Intercropping Winter Oilseed Rape (Brassica napus L.) Has the Potential to Lessen the Impact of the Insect Pest Complex
Abstract
1. Introduction
2. Materials and Methods
2.1. Field Site and Set-Up
2.2. Impact of the Companion Plants on WOR
2.3. Impact of Intercropping on the Insect Pest Complex
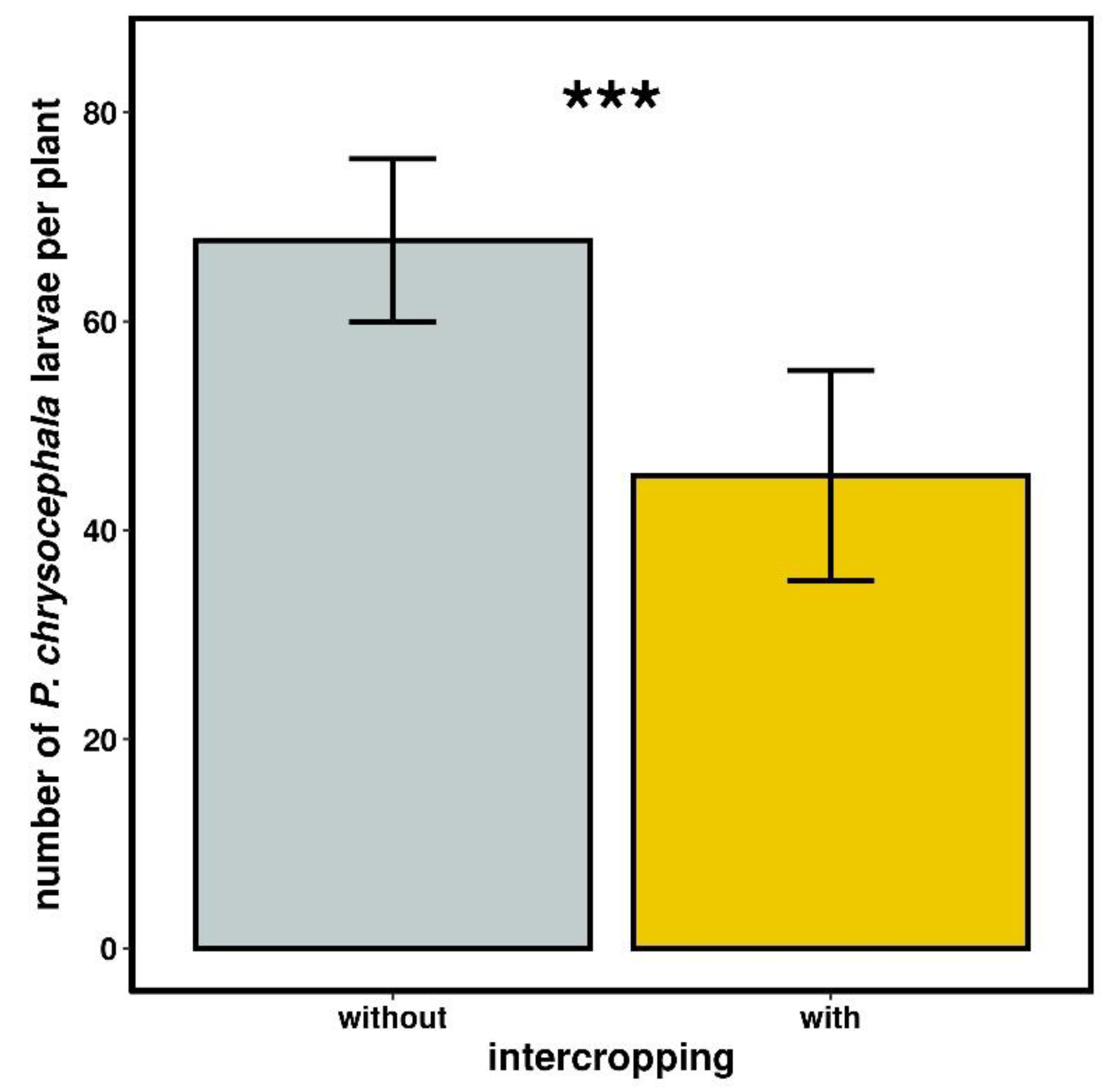
