Bacillus velezensis: A Beneficial Biocontrol Agent or Facultative Phytopathogen for Sustainable Agriculture
Abstract
1. Introduction
2. Role of B. velezensis in Controlling Plant Pathogens
2.1. Antimicrobial Substances Synthesized by B. velezensis to Control Plant Pathogens
2.1.1. Phytopathogens Controlled by Difficidin and Bacilysin
2.1.2. Phytopathogens Controlled by Bacillomycin D
2.1.3. Phytopathogens Controlled by Fengycins
| B. velezensis Strains | Antimicrobial Compounds | Target Pathogens (Plant Diseases) | References |
|---|---|---|---|
| B. velezensis FZB42 | Bacillomycin D | F. graminearum (Fusarium head blight) | [37] |
| B. velezensis FZB42 | Difficidin and bacilysin | X. oryzae pv. oryzae (bacterial blight of rice); X. oryzae pv. oryzicola (bacterial leaf streak) | [28] |
| B. velezensis FZB42 | Bacilysin | P. sojae (soybean root rot disease) | [31] |
| B. velezensis Y6 and F7 | Surfactin, Iturin and Fengycin | R. solanacearum (tomato wilt); F. oxysporum (banana wilt) | [20] |
| B. velezensis SQR9 | Bacillomycin D | F. oxysporum (cucumber vascular wilt) | [33] |
| B. velezensis MEP218 | Fengycins | X. axonopodis pv. vesicatoria (bacterial spot disease) | [44] |
| B. velezensis G341 | Bacillomycin L and Fengycin A | Alternaria panax, F. oxysporum, B. cineria, Phytophthroa capsici | [45] |
| B. velezensis WRB-ZX-001 and WRB-ZX-002 | Iturin A | Aspergillus fumigatus | [46] |
| B. velezensis CC09 | Iturin A | G. graminis var. tritici (take-all disease) and B. sorokiniana (spot blotch of wheat) | [24] |
| B. velezensis FJAT-46737 | Fengycins | R. solanacearum | [47] |
| B. velezensis | Fengycins | F. solani (basal stem rot in passion fruit) | [41] |
| B. velezensis NJN-6 | Bacillomycin D | F. oxysporum | [48] |
| B. velezensis NJN-6 | Macrolactin | R. solanacearum | [48] |
2.1.4. Bacteriocins Produced by B. velezensis
3. Bioactive Enzymes Produced by B. velezensis
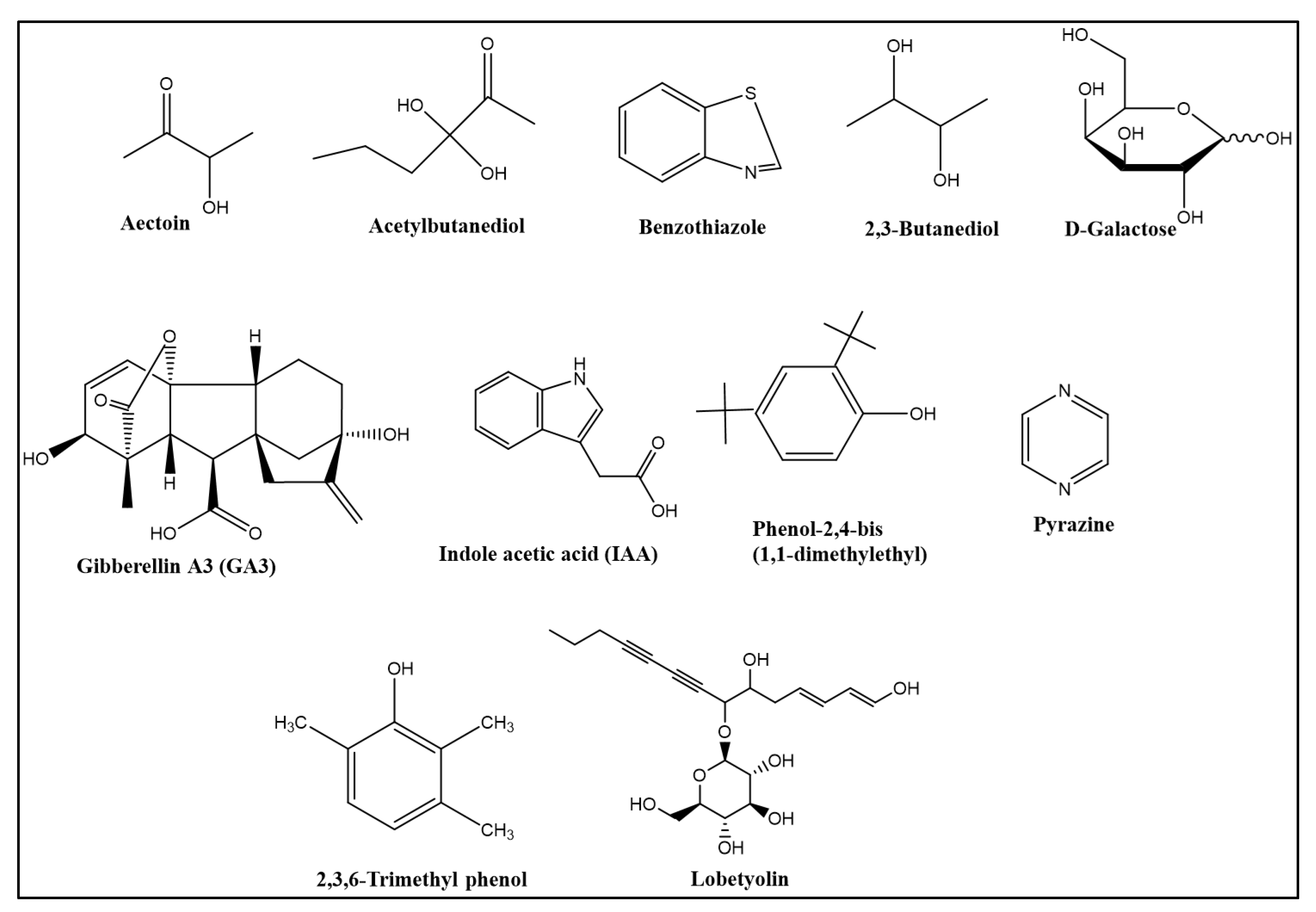
4. Production of Volatile Organic Compounds (VOCs) and Induction of ISR by B. velezensis
5. Plant Health and Growth Promotion by B. velezensis
6. Deleterious Nature of B. velezensis in Agriculture
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, L.; Wu, H.J.; Qiao, J.; Gao, X.; Borriss, R. Novel routes for improving biocontrol activity of Bacillus based bioinoculants. Front. Microbiol. 2015, 6, 1395. [Google Scholar] [CrossRef] [PubMed]
- Boyd, L.A.; Ridout, C.; O’Sullivan, D.M.; Leach, J.E.; Leung, H. Plant-pathogen interactions: Disease resistance in modern agriculture. Trends Genet. 2013, 29, 233–240. [Google Scholar] [CrossRef] [PubMed]
- Berini, F.; Katz, C.; Gruzdev, N.; Casartelli, M.; Tettamanti, G.; Marinelli, F. Microbial and viral chitinases: Attractive biopesticides for integrated pest management. Biotechnol. Adv. 2018, 36, 818–838. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Kumar, V.; Shahzad, B.; Tanveer, M.; Sidhu, G.P.S.; Handa, N.; Kohli, S.K.; Yadav, P.; Bali, A.S.; Parihar, R.D.; et al. Worldwide pesticide usage and its impacts on ecosystem. SN Appl. Sci. 2019, 1, 1446. [Google Scholar] [CrossRef]
- Marian, M.; Shimizu, M. Improving performance of microbial biocontrol agents against plant diseases. J. Gen. Plant Pathol. 2019, 85, 329–336. [Google Scholar] [CrossRef]
