Effect of Water Deficit-Induced at Vegetative and Reproductive Stages on Protein and Oil Content in Soybean Grains
Abstract
:1. Introduction
2. Material and Methods
2.1. Experimental Design and Assay
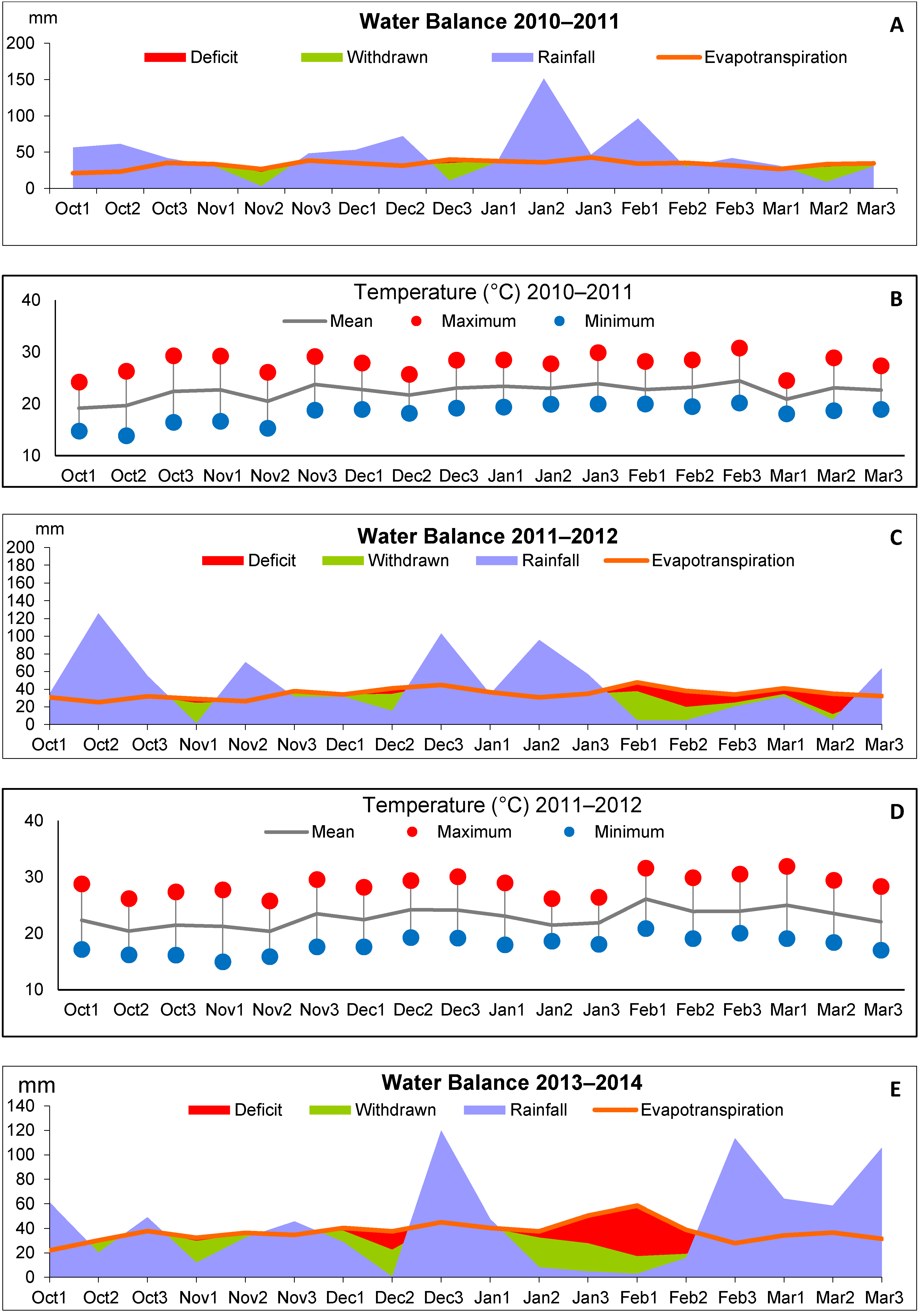
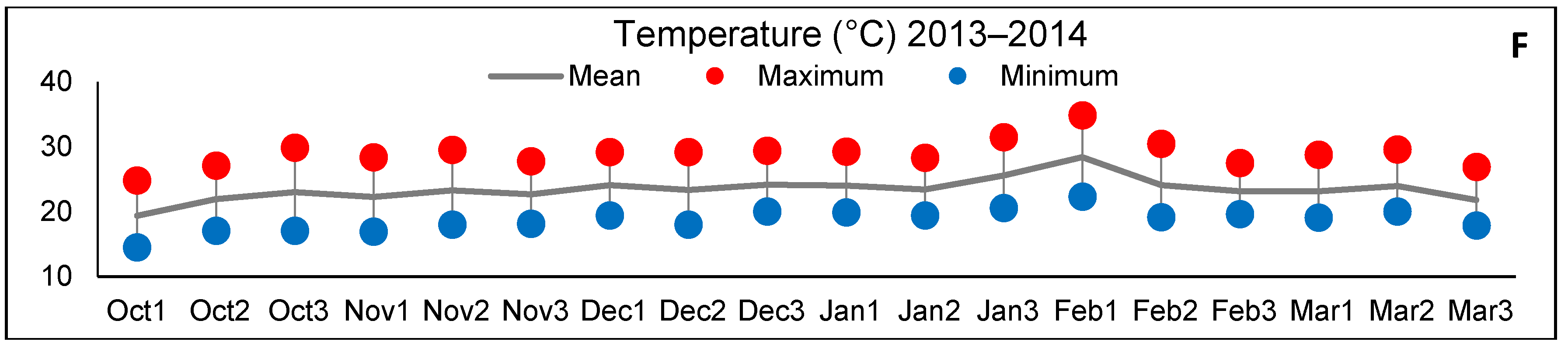
2.2. Control of Water Conditions
2.3. Traits
2.4. Statistical Analysis
3. Results and Discussion
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- United States Department of Agriculture (USDA)—World Agricultural Production. Available online: https://apps.fas.usda.gov/psdonline/circulars/production.pdf (accessed on 12 October 2017).
- CONAB. Acompanhamento da Safra Brasileira de Grãos Safra 2016/2017—Décimo Levantamento; CONAB: Brasília, Brazil, 2017; pp. 1–171.
- Wilson, R.F. Seed composition. In Soybeans: Improvement, Production, and Uses, 3rd ed.; Boerma, H.R., Specht, J.E., Eds.; ASA, CSSA, and SSSA: Madison, WI, USA, 2004; pp. 621–668. [Google Scholar]
- Kim, M.Y.; Van, K.; Kang, Y.J.; Kim, K.H.; Lee, S.H. Tracing soybean domestication history: From nucleotide to genome. Breed. Sci. 2012, 61, 445–452. [Google Scholar] [CrossRef] [PubMed]
- National Genetic Resources Program (NGRP)—Germplasm Resources Information Network (GRIN). Available online: https://www.ars-grin.gov/ (accessed on 12 October 2017).
