The ALMT Gene Family Performs Multiple Functions in Plants
Abstract
1. Introduction
2. ALMT Genes Perform Various Functions among Plants
3. ALMTs Are Widely Distributed in the Genomes of Higher Plants
3.1. ALMT Members in Poaceae
3.2. ALMT Members in Brassicaceae
3.3. ALMT Members in Fabaceae
3.4. ALMT Members in Fruit Plants
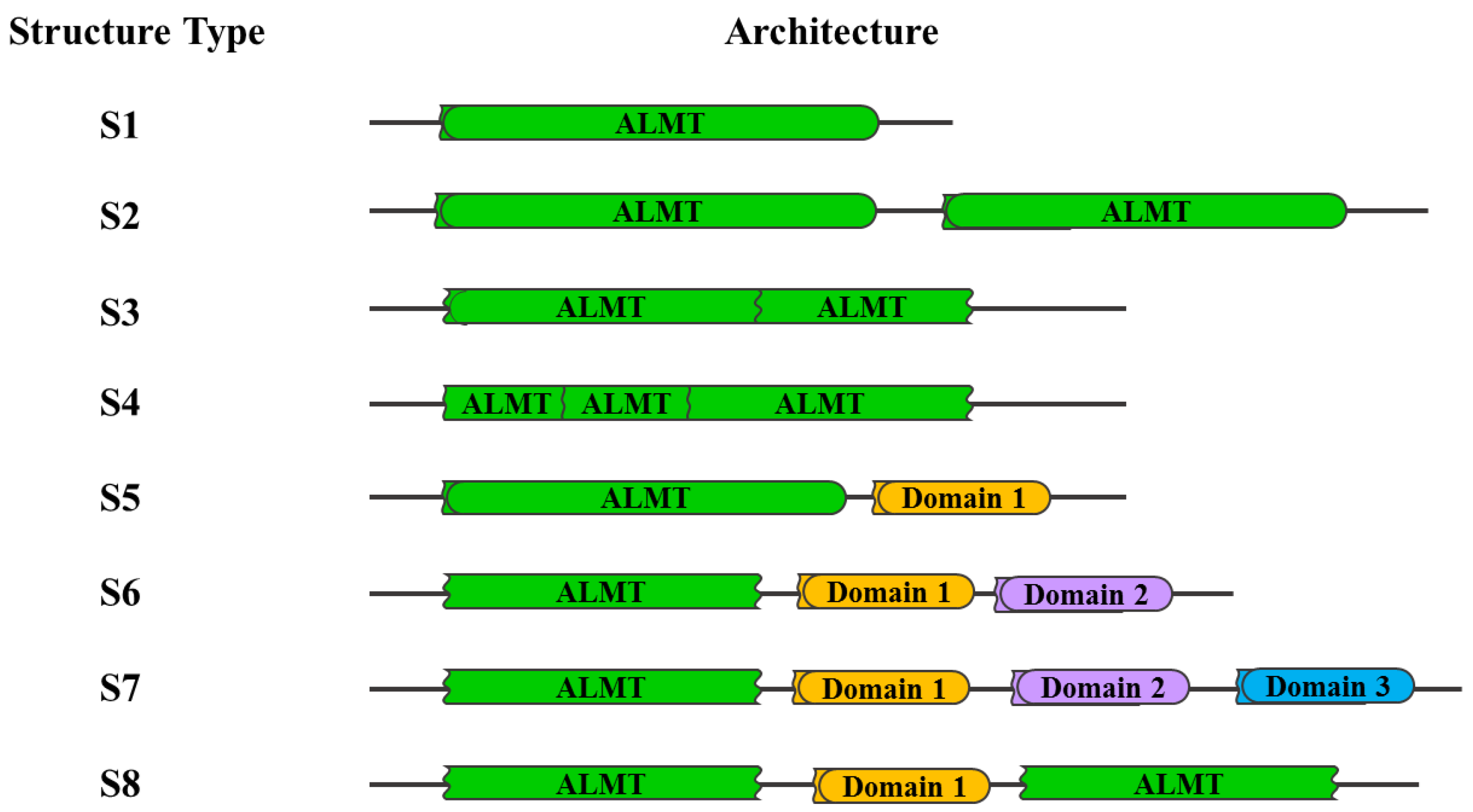
4. Structural Analysis and Evolution of the ALMT Family
4.1. Phylogeny of the ALMT Family
4.2. Secondary Structure
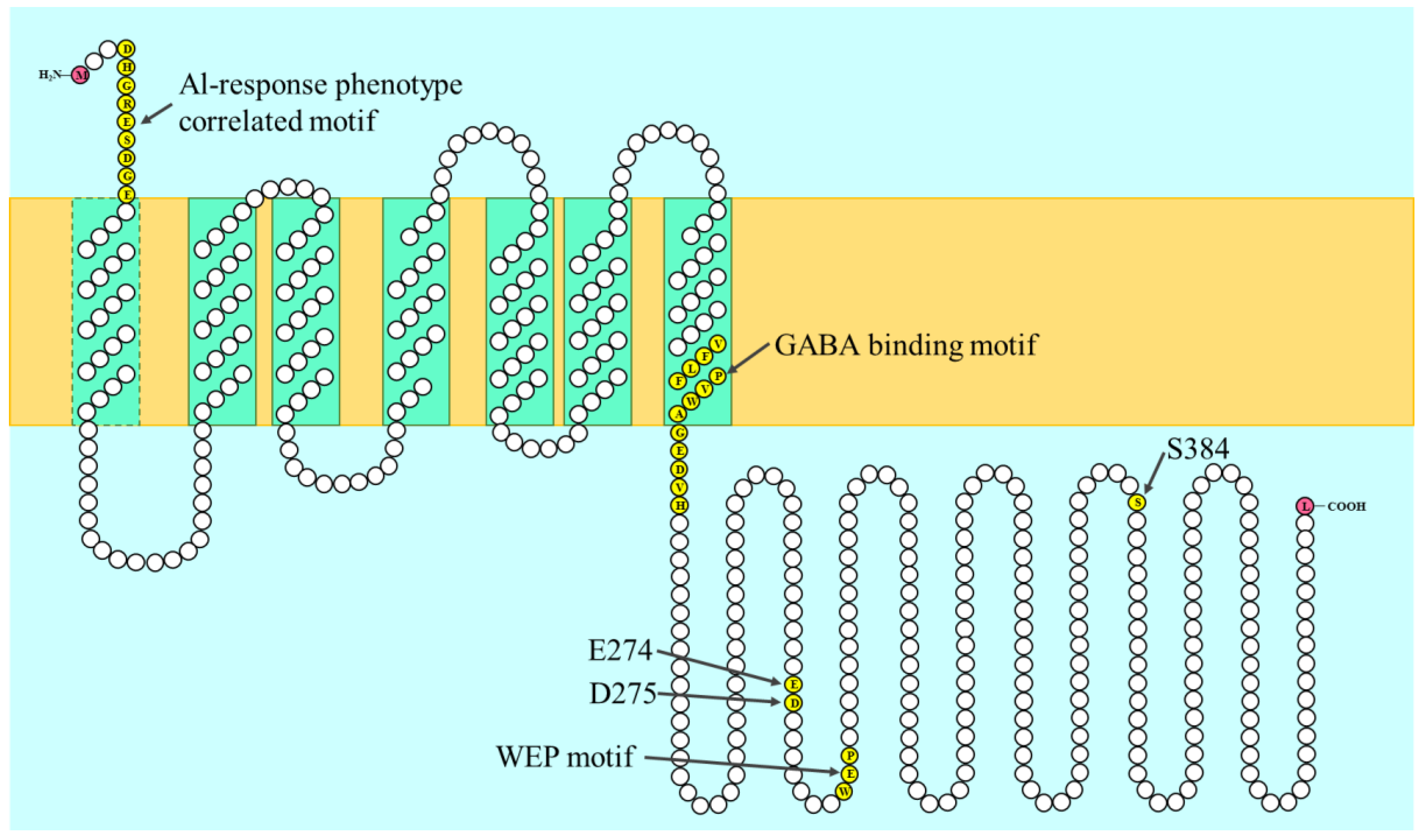
4.3. The Structure-Function Relationship of ALMT Proteins
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Sasaki, T.; Yamamoto, Y.; Ezaki, B.; Katsuhara, M.; Ahn, S.J.; Ryan, P.R.; Delhaize, E.; Matsumoto, H. A wheat gene encoding an aluminum-activated malate transporter. Plant J. 2004, 37, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Collins, N.C.; Shirley, N.J.; Saeed, M.; Pallotta, M.; Gustafson, J.P. An ALMT1 gene cluster controlling aluminum tolerance at the Alt4 locus of rye (Secale cereale L.). Genetics 2008, 179, 669–682. [Google Scholar] [CrossRef] [PubMed]
- Hoekenga, O.A.; Maron, L.G.; Pineros, M.A.; Cancado, G.M.A.; Shaff, J.; Kobayashi, Y.; Ryan, P.R.; Dong, B.; Delhaize, E.; Sasaki, T.; et al. AtALMT1, which encodes a malate transporter, is identified as one of several genes critical for aluminum tolerance in Arabidopsis. PNAS 2006, 103, 9738–9743. [Google Scholar] [CrossRef] [PubMed]
- Ligaba, A.; Katsuhara, M.; Ryan, P.R.; Shibasaka, M.; Matsumoto, H. The BnALMT1 and BnALMT2 genes from rape encode aluminum-activated malate transporters that enhance the aluminum resistance of plant cells. Plant Physiol. 2006, 142, 1294–1303. [Google Scholar] [CrossRef] [PubMed]
- Gruber, B.D.; Ryan, P.R.; Richardson, A.E.; Tyerman, S.D.; Ramesh, S.; Hebb, D.M.; Howitt, S.M.; Delhaize, E. HvALMT1 from barley is involved in the transport of organic anions. J. Exp. Bot. 2010, 61, 1455–1467. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; Mumm, P.; Imes, D.; Endler, A.; Weder, B.; Al-Rasheid, K.A.S.; Geiger, D.; Marten, I.; Martinoia, E.; Hedrich, R. AtALMT12 represents an R-type anion channel required for stomatal movement in Arabidopsis guard cells. Plant J. 2010, 63, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Mori, I.C.; Furuichi, T.; Munemasa, S.; Toyooka, K.; Matsuoka, K.; Murata, Y.; Yamamoto, Y. Closing plant stomata requires a homolog of an aluminum-activated malate transporter. Plant Cell Physiol. 2010, 51, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; Scholz-Starke, J.; De Angeli, A.; Kovermann, P.; Burla, B.; Gambale, F.; Martinoia, E. Malate transport by the vacuolar AtALMT6 channel in guard cells is subject to multiple regulation. Plant J. 2011, 67, 247–257. [Google Scholar] [CrossRef] [PubMed]
