On-Farm Demonstrations with a Set of Good Agricultural Practices (GAPs) Proved Cost-Effective in Reducing Pre-Harvest Aflatoxin Contamination in Groundnut
Abstract
1. Introduction
2. Results
2.1. Soil Chemical Properties
2.2. Extent of Farmers’ Awareness and Adoption of Good Agricultural Practices (GAPs) and Technologies in Groundnut
2.3. On-Farm Demonstrations
2.4. Cost Economics of GAPs Vis-à-Vis Farmers’ Practices
2.5. Post-Harvest Losses in Groundnut
3. Discussion
4. Materials and Methods
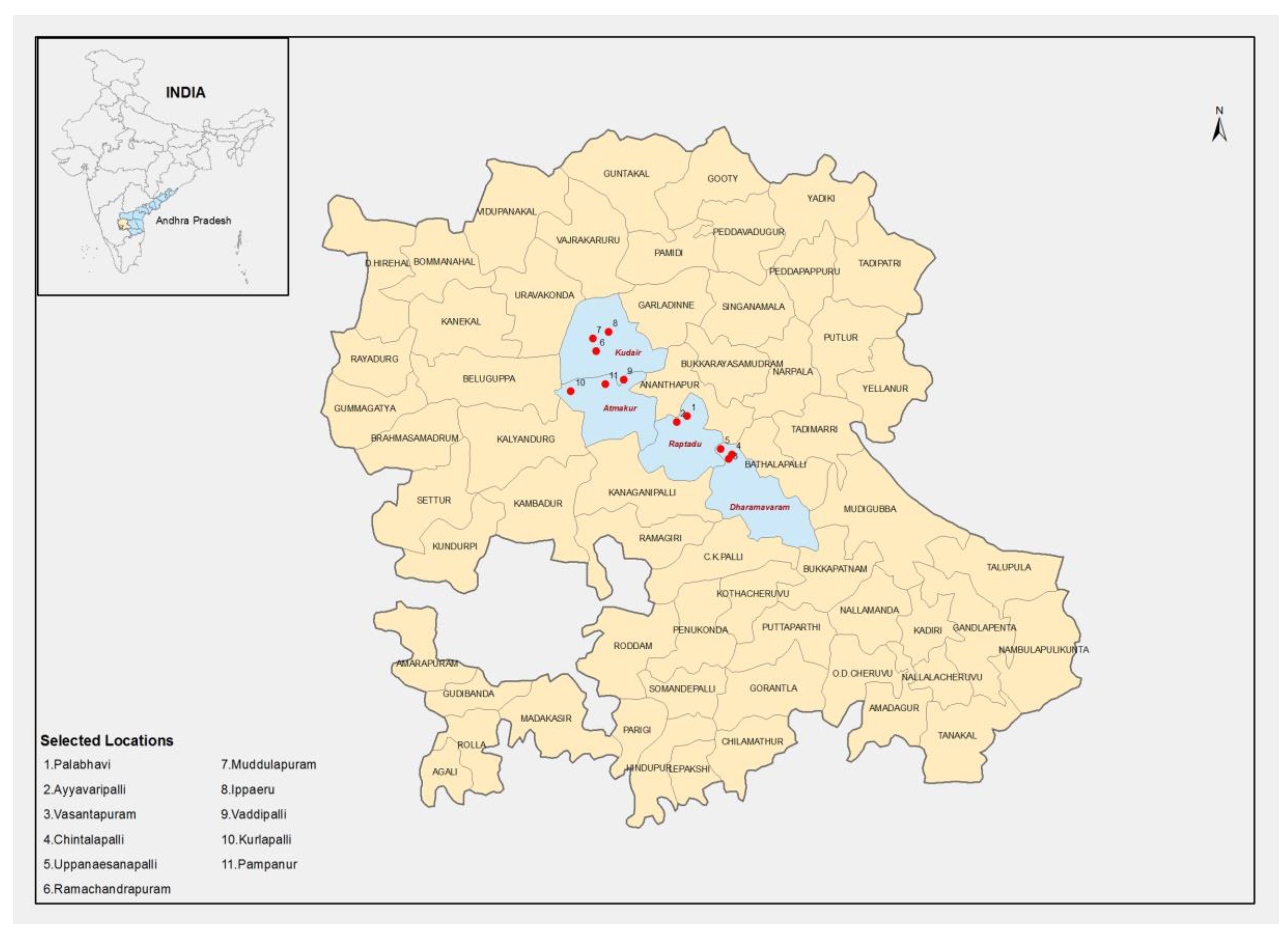
4.1. Selection and Details of Sites for On-Farm Demonstrations
4.2. Extent of Farmers’ Awareness and Adoption of Good Agricultural Practices (GAPs) during Pre- and Post-Harvest Stages of Groundnut
4.3. On-Farm Demonstrations
4.4. Cost Economics of GAPs Vis-à-Vis Farmers’ Practices
4.5. Estimation of Kernel A. flavus Infection and Aflatoxin Contamination
4.6. Data Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Waliyar, F.; Kumar, P.L.; Traore, A.; Ntare, B.R.; Diarra, B.; Kodio, O. Pre and post-harvest management of aflatoxin contamination in peanuts. In Mycotoxins Detection Methods, Management, Public Health and Agricultural Trade; Leslie, J., Bandyopadhyay, R., Visconti, A., Eds.; CABI: Waringford, UK, 2008; pp. 209–218. [Google Scholar]
- Monyo, E.S.; Njoroge, S.M.C.; Coe, R.; Osiru, M.; Madinda, F.; Waliyar, F.; Thakur, R.P.; Chilunjika, T.; Anitha, S. Occurrence and distribution of aflatoxin contamination in groundnuts (Arachis hypogaea L.) and population density of Aflatoxigenic Aspergilli in Malawi. Crop Prot. 2012, 42, 149–155. [Google Scholar] [CrossRef]
- Waliyar, F.; Umeh, V.C.; Traore, A.; Osiru, M.; Ntare, B.R.; Diarra, B.; Kodio, O.; Vijay Krishna Kumar, K.; Sudini, H. Prevalence and distribution of aflatoxin contamination in groundnut (Arachis hypogaea L.) in Mali, West Africa. Crop Prot. 2015, 70, 1–7. [Google Scholar] [CrossRef]
- VanEgmond, H.P. (Ed.) Aflatoxin M1: Occurrence, toxicity, regulation. In Mycotoxins in Dairy Products; Elsevier Applied Science: London, UK, 1989; pp. 11–55. [Google Scholar]
