Effect of Ulvan on the Biocontrol Activity of Debaryomyces hansenii and Stenotrophomonas rhizophila against Fruit Rot of Cucumis melo L.
Abstract
1. Introduction
2. Materials and Methods
2.1. Microorganisms and Fruit Materials
2.1.1. Fruit
2.1.2. Pathogen Inoculum
2.1.3. Antagonist Microorganisms
2.1.4. Chemical Treatments
2.2. Effect of Ulvan, D. hansenii, and S. rhizophila against F. proliferatum In Vivo
2.3. Effect of Ulvan, D. hansenii, and S. rhizophila against F. proliferatum In Vitro
2.3.1. Effect on Mycelial Growth
2.3.2. Effect on Spore Germination
2.4. Effect of Ulvan on Populations of D. hansenii and S. rhizophila in Vivo
2.5. Effect of Ulvan on the Growth of D. hansenii and S. rhizophila In Vitro
2.6. Effect of D. hansenii or S. rhizophila in Combination with Ulvan on Natural Disease Incidence
Determination of Quality Parameters
2.7. Statistical Analysis
3. Results
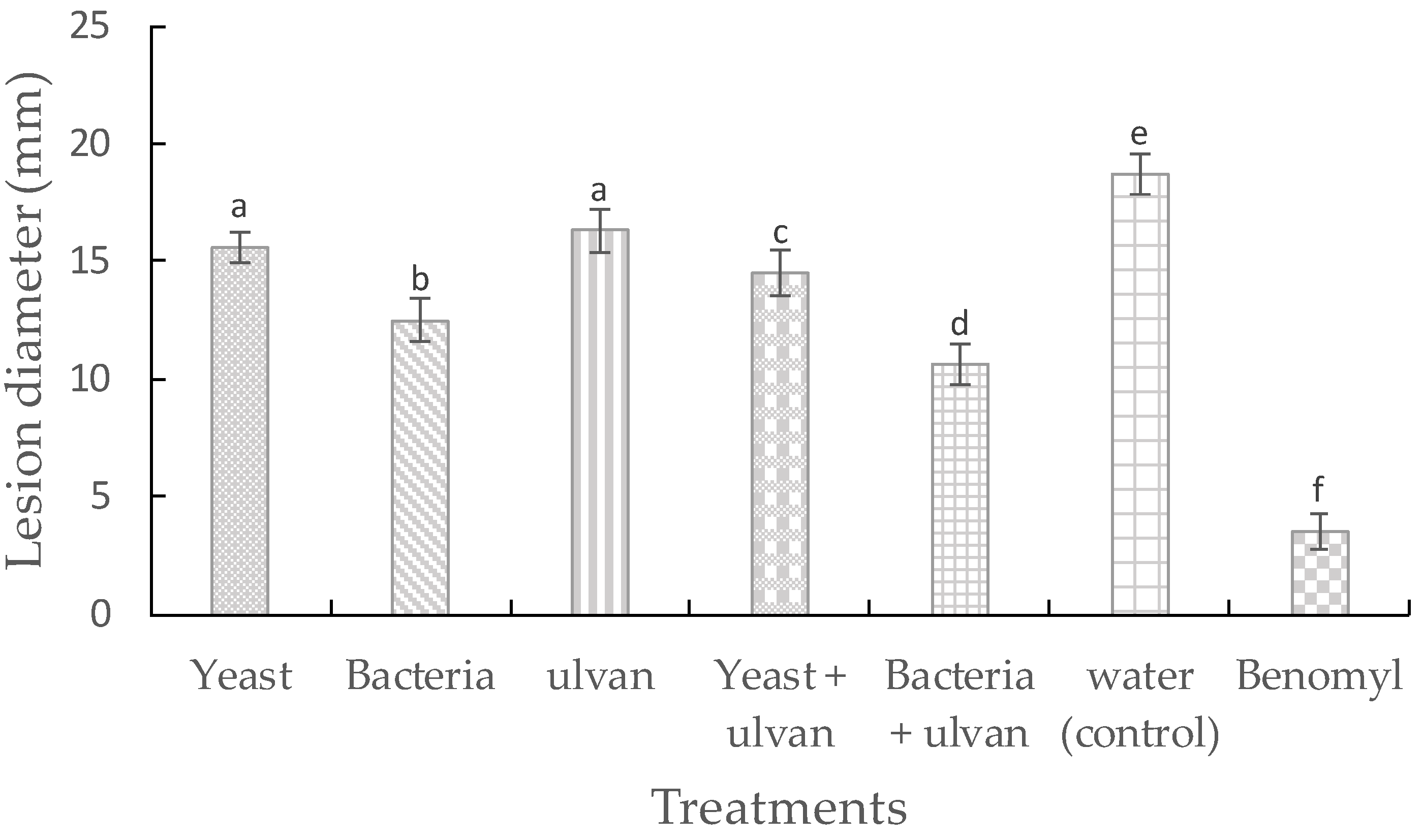
3.1. Effect of Ulvan, D. hansenii, and S. rhizophila on Muskmelon Fruit Rot by F. proliferatum
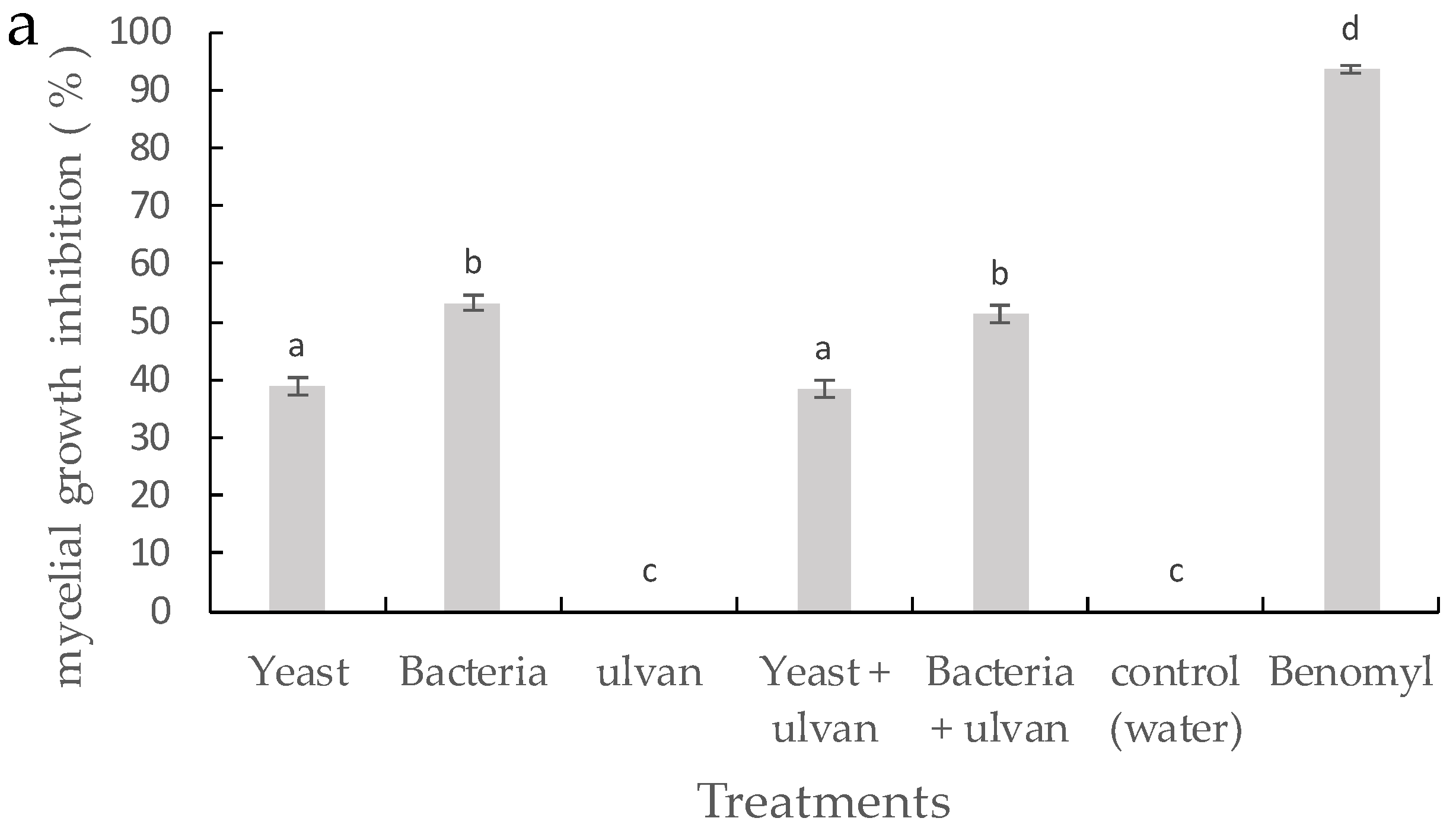
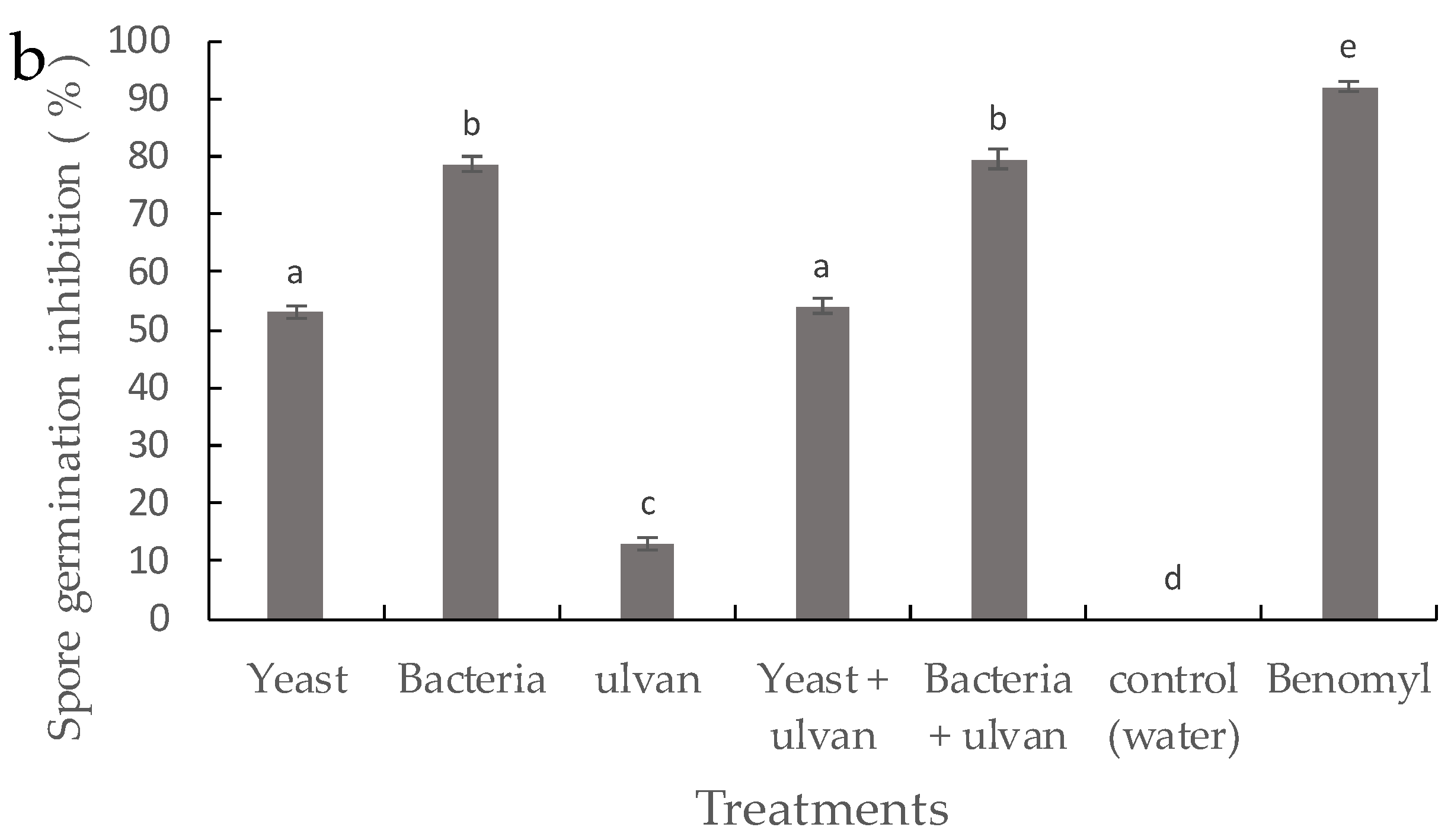
3.2. Effect of D. hansenii, S. rhizophila, and Ulvan on Mycelial Growth and Spore Germination of F. proliferatum
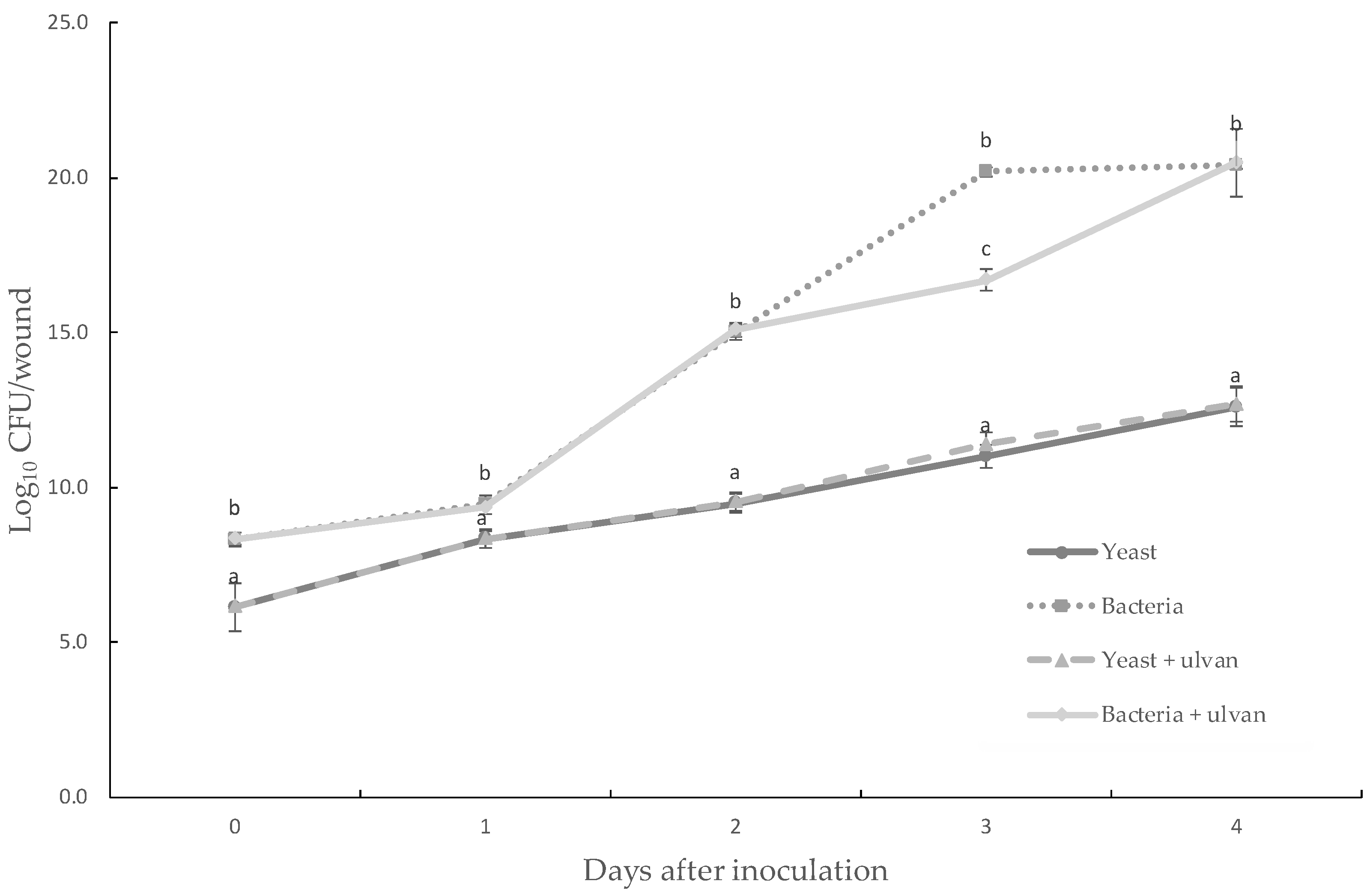
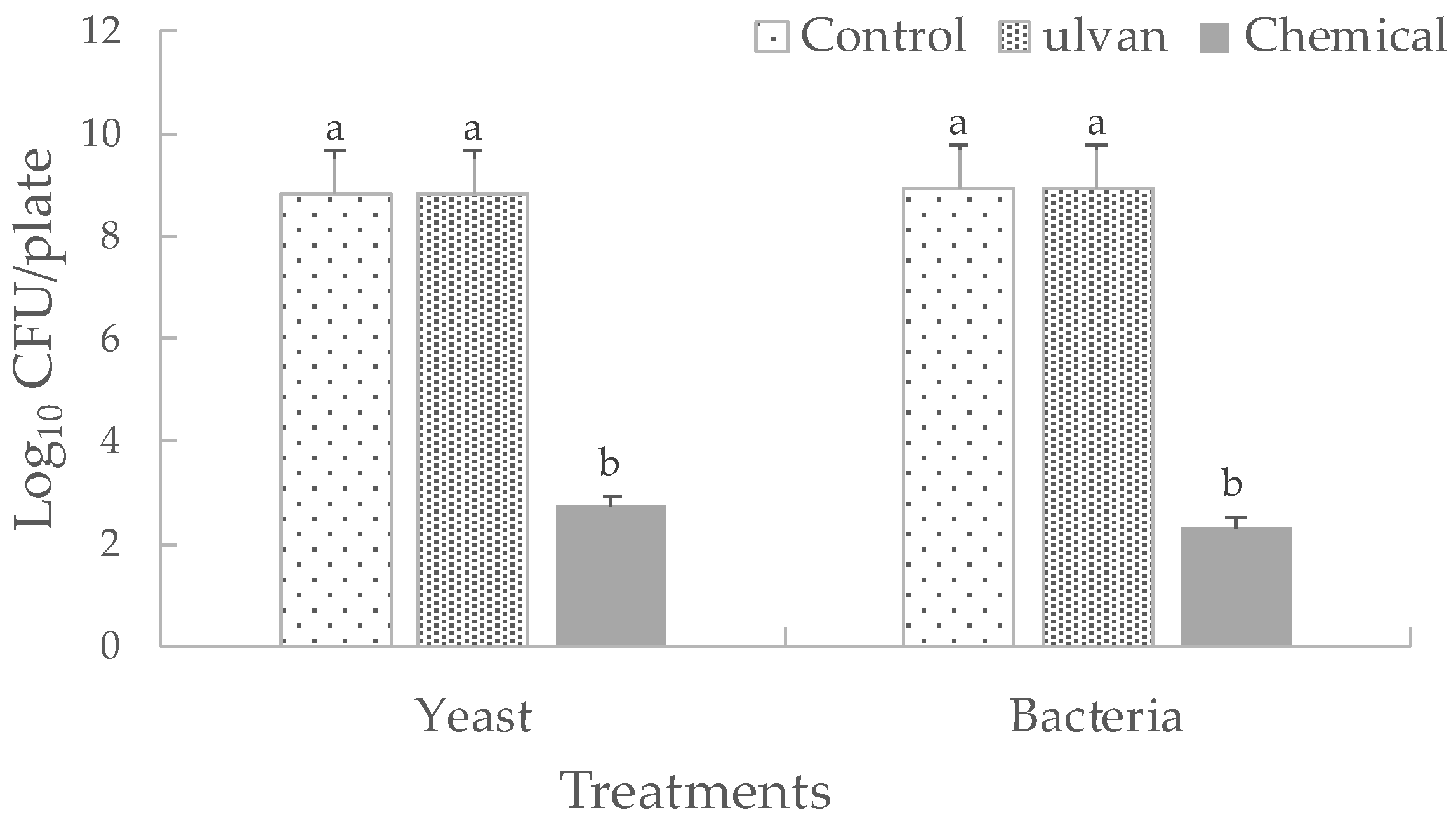
3.3. Effect of Ulvan on the In Vivo Population Dynamics or in the In Vitro Growth of D. hansenii and S. rhizophila
3.4. Effect of D. hansenii, S. rhizophila, and Ulvan on Natural Disease Incidence and Quality Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sánchez-Estrada, A.; Tiznado-Hernández, M.E.; Ojeda-Contreras, A.J.; Valenzuela-Quintanar, A.I.; Troncoso-Rojas, R. Induction of enzymes and phenolic compounds related to the natural defense response of netted melon fruit by a bio-elicitor. J. Phytopathol. 2009, 157, 24–32. [Google Scholar] [CrossRef]
