Little Potential of Spring Wheat Genotypes as a Strategy to Reduce Nitrogen Leaching in Central Europe
Abstract
:1. Introduction
2. Experimental Section
2.1. Experimental Conditions
2.2. Data Sampling
2.3. Calculations and Data Analysis
2.4. Statistical Analysis
3. Results
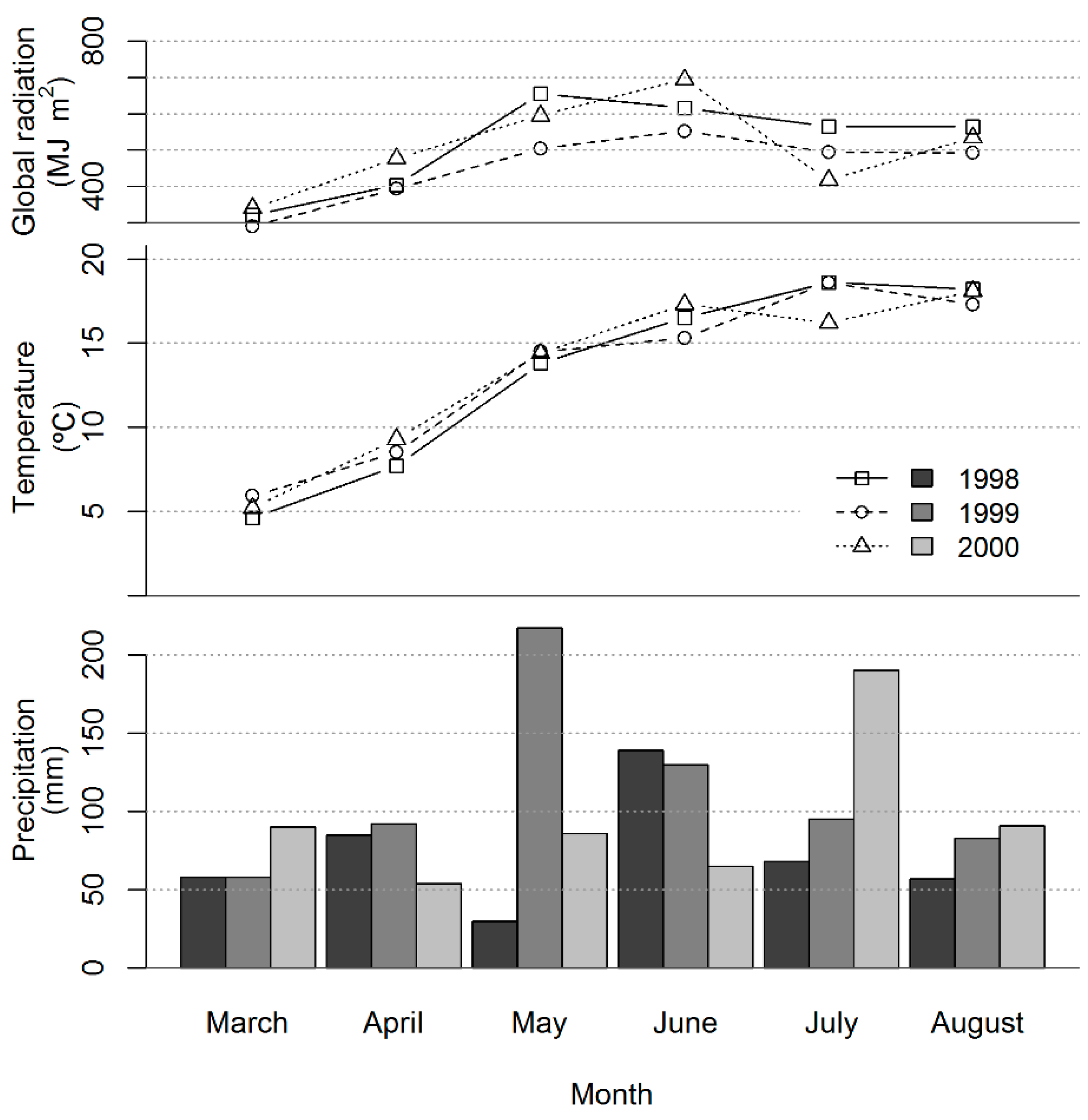
3.1. Growth Conditions
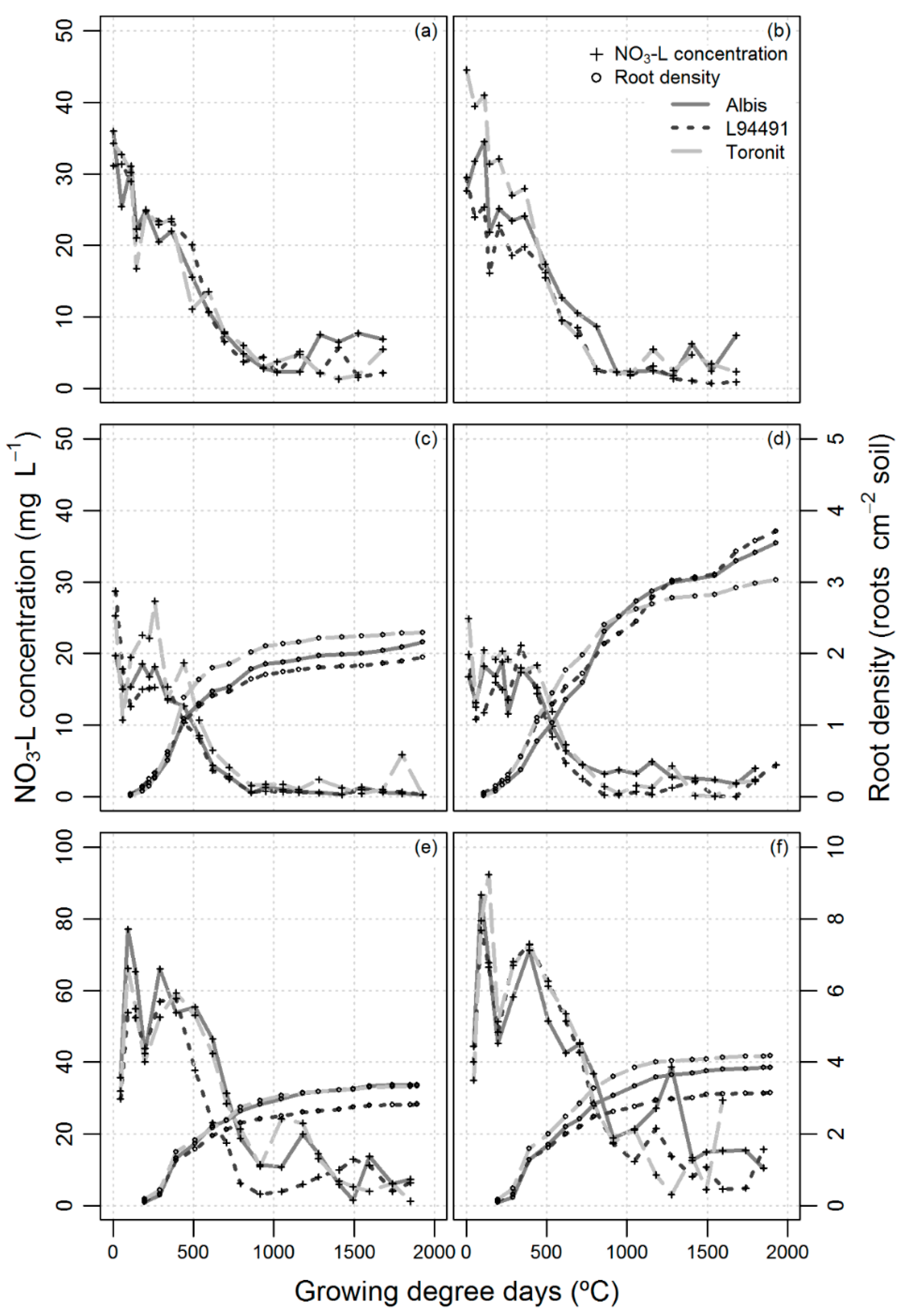
3.2. Crop Growth
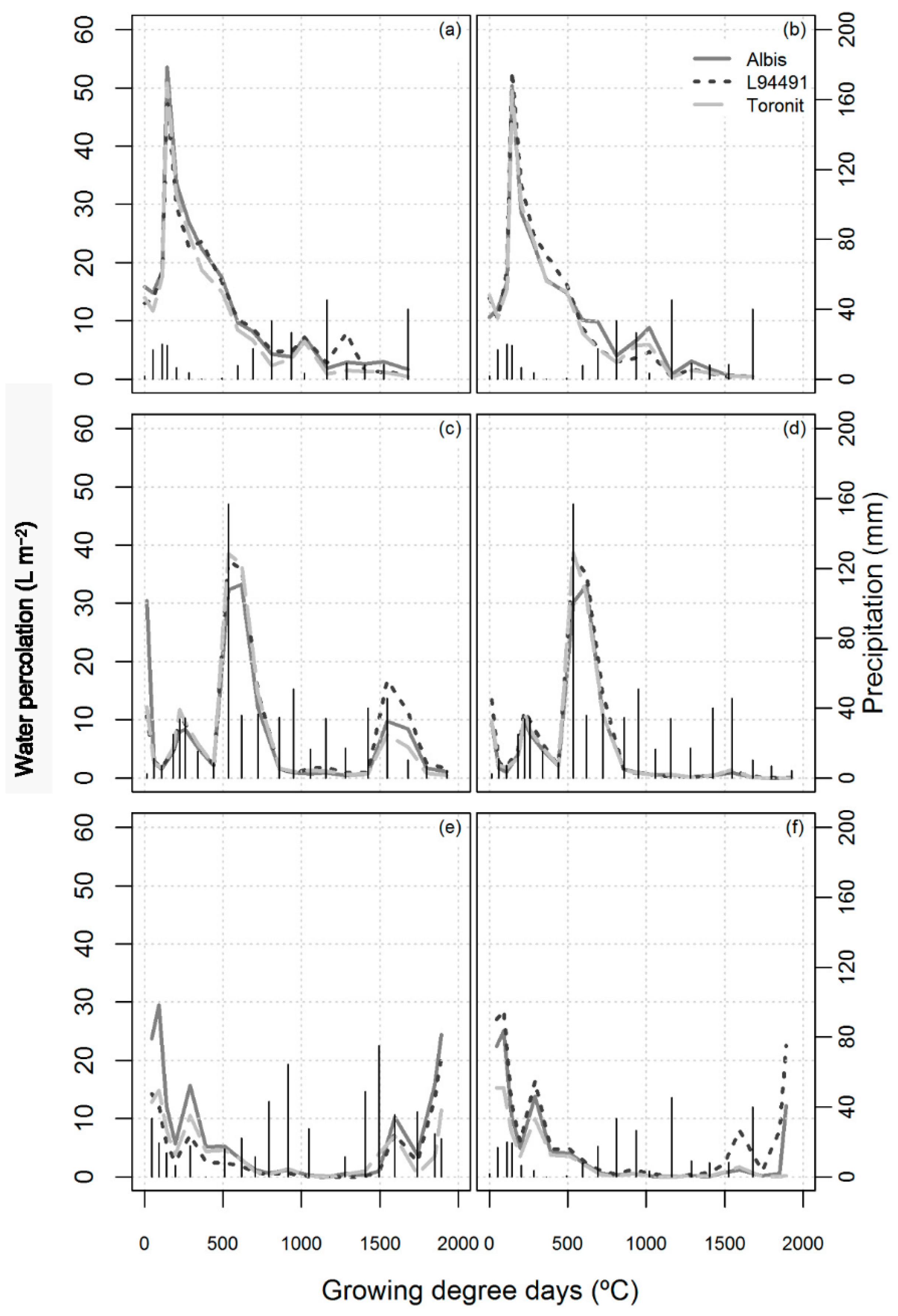
3.3. Water Percolation
3.4. NO3-N Leaching
3.5. Fertilizer N Recovery
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| ANR | apparent fertilizer N recovery |
| GDD | growing degree days |
| HN | high N supply |
| LN | low N supply |
| NO3ˉ | nitrate |
| NO3-N | nitrogen in the form of nitrate |
| NR | isotopic fertilizer N recovery efficiency |
| NO3-L | nitrate concentration in the leachate solution |
References
- David, M.B.; Gentry, L.E.; Kovacic, D.A.; Smith, K.M. Nitrogen balance in and export from an agricultural watershed. J. Environ. Qual. 1997, 26, 1038–1048. [Google Scholar] [CrossRef]
- Mitsch, W.J.; Day, J.W. Restoration of wetlands in the Mississippi-Ohio-Missouri (MOM) River Basin: Experience and needed research. Ecol. Eng. 2006, 26, 55–69. [Google Scholar] [CrossRef]
- Galloway, J.N.; Townsend, A.R.; Erisman, J.W.; Bekunda, M.; Cai, Z.; Freney, J.R.; Martinelli, L.A.; Seitzinger, S.P.; Sutton, M.A. Transformation of the nitrogen cycle: Recent trends, questions, and potential solutions. Science 2008, 320, 889–892. [Google Scholar] [CrossRef] [PubMed]
- Schindler, F.V.; Knighton, R.E. Fate of fertilizer nitrogen applied to corn as estimated by the isotopic and difference methods. Soil Sci. Soc. Am. J. 1999, 63, 1734–1740. [Google Scholar] [CrossRef]
