Variation for Concentrations of Various Phytoestrogens and Agronomic Traits Among a Broad Range of Red Clover (Trifolium pratense) Cultivars and Accessions
Abstract
:1. Introduction
2. Results
2.1 Range and Variance of Agronomic Traits
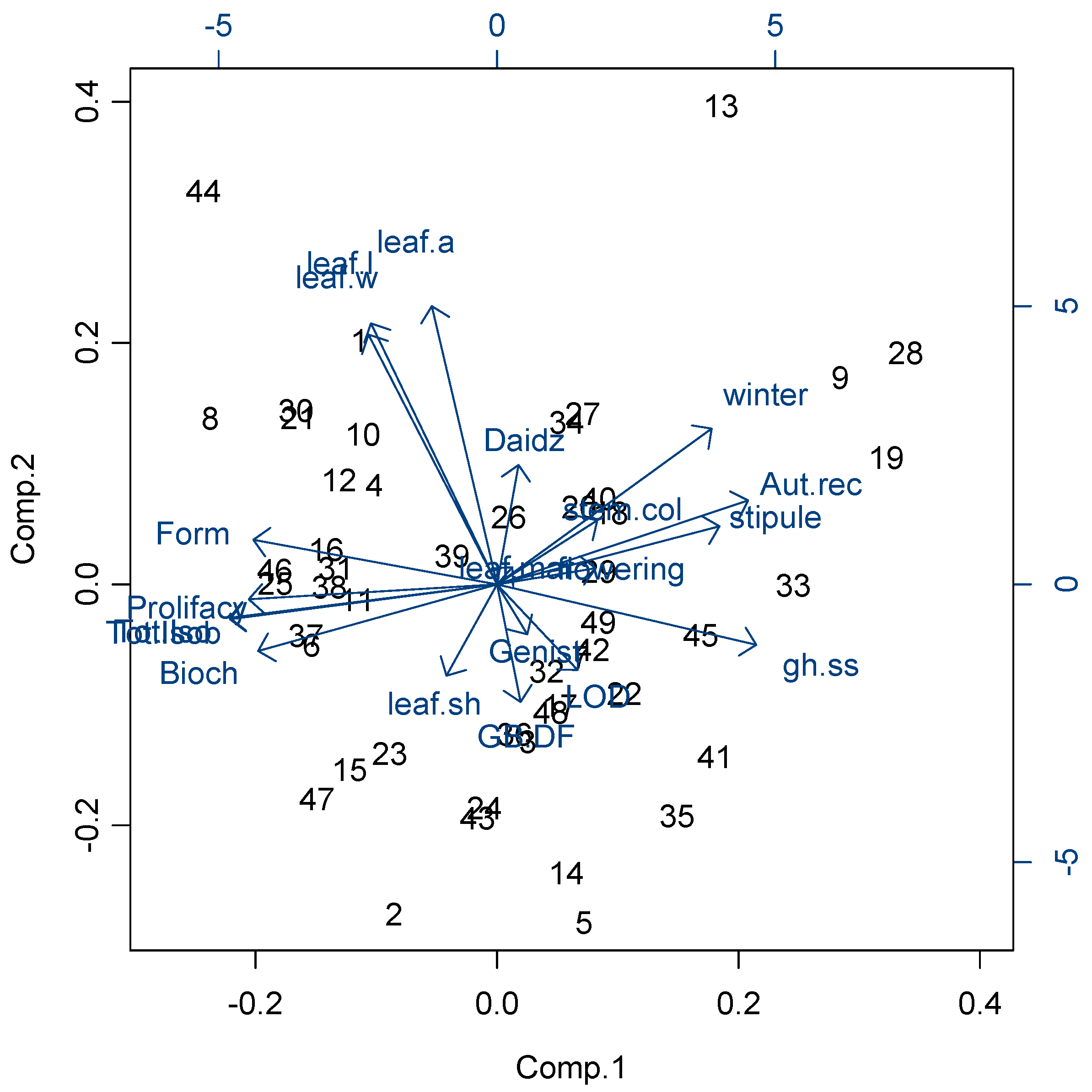
2.2 Principal Component Analysis of Phenotypic and Biochemical Variance among Red Clover Accessions
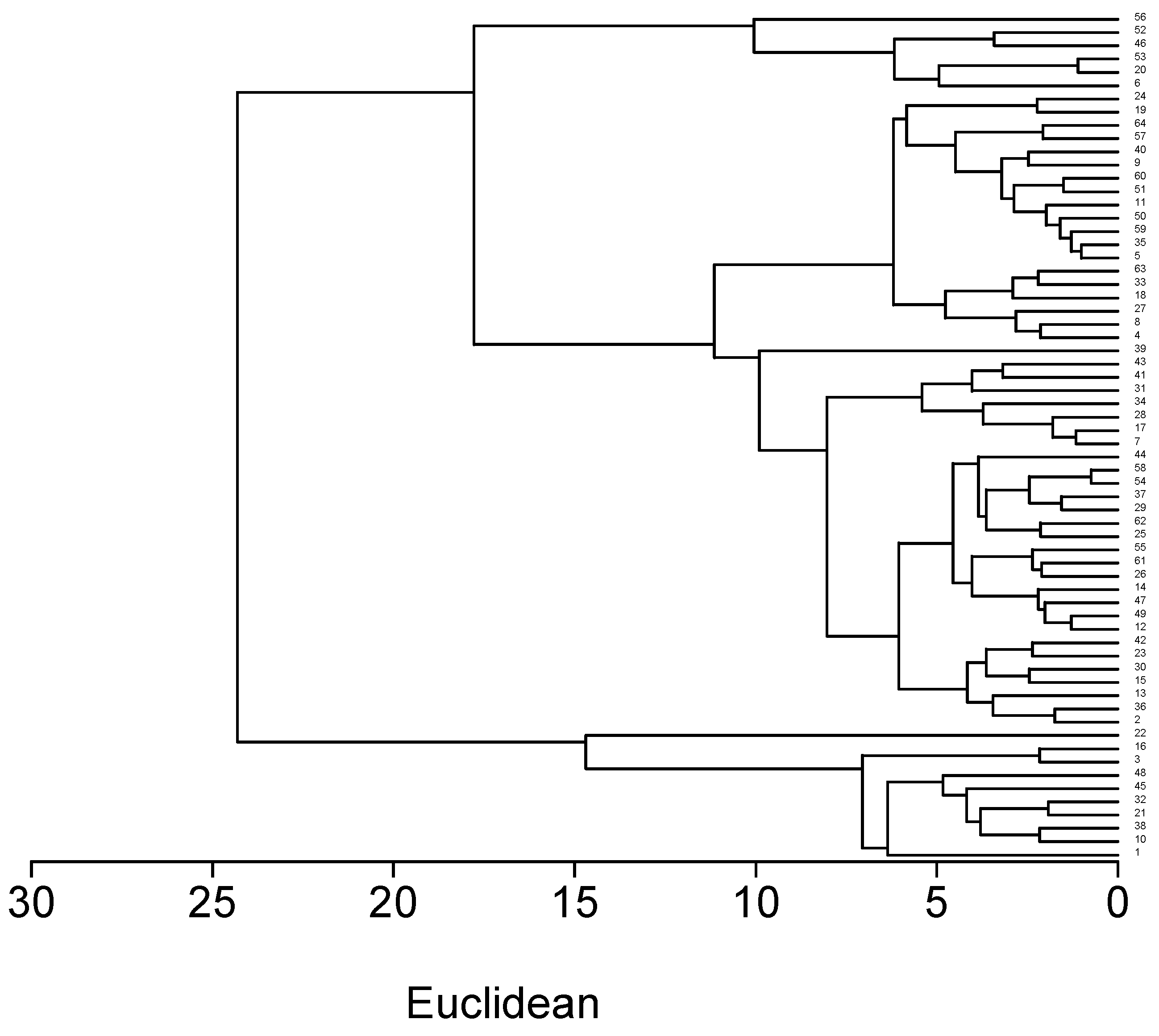
2.3. Cluster Analysis of Red Clover Accessions
3. Discussion
4. Materials and Methods
4.1. Germplasm
4.2. Establishment
4.3. Trial Design
4.4. Plant Measurements
4.5. Biochemical Analysis
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Smith, R.S. Red Clover (Trifolium pratense). In Alternative Pasture Legumes 1993, Proceedings of the 2nd National Alternative Pasture Legumes Workshop, Coonawarra, Australia, 25–28 July 1993; Michalk, D.L., Craig, A.D., Collins, W.J., Eds.; South Australian Research and Development Institute: Urrbrae, Australia, 1994. [Google Scholar]
- Fergus, E.N.; Hollowell, E.A. Red Clover. Adv. Agron. 1960, 12, 365–436. [Google Scholar]