2.3.1. Psylliodes chrysocephala Larval Density
2.3.2. Ceutorhynchus napi Oviposition Punctures and Damage
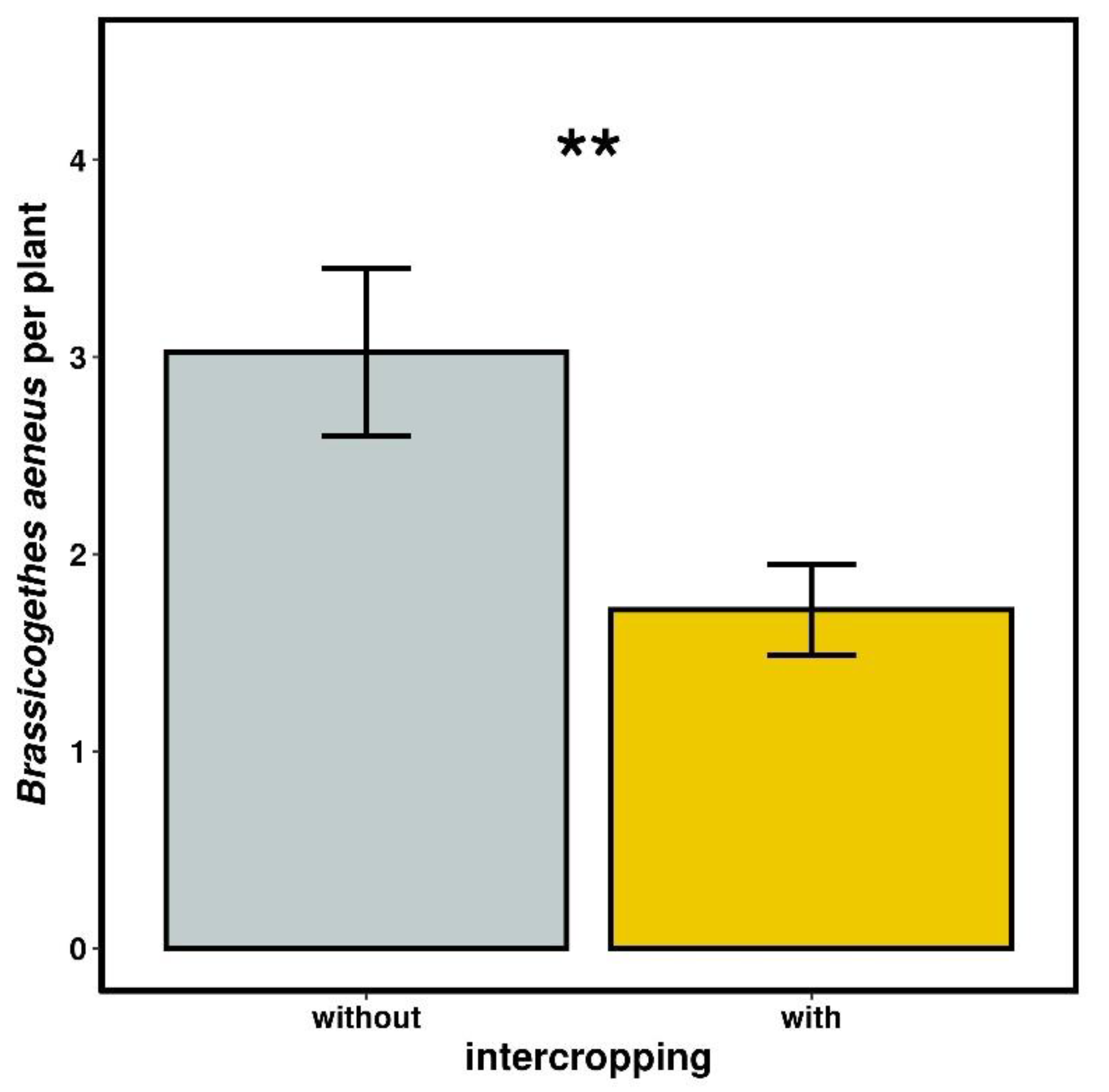
2.3.3. Number of Brassicogethes aeneus
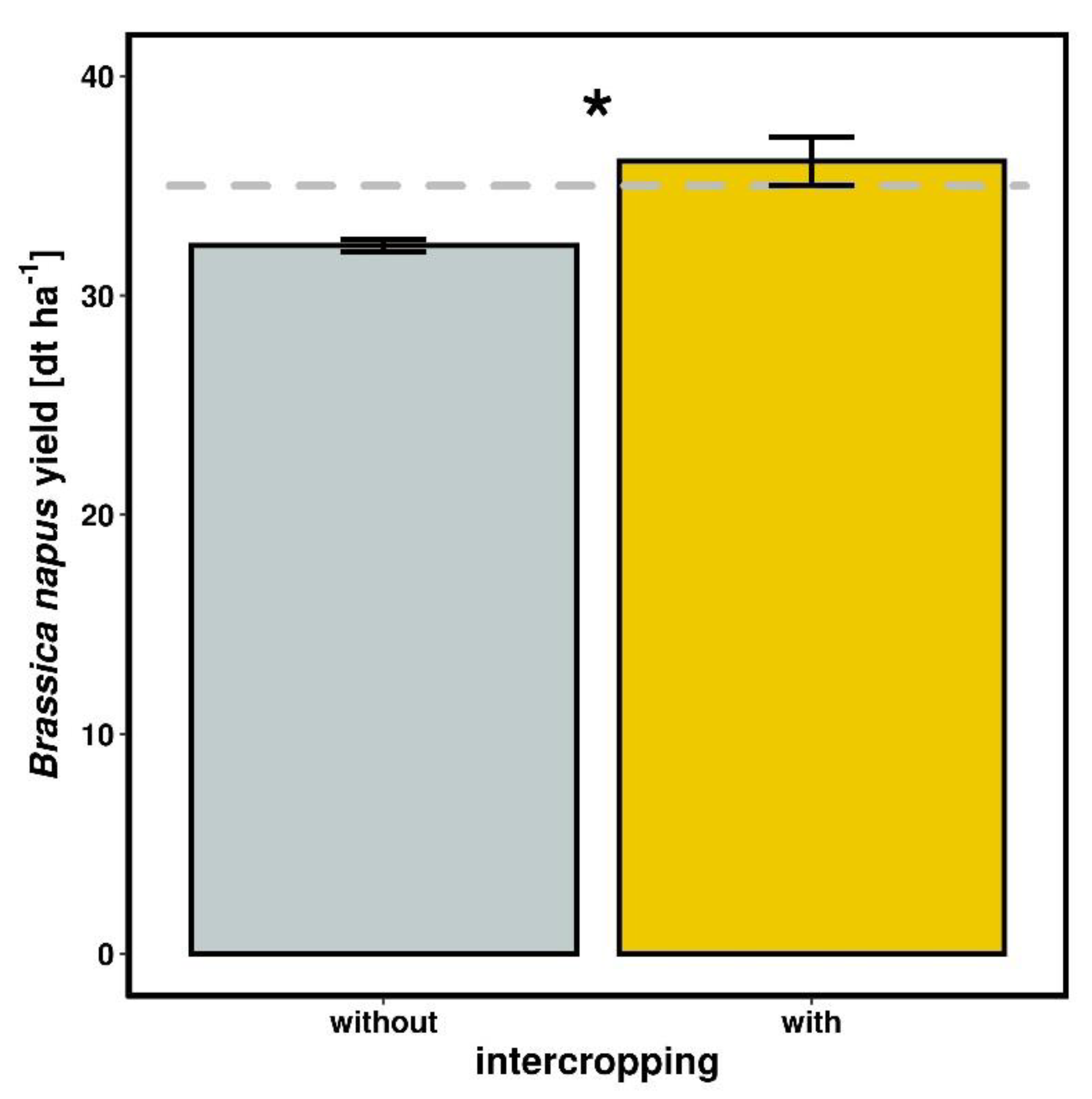
2.4. Yield
2.5. Statistical Analyses
3. Results
3.1. Impact of the Companion Plants on WOR
3.2. Impact of the Companion Plants on the Insect Pest Complex
3.2.1. Psylliodes chrysocephala Larval Density
3.2.2. Ceutorhynchus napi Oviposition Punctures and Damage
3.2.3. Number of Brassicogethes aeneus
3.3. Yield
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. FAOSTAT. Available online: http://www.fao.org/faostat (accessed on 26 August 2020).