- De Angeli, A.; Zhang, J.B.; Meyer, S.; Martinoia, E. AtALMT9 is a malate-activated vacuolar chloride channel required for stomatal opening in Arabidopsis. Nat. Commun. 2013, 4, 1804. [Google Scholar] [CrossRef] [PubMed]
- Kovermann, P.; Meyer, S.; Hortensteiner, S.; Picco, C.; Scholz-Starke, J.; Ravera, S.; Lee, Y.; Martinoia, E. The Arabidopsis vacuolar malate channel is a member of the ALMT family. Plant J. 2007, 52, 1169–1180. [Google Scholar] [CrossRef] [PubMed]
- Gruber, B.D.; Delhaize, E.; Richardson, A.E.; Roessner, U.; James, R.A.; Howitt, S.M.; Ryan, P.R. Characterisation of HvALMT1 function in transgenic barley plants. Funct. Plant Biol. 2011, 38, 163–175. [Google Scholar] [CrossRef]
- Pineros, M.A.; Cancado, G.M.A.; Maron, L.G.; Lyi, S.M.; Menossi, M.; Kochian, L.V. Not all ALMT1-type transporters mediate aluminum-activated organic acid responses: The case of ZmALMT1—An anion-selective transporter. Plant J. 2008, 53, 352–367. [Google Scholar] [CrossRef] [PubMed]
- Ligaba, A.; Maron, L.; Shaff, J.; Kochian, L.; Pineros, M. Maize ZmALMT2 is a root anion transporter that mediates constitutive root malate efflux. Plant Cell Environ. 2012, 35, 1185–1200. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Dougherty, L.; Li, M.J.; Fazio, G.; Cheng, L.L.; Xu, K.N. A natural mutation-led truncation in one of the two aluminum-activated malate transporter-like genes at the Ma locus is associated with low fruit acidity in apple. Mol. Genet. Genom. 2012, 287, 663–678. [Google Scholar] [CrossRef] [PubMed]
- De Angeli, A.; Baetz, U.; Francisco, R.; Zhang, J.; Chaves, M.M.; Regalado, A. The vacuolar channel VvALMT9 mediates malate and tartrate accumulation in berries of Vitis vinifera. Planta 2013, 238, 283–291. [Google Scholar] [CrossRef] [PubMed]
- Rudrappa, T.; Czymmek, K.J.; Pare, P.W.; Bais, H.P. Root-secreted malic acid recruits beneficial soil bacteria. Plant Physiol. 2008, 148, 1547–1556. [Google Scholar] [CrossRef] [PubMed]
- Takanashi, K.; Sasaki, T.; Kan, T.; Saida, Y.; Sugiyama, A.; Yamamoto, Y.; Yazaki, K. A dicarboxylate transporter, LjALMT4, mainly expressed in nodules of Lotus japonicus. Mol. Plant Microbe Interact. 2016, 29, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.; Gruber, B.D.; Delhaize, E.; White, R.G.; James, R.A.; You, J.; Yang, Z.; Ryan, P.R. The barley anion channel, HvALMT1, has multiple roles in guard cell physiology and grain metabolism. Physiol. Plant 2015, 153, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Delhaize, E.; Gruber, B.D.; Ryan, P.R. The roles of organic anion permeases in aluminium resistance and mineral nutrition. FEBS Lett. 2007, 581, 2255–2262. [Google Scholar] [CrossRef] [PubMed]
- Dreyer, I.; Gomez-Porras, J.L.; Riano-Pachon, D.M.; Hedrich, R.; Geiger, D. Molecular evolution of slow and quick anion channels (SLACs and QUACs/ALMTs). Front. Plant Sci. 2012, 3, 263. [Google Scholar] [CrossRef] [PubMed]