- Hauck, R.D.; Bremner, J.M. Use of tracers for soil and fertilizer nitrogen research. In Advances in Agronomy; Academic Press: Philadelphia, PA, USA, 1976; Volume 28, pp. 219–266. [Google Scholar]
- Rao, A.C.S.; Smith, J.L.; Parr, J.F.; Papendick, R.I. Considerations in estimating nitrogen recovery efficiency by the difference and isotopic dilution methods. Fertil. Res. 1992, 33, 209–217. [Google Scholar] [CrossRef]
- Roberts, T.L.; Janzen, H.H. Comparison of direct and indirect methods of measuring fertilizer N uptake in winter-wheat. Can. J. Soil Sci. 1990, 70, 119–124. [Google Scholar] [CrossRef]
- Smil, V. Nitrogen in crop production: An account of global flows. Glob. Biogeochem. Cy. 1999, 13, 647–662. [Google Scholar] [CrossRef]
- Cassman, K.G.; Dobermann, A.; Walters, D.T. Agroecosystems, nitrogen-use efficiency, and nitrogen management. Ambio 2002, 31, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Olson, R.V.; Swallow, C.W. Fate of labeled nitrogen-fertilizer applied to winter-wheat for 5 years. Soil Sci. Soc. Am. J. 1984, 48, 583–586. [Google Scholar] [CrossRef]
- Robinson, D. The responses of plants to non-uniform supplies of nutrients. New Phytol. 1994, 127, 635–674. [Google Scholar] [CrossRef]
- Raun, W.R.; Johnson, G.V. Improving nitrogen use efficiency for cereal production. Agron. J. 1999, 91, 357–363. [Google Scholar] [CrossRef]
- Ladha, J.K.; Pathak, H.; Krupnik, T.J.; Six, J.; van Kessel, C. Efficiency of fertilizer nitrogen in cereal production: Retrospects and prospects. Adv. Agron. 2005, 87, 85–156. [Google Scholar]