- D’Mello, J.P.F.; MacDonald, A.M.C. Mycotoxins. Anim. Feed Sci. Technol. 1997, 69, 155–166. [Google Scholar] [CrossRef]
- Boutrif, E.; Canet, C. Mycotoxin prevention and control: FAO Programmes. Revue de Médecine Vétérinaire 1998, 149, 681–694. [Google Scholar]
- Abdin, M.Z.; Ahmad, M.M.; Javed, S. Advances in molecular detection of Aspergillus: An update. Arch. Microbiol. 2010, 192, 409–425. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S. Aflatoxin and food safety: Recent African Perspectives. J. Toxicol. Toxin Rev. 2003, 22, 267–286. [Google Scholar] [CrossRef]
- Williams, J.H.; Phillips, T.D.; Jolly, P.E.; Stiles, J.K.; Jolly, C.M.; Aggarwal, D. Human aflatoxicosis in developing countries: A review of toxicology, exposure, potential health consequences, and interventions. Am. J. Clin. Nutr. 2004, 80, 1106–1122. [Google Scholar] [PubMed]
- Strosnider, H.; Azziz-Baumgartner, E.; Banziger, M.; Bhat, R.V.; Breiman, R.; Brune, M.N.; DeCock, K.; Dilley, A.; Groopman, J.; Hell, K.; et al. Workgroup report: Public health strategies for reducing aflatoxin exposure in developing countries. Environ. Health Perspect. 2006, 114, 1898–1903. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wu, F. Global burden of aflatoxin-induced hepatocellular carcinoma: A risk assessment. Environ. Health Perspect. 2010, 118, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Diener, U.L.; Cole, R.J.; Sanders, T.H.; Payne, G.A.; Lee, L.S.; Klich, M.A. Epidemiology of aflatoxin formation by Aspergillus flavus. Annu. Rev. Phytopathol. 1987, 25, 249–270. [Google Scholar] [CrossRef]
- Torres, A.M.; Barros, G.G.; Palacios, S.A.; Chulze, S.N.; Battilani, P. Review on pre- and post-harvest management of peanuts to minimize aflatoxin contamination. Food Res. Int. 2014, 62, 11–19. [Google Scholar] [CrossRef]
- FAOSTAT. Food and Agricultural Organization Statistics Database. 2013. (FAOSTAT). Available online: http://faostat3.fao.org/home/E (accessed on 29 January 2016).
