Salt Stress Effects on Avocado (Persea americana Mill.) Plants with and without Seaweed Extract (Ascophyllum nodosum) Application
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Environmental Conditions
2.2. Experimental Design and Treatments
2.3. Measurements on Plant Growth
2.4. Measurements on Nutrient Composition
2.5. Data Analysis
3. Results
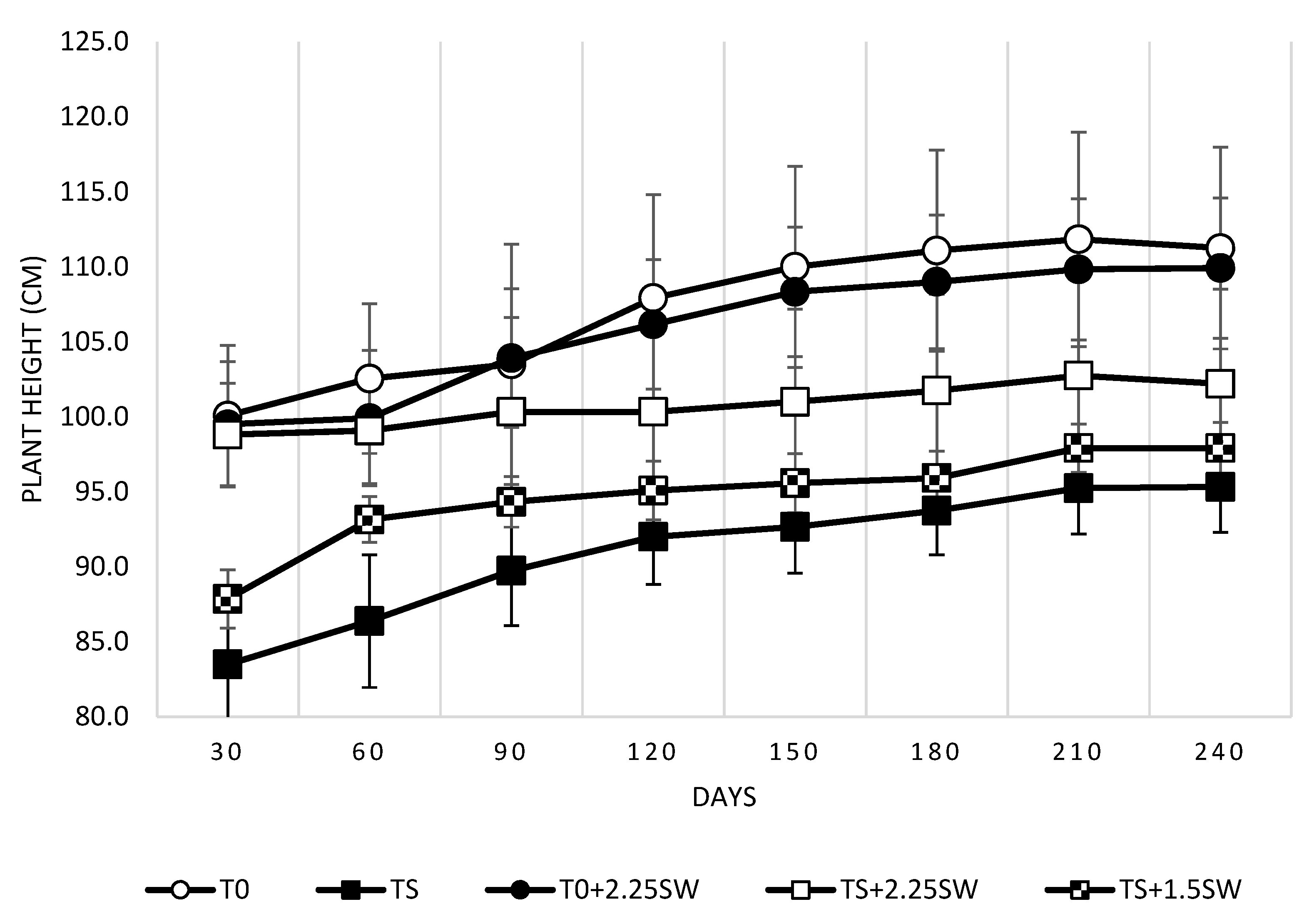
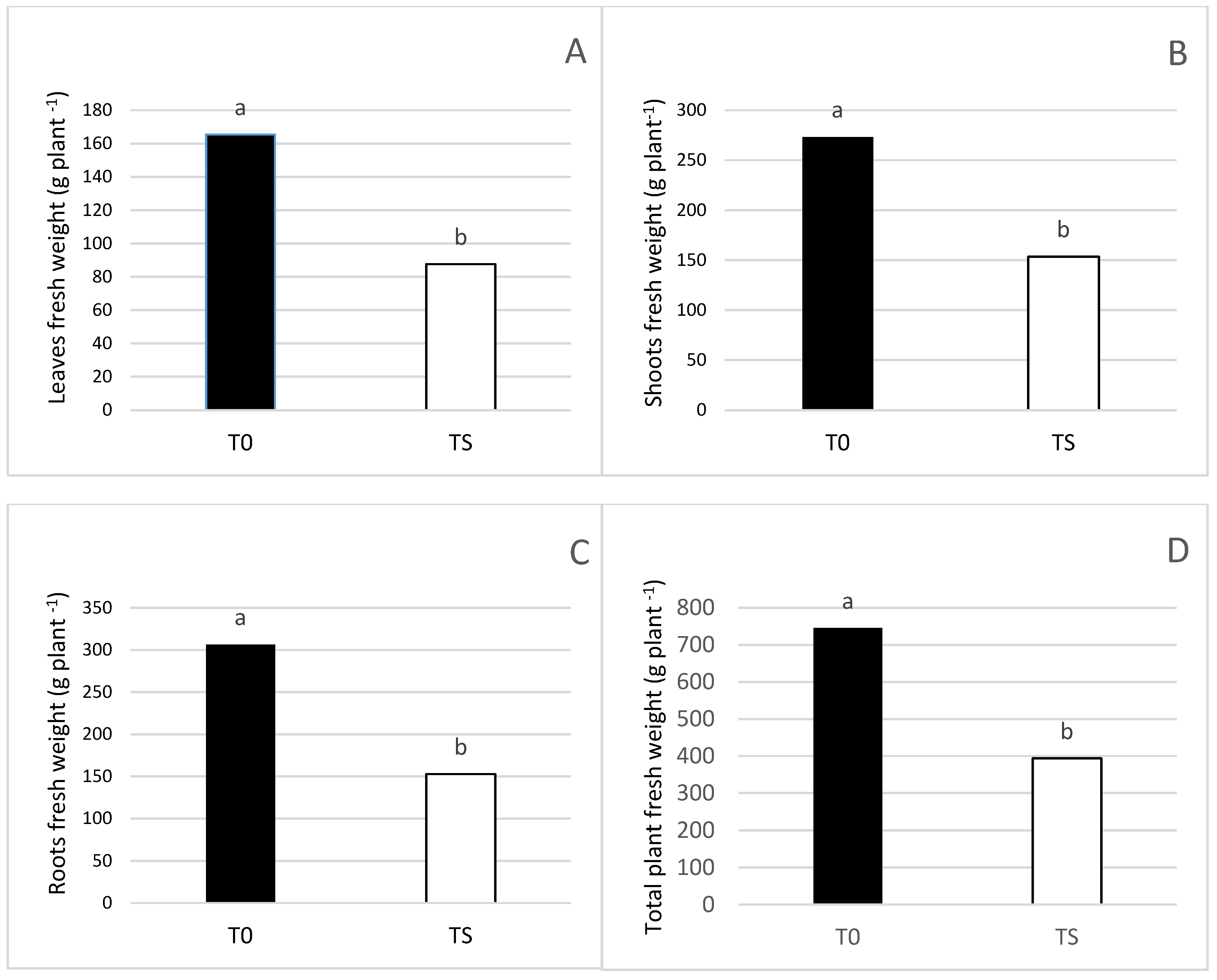
3.1. Effect of Salt Irrigation and Seaweed Extract (SW) on Growth Parameters and Leaf Necrosis on Avocado Plants
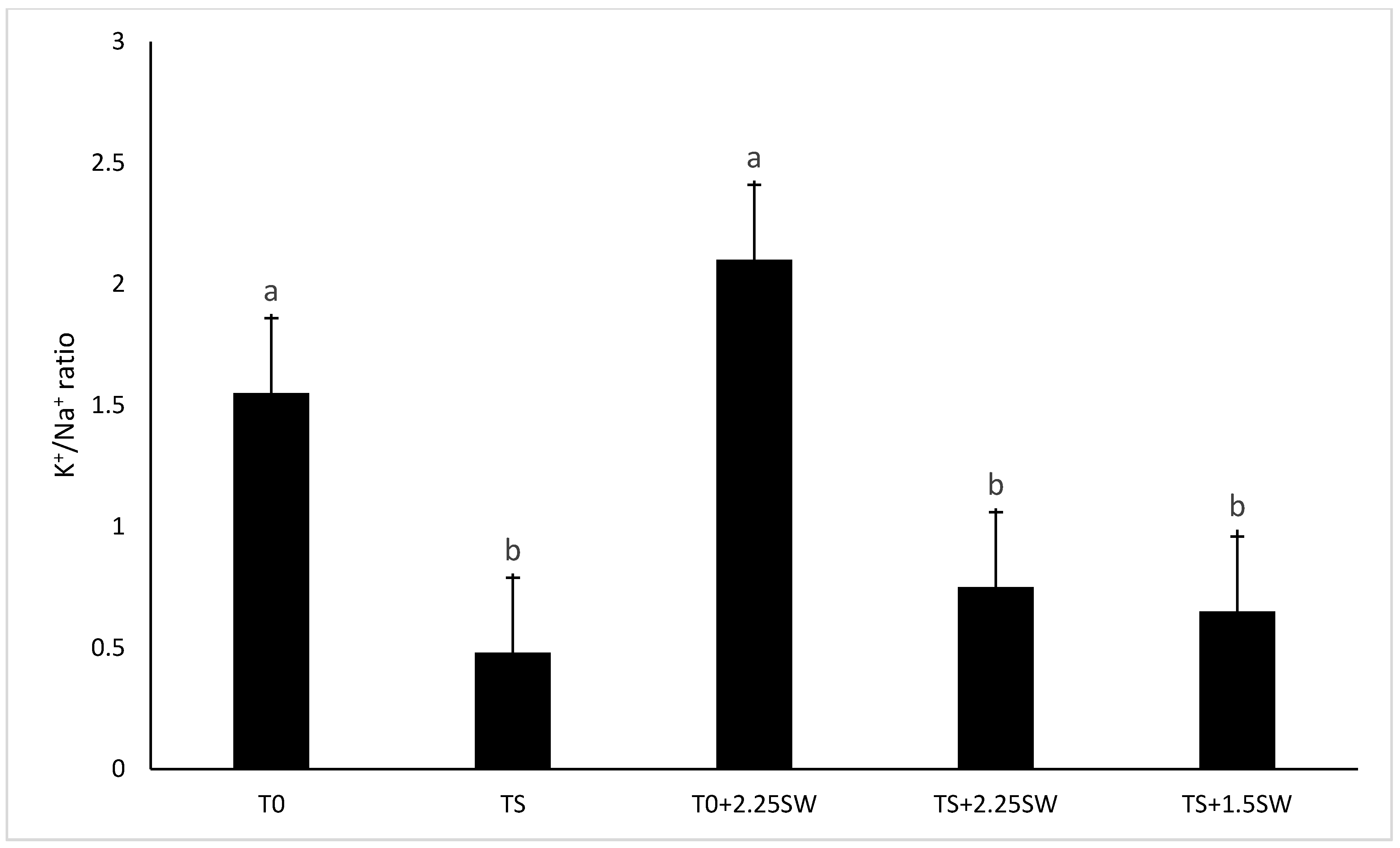
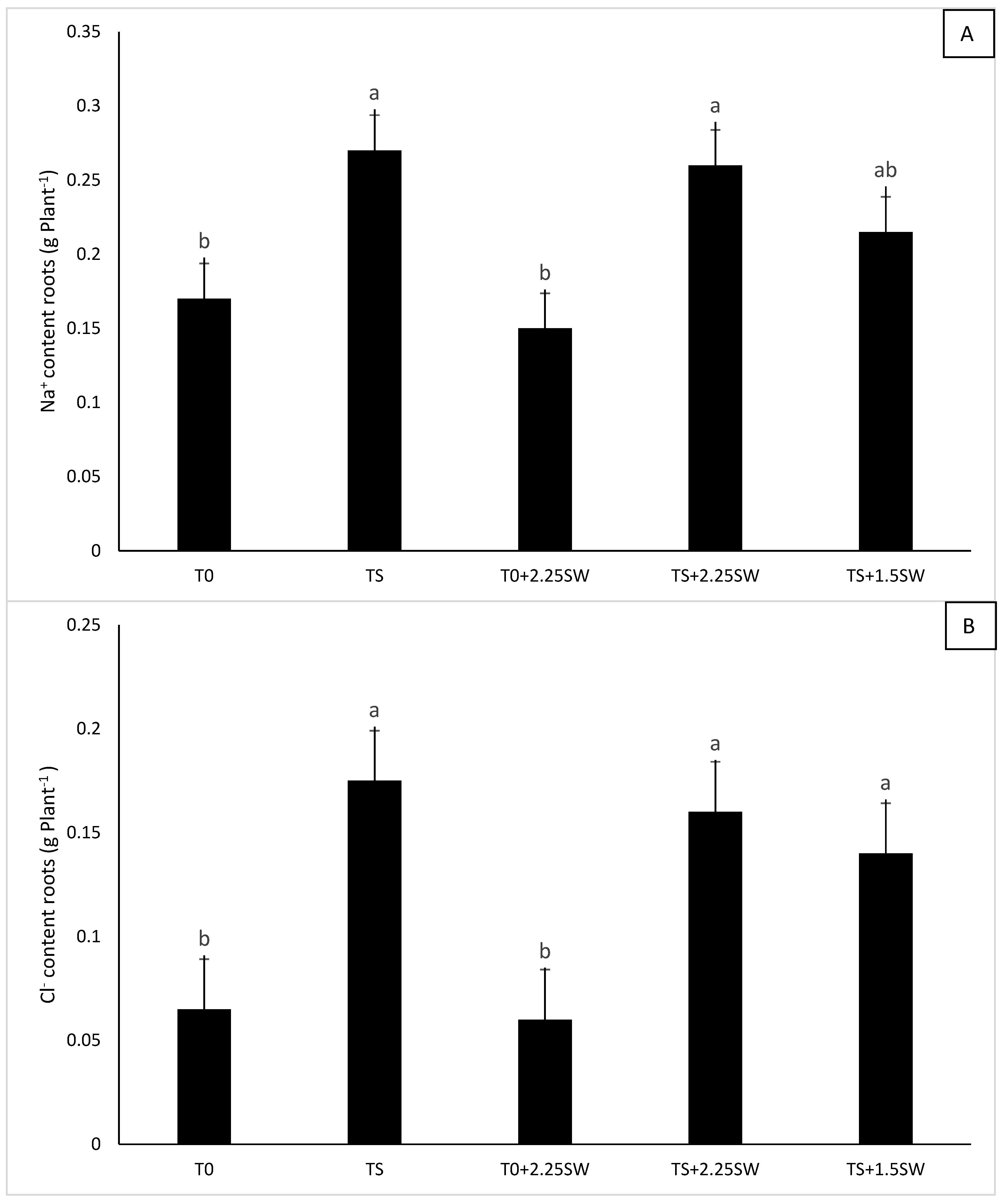
3.2. Effect of Salt Irrigation and Seaweed Extract (SE) on Nutrient Composition and Biochemical Parameters
4. Discussion
5. Conclusions
Author Contributions
Conflicts of Interest
References