- Pípolo, A.E.; Hungria, M.; Franchini, J.C.; Balbinot, A.A., Jr.; Debiasi, H.; Mandarino, J.M.G. Teores de Óleo e Proteína em Soja: Fatores Envolvidos e Qualidade Para a Indústria—Comunicado Técnico 86; Embrapa Soja: Londrina, Brazil, 2015; pp. 1–15. ISSN 2176-2889. [Google Scholar]
- Wang, J.; Chen, P.; Wrang, D.; Shannon, G.; Zeng, A.; Orazaly, M.; Wu, C. Identification and mapping of stable QTL for protein content in soybean seeds. Mol. Breed. 2015, 35, 92. [Google Scholar] [CrossRef]
- Piper, E.L.; Boote, K.I. Temperature and cultivar effects on soybean seed oil and protein concentration. J. Am. Oil Chem. Soc. 1999, 76, 1233–1241. [Google Scholar] [CrossRef]
- Pípolo, A.E.; Sinclair, T.R.; Camara, G.M.S. Protein and oil concentration of soybean seed cultured in vitro using nutrient solutions of differing glutamine concentration. Ann. Appl. Biol. 2004, 144, 223–227. [Google Scholar] [CrossRef]
- Thakur, M.; Hurburgh, C.R. Quality of US soybean meal compared to the quality of soybean meal from other origins. J. Am. Oil Chem. Soc. 2007, 84, 835–843. [Google Scholar] [CrossRef]
- Dornbos, D.L., Jr.; Mullen, R.E. Soybean seed protein and oil contents and fatty acid composition adjustments by drought and temperature. J. Am. Oil Chem. Soc. 1992, 69, 228–231. [Google Scholar] [CrossRef]
- Food and Agriculture Organization of the United Nations (FAO)—The State of Food and Agriculture. Available online: http://www.fao.org/publications/sofa/2016/en/ (accessed on 13 October 2017).
- Ku, Y.S.; Au-Yeung, W.K.; Yung, Y.L.; Li, M.W.; Wen, C.Q.; Liu, X.; Lam, H.M. Drought stress and tolerance in soybean. In A Comprehensive Survey of International Soybean Research—Genetics, Physiology, Agronomy and Nitrogen Relationships; Board, J.E., Ed.; InTech: Rijeka, Croatia, 2013; pp. 209–237. [Google Scholar] [CrossRef]
- Thornthwaite, C.W.; Mather, J.R. The Water Balance, 1st ed.; Drexel Institute of Technology—Laboratory of Climatology: Centerton, AR, USA, 1955; pp. 1–104. [Google Scholar]
- Crusiol, L.G.T.; Carvalho, J.F.C.; Sibaldelli, R.N.R.; Neiverth, W.; Rio, A.; Ferreira, L.C.; Procópio, S.O.; Mertz-Henning, L.M.; Nepomuceno, A.L.; Neumaier, N.; et al. NDVI variation according to the time of measurement, sampling size, positioning of sensor and water regime in different soybean cultivars. Precis. Agric. 2017, 4, 470–490. [Google Scholar] [CrossRef]
- Embrapa Soja. Tecnologias de Produção de Soja—Região Central do Brasil 2012 e 2013—Sistemas de Produção 15; Embrapa Soja: Londrina, Brazil, 2011; pp. 1–261. ISSN 2176-2902. [Google Scholar]
- Heil, C. Rapid, Multi-Component Analysis of Soybeans by FT-NIR Spectroscopy; Thermo Fisher Scientific: Madison, WI, USA, 2010; Available online: https://tools.thermofisher.com/content/sfs/brochures/AN-51954-Rapid-Multi-Component-Analysis-Soybeans-AN51954-EN.pdf (accessed on 20 November 2017).