- Huang, K.; Zou, Y.; Luo, J.; Liu, Y. Combining UV-C treatment with biocontrol yeast to control postharvest decay of melon. Environ. Sci. Pollut. Res. 2015, 22, 14307–14313. [Google Scholar] [CrossRef] [PubMed]
- Jamalizadeh, M.; Etebarian, H.R.; Aminian, H.; Alizadeh, A. A review of mechanisms of action of biological control organisms against post-harvest fruit spoilage. EPPO Bull. 2011, 41, 65–71. [Google Scholar] [CrossRef]
- Mari, M.; Di Francesco, A.; Bertolini, P. Control of fruit postharvest diseases: old issues and innovative approaches. Stewart Postharvest Rev. 2014, 10, 1–4. [Google Scholar]
- Droby, S.; Wisniewski, M.; Macarisin, D.; Wilson, C. Twenty years of postharvest biocontrol research: Is it time for a new paradigm? Postharvest Biol. Technol. 2009, 52, 137–145. [Google Scholar] [CrossRef]
- Medina-Córdova, N.; López-Aguilar, R.; Ascencio, F.; Castellanos, T.; Campa-Córdova, A.I.; Angulo, C. Biocontrol activity of the marine yeast Debaryomyces hansenii against phytopathogenic fungi and its ability to inhibit mycotoxins production in maize grain (Zea mays L.). Biol. Control 2016, 97, 70–79. [Google Scholar] [CrossRef]
- Hernández-Montiel, L.G.; Ochoa, J.L.; Troyo-Diéguez, E.; Larralde-Corona, C.P. Biocontrol of postharvest blue mold (Penicillium italicum Wehmer) on Mexican lime by marine and citrus Debaryomyces hansenii isolates. Postharvest Biol. Technol. 2010, 56, 181–187. [Google Scholar] [CrossRef]
- Hernandez-Montiel, L.G.; Zulueta-Rodriguez, R.; Angulo, C.; Rueda-Puente, E.O.; Quiñonez-Aguilar, E.E.; Galicia, R. Marine yeasts and bacteria as biological control agents against anthracnose on mango. J. Phytopathol. 2017, 165, 833–840. [Google Scholar] [CrossRef]