- Clark, S.G.; Reed, K.F.M. Performance of Grassland Hamua × Moroccan red clover crosses in a marginal environment. In Proceedings of the 4th Australian Society of Agronomy Conference, Melbourne, Australia, August 1987; Australian Society of Agrononoy: Melbourne, Australia, 1987. [Google Scholar]
- Moot, D.J. An overview of dryland legume research in New Zealand. Crop Pasture Sci. 2012, 63, 726–733. [Google Scholar] [CrossRef]
- Nichols, P.G.H.; Revell, C.K.; Humphries, A.W.; Howie, J.H.; Hall, E.J.; Sandral, G.A.; Ghamkhar, K.; Harris, C.A. Temperate pasture legumes in Australia—Their history, current use, and future prospects. Crop Pasture Sci. 2012, 63, 691–725. [Google Scholar] [CrossRef]
- Kenny, P.T.; Reed, K.F.M. Effects of pasture type on the growth and wool production of weaner sheep during summer and autumn. Aust. J. Exp. Agric. Anim. Husb. 1984, 24, 322–331. [Google Scholar] [CrossRef]
- McDonald, M.F. Effects of plant oestrogens in ruminants. Proc. Nutr. Soc. 1995, NZ 20, 43–51. [Google Scholar] [PubMed]
- Oram, R.N. Register of Australian Herbage Plant Cultivars; CSIRO: Melbourne, Australia, 1990. [Google Scholar]
- IP Australia. Plant Breeders Rights Database. IP Australia: Australian Government: Canberra. Available online: www.ipaustralia.gov.au/get-the-right-ip/plant-breeders-rights (accessed on 1 May 2017).
