Examination of the Effectiveness of Controlled Release Fertilizer to Balance Sugarcane Yield and Reduce Nitrate Leaching to Groundwater
Abstract
1. Introduction
- Evaluate the effectiveness of CRF on sugarcane yield enhancement by analyzing growth and yield parameters;
- Analyze the characteristics of nitrate-N leaching related to different fertilizer rates; and
- Calculate the WFs of sugarcane cultivation and evaluate the potential impact of nitrate-N leaching on GW.
2. Materials and Methods
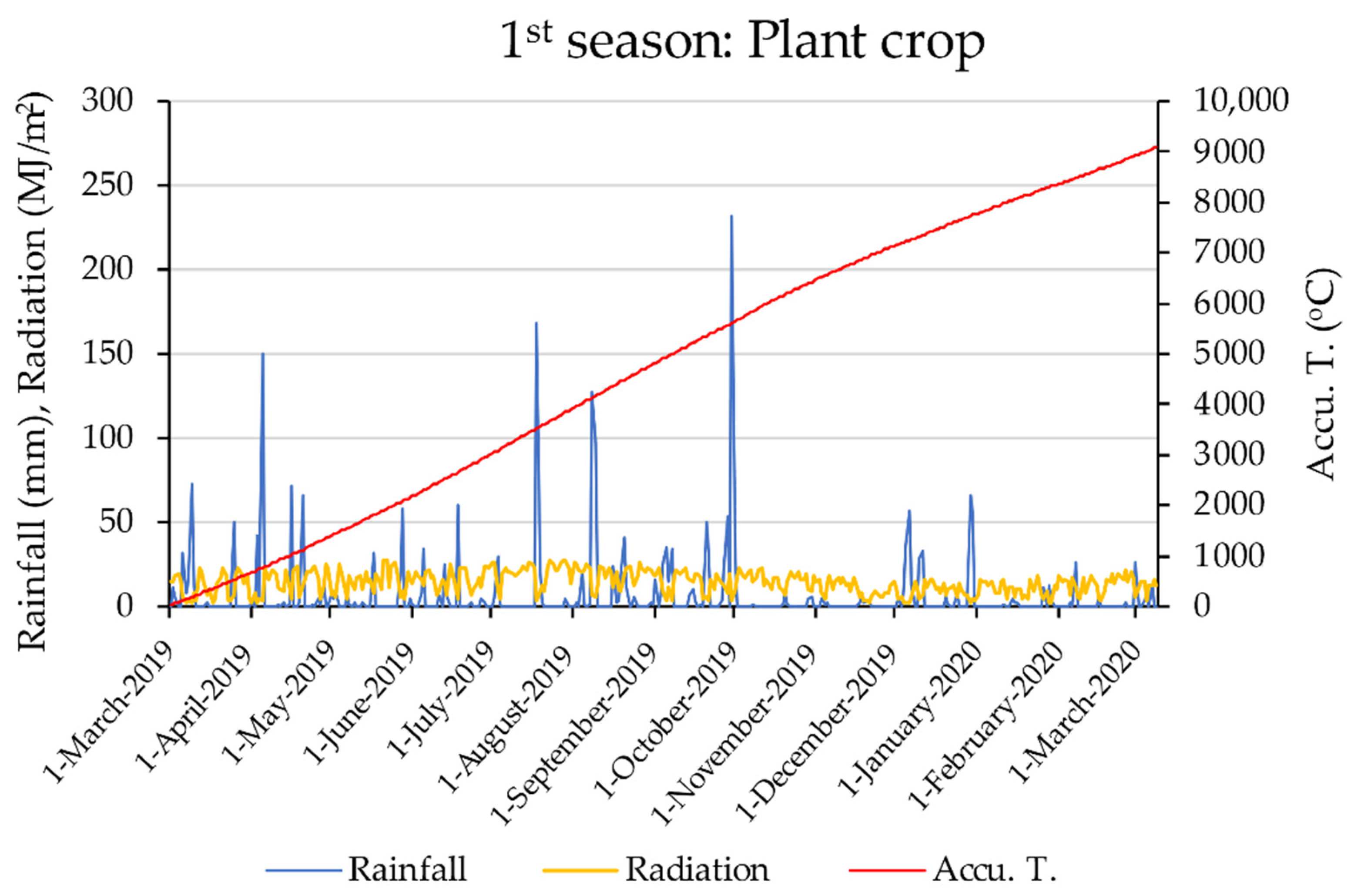
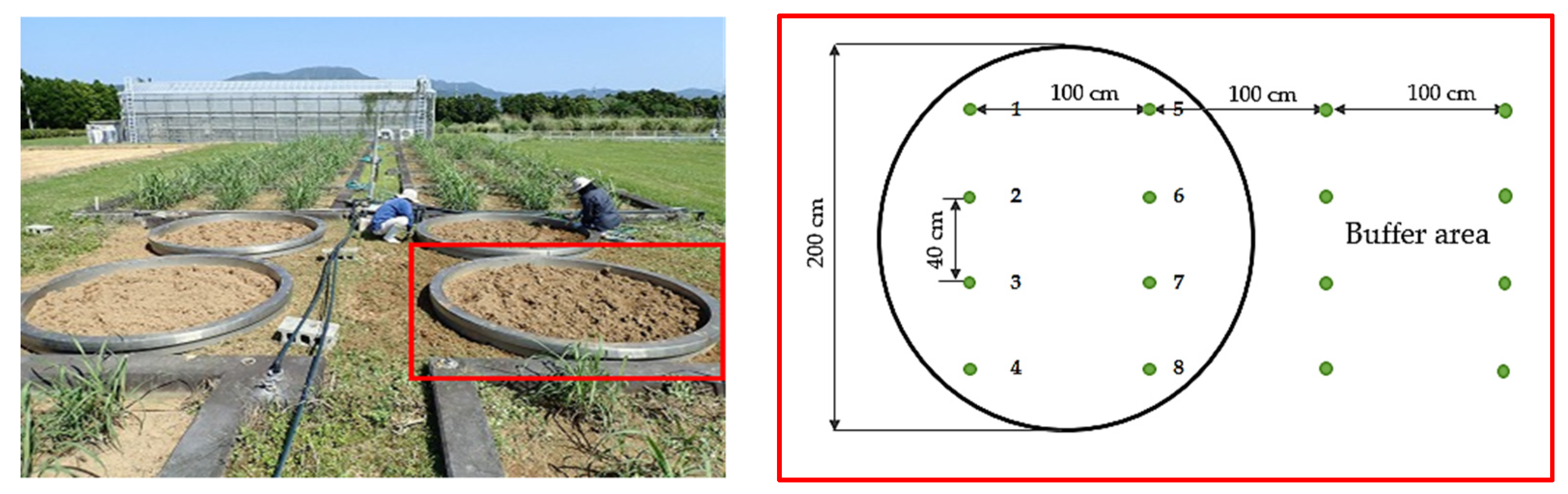
2.1. Experimental Site
2.2. Data Collection
2.3. Statistical Analysis and Data Interpretation
2.3.1. Analysis of Growth and Harvesting Parameters
2.3.2. Analysis of Nitrate-N Leaching Data
2.4. WF Assessment
3. Results
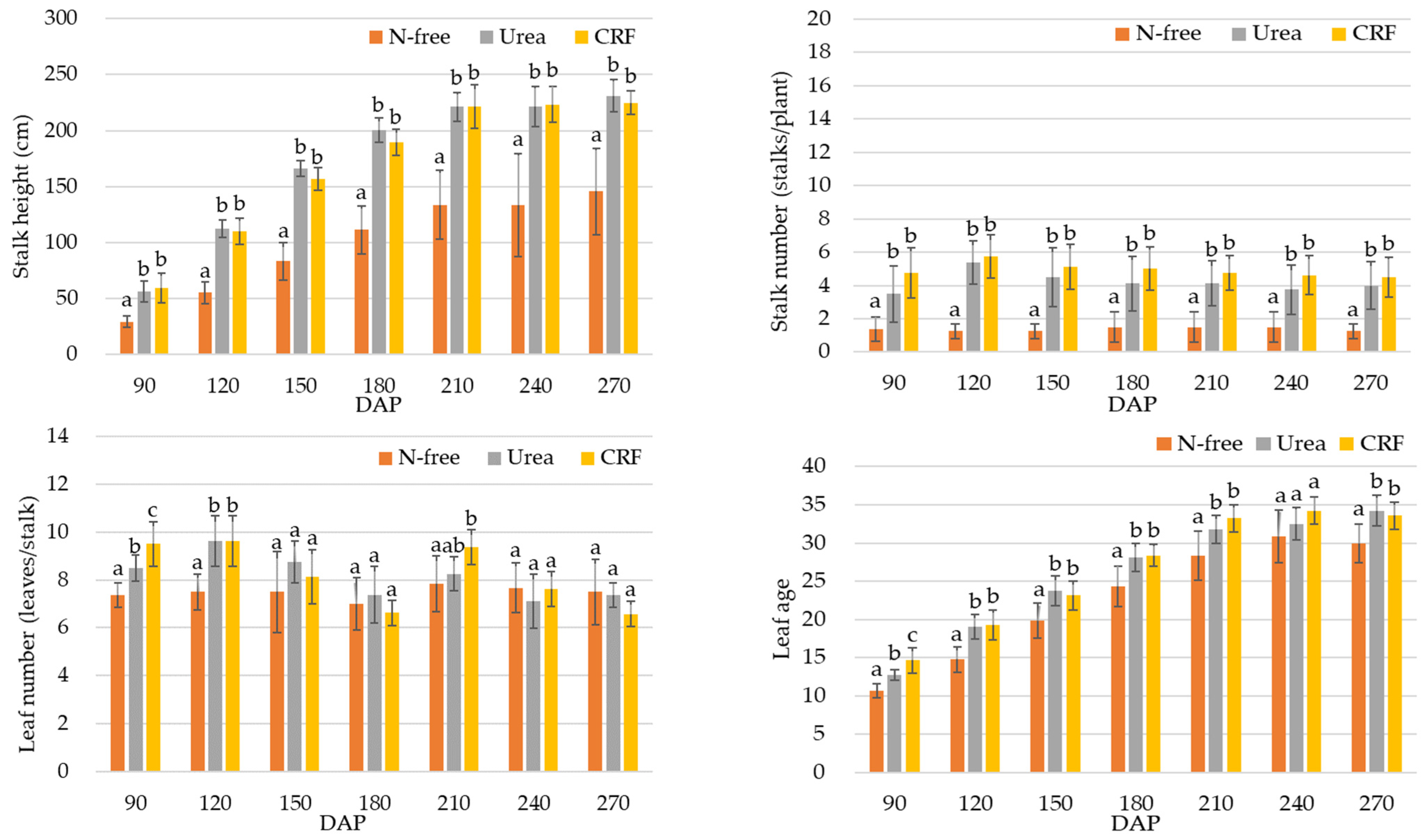
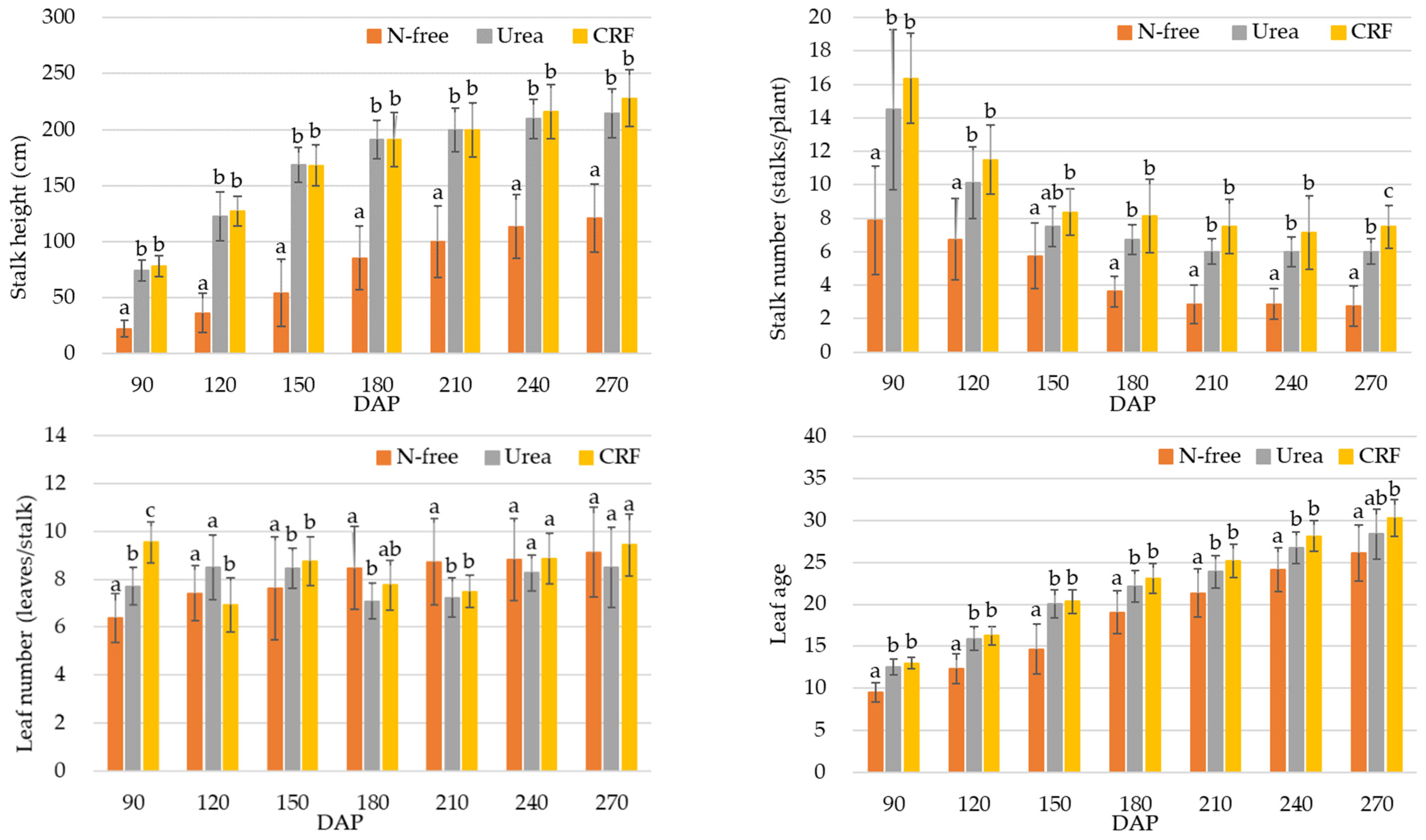
3.1. Results of the Growth and Harvesting Surveys
3.1.1. Results of the Growth Survey
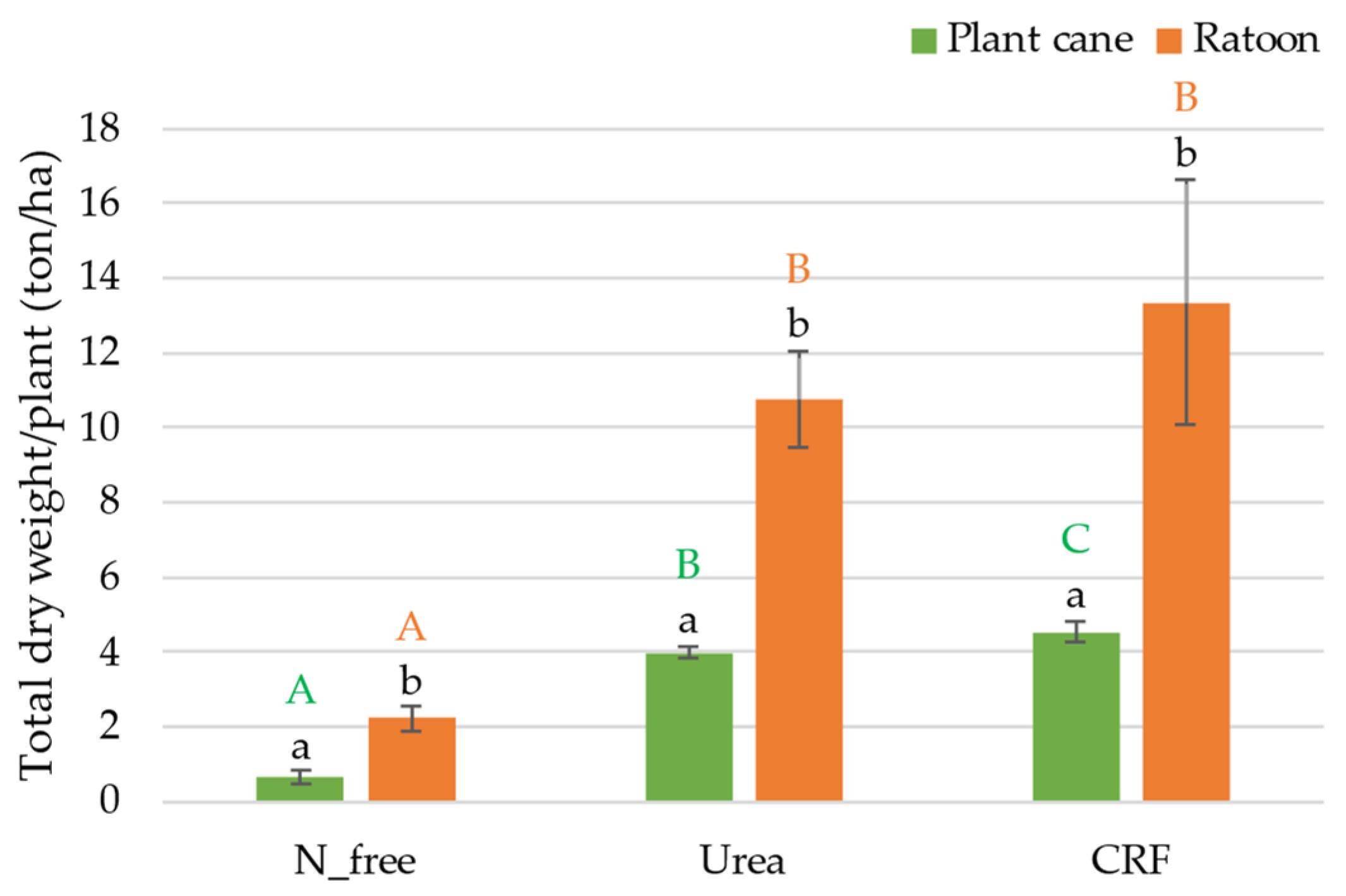
3.1.2. Results of the Harvesting Survey
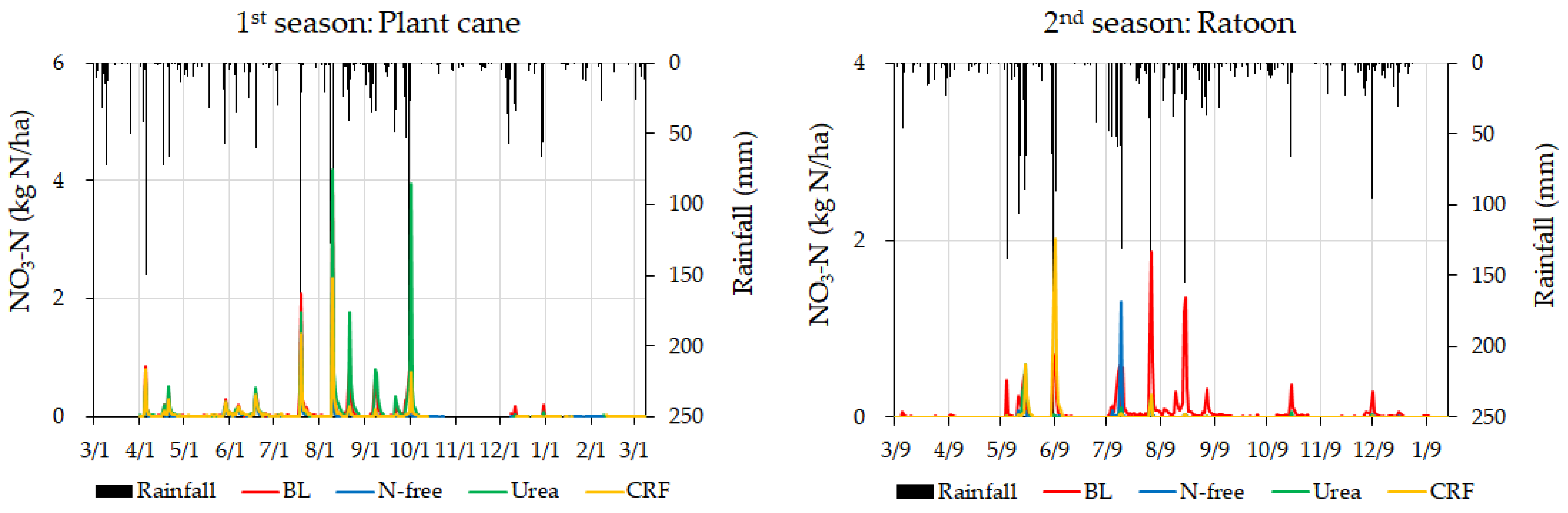
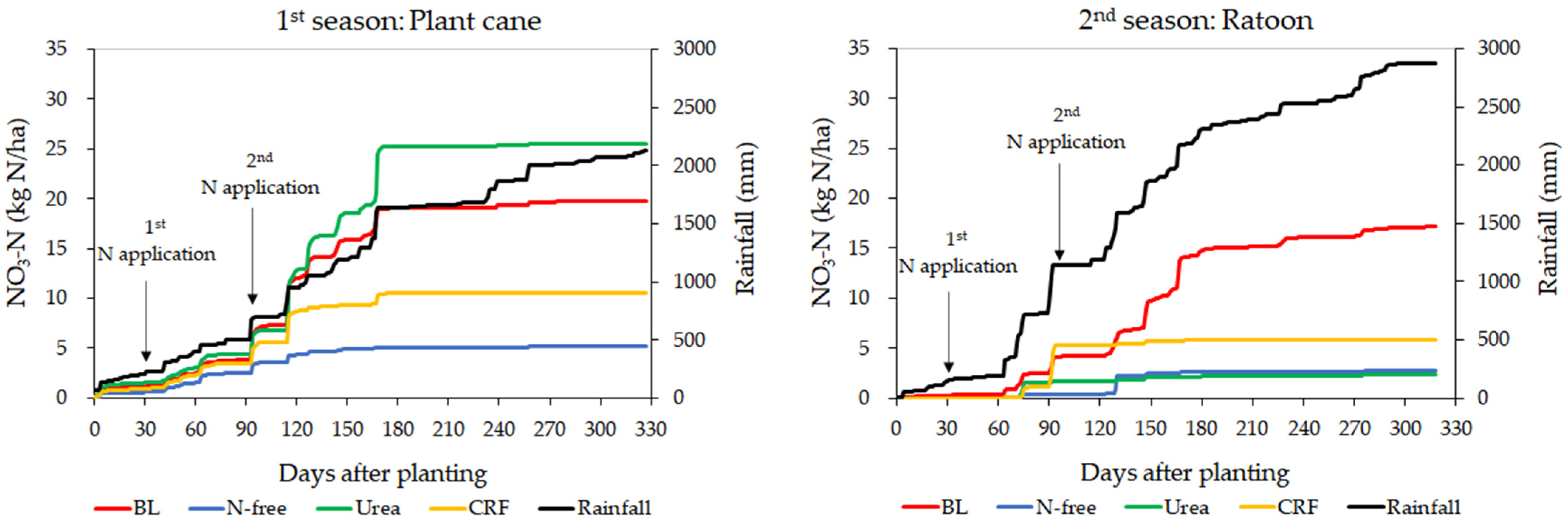
3.2. Variation of Leached Nitrate-N
3.3. WF of Sugarcane Cultivation