- Hernandez-Montiel, L.G.; Gutierrez-Perez, E.D.; Murillo-Amador, B.; Vero, S.; Chiquito-Contreras, R.G.; Rincon-Enriquez, G. Mechanisms employed by Debaryomyces hansenii in biological control of anthracnose disease on papaya fruit. Postharvest Biol. Technol. 2018, 139, 31–37. [Google Scholar] [CrossRef]
- Hernandez-Montiel, L.G.; Rivas-Garcia, T.R.; Romero-Bastidas, M.; Chiquito-Contreras, C.J.; Ruiz-Espinoza, F.H.; Chiquito-Contreras, R.G. Potencial antagónico de bacterias y levaduras marinas para el control de hongos fitopatógenos. REMEXCA 2018, 20, 4311–4321. [Google Scholar] [CrossRef]
- Medina-Córdova, N.; Rosales-Mendoza, S.; Hernández-Montiel, L.G.; Angulo, C. The potential use of Debaryomyces hansenii for the biological control of pathogenic fungi in food. Biol. Control 2018, 121, 216–222. [Google Scholar] [CrossRef]
- Berg, G.; Egamberdieva, D.; Lugtenberg, B.; Hagemann, M. Symbiotic plant–microbe interactions: Stress protection, plant growth promotion, and biocontrol by Stenotrophomonas. In Symbioses and Stress; Springer: Dordrecht, The Netherlands, 2010; pp. 445–460. [Google Scholar]
- Schmidt, C.S.; Alavi, M.; Cardinale, M.; Müller, H.; Berg, G. Stenotrophomonas rhizophila DSM14405T promotes plant growth probably by altering fungal communities in the rhizosphere. Biol. Fertil. Soils 2012, 48, 947–960. [Google Scholar]
- Nunes, C.A. Biological control of postharvest diseases of fruit. Eur. J. Plant Pathol. 2012, 133, 181–196. [Google Scholar] [CrossRef]
- Dukare, A.S.; Paul, S.; Nambi, V.E.; Gupta, R.K.; Singh, R.; Sharma, K.; Vishwakarma, R.K. Exploitation of microbial antagonists for the control of postharvest diseases of fruits: A review. Crit. Rev. Food Sci. Nutr. 2018, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hong, P.; Hao, W.; Luo, J.; Chen, S.; Hu, M.; Zhong, G. Combination of hot water, Bacillus amyloliquefaciens HF-01 and sodium bicarbonate treatments to control postharvest decay of mandarin fruit. Postharvest Biol. Technol. 2014, 88, 96–102. [Google Scholar] [CrossRef]