- Barbier-Brygoo, H.; De Angeli, A.; Filleur, S.; Frachisse, J.M.; Gambale, F.; Thomine, S.; Wege, S. Anion channels/transporters in plants: From molecular bases to regulatory networks. Annu. Rev. Plant Biol. 2011, 62, 25–51. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.R.; Delhaize, E.; Jones, D.L. Function and mechanism of organic anion exudation from plant roots. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2001, 52, 527–560. [Google Scholar] [CrossRef] [PubMed]
- Mariano, E.D.; Jorge, R.A.; Keltjens, W.G.; Menossi, M. Metabolism and root exudation of organic acid anions under aluminium stress. Braz. J. Plant Physiol. 2005, 17, 157–172. [Google Scholar] [CrossRef]
- Maurino, V.G.; Engqvist, M.K.M. 2-hydroxy acids in plant metabolism. The Arabidopsis Book/American. Soc. Plant Biol. 2015, 13, e0182. [Google Scholar] [CrossRef]
- Roberts, S.K. Plasma membrane anion channels in higher plants and their putative functions in roots. New Phytol. 2006, 169, 647–666. [Google Scholar] [CrossRef] [PubMed]
- Roelfsema, M.R.; Hedrich, R.; Geiger, D. Anion channels: Master switches of stress responses. Trends Plant Sci. 2012, 17, 221–229. [Google Scholar] [CrossRef] [PubMed]
- Isayenkov, S.; Isner, J.C.; Maathuis, F.J. Vacuolar ion channels: Roles in plant nutrition and signalling. FEBS Lett. 2010, 584, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, J.I.; Keller, B.U. Two types of anion channel currents in guard-cells with distinct voltage regulation. PNAS 1992, 89, 5025–5029. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.R.; Shaff, J.E.; Kochian, L.V. Aluminum toxicity in roots: Correlation among ionic currents, ion fluxes, and root elongation in aluminum-sensitive and aluminum-tolerant wheat cultivars. Plant Physiol. 1992, 99, 1193–1200. [Google Scholar] [CrossRef] [PubMed]
- Kinraide, T.B.; Parker, D.R.; Zobel, R.W. Organic acid secretion as a mechanism of aluminium resistance: A model incorporating the root cortex, epidermis, and the external unstirred layer. J. Exp. Bot. 2005, 56, 1853–1865. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Lakshmanan, V.; Kobayashi, Y.; Asai, M.; Iuchi, S.; Kobayashi, M.; Bais, H.P.; Koyama, H. Overexpression of AtALMT1 in the Arabidopsis thaliana ecotype columbia results in enhanced al-activated malate excretion and beneficial bacterium recruitment. Plant Signal. Behav. 2013, 8, e25565. [Google Scholar] [CrossRef] [PubMed]
- Liang, C.; Pineros, M.A.; Tian, J.; Yao, Z.; Sun, L.; Liu, J.; Shaff, J.; Coluccio, A.; Kochian, L.V.; Liao, H. Low pH, aluminum, and phosphorus coordinately regulate malate exudation through gmalmt1 to improve soybean adaptation to acid soils. Plant Physiol. 2013, 161, 1347–1361. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Wu, K.H.; Wang, P.; Yi, J.; Li, K.Z.; Yu, Y.X.; Chen, L.M. Overexpression of MsALMT1, from the aluminum-sensitive Medicago sativa, enhances malate exudation and aluminum resistance in tobacco. Plant Mol. Biol. Rep. 2013, 31, 769–774. [Google Scholar] [CrossRef]
