Oligonychus perseae (Tetranychidae) Invasion in the Canary Islands: History, Management and Current Situation
Abstract
1. Introduction
2. Persea Mite Establishment, Distribution and Main Host Plants in the Canary Islands
3. Persea Mite Management Approaches and Their Limitations
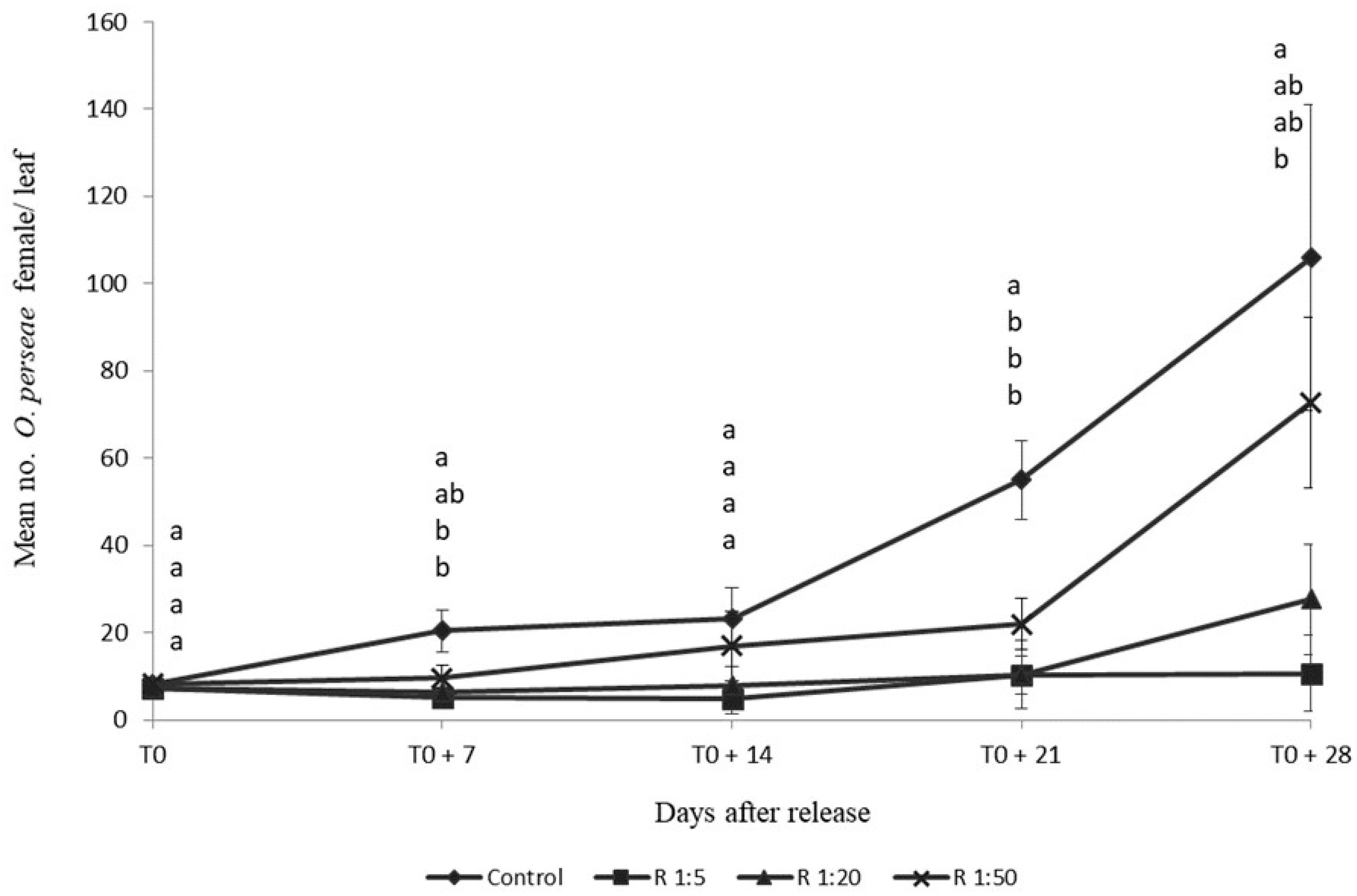
4. Monitoring of O. perseae
5. Preventive Cultural Management
6. Biological Control
7. Chemical Control of Oligonychus perseae
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAOSTAT. Food and Agriculture Organization of the United Nations. Available online: http://www.fao.org/faostat/en/ (accessed on 25 April 2023).