- Alvarez, C.; Marrero, A.; Gallo, A.; Gonzalez, A. Physiological response of selected avocados (Persea americana) subjected to NaCl and NaHCO3 stress. Sci. Hortic. 2018, 237, 81–88. [Google Scholar] [CrossRef]
- Haas, A.R.C. Effect of sodium chloride on Mexican, Guatemalan and West Indian avocado seedlings. Calif. Avocado Soc. Yearb. 1950, 34, 153–160. [Google Scholar]
- Poltronieri, P.; Bonsegna, B.; De-Domenico, S.; Santino, A. Molecular mechanisms in plant abiotic stress responde. Field Veg. Crops Res. 2011, 48, 15–24. [Google Scholar] [CrossRef]
- Rahnama, A.; James, R.A.; Poustini, K.; Munns, R. Stomatal conductance as a screen for osmotic stress tolerance in durum wheat growing in saline soil. Funct. Plant Biol. 2010, 37, 255–263. [Google Scholar] [CrossRef]
- Al Hassan, M.; Martínez, M.; Ramos, F.J.; Boscaiu, M. Effects of Salt and Water Stress on Plant Growth and on Accumulation of Osmolytes and Antioxidant Compounds in Cherry Tomato. Not. Bot. Horti Agrobot. Cluj-Napoca 2015, 43, 1–11. [Google Scholar] [CrossRef][Green Version]
- Nandy, P.; Das, S.; Ghose, M.; Spooner-Hart, R. Effects of salinity on photosynthesis, leaf anatomy, ion accumulation and photosynthetic nitrogen use efficiency in five Indian mangroves. Wetl. Ecol. Manag. 2007, 15, 47–57. [Google Scholar] [CrossRef]
- Craigie, J.S. Seaweed extract stimuli in plant science and agriculture. J. Appl. Phycol. 2011, 23, 371–393. [Google Scholar] [CrossRef]
- Airoldi, L.; Beck, M. Oceanography and Marine Biology: An Annual Review; CRC Press: New York, NY, USA, 2007; Volume 45, pp. 345–405. ISBN 978-1-4200-5094-3. [Google Scholar]
- Dos Reis, S.P.; Lima, A.M.; De Souza, C.R.B. Recent molecular advances on downstream plant responses to abiotic stress. Int. J. Mol. Sci. 2012, 13, 8628–8647. [Google Scholar] [CrossRef] [PubMed]
