption and grain yield [38,67].
Within plants, ammonium is incorporated into amino acids through glutamine synthetase and glutamate synthase [68]. These are the main enzymes that convert inorganic N to organic molecules for other metabolic activities [69]. Glutamine synthetase concentration is a key regulator of plant growth that is controlled by N accumulation in plants and can be used as an indicator of grain yield and to differentiate sink leaves versus source leaves [70,71]. The transformation of glutamate to glutamine in leaves by glutamine synthetase is active in new vegetative tissues and is positively correlated with post-anthesis N uptake and negatively correlated with the percentage of N in grain from remobilization [72]. All assimilated N is directed through the processes catalyzed by glutamine synthetase, and nitrate accumulation catalyzed by nitrate reductase and glutamine synthetase is a key factor regulating NUE in maize [56,67].
Maize growth is dependent on the quantity of photosynthetically captured active radiation and the efficiency by which it is converted to aboveground biomass [73]. Enhanced photosynthetic capacity can contribute to greater biomass and grain yield [74]. The amount of intercepted photosynthetically active radiation is influenced by the size and architecture of the crop canopy [75]. The distribution of N within the canopy, with its significance for leaf and canopy photosynthesis, is a foremost factor regulating radiation use efficiency in maize [76]. Following anthesis, the rate of maize N uptake declines, and hybrids with greater N uptake have delayed leaf senescence (Figure 1) and prolonged effective leaf area for photosynthesis [77].
Maize hybrids with delayed senescence have a greater ability to take up N during the grain-filling period since continued leaf activity stimulates uptake of N [78]. During the grain-filling period, a decline in N supply decreases dry matter partitioning to grain [79]. Maize genotypes with longer stay-green maintain leaf chlorophyll for a greater duration, which can increase grain yield by 10% to 12% [80]. Figure 1 is a simple diagram illustrating the mechanisms of N use in maize.
4. Nitrogen Remobilization in Maize
Efficient N remobilization and leaf longevity are key features contributing to an improved yield of contemporary maize hybrids [81,82]. The magnitude and duration of active leaf tissue affects N uptake and the quantity of N accessible for remobilization [77]. Remobilization of N is greater when post-anthesis N accumulation is less and is negatively correlated with leaf senescence [83]. Extended duration of leaf metabolic activity enhances the ratio of source leaves to sink leaves throughout the grain-filling period [81,82]. When maize N uptake is sustained throughout the grain-filling period, less N is mobilized from vegetative organs, thereby increasing leaf area duration, delaying senescence, and enhancing dry matter accumulation (Figure 1) [13]. High maize yield is attributed to the maintenance of photosynthate source and N uptake throughout the grain-filling period [84]. During maize reproductive growth, increasing and sustaining the capacity for N uptake in soils with low N supply is an avenue for improving NUE, and is dependent on the ability of the plant to supply the root system with the needed assimilates for root development and N uptake for sustained photosynthesis [85]. At a given level of N supply, increases in grain yield among maize genotypes are not always due to improvements in N assimilation and usage, but rather enhanced NUE as a result of a more effective N remobilization [86].
Remobilization of N by maize early in the growing season contributes to vegetative development and formation of the reproductive sink capacity, which affects grain yield [87]. Improved synchrony between N supply and crop N uptake is key to enhancing NUE [88]. Shortage of N uptake due to physiological restraints can arise even when there is a large sink capacity for grain protein synthesis, resulting in N loss, enhanced remobilization of N from vegetative tissues to grain, and low NUE [89]. Relationships between N accessibility, late-season N uptake by maize, and remobilization of N are influenced by soil N supply [84,90]. Low N supply following anthesis can lead to early leaf senescence since the development of grain requires more N than the maintenance of vegetative tissues [91].
Maize hybrids with rapid early-season N uptake, efficient N remobilization, and complete leaf senescence can perform well in environments with low N supply late in the growing season, while hybrids that are proficient in late-season N uptake require conditions with greater soil N availability late in the growing season to optimize grain yield [80]. Maize hybrids with enhanced NUE have the capacity to achieve greater yield at a given level of N supply, particularly at low levels, through enhanced N uptake and more efficient translocation of N to grain [19,91,92]. There is an inverse relationship between N remobilization and N assimilation in maize, which results from a correlation between photosynthesis and N uptake [83]. Growing environments that are favorable for post-anthesis photosynthesis promote N uptake and are therefore associated with reduced N remobilization. When growing environments are unfavorable for photosynthesis, post-anthesis N uptake in maize is reduced; therefore, the quantity of N remobilization is positively associated with aboveground N content at anthesis, and grain N content is associated with the level of post-anthesis N uptake and assimilation [83]. Disparities in post-anthesis N uptake in grain are mainly due to differences in total aboveground maize N uptake following anthesis [93].
During senescence, photosynthesis is reduced and translocation of carbohydrates to roots declines, contributing to a reduction in N uptake and an upsurge in N remobilization [94]. Genotypic differences in the photosynthetic rate at the cellular level of maize are associated with differences in N remobilization and can be used in breeding programs to improve NUE [95]. A substantial fraction of the N in maize leaf tissue is involved in photosynthetic processes, and the photosynthetic rate is closely associated with leaf N content [96]. The level of N remobilization from the stalk of maize is related to the level of N remobilization from leaves and N accumulation at flowering [97]. Following anthesis, leaf N content quickly declines due to remobilization to grain, especially in conditions with low N supply [98]. With greater soil N supply, there is generally no relationship between leaf N content and N remobilization [98]. Nitrogen supply from floral initiation to anthesis regulates kernel number and subsequently kernel weight [99]. Nitrogen uptake and remobilization are independently inherited traits, so favorable alleles could be combined when breeding for NUE [100].
5. Nitrogen Nutrition Index
Nitrogen nutrition index defines the N status of a crop and is the ratio between aboveground N uptake and the critical N uptake required for maximum aboveground biomass. Aboveground N accumulation in maize occurs most rapidly from mid-vegetative growth until anthesis and then decreases until senescence [101]. During maize vegetative growth, the amount of structural tissues increases more rapidly compared to metabolic tissues, such that N content is greatest in young leaves [102,103]. Nitrogen content in leaves declines as leaf area increases, demonstrating that N content depends on plant biomass [76,104]. Optimal N fertilizer application in maize requires knowledge of the adequate N content for a given amount of maize biomass, along with a rapid and accurate method to determine N content and biomass [105]. Nitrogen nutrition index provides the critical N content for a given amount of biomass [106].
Leaf and canopy N content in maize are positively correlated with their chlorophyll content and are indicators of photosynthetic ability [107,108]. Canopy chlorophyll can be used to estimate canopy-level N content in maize [107]. However, at the canopy level, the estimation of chlorophyll and N content is much more challenging [109,110]. There is a close relationship between leaf N content and maize grain yield [111]. Critical values or ranges of leaf chlorophyll meter readings in maize have been used to determine N deficiency to improve N management [112,113,114]. However, knowledge about the relationship between the N nutrition index and spectral canopy reflectance is limited [115].
6. Grain Nitrogen Harvest in Maize
Nitrogen harvest index is the proportion of N in grain relative to total aboveground biomass and is an indicator of N translocation efficiency [116]. Nitrogen harvest index is not directly linked to grain yield [45]. However, grain yield and grain N content are important for assessing NUE and serve as selection criteria for breeding programs and N management practices [117]. Nitrogen harvest index is greater in maize hybrids that sustain a high rate of photosynthesis during the grain-filling period, leading to greater NUE [118].