- MAPA. Statistical Yearbook of Agrarian Markets 2021; Ministerio de Agricultura, Pesca y Alimentación: Madrid, Spain, 2021; Available online: https://www.mapa.gob.es/en/estadistica/temas/publicaciones/anuario-de-estadistica/default.aspx (accessed on 25 April 2023).
- Gobierno de Canarias. Anuario Estadístico de Agricultura, Ganadería, Pesca y Agua de Canarias. 2021. Available online: http://www.gobiernodecanarias.org/agricultura/anuarioestadistico/ (accessed on 27 April 2023).
- Migeon, A.; Dorkeld, F. Spider Mites Web: A Comprehensive Database for the Tetranychidae. 2022. Available online: https://www1.montpellier.inra.fr/CBGP/spmweb (accessed on 8 November 2022).
- Vacante, V. The Handbook of Mites of Economic Plants: Identification, Bio-Ecology and Control; CABI Publishing: Wallingford, UK, 2016; 890p. [Google Scholar]
- Orozco, J.; Duque, M.; Mesa, N.C. Efecto de la temperatura sobre la tabla de vida de Oligonychus yothersi en Coffea arabica. Rev. Cent. Nac. Investig. Café Chinchiná Caldas Colomb. 1990, 41, 5–18. [Google Scholar]
- Chavez-Espinoza, A.; Rodríguez, H.; Estrada-Venegas, E. Biology of Oligonychus cubensis Livschitz (Acari: Tetranychidae) in avocado (Persea americana Miller). Rev. Prot. Veg. 2017, 32, 1–7. [Google Scholar]
- Baker, E.W. Spider mites (Tetranychidae: Acarina) from Southeast Asia and Japan. Coop. Econ. Insect Rep. 1975, 25, 911–922. [Google Scholar]
- Pande, Y.D.; Carnero, A.; Hernández, M. Notes on biological observations on some unrecorded species of phytophagus and predatory mites in Canary Islands. Investig. Agrar. Prod. Proteccióon Veg. 1989, 4, 275–281. [Google Scholar]
- Hernández García, M. Control Natural en Cultivo Biológico de Aguacate. Master’s thesis, Trabajo Fin de Carrera de Ingeniero Técnico Agrícola, Escuela Técnica Superior de Ingeniería Agraria; Universidad de La Laguna: La Laguna, Spain, 1987. [Google Scholar]
- Mushtaq, H.M.S.; Alatawi, F.J.; Kamran, M.; Flechtmann, C.H.W. The genus Oligonychus Berlese (Acari, Prostigmata, Tetranychidae): Taxonomic assessment and a key to subgenera, species groups, and subgroups. ZooKeys 2021, 1079, 89–127. [Google Scholar] [CrossRef] [PubMed]
- Baker, E.W.; Tuttle, D.M. A Guide to the Spider Mites (Tetranychidae) of the United States; Indira Publishing House: West Bloomfield, MI, USA, 1994; ISBN 9780930337124. [Google Scholar]
- European and Mediterranean Plan Protection Organization (EPPO). EPPO Global Database. Oligonychus perseae (OLIGPA); EPPO: Paris, France, 2023; Available online: https://gd.eppo.int/taxon/OLIGPA/categorization (accessed on 24 May 2023).