- Collinge, D.B.; Jensen, D.F.; Rabiey, M.; Sarrocco, S.; Shaw, M.W.; Shaw, R.H. Biological control of plant diseases—What has been achieved and what is the direction? Plant Pathol. 2022, 71, 1024–1047. [Google Scholar] [CrossRef]
- Deng, Y.; Chen, H.; Li, C.; Xu, J.; Qi, Q.; Xu, Y.; Zhu, Y.; Zheng, J.; Peng, D.; Ruan, L.; et al. Endophyte Bacillus subtilis evade plant defense by producing lantibiotic subtilomycin to mask self-produced flagellin. Commun. Biol. 2019, 2, 368. [Google Scholar] [CrossRef]
- Molina-Santiago, C.; Pearson, J.R.; Navarro, Y.; Berlanga-Clavero, M.V.; Caraballo-Rodriguez, A.M.; Petras, D.; García-Martín, M.L.; Lamon, G.; Haberstein, B.; Cazorla, F.M.; et al. The extracellular matrix protects Bacillus subtilis colonies from Pseudomonas invasion and modulates plant co-colonization. Nat. Commun. 2019, 10, 1919. [Google Scholar] [CrossRef]
- Liu-xu, L.; Vicedo, B.; García-agustín, P.; Llorens, E.; Liu-xu, L. Advances in endophytic fungi research: A data analysis of 25 years of achievements and challenges. J. Plant Interact. 2022, 17, 244–266. [Google Scholar] [CrossRef]
- Mousa, W.K.; Shearer, C.; Limay-Rios, V.; Ettinger, C.L.; Eisen, J.A.; Raizada, M.N. Root-hair endophyte stacking in finger millet creates a physicochemical barrier to trap the fungal pathogen Fusarium graminearum. Nat. Microbiol. 2016, 1, 16167. [Google Scholar] [CrossRef]
- White, J.F.; Kingsley, K.L.; Zhang, Q.; Verma, R.; Obi, N.; Dvinskikh, S.; Elmore, M.T.; Verma, S.K.; Gond, S.K.; Kowalski, K.P. Review: Endophytic microbes and their potential applications in crop management. Pest Manag. Sci. 2019, 75, 2558–2565. [Google Scholar] [CrossRef] [PubMed]
- Radhakrishnan, R.; Hashem, A.; Abd Allah, E.F. Bacillus: A biological tool for crop improvement through bio-molecular changes in adverse environments. Front. Physiol. 2017, 8, 667. [Google Scholar] [CrossRef] [PubMed]
- Morales-Cedeño, L.R.; Orozco-Mosqueda, M.d.C.; Loeza-Lara, P.D.; Parra-Cota, F.I.; De los Santos-Villalobos, S.; Santoyo, G. Plant growth-promoting bacterial endophytes as biocontrol agents of pre- and post-harvest diseases: Fundamentals, methods of application and future perspectives. Microbiol. Res. 2021, 242, 126612. [Google Scholar] [CrossRef] [PubMed]
- Clardy, J.; Fischbach, M.A.; Walsh, C.T. New antibiotics from bacterial natural products. Nat. Biotechnol. 2006, 24, 1541–1550. [Google Scholar] [CrossRef]
- Maheshwari, D.K. Bacteria in Agrobiology: Disease Management; Springer: Cham, Switzerland, 2012; pp. 1–495. [Google Scholar]
- Shafi, J.; Tian, H.; Ji, M. Bacillus species as versatile weapons for plant pathogens: A review. Biotechnol. Biotechnol. Equip. 2017, 31, 446–459. [Google Scholar] [CrossRef]
- Zaid, D.S.; Cai, S.; Hu, C.; Li, Z.; Li, Y.; Gralnick, J.A. Comparative genome analysis reveals phylogenetic identity of Bacillus velezensis HNA3 and genomic insights into its plant growth promotion and biocontrol effects. Microbiol. Spectr. 2022, 10, e02169-21. [Google Scholar] [CrossRef]
- Rabbee, M.F.; Ali, M.S.; Choi, J.; Hwang, B.S.; Jeong, S.C.; Baek, K. Bacillus velezensis: A valuable member of bioactive molecules within plant microbiomes. Molecules 2019, 24, 1046. [Google Scholar] [CrossRef]
- Pandin, C.; Le Coq, D.; Deschamps, J.; Védie, R.; Rousseau, T.; Aymerich, S.; Briandet, R. Complete genome sequence of Bacillus velezensis QST713: A biocontrol agent that protects Agaricus bisporus crops against the green mould disease. J. Biotechnol. 2018, 278, 10–19. [Google Scholar] [CrossRef]