- Chen, Z.C.; Yokosho, K.; Kashino, M.; Zhao, F.J.; Yamaji, N.; Ma, J.F. Adaptation to acidic soil is achieved by increased numbers of cis-acting elements regulating ALMT1 expression in Holcus lanatus. Plant J. 2013. [Google Scholar] [CrossRef] [PubMed]
- Delhaize, E.; Ma, J.F.; Ryan, P.R. Transcriptional regulation of aluminium tolerance genes. Trends Plant Sci. 2012, 17, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Sharma, T.; Dreyer, I.; Kochian, L.; Pineros, M.A. The ALMT family of organic acid transporters in plants and their involvement in detoxification and nutrient security. Front Plant Sci. 2016, 7. [Google Scholar] [CrossRef] [PubMed]
- Hedrich, R. Ion channels in plants. Physiol. Rev. 2012, 92, 1777–1811. [Google Scholar] [CrossRef] [PubMed]
- Eisenach, C.; Baetz, U.; Huck, N.V.; Zhang, J.; De, A.A.; Beckers, G.; Martinoia, E. ABA-induced stomatal closure involves ALMT4, a phosphorylation-dependent vacuolar anion channel of arabidopsis. Plant Cell 2017, 29, 2552–2569. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhou, M.; Delhaize, E.; Ryan, P.R. Altered expression of a malate-permeable anion channel, OsALMT4, disrupts mineral nutrition. Plant Physiol. 2017, 175, 1745–1759. [Google Scholar] [CrossRef] [PubMed]
- Raman, H.; Zhang, K.R.; Cakir, M.; Appels, R.; Garvin, D.F.; Maron, L.G.; Kochian, L.V.; Moroni, J.S.; Raman, R.; Imtiaz, M.; et al. Molecular characterization and mapping of ALMT1, the aluminium-tolerance gene of bread wheat (Triticum aestivum L.). Genome 2005, 48, 781–791. [Google Scholar] [CrossRef] [PubMed]
- Pineros, M.A.; Cancado, G.M.A.; Kochian, L.V. Novel properties of the wheat aluminum tolerance organic acid transporter (TaALMT1) revealed by electrophysiological characterization in Xenopus oocytes: Functional and structural implications. Plant Physiol. 2008, 147, 2131–2146. [Google Scholar] [CrossRef] [PubMed]
- Delhaize, E.; Ryan, P.R.; Hebb, D.M.; Yamamoto, Y.; Sasaki, T.; Matsumoto, H. Engineering high-level aluminum tolerance in barley with the ALMT1 gene. PNAS 2004, 101, 15249–15254. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Ryan, P.R.; Delhaize, E.; Hebb, D.M.; Ogihara, Y.; Kawaura, K.; Noda, K.; Kojima, T.; Toyoda, A.; Matsumoto, H.; et al. Sequence upstream of the wheat (Triticum aestivum L.) ALMT1 gene and its relationship to aluminum resistance. Plant Cell Physiol. 2006, 47, 1343–1354. [Google Scholar] [CrossRef] [PubMed]
- Gallego, F.J.; Benito, C. Genetic control of aluminium tolerance in rye (Secale cereale L.). Theor. Appl. Genet. 1997, 95, 393–399. [Google Scholar] [CrossRef]