- Flénet, F.; Wagner, D.; Simonin, P. Examination of an attempt to improve rapeseed cultivation in France in order to reduce the greenhouse gas emissions of biodiesel. OCL 2020, 27, 69. [Google Scholar] [CrossRef]
- Bouchet, A.-S.; Laperche, A.; Bissuel-Belaygue, C.; Snowdon, R.; Nesi, N.; Stahl, A. Nitrogen use efficiency in rapeseed. A review. Agron. Sustain. Dev. 2016, 36, 38. [Google Scholar] [CrossRef]
- Theunissen, J. Intercropping in field vegetable crops: Pest management by agrosystem diversification—An overview. Pest Manag. Sci. 1994, 42, 65–68. [Google Scholar] [CrossRef]
- Verret, V.; Gardarin, A.; Makowski, D.; Lorin, M.; Cadoux, S.; Butier, A.; Valantin-Morison, M. Assessment of the benefits of frost-sensitive companion plants in winter rapeseed. Eur. J. Agron. 2017, 91, 93–103. [Google Scholar] [CrossRef]
- Lorin, M.; Jeuffroy, M.H.; Butier, A.; Valantin-Morison, M. Undersowing winter oilseed rape with frost-sensitive legume living mulch: Consequences for cash crop nitrogen nutrition. Field Crop. Res. 2016, 193, 24–33. [Google Scholar] [CrossRef]
- Lorin, M.; Jeuffroy, M.H.; Butier, A.; Valantin-Morison, M. Undersowing winter oilseed rape with frost-sensitive legume living mulches to improve weed control. Eur. J. Agron. 2015, 71, 96–105. [Google Scholar] [CrossRef]
- Cadoux, S.; Sauzet, G.; Valantin-Morison, M.; Pontet, C.; Champolivier, L.; Robert, C.; Lieven, J.; Flénet, F.; Mangenot, O.; Fauvin, P.; et al. Intercropping frost-sensitive legume crops with winter oilseed rape reduces weed competition, insect damage, and improves nitrogen use efficiency. Oilseed Facts Crop. Lipids 2015, 22, D302. [Google Scholar] [CrossRef]
- Balachowsky, A.S. Entomologie Appliquée à L’agriculture, Tome 1; Masson et Cie Editeurs: Paris, France, 1963; Volume 2. [Google Scholar]
- Alford, D.; Nilsson, C.; Ulber, B. Insect pests of oilseed rape crops. In Biocontrol of Oilseed Rape Pests; Alford, D.V., Ed.; Blackwell: Oxford, UK, 2003; pp. 9–41. [Google Scholar]
- Williams, I.H. The major insect pests of oilseed rape in Europe and their management—An overview. In Biocontrol-Based Integrated Management of Oilseed Rape Pests; Williams, I.H., Ed.; Springer Dordrecht: Heidelberg, Germany; London UK; New York, NY, USA, 2010; pp. 1–44. [Google Scholar]
- Bonnemaison, L. Insect pests of crucifers and their control. Annu. Rev. Entomol. 1965, 10, 233–256. [Google Scholar] [CrossRef]
- Dewar, A.M. The adverse impact of the neonicotinoid seed treatment ban on crop protection in oilseed rape in the United Kingdom. Pest Manag. Sci. 2017, 73, 1305–1309. [Google Scholar] [CrossRef][Green Version]
- Heimbach, U.; Müller, A. Incidence of pyrethroid-resistant oilseed rape pests in Germany. Pest Manag. Sci. 2013, 69, 209–216. [Google Scholar] [CrossRef]
- Breitenmoser, S.; Steinger, T.; Hiltpold, I.; Grosjean, Y.; Nussbaum, V.; Bussereau, F.; Klötzli, F.; Widmer, N.; Baux, A. Companion planting in oilseed rape to control adult flea beetle. Swiss Agric. Res. 2020, 11, 11–16. [Google Scholar]
- Walters, K.F.A.; Lane, A.; Cooper, D.A.; Morgan, D. A commercially acceptable assessment technique for improved control of cabbage stem flea beetle feeding on winter oilseed rape. J. Crop Prot. 2001, 20, 907–912. [Google Scholar] [CrossRef]
- Bonnemaison, L. Les Ennemis Animaux des Plantes Cultivées et des Forêts II. Ordre des Coléoptères et Lépidoptères; Editions Sep: Paris, France, 1962; p. 504. [Google Scholar]
