Transferring a Biomass Enhancement Biotechnology from Glasshouse to Field: A Case Study on Wheat GWD RNAi
Abstract
1. Introduction
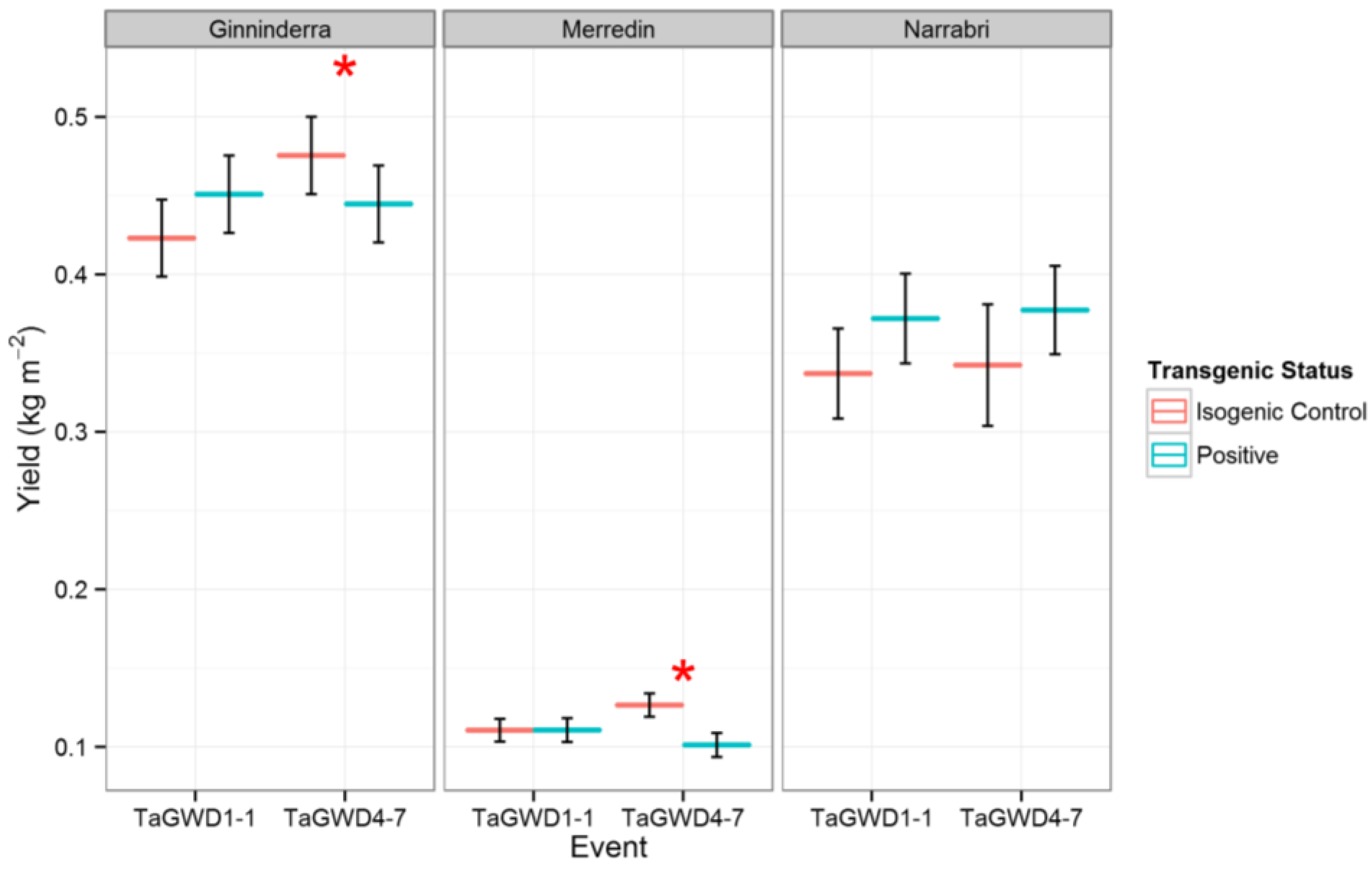
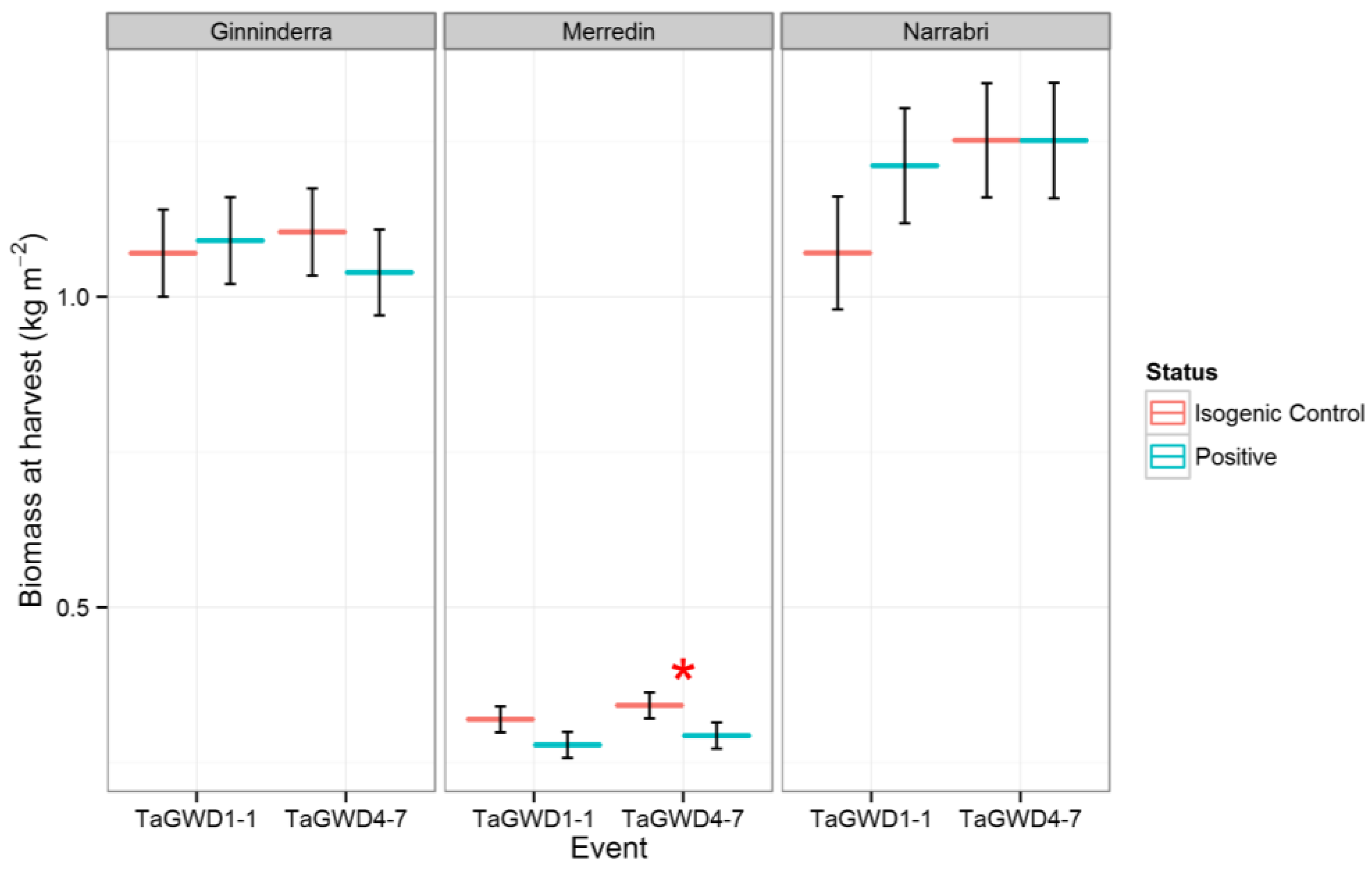
2. Results and Discussion
3. Experimental Section
3.1. Field Design
3.2. Measurements
3.3. Statistical Analysis
Supplementary Files
Supplementary File 1Acknowledgments
Author Contributions
Conflicts of Interest
Appendix A
| Trait (Units) | Event | Site | Negative Segregant | GWD RNAi | ||
|---|---|---|---|---|---|---|
| Mean | SE | Mean | SE | |||
| TGW (g) | G1.1 | GES | 40.05 | 2.55 | 35.06 | 2.56 |
| TGW (g) | G4.7 | GES | 40.12 | 2.54 | 36.63 | 2.51 |
| TGW (g) | G1.1 | Merredin | 44.95 | 0.60 | 45.33 | 0.60 |
| TGW (g) * | G4.7 | Merredin | 43.20 | 0.60 | 45.36 | 0.60 |
| TGW (g) | G1.1 | Narrabri | 42.90 | 0.73 | 43.45 | 0.73 |
| TGW (g) | G4.7 | Narrabri | 42.71 | 0.73 | 43.48 | 0.73 |
| HC biomass (g) | G1.1 | GES | 236.38 | 20.04 | 212.43 | 19.99 |
| HC biomass (g) | G4.7 | GES | 248.63 | 20.07 | 224.68 | 26.96 |
| HC biomass (g) | G1.1 | Merredin | 319.75 | 21.03 | 278.55 | 21.03 |
| HC biomass (g) * | G4.7 | Merredin | 342.18 | 21.03 | 293.59 | 21.03 |
| HC biomass (g) | G1.1 | Narrabri | 303.77 | 25.27 | 320.78 | 40.72 |
| HC biomass (g) | G4.7 | Narrabri | 303.77 | 25.27 | 339.1 | 25.3 |
| Yield/spike (g) | G1.1 | GES | 1.59 | 0.06 | 1.54 | 0.06 |
| Yield/spike (g) | G4.7 | GES | 1.58 | 0.06 | 1.52 | 0.1 |
| Yield/spike (g) | G1.1 | Merredin | 1.42 | 0.06 | 1.38 | 0.06 |
| Yield/spike (g) | G4.7 | Merredin | 1.33 | 0.06 | 1.45 | 0.06 |
| Yield/spike (g) | G1.1 | Narrabri | 1.53 | 0.1 | 1.65 | 0.1 |
| Yield/spike (g) | G4.7 | Narrabri | 1.62 | 0.1 | 1.77 | 0.11 |
| Leaf area (cm2) * | G1.1 | Narrabri | 48.25 | 2.98 | 57.87 | 2.97 |
| Leaf area (cm2) * | G4.7 | Narrabri | 49.99 | 2.97 | 58.27 | 2.94 |
| Rachis nodes * | G1.1 | GES | 18.26 | 0.24 | 19.32 | 0.24 |
| Rachis nodes | G4.7 | GES | 18.33 | 0.24 | 18.67 | 0.24 |
| NDVI (Z14) | G1.1 | Narrabri | 0.38 | 0.06 | 0.42 | 0.06 |
