Hyperspectral Non-Imaging Measurements and Perceptron Neural Network for Pre-Harvesting Assessment of Damage Degree Caused by Septoria/Stagonospora Blotch Diseases of Wheat
Abstract
1. Introduction
2. Materials and Methods
2.1. Wheat Cultivation and Pathogen Inoculation
2.2. Hyperspectral Non-Imaging Measurements
2.3. Hyperspectral Data Analysis and Neural Network
3. Results
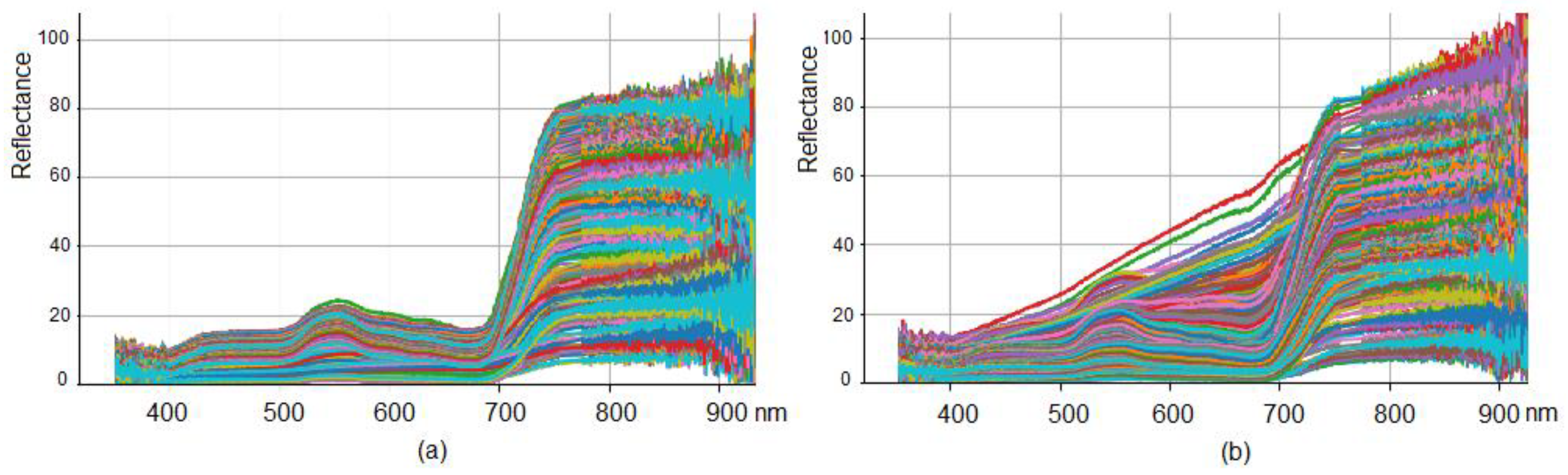
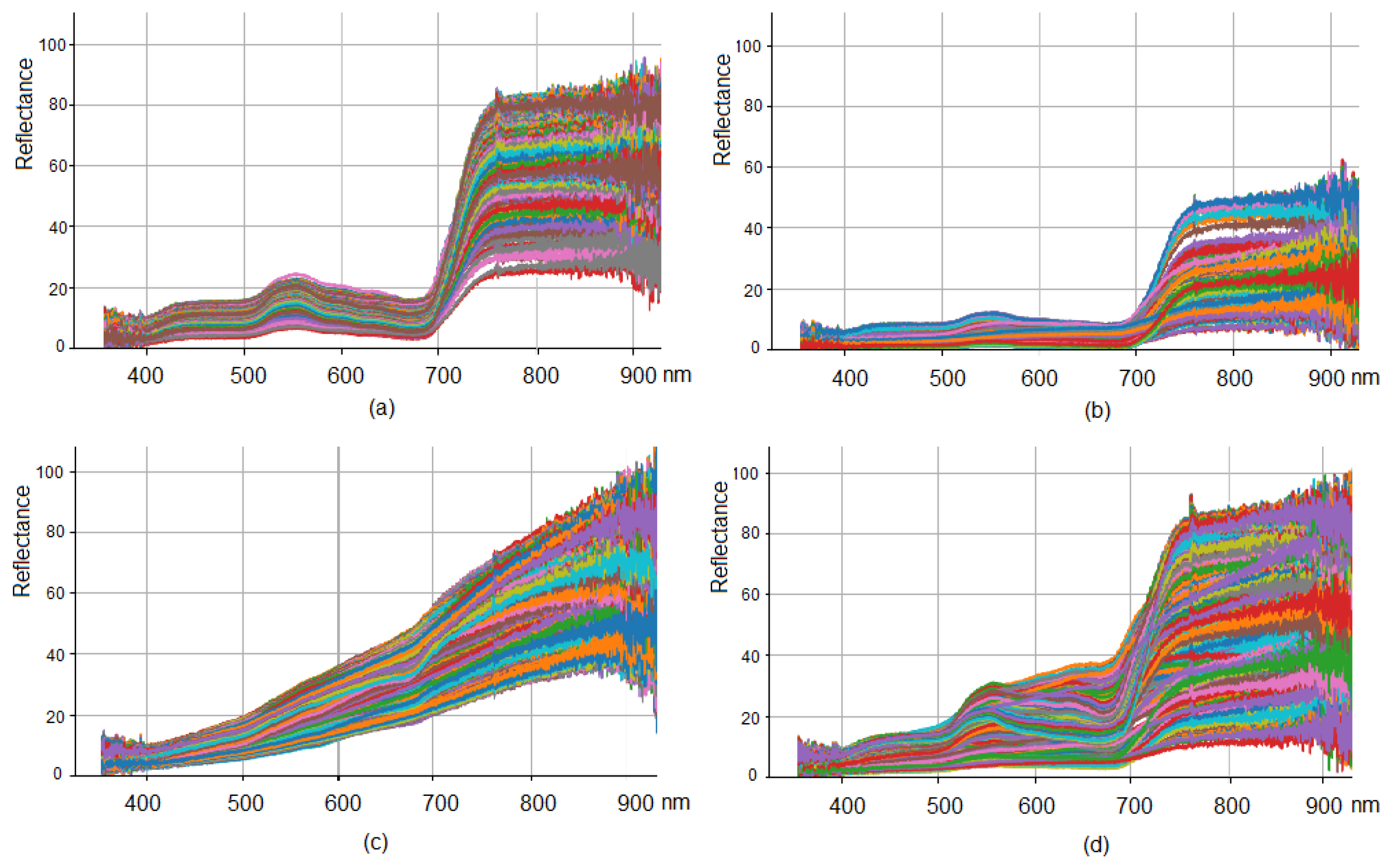
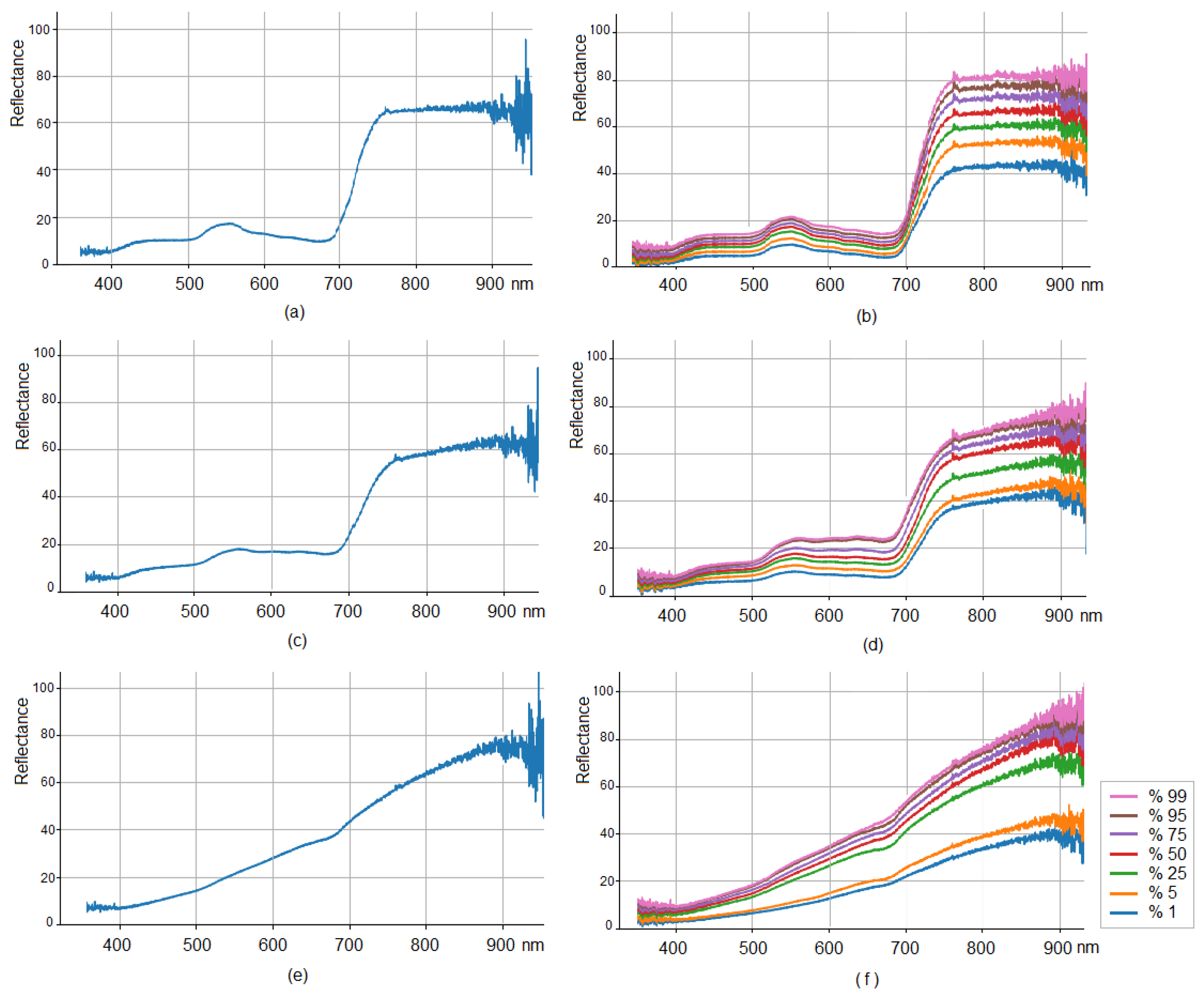
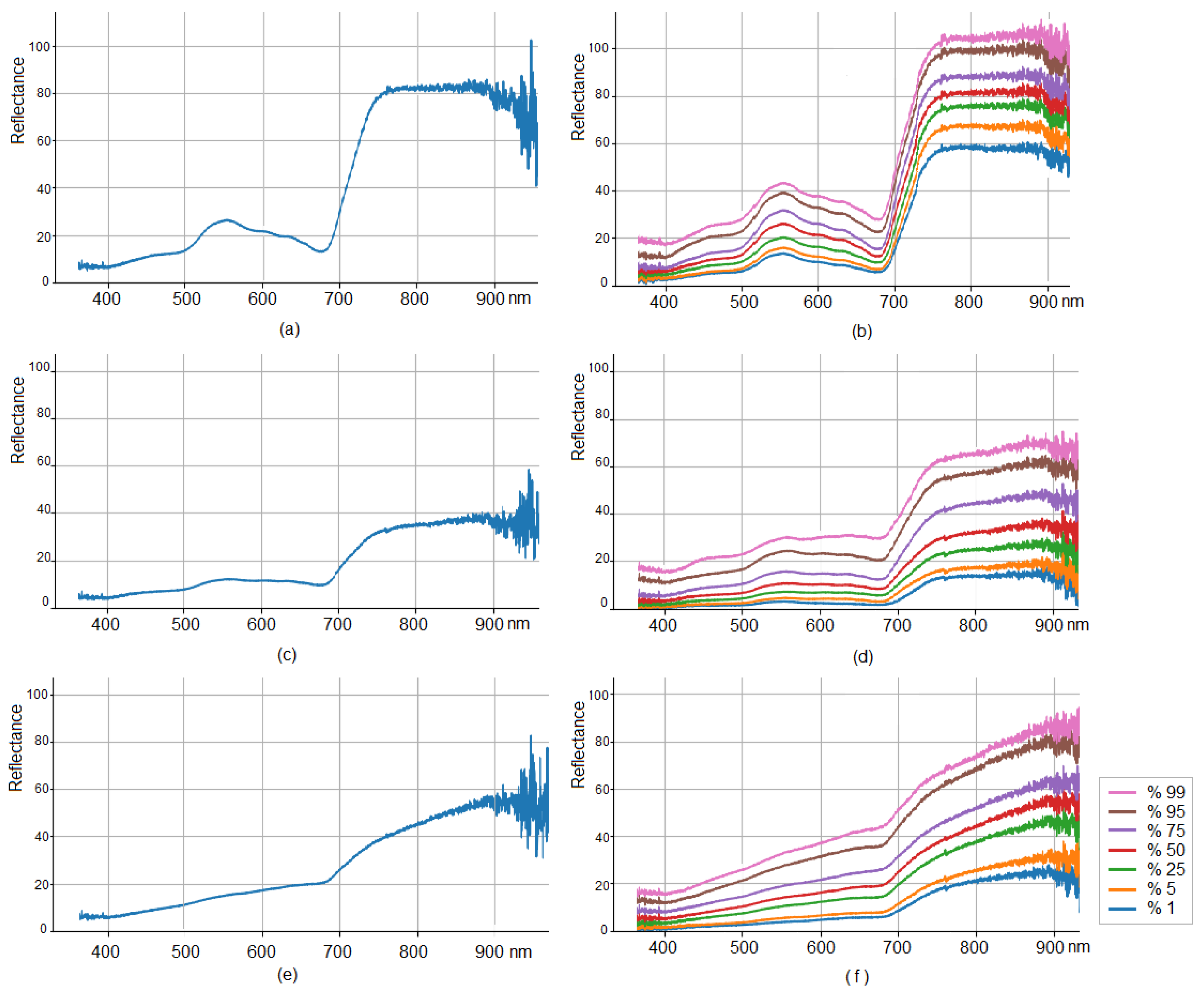
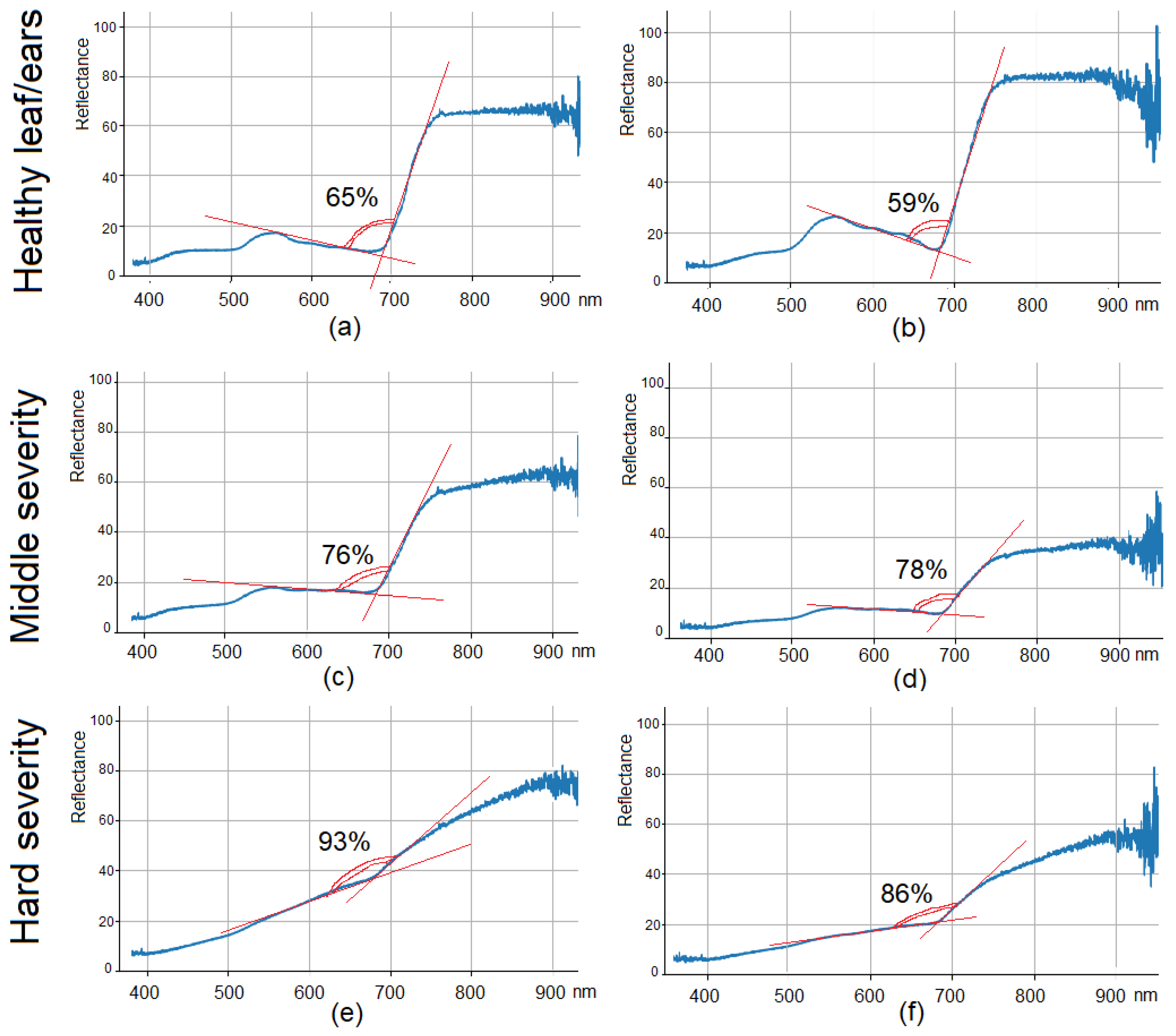
3.1. Hyperspectral Data Visualization and Distribution Quantile Analysis
3.2. Classification of Diseased Plants by Non-Imaging Hyperspectral Signatures with Neural Network
3.3. Estimation of Yield Losses Due to Septoriosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Suffert, F.; Sache, I.; Lannou, C. Early stages of septoria tritici blotch epidemics of winter wheat: Build-up, overseasoning, and release of primary inoculum. Plant Pathol. 2011, 60, 166–177. [Google Scholar] [CrossRef]
- Fones, H.; Gurr, S. The impact of Septoria tritici Blotch disease on wheat: An EU perspective. Fungal Genet. Biol. 2015, 79, 3–7. [Google Scholar] [CrossRef] [PubMed]
- Chedli, R.B.H.; M’barek, S.B.; Yahyaoui, A.; Kehel, Z.; Rezgui, S. Occurrence of Septoria tritici blotch (Zymoseptoria tritici) disease on durum wheat, triticale, and bread wheat in northern Tunisia. Chil. J. Agric. Res. 2018, 78, 559–568. [Google Scholar] [CrossRef]