- Palevsky, E.; Maoz, Y.; Gal, S.; Argov, Y.; Zilberstein, M.; Noy, M.; Izhar, Y.; Alchanatis, V. Developing an action threshold for the persea mite on avocado. In Proceedings of the 6th World Avocado Congress (Actas VI Congreso Mundial del Aguacate), Viña Del Mar, Chile, 12–16 November 2007. [Google Scholar]
- Alcázar, M.D.; Aranda, G.; Márquez, A.L.; Sánchez, L.; Ruiz, C. Oligonychus perseae (Acari; Tetranychidae), una nueva plaga en el aguacate en el Sur de España. In Proceedings of the IV Congreso Nacional de Entomología Aplicada, Braganza, Portugal, 17–21 October 2005; p. 213. [Google Scholar]
- Borges, P.A.V.; Aguiar, A.M.F.; Boieiro, M.; Carles-Tolra, M.S. A List of the Terrestrial Fungi, Flora and Fauna of Madeira and Selvagens Archipelagos; Direcção Reg do Ambient da Madeira Univ dos Açores, Funchal Angra do Heroísmo: Horta, Portugal, 2008; Port: 440. [Google Scholar]
- Hernández Suárez, E.; Torres, E.; Velásquez Hernández, Y.; Perera González, S. Araña Cristalina del Aguacate. Nueva Plaga en Canarias; Cabildo Insular de Tenerife, Servicio Técnico de Agricultura y Desarrollo Rural: Santa Cruz de Tenerife, Spain, 2007; 6p. [Google Scholar]
- MAPA. Informe de la reunión del grupo de trabajo de cítricos y subtropicales. Ministerio de Agricultura, Pesca y Alimentación. In Proceedings of the España En: Reuniones Anuales de los Grupos de Trabajo Fitosanitarios, Tolosa, Spain, 21–26 February 2006; pp. 67–92. [Google Scholar]
- Torres, E.; Perera, S.; Ramos, C.; Álvarez, C.; Carnero, A.; Boyero, J.R.; Vela, J.M.; Wong, M.E.; Hernández-Suárez, E. Avances en el Manejo Integrado de Oligonychus perseae Tuttle, Baker & Abatiello en Canarias; Manual Técnico No 2; Instituto Canario de Investigaciones Agrarias: Santa Cruz de Tenerife, Spain, 2018; 70p. [Google Scholar]
- Naves, P.; Nóbrega, F.; Auger, P. Updated and annotated review of Tetranychidae occurring in mainland Portugal, the Azores, and Madeira Archipelagos. Acarologia 2021, 61, 380–393. [Google Scholar] [CrossRef]
- Zappalà, L.; Kreiter, S.; Russo, A.; Tropea Garzia, G.; Auger, P. First record of the Persea Mite Oligonychus perseae (Acari: Tetranychidae) in Italy with a review of the literature. Int. J. Acarol. 2015, 41, 97–99. [Google Scholar] [CrossRef]
- Smaili, M.C.; Benyahia, H. Alerte! Invasion et la recrudescence des dégâts d’un nouveau ravageur émergent sur avocatier au Maroc: Oligonychus perseae (Acari: Tetranychidae) [Alarm! The invasion of a new emerging pest in Morocco’s avocado orchards and its increasing damages]. Agric. Du Maghreb 2018, 115, 88–89. (In French) [Google Scholar]
- Auger, P.; Navia, D.; Migeon, A. Three new alien spider mites (Prostigmata, Tetranychidae) from south-eastern France. Acarologia 2023, 63, 826–833. [Google Scholar] [CrossRef]
- Aponte, O.; McMurtry, J.A. Damage on “Hass” avocado leaves, webbing and nesting behavior of Oligonychus perseae (Acari: 491 Tetranychidae). Exp. Appl. Acarol. 1997, 21, 265–272. [Google Scholar] [CrossRef]
- Bender, G.S. A new mite problem in avocados. Calif. Avocado Soc. Yearb. 1993, 77, 73–77. [Google Scholar]
- Hoddle, M.S. Biology and management of the Persea Mite. Calif. Avocado Soc. Yearb. 1998, 82, 75–85. [Google Scholar]
- Kerguelen, V.; Hoddle, M.S. Biological control of Oligonychus perseae (Acari: Tetranychidae) on avocado: I. Evaluating release timings, recovery and efficacy of six commercially available phytoseiids. Int. J. Acarol. 1999, 25, 211–219. [Google Scholar] [CrossRef]