- Banziger, M.; Feil, B.; Schmid, J.E.; Stamp, P. Genotypic variation in grain N content of wheat as affected by mineral n supply in the soil. Eur. J. Agron. 1992, 1, 155–162. [Google Scholar] [CrossRef]
- Le Gouis, J.; Beghin, D.; Heumez, E.; Pluchard, P. Genetic differences for nitrogen uptake and nitrogen utilisation efficiencies in winter wheat. Eur. J. Agron. 2000, 12, 163–173. [Google Scholar] [CrossRef]
- Rodgers, C.O.; Barneix, A.J. Cultivar differences in the rate of nitrate uptake by intact wheat plants as related to growth-rate. Physiol. Plant. 1988, 72, 121–126. [Google Scholar] [CrossRef]
- Caspari, H.W.; Behboudian, M.H.; Chalmers, D.J.; Renquist, A.R. Pattern of seasonal water-use of Asian pears determined by lysimeters and the heat-pulse technique. J. Am. Soc. Hort. Sci. 1993, 118, 562–569. [Google Scholar]
- Wegehenkel, M.; Zhang, Y.Q.; Zenker, T.; Diestel, H. The use of lysimeter data for the test of two soil-water balance models: A case study. J. Plant Nutr. Soil Sci. 2008, 171, 762–776. [Google Scholar] [CrossRef]
- Eriksen, J.; Pedersen, L.; Jorgensen, J.R. Nitrate leaching and bread-making quality of spring wheat following cultivation of different grasslands. Agric. Ecosyst. Environ. 2006, 116, 165–175. [Google Scholar] [CrossRef]
- Knappe, S.; Haferkorn, U.; Meissner, R. Influence of different agricultural management systems on nitrogen leaching: Results of lysimeter studies. J. Plant Nutr. Soil Sci. 2002, 165, 73–77. [Google Scholar] [CrossRef]
- Uhlen, G. The leaching behavior and balances of nitrogen and other elements under spring wheat in lysimeter experiment 1985–92. Acta Agric. Scand. Sect. B-Soil Plant Sci. 1994, 44, 201–207. [Google Scholar]
