Polypropylene Microplastics and Cadmium: Unveiling the Key Impacts of Co-Pollution on Wheat–Soil Systems from Multiple Perspectives
Abstract
1. Introduction
2. Materials and Methods
2.1. MPs and Their Characterization
2.2. Plant Used in This Study and Their Pretreatment
2.3. Seed Germination Experiment
2.3.1. Experimental Design and Procedure
2.3.2. Method of Index Determination
2.4. Pot Experiment
2.4.1. Soil Used in This Study
2.4.2. Experimental Design and Procedure
2.4.3. Determination and Analysis of Plant and Soil Indexes
- (1)
- Plant indexes
- (2)
- Soil indexes
- (3)
- Cd analysis
2.5. Statistical Analysis
3. Results
3.1. Microplastics Characterization
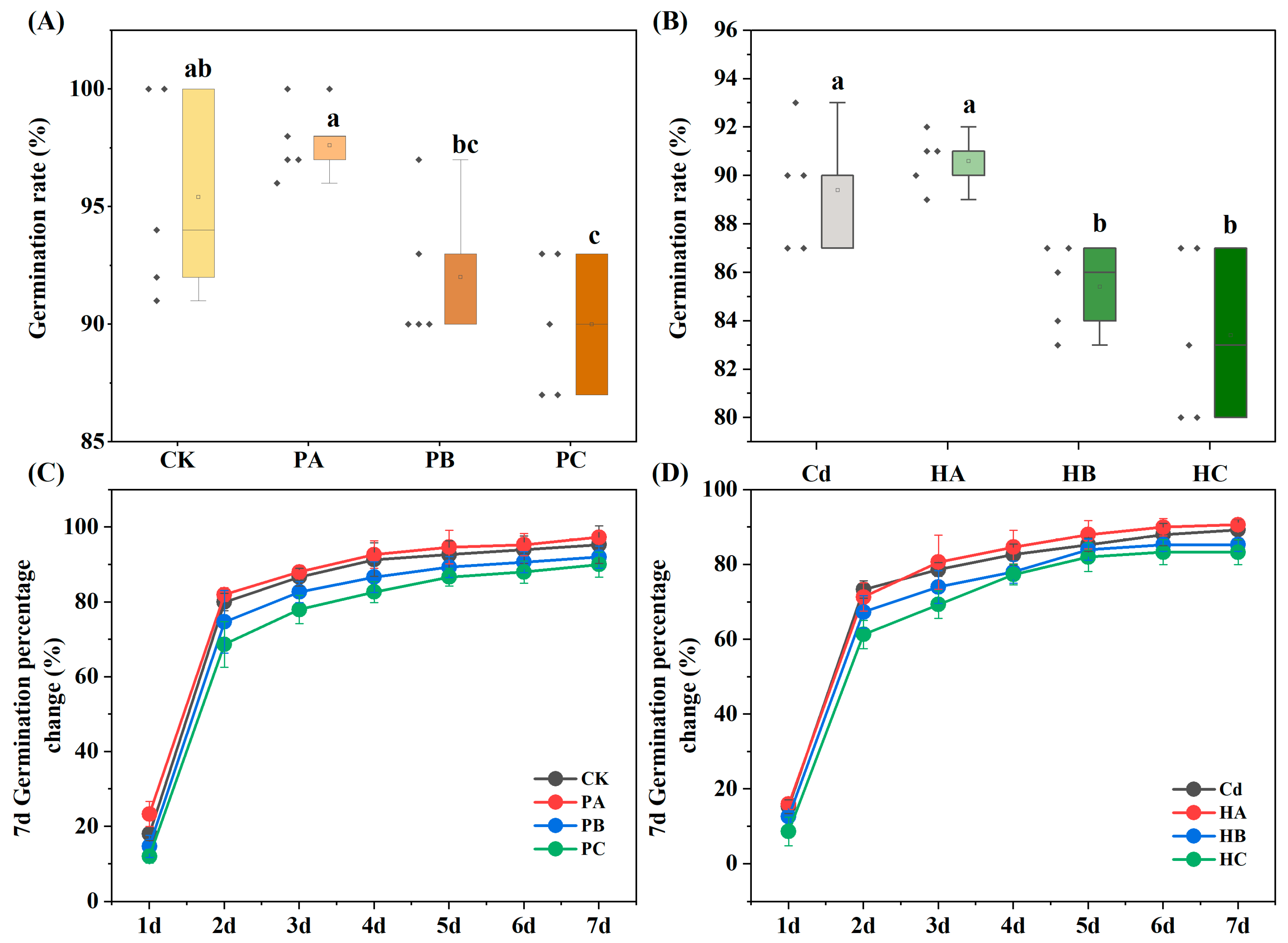
3.2. Seed Germination Rate
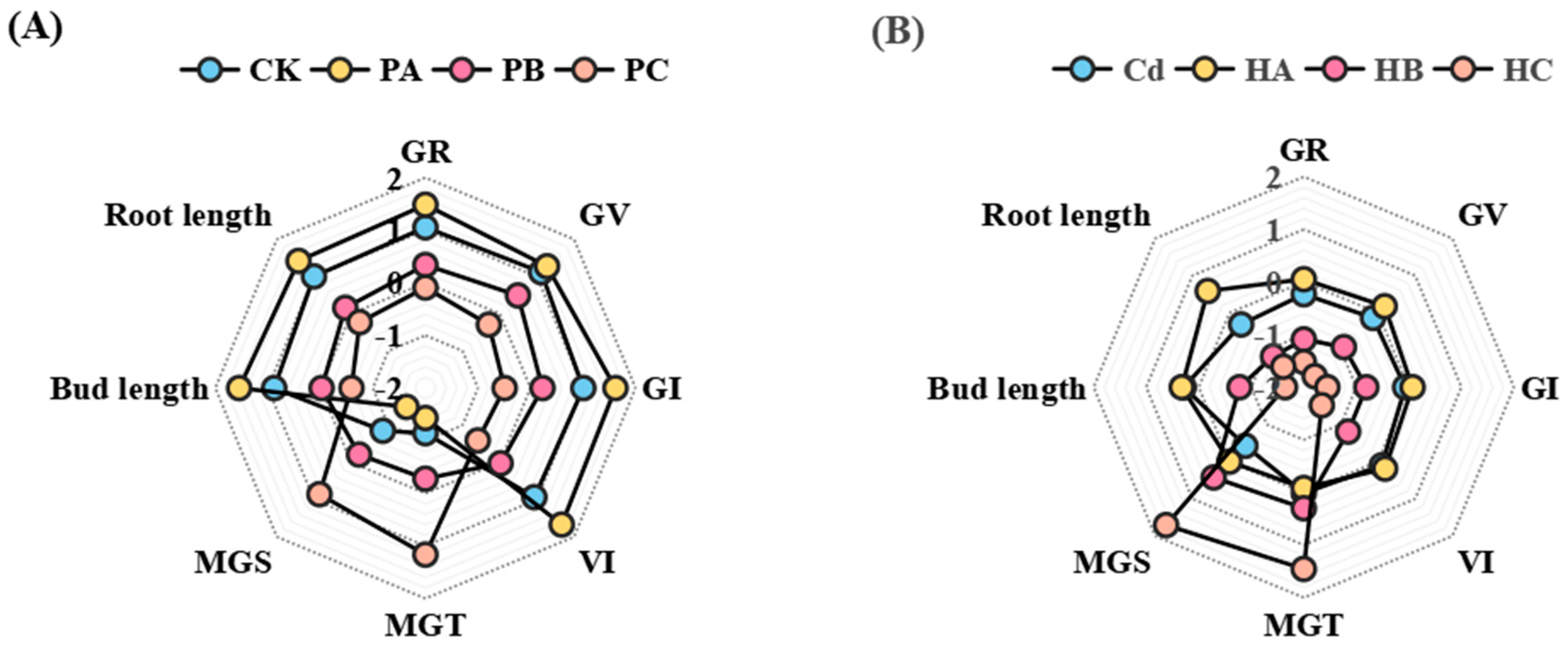
3.3. Seed Growth Characteristics