- Kunert, K.J.; Vorster, B.J.; Fenta, B.A.; Kibido, T.; Dionisio, G.; Foyer, C.H. Drought stress responses in soybean roots and nodules. Front. Plant Sci. 2016, 7, 1015. [Google Scholar] [CrossRef] [PubMed]
- Saitoh, K.; Mahmood, T.; Kuroda, T. Effect of moisture stress at different growth stages on flowering and pod set in determinate and indeterminate soybean cultivars. Jpn. J. Crop Sci. 1999, 68, 537–544. [Google Scholar] [CrossRef]
- Oya, T.; Nepomuceno, A.L.; Neumaier, N.; Farias, J.R.B.; Tobita, S.; Ito, O. Drought tolerance characteristics of Brazilian soybean cultivars—Evaluation and characterization of drought tolerance of various Brazilian soybean cultivars in the field. Plant Prod. Sci. 2004, 7, 129–137. [Google Scholar] [CrossRef]
- Wilson, E.W.; Rowntree, S.C.; Suhre, J.J.; Weidenbenner, N.H.; Conley, S.P.; Davis, V.M.; Diers, B.W.; Esker, P.D.; Naeve, S.L.; Specht, J.E.; et al. Genetic gain x management interaction in soybean: II. Nitrogen utilization. Crop Sci. 2014, 54, 340–348. [Google Scholar] [CrossRef]
- Lorini, I. Qualidade de Sementes e Grãos Comerciais de Soja no Brasil—Safra 2014/2015—Documentos 378; Embrapa Soja: Londrina, Brazil, 2016; pp. 1–190. [Google Scholar]
- Foroud, N.; Miindel, H.H.; Saindon, G.; Entz, T. Effect of level and timing of moisture stress on soybean yield, protein, and oil responses. Field Crops Res. 1993, 31, 195–209. [Google Scholar] [CrossRef]
- Ghassemi-Golezani, K.; Lotfi, R. Influence of water stress and pod position on oil and protein accumulation in soybean grains. Int. J. Agron. Plant Prod. 2013, 4, 2341–2345. [Google Scholar]
- Angra, S.; Kaur, S.; Singh, K.; Pathania, D.; Kaur, N.; Sharma, S.; Nayyar, H. Water-deficit stress during seed filling in contrasting soybean genotypes: Association of stress sensitivity with profiles of osmolytes and antioxidants. Int. J. Agric. Res. 2010, 5, 328–345. [Google Scholar] [CrossRef]
- Hajheidari, M.; Abdollahian-Noghabi, M.; Askari, H.; Heidari, M.; Sadeghian, S.Y.; Ober, E.S.; Salekdeh, G.H. Proteome analysis of sugar beet leaves under drought stress. Proteomics 2005, 5, 950–960. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, J.N.; Nelson, R.L.; Song, Q.; Cregan, P.B.; Li, Z. The genetic architecture of seed composition in soybean is refined by genome-wide association scans across multiple populations. G3 (Bethesda) 2014, 4, 2283–2294. [Google Scholar] [CrossRef] [PubMed]
- Eskandari, M.; Cober, E.R.; Rajcan, I. Genetic control of soybean seed oil: II. QTL and genes that increase oil concentration without decreasing protein or with increased seed yield. Theor. Appl. Genet. 2013, 126, 1677–1687. [Google Scholar] [CrossRef] [PubMed]
- Akinci, Ş.; Lösel, D.M. Plant water-stress response mechanisms. In Environmental Sciences—Water Stress; Rahman, I.M.M., Hasegawa, H., Eds.; InTech: Rijeka, Croatia, 2012; pp. 15–42. [Google Scholar] [CrossRef]