7. Nitrogen Supply
Nitrogen availability is one of the most significant constraints to crop growth [119], and the application of N through mineral and organic fertilizers plays a vital role in sustaining crop production [12]. Nitrogen application can play a significant role in improving soil fertility [120]. Fertilization of N can increase grain yield and biomass in maize [121,122]. The leading source of applied N worldwide is synthetic fertilizer, followed closely by livestock manure [123,124,125]. Application of livestock manure with a high carbon-to-N ratio can lead to N immobilization in the soil, thereby restricting maize N uptake and potentially yield in the short-term [126]. Soil organic matter can serve as a temporary sink for available soil N not utilized by maize to reduce N losses [127]. Organic fertilizers can lead to greater soil total N than mineral fertilizers after several cropping seasons [128,129], increase organic matter in soil, enhance soil porosity, and improve soil water holding capacity [130], thereby enhancing maize growth and nutrient uptake [131]. Application of composted manure for maize production has been shown to produce less greenhouse gas emissions compared to mineral N fertilizer in maize cropland due to slower release of available N [126]. Additionally, the application of organic N (manure) has been shown to increase uptake of N, P, K, Ca, Mg, Zn, Fe, and Cu of maize grown on Alfisol soils compared to NPK [131].
Mineral fertilizers can enhance maize grain yield and N uptake but also result in greater residual NO3−-N levels in the soil compared to organic fertilizers [132]. The application of mineral fertilizers can directly or indirectly cause changes to the chemical, physical, and biological properties of the soil [133]. The usage of mineral N, P, and K fertilizers can increase the availability of N, P, K, Ca, Mg, Fe, Cu, Mn, and Zn to maize [134,135]. The application of Zn has been shown to facilitate the tasseling/silking of maize in combination with greater N doses [136,137]. Magnesium stimulates a large number of enzymes in maize, can increase the rate of mineral N transformation into proteins [138], and plays a key role in chlorophyll and the development of kernels in maize [139]. Potassium deficiency strongly affects N metabolism and photosynthesis in maize [140]. The application of inorganic N fertilizers has been associated with loss of soil organic carbon through enhanced respiration and greenhouse gas emissions [141,142]. This loss reduces soil productivity and agronomic efficiency of applied N and changes the balance of N and carbon in soil in favor of greenhouse gas emissions [143]. However, there can be a positive collaboration between organic manures and urea as a N source [144], and integrated organic and inorganic N application can lead to greater maize yield compared to sole application of organic or inorganic N sources [145].
Due to changes in the form of N in the soil, maize NUE is low and rarely exceeds 60% [146,147]. Nitrogen takes on nine different forms in the soil, conforming to different oxidative conditions (nitrate, nitrogen dioxide, nitrite, nitric oxide, nitrous oxide, dinitrogen, ammonia, ammonium, and organic N) [148]. Nitrogen mineralization is the conversion of organic N to inorganic forms, immobilization is the uptake or assimilation of inorganic forms of N by microbes and other soil heterotrophs, nitrification is the conversion of ammonium to nitrite and then nitrate, and denitrification is the conversion of nitrate to nitrous oxide and then dinitrogen gas [148]. Most of the N in the soil originates as dinitrogen gas, and this inert N cannot be used by plants until it is transformed to ammonium or nitrate [149]. Investigations using ion-selective microelectrode methods demonstrated that maize absorption is suppressed from the root apex to 60 mm behind the root apex when ammonium and nitrate are both supplied at the same time [150]
Mineralization of N is influenced by several factors, including the source and carbon-to-N ratio of organic material, soil texture, temperature, water content, and pH, the microbial community, and agronomic practices [151]. Mineralization of organic amendments intensifies available N and, depending on the timing of mineralization, crop requirements for N, and sources and amounts of applied N can be a key component of an integrated N management approach to achieve high NUE while minimizing N loss [152]. However, accurate estimation of N mineralization from soil and organic sources of applied N is challenging due to the large number of site, environmental, and agronomic factors influencing it [153]. Integrated application of organic and mineral fertilizers at appropriate rates can be an effective strategy for improving maize N uptake and yield [154,155,156].
8. Maize Adaptation to Low Nitrogen Supply
Nitrogen supply affects root mass and morphology, which are important for acquiring spatially heterogeneous N in soil, particularly for newly mineralized N [157]. There is a close relationship between root length and N accumulation in maize seedlings [158,159]. Root size is vital for N uptake over the entire growth period of maize, but the initial establishment of a large root system is indispensable [159]. In response to low N supply, maize develops a greater root-to-shoot ratio and undergoes a slower rate of phenological development, with a greater proportion of root biomass enhancing the absorption capacity of N [160].
At the same quantity of accumulated N, maize genotypes with enhanced NUE typically allocate a greater proportion of N to the root system and develop a greater root system compared to less efficient genotypes [161,162,163]. This may be related to their capacity to sustain root development after anthesis, especially under conditions of low N supply [164]. Grain yield of maize is influenced by N supply, level of N uptake, and capacity to effectively partition N to grain during the grain-filling period [165]. High-yielding maize hybrids typically have greater yield response to the N application rate than low-yielding hybrids [19]. Sustainable post-anthesis uptake of N and maintenance of green leaf area until maturity are key traits of maize hybrids with high yield potential [166]. These traits are especially important for root longevity and N uptake in conditions of low N supply [166,167].
The efficiency of maize to utilize soil N depends on its ability to acquire, utilize, and translocate N, which is influenced by root morphology and the biochemical and physiological processes involved in nitrate assimilation [168]. Nitrogen uptake efficiency (total aboveground N accumulation at a given level of N supply) is separated into constituent traits, which describe root characteristics associated with genetic variation in N uptake [169]. Root tissue that is active in N acquisition, and the absorption rate per unit of root mass, influence the rate of N uptake, and both component traits differ genetically [169]. Morphological development of the root system affects the processes involved in nitrate acquisition [169]. Differential lateral root development between genotypes affects the translocation of solutes from the root system [170], as there is greater activity of nitrate reduction in root apices compared to basal parts [171].
9. Strategies to Improve Nitrogen Use Efficiency in Maize
Improvement in maize grain yield since the 1930s has been one of the greatest achievements in agriculture [46]. High NUE is critical for sustainable maize production [151], but globally, maize RE is only about 35% to 55% [61,62]. There is an opportunity to increase the NUE of maize through enhanced understanding of the mechanisms of N use to guide appropriate N application. Nitrogen use efficiency in maize is affected by soil N supply; hence, understanding the factors influencing N absorption, assimilation, and mobilization are key to increasing NUE to circumvent excessive N application and the subsequent negative environmental impacts (Figure 1) [172]. Increasing N supply results in reductions in all indices of NUE (i.e., RE, PE, and AE) [173]. One strategy to reduce N losses is to increase NUE through management practices to enhance the synchrony between N supply and crop demand throughout the growing season [174,175,176]. It is also important to recognize the critical stages of maize phenological development that are associated with NUE [125]. Further research should be conducted to assess the effects of the different fertilizer types on the mechanisms of N use in maize.
Since the maize harvest index is already high, increasing N uptake, aboveground biomass, and grain yield are paths to increasing NUE [177]. Developing hybrids capable of taking up more N or utilizing assimilated N more effectively can improve NUE, while knowledge on the physiology and genetics of N uptake and utilization is critical for the development of N-efficient hybrids [19,38,54]. Nitrogen use efficiency is controlled by a complex set of interactions among genotype, growing environment, and agronomic management [20,178]. Therefore, more extensive screening of a wide range of genotypes covering genetic diversity is needed to identify specific elements controlling NUE and productivity [38,178].
Sustainability is founded on the standard that present needs are met without compromising the needs of the future. Integrated collaboration between public and private sectors will improve the understanding and management of the biological and agronomic factors controlling NUE [20]. This will require a multidisciplinary approach, integrating expertise from crop developmental biology, physiology, genomics, genetics, breeding, simulation modeling, and agronomy [179]. Therefore, rigorous collaboration among stakeholders (i.e., environmental agencies, policy-makers, researchers, farm advisors, and farmers) and skillful implementation of indicators and guidelines are needed to improve NUE in maize production [180].