3.4. Interaction Between PP and Cd
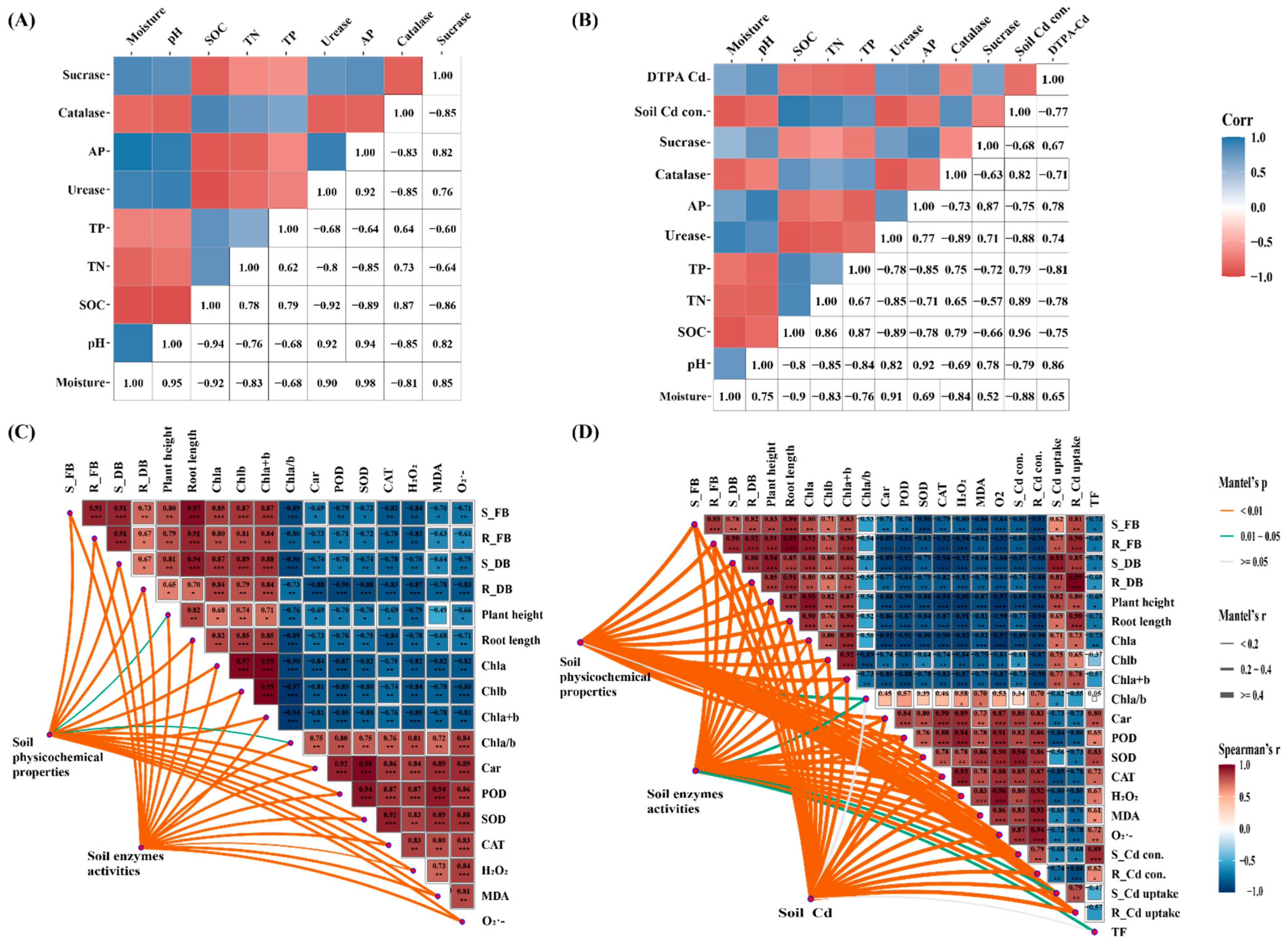
3.5. Soil Physicochemical Properties and Enzyme Activities
3.6. Plant Height, Root Length, Plant Biomass, and Chlorophyll Content
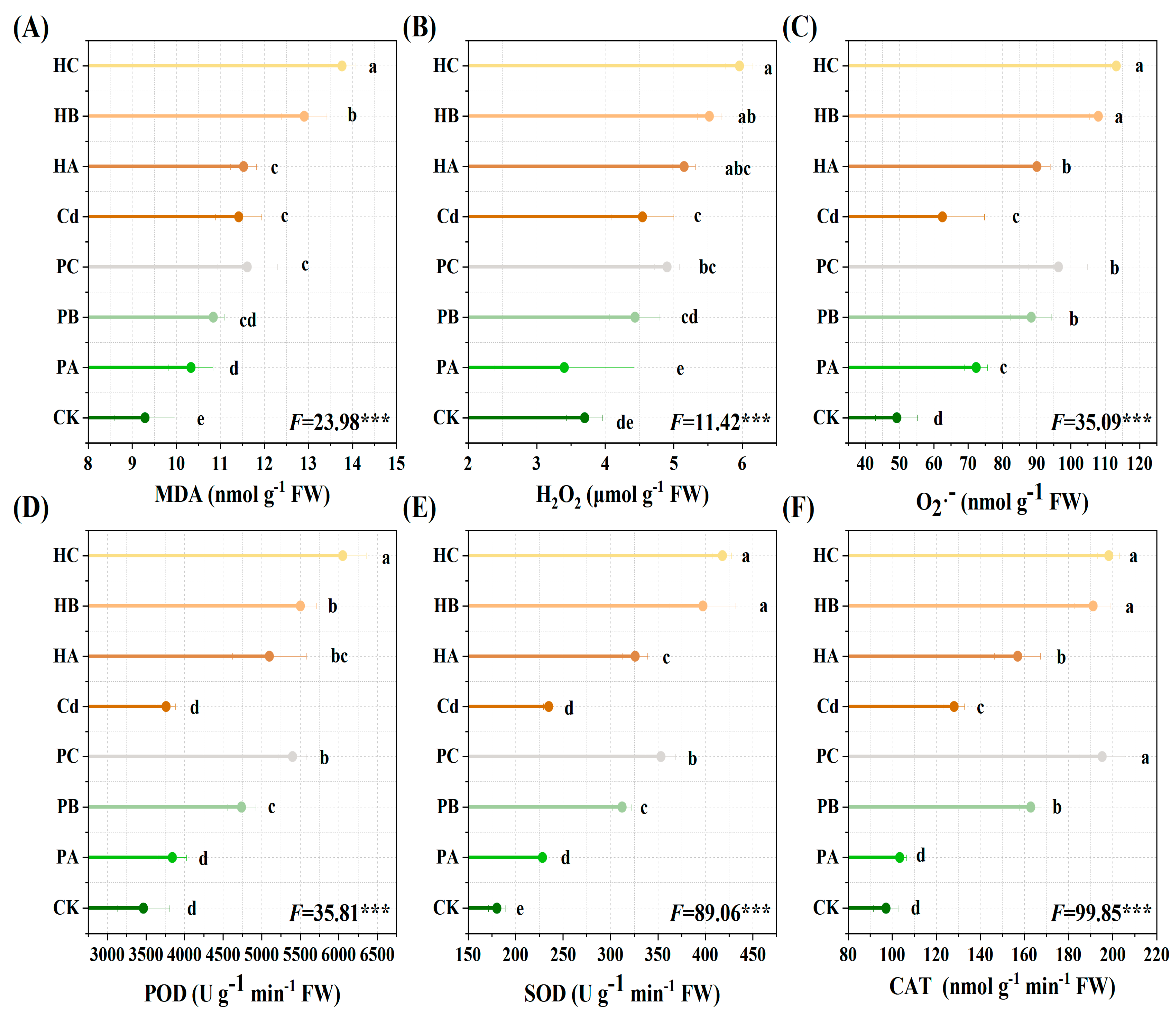
3.7. Plant Oxidative Damage and Antioxidant Enzyme Activity
3.8. Cd Concentration in Plant and Soil
3.9. Factors Affecting Cd Concentration in Plants
4. Discussion
4.1. Effects on Seed Germination
4.2. Effects on Soil Properties
4.3. Effects on Plant Growth
4.4. Driving Factors on Plant Cd Enrichment
5. Conclusions
- (1)
- The results of the seed germination experiment showed that the addition of low concentration PP could promote the growth of seeds. However, medium and high concentrations of PP had significant inhibitory effects on the growth characteristics of seeds. For PP + Cd co-pollution, the addition of low-concentration PP could partially alleviate the single stress of Cd. However, with the increase in PP concentration, the co-pollution showed stronger toxicity to the growth of wheat seeds than the single pollution. Moreover, the synergistic effect of PP and Cd was greater than the antagonistic effect; both of them aggravated the stress on wheat.
- (2)
- The results of the pot experiment showed that both single PP and combined Cd pollution had a significant impact on the soil microenvironment. It was manifested as reducing soil moisture and pH, affecting soil nutrient cycling, and inhibiting the activities of soil urease, sucrase, and alkaline phosphatase, while increasing the activity of catalase. And the combined pollution had a stronger adverse effect on the soil.