| NDVI (Z14) | G4.7 | Narrabri | 0.44 | 0.06 | 0.37 | 0.06 |
| Field Trial Characteristics | Narrabri (NSW) | Ginnindera (NSW) | Merredin (WA) |
|---|---|---|---|
| Sowing density (seeds per m2) | 150 | 170 | 100 |
| Plot size (m2) | 10 | 10 | 8 |
| Nitrogen application (kg·Ha−1) | 40 (sowing) | 15 (sowing) | 7.5 (sowing) |
| 40 (anthesis) | 75 (anthesis) | 7.5 (anthesis) | |
| Planting date | 16/05/12 | 11/06/12 | 14/06/12 |
| Emergence | 26/05/12 | 26/06/12 | 07/07/12 |
| Anthesis | 11/10/12 | 10/11/12 | 07/09/12 |
| Harvest | 27/11/12 | 15/01/13 | 08/11/12 |
References
- Tanno, K.-I.; Willcox, G. How fast was wild wheat domesticated? Science 2006, 311, 1886. [Google Scholar] [CrossRef] [PubMed]
- Godfray, H.C.J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The Challenge of Feeding 9 Billion People. Science 2010, 327, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Millet, E.; Rong, J.K.; Qualset, C.O.; Mcguire, P.E.; Bernard, M.; Sourdille, P.; Feldman, M. Grain yield and grain protein percentage of common wheat lines with wild emmer chromosome-arm substitutions. Euphytica 2014, 195, 69–81. [Google Scholar] [CrossRef]
- Yang, Z.; Bai, Z.; Li, X.; Wang, P.; Wu, Q.; Yang, L.; Li, L.; Li, X. SNP identification and allelic-specific PCR markers development for TaGW2, a gene linked to wheat kernel weight. Theor. Appl. Genet. 2012, 125, 1057–1068. [Google Scholar] [CrossRef] [PubMed]
- Uauy, C. Wheat genomics comes of age. Curr. Opin. Plant Biol. 2017, 36, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Whan, A.; Dielen, A.S.; Mieog, J.; Bowerman, A.F.; Robinson, H.M.; Byrne, K.; Colgrave, M.; Larkin, P.J.; Howitt, C.A.; Morell, M.K.; et al. Engineering alpha-amylase levels in wheat grain suggests a highly sophisticated level of carbohydrate regulation during development. J. Exp. Bot. 2014, 65, 5443–5457. [Google Scholar] [CrossRef] [PubMed]
- Bowerman, A.F.; Newberry, M.; Dielen, A.; Whan, A.; Larroque, O.; Pritchard, J.; Gubler, F.; Howitt, C.A.; Pogson, B.J.; Morell1, M.K.; et al. Suppression of glucan, water dikinase in the endosperm alters wheat grain properties, germination and coleoptile growth. Plant Biotechnol. J. 2016, 14, 398–408. [Google Scholar] [CrossRef] [PubMed]