Author Contributions
Conceptualization, A.E.A., Q.C., and J.A.C.; methodology, A.E.A.; software formatting/writing style, J.A.C.; validation, Q.C. and J.A.C.; formal analysis, Q.C. and J.A.C.; investigation, A.E.A.; resources, Q.C.; data collection/information, A.E.A.; writing, A.E.A.; editing, J.A.C.; visualization, A.E.A. and J.A.C.; supervision, Q.C. and J.A.C.; project administration, Q.C. and J.A.C.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| agronomic efficiency of applied nitrogen | AE |
| nitrogen | N |
| nitrogen use efficiency | NUE |
| physiological efficiency of applied nitrogen | PE |
| recovery efficiency of applied nitrogen | RE |
References
- Smil, V. Nitrogen in crop production: An account of global flows. Glob. Biogeochem. Cycles 1999, 13, 647–662. [Google Scholar] [CrossRef]
- UNEP. Reactive Nitrogen in the Environment, Too Much or Too Little of a Good Thing; The Woods Hole Research Center: Falmouth, MA, USA; UNEP DTIE Sustainable Consumption and Production Branch: Paris, France, 2007; Volume 51, ISBN 978 92 807 2783 8. [Google Scholar]
- Spiertz, J.H.J. Nitrogen, sustainable agriculture and food security: A review. In Sustainable Agriculture; Lighthouse, E., Navarrete, M., Debaeke, P., Véronique, S., Alberola, C., Eds.; Springer: Dordrecht, The Netherlands, 2009; Volume 30, pp. 635–651. [Google Scholar]
- Frink, C.R.; Waggoner, P.E.; Ausubel, J.H. Nitrogen fertilizer: Retrospect and prospect. Proc. Natl. Acad. Sci. USA 1999, 96, 1175–1180. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.H.; Liu, X.J.; Zhang, Y.; Shen, J.L.; Han, W.X.; Zhang, W.F. Significant acidification in major Chinese croplands. Science 2010, 327, 1008–1010. [Google Scholar] [CrossRef] [PubMed]
- Hoang, V.N.; Alauddin, M. Assessing the eco-environmental performance of agricultural production in OECD countries: The use of nitrogen flows and balance. Nutr. Cycl. Agroecosyst. 2010, 87, 353–368. [Google Scholar] [CrossRef]
- Ju, X.; Lu, X.; Gao, Z.; Chen, X.; Su, F.; Kogge, M. Processes and factors controlling N2O production in an intensively managed low carbon calcareous soil under sub-humid monsoon conditions. Environ. Pollut. 2011, 159, 1007–1016. [Google Scholar] [CrossRef]
- FAO. World Fertilizer Trends and Outlook to 2018; Food and Agriculture Organization of the United Nations-Rome: Rome, Italy, 2015; Available online: http://www.fao.org/3/a-i4324e.pdf (accessed on 20 July 2019).
- Tilman, D. Global environmental impacts of agricultural expansion: The need for sustainable and efficient practices. Proc. Natl. Acad. Sci. USA 1999, 96, 5995–6000. [Google Scholar] [CrossRef]
- Goulding, K.; Jarvis, S.; Whitmore, A. Optimizing nutrient management for farm systems. Philos. Trans. R. Soc. B 2008, 363, 667–680. [Google Scholar] [CrossRef]
- Zahoor, A.; Otten, A.; Wendisch, V.F. Metabolic engineering of Corynebacterium glutamicum for glycolate production. J. Biotechnol. 2014, 192, 366–375. [Google Scholar] [CrossRef]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef]
- Glass, A.D.M. Nitrogen use efficiency of crop plants: Physiological constraints upon nitrogen absorption. Crit. Rev. Plant Sci. 2003, 22, 453–470. [Google Scholar] [CrossRef]
- Hodge, A.; Robinson, D.; Fitter, A. Are microorganisms more effective than plants at competing for nitrogen? Trends Plant Sci. 2000, 5, 304–308. [Google Scholar] [CrossRef]
- Asghari, H.R.; Cavagnaro, T.R. Arbuscular mycorrhizas enhance plant interception of leached nutrients. Funct. Plant Biol. 2011, 38, 219–226. [Google Scholar] [CrossRef]
- Modolo, L.V.; Da-Silva, C.J.; Brandão, D.S.; Chaves, I.S. A mini review on what we have learned about urease inhibitors of agricultural interest since mid-2000s. J. Adv. Res. 2018, 13, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, M.K.; Tahir, M.M.; Rahim, N. Effect of N fertilizer source and timing on yield and N use efficiency of rainfed maize (Zea mays L.) in Kashmir-Pakistan. Geoderma 2013, 195, 87–93. [Google Scholar] [CrossRef]
- Conant, R.T.; Berdanier, A.B.; Grace, P.R. Patterns and trends in nitrogen use and nitrogen recovery efficiency in world agriculture. Glob. Biogeochem. Cycles 2013, 27, 558–566. [Google Scholar] [CrossRef]
- Mi, G.; Chen, F.; Zhang, F. Physiological and genetic mechanisms for nitrogen-use efficiency in maize. J. Crop Sci. Biotechnol. 2007, 10, 57–63. [Google Scholar]
- Hirel, B.; Tétu, T.; Lea, P.J.; Dubois, F. Improving nitrogen use efficiency in crops for sustainable agriculture. Sustainability 2011, 3, 1452–1485. [Google Scholar] [CrossRef]
- Sutton, M.A.; Oenema, O.; Erisman, J.W.; Leip, A.; Van Grinsven, H.; Winiwarter, W. Too much of a good thing. Nature 2011, 472, 159–161. [Google Scholar] [CrossRef]
- Kaur, A.; Bedi, S. Nitrogen use Efficiency and Source-sink Relations in Maize. J. Plant Sci. Res. 2012, 28, 219. [Google Scholar]
- Schröder, J.J.; Neeteson, J.J.; Withagen, J.C.M.; Noij, I.G.A.M. Effects of N application on agronomic and environmental parameters in silage maize production on sandy soils. Field Crop. Res. 1998, 58, 55–67. [Google Scholar] [CrossRef]
- Reganold, J.P.; Papendick, R.I.; Parr, F.F. Sustainable agriculture. Sci. Am. 1990, 262, 112–120. [Google Scholar] [CrossRef]
- Wichern, F.; Mayer, J.; Joergensen, R.G.; Müller, T. Release of C and N from roots of peas and oats and their availability to soil microorganisms. Soil Biol. Biochem. 2007, 39, 2829–2839. [Google Scholar] [CrossRef]
- Fustec, J.; Lesuffleur, F.; Mahieu, S.; Cliquet, J.B. Nitrogen rhizodeposition of legumes. A review. Agron. Sustain. Dev. 2010, 30, 57–66. [Google Scholar] [CrossRef]
- Bundy, L.G.; Kelling, K.A.; Good, L.W. Using Legumes as a Nitrogen Source; University of Wisconsin Extension, Cooperative Extension, Mifflin St.: Madison, WI, USA, 1992; Volume 3517, pp. 1–3. [Google Scholar]
- Coulter, J.A.; Sheaffer, C.C.; Wyse, D.L.; Haar, M.J.; Porter, P.M.; Quiring, S.R.; Klossner, L.D. Agronomic performance of cropping systems with contrasting crop rotations and external inputs. Agron. J. 2011, 103, 182–192. [Google Scholar] [CrossRef]
- Ranum, P.; Peña-Rosas, J.P.; Garcia-Casal, M.N. Global maize production, utilization, and consumption. Ann. N. Y. Acad. Sci. 2014, 1312, 105–112. [Google Scholar] [CrossRef]
- Dowswell, C. Maize in the Third World; CRC Press-Taylor and Francis Group: New York, NY, USA; Routledge: Abingdon, UK; New York, NY, USA, 2019; pp. 1–2. [Google Scholar]
- Chen, X.P.; Cui, Z.L.; Vitousek, P.M.; Cassman, K.G.; Matson, P.A.; Bai, J.S. Integrated soil–crop system management for food security. Proc. Natl. Acad. Sci. USA 2011, 108, 6399–6404. [Google Scholar] [CrossRef]