- (3)
- In addition, the MPs-Cd system significantly affected the physiological characteristics of plants. Specifically, compared with the CK treatment, the addition of low concentration PP alone promoted or had no significant effect on plant growth. However, with the increase in PP concentration, the biomass and chlorophyll content of plants decreased significantly, while carotenoids, oxidative damage, and antioxidant enzyme activities increased significantly. Moreover, PP + Cd co-pollution led to stronger phytotoxicity. In addition, PP exposure caused an increase in plant shoot and root Cd concentrations, promoting Cd transport from roots to shoots. Correlation heat maps and RDA analysis revealed that plant Cd concentration was significantly correlated with soil environmental factors and plant physiological indicators. Moreover, the results of the linear model (%) of relative importance indicated that soil pH and plant MDA content were important soil and plant variables affecting the increase in Cd concentration in plant tissues.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Thompson, R.C.; Swan, S.H.; Moore, C.J.; vom Saal, F.S. Our plastic age. Philos. Trans. R. Soc. B Biol. Sci. 2009, 364, 1973–1976. [Google Scholar] [CrossRef]
- Geyer, R.; Jambeck, J.R.; Law, K.L. Production, use, and fate of all plastics ever made. Sci. Adv. 2017, 3, e1700782. [Google Scholar] [CrossRef]
- Lim, X.Z. Microplastics are everywhere-but are they harmful? Nature 2021, 593, 22–25. [Google Scholar] [CrossRef]
- Nizzetto, L.; Futter, M.; Langaas, S. Are agricultural soils dumps for microplastics of urban origin? Environ. Sci. Technol. 2016, 50, 10777–10779. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Cui, W.Z.; Li, W.G.; Xu, S.; Sun, Y.H.; Xu, G.J.; Wang, F.Y. Effects of microplastics on cadmium accumulation by rice and arbuscular mycorrhizal fungal communities in cadmium-contaminated soil. J. Hazard. Mater. 2023, 442, 130102. [Google Scholar] [CrossRef] [PubMed]
- Khalid, N.; Aqeel, M.; Noman, A. Microplastics could be a threat to plants in terrestrial systems directly or indirectly. Environ. Pollut. 2020, 267, 115653. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Q.; Cui, Q.L.; Chen, L.; Zhu, X.Z.; Zhao, S.L.; Duan, C.J.; Zhang, X.C.; Song, D.X.; Fang, L.C. A critical review of microplastics in the soil-plant system: Distribution, uptake, phytotoxicity and prevention. J. Hazard. Mater. 2022, 424, 127750. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.Y.; Guo, R.; Zhang, S.W.; Sun, Y.H.; Wang, F.Y. Uptake and translocation of nano/microplastics by rice seedlings: Evidence from a hydroponic experiment. J. Hazard. Mater. 2022, 421, 126700. [Google Scholar] [CrossRef]
- Vethaak, A.D.; Legler, J. Microplastics and human health. Science 2021, 371, 672–674. [Google Scholar] [CrossRef]
- Geng, Z.; Zhao, B.; Duan, Y.; Xia, W.; Chu, J.; Yao, X. Phosphorus mitigates the adverse effects of microplastics pollution on wheat and maize: Impacts on growth, photosynthesis, and antioxidant defense. Environ. Exp. Bot. 2024, 228, 105993. [Google Scholar] [CrossRef]
- Wang, F.Y.; Zhang, X.Q.; Zhang, S.Q.; Zhang, S.W.; Sun, Y.H. Interactions of microplastics and cadmium on plant growth and arbuscular mycorrhizal fungal communities in an agricultural soil. Chemosphere 2020, 254, 126791. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.; Liu, G.; Huang, J.; Zeng, Z.; Zeng, Y.; Qing, T.; Zhang, P.; Feng, B. Comparison of the toxic effects of polystyrene and sulfonated polystyrene on wheat under cadmium stress. J. Hazard. Mater. 2024, 474, 134844. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Hao, S.; Li, P.; Du, Z.; Zhou, Y.; Wang, G.; Liang, Z.; Dou, M. Effects of Different Microplastics on Wheat’s (Triticum aestivum L.) Growth Characteristics and Rhizosphere Soil Environment. Plants 2024, 13, 3483. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Chen, S.; Li, L.; Liang, X.; Liang, F.; Lv, M. Effects of single and composite contamination of polylactic acid microplastics and cadmium on wheat seed germination before and after Ultraviolet aging. J. Agro-Environ. Sci. 2025, 44, 1477–1485. [Google Scholar] [CrossRef]