- Gallego, F.J.; Calles, B.; Benito, C. Molecular markers linked to the aluminium tolerance gene Alt1 in rye (Secale cereale L.). Theor. Appl. Genet. 1998, 97, 1104–1109. [Google Scholar] [CrossRef]
- Gallego, F.J.; Lopez-Solanilla, E.; Figueiras, A.M.; Benito, C. Chromosomal location of PCR fragments as a source of DNA markers linked to aluminium tolerance genes in rye. Theor. Appl. Genet. 1998, 96, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Taketa, S.; Yang, Z.M. Aluminum tolerance genes on the short arm of chromosome 3R are linked to organic acid release in triticale. Plant Physiol. 2000, 122, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Matos, M.; Camacho, M.V.; Perez-Flores, V.; Pernaute, B.; Pinto-Carnide, O.; Benito, C. A new aluminum tolerance gene located on rye chromosome arm 7RS. Theor. Appl. Genet. 2005, 111, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Fontecha, G.; Silva-Navas, J.; Benito, C.; Mestres, M.A.; Espino, F.J.; Hernandez-Riquer, M.V.; Gallego, F.J. Candidate gene identification of an aluminum-activated organic acid transporter gene at the alt4 locus for aluminum tolerance in rye (Secale cereale L.). Theor. Appl. Genet. 2007, 114, 249–260. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.R.; Raman, H.; Gupta, S.; Sasaki, T.; Yamamoto, Y.; Delhaize, E. The multiple origins of aluminium resistance in hexaploid wheat include Aegilops tauschii and more recent cis mutations to TaALMT1. Plant J. 2010, 64, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Krill, A.M.; Kirst, M.; Kochian, L.V.; Buckler, E.S.; Hoekenga, O.A. Association and linkage analysis of aluminum tolerance genes in maize. PLoS ONE 2010, 5, e9958. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Hoekenga, O.A.; Itoh, H.; Nakashima, M.; Saito, S.; Shaff, J.E.; Maron, L.G.; Pineros, M.A.; Kochian, L.V.; Koyama, H. Characterization of AtALMT1 expression in aluminum-inducible malate release and its role for rhizotoxic stress tolerance in Arabidopsis. Plant Physiol. 2007, 145, 843–852. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Tsuchiya, Y.; Ariyoshi, M.; Ryan, P.R.; Furuichi, T.; Yamamoto, Y. A domain-based approach for analyzing the function of aluminum-activated malate transporters from wheat (Triticum aestivum) and Arabidopsis thaliana in xenopus oocytes. Plant Cell Physiol. 2014, 55, 2126–2138. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, Y.; Kobayashi, Y.; Sugimoto, M.; Lakshmanan, V.; Iuchi, S.; Kobayashi, M.; Bais, H.P.; Koyama, H. Characterization of the complex regulation of AtALMT1 expression in response to phytohormones and other inducers. Plant Physiol. 2013, 162, 732–740. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, V.; Kitto, S.L.; Caplan, J.L.; Hsueh, Y.H.; Kearns, D.B.; Wu, Y.S.; Bais, H.P. Microbe-associated molecular patterns-triggered root responses mediate beneficial rhizobacterial recruitment in Arabidopsis. Plant Physiol. 2012, 160, 1642–1661. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, R.; Li, D.; Jia, X.; Zhou, D.; Li, J.; Lyi, S.M.; Hou, S.; Huang, Y.; Kochian, L.V.; et al. Nip1;2 is a plasma membrane-localized transporter mediating aluminum uptake, translocation, and tolerance in arabidopsis. PNAS 2017, 114, 5047–5052. [Google Scholar] [CrossRef] [PubMed]