- Turgay, E.B.; Büyük, O.; Yıldırım, A.F.; Ölmez, F.; Akci, N.; Karakaya, A. Pathotype diversity of Zymoseptoria tritici (Desm. Quaedvlieg & Crous) isolates collected from Central Anatolia, Turkey. J. Phytopathol. 2022, 170, 588–597. [Google Scholar] [CrossRef]
- Ponomarenko, A.; Goodwin, S.B.; Kema, G. Septoria tritici blotch (STB) of wheat. Plant Health Instr. 2011. [Google Scholar] [CrossRef]
- McDonald, M.C.; Renkin, M.; Spackman, M.; Orchard, B.; Croll, D.; Solomon, P.S.; Milgate, A. Rapid parallel evolution of azole fungicide resistance in Australian populations of the Wheat Pathogen Zymoseptoria tritici. Appl. Environ. Microbiol. 2019, 85, e01908-18. [Google Scholar] [CrossRef]
- O’Driscoll, A.; Doohan, F.; Mullins, E. Exploring the utility of Brachypodium distachyon as a model pathosystem for the wheat pathogen Zymoseptoria tritici Plant Biology. BMC Res. Notes 2015, 8, 132. [Google Scholar] [CrossRef]
- Sanin, S.S.; Korneva, L.G.; Polyakova, T.M. Prognosis of the risk of epiphytotic septoria of wheat leaves and ears. Plant Prot. Quar. 2015, 33–36. [Google Scholar]
- Kononenko, O.S.; Shishkin, N.V.; Derova, T.G. Winter wheat septoria blotch (Zymoseptoria tritici) (literature review). Grain Econ. Russ. 2021, 6, 104–108. [Google Scholar] [CrossRef]
- Sanin, S.S.; Zhohova, T.P. The influence of diseases and plant protection products on the quality of wheat grain. Защита и карантин растений 2012, 11, 16–20. (In Russian) [Google Scholar]
- Bock, C.H.; Barbedo, J.G.A.; Del Ponte, E.M.; Bohnenkamp, D.; Mahlein, A.-K. From visual estimates to fully automated sensor-based measurements of plant disease severity: Status and challenges for improving accuracy. Phytopathol. Res. 2020, 2, 9. [Google Scholar] [CrossRef]