- Fan, D.; Hodges, D.; Critchley, A.; Prithiviraj, B. A commercial extract of Brown Macroalga (Ascophyllum nodosum) affects yield and the nutritional quality of spinach in vitro. Commun. Soil Sci. Plant Anal. 2013, 44, 1873–1884. [Google Scholar] [CrossRef]
- Yang, S.L.; Lan, S.S.; Gong, M. Hydrogen peroxide-induce proline and metabolic pathway of its accumulation in maize seedlings. J. Plant Physiol. 2009, 166, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Nounjan, N.; Theerakulpisut, P. Effects of exogenous proline and trehalose on physiological responses in rice seedlings during salt-stress and after recovery. Plant Soil Environ. 2012, 58, 309–315. [Google Scholar] [CrossRef]
- Morales-Payan, J.P.; Candelas, C.D. Increasing Organic Avocado Fruit Yield Using an Ascophyllum nodosum Biostimulant and Fertilization. Acta Hortic. ISHS 2014, 1042, 121–124. [Google Scholar] [CrossRef]
- Hameed, M.; Ashraf, M. Physiological and biochemical adaptations of Cynodon dactylon (L.) Pers. from the Salt Range (Pakistan) to salinity stress. Flora 2008, 203, 683–694. [Google Scholar] [CrossRef]
- James, R.A.; Blake, C.; Byrt, C.S.; Munns, R. Major genes for Na+ exclusion, Nax1 and Nax2 (wheat HKT1;4 and HKT1;5), decrease Na+ accumulation in bread wheat leaves under saline and waterlogged conditions. J. Exp. Bot. 2011, 62, 2939–2947. [Google Scholar] [CrossRef] [PubMed]
- Greenway, H.; Munns, R. Mechanism of salt tolerance in nonhalophytes. Ann. Rev. Plant Physiol. 1980, 31, 149–190. [Google Scholar] [CrossRef]
- Meteorological Office of Chile. Weather Description of Metropolitana Region. Available online: http://www.meteochile.cl/PortalDMC-web/index.xhtml (accessed on 10 February 2018).