- Smidansky, E.D.; Clancy, M.; Meyer, F.D.; Lanning, S.P.; Blake, N.K.; Talbert, L.E.; Giroux, M.J. Enhanced ADP-glucose pyrophosphorylase activity in wheat endosperm increases seed yield. Proc. Natl. Acad. Sci. USA 2002, 99, 1724–1729. [Google Scholar] [CrossRef] [PubMed]
- Smidansky, E.D.; Meyer, F.D.; Blakeslee, B.; Weglarz, T.E.; Greene, T.W.; Giroux, M.J. Expression of a modified ADP-glucose pyrophosphorylase large subunit in wheat seeds stimulates photosynthesis and carbon metabolism. Planta 2007, 225, 965–976. [Google Scholar] [CrossRef] [PubMed]
- Tuncel, A.; Okita, T.W. Improving starch yield in cereals by over-expression of ADPglucose pyrophosphorylase: Expectations and unanticipated outcomes. Plant Sci. Int. J. Exp. Plant Biol. 2013, 211, 52–60. [Google Scholar] [CrossRef] [PubMed]
- Kang, G.; Liu, G.; Peng, X.; Wei, L.; Wang, C.; Zhu, Y.; Ma, Y.; Jiang, Y.; Guo, T. Increasing the starch content and grain weight of common wheat by overexpression of the cytosolic AGPase large subunit gene. Plant Physiol. Biochem. PPB 2013, 73, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Ral, J.-P.; Bowerman, A.F.; Li, Z.; Sirault, X.; Furbank, R.; Pritchard, J.R.; Bloemsma, M.; Cavanagh, C.R.; Howitt, C.A.; Morell, M.K. Down-regulation of Glucan, Water-Dikinase activity in wheat endosperm increases vegetative biomass and yield. Plant Biotechnol. J. 2012, 10, 871–882. [Google Scholar] [CrossRef] [PubMed]
- Ritte, G.; Heydenreich, M.; Mahlow, S.; Haebel, S.; Kotting, O.; Steup, M. Phosphorylation of C6- and C3-positions of glucosyl residues in starch is catalysed by distinct dikinases. FEBS Lett. 2006, 580, 4872–4876. [Google Scholar] [CrossRef] [PubMed]
- Ritte, G.; Scharf, A.; Eckermann, N.; Haebel, S.; Steup, M. Phosphorylation of transitory starch is increased during degradation. Plant Physiol. 2004, 135, 2068–2077. [Google Scholar] [CrossRef] [PubMed]