- Torres, E.; Álvarez-Acosta, C.; Del Pino, M.; Wong, M.E.; Boyero, J.R.; Hernández-Sárez, E.; Vela, J.M. Economic Impact of the Persea Mite in Spanish Avocados Crops. Agronomy 2023, 13, 668. [Google Scholar] [CrossRef]
- Boyero, J.R.; Calderon, E.; Vela, J.M. Presencia de Oligonychus punicae en España. Agrícola Vergel 2014, 376, 245–248. [Google Scholar]
- Vela, J.M.; González-Fernández, J.; Wong, E.; Montserrat, M.; Farré, J.M.; Boyero, J.R. El ácaro del aguacate (Oligonychus perseae): Estado actual del problema e investigación en Andalucía [The avocado mite (Oligonychus perseae): Present situation and research in Andalucia]. Agrícola Vergel 2007, 26, 301–308. [Google Scholar]
- Torres, E. Manejo Integrado de Oligonychus perseae Tuttle, Baker y Abbatiello (Acari: Tetranychidae), en el Cultivo del Aguacate. Master’s thesis, Trabajo Fin de Carrera, Ingeniero Agrónomo, Escuela Técnica Superior de Ingeniería Técnica Agrícola; Universidad de La Laguna: La Laguna, Spain, 2007. [Google Scholar]
- Bragard, A.; Baptista, C.; Chatzivassiliou, P.E.; Di Serio, F.; Gonthier, P.; Jaques, M.; Josep, A.; Justesen Annemarie, F.; Magnusson, C.S.; Milona, P.; et al. Pest Categorisation of Oligonychus perseae. EFSA J. 2022, 20, e07336. [Google Scholar] [CrossRef]
- Barzman, M.; Bàrberi, P.; Birch, A.N.E.; Boonekamp, P.; Dachbrodt-Saaydeh, S.; Graf, B.; Hommel, B.; Jensen, J.E.; Kiss, J.; Kudsk, P.; et al. Eight principles of integrated pest management. Agron. Sustain. Dev. 2015, 35, 1199–1215. [Google Scholar] [CrossRef]
- Norris, R.F.; Caswell-Chen, E.P.; Kogan, M. Concepts in Integrated Pest Management; Prentice Hall: Upper Saddle River, NJ, USA, 2003; ISBN 0130870161. [Google Scholar]
- Nutter, F.W.; Teng, P.S.; Royer, M.H. Terms and Concepts for Yield, Crop Loss, and Disease Thresholds. Plant Dis. 1993, 77, 211–215. [Google Scholar]
- McCown, R. Changing systems for supporting farmers’ decisions: Problems, paradigms, and prospects. Agric. Syst. 2002, 74, 179–220. [Google Scholar] [CrossRef]
- Goble, T. Integrated Pest Management: Concepts, Tactics, Strategies and Case Studies; Radclife, E., Hutchison, W., Cancelado, R., Eds.; Cambridge University Press: Cambridge, UK, 2009. [Google Scholar]
- Rossi, V.; Sperandio, G.; Caffi, T.; Simonetto, A.; Gilioli, G. Critical success factors for the adoption of decisión tolos in IPM. Agronomy 2019, 9, 710. [Google Scholar] [CrossRef]
- Rossi, V.; Ca, T.; Salinari, F. Helping farmers face the increasing complexity of decision-making for crop protection. Phytopathol. Mediterr. 2012, 51, 457–479. [Google Scholar]
- Torres, E.; Hernández-Suárez, E.; Alvarez-Acosta, C.; Ferragut, F. Oligonychus perseae Tuttle, Baker & Abbatiello (Acari: Tetranychidae) population dynamics and associated phytoseiid mites (Acari: Phytoseiidae) in avocado orchards in the Canary Islands (Spain). Int. J. Acarol. 2022, 48, 551–563. [Google Scholar] [CrossRef]
- Maoz, Y.; Gal, S.; Zilberstein, M.; Izhar, Y.; Alchanatis, V.; Coll, M.; Palevsky, E. Determining an economic injury level for the persea mite, Oligonychus perseae, a new pest of avocado in Israel. Entomol. Exp. Appl. 2010, 138, 110–116. [Google Scholar] [CrossRef]
- Takano-Lee, M.; Hoddle, M.S. Oligonychus perseae (Acari: Tetranychidae) population responses to cultural control attempts in an avocado orchard. Fla. Entomol. 2002, 85, 216–226. [Google Scholar] [CrossRef]
- Kerguelen, V.; Hoddle, M.S. Comparison of the susceptibility of several cultivars of avocado to the persea mite, Oligonychus perseae (Acari: Tetranychidae). Sci. Hortic. 2000, 84, 101–114. [Google Scholar] [CrossRef]