- Hughes, D.P.; Salathe, M. An open access repository of images on plant health to enable the development of mobile disease diagnostics. arXiv 2015, arXiv:1511.08060. [Google Scholar]
- Esgario, J.G.M.; Krohling, R.A.; Ventura, J.A. Deep learning for classification and severity estimation of coffee leaf biotic stress. Comput. Electron. Agric. 2020, 169, 105162. [Google Scholar] [CrossRef]
- Mahlein, A.K.; Steiner, U.; Hillnhütter, C.; Dehne, H.W.; Oerke, E.C. Hyperspectral imaging for small-scale analysis of symptoms caused by different sugar beet diseases. Plant Methods 2012, 8, 3. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.; Kuska, M.T.; Bohnenkamp, D.; Brugger, A.; Alisaac, E.; Wahabzada, M.; Behmann, J.; Mahlein, A.K. Benefits of hyperspectral imaging for plant disease detection and plant protection: A technical perspective. J. Plant Dis. Prot. 2018, 125, 5–20. [Google Scholar] [CrossRef]
- Abdulridha, J.; Ehsani, R.; De Castro, A. Detection and differentiation between laurel wilt disease, phytophthora disease, and salinity damage using a hyperspectral sensing technique. Agriculture 2016, 6, 56. [Google Scholar] [CrossRef]
- Wang, D.; Vinson, R.; Holmes, M.; Seibel, G.; Bechar, A.; Nof, S.; Tao, Y. Early Detection of Tomato Spotted Wilt Virus by Hyperspectral Imaging and Outlier Removal Auxiliary Classifier Generative Adversarial Nets (OR-AC-GAN). Sci. Rep. 2019, 9, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Lowe, A.; Harrison, N.; French, A.P. Hyperspectral image analysis techniques for the detection and classification of the early onset of plant disease and stress. Plant Methods 2017, 13, 80. [Google Scholar] [CrossRef]