- Grassini, P.; Cassman, K.G. High-yield maize with large net energy yield and small global warming intensity. Proc. Natl. Acad. Sci. USA 2012, 109, 1074–1079. [Google Scholar] [CrossRef]
- OECD-FAO Agricultural Outlook 2018–2027; OECD Publishing: Paris, France; Food and Agriculture Organization of the United Nations: Rome, Italy, 2018. [CrossRef]
- Worku, M.; Bänziger, M.; Friesen, D.; Horst, W.J. Nitrogen uptake and utilization in contrasting nitrogen efficient tropical maize hybrids. Crop Sci. 2007, 47, 519–528. [Google Scholar] [CrossRef]
- Kiniry, J.R.; McCauley, G.; Xie, Y.; Arnold, J.G. Rice parameters describing crop performance of four USA cultivars. Agron. J. 2001, 93, 1354–1361. [Google Scholar] [CrossRef]
- Peng, Y.; Niu, J.; Peng, Z.; Zhang, F.; Li, C. Shoot growth potential drives N uptake in maize plants and correlates with root growth in the soil. Field Crop. Res. 2010, 115, 85–93. [Google Scholar] [CrossRef]
- Byers, D.L. Evolution in heterogeneous environments and the potential of maintenance of genetic variation in traits of adaptive significance. Genetica 2005, 123, 107. [Google Scholar] [CrossRef] [PubMed]
- Hirel, B.; Le Gouis, J.; Ney, B.; Gallais, A. The challenge of improving nitrogen use efficiency in crop plants: Towards a more central role for genetic variability and quantitative genetics within integrated approaches. J. Exp. Bot. 2007, 58, 2369–2387. [Google Scholar] [CrossRef] [PubMed]
- Masclaux-Daubresse, C.; Daniel-Vedele, F.; Dechorgnat, J.; Chardon, F.; Gaufichon, L.; Suzuki, A. Nitrogen uptake, assimilation and remobilization in plants: Challenges for sustainable and productive agriculture. Ann. Bot. 2010, 105, 1141–1157. [Google Scholar] [CrossRef] [PubMed]
- Hammad, H.M.; Farhad, W.; Abbas, F.; Fahad, S.; Saeed, S.; Nasim, W.; Bakhat, H.F. Maize plant nitrogen uptake dynamics at limited irrigation water and nitrogen. Environ. Sci. Pollut. Res. 2017, 24, 2549–2557. [Google Scholar] [CrossRef] [PubMed]
- Duvick, D.N. The contribution of breeding to yield advances in maize (Zea mays L.). Adv. Agron. 2005, 86, 83–145. [Google Scholar]
- Ciampitti, I.A.; Vyn, T.J. A comprehensive study of plant density consequences on nitrogen uptake dynamics of maize plants from vegetative to reproductive stages. Field Crop. Res. 2011, 121, 2–18. [Google Scholar] [CrossRef]
- Ciampitti, I.A.; Vyn, T.J. Physiological perspectives of changes over time in maize yield dependency on nitrogen uptake and associated nitrogen efficiencies: A review. Field Crop. Res. 2012, 133, 48–67. [Google Scholar] [CrossRef]
- Echarte, L.; Andrade, F.H. Harvest index stability of Argentinean maize hybrids released between 1965 and 1993. Field Crop. Res. 2003, 82, 1–12. [Google Scholar] [CrossRef]
- Sinclair, T.R. Historical changes in harvest index crop nitrogen accumulation. Crop. Sci. 1998, 38, 638–643. [Google Scholar] [CrossRef]
- Tollenaar, M.; Lee, E.A. Dissection of physiological processes underlying grain yield in maize by examining genetic improvement and heterosis. Maydica 2006, 51, 399. [Google Scholar]
- Vega, C.R.C.; Sadras, V.O.; Andrade, F.H.; Uhart, S.A. Reproductive allometry in soybean, maize and sunflower. Ann. Bot. 2000, 85, 461–468. [Google Scholar] [CrossRef]
- Echarte, L.; Luque, S.; Andrade, F.H.; Sadras, V.O.; Cirilo, A.; ME Otegui, M.E.; Vega, C.R.C. Response of maize kernel number to plant density in Argentinean hybrids released between 1965 and 1995. Field Crop. Res. 2000, 68, 1–8. [Google Scholar] [CrossRef]
- Tollenaar, M.; Dwyer, L.M.; Stewart, D.W.; Ma, B.L. Physiological parameters associated with differences in kernel set among maize hybrids. In Physiology and Modeling Kernel Set in Maize; Westgate, M.A., Boote, K.J., Eds.; CSSA Spec. CSSA/ASA/SSSA: Madison, WI, USA, 2000; Volume 51, pp. 115–130. [Google Scholar]
- Andrade, F.H.; Vega, C.; Uhart, S.; Cirilo, A.; Cantarero, M.; Valentinuz, O. Kernel number determination in maize. Crop Sci. 1999, 39, 453–459. [Google Scholar] [CrossRef]
- Vega, C.R.C.; Andrade, F.H.; Sadras, V.O.; Uhart, S.A.; Valentinuz, O.R. Seed number as a function of growth. A comparative study in soybean, sunflower and maize. Crop Sci. 2001, 41, 748–754. [Google Scholar] [CrossRef]
- Echarte, L.; Andrade, F.H.; Vega, C.R.C.; Tollenaar, M. Kernel number determination in Argentinean maize hybrids released between 1965 and 1993. Crop Sci. 2004, 44, 1654–1661. [Google Scholar] [CrossRef]
- Moll, R.H.; Kamprath, E.J.; Jackson, W.A. Analysis and interpretation of factors which contribute to efficiency of nitrogen utilization. Agron. J. 1982, 74, 562–564. [Google Scholar] [CrossRef]
- Good, A.G.; Shrawat, A.K.; Muench, D.G. Can less yield more? Is reducing nutrient input into the environment compatible with maintaining crop production? Trends Plant Sci. 2004, 9, 597–605. [Google Scholar] [CrossRef]
- Seyfried, M.S.; Rao, P.S.C. Kinetics of nitrogen mineralization in Costa Rican soils: Model evaluation and pretreatment effects. Plant Soil 1988, 106, 159–169. [Google Scholar] [CrossRef]
- Hirel, B.; Lea, P.J. Ammonium assimilation. In Plant Nitrogen; Springer: Belin/Heidelberg, Germany, 2001; pp. 79–99. [Google Scholar]
- Huggins, D.R.; Pan, W.L. Key indicators for assessing nitrogen use efficiency in cereal-based agro-ecosystems. J. Crop Prod. 2003, 8, 157–185. [Google Scholar] [CrossRef]
- Dobermann, A. Nitrogen use efficiency: State of the art. In IFA International Workshop on Enhanced-Efficiency Fertilizers, Frankfurt; International Fertilizer Industry Association: Paris, France, 2005; pp. 28–30. [Google Scholar]
- Yan, L.; Zhang, Z.D.; Zhang, J.J.; Gao, Q.; Feng, G.Z.; Abelrahman, A.M.; Chen, Y. Effects of improving nitrogen management on nitrogen utilization, nitrogen balance, and reactive nitrogen losses in a Mollisol with maize monoculture in Northeast China. Environ. Sci. Pollut. Res. 2016, 23, 4576–4584. [Google Scholar] [CrossRef]
- Wiesler, F.; Behrens, T.; Horst, W.J. The Role of N Efficient Cultivars in Sustainable Agriculture. Sci. World J. 2001, 1, 61–69. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Rajput, T.; Kumar, R.; Patel, N. Water and nitrate dynamics in baby corn (Zea mays L.). under different fertigation frequencies and operating pressures in semi-arid region of India. Agric. Water Manag. 2016, 163, 263–274. [Google Scholar] [CrossRef]
- Couto-Va´zquez, A.; Gonza´lez-Prieto, S.J. Fate of 15N-fertilizers in the soil-plant system of a forage rotation under conservation and plough tillage. Soil Tillage Res. 2016, 161, 10–18. [Google Scholar] [CrossRef]