- Webster, C.P.; Shepherd, M.A.; Goulding, K.W.T.; Lord, E. Comparisons of methods for measuring the leaching of mineral nitrogen from arable land. J. Soil Sci. 1993, 44, 49–62. [Google Scholar] [CrossRef]
- Liedgens, M.; Richner, W.; Stamp, P.; Soldati, A. A rhizolysimeter facility for studying the dynamics of crop and soil processes: Description and evaluation. Plant Soil 2000, 223, 87–97. [Google Scholar] [CrossRef]
- Lancashire, P.D.; Bleiholder, H.; Vandenboom, T.; Langeluddeke, P.; Stauss, R.; Weber, E.; Witzenberger, A. A uniform decimal code for growth-stages of crops and weeds. Ann. Appl. Biol. 1991, 119, 561–601. [Google Scholar] [CrossRef]
- Crocker, T.L.; Hendrick, R.L.; Ruess, R.W.; Pregitzer, K.S.; Burton, A.J.; Allen, M.F.; Shan, J.P.; Morris, L.A. Substituting root numbers for length: Improving the use of minirhizotrons to study fine root dynamics. Appl. Soil Ecol. 2003, 23, 127–135. [Google Scholar] [CrossRef]
- Upchurch, D.R.; Ritchie, J.T. Root observations using a video recording-system in mini-rhizotrons. Agron. J. 1983, 75, 1009–1015. [Google Scholar] [CrossRef]
- Smit, A.L.; George, E.; Groenwold, J. Root observations and measurements at (transparent) interfaces with soil. In Root Methods: A Handbook; Smit, A.L., Bengough, A.G., Engels, C., van Noordwijk, M., Pellerin, S., van de Geijn, S.C., Eds.; Springler-Verlag: Berlin, Germany, 2000; pp. 236–271. [Google Scholar]
- Hunt, R. Plant Growth Curves; Edward Arnold: London, UK, 1982. [Google Scholar]
- Meredith, M.P.; Stehman, S.V. Repeated measures experiments in forestry: Focus on analysis of response curves. Can. J. For. Res. 1991, 21, 957–965. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2007. [Google Scholar]