- Bebronne, R.; Michez, A.; Leemans, V.; Vermeulen, P.; Dumont, B.; Mercatoris, B. Characterisation of fungal diseases on winter wheat crop using proximal and remote multispectral imaging. In Proceedings of the Precision Agriculture’19; Wageningen Academic Publishers: Wageningen, The Netherlands, 2019; pp. 255–261. [Google Scholar]
- Amara, J.; Bouaziz, B.; Algergawy, A. A deep learning-based approach for banana leaf diseases classification. Lect. Notes Inform. (LNI), Proc. Ser. Ges. Fur Inform. 2017, 266, 79–88. [Google Scholar]
- Mohanty, S.P.; Hughes, D.P.; Salathé, M. Using deep learning for image-based plant disease detection. Front. Plant Sci. 2016, 7, 1419. [Google Scholar] [CrossRef]
- Wan, L.; Li, H.; Li, C.; Wang, A.; Yang, Y.; Wang, P. Hyperspectral Sensing of Plant Diseases: Principle and Methods. Agronomy 2022, 12, 1451. [Google Scholar] [CrossRef]
- Boulent, J.; Foucher, S.; Théau, J.; St-Charles, P.L. Convolutional Neural Networks for the Automatic Identification of Plant Diseases. Front. Plant Sci. 2019, 10, 941. [Google Scholar] [CrossRef]
- Bhupathi, P.; Sevugan, P. Application of Hyperspectral Remote Sensing Technology for Plant Disease Forecasting: An Applied Review. Ann. Rom. Soc. Cell Biol. 2021, 25, 4555–4566. [Google Scholar]