- Imes, D.; Mumm, P.; Bohm, J.; Al-Rasheid, K.A.; Marten, I.; Geiger, D.; Hedrich, R. Open stomata 1 (ost1) kinase controls R-type anion channel QUAC1 in Arabidopsis guard cells. Plant J. 2013, 74, 372–382. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Martinoia, E.; De Angeli, A. Cytosolic nucleotides block and regulate the Arabidopsis vacuolar anion channel AtALMT9. J. Biol. Chem. 2014, 289, 25581–25589. [Google Scholar] [CrossRef] [PubMed]
- Ligaba, A.; Shen, H.; Shibata, K.; Yamamoto, Y.; Tanakamaru, S.; Matsumoto, H. The role of phosphorus in aluminium-induced citrate and malate exudation from rape (Brassica napus). Physiol. Plant 2004, 120, 575–584. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Zhang, X.D.; Wang, S.S.; Wang, Q.F.; Wang, G.Q.; Nian, H.J.; Li, K.Z.; Yu, Y.X.; Chen, L.M. Transcriptional and physiological changes of alfalfa in response to aluminium stress. J. Agric. Sci. 2011, 149, 737–751. [Google Scholar] [CrossRef]
- Yang, L.T.; Jiang, H.X.; Qi, Y.P.; Chen, L.S. Differential expression of genes involved in alternative glycolytic pathways, phosphorus scavenging and recycling in response to aluminum and phosphorus interactions in citrus roots. Mol. Biol. Rep. 2012, 39, 6353–6366. [Google Scholar] [CrossRef] [PubMed]
- Ma, B.; Liao, L.; Zheng, H.; Chen, J.; Wu, B.; Ogutu, C.; Li, S.; Korban, S.S.; Han, Y. Genes encoding aluminum-activated malate transporter ii and their association with fruit acidity in apple. Plant Genome 2015, 8. [Google Scholar] [CrossRef]
- Etienne, A.; Genard, M.; Lobit, P.; Mbeguie, A.M.D.; Bugaud, C. What controls fleshy fruit acidity? A review of malate and citrate accumulation in fruit cells. J. Exp. Bot. 2013, 64, 1451–1469. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, T.; Tsuchiya, Y.; Ariyoshi, M.; Nakano, R.; Ushijima, K.; Kubo, Y.; Mori, I.C.; Higashiizummi, E.; Galis, I.; Yamamoto, Y. Two members of the aluminum-activated malate transporter family, SlALMT4 and SlALMT5, are expressed during fruit development and the overexpression of SlALMT5 alters organic acid contents in seeds in tomato (Solanum lycopersicum). Plant Cell Physiol. 2016, 57, 2367–2379. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Wang, X.; Hu, T.; Zhang, F.; Wang, B.; Li, C.; Yang, T.; Li, H.; Lu, Y.; Giovannoni, J.; et al. An InDel in the promoter of Al-activated malate transporter9 selected during tomato domestication determines fruit malate contents and aluminum tolerance. Plant J. 2017, 29, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Punta, M.; Coggill, P.C.; Eberhardt, R.Y.; Mistry, J.; Tate, J.; Boursnell, C.; Pang, N.; Forslund, K.; Ceric, G.; Clements, J. The pfam protein families database. Nucleic Acids Res. 2012, 40, 290–301. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Baetz, U.; Krugel, U.; Martinoia, E.; De Angeli, A. Identification of a probable pore-forming domain in the multimeric vacuolar anion channel AtALMT9. Plant Physiol. 2013, 163, 830–843. [Google Scholar] [CrossRef] [PubMed]
- Harley, K.T.; Saier, M.H. A novel ubiquitous family of putative efflux transporters. J. Mol. Microbiol. Biotechnol. 2000, 2, 195–198. [Google Scholar] [PubMed]
- Tamura, K.; Peterson, D.; Peterson, N.; Stecher, G.; Nei, M.; Kumar, S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 2011, 28, 2731–2739. [Google Scholar] [CrossRef] [PubMed]
- Motoda, H.; Sasaki, T.; Kano, Y.; Ryan, P.R.; Delhaize, E.; Matsumoto, H.; Yamamoto, Y. The membrane topology of ALMT1, an aluminum-activated malate transport protein in wheat (Triticum aestivum). Plant Signal. Behav. 2007, 2, 467–472. [Google Scholar] [CrossRef] [PubMed]
- Meyer, S.; De Angeli, A.; Fernie, A.R.; Martinoia, E. Intra- and extra-cellular excretion of carboxylates. Trends Plant Sci. 2010, 15, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Mumm, P.; Imes, D.; Martinoia, E.; Al-Rasheid, K.A.; Geiger, D.; Marten, I.; Hedrich, R. C-terminus-mediated voltage gating of Arabidopsis guard cell anion channel QUAC1. Mol. Plant 2013, 6, 1550–1563. [Google Scholar] [CrossRef] [PubMed]
- Ligaba, A.; Dreyer, I.; Margaryan, A.; Schneider, D.J.; Kochian, L.; Piñeros, M. Functional, structural and phylogenetic analysis of domains underlying the Al-sensitivity of the aluminium-activated malate/anion transporter, TaALMT1. Plant J. 2013, 76, 766–780. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.R.; Delhaize, E. The convergent evolution of aluminium resistance in plants exploits a convenient currency. Funct. Plant Biol. 2010, 37, 275–284. [Google Scholar] [CrossRef]
- Osawa, H.; Matsumoto, H. Possible involvement of protein phosphorylation in aluminum-responsive malate efflux from wheat root apex. Plant Physiol. 2001, 126, 411–420. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, M.; Ohura, I.; Kawakita, K.; Yokota, N.; Fujiwara, M.; Shimamoto, K.; Doke, N.; Yoshioka, H. Calcium-dependent protein kinases regulate the production of reactive oxygen species by potato nadph oxidase. Plant Cell 2007, 19, 1065–1080. [Google Scholar] [CrossRef] [PubMed]