- Dornstauder, E.; Jisa, E.; Unterrieder, I.; Krenn, L.; Kubelka, W.; Jungbauer, A. Estrogenic activity of two standardized red clover extracts (Menoflavon®) intended for large scale use in hormone replacement therapy. J. Steroid Biochem. Mol. Biol. 2001, 78, 67–75. [Google Scholar] [CrossRef]
- Alimoradi, A.; Mahdizad, H.F. Effects of phytoestrogens on bone mineral density during the menopause transition: A systematic review of randomized controlled trials. Climacteric 2016, 19, 535–545. [Google Scholar]
- Ghazanfarpour, M.; Sadeghi, R.; Roudsari, R.L.; Khorsand, I.; KhadivZedah, T.; Muoio, B. Red clover for treatment of hot flashes and menopausal symptoms: A systematic review and meta-analysis. J. Obstet. Gynaecol. 2016, 36, 301–311. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.N.; Lin, C.C.; Liu, C.F. Efficacy of phytoestrogens for menopausal symptoms: A meta-analysis and systematic review. Climacteric 2016, 18, 260–269. [Google Scholar] [CrossRef] [PubMed]
- Clifton-Bligh, P.B.; Nery, M.L.; Clifton-Bligh, R.J.; Visvalingam, S.; Fulcher, G.R.; Byth, K.; Baber, R. Red clover isoflavones enriched with formonetin lower serum LDL cholesterol—A randomized, double-blind, placebo-controlled study. Eur. J. Clin. Nutr. 2015, 69, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Francis, C.M.; Quinlivan, B.J. Selection for formononetin content in red clover (Trifolium pratense). In Plant Introduction, Breeding and Seed Production, Proceedings of the XII International Grassland Congress, Moscow, Soviet Union, 11–20 June 1974; Organizing Committee of the International Grassland Congress: Moscow, Soviet Union, 1974; pp. 84–88. [Google Scholar]
- Kouame, C.N.; Quesenberry, K.H. Cluster analysis of a collection of red clover germplasm. Genet. Resour. Crop Evol. 1993, 40, 39–47. [Google Scholar] [CrossRef]
- Williams, W.M. Genetics and Breeding. In White Clover; Barker, M.J., Williams, W.M., Eds.; CAB International: Wallingford, UK, 1987. [Google Scholar]
- Smith, R.S.; Bishop, D.J. Astred—A stoloniferous red clover. In Proceedings of the 17th International Grassland Congress, Palmerston North, New Zealand, 8–21 February 1993; Organising Committee International Grassland Congress: Palmerston North, New Zealand, 1993; pp. 421–423. [Google Scholar]
- Reed, K.F.M.; Cade, J.W.; Williams, A.E. The significance of Mediterranean plant introductions for increasing the winter growth of pasture in the high rainfall areas of Victoria. In Proceedings of the 1st Australian Agronomy Conference, Lawes, Australia, April 1980; Wood, I.M., Ed.; Australian Society of Agronomy: Sydney, Australia, 1980; p. 238. [Google Scholar]
- Reed, K.F.M.; Clement, S.L.; Feely, W.F.; Clark, B. Improving tall fescue for cool season vigour. Aust. J. Exp. Agric. 2004, 44, 873–881. [Google Scholar] [CrossRef]