- Masclaux-Daubresse, C.; Daniel-Vedele, F.; Dechorgnat, J.; Chardon, F.; Gaufichon, L.; Suzuki, A. Nitrogen uptake, assimilation and remobilization in plants: Challenges for sustainable and productive agriculture. Ann. Bot. 2010, 105, 1141–1157. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, R.N.; Ligterink, W.; França-Neto, J.B.; Hilhorst, H.W.M.; Silva, E.A.A. Gene expression profiling of the green seed problem in soybean. BMC Plant Biol. 2016, 16, 37. [Google Scholar] [CrossRef] [PubMed]
- Mandarino, J.M.G. Grãos Verdes: Influência na Qualidade dos Produtos à Base de Soja—Série Sementes—Circular Técnica 90; Embrapa Soja: Londrina, Brazil, 2012; pp. 1–5. [Google Scholar]

| Yield (kg/ha) | |||||
|---|---|---|---|---|---|
| 2010–2011 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 3527.18 Ab | 3300.33 Ab | 3116.53 Ab | 4197.91 Aa | 3535.5 A |
| RF | 3187.79 Ab | 3376.41 Ab | 2994.32 Ab | 4178.77 Aa | 3434.3 A |
| VS | 3492.06 Aa | 2853.34 Ab | 2715.51 Ab | 3048.44 Bab | 3027.3 B |
| RS | 1843.69 Bab | 1679.42 Bab | 1359.06 Bb | 1982.47 Ca | 1716.2 C |
| 2011–2012 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 3612.06 Aa | 3185.5 Aab | 3006.43 Ab | 3282.58 Aab | 3271.6 A |
| RF | 3449.23 Aa | 3059.63 Aab | 2992.97 Aab | 2835.72 Ab | 3084.4 A |
| VS | 1937.67 Bab | 1490.25 Bb | 1825 Bab | 1978.98 Ba | 1807.0 B |
| RS | 887.02 Cns | 568.3 C | 504.12 C | 537.81 C | 624.3 C |
| 2013–2014 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 3278.13 Aa | 2929.31 Aab | 2568.13 Ab | 2515.45 Ab | 2822.8 A |
| RF | 2169.68 Bns | 1938.75 B | 1868.04 B | 1902.01 B | 1969.6 B |
| VS | 2126.33 Ba | 1439.66 Bb | 1635.96 Bb | 1534.2 Bb | 1684.0 C |
| RS | 1252.61 Ca | 766.21 Cab | 724.38 Cb | 819.61 Cab | 890.7 D |
| HSW (g) | |||||
|---|---|---|---|---|---|
| 2010–2011 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 12.41 Ab | 13.97 Ab | 13.48 Ab | 15.82 Aa | 13.92 A |
| RF | 12.33 Ac | 14.55 Ab | 13.07 Abc | 16.72 Aa | 14.17 A |
| VS | 12.08 ABC | 15.49 Aab | 14.02 Ac | 15.8 Aa | 14.35 A |
| RS | 10.36 Bbc | 12.03 Bab | 9.66 Bc | 12.48 Ba | 11.13 B |
| 2011–2012 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 13.37 Ac | 15.24 Ab | 15.8 Aab | 17.08 Aa | 15.37 A |
| RF | 11.53 Abb | 14.09 Aa | 15.45 Aa | 14.76 Ba | 13.96 B |
| VS | 10.56 Bb | 10.24 Bb | 10.44 Bb | 12.52 Ca | 10.94 C |
| RS | 10.12 Bab | 9.91 Bb | 11.64 Ba | 10.67 Cab | 10.58 C |
| 2013–2014 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 10.96 Ans | 12.2 A | 11.37 A | 11.38 A | 11.48 A |
| RF | 8.92 Bns | 9.61 BC | 9.87 A | 9.55 A | 9.49 B |
| VS | 9.58 Abns | 9.98 B | 9.69 A | 10.07 A | 9.83 B |
| RS | 8.16 Bns | 7.78 C | 6.99 B | 7.64 B | 7.64 C |
| NP | NS | SDM (g) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| WC | Y1 | Y2 | Y3 | Y1 | Y2 | Y3 | Y1 | Y2 | Y3 |
| I | 41.1 Aab | 36.3 Ab | 41.5 Aa | 76.4 Aab | 70.5 Ab | 84.4 Aa | 10.5 Aa | 10.3 Aa | 9.9 Aa |
| RF | 44 Aa | 32.7 Ab | 36.6 Ab | 79 Aa | 63.5 Ab | 70.3 Bab | 10.8 Aa | 8.2 Bb | 6.7 Bc |
| VS | 34.7 Ba | 25.8 Bb | 27.4 Bb | 69.6 Aa | 48.4 Bb | 53.9 Cb | 9.8 Aa | 4.9 Cb | 5.5 Bb |