- Derron, J.O.; Goy, G. L’altise d’hiver du colza (Psylliodes chrysocephala L.): Biologie, nuisibilité et moyens de lutte. Swiss Agric. Res. 1991, 23, 5–9. [Google Scholar]
- Derron, J.; Breitenmoser, S.; Goy, G.; Grosjean, Y.; Pellet, D. Charançon de la tige du colza: Effet sur le rendement et seuil d’intervention. Rech. Agron. Suisse 2015, 6, 328–335. [Google Scholar]
- Schaefer, H.L.; Brandes, H.; Ulber, B.; Becker, H.C.; Vidal, S. Evaluation of nine genotypes of oilseed rape (Brassica napus L.) for larval infestation and performance of rape stem weevil (Ceutorhynchus napi Gyll.). PLoS ONE 2017, 12, e0180807. [Google Scholar] [CrossRef] [PubMed]
- Free, J.B.; Williams, I.H. The infestation of crops of oil-seed rape (Brassica napus L.) by insect pests. J. Agric. Sci. 1979, 92, 203–218. [Google Scholar] [CrossRef]
- Zlof, V. Recommendations and conclusions of the Ad hoc EPPO Workshop on insecticide resistance of Meligethes spp. (pollen beetle) on oilseed rape. EPPO Bull. 2008, 38, 65–67. [Google Scholar]
- Bothorel, S.; Robert, C.; Ruck, L.; Carpezat, J.; Lauvernay, A.; Leflon, M.; Siegwart, M. Resistance to pyrethroid insecticides in cabbage stem flea beetle (Psylliodes chrysocephala) and rape winter stem weevil (Ceutorhynchus picitarsis) populations in France. Integr. Control. Oilseed Crop. IOBC-WPRS 2018, 136, 89–104. [Google Scholar]
- Dosdall, L.M. Responses of the cabbage seedpod weevil, Ceutorhynchus obstrictus (Marsham) (Coleoptera: Curculionidae), to seed treatments of canola (Brassica napus L.) with the neonicotinoid compounds clothianidin and imidacloprid. Pest Manag. Sci. 2009, 65, 1329–1336. [Google Scholar] [CrossRef]
- Højland, D.H.; Nauen, R.; Foster, S.P.; Williamson, M.S.; Kristensen, M. Incidence, spread and mechanisms of pyrethroid resistance in European populations of the cabbage stem flea beetle, Psylliodes chrysocephala L. (Coleoptera: Chrysomelidae). PLoS ONE 2016, 10, e0146045. [Google Scholar] [CrossRef]
- Willis, C.E.; Foster, S.P.; Zimmer, C.T.; Elias, J.; Chang, X.; Field, L.M.; Williamson, M.S.; Davies, T.G.E. Investigating the status of pyrethroid resistance in UK populations of the cabbage stem flea beetle (Psylliodes chrysocephala). Crop Prot. 2020, 138, 105316. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, C.T.; Müller, A.; Heimbach, U.; Nauen, R. Target-site resistance to pyrethroid insecticides in German populations of the cabbage stem flea beetle, Psylliodes chrysocephala L. (Coleoptera: Chrysomelidae). Pestic. Biochem. Physiol. 2014, 108, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Conrad, N.; Brandes, M.; Heimbach, U. Passive extraction of cabbage stem flea beetle larvae (Psylliodes chrysocephala L.). J. Kult. 2016, 68, 249–252. [Google Scholar] [CrossRef]
- Schneider, C.A.; Rasband, W.S.; Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 2012, 9, 671–675. [Google Scholar] [CrossRef]
- Diepenbrock, W. Yield analysis of winter oilseed rape (Brassica napus L.): A review. Field Crop. Res. 2000, 67, 35–49. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021. [Google Scholar]
- Agridea. Seuils D’intervention Contre les Organismes Nuisibles en Grandes Cultures (PER). Available online: https://www.agridea.ch/fileadmin/AGRIDEA/Theme/Productions_vegetales/Grandes_cultures/bekaempfungsschwellen/1.33-39_2021.pdf (accessed on 15 November 2021).