- Pinheiro, J.; Bates, D. Mixed-effects Models in S and S-plus; Springer: New York, NY, USA, 2000. [Google Scholar]
- Thomsen, I.K. Nitrate leaching under spring barley is influenced by the presence of a ryegrass catch crop: Results from a lysimeter experiment. Agr. Ecosyst. Environ. 2005, 111, 21–29. [Google Scholar] [CrossRef]
- Hooker, K.V.; Coxon, C.E.; Hackett, R.; Kirwan, L.E.; O’Keeffe, E.; Richards, K.G. Evaluation of cover crop and reduced cultivation for reducing nitrate leaching in ireland. J. Environ. Qual. 2008, 37, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Banziger, M.; Feil, B.; Schmid, J.E.; Stamp, P. Utilization of late-applied fertilizer nitrogen by spring wheat genotypes. Eur. J. Agron. 1994, 3, 63–69. [Google Scholar] [CrossRef]
- Noulas, C.; Stamp, P.; Soldati, A.; Liedgens, M. Nitrogen use efficiency of spring wheat genotypes under field and lysimeter conditions. J. Agron. Crop. Sci. 2004, 190, 111–118. [Google Scholar] [CrossRef]
- Macdonald, A.J.; Powlson, D.S.; Poulton, P.R.; Jenkinson, D.S. Unused fertilizer nitrogen in arable soils—Its contribution to nitrate leaching. J. Sci. Food Agric. 1989, 46, 407–419. [Google Scholar] [CrossRef]
- Bergstrom, L.; Brink, N. Effects of differentiated applications of fertilizer N on leaching losses and distribution of inorganic n in the soil. Plant Soil 1986, 93, 333–345. [Google Scholar] [CrossRef]
- Herrera, J.M.; Liedgens, M. Leaching and utilization of nitrogen during a spring wheat catch crop succession. J. Environ. Qual. 2009, 38, 1410–1419. [Google Scholar] [CrossRef] [PubMed]