- Kerguelen, V.; Hoddle, M.S. Biological control of Oligonychus perseae (Acari: Tetranychidae) on avocado: II. Evaluating the efficacy of Galendromus helveolus and Neoseiulus californicus (Acari: Phytoseiidae). Int. J. Acarol. 1999, 25, 221–229. [Google Scholar] [CrossRef]
- Hoddle, M.S.; Robinson, L.; Virzi, J. Biological control of Oligonychus perseae (Acari: Tetranychidae) on avocado. III. Evaluating the efficacy of varying release rates and release frequency of Neoseiulus californicus (Acari: Phytoseiidae). Int. J. Acarol. 2000, 26, 203–214. [Google Scholar] [CrossRef]
- Takano, M.; Hoddle, M.S. Biological control of Oligonychus perseae (Acari: Tetranychidae) on avocado: IV Evaluating the efficacy of a modified mistblower to mechanically dispense Neoseiulus californicus (Acari: Phytoseiidae). Int. J. Acarol. 2001, 27, 157–169. [Google Scholar] [CrossRef]
- Maoz, Y.; Gal, S.; Argov, Y.; Coll, M.; Palevsky, E. Biocontrol of perseae mite, Oligonychus perseae, with an exotic spider mite predator and an indigenous pollen feeder. Biol. Control 2011, 59, 147–157. [Google Scholar] [CrossRef]
- Machlitt, D. Persea mite on avocados—A quick field counting method. Subtrop. Fruit Notes 1998, 6, 1–4. [Google Scholar]
- Strong, W.B.; Croft, B.A. Inoculative release of phytoseiid mites (Acarina: Phytoseidae) into rapidly expanding canopy of hops for control of Tetranychus urticae (Acarina: Tetranychidae). Environ. Entolmol. 1995, 24, 446–453. [Google Scholar] [CrossRef]
- Greco, N.M.; Lijesthröm, G.G.; Sánchez, N.E. Spatial distribution and coincidence of Neoseilus californicus and tetranychus urticae (Acari: Phytoseiidae) on strawberry. Exp. Appl. Acarol. 1999, 23, 567–579. [Google Scholar] [CrossRef]
- Gómez Moya, C.A.; Ferragut, F. Distribución en la planta y eficacia de Neoseiulus californicus y Phytoseiulus persimilis (Acari: Phytoseiidae) en el control de las arañas rojas de cultivos hortícolas en condiciones de semicampo. Boletín Sanid. Veg. Plagas 2009, 35, 377–390. [Google Scholar]
- Maoz, Y.; Gal, S.; Abrahams, J.; Gan-Mor, S.; Coll, M.; Palevsky, E. Pollen provisioning enhances Euseius scutalis (Phytoseiidae) populations and improves control of Oligonychus perseae (Tetranychidae). In Proceedings of the Third International Symposium on Biological Control of Arthropods, Christchurch, New Zealand, 8–13 February 2008; pp. 339–346. [Google Scholar]
- González- Fernández, J.J.; Vela, J.M.; de la Peña, F.; Wong, M.E.; Farré, J.M.; Boyero, J.R.; Montserrat, M. Dinámica poblacional de Oligonychus perseae (Tuttle, Baker & Abbatiello) y la fauna auxiliar asociada en cultivo de aguacate Hass en el sur de España. In Proceedings of the VI World Avocado Congress, Viña Del Mar, Chile, 12–16 November 2007. [Google Scholar]
- González-Fernández, J.J.; De La Peña, F.; Hormaza, J.I.; Boyero, J.R.; Vela, J.M.; Wong, E.; Trigo, M.M.; Montserrat, M. Alternative food improves the combined effect of an omnivore and a predator on biological pest control. A case study in avocado orchards. Bull. Entomol. Res. 2009, 99, 433–444. [Google Scholar] [CrossRef] [PubMed]
- MAPA—Ministerio de Agricultura, Pesca y Alimentación. Resolución Autorización Excepcional Para Comercialización y Uso de Productos Fitosanitarios Formulados a Base de Spirodiclofen 24% [SC] Como Insecticida Contra Oligonychus perseae en Aguacate. 2018. Available online: https://www.juntadeandalucia.es/export/drupaljda/MOD_RES%20_AUT_EXCEPCIONAL_SPIRODICLOFEN_ARA%C3%91A_AGUACATE.pdf (accessed on 25 January 2023).