4. Discussion
4.1. Analysis of the Growth and Harvesting Parameters
4.2. Analysis of Leached Nitrate-N
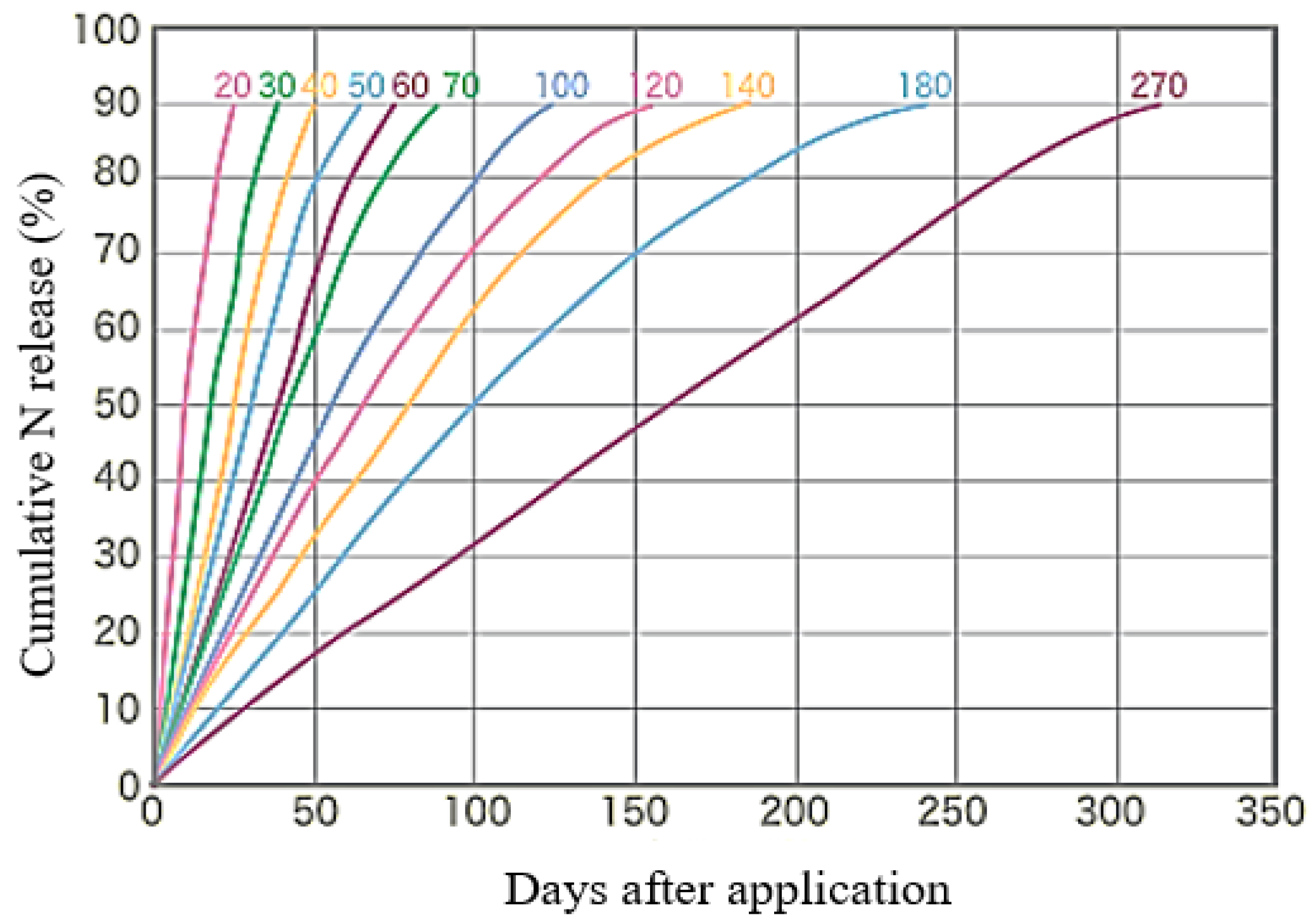
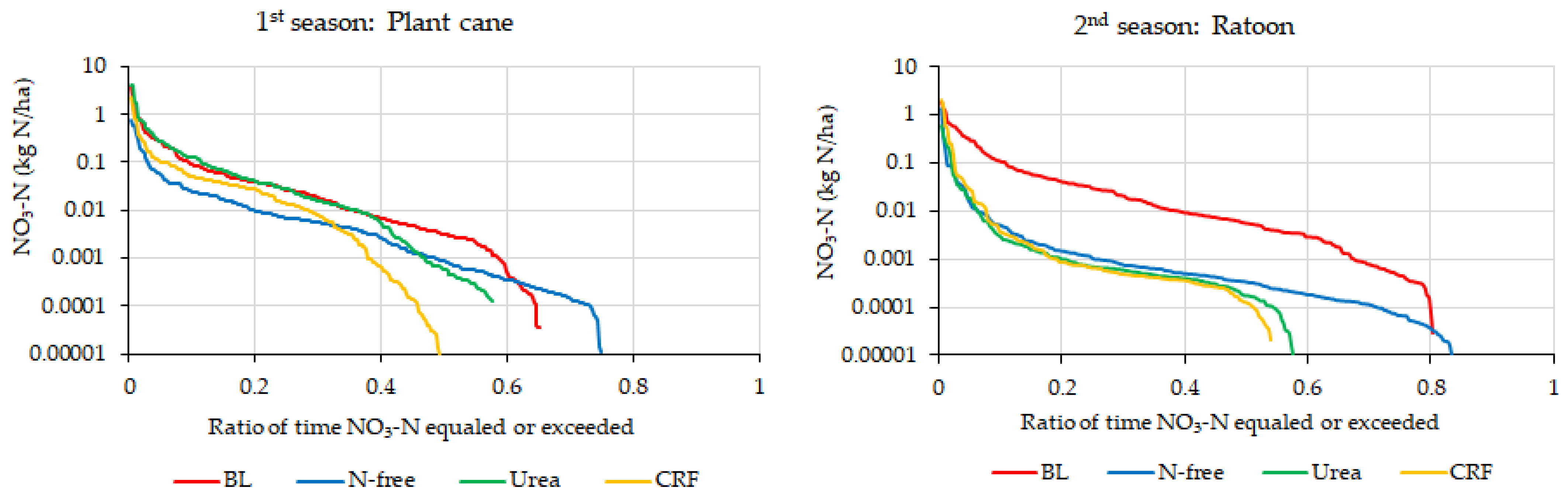
4.2.1. Evaluation of Daily and Cumulative Variation of Nitrate-N Leaching
Consideration of the Relationship between N Leaching and C:N Ratio
Consideration of the Relationship between N Leaching and Infiltration
Consideration of the Relationship between N Leaching and NHC
Consideration of the Relationship between N Leaching and N Uptake by Plants
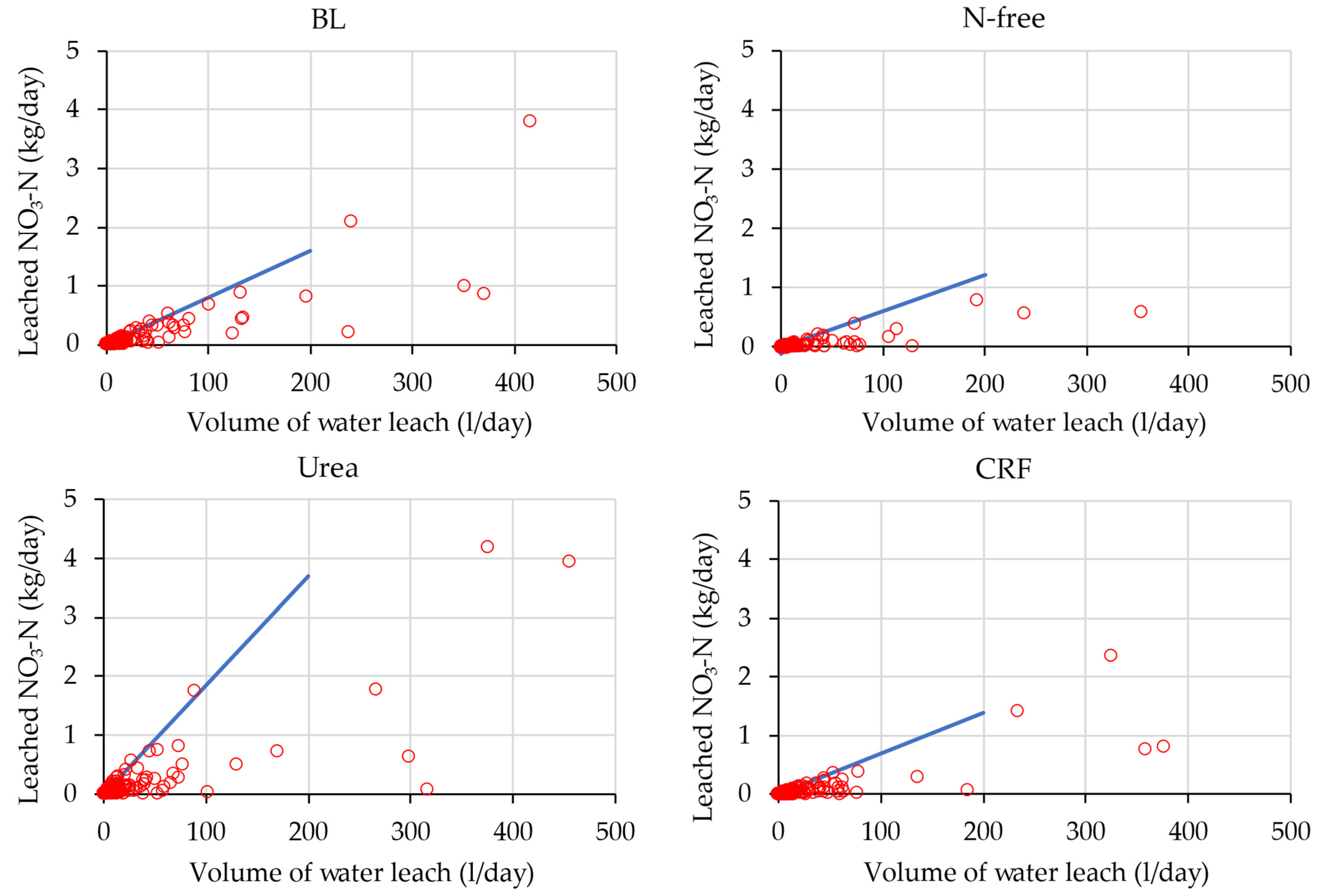
4.2.2. Evaluation of the Relationship between Water Leaching and Nitrate-N Leaching
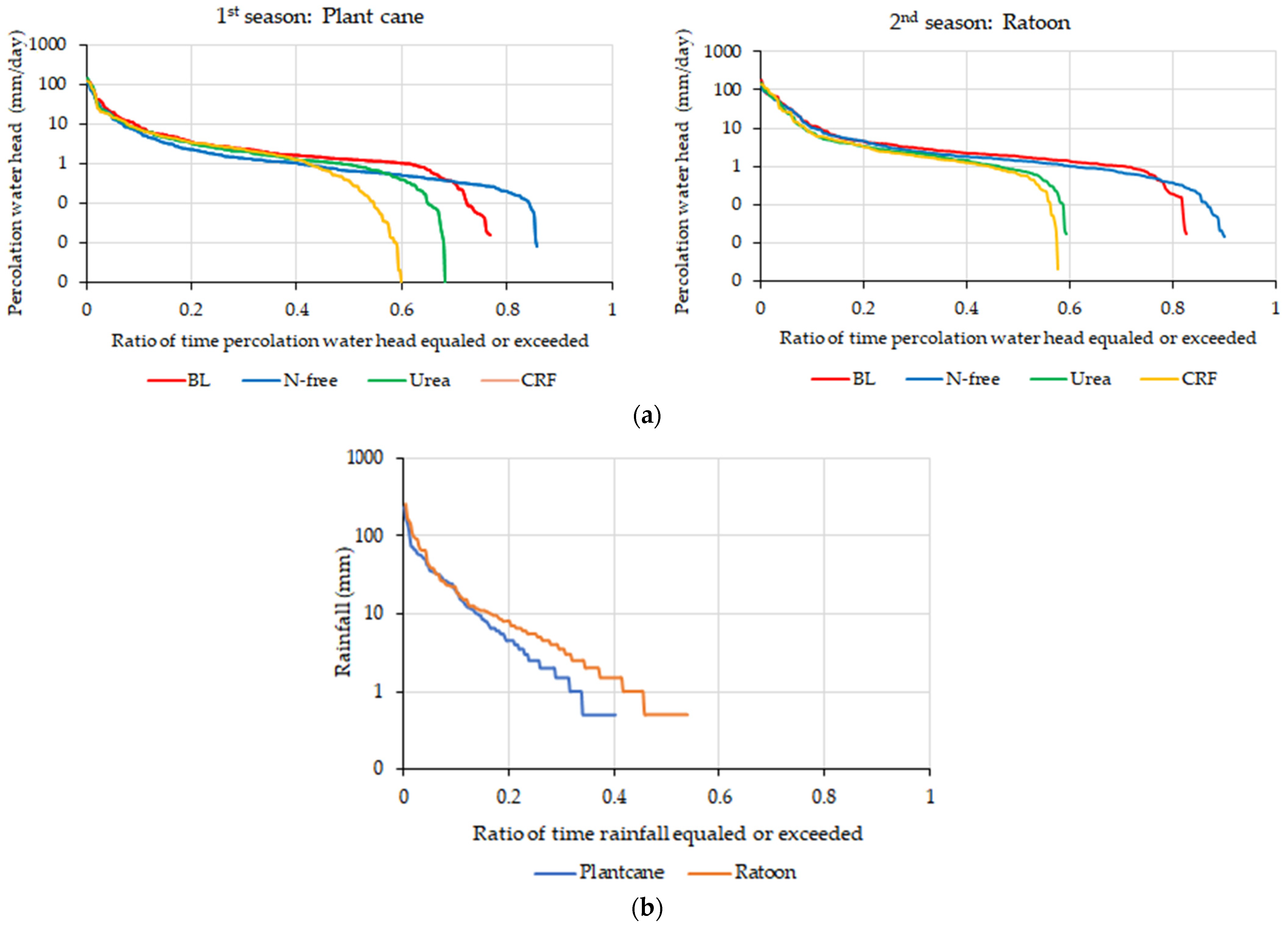
Evaluation of Duration Curves of Water Leaching and Nitrate-N Leaching
Evaluation of Envelop Lines of Water Leaching and Nitrate-N Leaching of Low Leaching Events
4.3. WF of Sugarcane Cultivation
Sustainability Assessment on Obtained WFs of Sugarcane Cultivation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prasertsak, P.; Freney, J.R.; Denmead, O.T.; Saffigna, P.G.; Prove, B.G.; Reghenzani, J.R. Effect of Fertilizer Placement on Nitrogen Loss from Sugarcane in Tropical Queensland. Nutr. Cycl. Agroecosyst. 2002, 62, 229–239. [Google Scholar] [CrossRef]