- Soltanpour, P.N.; Jones, J.B., Jr.; Workman, S.M. Optical Emission Spectrometry. In Methods of Soil Analysis. Part 2. Chemical and Microbiological Properties; Page, A., Ed.; American Society of Agronomy: Madison, WI, USA, 1982; pp. 29–65. [Google Scholar]
- Bates, L.; Waldren, P.; Teare, I. A rapid determination of free proline for water stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Yildirim, E.; Taylor, A.G.; Spittler, T.D. Ameliorative effects of biological treatments on growth of squash plants under salt stress. Sci. Hortic. 2006, 111, 1–6. [Google Scholar] [CrossRef]
- Oliveira, M.L.; Menezes, A.C.; Aragao, M.; Fernandes de Melo, D. Salt modulation of vacuolar H+ ATPase and H+-Pyrophosphatase activities in Vignaunguiculata. J. Plant Physiol. 2001, 158, 545–551. [Google Scholar] [CrossRef]
- Munns, R.; Rawson, H. Effect of salinity on salt accumulation and reproductive development in the apical meristem of wheat and barley. Aust. J. Plant Physiol. 1999, 26, 459–464. [Google Scholar] [CrossRef]
- Shi, H.; Quintero, F.J.; Pardo, J.M.; Zhu, J.-K. The putative plasma membrane Na+/H+ antiporter SOS1 controls long distance Na+ transport in plants. Plant Cell 2008, 14, 465–477. [Google Scholar] [CrossRef]
- Musyimi, D.M.; Netondo, G.W.; Ouma, G. Growth of Avocado Plants Under Saline Conditions. Int. J. Fruit Sci. 2008, 7, 67–69. [Google Scholar] [CrossRef]
- Bernstein, N.; Meiri, A.; Zilbersaine, M. Salt-stress effects on avocado rootstock growth. I. Establishing criteria for determination of shoot growth sensitivity to the stress. Plant Soil 2001, 233, 1–11. [Google Scholar] [CrossRef]
- Khan, M.S.; Zaidi, A.; Wani, P.A.; Oves, M. Role of plant growth promoting rhizobacteria in the remediation of metal contaminated soils. Environ. Chem. Lett. 2009, 7, 1–19. [Google Scholar] [CrossRef]
- Wahome, P.K.; Jesch, H.H.; Grittner, I. Effect of the NaCl on the vegetative growth and flower quality of roses. J. Appl. Bot. 2000, 74, 38–41. [Google Scholar]
- Ruiz, D.; Martinez, V.; Cerda, A. Demarcating specific ion (NaCl, Cl−, Na+) and osmotic effects in the response of two citrus rootstocks to salinity. Sci. Hortic. 1999, 80, 213–224. [Google Scholar] [CrossRef]