- Trenkel, M.E. Slow and Controlled-Release and Stabilized Fertilizers: An Option for Enhancing Nutrient Use Efficiency in Agriculture; International Fertilizer Association: Paris, France, 2010. [Google Scholar]
- Schmidt, E.L. Nitrification in soil. In Nitrogen in Agricultural Soils; Stevenson, F.J., Ed.; ASA, CSSA, and SSSA: Madison, WI, USA, 1982; Volume 22, pp. 253–288. [Google Scholar]
- Kaboneka, S.; Sabbe, W.E.; Mauromoustakos, A. Carbon decomposition kinetics and nitrogen mineralization from corn, soybean, and wheat. Commun. Soil Sci. Plant Anal. 1997, 28, 1359–1373. [Google Scholar] [CrossRef]
- Kelly, J.T.; Bacon, R.K.; Wells, B.R. Genetic variability in nitrogen utilization at four growth stages in soft red winter wheat. J. Plant Nutr. 1995, 18, 969–982. [Google Scholar] [CrossRef]
- Hirel, B.; Bertin, P.; Quillere, I.; Bourdoncle, W.; Attagnant, C.; Dellay, C.; Gouy, S.; Retailliau, C.; Falque, M.; Gallais, A. Towards a better understanding of the genetic and physiological basis for nitrogen use efficiency in maize. Plant Physiol. 2001, 125, 1258–1270. [Google Scholar] [CrossRef]
- Lea, P.J.; Miflin, B.J. Glutamate synthase and the synthesis of glutamate in plants. Plant Physiol. Biochem. 2003, 41, 555–564. [Google Scholar] [CrossRef]
- Fuentes, S.I.; Allen, D.J.; Ortiz-Lopez, A.; Hernández, G. Over-expression of cytosolic glutamine synthetase increases photosynthesis and growth at low nitrogen concentrations. J. Exp. Bot. 2001, 52, 1071–1081. [Google Scholar] [CrossRef]
- Kichey, T.; Heumez, E.; Pocholle, P.; Pageau, K.; Vanacker, H.; Dubois, F.; Le Gouis, J.; Hirel, B. Combined agronomic and physiological aspects of nitrogen management in wheat (Triticum aestivum L). Dynamic and integrated views highlighting the central role for the enzyme glutamine synthetase. New Phytol. 2006, 169, 265–278. [Google Scholar] [CrossRef]
- Forde, B.G.; Lea, P.J. Glutamate in plants: Metabolism, regulation and signaling. J. Exp. Biol. 2007, 58, 2339–2358. [Google Scholar] [CrossRef]
- Cren, M.; Hirel, B. Glutamine synthetase in higher plants: Regulation of gene and protein expression from the organ to the cell. Plant Cell Physiol. 1999, 40, 1187–1193. [Google Scholar] [CrossRef]
- Cirilo, A.G.; Dardanelli, J.; Balzarini, M.; Andrade, F.H.; Cantarero, M.; Luque, S.; Pedrol, H.M. Morpho-physiological traits associated with maize crop adaptations to environments differing in nitrogen availability. Field Crop. Res. 2009, 113, 116–124. [Google Scholar] [CrossRef]
- Borrell, A.; Hammer, G. Nitrogen dynamics and the physiological basis of stay-green in sorghum. Crop Sci. 2000, 40, 1295–1307. [Google Scholar] [CrossRef]
- Maddonni, G.A.; Otegui, M.E.; Cirilo, A.G. Plant population density, row spacing and hybrid effects on maize canopy architecture and light attenuation. Field Crop. Res. 2001, 71, 183–193. [Google Scholar] [CrossRef]
- Gastal, F.; Lemaire, G. Nitrogen uptake and distribution in crops: An agronomical and ecophysiological perspective. J. Exp. Bot. 2002, 53, 789–799. [Google Scholar] [CrossRef]
- Ma, B.L.; Dwyer, L.M. Nitrogen uptake and use in two contrasting maize hybrids differing in leaf senescence. Plant Soil 1998, 199, 283–291. [Google Scholar] [CrossRef]
- Hikosaka, K. Interspecific difference in the photosynthesis–nitrogen relationship: Patterns, physiological causes, and ecological importance. J. Plant Res. 2004, 117, 481–494. [Google Scholar] [CrossRef]
- Andrade, F.H.; Echarte, L.; Rizzalli, R.; Della, M.A.; Casanovas, M. Kernel number prediction in maize under nitrogen or water stress. Crop Sci. 2002, 42, 1173–1179. [Google Scholar] [CrossRef]
- Spano, G.; Di Fonzo, N.; Perrota, C.; Platani, C.; Ronga, G.; Lawlor, Q.W.; Napier, J.A.; Shewry, P.R. Physiological characterization of ‘stay green’ mutants in durum wheat. J. Exp. Bot. 2003, 54, 1415–1420. [Google Scholar] [CrossRef]
- Racjan, I.; Tollenaar, M. Source-sink ratio and leaf senescence in maize I. Dry matter accumulation and partitioning during grain filling. Field Crop. Res. 1999, 60, 245–253. [Google Scholar]
- Racjan, I.; Tollenaar, M. Source-sink ratio and leaf senescence in maize II. Nitrogen metabolism during grain filling. Field Crop. Res. 1999, 60, 255–265. [Google Scholar]
- Coque, M.; Gallais, A. Genetic variation for N-remobilization and post-silking N-uptake in a set of maize recombinant inbred lines I: Evaluation by 15N labeling, heritabilities and correlations among traits for test cross performance. Crop Sci. 2007, 47, 1787–1796. [Google Scholar] [CrossRef]
- Borras, L.; Slafer, G.A.; Otegui, M.E. Seed dry weight response to source-sink manipulations in wheat, maize, and soybean: A quantitative reappraisal. Field Crop. Res. 2004, 86, 131–146. [Google Scholar] [CrossRef]
- Pang, X.P.; Letey, J. Organic farming challenge of timing nitrogen availability to crop nitrogen requirements. Soil Sci. Soc. Am. J. 2000, 64, 247–253. [Google Scholar] [CrossRef]
- Purcino, A.A.C.; Arellano, C.; Athwal, G.S.; Huber, S.C. Nitrate effect on carbon and nitrogen assimilating enzymes of maize hybrids representing seven eras of maize breeding. Maydica 1998, 43, 83–94. [Google Scholar]
- Halvorson, A.D.; Nielsen, D.C.; Reule, C.A. Nitrogen fertilization and rotation effects on no-till dry land wheat production. Agron. J. 2004, 96, 1196–1201. [Google Scholar] [CrossRef]
- Peoples, M.B.; Freney, J.R.; Mosier, A.R. Minimizing gaseous losses of nitrogen. In Nitrogen Fertilization in the Environment; Bacon, P.E., Ed.; Marcel Dekker Inc.: New York, NY, USA, 1995; pp. 565–602. [Google Scholar]
- Rizzi, E.; Balconi, C.; Bosio, D.; Nembrini, L.; Morselli, A.; Motto, M. Accumulation and partitioning of nitrogen among plant parts in the high and low protein strains of maize. Maydica 1996, 41, 325–332. [Google Scholar]
- Subedi, K.D.; Ma, B.L. Nitrogen uptake and partitioning in stay-green and leafy maize hybrids. Crop Sci. 2005, 45, 740–747. [Google Scholar] [CrossRef]
- Sattelmacher, B.; Horst, W.J.; Becker, H.C. Factors that contribute to genetic variation for nutrient efficiency of crop plants. Zeitschrift für Pflanzenernährung und Bodenkunde 1994, 157, 215–224. [Google Scholar] [CrossRef]
- Akintoye, H.A.; Kling, J.; Gand, L.E.O. N-use efficiency of single, double and synthetic maize lines grown at four N levels in three ecological zones of West Africa. Field Crop. Res. 1999, 60, 189–199. [Google Scholar] [CrossRef]
- Gallais, A.; Coque, M.; Quilléré, I.; Le Gouis, J.; Prioul, J.L.; Hirel, B. Estimating proportions of N-remobilization and of post-silking N-uptake allocated to maize kernels by 15N labeling. Crop Sci. 2007, 47, 685–691. [Google Scholar] [CrossRef]