- Wang, W.; Park, G.; Reeves, S.; Zahmel, M.; Heenan, M.; Salter, B. Nitrous Oxide Emission and Fertiliser Nitrogen Efficiency in a Tropical Sugarcane Cropping System Applied with Different Formulations of Urea. Soil Res. 2016, 54, 572–584. [Google Scholar] [CrossRef]
- Buss, S.R.; Rivett, M.O.; Morgan, P.; Bemment, C.D. Using Science to Create a Better Place: Attenuation of Nitrate in the Sub-Surface Environment; Waterside Drive, Aztec West Almondsbury: Bristol, UK, 2005. [Google Scholar]
- Killpack, S.C.; Buchholz, D. Nitrogen in the Environment: Leaching. Available online: https://extension.missouri.edu/publications/wq262 (accessed on 5 March 2022).
- Fernandez, F.G.; Kaiser, D.E. Understanding Nitrogen in Soils. 2021. Available online: https://extension.umn.edu/nitrogen/understanding-nitrogen-soils#nitrification-761161 (accessed on 4 March 2022).
- University of Nebraska. Section D: What Happens When Nitrogen Is Applied to The Soil? Available online: https://water.unl.edu/documents/Section%20D.pdf (accessed on 5 March 2022).
- International Plant Nutrition Institute (IPNI). Nitrate Leaching. Available online: http://www.ipni.net/publication/nitrogen-en.nsf/0/FDEE48CFF7600CE585257C13004C7BB0/$FILE/NitrogenNotes-EN-03.pdf (accessed on 4 March 2022).
- Provin, T.L.; Hossner, L.R. What Happens to Nitrogen in Soils? Available online: https://agrilifeextension.tamu.edu/library/gardening/what-happens-to-nitrogen-in-soils/ (accessed on 5 March 2022).
- Johnson, C.; Albrecht, G.; Ketterings, Q.; Beckman, J.; Stockin, K. Nitrogen Basics—The Nitrogen Cycle Agronomy Fact Sheet Series; Cornell University: New York, NY, USA, 2005. [Google Scholar]
- Bernhard, A. The Nitrogen Cycle: Processes, Players, and Human Impact. Nat. Educ. Knowl. 2010, 3, 25. [Google Scholar]
- Norton, J.M. Nitrification in Agricultural Soils. In Nitrogen in Agricultural Systems; Schepers, J.S., Raun, W.R., Eds.; American Society of Agronomy, Crop Science Society of America, Soil Science Society of America: Madison, WI, USA, 2008; Volume 49, pp. 173–199. [Google Scholar] [CrossRef]
- Myrold, D.D.; Bottomley, P.J. Nitrogen Mineralization and Immobilization. In Nitrogen in Agricultural Systems; Schepers, J.S., Raun, W.R., Eds.; American Society of Agronomy, Crop Science Society of America, Soil Science Society of America: Madison, WI, USA, 2008; Volume 49, pp. 157–172. [Google Scholar] [CrossRef]