- Mahlein, A.K.; Kuska, M.T.; Thomas, S.; Bohnenkamp, D.; Alisaac, E.; Behmann, J.; Wahabzada, M.; Kersting, K. Plant disease detection by hyperspectral imaging: From the lab to the field. Adv. Anim. Biosci. 2017, 8, 238–243. [Google Scholar] [CrossRef]
- Whetton, R.L.; Waine, T.W.; Mouazen, A.M. Hyperspectral measurements of yellow rust and fusarium head blight in cereal crops: Part 2: On-line field measurement. Biosyst. Eng. 2018, 167, 144–158. [Google Scholar] [CrossRef]
- Odilbekov, F.; Armoniené, R.; Henriksson, T.; Chawade, A. Proximal phenotyping and machine learning methods to identify septoria tritici blotch disease symptoms in wheat. Front. Plant Sci. 2018, 9, 685. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Anderegg, J.; Mikaberidze, A.; Karisto, P.; Mascher, F.; McDonald, B.A.; Walter, A.; Hund, A. Hyperspectral canopy sensing of wheat septoria tritici blotch disease. Front. Plant Sci. 2018, 9, 1195. [Google Scholar] [CrossRef] [PubMed]
- Anderegg, J.; Hund, A.; Karisto, P.; Mikaberidze, A. In-Field Detection and Quantification of Septoria Tritici Blotch in Diverse Wheat Germplasm Using Spectral–Temporal Features. Front. Plant Sci. 2019, 10, 1355. [Google Scholar] [CrossRef]
- Cheshkova, A.F. A review of hyperspectral image analysis techniques for plant disease detection and identification. Vavilovskii Zhurnal Genet. Sel. 2022, 26, 202–213. [Google Scholar] [CrossRef]
- Doktor, D.; Brosinsky, A.; Lausch, A.; Merbach, I. Comparison of imaging and non-imaging hyperspectral sensors observing vegetation in the laboratory. Geophys. Res. Abstr. 2011, 13, 9251. [Google Scholar]