- Mickelbart, M.V.; Robinsona, P.W.; Witneya, G.; Arpaia, M.L. ‘Hass’ avocado tree growth on four rootstocks in California. II. Shoot and root growth. Sci. Hortic. 2012, 143, 205–210. [Google Scholar] [CrossRef]
- Mansour, M. Nitrogen containing compounds and adaptation of plants to salinity stress. Biol. Plant 2000, 43, 491–500. [Google Scholar] [CrossRef]
- Liu, J.; Xia, J.; Fang, Y.; Li, T. Effects of Salt-Drought Stress on Growth and Physiobiochemical Characteristics of Tamarix chinensis Seedlings. Sci. World J. 2014, 2014, 765840. [Google Scholar] [CrossRef] [PubMed]
- Du Jardin, P. Plant biostimulants: Definition, concept, main categories and regulation. Sci. Hortic. 2014, 196, 3–14. [Google Scholar] [CrossRef]
- Bernstein, N.; Meiri, A.; Zilbersaine, M. Root growth of avocado is more sensitive to salinity than shoot growth. J. Am. Soc. Hort. Sci. 2004, 2, 188–192. [Google Scholar]
- Shahbaz, M.; Mushtaq, Z.; Andaz, F.; Masood, A. Does proline application ameliorate adverse effects of salt stress on growth, ions and photosynthetic ability of eggplant (Solanum melongena L.)? Sci. Hortic. 2013, 164, 507–511. [Google Scholar] [CrossRef]
- Grattan, S.R.; Grieve, C.M. Salinity-mineral nutrient relations in horticultural crops. Sci. Hortic. 1999, 78, 127–157. [Google Scholar] [CrossRef]
- Silveira, J.A.; Melo, A.R.; Viegas, R.A.; Oliveira, J.T. Salinity-induced effects on nitrogen assimilation related to growth in cowpea plants. Environ. Exp. Bot. 2001, 46, 171–179. [Google Scholar] [CrossRef]
- Pardossi, A.; Malorgio, F.; Tognoni, F. Salt tolerance and mineral relations for celery. J. Plant Nutr. 1999, 22, 151–161. [Google Scholar] [CrossRef]
- Rubinigg, M.; Wenisch, J.; Elzenga, J.; Stulen, I. NaCl salinity affects lateral root development in Plantago maritima. Funct. Plant Biol. 2004, 8, 775–780. [Google Scholar] [CrossRef]
- Kamran, M.; Shahbaz, M.; Ashraf, N. Alleviation of drought-induced adverse effects in spring wheat (Triticum aestivum L.) using proline as a pre-sowing seed treatment. Pak. J. Bot. 2009, 41, 621–632. [Google Scholar]
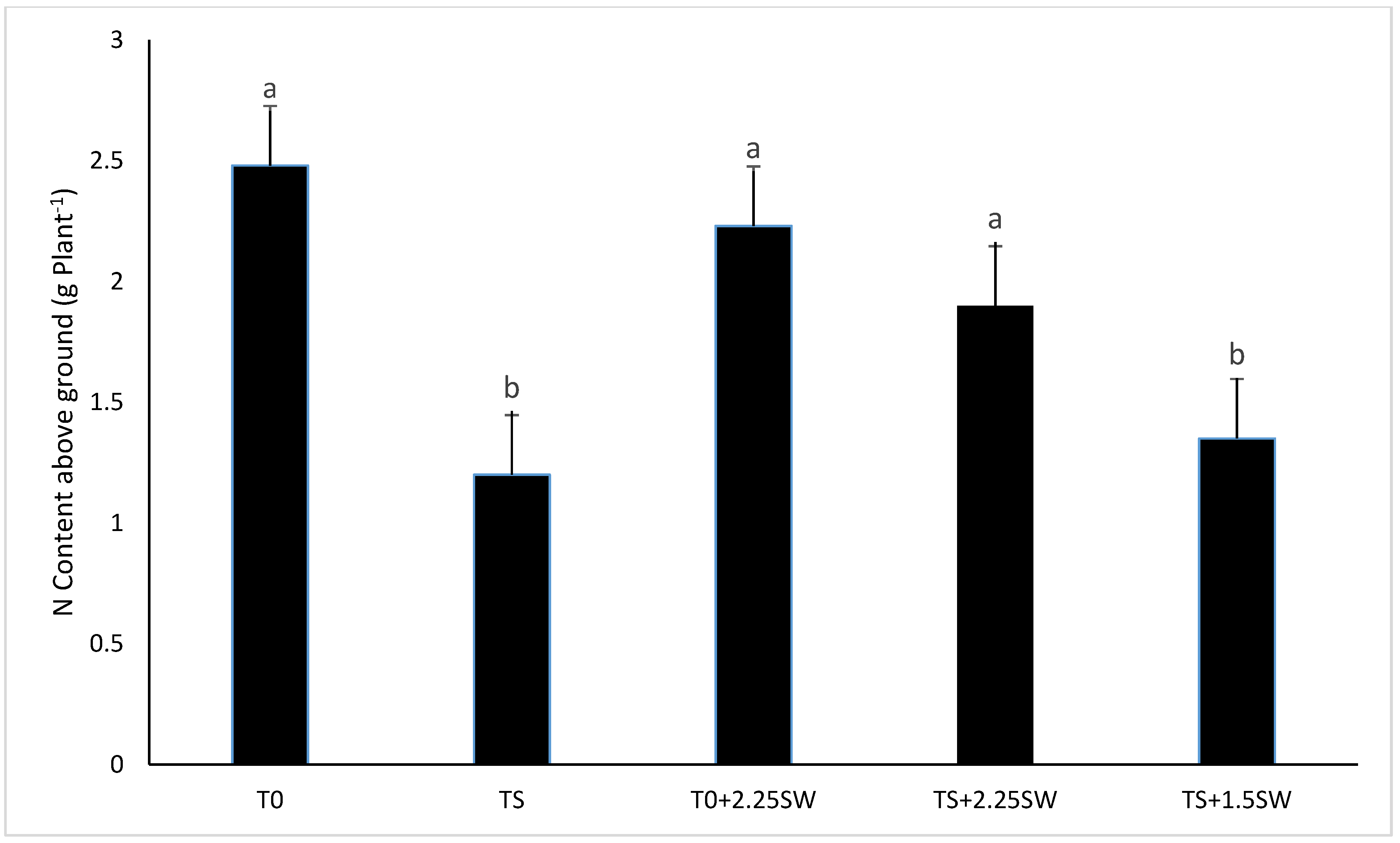
| Treatments | Parameters | |||
|---|---|---|---|---|
| Plant | ∆ Stem Diameter | Leaf | Necrosis (%) | |
| Height (mm) | (mm) | Number | ||