| RS | 21 Ca | 11.5 Cb | 22.1 Ba | 36.8 Ba | 21.6 Cb | 35.7 Da | 4.3 Ba | 2 Db | 2.7 Cb |
| Protein (%) | |||||
|---|---|---|---|---|---|
| 2010–2011 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 34.93 BCc | 36.77 Abab | 35.7 Bbc | 37.4 Aa | 36.62 B |
| RF | 34.18 Cc | 35.84 Bc | 35.29 Bbc | 37.59 Aa | 36.24 B |
| VS | 36.55 Abb | 37.34 Aba | 37.91 Aab | 38.66 Aa | 37.97 A |
| RS | 37.55 Ans | 37.55 A | 37.41 A | 37.26 A | 37.41 A |
| 2011–2012 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 34.33 Ac | 37.32 Bab | 36.29 Bb | 37.98 Aa | 36.48 BC |
| RF | 35.36 Bb | 37.69 Aba | 36.46 Bab | 37.21 Aba | 36.68 B |
| VS | 34.97 Bb | 36.73 Ba | 36.14 Bab | 36.03 Bab | 35.97 C |
| RS | 39.18 Ans | 39.08 A | 38.6 A | 38.53 A | 38.85 A |
| 2013–2014 | |||||
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Mean |
| I | 36.34 Bns | 37.55 B | 37.41 A | 37.07 B | 37.09 C |
| RF | 36.78 Bb | 38.17 Ba | 37.29 Aab | 38.35 Aba | 37.65 BC |
| VS | 36.56 Bb | 38.87 Ba | 37.99 Aa | 38.29 Aba | 37.93 B |
| RS | 39.78 Aab | 40.63 Aa | 38.59 Ab | 39.77 Aab | 39.69 A |
| Oil (%) | AHI | |||||||
|---|---|---|---|---|---|---|---|---|
| WC | Embrapa 48 | BR 16 | P58 | P2193 | Embrapa 48 | BR 16 | P58 | P2193 |
| I | 22.57 Aa | 21.58 Ab | 21.96 Ab | 21.81 Ab | 0.44 Bb | 0.47 Bab | 0.46 Bab | 0.48 Aba |
| RF | 22.19 Ans | 21.72 A | 21.94 A | 21.73 AB | 0.45 Bns | 0.46 B | 0.47 B | 0.46 B |
| VS | 22.23 Aa | 21.61 Ab | 21.81 Aab | 22.24 Aa | 0.52 Ans | 0.5 A | 0.51 A | 0.51 A |
| RS | 20.87 Bab | 20.63 Bb | 21 Bab | 21.23 Ba | 0.37 Ca | 0.33 Cb | 0.32 Cb | 0.33 Cb |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mertz-Henning, L.M.; Ferreira, L.C.; Henning, F.A.; Mandarino, J.M.G.; Santos, E.D.; Oliveira, M.C.N.D.; Nepomuceno, A.L.; Farias, J.R.B.; Neumaier, N. Effect of Water Deficit-Induced at Vegetative and Reproductive Stages on Protein and Oil Content in Soybean Grains. Agronomy 2018, 8, 3. https://doi.org/10.3390/agronomy8010003
Mertz-Henning LM, Ferreira LC, Henning FA, Mandarino JMG, Santos ED, Oliveira MCND, Nepomuceno AL, Farias JRB, Neumaier N. Effect of Water Deficit-Induced at Vegetative and Reproductive Stages on Protein and Oil Content in Soybean Grains. Agronomy. 2018; 8(1):3. https://doi.org/10.3390/agronomy8010003
Chicago/Turabian StyleMertz-Henning, Liliane M., Leonardo C. Ferreira, Fernando A. Henning, José M. G. Mandarino, Elizeu D. Santos, Maria C. N. D. Oliveira, Alexandre L. Nepomuceno, José R. B. Farias, and Norman Neumaier. 2018. "Effect of Water Deficit-Induced at Vegetative and Reproductive Stages on Protein and Oil Content in Soybean Grains" Agronomy 8, no. 1: 3. https://doi.org/10.3390/agronomy8010003
APA StyleMertz-Henning, L. M., Ferreira, L. C., Henning, F. A., Mandarino, J. M. G., Santos, E. D., Oliveira, M. C. N. D., Nepomuceno, A. L., Farias, J. R. B., & Neumaier, N. (2018). Effect of Water Deficit-Induced at Vegetative and Reproductive Stages on Protein and Oil Content in Soybean Grains. Agronomy, 8(1), 3. https://doi.org/10.3390/agronomy8010003