- Yellamanda Reddy, T.; Sulochanamma, B.N.; Subramanyam, A.; Balaguravaiah, D. Influence of weather, dry spells and management practices on aflatoxin contamination in groundnut. Indian Phytopathol. 2003, 56, 262–265. [Google Scholar]
- Hill, R.A.; Blankenship, P.D.; Cole, R.J.; Sanders, T.H. Effects of soil moisture and temperature on pre harvest invasion of peanuts by the Aspergillus flavus group and subsequent aflatoxin development. Appl. Environ. Microbiol. 1983, 45, 628–633. [Google Scholar] [PubMed]
- Cole, R.J.; Sanders, T.H.; Hill, R.A.; Blankenship, P.D. Mean geocarposphere temperatures that induce pre-harvest aflatoxin contamination of peanuts under drought stress. Mycopathologia 1985, 91, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.J.; Sanders, T.H.; Dorner, J.W.; Blankenship, P.D. Environmental conditions required to induce pre harvest aflatoxin contamination of groundnuts: Summary of six years’ research. In Aflatoxin Contamination of Groundnut: Proceedings of the International Workshop, 6–9 October 1987. ICRISAT Center, India; Hall, S.D., Ed.; ICRISAT: Patancelu, India, 1989; pp. 279–287. [Google Scholar]
- Wilson, D.M.; Walker, M.E. Effects of gypsum and irrigation on Aspergillus flavus group colonization of peanuts. In Proceedings of the American Peanut Research and Education Society, Tulsa, OK, USA, 12–15 July 1988; p. 24. [Google Scholar]
- Waliyar, F.; Ntare, B.R.; Diallo, A.T.; Kodio, O.; Diarra, B. On-Farm Management of Aflatoxin Contamination of Groundnut in West Africa: A Synthesis Report; International Crops Research Institute for the Semi-Arid Tropics: Patancelu, India, 2007; 24p. [Google Scholar]
- Arunyanark, A.; Jogloy, S.; Wongkaew, S.; Akkasaeng, C.; Vorasoot, N.; Kesmala, T.; Patanothai, A. Heritability of aflatoxin resistance traits and correlation with drought tolerance. Field Crops Res. 2010, 117, 258–264. [Google Scholar] [CrossRef]
- Hamidou, F.; Rathore, A.; Waliyar, F.; Vadez, V. Although drought intensity increases aflatoxin contamination, drought tolerance does not lead to less aflatoxin contamination. Field Crops Res. 2014, 156, 103–110. [Google Scholar] [CrossRef]
- Wilson, D.M.; Stansell, J.R. Effect of irrigation regimes on aflatoxin contamination of peanut pods. Peanut Sci. 1983, 10, 54–56. [Google Scholar] [CrossRef]
- Mixon, A.C.; Bell, D.K.; Wilson, D.M. Effect of chemical and biological agents on the incidence of Aspergillus flavus and aflatoxin contamination of peanut seed. Phytopathology 1984, 74, 1440–1444. [Google Scholar] [CrossRef]
- Reding, C.L.C.; Harrison, M.A.; Kvien, C.K. Aspergillus parasiticus growth and aflatoxin synthesis on florunner peanuts grown in gypsum-supplemented soil. J. Food Prot. 1993, 56, 593–611. [Google Scholar] [CrossRef]
- Lazarovits, G.; Tenuta, M.; Conn, K.L. Organic amendments as a disease control strategy for soilborne diseases of high-value agricultural crops. Australas. Plant Pathol. 2001, 30, 111–117. [Google Scholar] [CrossRef]
- Chhetry, G.K.N.; Mangang, H.C. Effect of soil amendments on soil borne pathogens of French bean (Phaseolus vulgaris L.) in organic farming system in Manipur. J. Agric. Sci. Technol. 2011, 1, 68–72. [Google Scholar]
- Wilson, D.M.; Walker, M.E. Calcium potential aflatoxin foe. South-East. Peanut Farmer 1981, 19, 6B. [Google Scholar]