- Cao, Y.; Pi, H.; Chandrangsu, P.; Li, Y.; Wang, Y.; Zhou, H.; Xiong, H.; Helmann, J.D.; Cai, Y. Antagonism of two plant-growth promoting Bacillus velezensis isolates against Ralstonia solanacearum and Fusarium oxysporum. Sci. Rep. 2018, 8, 4360. [Google Scholar] [CrossRef]
- Zhou, S.; Liu, G.; Zheng, R.; Sun, C.; Wu, S. Structural and functional insights into iturin W, a novel lipopeptide produced by the deep-sea bacterium Bacillus sp. strain wsm-1. Appl. Environ. Microbiol. 2020, 86, e01597-20. [Google Scholar] [CrossRef]
- Pandin, C.; Darsonval, M.; Mayeur, C.; Le Coq, D.; Aymerich, S.; Briandeta, R. Biofilm formation and synthesis of antimicrobial compounds by the biocontrol agent Bacillus velezensis QST713 in an Agaricus bisporus compost micromodel. Appl. Environ. Microbiol. 2019, 85, e00327-19. [Google Scholar] [CrossRef] [PubMed]
- Ge, B.; Liu, B.; Nwet, T.T.; Zhao, W.; Shi, L.; Zhang, K.; Thinn Nwet, T.; Zhao, W.; Shi, L.; Zhang, K.; et al. Bacillus methylotrophicus strain NKG-1, Isolated from changbai mountain, China, Has potential applications as a biofertilizer or biocontrol agent. PLoS ONE 2016, 11, e0166079. [Google Scholar] [CrossRef] [PubMed]
- Kang, X.; Zhang, W.; Cai, X.; Zhu, T.; Xue, Y.; Liu, C. Bacillus velezensis CC09: A potential ‘Vaccine’ for controlling wheat diseases. Mol. Plant-Microbe Interact. 2018, 31, 623–632. [Google Scholar] [CrossRef]
- Wang, D.; Xu, Z.; Zhang, G.; Xia, L.; Dong, X.; Li, Q.; Liles, M.R.; Shao, J.; Shen, Q.; Zhang, R. A genomic island in a plant beneficial rhizobacterium encodes novel antimicrobial fatty acids and a self-protection shield to enhance its competition. Environ. Microbiol. 2019, 21, 3455–3471. [Google Scholar] [CrossRef]
- Rabbee, M.F.; Ali, S.; Baek, K. Endophyte Bacillus velezensis isolated from citrus spp. controls streptomycin-resistant Xanthomonas citri subsp. citri that causes citrus bacterial canker. Agronomy 2019, 9, 470. [Google Scholar]
- Rabbee, M.F.; Islam, N.; Baek, K.H. Biocontrol of citrus bacterial canker caused by Xanthomonas citri subsp. citri by Bacillus velezensis. Saudi J. Biol. Sci. 2022, 29, 2363–2371. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Wu, H.; Chen, L.; Yu, X.; Borriss, R.; Gao, X. Difficidin and bacilysin from Bacillus amyloliquefaciens FZB42 have antibacterial activity against Xanthomonas oryzae rice pathogens. Sci. Rep. 2015, 5, 12975. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.A.; Göpel, Y.; Milewski, S.; Görke, B. Two small RNAs conserved in enterobacteriaceae provide intrinsic resistance to antibiotics targeting the cell wall biosynthesis enzyme glucosamine-6-phosphate synthase. Front. Microbiol. 2016, 7, 908. [Google Scholar] [CrossRef]
- Islam, T.; Rabbee, M.F.; Choi, J.; Baek, K. Biosynthesis, molecular regulation, and application of bacilysin produced by Bacillus species. Metabolites 2022, 12, 397. [Google Scholar] [CrossRef]
- Han, X.; Shen, D.; Xiong, Q.; Bao, B.; Zhang, W.; Dai, T.; Zhao, Y.; Borriss, R.; Fan, B. The plant-beneficial rhizobacterium Bacillus velezensis FZB42 controls the soybean pathogen phytophthora sojae due to bacilysin production. Appl. Environ. Microbiol. 2021, 87, e01601-21. [Google Scholar] [CrossRef]