- Dear, B.S.; Moore, G.A.; Hughes, S.J. Adaptation and potential contribution of temperate perennial legumes to the southern Australian wheatebelt: A review. Aust. J. Exp. Agric. 2003, 52, 973–983. [Google Scholar] [CrossRef]
- Cade, J.; Stapleton, P. How to Identify Sub Clover Varieties. Agricultural Notes—Agdex 137/33; Natural Resources and Environment: Victoria, Australia, 1984; pp. 1–4. [Google Scholar]
- Setchell, K.D.; Welsh, M.B.; Lim, C.K. High-performance liquid chromatographic analysis of phytoestrogens in soy protein preparations with ultraviolet, electrochemical and thermospray mass spectrometric detection. J. Chromatogr. 1987, 386, 315–323. [Google Scholar] [CrossRef]
- Franke, A.A.; Custer, L.J.; Cerna, C.M.; Narala, K. Rapid HPLC analysis of dietary phytoestrogens from legumes and from human urine. Proc. Soc. Exp. Biol. Med. 1995, 208, 18–26. [Google Scholar] [CrossRef]
- GenStat Committee. GenStat® Release 7.1; VSN International Ltd.: Oxford, UK, 2003. [Google Scholar]
| Trait | Range | Mean | σ2g ± s.e | R |
|---|---|---|---|---|
| Autumn Recovery (Aut rec) | 0.95–2.68 | 1.704 | 0.214 ± 0.044 | 0.694 |
| Flowering Time (FT) | 28.0–83.2 | 50.320 | 136.9 ± 25.60 | 0.877 |
| Growth Habit, Spring/Summer (gh.ss) | 2.19–5.84 | 3.976 | 1.580 ± 0.290 | 0.967 |
| Leaf Shape (leaf sh) | 1.10–2.40 | 2.166 | 0.035 ± 0.018 | 0.467 |
| Flowering Prolificacy (prolificacy) | 3.02–5.92 | 4.789 | 0.520 ± 0.104 | 0.820 |
| Stem Colour (stem) | 1.10–2.53 | 1.780 | 0.089 ± 0.033 | 0.447 |
| Stipule Pigmentation (stipule) | 1.50–3.73 | 2.278 | 0.314 ± 0.088 | 0.632 |
| Cool Season Yield (DM, g/plant) (winter) | 1.97–4.94 | 2.938 | 0.411 ± 0.086 | 0.734 |
| Leaf Width (mm) (leaf w) | 13.66–22.16 | 17.085 | 4.08 ± 1.11 | 0.658 |
| Leaf Length (mm) (leaf l) | 17.87–28.85 | 22.397 | 9.20 ± 2.66 | 0.638 |
| Leaf Area (cm2) (leaf a) | 1.56–2.44 | 1.944 | 0.063 ± 0.018 | 0.589 |
| Biochanin (g/kg DM) (Bioch) | 0.09–0.91 | 0.480 | 0.021 ± 0.01 | 0.656 |
| Daidzein (g/kg DM) (Diadz) | 0.003–0.060 | 0.010 | 0.0001 ± 0.0 | 0.791 |
| Formononetin (g/kg DM) (Form) | 0.06–0.86 | 0.354 | 0.011 ± 0.01 | 0.588 |
| Genistein (g/kg DM) (Genist) | 0.001–0.076 | 0.017 | 0.0001 ± 0.001 | 0.374 |
| Total Isoflavones (g/kg DM) | 0.14–1.45 | 0.865 | 0.051 ± 0.028 | 0.670 |
| Line No. | Cultivar/ Accession | Origin | Autumn Recovery | Flowering Time (days) | Growth Habit Spring/ Summer | Leaf Shape | Prolificacy of Flowering | Stem Colour | Winter DM Yield (kg) | Leaf Width (mm) | Leaf Length (mm) | Total Leaf Area (loge cm2) | Stipule Pigmentation |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | PAC 19 | Chile | 1.94 | 71.9 | 2.47 | 2.12 | 4.12 | 1.53 | 6.58 | 19.6 | 24.3 | 2.19 | 2.04 |