- Li, Q.J.; Yan, J.; Li, Y.L.; Liu, Y.W.; Andom, O.; Li, Z.J. Microplastics alter cadmium accumulation in different soil-plant systems: Revealing the crucial roles of soil bacteria and metabolism. J. Hazard. Mater. 2024, 474, 134768. [Google Scholar] [CrossRef]
- Li, F.P.; Yang, X.Y.; Zhang, Z.M.; Jiang, Y.C.; Gong, Y.F. Behaviour, ecological impacts of microplastics and cadmium on soil systems: A systematic review. Environ. Technol. Innov. 2024, 35, 103637. [Google Scholar] [CrossRef]
- Zong, X.Y.; Zhang, J.J.; Zhu, J.W.; Zhang, L.Y.; Jiang, L.J.; Yin, Y.; Guo, H.Y. Effects of polystyrene microplastic on uptake and toxicity of copper and cadmium in hydroponic wheat seedlings (Triticum aestivum L.). Ecotoxicol. Environ. Saf. 2021, 217, 112217. [Google Scholar] [CrossRef]
- Zeb, A.; Liu, W.T.; Meng, L.Z.; Lian, J.P.; Wang, Q.; Lian, Y.H.; Chen, C.H.; Wu, J.N. Effects of polyester microfibers (PMFs) and cadmium on lettuce (Lactuca sativa) and the rhizospheric microbial communities: A study involving physio-biochemical properties and metabolomic profiles. J. Hazard. Mater. 2022, 424, 127405. [Google Scholar] [CrossRef]
- Wang, F.L.; Wang, X.X.; Song, N.N. Polyethylene microplastics increase cadmium uptake in lettuce (Lactuca sativa L.) by altering the soil microenvironment. Sci. Total Environ. 2021, 784, 147133. [Google Scholar] [CrossRef]
- Cao, Y.X.; Zhao, M.J.; Ma, X.Y.; Song, Y.W.; Zuo, S.H.; Li, H.H.; Deng, W.Z. A critical review on the interactions of microplastics with heavy metals: Mechanism and their combined effect on organisms and humans. Sci. Total Environ. 2021, 788, 147620. [Google Scholar] [CrossRef]
- Wang, F.Y.; Zhang, X.Q.; Zhang, S.Q.; Zhang, S.W.; Adams, C.A.; Sun, Y.H. Effects of co-contamination of microplastics and Cd on plant growth and Cd accumulation. Toxics 2020, 8, 36. [Google Scholar] [CrossRef]
- Dong, Y.M.; Gao, M.L.; Qiu, W.W.; Song, Z.G. Effect of microplastics and arsenic on nutrients and microorganisms in rice rhizosphere soil. Ecotoxicol. Environ. Saf. 2021, 211, 111899. [Google Scholar] [CrossRef]
- Khalid, N.; Aqeel, M.; Noman, A.; Khan, S.M.; Akhter, N. Interactions and effects of microplastics with heavy metals in aquatic and terrestrial environments. Environ. Pollut. 2021, 290, 118104. [Google Scholar] [CrossRef]
- Chen, S.; Feng, T.; Lin, X.; Hou, Z.; Chao, L.; Zhang, X.; Liu, Y. Effects of microplastics and cadmium on the soil-wheat system as single and combined contaminants. Plant Physiol. Biochem. 2023, 196, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Farooq, U.; Kozinski, J.A.; Khan, M.A.; Athar, M. Biosorption of heavy metal ions using wheat based biosorbents--a review of the recent literature. Bioresour. Technol. 2010, 101, 5043–5053. [Google Scholar] [CrossRef] [PubMed]
- Fu, D.; Zhang, Q.; Fan, Z.; Qi, H.; Peng, L. Aged microplastics Polyvinyl chloride interact with copper and cause oxidative stress towards microalgae Chlorella vulgaris. Aquat. Toxicol. 2019, 216, 105319. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Z.; Yang, L.; Li, J.; Fu, D.D.; Hu, W.W.; Fan, Z.Q.; Peng, L.C. Single and combined effects of microplastics and cadmium on the germination characteristics of rice seeds. J. Agro-Environ. Sci. 2021, 40, 44–53. [Google Scholar] [CrossRef]
- Min, K.; Kim, G.; Lee, H.; Kim, Y.-K.; Lee, S.-E.; Lee, S.-R. Vulnerability of Brassica oleracea L. (cabbage) grown in microplastic-contaminated soil to extreme climatic events associated with freeze-thaw. Environ. Exp. Bot. 2025, 232, 106110. [Google Scholar] [CrossRef]
- Liu, Y.; Li, B.; Zhou, J.; Li, D.; Liu, Y.; Wang, Y.; Huang, W.; Ruan, Z.; Yao, J.; Qiu, R. Effects of naturally aged microplastics on arsenic and cadmium accumulation in lettuce: Insights into rhizosphere microecology. J. Hazard. Mater. 2025, 486, 136988. [Google Scholar] [CrossRef]