- Chen, L.; Heng, J.; Qin, S.; Bian, K. A comprehensive understanding of the biocontrol potential of Bacillus velezensis LM2303 against Fusarium head blight. PLoS ONE 2018, 13, e0198560. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Shao, J.; Li, B.; Yan, X.; Shen, Q.; Zhang, R. Contribution of bacillomycin D in Bacillus amyloliquefaciens SQR9 to antifungal activity and biofilm formation. Appl. Environ. Microbiol. 2013, 79, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Mandic-Mulec, I.; Zhang, H.; Zhang, N.; Xu, Z.; Mandic-Mulec, I.; Zhang, H.; Liu, Y.; Sun, X.; Feng, H.; et al. Antibiotic bacillomycin D Affects iron acquisition and biofilm formation in Bacillus velezensis through a Btr-mediated FeuABC-dependent pathway. Cell Rep. 2019, 29, 1192–1202. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Yang, N.; Zheng, S.; Yan, F.; Jiang, C.; Yu, Y.; Guo, J.; Chai, Y.; Chen, Y. The spo0A-sinI-sinR regulatory circuit plays an essential role in biofilm formation, nematicidal activities, and plant protection in Bacillus cereus AR156. Mol. Plant-Microbe Interact. 2017, 30, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Arnaouteli, S.; Bamford, N.C.; Stanley-Wall, N.R.; Kovács, Á.T. Bacillus subtilis biofilm formation and social interactions. Nat. Rev. Microbiol. 2021, 19, 600–614. [Google Scholar] [CrossRef] [PubMed]
- Gu, Q.; Yang, Y.; Yuan, Q.; Shi, G.; Wu, L.; Lou, Z.; Huo, R.; Wu, H.; Borriss, R.; Gao, X. Bacillomycin D produced by Bacillus amyloliquefaciens is involved in the antagonistic interaction with the plant-pathogenic fungus Fusarium graminearum. Appl. Environ. Microbiol. 2017, 83, e01075-17. [Google Scholar] [CrossRef]
- Wang, S.; Herrera-balandrano, D.D.; Wang, Y.; Shi, X.; Chen, X.; Jin, Y.; Liu, F.; Laborda, P. Biocontrol Ability of the Bacillus amyloliquefaciens group, B. amyloliquefaciens, B. velezensis, B. nakamurai, and B. siamensis, for the management of fungal postharvest diseases: A review. J. Agric. Food Chem. 2022, 70, 6591–6616. [Google Scholar] [CrossRef]
- Xiong, Q.; Liu, D.; Zhang, H.; Dong, X.; Zhang, G.; Liu, Y.; Zhang, R. Quorum sensing signal autoinducer-2 promotes root colonization of Bacillus velezensis SQR9 by affecting biofilm formation and motility. Appl. Microbiol. Biotechnol. 2020, 104, 7177–7185. [Google Scholar] [CrossRef]
- Rabbee, M.F.; Baek, K.H. Antimicrobial Activities of lipopeptides and polyketides of Bacillus velezensis for agricultural applications. Molecules 2020, 25, 4973. [Google Scholar] [CrossRef]
- Ye, X.; Wang, C.; Ng, T.B.; Zhang, W. Study on the biocontrol potential of antifungal peptides produced by Bacillus velezensis against Fusarium solani that infects the passion fruit Passiflora edulis. J. Agric. Food Chem. 2021, 69, 2051–2061. [Google Scholar]
- Deleu, M.; Paquot, M.; Nylander, T. Fengycin interaction with lipid monolayers at the air-aqueous interface—Implications for the effect of fengycin on biological membranes. J. Colloid Interface Sci. 2005, 283, 358–365. [Google Scholar] [CrossRef]
- Moreno-Velandia, C.A.; Ongena, M.; Cotes, A.M. Effects of fengycins and iturins on Fusarium oxysporum f. sp. physali and root colonization by Bacillus velezensis Bs006 protect golden berry against vascular wilt. Phytopathology 2021, 111, 2227–2237. [Google Scholar]
- Rautenbach, M.; Martins, J.C.; Quintieri, L.; Jofré, E.; Medeot, D.B.; Fernandez, M.; Morales, G.M.; Jofré, E. Fengycins from Bacillus amyloliquefaciens MEP218 exhibit antibacterial activity by producing alterations on the cell surface of the pathogens Xanthomonas axonopodis pv. vesicatoria and Pseudomonas aeruginosa PA01. Front. Microbiol. 2020, 10, 3107. [Google Scholar]