- Ligaba, A.; Kochian, L.; Pineros, M. Phosphorylation at S384 regulates the activity of the TaALMT1 malate transporter that underlies aluminum resistance in wheat. Plant J. 2009, 60, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Furuichi, T.; Sasaki, T.; Tsuchiya, Y.; Ryan, P.R.; Delhaize, E.; Yamamoto, Y. An extracellular hydrophilic carboxy-terminal domain regulates the activity of TaALMT1, the aluminum-activated malate transport protein of wheat. Plant J. 2010, 64, 47–55. [Google Scholar] [CrossRef] [PubMed]
- Ryan, P.R.; Tyerman, S.D.; Sasaki, T.; Furuichi, T.; Yamamoto, Y.; Zhang, W.H.; Delhaize, E. The identification of aluminium-resistance genes provides opportunities for enhancing crop production on acid soils. J. Exp. Bot. 2011, 62, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, S.A.; Tyerman, S.D.; Xu, B.; Bose, J.; Kaur, S.; Conn, V.; Domingos, P.; Ullah, S.; Wege, S.; Shabala, S.; et al. GABA signalling modulates plant growth by directly regulating the activity of plant-specific anion transporters. Nat. Commun. 2015, 6, 7879. [Google Scholar] [CrossRef] [PubMed]
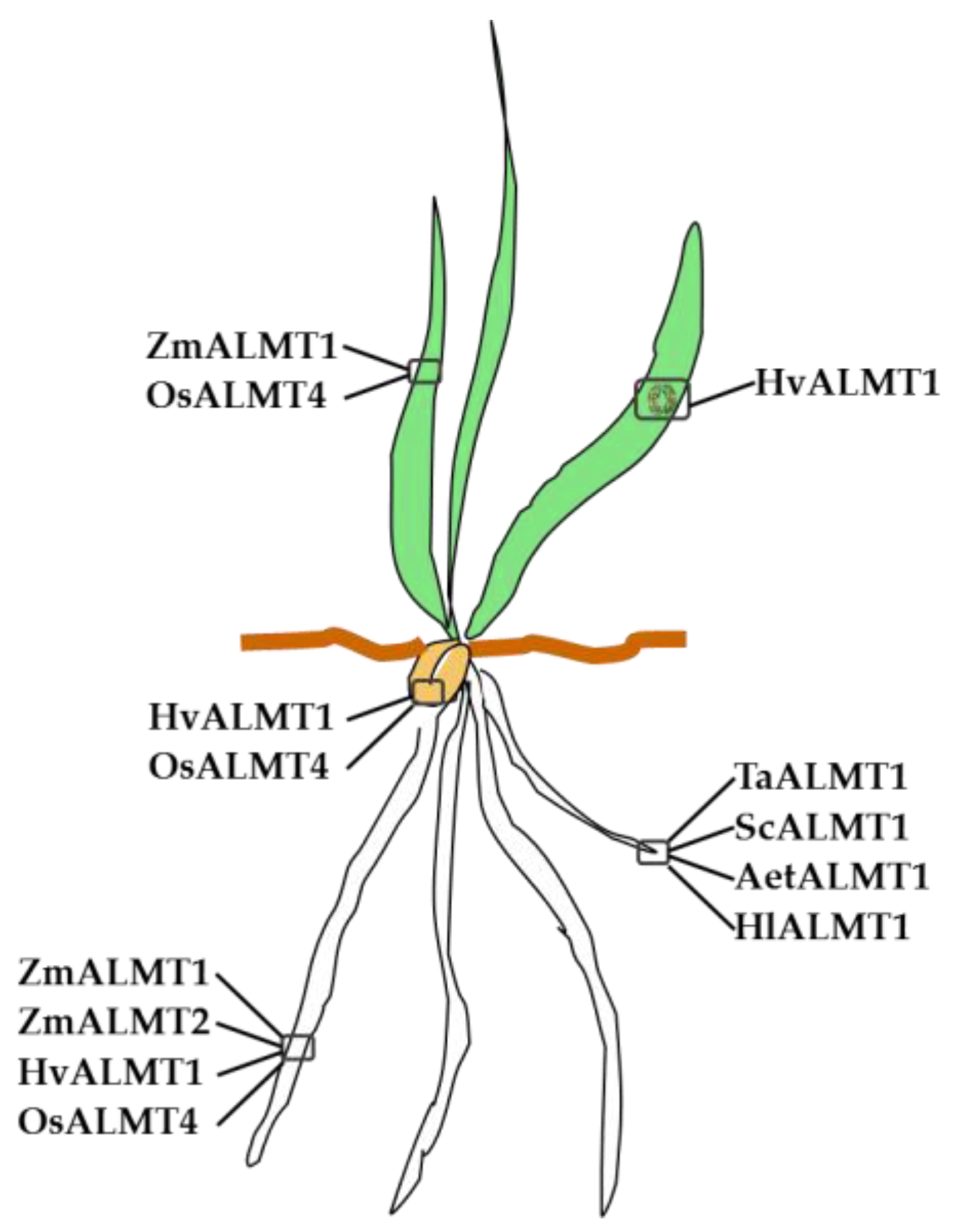
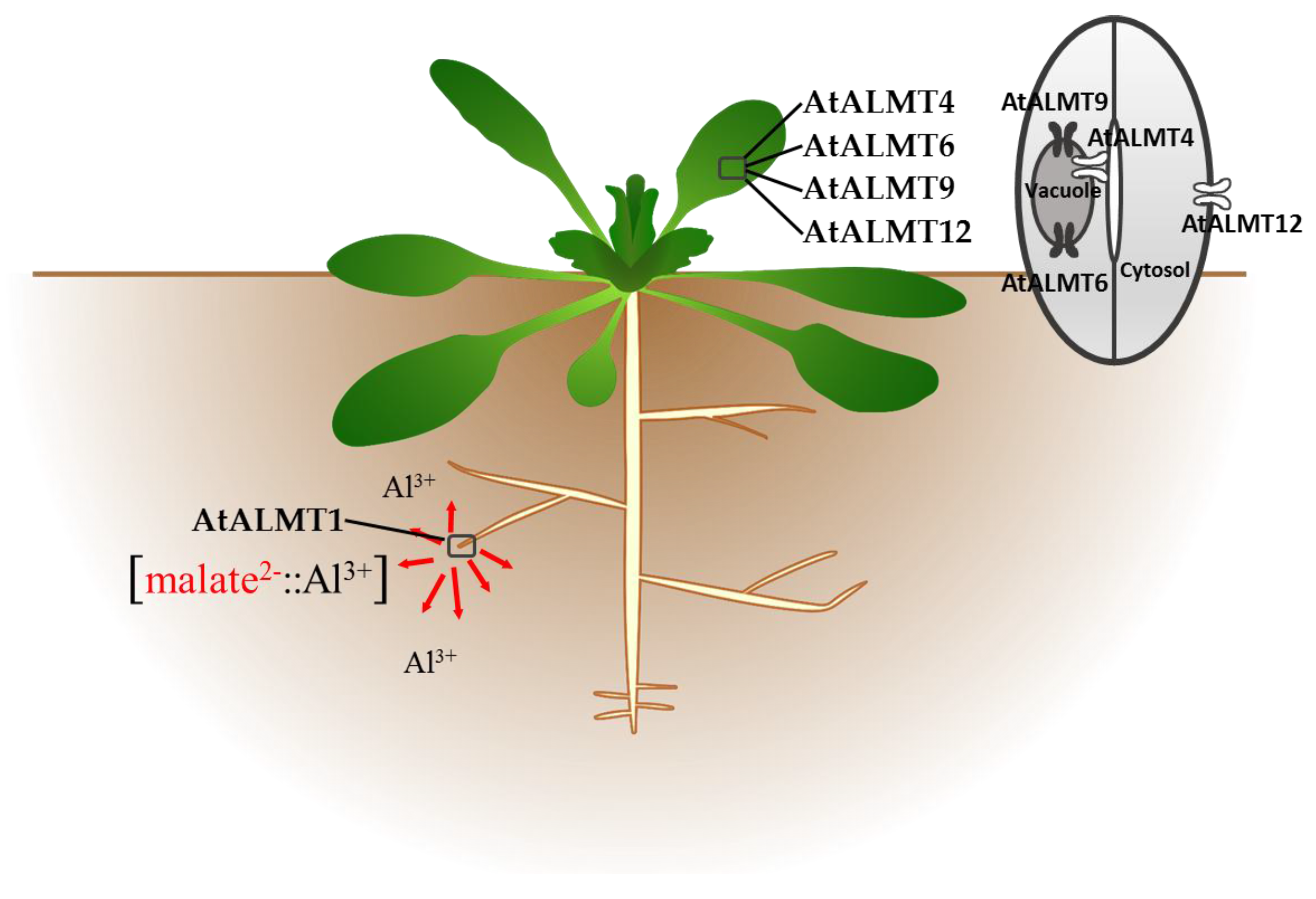
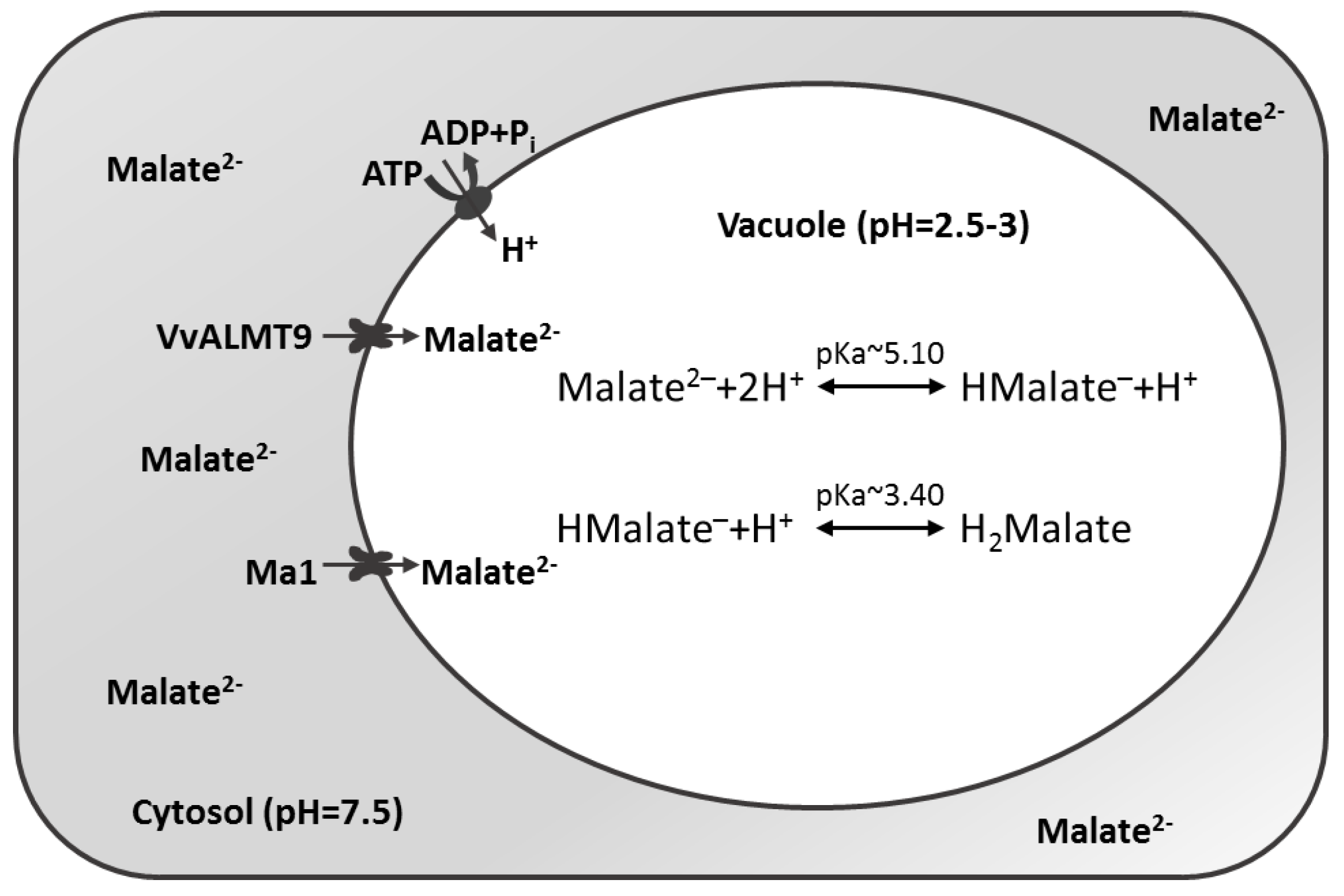
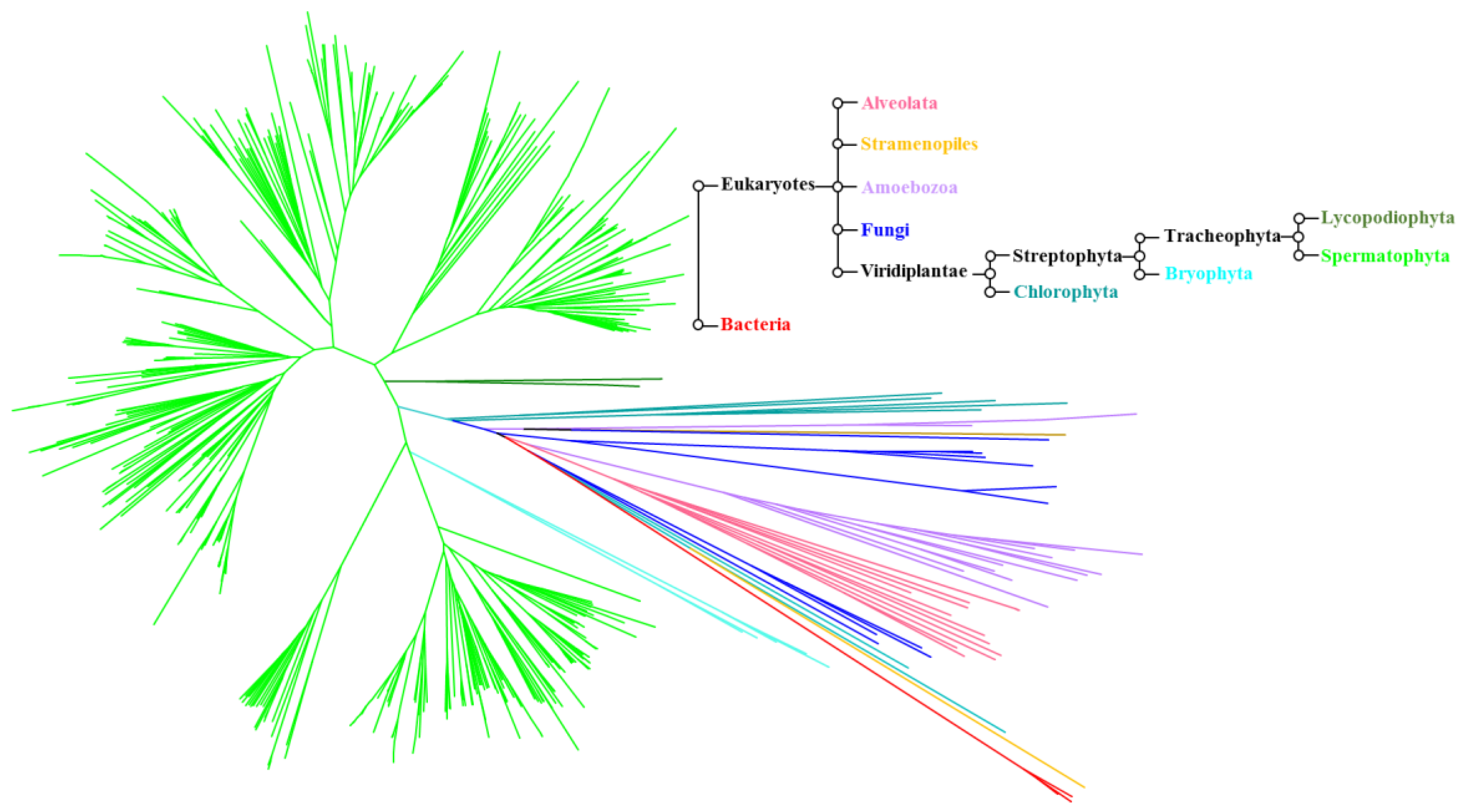
| Family | Species | Gene | Function(s) |
|---|---|---|---|
| Poaceae | Wheat | TaALMT1 | Al Resistance |
| Rye | ScALMT1 | Al Resistance | |
| Goatgrass | AetALMT1 | Al Resistance | |
| Barley | HvALMT1 | Stomatal Functions, Seed development | |
| Yorkshire fog | HlALMT1 | Al Resistance | |
| Maize | ZmALMT1 | Mineral Nutrition | |
| ZmALMT2 | Mineral Nutrition | ||
| Rice | OsALMT4 | Mineral Nutrition | |
| Brassicaceae | Arabidopsis | AtALMT1 | Al Resistance, Microbe interactions |
| AtALMT4 | Stomatal Functions | ||
| AtALMT12 | Stomatal Functions | ||
| AtALMT6 | Stomatal Functions | ||
| AtALMT9 | Stomatal Functions | ||
| Rape | BnALMT1 | Al Resistance | |
| BnALMT2 | Al Resistance | ||
| Fabaceae | Alfalfa | MsALMT1 | Al Resistance |
| Soybean | GmALMT1 | Al Resistance | |
| Lotus japonicus L. | LjALMT4 | Microbe interactions | |
| Rutaceae | Citrus | CsALMT1/CgALMT1 | Al Resistance |
| Rosaceae | Apple | Ma1/Ma2 | Fruit acidity |
| Vitaceae | Grape | VvALMT9 | Fruit acidity |
| Solanaceas | Tomato | SlALMT4 | Unclear |
| SlALMT5 | Seed malate content | ||
| SlALMT9 | fruit malate accumulation, Al Resistance |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, J.; Zhou, M. The ALMT Gene Family Performs Multiple Functions in Plants. Agronomy 2018, 8, 20. https://doi.org/10.3390/agronomy8020020
Liu J, Zhou M. The ALMT Gene Family Performs Multiple Functions in Plants. Agronomy. 2018; 8(2):20. https://doi.org/10.3390/agronomy8020020
Chicago/Turabian StyleLiu, Jie, and Meixue Zhou. 2018. "The ALMT Gene Family Performs Multiple Functions in Plants" Agronomy 8, no. 2: 20. https://doi.org/10.3390/agronomy8020020
APA StyleLiu, J., & Zhou, M. (2018). The ALMT Gene Family Performs Multiple Functions in Plants. Agronomy, 8(2), 20. https://doi.org/10.3390/agronomy8020020