- Humeres, E.C.; Morse, J.G. Baseline susceptibility of Persea mite (Acari: Tetranychidae) to abamectin and milbemectin in avocado groves in Southern California. Exp. Appl. Acarol. 2005, 36, 51–59. [Google Scholar] [CrossRef] [PubMed]
- Hoddle, M.S.; Morse, J. The Persea mite invasion into California: History, biology, management, and current status. Calif. Avocado Soc. Yearb. 2012, 95, 106–136. [Google Scholar]
- Verónica, A.; Diaz, L.; Francisco, J.; Dávila, R. Modelización espacial de Oligonychus perseae (Tuttle, Baker & Abbatiello) mediante técnicas geoestadísticas. Rev. Mex. Cienc. Agric. 2019, 10, 1405–1416. [Google Scholar]
- Manjunatha Reddy, G.V.; Srinivasa, S.; Muralidhara, M.S. Potentiality of Cinnamomum extracts to two spotted spider mite, Tetranychus urticae Koch and its predator Neoseiulus longispinosus (Evans). J. Biopestic. 2014, 7, 11–14. [Google Scholar]
- Holden, D.; Ross, R.E. A commercial extract of the brown seaweed Ascophyllum nodosum suppresses avocado Thrips and Persea mites in field-grown “Hass” avocados, a practical field perspective. Acta Hortic. 2012, 1009, 137–142. [Google Scholar] [CrossRef]

| Field Trial | Coordinates | Phytoseiid Species Released | Maximum Percentage of Reduction of PM | Maximum Number of Phytoseiids/Leaf Observed | Phytoseiid Species Identified (%) |
|---|---|---|---|---|---|
| Latitude (N) Longitude (W) | |||||
| N° 1 (2007) | 28°23′13.58″ (N) 16°32′41.53″ (W) | N. californicus (Koppert Biological Systems) | 0.00 * | 4.1 | Phytoseiids were not identified |
| 36.61 ** | 7.27 | ||||
| N° 2 (2007) | 28°24′31.09″ (N) 16°30′43.22″ (W) | N. californicus (Koppert B.S.) | 10.87 * | 2.63 | E. stipulatus (89.2) |
| 29.37 ** | 2.08 | E. stipulatus (54.5) | |||
| A. swirskii (Koppert B.S.) | 0.00 * | 0.83 | I. degenerans (40) | ||
| 15.53 ** | 0.55 | A. swirskii (46.4) | |||
| A. swirskii (Syngenta Bioline) | 49 ** | 1.25 | E. stipulatus (55.6) | ||
| N° 3 (2008) | 28°23′27.10″ (N) 16°36′1.19″ (W) | N. californicus | 84 ** | 0.88 | E. stipulatus (100) |
| 44.30 W ** | 0.93 | E. stipulatus (90.5) | |||
| A. swirskii | 53.37 ** | 0.53 | E. stipulatus (87.5) | ||
| 68.53 W ** | 0.65 | E. stipulatus (93.5) | |||
| I. degenerans | 66.32 *** | 0.65 | E. stipulatus (50) N. californicus (50) | ||
| 39.10 W *** | 0.93 | E. stipulatus (80) | |||
| N° 4 (2009) | 28°24′20.47″ (N) 16°31′20.47″ (W) | N. californicus | 86.11 ** | 4.35 | E. stipulatus (52.5) N. californicus (47.5) |
| 63.59 ** | 2.98 | E. stipulatus (57.5) N. californicus (42.5) | |||
| N° 5 (2010) | 28°18′36.50″ (N) 16°25′22.15″ (W) | N. californicus | 51 ** | 5.08 | Euseius spp. (53.3) E. scutalis (46.7) N. californicus (45) E. stipulatus (5.6) |
| Phytoseiids | Avocado Trees | Resident Ground Cover |
|---|---|---|
| Euseius scutalis (Athias-Henriot) | Present | Present |
| Euseius stipulatus (Athias-Henriot) | Present | Present |
| Neoseiulus californicus (McGregor) | Present | Present |
| Typhlodromus phialatus Athias-Henriot | Present | Present |
| Iphiseius degenerans (Berlese) | Present | Present |
| Neoseiulus barkeri Hughes | Present | Present |