- Plant and Soil Sciences eLibrary. Soils—Part 5: Nitrogen as a Nutrient. Available online: https://passel2.unl.edu/view/lesson/3176eba1ba31/2 (accessed on 4 March 2022).
- Sousa, J.R.; Cabral, F.; Coutinho, J. Assessment of N Mineralization and N Leaching in Soil Using a New in situ Incubation Method. Commun. Soil Sci. Plant Anal. 2016, 47, 2157–2167. [Google Scholar] [CrossRef]
- Dawes, M.A.; Schleppi, P.; Hättenschwiler, S.; Rixen, C.; Hagedorn, F. Soil warming opens the nitrogen cycle at the alpine treeline. Glob. Change Biol. 2017, 23, 421–434. [Google Scholar] [CrossRef]
- Vitousek, P.M.; Porder, S.; Houlton, B.Z.; Chadwick, O.A. Terrestrial phosphorus limitation: Mechanisms, implications, and nitrogen-phosphorus interactions. Ecol. Appl. 2010, 20, 5–15. [Google Scholar] [CrossRef]
- Gan, H.Y.; Schöning, I.; Schall, P.; Ammer, C.; Schrumpf, M. Soil Organic Matter Mineralization as Driven by Nutrient Stoichiometry in Soils Under Differently Managed Forest Stands. Front. For. Glob. Change 2020, 3, 99:1–99:15. [Google Scholar] [CrossRef]