- Thakur, R.P.; Rao, V.P.; Subramanyam, K. Influence of biocontrol agents on population density of Aspergillus flavus as well as kernel infection in groundnut. Indian Phytopathol. 2003, 56, 408–412. [Google Scholar]
- Sanders, T.H.; Cole, R.J.; Blankenship, P.D.; Dorner, J.W. Aflatoxin concentration of peanuts from plants drought stressed in pod or root zones. Peanut Sci. 1993, 20, 5–8. [Google Scholar] [CrossRef]
- Craufurd, P.Q.; Prasad, P.V.V.; Waliyar, F.; Taheri, A. Drought, pod yield, pre-harvest Aspergillus infection and aflatoxin contamination on peanut in Niger. Field Crops Res. 2006, 98, 20–29. [Google Scholar] [CrossRef]
- Payne, G.A.; Hagler, W.M., Jr. Effect of specific amino acids on growth and aflatoxin production by Aspergillus parasiticus and Aspergillus flavus in defined media. Appl. Environ. Microbiol. 1983, 46, 805–812. [Google Scholar] [PubMed]
- Middleton, K.J.; Pande, S.S.; Sharma, S.B.; Smith, D.H. Diseases. In Groundnut Crop: A Scientific Basis for Improvement; Smart, J.S., Ed.; Chapman and Hall: London, UK, 1994; pp. 336–378. [Google Scholar]
- Mutegi, C.K.; Ngugi, H.K.; Hendriks, S.L.; Jones, R.B. Factors associated with the incidence of Aspergillus section Flavi and aflatoxin contamination of peanuts in the Busia and Homa bay districts of western Kenya. Plant Pathol. 2012, 61, 1143–1153. [Google Scholar] [CrossRef]
- Wu, F.; Liu, Y.; Bhatnagar, D. Cost-effectiveness of aflatoxin control methods: Economic Incentives. Toxin Rev. 2008, 27, 203–225. [Google Scholar] [CrossRef]
- Reddy, P.S.; Basu, M.S.; Khaleque, M.A.; Haque, M.S.; Ali, A.; Malek, H.; Than, H.; Soe, T.; Regunathan, B.; Mishra, B.; et al. Status of groundnut research and production in South Asia. In Groundnut—A Global Perspective. Proceedings of an International Workshop 25–29 November 1991, ICRIST Center; Nigam, S.N., Ed.; ICRISAT: Patancelu, India, 1992; pp. 133–147. [Google Scholar]
- Sudini, H.; Ranga Rao, G.V.; Gowda, C.L.L.; Chandrika, R.; Margam, V.; Rathore, A.; Murdock, L.L. Purdue Improved Crop Storage (PICS) bags for safe storage of groundnuts. J. Stored Prod. Res. 2015, 64, 133–138. [Google Scholar] [CrossRef]
- Gong, Y.Y.; Hounsa, A.; Egal, S.; Turner, P.C.; Sutcliffe, A.E.; Hall, A.J.; Cardwell, K.; Wild, C.P. Post-weaning exposure to aflatoxin results in impaired child growth: A longitudinal study in Benin, West Africa. Environ. Health Perspect. 2004, 112, 1334–1338. [Google Scholar] [CrossRef] [PubMed]
- Kumar, G.D.S.; Popat, M.N. Assessment of adoption gaps in management of aflatoxin contamination of groundnut (Arachis hypogaea L.). J. Agric. Educ. Ext. 2010, 16, 309–319. [Google Scholar] [CrossRef]
- Kumar, G.D.S.; Popat, M.N. Farmers’ perceptions, knowledge and management of aflatoxins in groundnuts (Arachis hypogaea L.) in India. Crop Prot. 2010, 29, 1534–1541. [Google Scholar] [CrossRef]
- Kumar, G.D.S.; Popat, M.N. Factors influencing the adoption of aflatoxin management practices in groundnut (Arachis hypogaea L.). Int. J. Pest Manag. 2010, 56, 165–171. [Google Scholar] [CrossRef]
- Kishore, G.K.; Pande, S.; Manjula, K.; Narayana Rao, J.; Thomas, D. Occurrence of mycotoxins and toxigenic fungi in groundnut (Arachis hypogaea L.) seeds in Andhra Pradesh, India. Plant Pathol. J. 2002, 18, 204–209. [Google Scholar] [CrossRef]