- Tang, J.; Liu, Y.; Li, H.; Wang, L.; Huang, K.; Chen, Z. Combining an antagonistic yeast with harpin treatment to control postharvest decay of kiwifruit. Biol. Control 2015, 89, 61–67. [Google Scholar] [CrossRef]
- Yu, T.; Yu, C.; Chen, F.; Sheng, K.; Zhou, T.; Zunun, M.; Abudu, O.; Yang, S.; Zheng, X. Integrated control of blue mold in pear fruit by combined application of chitosan, a biocontrol yeast and calcium chloride. Postharvest Biol. Technol. 2012, 69, 49–53. [Google Scholar] [CrossRef]
- Abouraïcha, E.; El Alaoui-Talibi, Z.; El Boutachfaiti, R.; Petit, E.; Courtois, B.; Courtois, J.; El Modafar, C. Induction of natural defense and protection against Penicillium expansum and Botrytis cinerea in apple fruit in response to bioelicitors isolated from green algae. Sci. Hort. 2015, 181, 121–128. [Google Scholar] [CrossRef]
- Stadnik, M.J.; Freitas, M.B.D. Algal polysaccharides as source of plant resistance inducers. Trop. Plant Pathol. 2014, 39, 111–118. [Google Scholar] [CrossRef]
- Siah, A.; Magnin-Robert, M.; Randoux, B.; Choma, C.; Rivière, C.; Halama, P.; Reignault, P. Natural agents inducing plant resistance against pests and diseases. In Natural Antimicrobial Agents; Merillon, J.M., Riviere, C., Eds.; Springer: Cham, Switzerland, 2018; ISBN 978-3-319-67045-4. [Google Scholar]
- Araujo, L.; Stadnik, M.J. Cultivar-specific and ulvan-induced resistance of apple plants to Glomerella leaf spot are associated with enhanced activity of peroxidases. Acta Sci. Agron. 2013, 35, 287–293. [Google Scholar] [CrossRef]
- de Freitas, M.B.; Stadnik, M.J. Ulvan-induced resistance in Arabidopsis thaliana against Alternaria brassicicola requires reactive oxygen species derived from NADPH oxidase. Physiol. Mol. Plant Pathol. 2015, 90, 49–56. [Google Scholar] [CrossRef]