- Kalembasa, S.J.; Jenkinson, D.S. A comparative study of titrimetric and gravimetric methods for the determination of organic carbon in soil. J. Sci. Food Agric. 1973, 24, 1085–1090. [Google Scholar] [CrossRef]
- Cui, Y.X.; Wang, X.; Zhang, X.C.; Ju, W.L.; Duan, C.J.; Guo, X.B.; Wang, Y.Q.; Fang, L.C. Soil moisture mediates microbial carbon and phosphorus metabolism during vegetation succession in a semiarid region. Soil Biol. Biochem. 2020, 147, 107814. [Google Scholar] [CrossRef]
- Duan, C.J.; Fang, L.C.; Yang, C.L.; Chen, W.B.; Cui, Y.X.; Li, S.Q. Reveal the response of enzyme activities to heavy metals through in situ zymography. Ecotox. Environ. Saf. 2018, 156, 106–115. [Google Scholar] [CrossRef]
- He, M.; Feng, Z.; Xu, Y.; Ding, H.; Ying, C.; Cai, Y.; Zhang, H. Macro- and microplastic leachates show a slightly toxic effect on seed germination of cotton. Chemosphere 2023, 335, 139081. [Google Scholar] [CrossRef]
- Lian, J.P.; Wu, J.N.; Xiong, H.X.; Zeb, A.; Yang, T.Z.; Su, X.M.; Su, L.J.; Liu, W.T. Impact of polystyrene nanoplastics (PSNPs) on seed germination and seedling growth of wheat (Triticum aestivum L.). J. Hazard. Mater. 2020, 385, 121620. [Google Scholar] [CrossRef] [PubMed]
- Bosker, T.; Bouwman, L.J.; Brun, N.R.; Behrens, P.; Vijver, M.G. Microplastics accumulate on pores in seed capsule and delay germination and root growth of the terrestrial vascular plant Lepidium sativum. Chemosphere 2019, 226, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.E.; Pearce, C.I.; Sanguinet, K.A.; Hu, D.H.; Chrisler, W.B.; Kim, Y.M.; Wang, Z.; Flury, M. Polystyrene nano- and microplastic accumulation at Arabidopsis and wheat root cap cells, but no evidence for uptake into roots. Environ. Sci. Nano 2020, 7, 1942–1953. [Google Scholar] [CrossRef]
- Wang, F.Y.; Wang, Q.L.; Adams, C.A.; Sun, Y.H.; Zhang, S.W. Effects of microplastics on soil properties: Current knowledge and future perspectives. J. Hazard. Mater. 2022, 424, 127531. [Google Scholar] [CrossRef]
- Wan, Y.; Wu, C.X.; Xue, Q.; Hui, X.M.N. Effects of plastic contamination on water evaporation and desiccation cracking in soil. Sci. Total Environ. 2019, 654, 576–582. [Google Scholar] [CrossRef]
- Rillig, M.C. Microplastic disguising as soil carbon storage. Environ. Sci. Technol. 2018, 52, 6079–6080. [Google Scholar] [CrossRef]
- de Souza Machado, A.A.; Lau, C.W.; Kloas, W.; Bergmann, J.; Bacheher, J.B.; Faltin, E.; Becker, R.; Goerlich, A.S.; Rillig, M.C. Microplastics can change soil properties and affect plant performance. Environ. Sci. Technol. 2019, 53, 6044–6052. [Google Scholar] [CrossRef]
- Seeley, M.E.; Song, B.; Passie, R.; Hale, R.C. Microplastics affect sedimentary microbial communities and nitrogen cycling. Nat. Commun. 2020, 11, 2372. [Google Scholar] [CrossRef]
- Zhang, P.; Yuan, Y.; Zhang, J.; Wen, T.; Wang, H.; Qu, C.; Tan, W.; Xi, B.; Hui, K.; Tang, J. Specific response of soil properties to microplastics pollution: A review. Environ. Res. 2023, 232, 116427. [Google Scholar] [CrossRef]
- Yu, H.; Fan, P.; Hou, J.H.; Dang, Q.L.; Cui, D.Y.; Xi, B.D.; Tan, W.B. Inhibitory effect of microplastics on soil extracellular enzymatic activities by changing soil properties and direct adsorption: An investigation at the aggregate-fraction level. Environ. Pollut. 2020, 267, 115544. [Google Scholar] [CrossRef] [PubMed]
- Li, G.L.; Cui, X.R.; Tariq, M.; Khan, I.; Khan, A.R.; Al Obaid, S.; Ansari, M.J.; Zhou, H.; Iqbal, B.; Zhao, X. Microplastic and cadmium contamination: Impact on the soil by inhibiting the growth of pak choi (Brassica rapa subsp. chinensis). Process Saf. Environ. Prot. 2024, 189, 714–727. [Google Scholar] [CrossRef]
- Li, K.; He, Q.H.; Zhu, J.; Wang, J.Y.; Sun, C.; Tan, A.; Zhao, X.Q.; Peng, Y.H.; Huang, C.; Cai, J.J.; et al. Responses of microbial communities to the addition of different types of microplastics in agricultural soils. Environ. Pollut. 2025, 364, 125220. [Google Scholar] [CrossRef]