| 2 | A9807 | Unknown | 1.71 | 49.1 | 2.54 | 1.89 | 4.86 | 1.68 | 5.23 | 17.8 | 24.6 | 2.19 | 1.76 |
| 3 | Rajah | Denmark | 1.51 | 71.7 | 4.73 | 2.24 | 4.79 | 1.59 | 3.90 | 14.6 | 17.9 | 1.61 | 1.62 |
| 4 | SA 26.449 | Turkey | 2.13 | 45.3 | 4.95 | 2.24 | 4.73 | 1.59 | 7.41 | 15.8 | 19.6 | 1.78 | 2.09 |
| 5 | PI 4379 | Unknown | 1.60 | 43.3 | 3.09 | 2.16 | 5.41 | 1.87 | 5.78 | 17.3 | 23.7 | 2.11 | 2.33 |
| 6 | SA 20.017 | Turkey | 1.86 | 30.4 | 5.45 | 2.16 | 4.65 | 1.72 | 6.10 | 13.7 | 18.0 | 1.58 | 2.92 |
| 7 | PI 4377 | Unknown | 1.56 | 56.6 | 3.03 | 2.28 | 5.10 | 1.66 | 5.08 | 16.5 | 21.6 | 1.88 | 1.87 |
| 8 | S48/9732 | Turkey | 1.75 | 45.4 | 5.61 | 2.15 | 4.65 | 1.75 | 6.07 | 16.1 | 21.3 | 1.89 | 2.67 |
| 9 | El Sureno | Argentina | 0.95 | 43.2 | 2.76 | 2.15 | 5.31 | 1.66 | 4.36 | 19.5 | 25.8 | 2.23 | 2.41 |
| 10 | SA 758 | Morocco | 2.68 | 69.7 | 5.34 | 2.12 | 3.24 | 1.72 | 7.95 | 17.6 | 22.8 | 2.14 | 3.37 |
| 11 | Renegade | USA | 1.67 | 43.7 | 2.53 | 2.11 | 4.93 | 1.93 | 5.37 | 17.6 | 24.9 | 2.05 | 2.04 |
| 12 | H: 4949 | Unknown | 1.67 | 51.2 | 3.06 | 2.24 | 5.76 | 1.55 | 6.33 | 17.6 | 22.5 | 2.09 | 1.76 |
| 13 | Krano | Denmark | 1.82 | 46.8 | 2.81 | 2.06 | 5.27 | 1.62 | 5.67 | 17.5 | 22.8 | 1.99 | 1.82 |
| 14 | PI 5290 | Unknown | 1.47 | 52.7 | 2.95 | 2.14 | 5.35 | 2.02 | 5.92 | 18.3 | 23.6 | 1.91 | 2.39 |
| 15 | T 106 | Tunisia | 2.54 | 46.4 | 4.91 | 2.06 | 4.71 | 2.53 | 9.88 | 17.6 | 23.8 | 2.15 | 3.08 |
| 16 | SA 15.891 | Turkey | 1.74 | 71.5 | 4.45 | 2.14 | 3.43 | 1.89 | 5.29 | 14.2 | 19.1 | 1.56 | 2.43 |
| 17 | H: 7565 | Unknown | 1.25 | 56.5 | 3.46 | 2.35 | 5.11 | 1.73 | 5.41 | 17.4 | 21.2 | 1.97 | 2.13 |
| 18 | SA 22.825 | UK | 1.25 | 42.2 | 4.22 | 2.17 | 5.53 | 1.63 | 5.11 | 15.5 | 21.7 | 1.85 | 1.78 |
| 19 | Cherokee | USA | 1.42 | 38.3 | 2.90 | 2.11 | 5.14 | 1.62 | 5.89 | 17.5 | 24.1 | 2.05 | 1.84 |
| 20 | SA 32.382 | Turkey | 1.92 | 34.7 | 4.99 | 2.13 | 4.45 | 1.80 | 6.42 | 15.7 | 20.2 | 1.73 | 2.61 |
| 21 | S42/9635 | Turkey | 2.27 | 66.2 | 5.64 | 2.02 | 4.37 | 2.34 | 6.91 | 16.1 | 22.0 | 1.94 | 3.19 |
| 22 | M 98 | Morocco | 2.75 | 83.2 | 5.65 | 2.11 | 3.39 | 1.73 | 8.64 | 17.2 | 22.1 | 2.05 | 2.93 |
| 23 | SA 32.377 | Turkey | 2.02 | 48.3 | 5.23 | 2.28 | 4.17 | 1.85 | 6.87 | 17.4 | 22.5 | 2.05 | 2.02 |
| 24 | SA 21.963 | Uruguay | 1.40 | 36.9 | 2.63 | 2.04 | 5.35 | 1.76 | 5.75 | 17.5 | 25.7 | 2.16 | 2.20 |
| 25 | S36 | Sardinia | 2.48 | 55.2 | 5.71 | 2.10 | 3.79 | 1.97 | 8.53 | 15.9 | 20.5 | 1.82 | 2.48 |
| 26 | SA 901 | UK | 1.61 | 49.3 | 3.36 | 2.34 | 5.19 | 1.71 | 5.54 | 17.0 | 21.1 | 1.84 | 2.20 |
| 27 | SA 8.440 | Russia | 1.48 | 44.3 | 3.17 | 2.33 | 4.60 | 1.57 | 5.39 | 15.5 | 19.7 | 1.72 | 2.20 |
| 28 | Colenso | New Zealand | 1.34 | 58.0 | 2.89 | 2.28 | 5.31 | 1.59 | 5.08 | 17.5 | 22.2 | 2.08 | 1.91 |