- Okalebo, J.R.; Gathua, K.W.; Woomer, P.L. Exchangeable bases in soils. In Laboratory Methods of Soil and Plant Analysis: A Working Manual; Tropical Soil Biology and Fertility Programme: Nairobi, Kenya, 1993; pp. 52–54. [Google Scholar]
- Nelson, D.W.; Sommers, L.E. Total carbon, Organic carbon and organic matter. In Methods of Soil Analysis, Part 3: Chemical Methods; Sparks, D.L., Page, A.L., Eds.; Soil Science Society of America & American Society of Agronomy: Madison, WI, USA, 1996; pp. 961–1010. [Google Scholar]
- Thomas, G.W. Soil pH and soil acidity. In Methods of Soil Analysis, Part 3: Chemical Methods; Sparks, D.L., Page, A.L., Eds.; Soil Science Society of America & American Society of Agronomy: Madison, WI, USA, 1996; pp. 475–490. [Google Scholar]
- Waliyar, F.; Bockelee-Morvan, A. Resistance of groundnut varieties to Aspergillus flavus in Senegal. In Proceedings of the International Workshop on Aflatoxin Contamination Groundnut, Patancheru, India, 6–9 October 1987; pp. 305–310. [Google Scholar]
- Raper, K.B.; Fennell, D.I. The Genus Aspergillus; Williams and Wilkins: Baltimore, MD, USA, 1965; p. 686. [Google Scholar]
- Reddy, S.V.; Kiran Mayi, D.; Uma Reddy, M.; Thirumala Devi, K.; Reddy, D.V.R. Aflatoxin B1 in different grades of chilies (Capsicum annum) as determined by indirect competitive-ELISA. Food Addit. Contam. 2001, 18, 553–558. [Google Scholar] [CrossRef] [PubMed]
| Mandal/Location | Soil Parameters | ||
|---|---|---|---|
| pH | Organic Carbon (%) | Calcium (ppm) | |
| Rapthadu | 7.9 (±0.09) | 0.32 (±0.03) | 1501 (±116.4) |
| Dharmavaram | 7.0 (±0.22) | 0.48 (±0.04) | 1612 (±152.0) |
| Kudaeru | 7.1 (±0.17) | 0.44 (±0.02) | 2289 (±286.3) |
| Atmakur | 7.2 (±0.17) | 0.49 (±0.17) | 2039 (±170.7) |
| Serial No. | Parameter Assessed | Prevalence/Extent of Adoption among Farmers |
|---|---|---|
| Awareness on Technical Aspects | ||
| 1 | Aflatoxin problem | 1.4% |
| 2 | Crop rotation | Nil |
| 3 | Crop varietal preference | 36.7% |
| 4 | Extent of adoption of resistant/tolerant/other improved varieties | 36.8% (drought) |
| 44.3% (high yields) | ||
| 13.2% (high oil content) | ||
| Adoption of On-Farm Practices | ||
| 5 | Application of Farm Yard Manure (FYM) | 7.1% |
| 6 | Application of chemical fertilizers | 78.2% |
| 7 | Gypsum application | 3% |
| 8 | Crop harvesting at right maturity | 100% |
| 9 | Major abiotic stresses | Drought (100%) |
| 10 | Major insect pests | Spodoptera sp., Helicoverpa sp., RHC, leaf folder |
| 11 | Major plant diseases | Leaf spots, collar rot, stem rot, bud and stem necrosis diseases |
| Adoption of Off-Farm/Post-Harvest Practices | ||
| 12 | Separation of healthy pods from diseased pods prior to storage | 100% |
| 13 | Major storage pests | Bruchids |
| 14 | Storage system | Jute bags (Traditional storage) |
| 15 | Marketing | Local traders in village |
| Practice/Characteristic | 2013 | 2014 | 2015 | Pooled (2013–2015) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aflatoxins (µg/kg) | Seed Infection with A. flavus (%) | Pod Yield (Q/acre) | Aflatoxins (µg/kg) | Seed Infection with A. flavus (%) | Pod Yield (Q/acre) | Aflatoxins (µg/kg) | Seed Infection with A. flavus (%) | Pod Yield (Q/acre) | Aflatoxins (µg/kg) | Seed Infection with A. flavus (%) | Pod Yield (Q/acre) | |