- Bohnenkamp, D.; Behmann, J.; Paulus, S.; Steiner, U.; Mahlein, A.K. A Hyperspectral Library of Foliar Diseases of Wheat. Phytopathology 2021, 111, 1583–1593. [Google Scholar] [CrossRef] [PubMed]
- Paulus, S.; Mahlein, A.K. Technical workflows for hyperspectral plant image assessment and processing on the greenhouse and laboratory scale. Gigascience 2020, 9, giaa090. [Google Scholar] [CrossRef] [PubMed]
- Dammer, K.H. Proof of concept study: A novel mobile in-canopy imaging system for detecting symptoms of fungal diseases in cereals. J. Plant Dis. Prot. 2022, 129, 769–773. [Google Scholar] [CrossRef]
- Iori, A.; Scala, V.; Cesare, D.; Pinzari, F.; D’Egidio, M.G.; Fanelli, C.; Fabbri, A.A.; Reverberi, M.; Serranti, S. Hyperspectral and molecular analysis of Stagonospora nodorum blotch disease in durum wheat. Eur. J. Plant Pathol. 2015, 141, 689–702. [Google Scholar] [CrossRef]
- Singh, V.; Sharma, N.; Singh, S. A review of imaging techniques for plant disease detection. Artif. Intell. Agric. 2020, 4, 229–242. [Google Scholar] [CrossRef]
- Meier, U.; Bleiholder, H.; Buhr, L.; Feller, C.; Hack, H.; Heß, M.; Lancashire, P.; Schnock, U.; Stauß, R.; Van den Boom, T.; et al. The BBCH system to coding the phenological growth stages of plants-history and publications. J. Für Kult. 2009, 61, 41–52. [Google Scholar] [CrossRef]
- Ocean Optics, Ocean View Installation & Operation Manual United States America, 2016. Available online: https://www.oceaninsight.com/globalassets/catalog-blocks-and-images/manuals--instruction-ocean-optics/software/oceanviewio.pdf (accessed on 6 November 2022).