- Tolley-Henry, L.; Raper, C.D., Jr. Soluble carbohydrate allocation to roots, photosynthetic rate of leaves, and nitrate assimilation as affected by nitrogen stress and irradiance. Bot. Gaz. 1991, 152, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Pons, T.L.; van der Werf, A.; Lambers, H. Photosynthetic nitrogen use efficiency of inherently low- and fast-growing species: Possible explanations for observed differences. In A Whole Plant Perspective on Carbon-Nitrogen Interactions; Roy, J., Garnier, E., Eds.; SPB Academic Publishing: The Hague, The Netherlands, 1994; pp. 61–77. [Google Scholar]
- Evans, J.R. Photosynthesis and nitrogen relationships in leaves of C3 plants. Oecologia 1989, 78, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Gallais, A.; Coque, M.; Quilléré, I.; Prioul, J.L.; Hirel, B. Modelling post-silking N-fluxes in maize using 15N-labeling-field experiments. New Phytol. 2006, 172, 696–707. [Google Scholar] [CrossRef] [PubMed]
- Paponov, I.A.; Engels, C. Effect of nitrogen supply on leaf traits related to photosynthesis during grain filling in two maize genotypes with different N efficiency. J. Plant Nutr. Soil Sci. 2003, 166, 756–763. [Google Scholar] [CrossRef]
- Pearson, C.J.; Jacobs, B.C. Yield components and nitrogen partitioning of maize in response to nitrogen before and after anthesis. Aust. J. Agric. Res. 1987, 38, 1001–1009. [Google Scholar] [CrossRef]
- McKendry, A.L.; McVetty, P.B.E.; Evans, L.E. Selection criteria for combining high grain yield and high grain protein concentration in bread wheat. Crop Sci. 1995, 35, 1597–1602. [Google Scholar] [CrossRef]
- Mistele, B.; Schmidhalter, U. Estimating the nitrogen nutrition index using spectral canopy reflectance measurements. Eur. J. Agron. 2008, 29, 184–190. [Google Scholar] [CrossRef]
- Grindlay, D.J.C.; Sylvester-Bradley, R.; Scott, R.K. The relationship between canopy green area and nitrogen in the shoot. In Diagnostic Procedures for Crop N Management; Lemaire, G., Burns, I.G., Eds.; INRA Editions: Paris, France, 1995; pp. 53–60. [Google Scholar]
- Lemaire, G.; Gastal, F.; Plenet, D. Dynamics of N uptake and N distribution in plant canopies. Use of crop N status index in crop modeling. In Diagnostic Procedures for Crop N Management; Lemaire, G., Bruns, I.G., Eds.; INRA Editions: Paris, France, 1995; pp. 15–29. [Google Scholar]
- Eichelmann, H.; Oja, V.; Rasulov, B.; Padu, E.; Bichele, I.; Pettai, P.; Mänd, P.; Kull, O.; Laisk, A. Adjustment of leaf photosynthesis to shade in a natural canopy: Reallocation of nitrogen. Plant Cell Environ. 2005, 28, 389–401. [Google Scholar] [CrossRef]
- Schmidhalter, U. Development of a quick on-farm test to determine nitrate levels in soil. J. Plant Nutr. Soil Sci. 2005, 168, 432–438. [Google Scholar] [CrossRef]
- Houlès, V.; Guérif, M.; Mary, B. Elaboration of a nitrogen nutrition indicator for winter wheat based on leaf area index and chlorophyll content for making nitrogen recommendations. Eur. J. Agron. 2007, 27, 1–11. [Google Scholar] [CrossRef]
- Baret, F.; Houlès, V.; Guérif, M. Quantification of plant stress using remote sensing observations and crop models: The case of nitrogen management. J. Exp. Bot. 2007, 58, 869–880. [Google Scholar] [CrossRef] [PubMed]
- Schlemmer, M.; Gitelson, A.; Schepers, J.; Ferguson, R.; Peng, Y.; Shanahan, J.; Rundquist, D. Remote estimation of nitrogen and chlorophyll contents in maize at leaf and canopy levels. Int. J. Appl. Earth Obs. Geoinf. 2013, 25, 47–54. [Google Scholar] [CrossRef]
- Sripada, R.P.; Schmidt, J.P.; Dellinger, A.E.; Beegle, D.B. Evaluating multiple indices from a canopy reflectance sensor to estimate corn N requirements. Agron. J. 2008, 100, 1553–1561. [Google Scholar] [CrossRef]
- Gitelson, A.A. Remote Sensing estimation of crop biophysical characteristics at various scales. In Chapter 15 in Hyperspectral Remote Sensing of Vegetation; Thenkabail, P.S., Lyon, J.G., Huete, A., Eds.; CRC Press-Taylor and Francis Group: Boca Raton, FL, USA; London, UK; New York, NY, USA, 2011; pp. 329–358. [Google Scholar]
- Walters, D.T. Diagnosis of nitrogen deficiency in maize and the influence of hybrid and plant density. In Proceedings of the North Central Extension-Industry Soil Fertility Conference, Des Moines, IA, USA, 19–20 November 2003; Volume 19. [Google Scholar]
- Zebarth, B.J.; Younie, M.; Paul, J.W.; Bittman, S. Evaluation of leaf chlorophyll index for making fertilizer nitrogen recommendations for silage corn in a high fertility environment. Commun. Soil Sci. Plant Anal. 2002, 33, 665–684. [Google Scholar] [CrossRef]
- Argenta, G.; Ferreira da Silva, P.R.; Sangoi, L. Leaf relative chlorophyll content as an indicator parameter to predict nitrogen fertilization in maize. Ciênc Rural 2004, 34, 1379–1387. [Google Scholar] [CrossRef]
- Rashid, M.T.; Voroney, P.; Parkin, G. Predicting nitrogen fertilizer requirements for corn by chlorophyll meter under different N availability conditions. Can. J. Soil Sci. 2005, 85, 149–159. [Google Scholar] [CrossRef]
- Link, A.; Jasper, J. Site-specific N fertilization based on remote sensing: Is it necessary to take yield variability into account? In Proceedings of the 4th European Conference on Precision Agriculture, Berlin, Germany, 15–19 June 2003; Stafford, J., Werner, A., Eds.; Academic Publishers: Wageningen, The Netherlands, 2003; pp. 353–359. [Google Scholar]
- Soon, Y.K.; Clayton, G.W. Eight years of crop rotation and tillage effects on crop production and N fertilizer use. Can. J. Soil Sci. 2002, 82, 165–172. [Google Scholar] [CrossRef]
- Silla, F.; Escudero, A. Nitrogen-use efficiency: Trade-offs between N productivity and mean residence time at organ, plant and population levels. Funct. Ecol. 2004, 18, 511–521. [Google Scholar] [CrossRef]
- Echarte, L.; Rothstein, S.; Tollenaar, M. The response of leaf photosynthesis and dry matter accumulation to nitrogen supply in an older and a newer maize hybrid. Crop Sci. 2008, 48, 656–665. [Google Scholar] [CrossRef]
- Masclaux, C.; Quilleré, I.; Gallais, A.; Hirel, B. The challenge of remobilization in plant nitrogen economy. A survey of physio-agronomic and molecular approaches. Ann. Appl. Biol. 2001, 138, 69–81. [Google Scholar] [CrossRef]
- Habtegebrial, K.; Singh, B.R.; Haile, M. Impact of tillage and nitrogen fertilization on yield, nitrogen use efficiency of tef (Eragrostis tef (Zucc.) Trotter) and soil properties. Soil Tillage Res. 2007, 94, 55–63. [Google Scholar] [CrossRef]
- Ogola, J.B.O.; Wheeler, T.R.; Harris, P.M. Effects of nitrogen and irrigation on water use of maize crops. Field Crop. Res. 2002, 78, 105–117. [Google Scholar] [CrossRef]