- Streb, S.; Eicke, S.; Zeeman, S.C. The simultaneous abolition of three starch hydrolases blocks transient starch breakdown in Arabidopsis. J. Biol. Chem. 2012, 287, 41745–41756. [Google Scholar] [CrossRef] [PubMed]
- Blennow, A.; Engelsen, S.B. Helix-breaking news: Fighting crystalline starch energy deposits in the cell. Trends Plant Sci. 2010, 15, 236–240. [Google Scholar] [CrossRef] [PubMed]
- Smith, A.M.; Zeeman, S.C.; Smith, S.M. Starch degradation. Annu. Rev. Plant Biol. 2005, 56, 73–98. [Google Scholar] [CrossRef] [PubMed]
- MacNeill, G.J.; Mehrpouyan, S.; Minow, M.A.A.; Patterson, J.A.; Tetlow, I.J.; Emes, M.J. Starch as a source, starch as a sink: The bifunctional role of starch in carbon allocation. J. Exp. Bot. 2047, 68, 4433–4453. [Google Scholar] [CrossRef] [PubMed]
- Zeller, S.L.; Kalinina, O.; Brunner, S.; Keller, B.; Schmid, B. Transgene × Environment Interactions in Genetically Modified Wheat. PLoS ONE 2010, 5, e11405. [Google Scholar] [CrossRef] [PubMed]


© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Whan, A.P.; Verbyla, A.P.; Mieog, J.C.; Howitt, C.A.; Ral, J.-P. Transferring a Biomass Enhancement Biotechnology from Glasshouse to Field: A Case Study on Wheat GWD RNAi. Agronomy 2017, 7, 82. https://doi.org/10.3390/agronomy7040082
Whan AP, Verbyla AP, Mieog JC, Howitt CA, Ral J-P. Transferring a Biomass Enhancement Biotechnology from Glasshouse to Field: A Case Study on Wheat GWD RNAi. Agronomy. 2017; 7(4):82. https://doi.org/10.3390/agronomy7040082
Chicago/Turabian StyleWhan, Alex P., Arunas P. Verbyla, Jos C. Mieog, Crispin A. Howitt, and Jean-Philippe Ral. 2017. "Transferring a Biomass Enhancement Biotechnology from Glasshouse to Field: A Case Study on Wheat GWD RNAi" Agronomy 7, no. 4: 82. https://doi.org/10.3390/agronomy7040082
APA StyleWhan, A. P., Verbyla, A. P., Mieog, J. C., Howitt, C. A., & Ral, J.-P. (2017). Transferring a Biomass Enhancement Biotechnology from Glasshouse to Field: A Case Study on Wheat GWD RNAi. Agronomy, 7(4), 82. https://doi.org/10.3390/agronomy7040082