- Perceptron Neural Networks Description. Available online: https://www.mathworks.com/help/deeplearning/ug/perceptron-neural-networks.html (accessed on 24 February 2023).
- Bohnenkamp, D.; Behmann, J.; Mahlein, A.K. In-field detection of Yellow Rust in Wheat on the Ground Canopy and UAV Scale. Remote Sens. 2019, 11, 2495. [Google Scholar] [CrossRef]
- Golhani, K.; Balasundram, S.K.; Vadamalai, G.; Pradhan, B. Red-Edge indices to diagnose orange spotting disease of oil palm in Malaysia. In Proceedings of the 38th Asian Conference on Remote Sensing - Space Applications: Touching Human Lives, ACRS 2017, New Delhi, India, 23–27 October 2017. [Google Scholar]
- Kristoffersen, R.; Eriksen, L.B.; Nielsen, G.C.; Jørgensen, J.R.; Jørgensen, L.N. Management of Septoria Tritici Blotch Using Cultivar Mixtures. Plant Dis. 2022, 106, 1341–1349. [Google Scholar] [CrossRef]
- Castro, A.C.; Simón, M.R. Effect of tolerance to Septoria tritici blotch on grain yield, yield components and grain quality in Argentinean wheat cultivars. Crop Prot. 2016, 90, 66–76. [Google Scholar] [CrossRef]
- Ficke, A.; Cowger, C.; Bergstrom, G.; Brodal, G. Understanding yield loss and pathogen biology to improve disease management: Septoria nodorum blotch—A case study in wheat. Plant Dis. 2018, 102, 696–707. [Google Scholar] [CrossRef]
- Ghaffary, S.M.T.; Chawade, A.; Singh, P.K. Practical breeding strategies to improve resistance to Septoria tritici blotch of wheat. Euphytica 2018, 214, 122. [Google Scholar] [CrossRef]
- Cunfer, B.M. Stagonospora and Septoria diseases of barley, oat, and rye. Can. J. Plant Pathol. 2000, 22, 332–348. [Google Scholar] [CrossRef]
- Simko, I.; Jimenez-Berni, J.A.; Sirault, X.R.R. Phenomic approaches and tools for phytopathologists. Phytopathology 2017, 107, 6–17. [Google Scholar] [CrossRef]
- Mahlein, A.K.; Kuska, M.T.; Thomas, S.; Wahabzada, M.; Behmann, J.; Rascher, U.; Kersting, K. Quantitative and qualitative phenotyping of disease resistance of crops by hyperspectral sensors: Seamless interlocking of phytopathology, sensors, and machine learning is needed! Curr. Opin. Plant Biol. 2019, 50, 156–162. [Google Scholar] [CrossRef]
- Zahir, S.A.D.M.; Omar, A.F.; Jamlos, M.F.; Azmi, M.A.M.; Muncan, J. A review of visible and near-infrared (Vis-NIR) spectroscopy application in plant stress detection. Sens. Actuators A Phys. 2022, 338, 113468. [Google Scholar] [CrossRef]
- Ryckewaert, M.; Héran, D.; Simonneau, T.; Abdelghafour, F.; Boulord, R.; Saurin, N.; Moura, D.; Mas-Garcia, S.; Bendoula, R. Physiological variable predictions using VIS–NIR spectroscopy for water stress detection on grapevine: Interest in combining climate data using multiblock method. Comput. Electron. Agric. 2022, 197, 106973. [Google Scholar] [CrossRef]
- Kremneva, O.Y.; Danilov, R.Y.; Sereda, I.I.; Tutubalina, O.V.; Pachkin, A.A.; Zimin, M. V Spectral characteristics of winter wheat varieties depending on the development degree of Pyrenophora tritici-repentis. Precis. Agric. 2022. [Google Scholar] [CrossRef]
- Wegulo, S.N.; Breathnach, J.A.; Baenziger, P.S. Effect of growth stage on the relationship between tan spot and spot blotch severity and yield in winter wheat. Crop Prot. 2009, 28, 696–702. [Google Scholar] [CrossRef]