| FP | 0.0–1772.1 | 0.0–96.7 | 3.0–6.5 | 0.0–10,169.3 | 0.0–60.0 | 3.0–6.0 | 12.2–2282.2 | 3.3–53.3 | 4.0–7.0 | 0.0–10,169.3 | 0.0–96.7 | 3.0–7.0 |
| GAPs | 0.0–301.3 | 0.0–20.0 | 6.0–7.0 | 0.0–71.5 | 0.0–66.7 | 5.0–7.0 | 12.6–113.5 | 0.0–16.7 | 5.0–7.5 | 0.0–301.3 | 0.0–66.7 | 5.0–7.5 |
| FP a | 114.8 | 10.3 | 4.7 | 388.3 | 11.2 | 4.3 | 326.1 | 15.4 | 5.5 | 276.4 | 12.3 | 4.8 |
| GAPs b | 43.9 | 4.4 | 6.7 | 24.7 | 9.8 | 6.2 | 27.1 | 7.9 | 6.7 | 31.9 | 7.4 | 6.5 |
| Percent reduction/gain over FP | 61.8 | 57.6 | 30.0 | 93.6 | 12.6 | 30.4 | 91.7 | 48.7 | 18.6 | 88.5 | 40.1 | 26.3 |
| Non-Parametric Test | 2013 | 2014 | 2015 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| % Seed Infection | Aflatoxins (µg/kg) | Pod Yield (Q/acre) | % Seed Infection | Aflatoxins (µg/kg) | Pod Yield (Q/acre) | % Seed Infection | Aflatoxins (µg/kg) | Pod Yield (Q/acre) | |
| Mann–Whitney U test | 29.5 ** | 5 * | 0 * | 648 ** | 384.5 * | 58 * | 415.5 * | 90 * | 119 * |
| Z scores | 0.93 | 3.1 | −3.53 | 1.46 | 3.99 | −7.13 | 3.7 | 6.83 | −6.55 |
| Agronomic Practices/Field Operations | Expenses Incurred (USD/acre) | |
|---|---|---|
| Plots with FP | Plots with GAP | |
| Ploughing | $41.53 | $41.53 |
| Basal applications | ||
| (a) Fertilizers | $23.07 | $23.07 |
| (b) Seed cost | $83.07 | $83.07 |
| (c) Seed treatment (ST) | $3.84 | $3.84 |
| Weed management (Post-emergence) | ||
| (a) 20 DAS | $7.69 | $7.69 |
| (b) 40 DAS | $11.53 | $11.53 |
| Plant protection costs | $23.07 | $23.07 |
| Harvesting & Drying | $21.53 | $21.53 |
| Stripping | $15.38 | $15.38 |
| GAP | ||
| (a) Farmyard manure (FYM) | - | $46.15 |
| (b) Gypsum application | - | $19.23 |
| (c) Protective irrigation | - | $7.69 |
| (d) Tarpaulin sheet for pod drying | - | $1.15 |
| Total cost | $230.71 | $304.93 |
| Average pod yield | 5.5 Q (at the rate of $61.53/Q) = $338.41 | 7 Q (at the rate of $61.53/Q) = $430.71 |
| Haulm yield (t) | 1.5 t (at the rate of $9.23/t) = $13.8 | 2 t (at the rate of $9.23/t) = $18.46 |
| Total income | =$352.26 | =$449.17 |
| Net gain | $121.55 | $144.24 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Parimi, V.; Kotamraju, V.K.K.; Sudini, H.K. On-Farm Demonstrations with a Set of Good Agricultural Practices (GAPs) Proved Cost-Effective in Reducing Pre-Harvest Aflatoxin Contamination in Groundnut. Agronomy 2018, 8, 10. https://doi.org/10.3390/agronomy8020010
Parimi V, Kotamraju VKK, Sudini HK. On-Farm Demonstrations with a Set of Good Agricultural Practices (GAPs) Proved Cost-Effective in Reducing Pre-Harvest Aflatoxin Contamination in Groundnut. Agronomy. 2018; 8(2):10. https://doi.org/10.3390/agronomy8020010
Chicago/Turabian StyleParimi, Vijayaraju, Vijay Krishna K. Kotamraju, and Hari K. Sudini. 2018. "On-Farm Demonstrations with a Set of Good Agricultural Practices (GAPs) Proved Cost-Effective in Reducing Pre-Harvest Aflatoxin Contamination in Groundnut" Agronomy 8, no. 2: 10. https://doi.org/10.3390/agronomy8020010
APA StyleParimi, V., Kotamraju, V. K. K., & Sudini, H. K. (2018). On-Farm Demonstrations with a Set of Good Agricultural Practices (GAPs) Proved Cost-Effective in Reducing Pre-Harvest Aflatoxin Contamination in Groundnut. Agronomy, 8(2), 10. https://doi.org/10.3390/agronomy8020010