- Ehdaie, B.; Merhaut, D.J.; Ahmadian, S.; Hoops, A.C.; Khuong, T.; Layne, A.P.; Waines, J.G. Root system size influences water-nutrient uptake and nitrate leaching potential in wheat. J. Agron. Crop. Sci. 2010, 196, 455–466. [Google Scholar] [CrossRef]
- Baethgen, W.E.; Alley, M.M. Optimizing soil and fertilizer nitrogen use by intensively managed winter-wheat.I. Crop nitrogen uptake. Agron. J. 1989, 81, 116–120. [Google Scholar] [CrossRef]
- Liao, M.T.; Palta, J.A.; Fillery, I.R.P. Root characteristics of vigorous wheat improve early nitrogen uptake. Aust. J. Agric. Res. 2006, 57, 1097–1107. [Google Scholar] [CrossRef]
- Macdonald, A.J.; Poulton, P.R.; Howe, M.T.; Goulding, K.W.T.; Powlson, D.S. The use of cover crops in cereal-based cropping systems to control nitrate leaching in SE England. Plant Soil 2005, 273, 355–373. [Google Scholar] [CrossRef]
- Abril, A.; Baleani, D.; Casado-Murillo, N.; Noe, L. Effect of wheat crop fertilization on nitrogen dynamics and balance in the humid pampas, Argentina. Agric. Ecosyst. Environ. 2007, 119, 171–176. [Google Scholar] [CrossRef]
- Below, F.E. Nitrogen metabolism and crop productivity. In Handbook of Plant and Crop Physiology; Pessarakli, M., Ed.; Marcel Dekker: New York, NY, USA, 2002; pp. 385–406. [Google Scholar]
- Jenkinson, D.S.; Fox, R.H.; Rayner, J.H. Interactions between fertilizer nitrogen and soil-nitrogen—The so-called priming effect. J. Soil Sci. 1985, 36, 425–444. [Google Scholar] [CrossRef]
- Welbank, P.J.; Gibb, M.J.; Taylor, P.J.; Williams, E.D. Root growth of cereal crops. In Rothamsted Experimental Station Report for 1973, Part 2; Rothamsted Press: Harpenden, UK, 1974; Volume 199, pp. 26–66. [Google Scholar]