| 29 | SA 32.376 | Turkey | 2.16 | 53.5 | 5.05 | 2.22 | 4.77 | 1.76 | 6.93 | 16.8 | 22.0 | 1.96 | 2.05 |
| 30 | SA 26.500 | Turkey | 2.05 | 46.0 | 4.93 | 2.15 | 4.66 | 2.08 | 6.72 | 16.3 | 24.3 | 2.05 | 2.66 |
| 31 | Hamidori | Japan | 1.50 | 60.4 | 2.78 | 2.26 | 4.54 | 1.71 | 4.60 | 15.7 | 18.9 | 1.67 | 1.50 |
| 32 | M 80 | Morocco | 2.64 | 66.9 | 5.48 | 2.10 | 3.75 | 1.70 | 9.17 | 16.7 | 21.8 | 2.12 | 3.73 |
| 33 | S44/9664 | Turkey | 1.93 | 40.9 | 5.54 | 2.08 | 5.31 | 2.29 | 6.68 | 16.1 | 22.2 | 1.88 | 2.89 |
| 34 | Quinqueli | Chile | 1.16 | 56.9 | 3.47 | 2.18 | 4.87 | 2.00 | 5.61 | 19.0 | 24.7 | 2.15 | 2.14 |
| 35 | SA 18.686 | Australia | 1.47 | 43.5 | 3.29 | 2.22 | 5.56 | 1.67 | 6.15 | 18.0 | 23.2 | 2.04 | 2.12 |
| 36 | SwisSelectn | Switzerland | 1.63 | 49.4 | 2.19 | 2.22 | 4.74 | 1.45 | 5.51 | 19.4 | 24.8 | 2.23 | 1.78 |
| 37 | S46/9679 | Turkey | 1.93 | 53.2 | 5.79 | 2.12 | 4.61 | 1.76 | 5.88 | 16.5 | 20.9 | 1.86 | 2.35 |
| 38 | M 154 | Morocco | 2.54 | 69.8 | 5.74 | 2.46 | 3.16 | 1.75 | 8.10 | 16.8 | 21.2 | 1.98 | 2.32 |
| 39 | A9806 | Turkey | 1.26 | 52.8 | 2.80 | 2.19 | 5.36 | 1.67 | 5.11 | 22.2 | 28.9 | 2.44 | 1.80 |
| 40 | Redquin | Australia | 1.45 | 45.1 | 2.82 | 2.22 | 5.42 | 1.71 | 4.91 | 19.8 | 24.4 | 2.14 | 1.79 |
| 41 | Makimidori | Japan | 1.51 | 57.2 | 2.76 | 2.18 | 4.75 | 1.96 | 5.17 | 15.4 | 18.6 | 1.70 | 2.24 |
| 42 | SA 32.381 | Turkey | 2.10 | 49.8 | 5.18 | 2.18 | 4.05 | 1.55 | 7.32 | 17.7 | 24.2 | 2.20 | 2.46 |
| 43 | S47/9706 | Turkey | 1.87 | 57.3 | 5.54 | 2.16 | 4.69 | 2.31 | 6.20 | 14.1 | 18.3 | 1.59 | 2.64 |
| 44 | Concorde | Unknown | 1.52 | 53.6 | 2.99 | 2.21 | 5.23 | 1.82 | 4.56 | 15.5 | 20.0 | 1.80 | 1.78 |
| 45 | Maneta | France | 1.69 | 68.3 | 2.57 | 2.24 | 3.92 | 1.71 | 5.65 | 17.8 | 22.2 | 1.92 | 2.13 |
| 46 | SA 19676 | Afghanistan | 1.12 | 28.0 | 2.90 | 2.13 | 4.89 | 1.71 | 4.07 | 16.1 | 21.8 | 1.89 | 1.91 |
| 47 | Hamua | New Zealand | 1.21 | 53.0 | 2.92 | 2.07 | 5.92 | 1.59 | 4.95 | 16.7 | 22.7 | 1.95 | 2.36 |
| 48 | PI 4383 | Unknown | 2.10 | 64.6 | 3.53 | 2.00 | 3.02 | 1.86 | 6.38 | 17.4 | 23.5 | 2.07 | 2.38 |
| 49 | PI 4378 | Unknown | 1.14 | 51.6 | 2.85 | 2.32 | 5.28 | 1.83 | 5.39 | 16.9 | 22.2 | 1.99 | 1.94 |
| 50 | SA 22.000 | USA | 1.14 | 43.6 | 2.57 | 2.24 | 5.51 | 1.66 | 4.59 | 18.7 | 23.8 | 2.00 | 2.36 |
| 51 | SA 32.380 | Turkey | 1.19 | 41.6 | 3.83 | 2.07 | 5.19 | 1.59 | 4.93 | 17.2 | 24.3 | 2.09 | 2.03 |
| 52 | SA 32.374 | Turkey | 2.09 | 29.5 | 5.29 | 1.88 | 4.36 | 1.84 | 6.91 | 16.8 | 22.2 | 1.90 | 2.11 |
| 53 | S59/9895 | Turkey | 1.56 | 34.2 | 5.73 | 2.12 | 4.35 | 1.70 | 5.97 | 15.5 | 19.8 | 1.71 | 2.40 |
| 54 | SA 32.061 | Russia | 2.16 | 51.0 | 5.85 | 2.22 | 4.28 | 1.91 | 6.83 | 16.5 | 21.0 | 1.88 | 2.23 |