- Cluzet, S.; Torregrosa, C.; Jacquet, C.; Lafitte, C.; Fournier, J.; Mercier, L.; Salamagne, S.; Briand, X.; Esquerre-Tugaye, M.T.; Dumas, B. Gene expression profiling and protection of Medicago truncatula against a fungal infection in response to an elicitor from green algae Ulva spp. Plant Cell Environ. 2004, 27, 917–928. [Google Scholar] [CrossRef]
- El Modafar, C.; Elgadda, M.; El Boutachfaiti, R.; Abouraicha, E.; Zehhar, N.; Petit, E.; El Alaoui-Talibi, Z.; Courtois, J. Induction of natural defense accompanied by salicylic acid-dependent systemic acquired resistance in tomato seedlings in response to bioelicitors isolated from green algae. Sci. Hort. 2012, 138, 55–63. [Google Scholar] [CrossRef]
- Rivas-Garcia, T.; Hernandez-Montiel, L.G.; Murillo-Amador, B.; Nieto-Garibay, A.; Chiquito-Contreras, R.G.; Rincon-Enriquez, G. Identification and characterization of Fusarium spp. from muskmelon in northwest Mexico. Biotecnia 2018, 20, 71–75. [Google Scholar]
- Zhou, Y.; Zhang, L.; Zeng, K. Efficacy of Pichia membranaefaciens combined with chitosan against Colletotrichum gloeosporioides in citrus fruits and possible modes of action. Biol. Control 2016, 96, 39–47. [Google Scholar] [CrossRef]
- Mattiuz, B.H.; Ducamp-Collin, M.N.; Mattiuz, C.F.M.; Vigneault, C.; Marques, K.M.; Sagoua, W.; Montet, D. Effect of propolis on postharvest control of anthracnose and quality parameters of ‘Kent’mango. Sci. Hort. 2015, 184, 160–168. [Google Scholar] [CrossRef]
- Levy, Y.; Benderly, M.; Cohen, Y.; Gisi, U.; Bassand, D. The joint action of fungicides in mixtures: Comparison of two methods for synergy calculation. EPPO Bulletin 1986, 16, 651–657. [Google Scholar] [CrossRef]