- Fan, P.; Tan, W.; Yu, H. Effects of different concentrations and types of microplastics on bacteria and fungi in alkaline soil. Ecotoxicol. Environ. Saf. 2022, 229, 113045. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Lehmann, A.; Yang, G.; Leifheit, E.F.; Rillig, M.C. Effects of Microplastic Fibers on Soil Aggregation and Enzyme Activities Are Organic Matter Dependent. Front. Environ. Sci. 2021, 9, 650155. [Google Scholar] [CrossRef]
- Zhang, Z.Q.; Li, Y.; Qiu, T.Y.; Duan, C.J.; Chen, L.; Zhao, S.L.; Zhang, X.C.; Fang, L.C. Microplastics addition reduced the toxicity and uptake of cadmium to Brassica chinensis L. Sci. Total Environ. 2022, 852, 158353. [Google Scholar] [CrossRef]
- Lee, Y.K.; Murphy, K.R.; Hur, J. Fluorescence signatures of dissolved organic matter leached from microplastics: Polymers and additives. Environ. Sci. Technol. 2020, 54, 11905–11914. [Google Scholar] [CrossRef]
- Men, C.; Xie, Z.; Li, K.; Xing, X.; Li, Z.; Zuo, J. Single and combined effect of polyethylene microplastics (virgin and naturally aged) and cadmium on pakchoi (Brassica rapa subsp. chinensis) under different growth stages. Sci. Total Environ. 2024, 951, 175602. [Google Scholar] [CrossRef]
- Wang, J.L.; Liu, W.T.; Wang, X.; Zeb, A.; Wang, Q.; Mo, F.; Shi, R.Y.; Liu, J.Z.; Yu, M.; Li, J.T. Assessing stress responses in potherb mustard (Brassica juncea var. multiceps) exposed to a synergy of microplastics and cadmium: Insights from physiology, oxidative damage, and metabolomics. Sci. Total Environ. 2024, 907, 167920. [Google Scholar] [CrossRef]
- Yu, Y.F.; Li, J.; Song, Y.; Zhang, Z.Y.; Yu, S.G.; Xu, M.L.; Zhao, Y.Y. Stimulation versus inhibition: The effect of microplastics on pak choi growth. Appl. Soil Ecol. 2022, 177, 104505. [Google Scholar] [CrossRef]
- Jia, H.; Wu, D.; Yu, Y.; Han, S.; Sun, L.; Li, M. Impact of microplastics on bioaccumulation of heavy metals in rape (Brassica napus L.). Chemosphere 2022, 288, 132576. [Google Scholar] [CrossRef] [PubMed]
- Adil, M.F.; Sehar, S.; Han, Z.; Lwalaba, J.L.W.; Jilani, G.; Zeng, F.; Chen, Z.-H.; Shamsi, I.H. Zinc alleviates cadmium toxicity by modulating photosynthesis, ROS homeostasis, and cation flux kinetics in rice. Environ. Pollut. 2020, 265, 114979. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Zhao, W.; Liang, Q.; Cai, M.; Fan, X.; Hu, S.; Zhu, Y.; Xie, H.; Peng, C.; Liu, J. Polystyrene microplastics enhanced the toxicity of cadmium to rice seedlings: Evidence from rice growth, physiology, and element metabolism. Sci. Total Environ. 2024, 945, 173931. [Google Scholar] [CrossRef]
- Dong, Y.M.; Gao, M.L.; Song, Z.G.; Qiu, W.W. Microplastic particles increase arsenic toxicity to rice seedlings. Environ. Pollut. 2020, 259, 113892. [Google Scholar] [CrossRef]
- Liu, J.; Gai, L.; Zong, H. Foliage application of chitosan alleviates the adverse effects of cadmium stress in wheat seedlings (Triticum aestivum L.). Plant Physiol. Biochem. 2021, 164, 115–121. [Google Scholar] [CrossRef]
- Gao, M.L.; Liu, Y.; Song, Z.G. Effects of polyethylene microplastic on the phytotoxicity of di-n-butyl phthalate in lettuce (Lactuca sativa L. var. ramosa Hort). Chemosphere 2019, 237, 124482. [Google Scholar] [CrossRef]
- Li, Y.; Chen, Y.; Li, P.; Huang, H.; Xue, K.; Cai, S.; Liao, X.; Jin, S.; Zheng, D. Microplastics in soil affect the growth and physiological characteristics of Chinese fir and Phoebe bournei seedlings. Environ. Pollut. 2024, 358, 124503. [Google Scholar] [CrossRef]
- Pinto-Poblete, A.; Retamal-Salgado, J.; Lopez, M.D.; Zapata, N.; Sierra-Almeida, A.; Schoebitz, M. Combined effect of microplastics and Cd alters the enzymatic activity of soil and the productivity of strawberry plants. Plants 2022, 11, 536. [Google Scholar] [CrossRef]
- Yang, R.C.; Cheng, L.; Li, Z.Q.; Cui, Y.L.; Liu, J.W.; Xu, D.; Liu, S.J.; Lin, Z.; Chen, J.G.; Zhang, Y.Q. Mechanism of microplastics in the reduction of cadmium toxicity in tomato. Ecotox. Environ. Saf. 2025, 289, 117621. [Google Scholar] [CrossRef]