- Lim, S.M.; Yoon, M.Y.; Choi, G.J.; Choi, Y.H.; Jang, K.S.; Shin, T.S.; Park, H.W.; Yu, N.H.; Kim, Y.H.; Kim, J.C. Diffusible and volatile antifungal compounds produced by an antagonistic Bacillus velezensis G341 against various phytopathogenic fungi. Plant Pathol. J. 2017, 33, 488–498. [Google Scholar] [CrossRef]
- Xiong, Z.R.; Cobo, M.; Whittal, R.M.; Snyder, A.B.; Worobo, R.W. Purification and characterization of antifungal lipopeptide produced by Bacillus velezensis isolated from raw honey. PLoS ONE 2022, 17, e0266470. [Google Scholar] [CrossRef]
- Chen, M.; Wang, J.; Liu, B.; Zhu, Y.; Xiao, R.; Yang, W.; Ge, C.; Chen, Z. Biocontrol of tomato bacterial wilt by the new strain Bacillus velezensis FJAT-46737 and its lipopeptides. BMC Microbiol. 2020, 20, 160. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Li, B.; Zhang, N.; Waseem, R.; Shen, Q.; Huang, Q. Production of bacillomycin- and macrolactin-type antibiotics by Bacillus amyloliquefaciens NJN-6 for suppressing soilborne plant pathogens. J. Agric. Food Chem. 2012, 60, 2976–2981. [Google Scholar] [CrossRef] [PubMed]
- Venugopal, H.; Edwards, P.J.B.; Schwalbe, M.; Claridge, J.K.; Libich, D.S.; Stepper, J.; Loo, T.; Patchett, M.L.; Norris, G.E.; Pascal, S.M. Structural, dynamic, and chemical characterization of a novel S-glycosylated bacteriocin. Biochemistry 2011, 50, 2748–2755. [Google Scholar] [CrossRef]
- Collins, F.W.J.; O’Connor, P.M.; O’Sullivan, O.; Gómez-Sala, B.; Rea, M.C.; Hill, C.; Ross, R.P. Bacteriocin Gene-Trait matching across the complete Lactobacillus Pan-genome. Sci. Rep. 2017, 7, 3481. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, Z.; Bai, X.; Zhang, D. A novel of new class II bacteriocin from Bacillus velezensis HN-Q-8 and its antibacterial activity on Streptomyces scabies. Front. Microbiol. 2022, 13, 943232. [Google Scholar] [CrossRef]
- Scholz, R.; Molohon, K.J.; Nachtigall, J.; Vater, J.; Markley, A.L.; Süssmuth, R.D.; Mitchell, D.A.; Borriss, R. Plantazolicin, a novel microcin B17/streptolysin S-like natural product from Bacillus amyloliquefaciens FZB42. J. Bacteriol. 2011, 193, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Scholz, R.; Vater, J.; Budiharjo, A.; Wang, Z.; He, Y.; Dietel, K.; Schwecke, T.; Herfort, S.; Lasch, P.; Borriss, R. Amylocyclicin, a novel circular bacteriocin produced by Bacillus amyloliquefaciens FZB42. J. Bacteriol. 2014, 196, 1842–1852. [Google Scholar] [CrossRef] [PubMed]
- Danilova, I.; Sharipova, M. The practical potential of bacilli and their enzymes for industrial production. Front. Microbiol. 2020, 11, 1782. [Google Scholar] [CrossRef] [PubMed]
- Langner, T.; Göhre, V. Fungal chitinases: Function, regulation, and potential roles in plant/pathogen interactions. Curr. Genet. 2016, 62, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Krolicka, M.; Hinz, S.W.A.; Koetsier, M.J.; Joosten, R.; Eggink, G.; Van Den Broek, L.A.M.; Boeriu, C.G. Chitinase Chi1 from Myceliophthora thermophila C1, a thermostable enzyme for chitin and chitosan depolymerization. J. Agric. Food Chem. 2018, 66, 1658–1669. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.M.; Huynh, T.U.; Nguyen, T.H.; Do, T.O.; Pentekhina, I.; Nguyen, Q.V.; Nguyen, A.D. Expression, purification, and basic properties of a novel domain structure possessing chitinase from Escherichia coli carrying the family 18 chitinase gene of Bacillus velezensis strain RB.IBE29. Mol. Biol. Rep. 2022, 49, 4141–4148. [Google Scholar] [CrossRef]