- Blackburn, D.; Shapiro-Ilan, D.I.; Adams, B.J. Biological control and nutrition: Food for thought. Biol. Control 2016, 97, 131–138. [Google Scholar] [CrossRef]
- Cao, S.; Yang, Z.; Hu, Z.; Zheng, Y. The effects of the combination of Pichia membranefaciens and BTH on controlling of blue mould decay caused by Penicillium expansum in peach fruit. Food Chem. 2011, 124, 991–996. [Google Scholar] [CrossRef]
- Sharma, R.R.; Singh, D.; Singh, R. Biological control of postharvest diseases of fruits and vegetables by microbial antagonists: A review. Biol. Control 2009, 50, 205–221. [Google Scholar] [CrossRef]
- Meng, X.H.; Qin, G.Z.; Tian, S.P. Influences of preharvest spraying Cryptococcus laurentii combined with postharvest chitosan coating on postharvest diseases and quality of table grapes in storage. LWT-Food Sci. Technol. 2010, 43, 596–601. [Google Scholar] [CrossRef]
- De Costa, D.M.; Gunawardhana, H.M.D.M. Effects of sodium bicarbonate on pathogenicity of Colletotrichum musae and potential for controlling postharvest diseases of banana. Postharvest Biol. Technol. 2012, 68, 54–63. [Google Scholar] [CrossRef]
- Araújo, L.; Gonçalves, A.E.; Stadnik, M.J. Ulvan effect on conidial germination and appressoria formation of Colletotrichum gloeosporioides. Phytoparasitica 2014, 42, 631–640. [Google Scholar] [CrossRef]
- Gonçalves, A.E.; Stadnik, M.J. Interferência de ulvana no desenvolvimento e melanização de apressórios de Colletotrichum gloeosporioides. Trop. Plant Pathol. 2012, 37, 431–437. [Google Scholar] [CrossRef]
- de Freitas, M.B.; Ferreira, L.G.; Hawerroth, C.; Duarte, M.E.R.; Noseda, M.D.; Stadnik, M.J. Ulvan induce resistance against plant pathogenic fungi independently of their sulfation degree. Carbohydr. Polym. 2015, 133, 384–390. [Google Scholar] [CrossRef] [PubMed]