- Pierre, B.; Marie-Odile, B.; François, C.; David, G. Identifying traits leading to tolerance of wheat to Septoria tritici blotch. Field Crop Res. 2015, 180, 176–185. [Google Scholar] [CrossRef]
- Jevtić, R.; Župunski, V.; Lalošević, M.; Župunski, L. Predicting potential winter wheat yield losses caused by multiple disease systems and climatic conditions. Crop Prot. 2017, 99, 17–25. [Google Scholar] [CrossRef]
- Budka, A.; Łacka, A.; Gaj, R.; Jajor, E.; Korbas, M. Predicting winter wheat yields by comparing regression equations. Crop Prot. 2015, 78, 84–91. [Google Scholar] [CrossRef]



| Defined with Neural Network | Cohen’s Kappa | |||
|---|---|---|---|---|
| Healthy, % | Damaged, % | Uncertain, % | ||
| Nominal scale | ||||
| Leaves | ||||
| Healthy leaves | 88.1 ± 2.4 | 10.6 ± 2.5 | 1.1 ± 0.1 | 0.90 |
| Light lesion of Septoria | 84.0 ± 5.5 | 10.8 ± 6.1 | 5.0 ± 0.1 | 0.64 |
| Medium lesion of Septoria | 60.5 ± 5.6 | 38.3 ± 5.9 | 1.2 ± 0.3 | 0.13 |
| Severe lesion of Septoria | 7.1 ± 2.9 | 92.7 ± 3.1 | 0.1 ± 0.1 | 0.98 |
| Dead leaves | 2.3 ± 3.0 | 97.7 ± 3.2 | 0.1 ± 0.1 | 0.99 |
| Ears | ||||
| Healthy ears | 94.9 ± 4.8 | 4.9 ± 4.7 | 0.1 ± 0.1 | 0.96 |
| Light lesion of Septoria | 70.2 ± 6.5 | 29.2 ± 6.3 | 0.6 ± 0.2 | 0.40 |
| Medium lesion of Septoria | 33.3 ± 7.6 | 65.6 ± 7.9 | 1.2 ± 0.2 | 0.24 |
| Severe lesion of Septoria | 0.6 ± 1.0 | 99.4 ± 1.0 | 0.0 ± 0.0 | 0.99 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhelezova, S.V.; Pakholkova, E.V.; Veller, V.E.; Voronov, M.A.; Stepanova, E.V.; Zhelezova, A.D.; Sonyushkin, A.V.; Zhuk, T.S.; Glinushkin, A.P. Hyperspectral Non-Imaging Measurements and Perceptron Neural Network for Pre-Harvesting Assessment of Damage Degree Caused by Septoria/Stagonospora Blotch Diseases of Wheat. Agronomy 2023, 13, 1045. https://doi.org/10.3390/agronomy13041045
Zhelezova SV, Pakholkova EV, Veller VE, Voronov MA, Stepanova EV, Zhelezova AD, Sonyushkin AV, Zhuk TS, Glinushkin AP. Hyperspectral Non-Imaging Measurements and Perceptron Neural Network for Pre-Harvesting Assessment of Damage Degree Caused by Septoria/Stagonospora Blotch Diseases of Wheat. Agronomy. 2023; 13(4):1045. https://doi.org/10.3390/agronomy13041045
Chicago/Turabian StyleZhelezova, Sofia V., Elena V. Pakholkova, Vladislav E. Veller, Mikhail A. Voronov, Eugenia V. Stepanova, Alena D. Zhelezova, Anton V. Sonyushkin, Timur S. Zhuk, and Alexey P. Glinushkin. 2023. "Hyperspectral Non-Imaging Measurements and Perceptron Neural Network for Pre-Harvesting Assessment of Damage Degree Caused by Septoria/Stagonospora Blotch Diseases of Wheat" Agronomy 13, no. 4: 1045. https://doi.org/10.3390/agronomy13041045
APA StyleZhelezova, S. V., Pakholkova, E. V., Veller, V. E., Voronov, M. A., Stepanova, E. V., Zhelezova, A. D., Sonyushkin, A. V., Zhuk, T. S., & Glinushkin, A. P. (2023). Hyperspectral Non-Imaging Measurements and Perceptron Neural Network for Pre-Harvesting Assessment of Damage Degree Caused by Septoria/Stagonospora Blotch Diseases of Wheat. Agronomy, 13(4), 1045. https://doi.org/10.3390/agronomy13041045