| 55 | FLMR7 | USA | 1.53 | 48.3 | 4.02 | 2.20 | 5.38 | 1.74 | 4.62 | 15.6 | 20.1 | 1.66 | 2.64 |
| 56 | LE 116 | Uruguay | 1.63 | 33.2 | 2.49 | 2.15 | 5.75 | 1.56 | 6.61 | 21.0 | 27.6 | 2.36 | 1.95 |
| 57 | PI 6435 | Unknown | 1.32 | 41.7 | 2.77 | 2.06 | 5.30 | 1.72 | 5.12 | 20.2 | 27.5 | 2.32 | 2.73 |
| 58 | Kenland | USA | 2.04 | 51.4 | 5.64 | 2.30 | 4.07 | 1.77 | 6.72 | 16.3 | 21.2 | 1.94 | 2.70 |
| 59 | Astred | Portugal | 1.28 | 43.2 | 2.70 | 2.40 | 5.30 | 1.74 | 5.12 | 18.0 | 22.7 | 2.04 | 1.78 |
| 60 | S49/9742 | Turkey | 0.99 | 40.9 | 2.88 | 2.01 | 5.46 | 2.05 | 4.89 | 17.4 | 24.9 | 2.16 | 2.33 |
| 61 | SA 12.274 | Turkey | 1.19 | 50.6 | 3.91 | 2.34 | 5.74 | 1.62 | 5.03 | 15.9 | 20.5 | 1.84 | 2.10 |
| 62 | P42 | Unknown | 1.90 | 55.2 | 5.51 | 2.08 | 5.00 | 2.03 | 5.95 | 15.4 | 20.1 | 1.84 | 3.13 |
| 63 | T 98 | Tunisia | 1.50 | 40.0 | 5.63 | 2.15 | 4.79 | 1.60 | 5.65 | 16.3 | 20.6 | 1.91 | 2.52 |
| 64 | 37796 | Unknown | 1.81 | 42.2 | 4.04 | 2.06 | 4.84 | 1.83 | 7.08 | 20.5 | 27.1 | 2.33 | 1.73 |
| S.e.d | 0.229 | 3.47 | 0.287 | 0.216 | 0.460 | 0.319 | 0.871 | 1.77 | 2.78 | 0.233 | 0.504 |
| Cultivar/Line No. | Cultivar/Accession | Origin | Daidzein | Genistein | Formononetin | Biochanin | Total Isoflavone |
|---|---|---|---|---|---|---|---|
| 1 | PAC 19 | Chile | 0.011 | 0.04 | 0.59 | 0.62 | 1.26 |
| 3 | Rajah | Denmark | 0.009 | 0.01 | 0.67 | 0.53 | 1.25 |
| 4 | SA 26.449 | Turkey | 0.006 | 0.00 | 0.28 | 0.59 | 0.88 |
| 5 | PI 4379 | Unknown | 0.019 | 0.04 | 0.47 | 0.57 | 1.10 |
| 6 | SA 20.017 | Turkey | 0.007 | 0.03 | 0.31 | 0.62 | 0.97 |
| 7 | PI 4377 | Unknown | 0.014 | 0.02 | 0.68 | 0.63 | 1.34 |
| 8 | S48/9732 | Turkey | 0.008 | 0.05 | 0.33 | 0.33 | 0.71 |
| 9 | El Sureno | Argentina | 0.005 | 0.08 | 0.59 | 0.78 | 1.45 |
| 10 | SA 758 | Morocco | 0.008 | 0.03 | 0.13 | 0.10 | 0.27 |
| 11 | Renegade | USA | 0.014 | 0.02 | 0.48 | 0.50 | 1.01 |
| 13 | Krano | Denmark | 0.007 | 0.01 | 0.27 | 0.78 | 1.06 |
| 14 | PI 5290 | Unknown | 0.008 | 0.01 | 0.51 | 0.67 | 1.20 |
| 15 | T 106 | Tunisia | 0.060 | 0.01 | 0.29 | 0.17 | 0.53 |
| 16 | SA 15.891 | Turkey | 0.005 | 0.03 | 0.41 | 0.56 | 0.99 |
| 18 | SA 22.825 | UK | 0.010 | 0.01 | 0.32 | 0.82 | 1.16 |
| 19 | Cherokee | USA | 0.010 | 0.01 | 0.37 | 0.74 | 1.13 |
| 20 | SA 32.382 | Turkey | 0.012 | 0.00 | 0.34 | 0.47 | 0.82 |
| 21 | S42/9635 | Turkey | 0.007 | 0.01 | 0.44 | 0.54 | 0.99 |
| 22 | M 98 | Morocco | 0.006 | 0.00 | 0.02 | 0.13 | 0.17 |
| 23 | SA 32.377 | Turkey | 0.013 | 0.03 | 0.22 | 0.31 | 0.58 |
| 24 | SA 21.963 | Uruguay | 0.009 | 0.01 | 0.57 | 0.62 | 1.20 |
| 25 | S36 | Sardinia | 0.009 | 0.02 | 0.16 | 0.91 | 1.09 |
| 26 | SA 901 | UK | 0.003 | 0.01 | 0.29 | 0.76 | 1.06 |
| 27 | SA 8.440 | Russia | 0.008 | 0.01 | 0.23 | 0.45 | 0.68 |