- Paulert, R.; Talamini, V.; Cassolato, J.E.F.; Duarte, M.E.R.; Noseda, M.D.; Smania, A.; Stadnik, M.J. Effects of sulfated polysaccharide and alcoholic extracts from green seaweed Ulva fasciata on anthracnose severity and growth of common bean (Phaseolus vulgaris L.). J. Plant Dis. Protect. 2009, 116, 263–270. [Google Scholar] [CrossRef]
- Borsato, L.C.; Di Piero, R.M.; Stadnik, M.J. Mecanismos de defesa eliciados por ulvana contra Uromyces appendiculatus em três cultivares de feijoeiro. Trop. Plant Pathol. 2010, 35, 318–322. [Google Scholar] [CrossRef]
- Jaulneau, V.; Lafitte, C.; Jacquet, C.; Fournier, S.; Salamagne, S.; Briand, X.; Esquerre-Tugaye, M.T.; Dumas, B. Ulvan, a sulfated polysaccharide from green algae, activates plant immunity through the jasmonic acid signaling pathway. J. Biomed. Biotechnol. 2010. [Google Scholar] [CrossRef] [PubMed]
- Paulert, R.; Ebbinghaus, D.; Urlass, C.; Moerschbacher, B.M. Priming of the oxidative burst in rice and wheat cell cultures by ulvan, a polysaccharide from green macroalgae, and enhanced resistance against powdery mildew in wheat and barley plants. Plant Pathol. 2010, 59, 634–642. [Google Scholar] [CrossRef]
- Janisiewicz, W.J.; Korsten, L. Biological control of postharvest diseases of fruits. Annu. Rev. Phytopathol. 2002, 40, 411–441. [Google Scholar] [CrossRef] [PubMed]
- Grzegorczyk, M.; Żarowska, B.; Restuccia, C.; Cirvilleri, G. Postharvest biocontrol ability of killer yeasts against Monilinia fructigena and Monilinia fructicola on stone fruit. Food Microbiol. 2017, 61, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Rivas-Garcia, T.; Hernandez-Montiel, L.G.; Murillo-Amador, B.; Nieto-Garibay, A.; Chiquito-Contreras, R.G.; Rincon-Enriquez, G. Enhanced biocontrol of fruit rot on muskmelon by marine Debaryomyces hansenii and Stenotrophomonas rhizophila and their potential modes of action. Postharvest Biol. Technol. in press.