- Weisskopf, L.; Schulz, S.; Garbeva, P. Microbial volatile organic compounds in intra-kingdom and inter-kingdom interactions. Nat. Rev. Microbiol. 2021, 19, 391–404. [Google Scholar] [CrossRef]
- Yuan, J.; Raza, W.; Shen, Q.; Huang, Q. Antifungal activity of Bacillus amyloliquefaciens NJN-6 volatile compounds against Fusarium oxysporum f. sp. cubense. Appl. Environ. Microbiol. 2012, 78, 5942–5944. [Google Scholar] [CrossRef]
- Massawe, V.C.; Hanif, A.; Farzand, A.; Mburu, D.K.; Ochola, S.O.; Wu, L.; Tahir, H.A.S.; Gu, Q.; Wu, H.; Gao, X. Volatile compounds of endophytic Bacillus spp. have biocontrol activity against Sclerotinia sclerotiorum. Phytopathology 2018, 108, 1373–1385. [Google Scholar] [CrossRef]
- Gao, Z.; Zhang, B.; Liu, H.; Han, J.; Zhang, Y. Identification of endophytic Bacillus velezensis ZSY-1 strain and antifungal activity of its volatile compounds against Alternaria solani and Botrytis cinerea. Biol. Control 2017, 105, 27–39. [Google Scholar] [CrossRef]
- Harun-Or-Rashid, M.; Kim, H.J.; Yeom, S.I.; Yu, H.A.; Manir, M.M.; Moon, S.S.; Kang, Y.J.; Chung, Y.R. Bacillus velezensis YC7010 enhances plant defenses against brown planthopper through transcriptomic and metabolic changes in rice. Front. Plant Sci. 2018, 9, 1904. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.; Zhao, X.; Li, Y.; Wang, R.; Huang, Y.; Qi, G. Engineering Bacillus velezensis with high production of acetoin primes strong induced systemic resistance in Arabidopsis thaliana. Microbiol. Res. 2019, 227, 126297. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.H.; Liao, M.J.; Wang, H.K.; Zheng, M.Z.; Xu, J.J.; Guo, J.H. Bacillus velezensis, a potential and efficient biocontrol agent in control of pepper gray mold caused by Botrytis cinerea. Biol. Control 2018, 126, 147–157. [Google Scholar] [CrossRef]
- Lahlali, R.; Peng, G.; Gossen, B.D.; McGregor, L.; Yu, F.Q.; Hynes, R.K.; Hwang, S.F.; McDonald, M.R.; Boyetchko, S.M. Evidence that the biofungicide serenade (Bacillus subtilis) suppresses clubroot on canola via antibiosis and induced host resistance. Phytopathology 2013, 103, 245–254. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Shi, B.; Wang, L.; Huang, T.; Zhou, Z.; Hou, H.; Tu, H. Isolation and characterization of Bacillus velezensis strain p2-1 for biocontrol of apple postharvest decay caused by Botryosphaeria dothidea. Front. Microbiol. 2022, 12, 808938. [Google Scholar] [CrossRef]
- Lee, S.A.; Kim, S.Y.; Sang, M.K.; Song, J.; Weon, H.Y. Complete genome sequence of Bacillus velezensis T20E-257, a plant growth-promoting bacterium, isolated from tomato (Solanum lycopersicum L.) root. Korean J. Microbiol. 2017, 53, 342–343. [Google Scholar]
- Wang, A.; Hua, J.; Wang, Y.; Zhang, G.; Luo, S. Stereoisomers of nonvolatile acetylbutanediol metabolites produced by Bacillus velezensis WRN031 improved root elongation of maize and rice. J. Agric. Food Chem. 2020, 68, 6308–6315. [Google Scholar] [CrossRef]