- Kloen, H.; Altieri, M.A. Effect of mustard (Brassica hirta) as a non-crop plant on competition and insect pests in broccoli (Brassica oleracea). Crop Prot. 1990, 9, 90–96. [Google Scholar] [CrossRef]
- Jamont, M.; Crépellière, S.; Jaloux, B. Effect of extrafloral nectar provisioning on the performance of the adult parasitoid Diaeretiella rapae. Biol. Control 2013, 65, 271–277. [Google Scholar] [CrossRef]
- Rudgers, J.A.; Gardener, M.C. Extrafloral nectar as a resource mediating multispecies interactions. Ecology 2004, 85, 1495–1502. [Google Scholar] [CrossRef]
- Wäckers, F.L. Suitability of (extra-) floral nectar, pollen, and honeydew as insect food sources. In Plant-Provided Food for Carnivorous Insects: A Protective Mutualism and Its Applications; Wäckers, F.L., Van Rijn, P.C.J., Bruin, J., Eds.; Cambrige University Press: Cambrige, UK, 2005; pp. 17–74. [Google Scholar]
- Faria, C.A.; Wäckers, F.L.; Turlings, T.C.J. The nutritional value of aphid honeydew for non-aphid parasitoids. Basic Appl. Ecol. 2008, 9, 286–297. [Google Scholar] [CrossRef]
- Lai, R.; Hu, H.; Wu, X.; Bai, J.; Gu, G.; Bai, J.; Zhou, T.; Lin, T.; Zhong, X. Intercropping oilseed rape as a potential relay crop for enhancing the biological control of green peach aphids and aphid-transmitted virus diseases. Entomol. Exp. Et Appl. 2019, 167, 969–976. [Google Scholar] [CrossRef]
- Gardarin, A.; Pigot, J.; Valantin-Morison, M. The hump-shaped effect of plant functional diversity on the biological control of a multi-species pest community. Sci. Rep. 2021, 11, 21635. [Google Scholar] [CrossRef]
- Dong, N.; Tang, M.M.; Zhang, W.P.; Bao, X.G.; Wang, Y.; Christie, P.; Li, L. Temporal differentiation of crop growth as one of the drivers of intercropping yield advantage. Sci. Rep. 2018, 8, 3110. [Google Scholar] [CrossRef] [PubMed]
- Momoh, E.J.J.; Zhou, W. Growth and yield responses to plant density and stage of transplanting in winter oilseed rape (Brassica napus L.). J. Agron. Crop Sci. 2001, 186, 253–259. [Google Scholar] [CrossRef]
- de Baan, L.; Spycher, S.; Daniel, O. Utilisation des produits phytosanitaires en Suisse de 2009 à 2012. Rech. Agron. Suisse 2015, 2, 48–55. [Google Scholar]
- Dowling, A.; O Sadras, V.; Roberts, P.; Doolette, A.; Zhou, Y.; Denton, M.D. Legume-oilseed intercropping in mechanised broadacre agriculture—A review. Field Crop. Res. 2021, 260, 107980. [Google Scholar] [CrossRef]
- Tian, X.L.; Wang, C.B.; Bao, X.G.; Wang, P.; Li, X.F.; Yang, S.C.; Ding, G.C.; Christie, P.; Li, L. Crop diversity facilitates soil aggregation in relation to soil microbial community composition driven by intercropping. Plant Soil 2019, 436, 173–192. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Breitenmoser, S.; Steinger, T.; Baux, A.; Hiltpold, I. Intercropping Winter Oilseed Rape (Brassica napus L.) Has the Potential to Lessen the Impact of the Insect Pest Complex. Agronomy 2022, 12, 723. https://doi.org/10.3390/agronomy12030723
Breitenmoser S, Steinger T, Baux A, Hiltpold I. Intercropping Winter Oilseed Rape (Brassica napus L.) Has the Potential to Lessen the Impact of the Insect Pest Complex. Agronomy. 2022; 12(3):723. https://doi.org/10.3390/agronomy12030723
Chicago/Turabian StyleBreitenmoser, Stève, Thomas Steinger, Alice Baux, and Ivan Hiltpold. 2022. "Intercropping Winter Oilseed Rape (Brassica napus L.) Has the Potential to Lessen the Impact of the Insect Pest Complex" Agronomy 12, no. 3: 723. https://doi.org/10.3390/agronomy12030723
APA StyleBreitenmoser, S., Steinger, T., Baux, A., & Hiltpold, I. (2022). Intercropping Winter Oilseed Rape (Brassica napus L.) Has the Potential to Lessen the Impact of the Insect Pest Complex. Agronomy, 12(3), 723. https://doi.org/10.3390/agronomy12030723