- Anatoliy, G.K.; Thelen, K.D. Effect of winter wheat crop residue on no-till corn growth and development. Agron. J. 2007, 99, 549–555. [Google Scholar]
- Hooda, P.S.; Edwards, A.C.; Anderson, H.A.; Miller, A. A review of water quality concerns in livestock farming areas. Sci. Total Environ. 2000, 250, 143–167. [Google Scholar] [CrossRef]
- Robertson, G.P.; Vitousek, P.M. Nitrogen in agriculture: Balancing the cost of an essential resource. Ann. Rev. Environ. Resour. 2009, 34, 97–125. [Google Scholar] [CrossRef]
- Lucas, F.T.; Borges, B.M.M.N.; Coutinho, E.L.M. Nitrogen fertilizer management for maize production under tropical climate. Agron. J. 2019, 111, 2031–2037. [Google Scholar] [CrossRef]
- Nyamangara, J.; Piha, M.I.; Giller, K.E. Effects of combined cattle manure and mineral nitrogen on maize N uptake and grain yield. Afr. Crop Sci. J. 2003, 11, 289–300. [Google Scholar] [CrossRef][Green Version]
- Rimski-Korsakov, H.; Rubio, G.; Lavado, R.S. Fate of the nitrogen from fertilizers in field-grown maize. Nutr. Cycl. Agroecosyst. 2012, 93, 253–263. [Google Scholar] [CrossRef]
- Huang, B.; Sun, W.; Zhao, Y.; Zhu, J.; Yang, R.; Zou, Z.; Su, J. Temporal and spatial variability of soil organic matter and total nitrogen in an agricultural ecosystem as affected by farming practices. Geoderma 2007, 139, 336–345. [Google Scholar] [CrossRef]
- Khan, Z.; Shah, P.; Arif, M. Management of organic farming: Effectiveness of farm yard manure (FYM) and nitrogen for maize productivity. Sarhad J. Agric. 2000, 16, 461–465. [Google Scholar]
- Gangwar, K.S.; Singh, K.K.; Sharma, S.K.; Tomar, O.K. Alternative tillage and crop residue management in wheat after rice in sandy loam soils of Indo-Gengetic plains. Soil Tillage Res. 2006, 88, 242–252. [Google Scholar] [CrossRef]
- Ayeni, L.S.; Adetunji, M.T.; Ojeniyi, S.O.; Ewulo, B.S.; Adeyemo, A.J. Comparative and cumulative effect of cocoa pod husk ash and poultry manure on soil and maize nutrient contents and yield. Am.–Euras. J. Sustain. Agric. 2008, 2, 92–97. [Google Scholar]
- Biau, A.; Santiveri, F.; Mijangos, I.; Lloveras, J. The impact of organic and mineral fertilizers on soil quality parameters and the productivity of irrigated maize crops in semiarid regions. Eur. J. Soil Biol. 2012, 53, 56–61. [Google Scholar] [CrossRef]
- Belay, A.; Claassens, A.; Wehner, F.C. Effect of direct nitrogen and potassium and residual phosphorus fertilizers on soil chemical properties, microbial components and maize yield under long-term crop rotation. Biol. Fertil. Soils 2002, 35, 420–427. [Google Scholar]
- Ayeni, L.S.; Adetunji, M.T. Integrated application of poultry manure and mineral fertilizer on soil chemical properties, nutrient uptake, yield and growth components of maize. Nat. Sci. 2010, 8, 60–67. [Google Scholar]
- Ali, K.; Munsif, F.; Zubair, M.; Hussain, Z.; Shahid, M.; Din, I.U.; Khan, N. Management of organic and inorganic nitrogen for different maize varieties. Sarhad J. Agric. 2011, 27, 525–529. [Google Scholar]
- Rafiq, M.A.; Ali, A.; Malik, M.A.; Hussain, M. Effect of fertilizer levels and plant densities on yield and protein contents of autumn planted maize. Pak. J. Agric. Sci. 2010, 47, 201–208. [Google Scholar]
- Asif, M.; Saleem, M.F.; Anjum, S.A.; Wahid, M.A.; Bilal, M.F. Effect of nitrogen and zinc sulphate on growth and yield of maize (Zea mays L.). J. Agric. Res. 2013, 51, 03681157. [Google Scholar]
- Pessarakli, M. (Ed.) Handbook of Plant and Crop Physiology; Eastern Hemisphere Distribution, Marcel Dekker Inc.: New York, NY, USA; Basel, Switzerland, 2002; Volume 940. [Google Scholar]
- Potarzycki, J. Effect of magnesium or zinc supplementation at the background of nitrogen rate on nitrogen management by maize canopy cultivated in monoculture. Plant Soil Environ. 2011, 57, 19–25. [Google Scholar] [CrossRef]
- Qu, C.; Liu, C.; Ze, Y.; Gong, X.; Hong, M.; Wang, L.; Hong, F. Inhibition of nitrogen and photosynthetic carbon assimilation of maize seedlings by exposure to a combination of salt stress and potassium-deficient stress. Biol. Trace Elem. Res. 2011, 144, 1159–1174. [Google Scholar] [CrossRef] [PubMed]
- Mulvaney, R.L.; Khan, S.A.; Ellsworth, T.R. Synthetic nitrogen fertilizers deplete soil nitrogen: A global dilemma for sustainable cereal production. J. Environ. Qual. 2009, 38, 2295–2314. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.A.; Mulvaney, R.L.; Ellsworth, T.R.; Boast, C.W. The myth of nitrogen fertilization for soil carbon sequestration. J. Environ. Qual. 2007, 36, 1821–1832. [Google Scholar] [CrossRef] [PubMed]
- Mapanda, F.; Wuta, M.; Nyamangara, J.; Rees, R.M. Effects of organic and mineral fertilizer nitrogen on greenhouse gas emissions and plant-captured carbon under maize cropping in Zimbabwe. Plant Soil 2011, 343, 67–81. [Google Scholar] [CrossRef]
- Yang, J.Y.; Huffman, E.C.; Jong, R.D.; Kirkwood, V.; MacDonald, K.B.; Drury, C.F. Residual soil nitrogen in soil landscapes of Canada as affected by land use practices and agricultural policy scenarios. Land Use Policy 2007, 24, 89–99. [Google Scholar] [CrossRef]
- Khan, H.Z.; Malik, M.A.; Saleem, M.F. Effect of rate and source of organic material on the production potential of spring maize (Zea mays L.). Pak. J. Agric. Sci. 2008, 45, 40–43. [Google Scholar]
- Grant, C. Policy aspects related to the use of enhanced-efficiency fertilizers: View point of the scientific community. In IFA International Workshop on Enhanced-Efficiency Fertilizers, Frankfurt; International Fertilizer Industry Association: Paris, France, 2005; pp. 28–30. [Google Scholar]
- Stevens, W.B.; Hoeft, R.G.; Mulvaney, R.L. Fate of Nitrogen15 in a long-term nitrogen rate study: II. Nitrogen uptake efficiency. Agron. J. 2005, 97, 1046–1053. [Google Scholar] [CrossRef]
- Robertson, G.P.; Groffman, P.M. Nitrogen transformations. In Soil Microbiology, Ecology and Biochemistry; Academic Press: New York, NY, USA, 2007; pp. 341–364. [Google Scholar]
- Provin, T.; Hossner, L.R. What Happens to Nitrogen in Soils? In Texas A&M AgriLife Extension; The Texas A&M University Systems: College Station, TX, USA, 2001. [Google Scholar]
- Jackson, L.E.; Burger, M.; Cavagnaro, T.R. Roots, nitrogen transformations, and ecosystem services. Annu. Rev. Plant Biol. 2008, 59, 341–363. [Google Scholar] [CrossRef]
- Griffin, T.S. Nitrogen availability. In Nitrogen in Agricultural Soils; Schepers, J.S., Raun, W.R., Eds.; ASA, CSSA, and SSSA: Madison, WI, USA, 2008; Volume 49, pp. 616–646. [Google Scholar]