| 28 | Colenso | New Zealand | 0.007 | 0.02 | 0.56 | 0.71 | 1.30 |
| 29 | SA 32.376 | Turkey | 0.012 | 0.01 | 0.39 | 0.39 | 0.80 |
| 30 | SA 26.500 | Turkey | 0.014 | 0.01 | 0.28 | 0.27 | 0.57 |
| 32 | M 80 | Morocco | 0.006 | 0.00 | 0.58 | 0.07 | 0.14 |
| 33 | S44/9664 | Turkey | 0.008 | 0.05 | 0.19 | 0.37 | 0.62 |
| 34 | Quinqueli | Chile | 0.005 | 0.01 | 0.62 | 0.66 | 1.23 |
| 35 | SA 18.686 | Australia | 0.004 | 0.01 | 0.25 | 0.78 | 1.05 |
| 37 | S46/9679 | Turkey | 0.006 | 0.01 | 0.35 | 0.44 | 0.80 |
| 38 | M 154 | Morocco | 0.008 | 0.00 | 0.07 | 0.18 | 0.27 |
| 42 | SA 32.381 | Turkey | 0.009 | 0.01 | 0.31 | 0.35 | 0.67 |
| 43 | S47/9706 | Turkey | 0.018 | 0.03 | 0.37 | 0.23 | 0.65 |
| 46 | SA 19676 | Afghanistan | 0.005 | 0.00 | 0.08 | 0.23 | 0.31 |
| 47 | Hamua | New Zealand | 0.005 | 0.01 | 0.52 | 0.70 | 1.23 |
| 49 | PI 4378 | Unknown | 0.026 | 0.03 | 0.55 | 0.57 | 1.17 |
| 51 | SA 32.380 | Turkey | 0.007 | 0.02 | 0.17 | 0.34 | 0.53 |
| 52 | SA 32.374 | Turkey | 0.013 | 0.05 | 0.30 | 0.21 | 0.57 |
| 53 | S59/9895 | Turkey | 0.004 | 0.01 | 0.09 | 0.09 | 0.19 |
| 54 | SA 32.061 | Russia | 0.010 | 0.01 | 0.27 | 0.49 | 0.78 |
| 55 | FLMR7 | USA | 0.006 | 0.00 | 0.28 | 0.50 | 0.79 |
| 56 | LE 116 | Uruguay | 0.009 | 0.01 | 0.86 | 0.51 | 1.39 |
| 58 | Kenland | USA | 0.007 | 0.04 | 0.12 | 0.19 | 0.36 |
| 59 | Astred | Portugal | 0.018 | 0.02 | 0.55 | 0.68 | 1.26 |
| 61 | SA 12.274 | Turkey | 0.004 | 0.01 | 0.56 | 0.72 | 1.29 |
| 62 | P42 | Unknown | 0.010 | 0.01 | 0.38 | 0.66 | 1.06 |
| 63 | T 98 | Tunisia | 0.013 | 0.00 | 0.25 | 0.16 | 0.43 |
| S.e.d | 0.011 | 0.340 | 0.231 | 0.271 | 0.408 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Little, V.; Reed, K.F.M.; Smith, K.F. Variation for Concentrations of Various Phytoestrogens and Agronomic Traits Among a Broad Range of Red Clover (Trifolium pratense) Cultivars and Accessions. Agronomy 2017, 7, 34. https://doi.org/10.3390/agronomy7020034
Little V, Reed KFM, Smith KF. Variation for Concentrations of Various Phytoestrogens and Agronomic Traits Among a Broad Range of Red Clover (Trifolium pratense) Cultivars and Accessions. Agronomy. 2017; 7(2):34. https://doi.org/10.3390/agronomy7020034
Chicago/Turabian StyleLittle, Valerie, Kevin F.M. Reed, and Kevin F. Smith. 2017. "Variation for Concentrations of Various Phytoestrogens and Agronomic Traits Among a Broad Range of Red Clover (Trifolium pratense) Cultivars and Accessions" Agronomy 7, no. 2: 34. https://doi.org/10.3390/agronomy7020034
APA StyleLittle, V., Reed, K. F. M., & Smith, K. F. (2017). Variation for Concentrations of Various Phytoestrogens and Agronomic Traits Among a Broad Range of Red Clover (Trifolium pratense) Cultivars and Accessions. Agronomy, 7(2), 34. https://doi.org/10.3390/agronomy7020034