- Liu, Y.; Feng, H.; Fu, R.; Zhang, N.; Du, W.; Shen, Q.; Zhang, R. Induced root-secreted d-galactose functions as a chemoattractant and enhances the biofilm formation of Bacillus velezensis SQR9 in an McpA-dependent manner. Appl. Microbiol. Biotechnol. 2020, 104, 785–797. [Google Scholar] [CrossRef]
- Sun, X.; Xu, Z.; Xie, J.; Hesselberg-Thomsen, V.; Tan, T.; Zheng, D.; Strube, M.L.; Dragoš, A.; Shen, Q.; Zhang, R.; et al. Bacillus velezensis stimulates resident rhizosphere Pseudomonas stutzeri for plant health through metabolic interactions. ISME J. 2022, 16, 774–787. [Google Scholar] [CrossRef]
- Xu, Z.; Xie, J.; Zhang, H.; Wang, D.; Shen, Q.; Zhang, R. Enhanced control of plant wilt disease by a xylose-inducible DegQ gene engineered into Bacillus velezensis strain SQR9XYQ. Phytopathology 2019, 109, 36–43. [Google Scholar] [CrossRef]
- Balderas-Ruíz, K.A.; Gómez-Guerrero, C.I.; Trujillo-Roldán, M.A.; Valdez-Cruz, N.A.; Aranda-Ocampo, S.; Juárez, A.M.; Leyva, E.; Galindo, E.; Serrano-Carreón, L. Bacillus velezensis 83 increases productivity and quality of tomato (Solanum lycopersicum L.): Pre and postharvest assessment. Curr. Res. Microb. Sci. 2021, 2, 100076. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Q.; Wu, Y.N.; Fan, Q.; Han, Q.Q.; Paré, P.W.; Xu, R.; Wang, Y.Q.; Wang, S.M.; Zhang, J.L. Improved growth and metabolite accumulation in Codonopsis pilosula (Franch.) Nannf. by inoculation of Bacillus amyloliquefaciens GB03. J. Agric. Food Chem. 2016, 64, 8103–8108. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Ruan, Y.; Wang, B.; Zhang, J.; Waseem, R.; Huang, Q.; Shen, Q. Plant growth-promoting rhizobacteria strain Bacillus amyloliquefaciens NJN-6-enriched bio-organic fertilizer suppressed fusarium wilt and promoted the growth of banana plants. J. Agric. Food Chem. 2013, 61, 3774–3780. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Liu, Y.; Xie, J.; Štefanic, P.; Lv, Y.; Fan, B.; Mandic-Mulec, I.; Zhang, R.; Shen, Q.; Xu, Z. Annulment of bacterial antagonism improves plant beneficial activity of a Bacillus velezensis consortium. Appl. Environ. Microbiol. 2022, 88, e00240-22. [Google Scholar] [CrossRef] [PubMed]
- Torres, M.; Llamas, I.; Torres, B.; Toral, L.; Sampedro, I.; Béjar, V. Growth promotion on horticultural crops and antifungal activity of Bacillus velezensis XT1. Appl. Soil Ecol. 2020, 150, 103453. [Google Scholar] [CrossRef]
- Kim, Y.S.; Lee, Y.; Cheon, W.; Park, J.; Kwon, H.T.; Balaraju, K.; Kim, J.; Yoon, Y.J.; Jeon, Y. Characterization of Bacillus velezensis AK-0 as a biocontrol agent against apple bitter rot caused by Colletotrichum gloeosporioides. Sci. Rep. 2021, 11, 626. [Google Scholar] [CrossRef]
- Bulgari, D.; Filisetti, S.; Montagna, M.; Gobbi, E.; Faoro, F. Pathogenic potential of bacteria isolated from commercial biostimulants. Arch. Microbiol. 2022, 204, 162. [Google Scholar] [CrossRef]
- Hwang, S.K.; Back, C.G.; Win, N.K.K.; Kim, M.K.; Kim, H.D.; Kang, I.K.; Lee, S.C.; Jung, H.Y. Occurrence of bacterial rot of onion caused by Bacillus amyloliquefaciens in Korea. J. Gen. Plant Pathol. 2012, 78, 227–232. [Google Scholar] [CrossRef]
- Wang, L.; Li, X.B.; Suo, H.C.; An, K.; Luo, H.M.; Liu, X.J. Soft rot of potatoes caused by Bacillus amyloliquefaciens in Guangdong province, China. Can. J. Plant Pathol. 2017, 39, 533–539. [Google Scholar] [CrossRef]
- Zeng, Y.; Yu, Q.; Cheng, S. Effects of Bacillus velezensis ZK1 on the physiology and metabolism of peaches. Int. J. Food Sci. Technol. 2022, 57, 2203–2215. [Google Scholar] [CrossRef]