| T0 | 112 ± 6.72 a | 1.9 ± 0.75 a | 148 ± 8.55 a | 8 b |
| TS | 93 ± 4.67 b | 0.2 ± 0.58 b | 95 ± 15.70 b | 24 a |
| T0 + 2.25SW | 110 ± 6.32 a | 1.4 ± 0.21 a | 144 ± 9.08 a | 12 b |
| TS + 2.25SW | 101 ± 1.71 ab | 0.6 ± 0.89 ab | 119 ± 12.76 ab | 25 a |
| TS + 1.5SW | 97 ± 3.02 ab | 0.8 ± 0.28 ab | 117 ± 6.87 ab | 23 a |
| Treatments | Aboveground | Belowground |
|---|---|---|
| T0 | 155 ± 9.78 a | 48 ± 4.57 a |
| TS | 67 ± 7.10 b | 19 ± 1.39 b |
| T0 + 2.25SW | 137 ± 6.63 a | 44 ± 4.02 a |
| TS + 2.25SW | 83 ± 6.35 b | 18 ± 1.72 b |
| TS + 1.5SW | 78 ± 3.42 b | 15 ± 0.98 b |
| Treatments | Ion Concentration | |
|---|---|---|
| K+ (%) | Ca2+ (%) | |
| T0 | 1.07 ± 0.06 b | 1.10 ± 0.21 bc |
| TS | 0.93 ± 0.03 c | 0.88 ± 0.10 c |
| T0+2.25SW | 1.40 ± 0.05 a | 1.47 ± 0.06 a |
| TS+2.25SW | 1.47 ± 0.08 a | 1.07 ± 0.12 bc |
| TS+1.5SW | 1.50 ± 0.10 a | 0.97 ± 0.26 b |
| Element | Value | |
|---|---|---|
| P | % | 0.03 |
| K | % | 2.99 |
| Ca | % | 0.05 |
| Mg | % | 0.1 |
| Cu | mg/kg | 1.28 |
| Zn | mg/kg | 6.6 |
| Fe | mg/kg | 19.5 |
| Mn | mg/kg | 3.18 |
| B | mg/kg | 4.14 |
| Proline | µg/g | 56.31 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bonomelli, C.; Celis, V.; Lombardi, G.; Mártiz, J. Salt Stress Effects on Avocado (Persea americana Mill.) Plants with and without Seaweed Extract (Ascophyllum nodosum) Application. Agronomy 2018, 8, 64. https://doi.org/10.3390/agronomy8050064
Bonomelli C, Celis V, Lombardi G, Mártiz J. Salt Stress Effects on Avocado (Persea americana Mill.) Plants with and without Seaweed Extract (Ascophyllum nodosum) Application. Agronomy. 2018; 8(5):64. https://doi.org/10.3390/agronomy8050064
Chicago/Turabian StyleBonomelli, Claudia, Valentina Celis, Gian Lombardi, and Johanna Mártiz. 2018. "Salt Stress Effects on Avocado (Persea americana Mill.) Plants with and without Seaweed Extract (Ascophyllum nodosum) Application" Agronomy 8, no. 5: 64. https://doi.org/10.3390/agronomy8050064
APA StyleBonomelli, C., Celis, V., Lombardi, G., & Mártiz, J. (2018). Salt Stress Effects on Avocado (Persea americana Mill.) Plants with and without Seaweed Extract (Ascophyllum nodosum) Application. Agronomy, 8(5), 64. https://doi.org/10.3390/agronomy8050064