- Fu, M.H.; Xu, X.C.; Tabatabai, M.A. Effect of pH on nitrogen mineralization in crop-residue-treated soils. Biol. Fertil. Soils 1987, 5, 115–119. [Google Scholar] [CrossRef]
- Matsuoka, M. Sugarcane Cultivation and Sugar Industry in Japan. Sugar Tech. 2006, 8, 3–9. [Google Scholar] [CrossRef]
- Verburg, K.; Harvey, T.G.; Muster, T.H.; Brennan McKellar, L.; Thorburn, P.; Biggs, J.; Di Bella, L.; Wang, W. Use of Enhanced Efficiency Fertilisers to Increase Fertiliser Nitrogen Use Efficiency in Sugarcane. In A Review of Nitrogen Use Efficiency in Sugarcane; Bell, M.J., Ed.; Sugar Research Australia: Indooroopilly, Australia, 2015; pp. 229–280. [Google Scholar]
- Yoshimoto, S.; Tsuchihara, T.; Ishida, S.; Masumoto, T.; Imaizumi, M. Groundwater Flow and Transport and Potential Sources of Groundwater Nitrates in the Ryukyu Limestone as a Mixed Flow Aquifer in Okinawa Island, Japan. Paddy Water Environ. 2011, 9, 367–384. [Google Scholar] [CrossRef]
- Seiko, S. A Report from Miyako Island: What Is Happening on the Small Islands of Okinawa? Available online: https://www.ecojesuit.com/a-report-from-miyako-island-what-is-happening-on-the-small-islands-of-okinawa/ (accessed on 10 January 2021).