- Jin, S.; Wang, N.; Zhou, Z.; Ji, Z.; Zhang, J. Identification of Bacillus species causing bacterial leaf spot of peach in China. J. Phytopathol. 2022, 170, 811–819. [Google Scholar] [CrossRef]
- Song, J.H.; Zhang, L.X.; Tan, G.J.; Wang, S.; Wang, J.J. First report of Bacillus pumilus causing fruit rot on muskmelon (Cucumis melo) in China. Plant Dis. 2017, 102, 439. [Google Scholar] [CrossRef]
- Su, Y.; Liu, C.; Fang, H.; Zhang, D. Bacillus subtilis: A universal cell factory for industry, agriculture, biomaterials and medicine. Microb. Cell Fact. 2020, 19, 173. [Google Scholar] [CrossRef]
- Gu, H.J.; Sun, Q.L.; Luo, J.C.; Zhang, J.; Sun, L. A first study of the virulence potential of a Bacillus subtilis isolate from deep-sea hydrothermal vent. Front. Cell. Infect. Microbiol. 2019, 9, 183. [Google Scholar] [CrossRef] [PubMed]
- Zhu, T.F.; Chen, F.F.; Li, J.C. A strain of pathogenic Bacillus subtilis results in brain damage in ducklings when co-infected with Riemerella anatipestifer. Pol. J. Vet. Sci. 2020, 20, 803–809. [Google Scholar]
- Enosi Tuipulotu, D.; Mathur, A.; Ngo, C.; Man, S.M. Bacillus cereus: Epidemiology, virulence factors, and host–pathogen interactions. Trends Microbiol. 2021, 29, 458–471. [Google Scholar] [CrossRef] [PubMed]
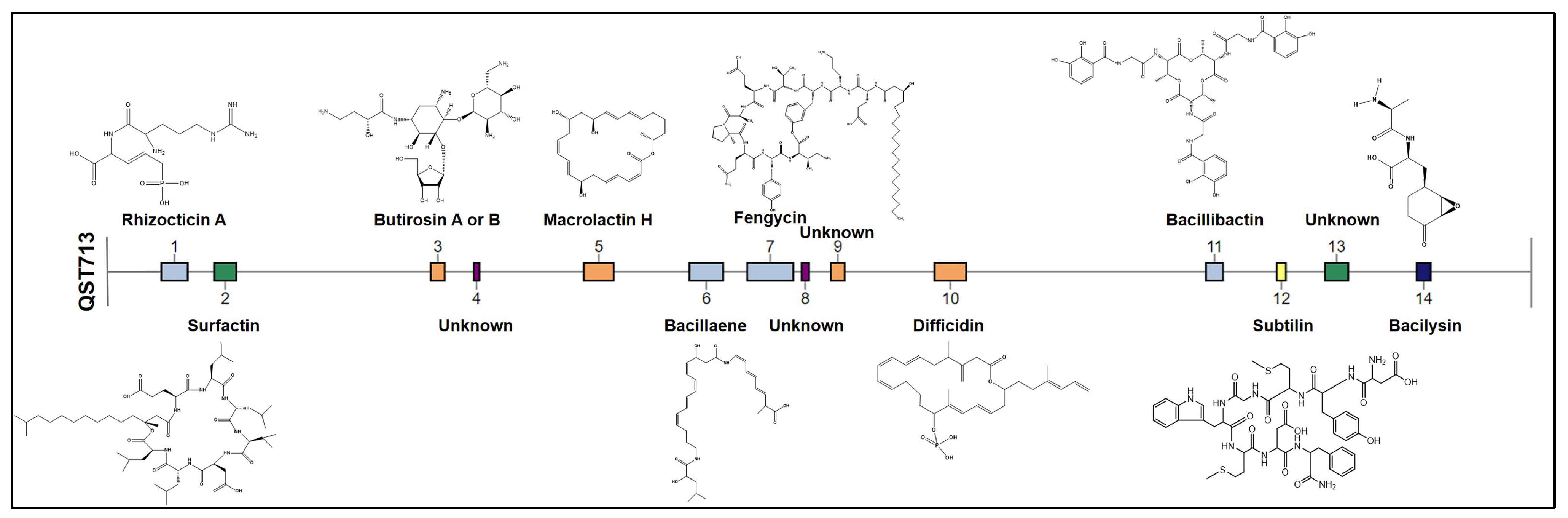

| Gene Clusters of B. velezensis QST713 (NCBI Accession Number: CP025079.1) | Bioactive Compounds Presence (+)/Absence (−) | |||||
|---|---|---|---|---|---|---|
| Region | Bioactive Compounds | Type | Size (nt) | Similarity | GB03 (CP049904.1) | 83 (CP034203.1) |
| 1 | Rhizocticin A | NRPS/PKS | 78,608 | 22% | - | - |
| 2 | Surfactin | NRPS | 66,095 | 86% | + | + |
| 3 | Butirosin A/Butirosin B | PKS | 42,244 | 7% | + | + |
| 4 | Unknown | Terpene | 18,104 | - | - | - |
| 5 | Macrolactin H | PKS | 88,814 | 100% | + | + |
| 6 | Bacillaene | PKS/NRPS | 100,882 | 100% | + | + |
| 7 | Fengycin | NRPS | 137,406 | 100% | + | + |
| 8 | Unknown | Terpene | 22,883 | - | - | - |
| 9 | Unknown | PKS | 42,100 | - | - | - |
| 10 | Difficidin | PKS | 94,798 | 100% | + | + |
| 11 | Bacillibactin | NRPS | 51,501 | 100% | + | + |
| 12 | Subtilin | Lanthipeptide class-I | 27,785 | 100% | Lanthipeptide class II: Mersacidin | Lanthipeptide class III: Locillomycin |
| 13 | Unknown | NRPS | 69,429 | - | - | - |
| 14 | Bacilysin | NRPS/PKS | 42,418 | 100% | + | + |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rabbee, M.F.; Hwang, B.-S.; Baek, K.-H. Bacillus velezensis: A Beneficial Biocontrol Agent or Facultative Phytopathogen for Sustainable Agriculture. Agronomy 2023, 13, 840. https://doi.org/10.3390/agronomy13030840
Rabbee MF, Hwang B-S, Baek K-H. Bacillus velezensis: A Beneficial Biocontrol Agent or Facultative Phytopathogen for Sustainable Agriculture. Agronomy. 2023; 13(3):840. https://doi.org/10.3390/agronomy13030840
Chicago/Turabian StyleRabbee, Muhammad Fazle, Buyng-Su Hwang, and Kwang-Hyun Baek. 2023. "Bacillus velezensis: A Beneficial Biocontrol Agent or Facultative Phytopathogen for Sustainable Agriculture" Agronomy 13, no. 3: 840. https://doi.org/10.3390/agronomy13030840
APA StyleRabbee, M. F., Hwang, B.-S., & Baek, K.-H. (2023). Bacillus velezensis: A Beneficial Biocontrol Agent or Facultative Phytopathogen for Sustainable Agriculture. Agronomy, 13(3), 840. https://doi.org/10.3390/agronomy13030840