| Neoseiulella canariensis Ferragut and Peña-Estévez | Present | Present |
| Typhlodromus rhenanoides Athias-Henriot | Present | Present |
| Typhlodromina tropica (Chant) | Present | Absent |
| Typhlodromus pyri (Scheuten) | Present | Absent |
| Field Trial | Coordinates Plot | Treatment | Dose | Maximum Efficacy (%) | Maximum Reduction of Phytoseiids (%) | |
|---|---|---|---|---|---|---|
| Latitude (N) Longitude (W) | Active Ingredient | Trade Name Manufacturer (Register N°) | ||||
| N°1 1 (2007) | 28°24′25.55″ (N) 16°31′3.37″ (W) | Summer oil 83% EC | Oil Oro® 1 Químicas Oro (12,797) | 1% | 62 | 24 |
| Ricinus oil and Sulphur 30% SC | Melamyel and Gescen® 2 Myelosgreen | 0.3% 0.20% | 3 | 41 | ||
| Sulphur 80% SC | Sufrevit® 3 Sipcam (16,419) | 0.50% | 84 | 56 | ||
| N°2 2 (2008) | 28°23′27.10″ (N) 16°36′1.19″ (W) | Azadirachtin 2.6% EC | Align® 3 Sipcam (23,291) | 0.15% | 65 | 50 |
| Potassium soap 50% SC | Kabon® 3 Atlántica A. (4840) | 0.5% | 64 | 55 | ||
| Abamectin 1.8% EC | Vertimec® 1 Syngenta (16,784) | 0.05% | 4.2 | 10 | ||
| Sulphur 80% SC | Sufrevit® 3 Sipcam (16,419) | 0.50% | 53 | 70 | ||
| N°3 3 (2010) | 28°18′36.50″ (N) 16° 25′22.15″ (W) | Abamectin 1.8% EC | Vertimec® 1 Syngenta (16,784) | 0.05% | 99 | 96 |
| Abamectin 1.8% EC | Apache® 1 Afrasa (21,854) | 0.05% | 98 | 87 | ||
| Sulphur 80% SC | Sufrevit® 3 Sipcam (16,419) | 0.50% | 99 | 82 | ||
| Spirodiclofen 24% SC | Envidor® 1 Bayer (23,972) | 0.02% | 98 | 89 | ||
| N°4 4 (2015) | 28°24′23.19″ (N) 16°30′58.35″ (W) | Ascophyllum nodosum algae extract 100% (PS) | Stimul® 3 Sipcam | 0.06% | 65.08 | 29.4 |
| Zinc soluble 25% (w/w) and plant adjuvants at 13% | Cinamite® 2 Biocolor | 0.20% | 93.10 | 67.8 | ||
| Cinnamomum zeylanicum and other plant extracts. 90% organic matter and Citrus oil 40% (w/w) | Cinatec® and Olitec® 3 Agrotecnologia Group | 0.30% 0.20% | 68.5 | 52.1 | ||
| Spirodiclofen 24% SC | Envidor® 1 Bayer (23,972) | 0.02% | 98.28 | 60 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Torres, E.; Álvarez-Acosta, C.; Ferragut, F.; Hernández-Suárez, E.M. Oligonychus perseae (Tetranychidae) Invasion in the Canary Islands: History, Management and Current Situation. Agronomy 2024, 14, 920. https://doi.org/10.3390/agronomy14050920
Torres E, Álvarez-Acosta C, Ferragut F, Hernández-Suárez EM. Oligonychus perseae (Tetranychidae) Invasion in the Canary Islands: History, Management and Current Situation. Agronomy. 2024; 14(5):920. https://doi.org/10.3390/agronomy14050920
Chicago/Turabian StyleTorres, Eduardo, Carlos Álvarez-Acosta, Francisco Ferragut, and Estrella Marina Hernández-Suárez. 2024. "Oligonychus perseae (Tetranychidae) Invasion in the Canary Islands: History, Management and Current Situation" Agronomy 14, no. 5: 920. https://doi.org/10.3390/agronomy14050920
APA StyleTorres, E., Álvarez-Acosta, C., Ferragut, F., & Hernández-Suárez, E. M. (2024). Oligonychus perseae (Tetranychidae) Invasion in the Canary Islands: History, Management and Current Situation. Agronomy, 14(5), 920. https://doi.org/10.3390/agronomy14050920