- Shimoji, N.; Kawata, S.; Hiyane, M.; Kadekari, S. For Protecting the Life Sustaining Groundwater of Miyako Island: The Development of an Environmentally Friendly Organic Fertilizer Utilizing Phosphorus Accumulated in the Soil. In Proceedings of the 6th Pacific Islands Leaders Meeting (PALM6), Miyakojima, Japan, 23–26 May 2012. [Google Scholar]
- Miyakojima City Office. Miyakojima City Groundwater Quality Conservation Survey Report. 2011. (In Japanese). Available online: https://www.city.miyakojima.lg.jp/kurashi/seikatsu/kankyohozen/files/H26rep.pdf (accessed on 15 January 2021).
- Pastore, M. The Impact of Nitrogen Eutrophication on Caribbean Coral Reefs: A Review. Concept 2014, 37. Available online: https://concept.journals.villanova.edu/index.php/concept/article/view/1725/1592/ (accessed on 4 March 2022).
- Fujiie, R.; Nakagawa, Y.; Shima, T.; Shiono, T.; Shinogi, Y. Estimation of Leached Nitrate-Nitrogen in Groundwater Basin, Miyako Island [Japan]. AGRIS 2008, 127–138. Available online: https://agris.fao.org/agris-search/search.do?recordID=JP2008003564 (accessed on 4 March 2022).
- Agata, S. Chemical Characteristics and Isotopic Compositions of Spring and River Waters in Okinawa Island. Chikyukagaku 2001, 45, 27–41. [Google Scholar]
- Nakagawa, Y.; Yan, C.; Shiono, T.; Miyamoto, T.; Kameyama, K.; Shinogi, Y. Evaluating the Validity and Sensitivity of the DNDC Model for Shimajiri Dark Red Soil. Japan Agric. Res. Q. 2008, 42, 163–172. [Google Scholar] [CrossRef][Green Version]
- Shuhei, Y. Dynamics of Groundwater Nitrates in Limestone Aquifer of the Southern Okinawa Island. In Agricultural and Industrial Research Report; National Agriculture and Food Research Organization (NARO): Tsukuba, Japan, 2013; Volume 52, pp. 59–110. [Google Scholar]
- Hoekstra, A.Y.; Chapagain, A.K.; Aldaya, M.M.; Mekonnen, M.M. The Water Footprint Assessment Manual. Setting the Global Standard; Earthscan: Longdon, UK, 2011; ISBN 978-1-84971-279-8. [Google Scholar]
- Kongboon, R.; Sampattagul, S. The Water Footprint of Sugarcane and Cassava in Northern Thailand. Procedia—Soc. Behav. Sci. 2012, 40, 451–460. [Google Scholar] [CrossRef]
- Scarpare, F.V.; Hernandes, T.A.D.; Ruiz-Corrêa, S.T.; Kolln, O.T.; Gava, G.J.D.C.; Dos Santos, L.N.S.; Victoria, R.L. Sugarcane Water Footprint under Different Management Practices in Brazil: Tietê/Jacaré Watershed Assessment. J. Clean. Prod. 2016, 112, 4576–4584. [Google Scholar] [CrossRef]
- Del Milagro Jorrat, M.; Araujo, P.Z.; Mele, F.D. Sugarcane Water Footprint in the Province of Tucumán, Argentina. Comparison between Different Management Practices. J. Clean. Prod. 2018, 188, 521–529. [Google Scholar] [CrossRef]
- Hossain, M.A.; Ueno, M.; Maeda, K.; Kawamitsu, Y. Potential Evapotranspiration and Crop Coefficient Estimates for Sugarcane in Okinawa. J. Agric. Mereorol. 2005, 60, 573–576. [Google Scholar] [CrossRef]
- Bellido, J.; Sakai, K.; Nakamura, S.; Kazuro, M.; Okamoto, K.; Nakandakari, T. N2O Emissions from Shimajiri-Maji (Calcaric Dark Red Soil) after Applying Two Chemical Fertilizers. Appl. Ecol. Environ. Res. 2015, 13, 339–348. [Google Scholar] [CrossRef]
- Hamazaki, T. Diversity of Soils and Soil Management Measures in Tropical and Subtropical Islands. In Proceedings of the JIRCAS 2005 International Symposium, Ishigaki, Japan, 11 March 2005; pp. 83–96. [Google Scholar]
- Verburg, K.; Muster, T.H.; Zhao, Z.; Biggs, J.S.; Thorburn, P.J.; Kandulu, J.; Wittwer-Schmid, K.; McLachlan, G.; Bristow, K.L.; Poole, J.; et al. Role of Controlled Release Fertilizer in Australian Sugarcane Systems: Final Report 2014/011; Sugar Research Australia Limited: Indooroopilly, Australia, 2017. [Google Scholar]
- Visiongain. Controlled-Release Fertilizers Market Report to 2031. 2021. Available online: https://www.visiongain.com/controlled-release-fertilizers-market-analysis/ (accessed on 5 March 2022).
- Subbarao, C.V.; Kartheek, G.; Sirisha, D. Slow Release of Potash Fertilizer through Polymer Coating. Int. J. Appl. Sci. Eng. 2013, 11, 25–30. [Google Scholar]
- Trenkel, M.E. Slow and Controlled-Release and Stabilized Fertilizers: An Option for Enhancing Nutrient Efficiency in Agriculture, 2nd ed.; International Fertilizer Industry Association: Paris, France, 2010; ISBN 978-2-9523139-7-1. [Google Scholar]
- Kubotera, H.; Wada, S.-I. An experimental method for the direct measurement of nitrate retention of soils in conditions similar to the field. Jpn. Soc. Pedol. 2008, 52, 118–125. [Google Scholar] [CrossRef]
- Wong, M.T.; Hughes, R.; Rowell, D.L. Retarded leaching of nitrate in acid soils from the tropics: Measurement of the effective anion exchange capacity. J. Soil Sci. 1990, 41, 655–663. [Google Scholar] [CrossRef]
- Bray, R.H.; Kurt, L.T. Determination of total, organic, and available forms of phosphorus in soils. Soil Sci. 1945, 59, 39–45. [Google Scholar] [CrossRef]
- Japanese Ministry of Environment, Environmental Quality Standards for Water Pollution. Available online: https://www.env.go.jp/en/water/wq/wp.pdf (accessed on 24 January 2021).
- Nakanishi, Y.; Yamamoto, Y.; Park, K.L.; Kato, S.; Kumazawa, K. Estimation and Verification of Origins of Groundwater Nitrate by Using delts 15N Values. Jpn. J. Soil Sci. Plant Nutr. 1995, 66, 544–551. (In Japanese) [Google Scholar]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 4 March 2022).
- De Mendiburu, F. Agricolae: Statistical Procedures for Agricultural Research. R Package Version 1.3-5. 2021. Available online: https://cran.r-project.org/package=agricolae (accessed on 4 March 2022).
- Food and Agriculture Organization (FAO). CropWat. Available online: http://www.fao.org/land-water/databases-and-software/cropwat/en/ (accessed on 24 January 2021).
- Santos, F.; Diola, V. Chapter 2-Physiology. In Sugarcane: Agricultural Production, Bioenergy and Ethanol; Borem, A., Caldas, C., Eds.; Academic Press: San Diego, CA, USA, 2015; pp. 13–33. ISBN 978-0-12-802239-9. [Google Scholar]
- Dinh, T.H.; Watanabe, K.; Takaragawa, H.; Nakabaru, M.; Kawamitsu, Y. Photosynthetic Response and Nitrogen Use Efficiency of Sugarcane under Drought Stress Conditions with Different Nitrogen Application Levels. Plant Prod. Sci. 2017, 20, 412–422. [Google Scholar] [CrossRef]
- El-Hinnawy, H.H.; Masri, M.I. Crop Cycle Effects on Genetic Variability, Heritability, and Yield of Sugarcane. J. Plant Prod. 2009, 34, 6749–6761. [Google Scholar] [CrossRef]
- United States Department of Agriculture (USDA)-Natural Resources Conservation Service. Carbon to Nitrogen Ratios in Cropping Systems Introduction. Available online: https://www.nrcs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb1166766.pdf (accessed on 10 December 2020).