- Huang, F.Y.; Hu, J.Z.; Chen, L.; Wang, Z.; Sun, S.Y.; Zhang, W.M.; Jiang, H.; Luo, Y.; Wang, L.; Zeng, Y.; et al. Microplastics may increase the environmental risks of Cd via promoting Cd uptake by plants: A meta-analysis. J. Hazard. Mater. 2023, 448, 130887. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Q.; Zhao, S.L.; Chen, L.; Duan, C.J.; Zhang, X.C.; Fang, L.C. A review of microplastics in soil: Occurrence, analytical methods, combined contamination and risks. Environ. Pollut. 2022, 306, 119374. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.W.; Han, B.; Sun, Y.H.; Wang, F.Y. Microplastics influence the adsorption and desorption characteristics of Cd in an agricultural soil. J. Hazard. Mater. 2020, 388, 121775. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, S.; Moore, F.; Keshavarzi, B.; Hopke, P.K.; Naidu, R.; Rahman, M.M.; Oleszczuk, P.; Karimi, J. PET-microplastics as a vector for heavy metals in a simulated plant rhizosphere zone. Sci. Total Environ. 2020, 744, 140984. [Google Scholar] [CrossRef]
- Takahashi, R.; Ishimaru, Y.; Shimo, H.; Ogo, Y.; Senoura, T.; Nishizawa, N.K.; Nakanishi, H. The OsHMA2 transporter is involved in root-to-shoot translocation of Zn and Cd in rice. Plant Cell Environ. 2012, 35, 1948–1957. [Google Scholar] [CrossRef]
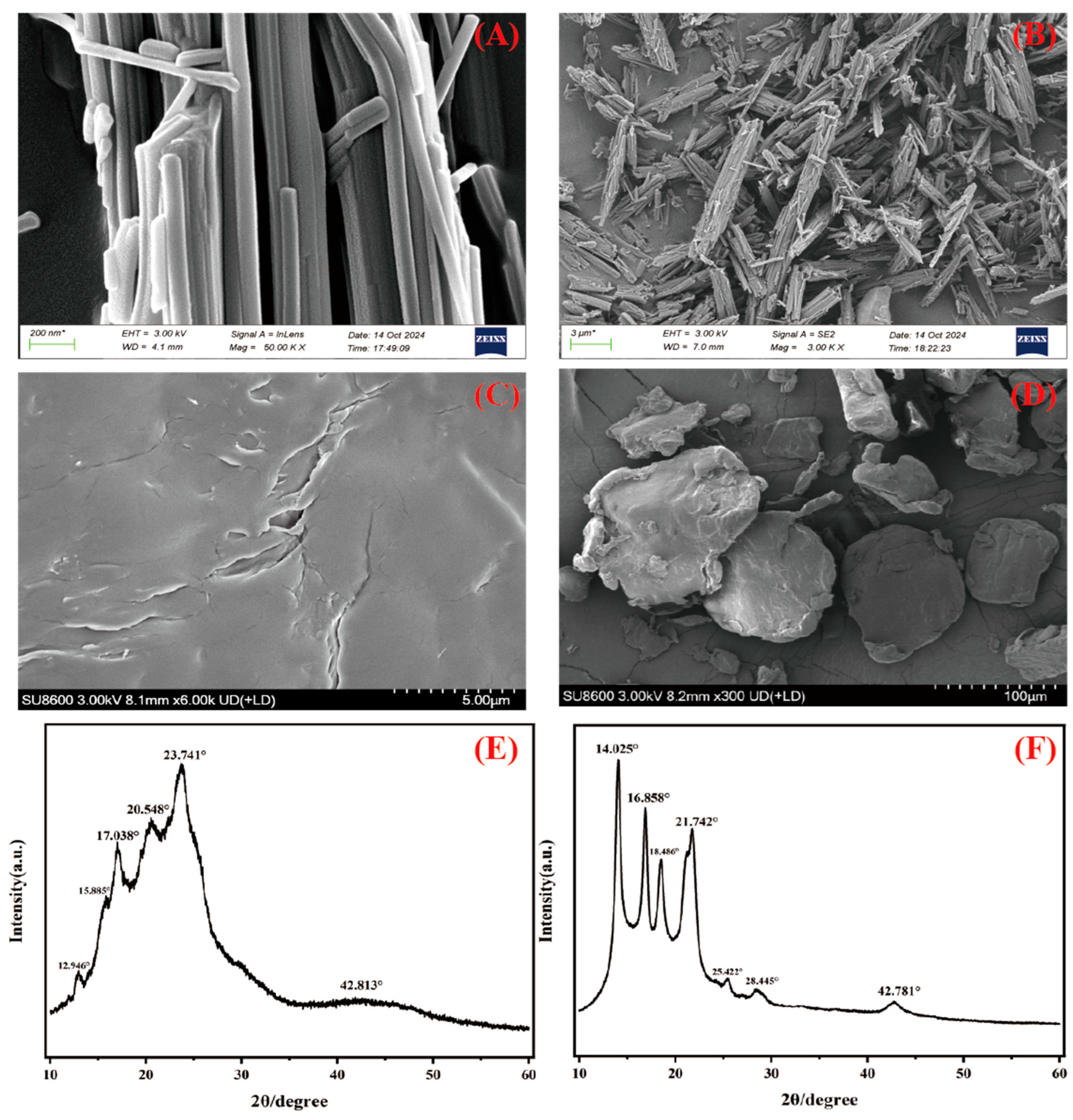

| Type | Treatment | mg L−1 | GV | GI | VI | MGT | MGS |
|---|---|---|---|---|---|---|---|
| PP | CK | 0 | 0.87 ± 0.02 ab | 47.26 ± 1.24 b | 609.81 ± 30.75 b | 2.14 ± 0.27 bc | 4.48 ± 0.02 cd |
| PA | 50 | 0.88 ± 0.02 a | 49.67 ± 0.94 a | 694.52 ± 38.67 a | 2.11 ± 0.11 c | 4.45 ± 0.02 d | |
| PB | 200 | 0.83 ± 0.03 bc | 44.20 ± 2.32 c | 505.05 ± 29.09 c | 2.23 ± 0.21 abc | 4.51 ± 0.06 bc | |
| PC | 500 | 0.78 ± 0.04 cd | 41.36 ± 1.78 de | 434.30 ± 57.66 d | 2.38 ± 0.11 ab | 4.56 ± 0.03 ab | |
| PP + Cd | Cd | 0 | 0.79 ± 0.02 cd | 43.02 ± 0.79 cd | 513.98 ± 49.17 c | 2.26 ± 0.16 abc | 4.50 ± 0.03 bc |
| HA | 50 | 0.81 ± 0.07 c | 43.58 ± 1.50 c | 524.07 ± 66.61 c | 2.25 ± 0.17 abc | 4.52 ± 0.03 bc | |
| HB | 200 | 0.74 ± 0.04 de | 40.11 ± 0.85 e | 409.59 ± 41.82 d | 2.29 ± 0.16 abc | 4.54 ± 0.05 b | |
| HC | 500 | 0.69 ± 0.04 e | 37.19 ± 0.80 f | 327.98 ± 20.07 e | 2.41 ± 0.06 a | 4.60 ± 0.03 a | |
| Factor (Df) | - | - | 13.36 | 40.71 | 33.93 | 1.94 | 8.08 |
| p | - | - | *** | *** | *** | ns | *** |
| Treatments | Cd Concentration (mg kg−1) | Total Uptake (μg pot−1) | TF | Soil Total Cd Concentration | DTPA-Cd Concentration | ||
|---|---|---|---|---|---|---|---|
| Shoot | Root | Shoot | Root | (mg kg−1) | (mg kg−1) | ||
| Cd | 0.64 ± 0.01 c | 2.82 ± 0.05 c | 0.56 ± 0.02 a | 1.33 ± 0.09 a | 0.23 ± 0.01 c | 0.64 ± 0.01 d | 0.36 ± 0.01 a |
| HA | 0.67 ± 0.01 b | 2.86 ± 0.02 bc | 0.48 ± 0.03 ab | 1.1 ± 0.15 b | 0.23 ± 0.01 bc | 0.68 ± 0.01 c | 0.36 ± 0.01 a |
| HB | 0.78 ± 0.02 a | 2.91 ± 0.02 b | 0.49 ± 0.07 ab | 1.01 ± 0.12 bc | 0.27 ± 0.01 a | 0.70 ± 0.01 b | 0.34 ± 0.01 b |
| HC | 0.79 ± 0.01 a | 3.23 ± 0.04 a | 0.41 ± 0.03 b | 0.84 ± 0.08 c | 0.24 ± 0.01 b | 0.72 ± 0.01 a | 0.34 ± 0.01 b |
| Factor (Df) | 71.71 | 82.92 | 6.23 | 9.81 | 23.47 | 33.38 | 26.26 |
| p | *** | *** | * | ** | *** | *** | *** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, Z.; He, H.; Chang, N.; Duan, C. Polypropylene Microplastics and Cadmium: Unveiling the Key Impacts of Co-Pollution on Wheat–Soil Systems from Multiple Perspectives. Agronomy 2025, 15, 2013. https://doi.org/10.3390/agronomy15082013
Zhang Z, He H, Chang N, Duan C. Polypropylene Microplastics and Cadmium: Unveiling the Key Impacts of Co-Pollution on Wheat–Soil Systems from Multiple Perspectives. Agronomy. 2025; 15(8):2013. https://doi.org/10.3390/agronomy15082013
Chicago/Turabian StyleZhang, Zhiqin, Haoran He, Nan Chang, and Chengjiao Duan. 2025. "Polypropylene Microplastics and Cadmium: Unveiling the Key Impacts of Co-Pollution on Wheat–Soil Systems from Multiple Perspectives" Agronomy 15, no. 8: 2013. https://doi.org/10.3390/agronomy15082013
APA StyleZhang, Z., He, H., Chang, N., & Duan, C. (2025). Polypropylene Microplastics and Cadmium: Unveiling the Key Impacts of Co-Pollution on Wheat–Soil Systems from Multiple Perspectives. Agronomy, 15(8), 2013. https://doi.org/10.3390/agronomy15082013