- Pech, J.C.; Bouzayen, M.; Latché, A. Climacteric fruit ripening: Ethylene-dependent and independent regulation of ripening pathways in melon fruit. Plant Sci. 2008, 175, 114–120. [Google Scholar] [CrossRef]
- Gupta, N.; Jain, S.K. Storage behavior of mango as affected by postharvest application of plant extracts and storage conditions. J. Food Sci. Technol. 2014, 51, 2499–2507. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Bruton, B.D.; Biles, C.L. Fusarium solani endo-polygalacturonase from decayed muskmelon fruit: Purification and characterization. Physiol. Mol. Plant Pathol. 1999, 54, 171–186. [Google Scholar] [CrossRef]
- Leslie, J.F.; Summerell, B.A. The Fusarium Laboratory Manual, 2nd ed.; Blackwell Pub.: Ames, IA, USA, 2006; p. 387. [Google Scholar]
| Treatments | Disease Control (%) | Ee | SF |
|---|---|---|---|
| D. hansenii | 28.6 ± 3.1 a * | - | - |
| S. rhizophila | 35.7 ± 2.8 b | - | - |
| ulvan | 14.3 ± 3.9 c | - | - |
| D. hansenii + ulvan | 57.1 ± 4.3 d | 38.8 | 1.5 |
| S. rhizophila + ulvan | 64.3 ± 4.1 e | 44.9 | 1.4 |
| Benomyl | 64.3a ± 2.4 e | - | - |
| Treatments | DI (%) | Weight Loss (gr) | Firmness (N) | TSS (%) | pH |
|---|---|---|---|---|---|
| D. hansenii | 33.3 ± 1.2 a * | 0.30 ± 0.02 a | 4.2 ± 0.5 a | 9.2 ± 0.08 ac | 6.5 ± 0.1 a |
| S. rhizophila | 26.7 ± 1.6 b | 0.30 ± 0.01 a | 4.2 ± 0.4 a | 9.2 ± 0.09 a | 6.5 ± 0.1 a |
| ulvan | 23.3 ± 0.8 c | 0.24 ± 0.03 a | 4.2 ± 0.4 a | 9.3 ± 0.06 b | 6.1 ± 0.1 b |
| D. hansenii + ulvan | 20.0 ± 1.2 c | 0.21 ± 0.03 a | 4.3 ± 0.3 b | 9.3 ± 0.08 bc | 6.2 ± 0.1 b |
| S. rhizophila + ulvan | 13.3 ± 0.7 d | 0.22 ± 0.02 a | 4.3 ± 0.3 b | 9.3 ± 0.06 b | 6.1 ± 0.1 b |
| Benomyl | 10.0 ± 0.4 d | 0.68 ± 0.05 b | 4.1 ± 0.8 c | 9.2 ± 0.08 a | 6.6 ± 0.1 c |
| Control (water) | 70.0 ± 1.4 e | 1.06 ± 0.08 c | 4.0 ± 0.6 d | 9.2 ± 0.07 a | 6.6 ± 0.08 c |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rivas-Garcia, T.; Murillo-Amador, B.; Nieto-Garibay, A.; Chiquito-Contreras, R.G.; Rincon-Enriquez, G.; Hernandez-Montiel, L.G. Effect of Ulvan on the Biocontrol Activity of Debaryomyces hansenii and Stenotrophomonas rhizophila against Fruit Rot of Cucumis melo L. Agronomy 2018, 8, 273. https://doi.org/10.3390/agronomy8120273
Rivas-Garcia T, Murillo-Amador B, Nieto-Garibay A, Chiquito-Contreras RG, Rincon-Enriquez G, Hernandez-Montiel LG. Effect of Ulvan on the Biocontrol Activity of Debaryomyces hansenii and Stenotrophomonas rhizophila against Fruit Rot of Cucumis melo L. Agronomy. 2018; 8(12):273. https://doi.org/10.3390/agronomy8120273
Chicago/Turabian StyleRivas-Garcia, Tomas, Bernardo Murillo-Amador, Alejandra Nieto-Garibay, Roberto G. Chiquito-Contreras, Gabriel Rincon-Enriquez, and Luis G. Hernandez-Montiel. 2018. "Effect of Ulvan on the Biocontrol Activity of Debaryomyces hansenii and Stenotrophomonas rhizophila against Fruit Rot of Cucumis melo L." Agronomy 8, no. 12: 273. https://doi.org/10.3390/agronomy8120273
APA StyleRivas-Garcia, T., Murillo-Amador, B., Nieto-Garibay, A., Chiquito-Contreras, R. G., Rincon-Enriquez, G., & Hernandez-Montiel, L. G. (2018). Effect of Ulvan on the Biocontrol Activity of Debaryomyces hansenii and Stenotrophomonas rhizophila against Fruit Rot of Cucumis melo L. Agronomy, 8(12), 273. https://doi.org/10.3390/agronomy8120273