| Year | 1998 | 1999 | 2000 | ||||
|---|---|---|---|---|---|---|---|
| N supply | LN | HN | LN | HN | LN | HN | |
| Grain yield (g m−2) | Albis | 679 | 758 | 464 a,† | 726 b | 664 c | 646 c |
| L94491 | 699 | 871 | 385 b | 835 ab | 685 b | 787 b | |
| Toronit | 844 | 853 | 471 a | 939 a | 795 a | 885 a | |
| Biomass yield (g m−2) | Albis | 1376 b | 1606 | 1083 b | 1878 b | 1572 a | 1681 b |
| L94491 | 1421 b | 1689 | 890 c | 1892 b | 1476 b | 1707 b | |
| Toronit | 1671 a | 1753 | 1257 a | 2240 a | 1666 a | 1936 a | |
| Shoot N concentration (%) | Albis | 1.41 ab | 1.49 ab | 0.95 ab | 1.53 ab | 1.37 ab | 1.81 ab |
| L94491 | 1.47 a | 1.53 a | 1.18 a | 1.58 a | 1.52 a | 2.01 a | |
| Toronit | 1.26 b | 1.38 b | 0.94 b | 1.44 b | 1.35 b | 1.71 b | |
| Biomass N yield (g N m−2) | Albis | 19.4 | 22.9 b | 9.28 | 24.57 b | 19.53 | 26.71 b |
| L94491 | 20.8 | 26.5 a | 9.44 | 27.83 a | 19.74 | 33.07 a | |
| Toronit | 21.5 | 24.0 a | 9.74 | 29.75 a | 20.16 | 32.03 a | |
| N supply | Parameter | Unit | 1999 | 2000 | ||||
|---|---|---|---|---|---|---|---|---|
| Albis | L99491 | Toronit | Albis | L99491 | Toronit | |||
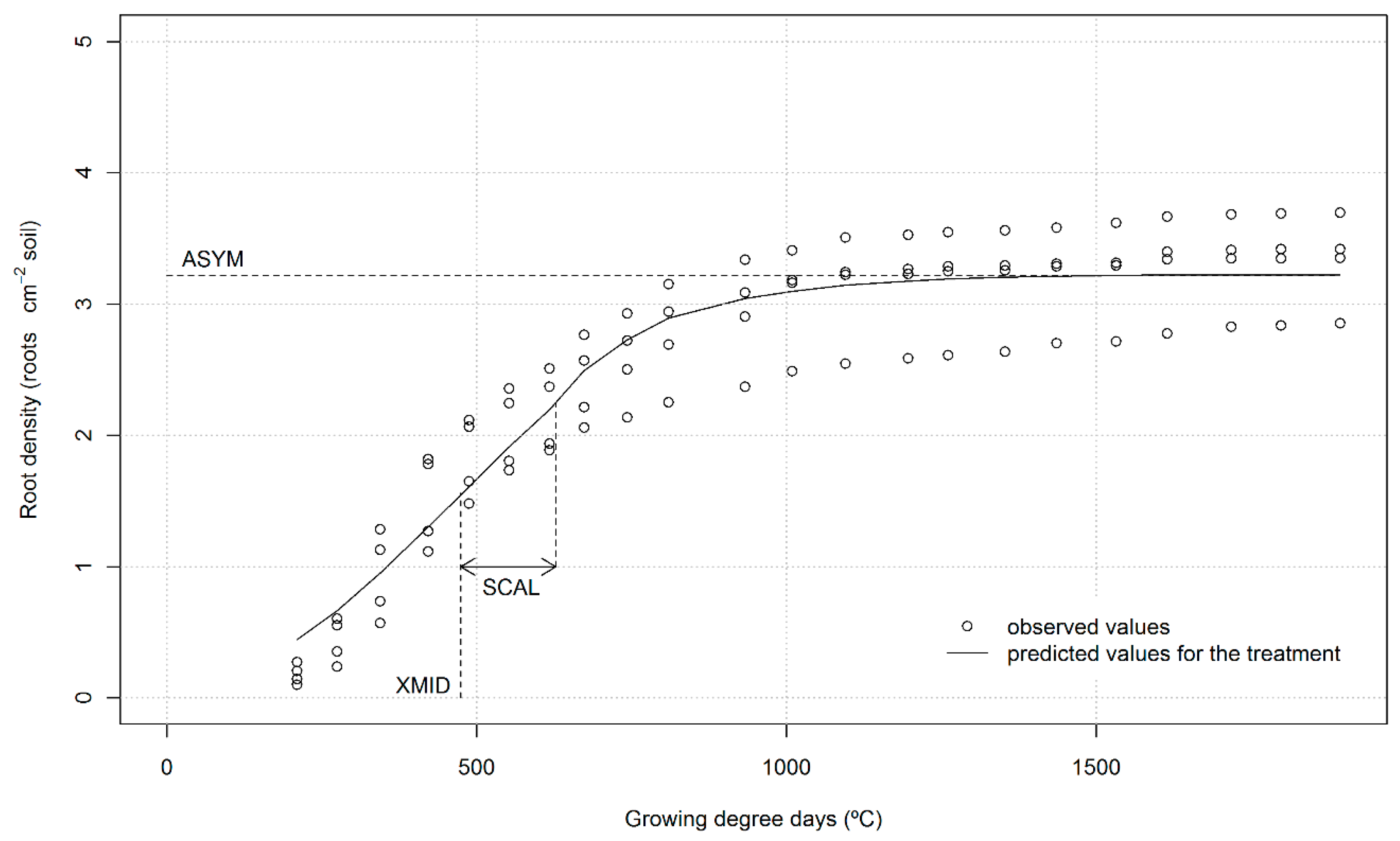
| LN | ASYM | roots cm−2 | 2.01 | 1.84 | 2.19 | 3.75 a,† | 3.05 b | 4.12 a |
| XMID | °C | 460 a | 429 ab | 411 b | 568 | 514 | 538 | |
| SCAL | °C | 132 a | 130 a | 97 b | 174 | 160 | 170 | |
| HN | ASYM | roots cm−2 | 3.37 | 3.84 | 2.88 | 3.23 | 2.68 | 3.22 |
| XMID | °C | 703 ab | 744 a | 546 b | 509 | 472 | 489 | |
| SCAL | °C | 202 ab | 260 a | 155 b | 153 | 139 | 154 | |
| Parameter | Year | SO-BT †,1 | BT-SE 2 | SE-AN 3 | AN-PM 4 | Total 5 |