- Ma, B.L.; Dwyer, L.M.; Gregorich, E.G. Soil nitrogen amendment effects on seasonal nitrogen mineralization and nitrogen cycling in maize production. Agron. J. 1999, 91, 1003–1009. [Google Scholar] [CrossRef]
- Schröder, J.J.; Neeteson, J.J.; Oenema, O.; Struik, P.C. Does the crop or the soil indicate how to save nitrogen in maize production? Reviewing the state of the art. Field Crop. Res. 2000, 66, 151–164. [Google Scholar] [CrossRef]
- Rusinamhodzi, L.; Corbeels, M.; Zingore, S.; Nyamangara, J.; Giller, K.E. Pushing the envelope? Maize production intensification and the role of cattle manure in recovery of degraded soils in smallholder farming areas of Zimbabwe. Field Crop. Res. 2013, 147, 40–53. [Google Scholar] [CrossRef]
- Dunjana, N.; Nyamugafata, P.; Shumba, A.; Nyamangara, J.; Zingore, S. Effects of cattle manure on selected soil physical properties of smallholder farms on two soils of Murewa, Zimbabwe. Soil Use Manag. 2012, 28, 221–228. [Google Scholar] [CrossRef]
- Negassa, W.; Gebrekidan, H.; Friesen, D.K. Integrated use of farmyard manure and NP fertilizers for maize on farmers’ fields. J. Agric. Rural Dev. Trop. Subtrop. 2005, 106, 131–141. [Google Scholar]
- Bengough, A.G.; Bransby, M.F.; Hans, J.; McKenna, S.J.; Roberts, T.J.; Valentine, T.A. Root response to soil physical conditions: Growth dynamics from field to cell. J. Exp. Bot. 2006, 57, 437–447. [Google Scholar] [CrossRef]
- Li, Y.; Mi, G.H.; Chen, F.; Lao, X.; Zhang, F. Genotypic difference of nitrogen recycling between root and shoot of maize seedlings. Acta Phytophysiol. Sin. 2001, 27, 226–230. [Google Scholar]
- Wang, Y.; Mi, G.H.; Chen, F.J.; Zhang, J.H.; Zhang, F.S. Response of root morphology to nitrate supply and its contribution to nitrogen accumulation in maize. J. Plant Nutr. 2004, 27, 2189–2202. [Google Scholar] [CrossRef]
- Marschner, H.; Kirkby, E.A.; Cakmak, I. Effect of mineral nutritional status on shoot-root partitioning of photoassimilates and cycling of mineral nutrients. J. Exp. Bot. 1996, 47, 1255–1263. [Google Scholar] [CrossRef]
- Chun, L.; Chen, F.; Zhang, F.; Mi, G.H. Root growth, nitrogen uptake and yield formation of hybrid maize with different N efficiency. Plant Nutr. Fert. Sci. 2005, 11, 615–619. [Google Scholar]
- Tian, Q.Y.; Chen, F.J.; Zhang, F.S.; Mi, G.H. Genotypic difference in nitrogen acquisition ability in maize plants is related to the coordination of leaf and root growth. J. Plant Nutr. 2006, 29, 317–330. [Google Scholar] [CrossRef]
- Niu, J.; Chen, F.J.; Mi, G.H.; Li, C.J.; Zhang, F.S. Transpiration, and nitrogen uptake and flow in two maize (Zea mays L.). inbred lines as affected by nitrogen supply. Ann. Bot. 2007, 99, 153–160. [Google Scholar] [CrossRef]
- Mackay, A.D.; Barber, S.A. Effects of nitrogen on root growth of two corn genotypes in the field. Agron. J. 1986, 78, 699–703. [Google Scholar] [CrossRef]
- Mkhabela, M.S.; Mkhabela, M.S.; Pali-Shikhulu, J. Response of maize (Zea mays L.) cultivars to different levels of nitrogen application in Swaziland. In Proceedings of the Seventh Eastern and Southern Africa Regional Maize Conference, Nairobi, Kenya, 5–11 February 2001; Volume 11, pp. 377–381. [Google Scholar]
- Mi, G.H.; Liu, J.A.; Chen, F.J.; Zhang, F.S.; Cui, Z.L.; Liu, X.S. Nitrogen uptake and remobilization in maize hybrids differing in leaf senescence. J. Plant Nutr. 2003, 26, 237–247. [Google Scholar] [CrossRef]
- Horst, W.J.; Behrens, T.; Heuberger, H.; Kamh, M.; Reidenbach, G.; Wiesler, F. Genotypic differences in nitrogen use efficiency in crop plants. In Innovative Soil-Plant Systems for Sustainable Agricultural Production; Lynch, J.M., Schepers, J.S., Unver, I., Eds.; Organisation for Economic Co-operation and Development: Paris, France, 2003; pp. 75–92. [Google Scholar]
- Miflin, B.J.; Habash, D.Z. The role of glutamine synthetase and glutamate dehydrogenase in nitrogen assimilation and possibilities for improvement in the nitrogen utilization of crops. J. Exp. Bot. 2002, 53, 979–987. [Google Scholar] [CrossRef] [PubMed]
- Pan, W.L.; Jackson, W.A.; Moll, R.H. Nitrate uptake and partitioning by corn (Zea mays L.). root systems and associated morphological differences among genotypes and stages of root development. J. Exp. Bot. 1985, 36, 1341–1351. [Google Scholar] [CrossRef]
- Peterson, C.A.; Emanuel, M.E.; Humphreys, G.B. Pathway of movement of apoplastic fluorescent dye tracers through the endodermis at the site of secondary root formation in corn (Zea mays L.) and broad bean (Vicia faba). Can. J. Bot. 1981, 59, 618–625. [Google Scholar] [CrossRef]
- Oaks, A.; Aslam, M.; Boesel, I. Ammonium and amino acids as regulators of nitrate reductase in corn roots. Plant Physiol. 1977, 59, 391–394. [Google Scholar] [CrossRef]
- Lewis, C.E.; Noctor, G.; Causton, D.; Foyer, C. Regulation of assimilate partitioning in leaves. Aust. J. Plant Physiol. 2000, 27, 507–519. [Google Scholar] [CrossRef]
- Ortiz-Monasterio, J.I.; Sayre, K.D.; Rajaram, S.; McMahon, M. Genetic progress in wheat yield and nitrogen use efficiency under four nitrogen regimes. Crop Sci. 1997, 37, 898–904. [Google Scholar] [CrossRef]
- Wivstad, M.; Dahlin, A.S.; Grant, C. Perspectives on nutrient management in arable farming systems. Soil Use Manag. 2005, 21, 113–121. [Google Scholar] [CrossRef]
- Djaman, K.; Irmak, S.; Martin, D.L.; Ferguson, R.B.; Bernards, M.L. Plant nutrient uptake and soil nutrient dynamics under full and limited irrigation and rainfed maize production. Agron. J. 2013, 105, 527–538. [Google Scholar] [CrossRef]
- Qiu, S.J.; He, P.; Zhao, S.C.; Li, W.J.; Xie, J.G.; Hou, Y.P.; Jin, J.Y. Impact of nitrogen rate on maize yield and nitrogen use efficiencies in northeast China. Agron. J. 2015, 107, 305–313. [Google Scholar] [CrossRef]
- Sinclair, T.R.; Vadez, V. Physiological traits for crop yield improvement in low N and P environments. Plant Soil 2002, 245, 1–15. [Google Scholar] [CrossRef]
- Cañas, R.A.; Amiour, N.; Quilleré, I.; Hirel, B. An integrated statistical analysis of the genetic variability of nitrogen metabolism in the ear of three maize inbred lines (Zea mays L.). J. Exp. Bot. 2010, 62, 2309–2318. [Google Scholar] [CrossRef]
- Stark, C.H.; Richards, K.G. The continuing challenge of agricultural nitrogen loss to the environment in the context of global change and advancing research. Dyn. Soil Dyn. Plant 2008, 2, 1–12. [Google Scholar]
- Delgado, J.A.; Shaffer, M.; Hu, C.; Lavado, R.; Cueto-Wong, J.; Joosse, P.; Sotomayer, D.; Colon, W.; Follett, R.; DelGrosso, S.; et al. An index approach to assess nitrogen losses to the environment. Ecol. Eng. 2008, 32, 108–120. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).