|---|---|---|---|---|---|---|
| Water percolation (L m−2) | 1998 | 150 a | 45 b | 23 b | 6 b | 225 a |
| 1999 | 34 c | 52 a | 50 a | 13 ab | 152 b | |
| 2000 | 59 b | 20 c | 5 c | 21 a | 126 b | |
| NO3-N leaching (g N m−2) | 1998 | 3.84 a | 0.82 b | 0.10 b | 0.01 b | 4.78 a |
| 1999 | 0.65 b | 0.60 b | 0.23 a | 0.01 b | 1.50 b | |
| 2000 | 3.23 a | 1.27 a | 0.15 b | 0.28 a | 4.77 a |
| Parameter | ANR | NR | |||
|---|---|---|---|---|---|
| 1998 | 1999 | 2000 | 1999 | 2000 | |
| Albis (%) | 14.0 | 60.6 b,† | 29.9 | 54.0 b | 36.2 b |
| L94491 (%) | 22.5 | 74.2 a | 46.1 | 56.6 b | 47.1 a |
| Toronit (%) | 10.1 | 81.4 a | 47.6 | 63.7 a | 48.1 a |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrera, J.M.; Noulas, C.; Stamp, P.; Pellet, D. Little Potential of Spring Wheat Genotypes as a Strategy to Reduce Nitrogen Leaching in Central Europe. Agronomy 2016, 6, 29. https://doi.org/10.3390/agronomy6020029
Herrera JM, Noulas C, Stamp P, Pellet D. Little Potential of Spring Wheat Genotypes as a Strategy to Reduce Nitrogen Leaching in Central Europe. Agronomy. 2016; 6(2):29. https://doi.org/10.3390/agronomy6020029
Chicago/Turabian StyleHerrera, Juan Manuel, Christos Noulas, Peter Stamp, and Didier Pellet. 2016. "Little Potential of Spring Wheat Genotypes as a Strategy to Reduce Nitrogen Leaching in Central Europe" Agronomy 6, no. 2: 29. https://doi.org/10.3390/agronomy6020029
APA StyleHerrera, J. M., Noulas, C., Stamp, P., & Pellet, D. (2016). Little Potential of Spring Wheat Genotypes as a Strategy to Reduce Nitrogen Leaching in Central Europe. Agronomy, 6(2), 29. https://doi.org/10.3390/agronomy6020029