- Swangjang, K. Soil Carbon and Nitrogen Ratio in Different Land Use. In Proceedings of the International Conference on Advances in Environment Research, Jeju, Korea, 29–30 July 2015; pp. 36–40. [Google Scholar]
- Brust, G.E. Chapter 9—Management Strategies for Organic Vegetable Fertility. In Safety and Practice for Organic Food; Biswas, D., Micallef, S.A., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 193–212. ISBN 978-0-12-812060-6. [Google Scholar]
- Meter Environment. How to Measure Soil Hydraulic Conductivity—Which Method Is Right for You? Available online: https://www.metergroup.com/en/meter-environment/measurement-insights/how-measure-soil-hydraulic-conductivity-which-method-right-you (accessed on 5 March 2022).
- Bagarello, V.; Castellini, M.; Iovino, M. Influence of the pressure head sequence on the soil hydraulic conductivity determined with tension infiltrometer. Appl. Eng. Agric. 2005, 21, 383–391. [Google Scholar] [CrossRef]
- Hallaq, A.H. The impact of soil texture on nitrates leaching into groundwater in the north governorate, Gaza strip. J. Soc. Sci. 2010, 38, 11–35. [Google Scholar]
- Magdoff, F.; Van Es, H. Building Soils for Better Crops: Ecological Management for Healthy Soils, 4th ed.; Sustainable Agriculture Research and Education (SARE) Program: Maryland, MD, USA, 2021; Volume 10. [Google Scholar]
- Kubotera, H.; Wada, S.-I. Factors influencing nitrate retention in 3 Andisol profiles in Kyushu, Japan. In Proceedings of the 19th World Congress of Soil Science for a Changing World, Brisbane, Australia, 1–6 August 2010; pp. 66–69. [Google Scholar]
- Nicol, G.W.; Leininger, S.; Schleper, C.; Prosser, J.I. The influence of soil pH on the diversity, abundance and transcriptional activity of ammonia oxidizing archaea and bacteria. Environ. Microbiol. 2008, 10, 2966–2978. [Google Scholar] [CrossRef]
- Soil Quality Pty Ltd. Soil Acidity|Fact Sheets. 2021. Available online: https://soilquality.org.au/factsheets/soil-acidity (accessed on 25 November 2021).
- Koumanov, K.S.; Stoilov, G.P.; Dochev, D.V. The ‘nitrate nitrogen—Electrical conductivity’ relationship in non-saline soils under fertigation. In Proceedings of the ICID 19th European Regional Conference “Sustainable Use of Land and Water”, Brno and Prague, Czech Republic, 4–8 June 2001. [Google Scholar]
- Verburg, K.; Keating, B.A.; Probert, M.E.; Bristow, K.L.; Huth, N.I. Nitrate Leaching under Sugarcane: Interactions between Crop Yield, Soil Type and Management Strategies. In Proceedings of the 9th Australian Agronomy Conference, Wagga, Australia, 20–23 July 1998; pp. 717–720. [Google Scholar]
- Iqbal, M.T. Study on Vertical and Lateral Leaching of Nitrate from a Wheat Field in China. Turkish J. Agric. For. 2006, 30, 59–65. [Google Scholar]
- Kameyama, K.; Miyamoto, T.; Shinogi, Y. Increases in Available Water Content of Soils by Applying Bagasse-Charcoals. In Proceedings of the 19th World Congress of Soil Science, Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010; pp. 105–108. [Google Scholar]
- Chen, J.; Wei, X. Controlled-Release Fertilizers as a Means to Reduce Nitrogen Leaching and Runoff in Container-Grown Plant Production. In Nitrogen in Agriculture: Updates; Amanullah, K., Fahad, S., Eds.; InTech: Rijeka, Croatia, 2018; pp. 33–52. ISBN 978-953-51-3769-6. [Google Scholar]
- Aranibar, J.N.; Otter, L.; Macko, S.A.; Feral, C.J.W.; Epstein, H.E.; Dowty, P.R.; Eckardt, F.; Shugart, H.H.; Swap, R.J. Nitrogen Cycling in the Soil-Plant System along a Precipitation Gradient in the Kalahari Sands. Glob. Change Biol. 2004, 10, 359–373. [Google Scholar] [CrossRef]
- Gu, C.; Riley, W.J. Combined Effects of Short Term Rainfall Patterns and Soil Texture on Soil Nitrogen Cycling—A Modeling Analysis. J. Contam. Hydrol. 2010, 112, 141–154. [Google Scholar] [CrossRef] [PubMed]
- Ridolfi, E.; Kumar, H.; Bárdossy, A. A methodology to estimate flow duration curves at partially ungauged basins. Hydrol. Earth Syst. Sci. 2020, 24, 2043–2060. [Google Scholar] [CrossRef]
- Cameron, K.C.H.; Di, J.; Moir, J.L. Nitrogen losses from the soil/plant system: A review. Ann. Appl. Biol. 2013, 162, 145–173. [Google Scholar] [CrossRef]
- Fito, J.; Tefera, N.; Demeku, S.; Kloos, H. Water Footprint as an Emerging Environmental Tool for Assessing Sustainable Water Use of the Bioethanol Distillery at Metahara Sugarcane Farm, Oromiya Region, Ethiopia. Water Conserv. Sci. Eng. 2017, 2, 165–176. [Google Scholar] [CrossRef]
- Mekonnen, M.M.; Hoekstra, A.Y. The Green, Blue and Grey Water Footprint of Crops and Derived Crop Products. Hydrol. Earth Syst. Sci. 2011, 15, 1577–1600. [Google Scholar] [CrossRef]
- Chico, D.; Zhang, G. Water Footprint Assessment of FMO’s Agribusiness Portfolio. 2015. Available online: https://waterfootprint.org/media/downloads/FMO_Sugar_supply_chain-WFN_-formatted.pdf (accessed on 4 March 2022).
- Chapagain, A.; Mathews, R. A Guide to Reducing the Water Footprint of Cotton Cultivation in India. 2017. Available online: https://waterfootprint.org/media/downloads/A_guide_to_reduce_water_footprint_of_cotton_cultivation.pdf (accessed on 4 March 2022).
- Borsato, E.; Sartori, L.; Tarolli, P.; Marinello, F. Decrease the Water Footprint using precision agriculture: A comparison between conventional and conservative agriculture. Geophys. Res. Abstr. 2018, 20, 769. [Google Scholar]



| Fertilizer Treatment | 1st Fertilization (at 30 DAP) | 2nd Fertilization (at 90 DAP) | ||||
|---|---|---|---|---|---|---|
| N | P | K | N | P | K | |
| (kg/ha) | (kg/ha) | |||||
| Non-vegetation/bare land (BL) | 0 | 0 | 0 | 0 | 0 | 0 |
| P and K fertilizer application without N (N-free) | 0 | 13 | 35 | 0 | 13 | 35 |
| Urea/normal released fertilizer application | 100 | 13 | 35 | 100 | 13 | 35 |
| Controlled release fertilizer (CRF) application | 200 | 25 | 69 | 0 | 0 | 0 |
| Treatment | Kh (cm/s) | Soil Depth | Air Dried MC (%) | Bulk Density (g/cm3) | Particle Size (%) | Soil Texture | |||
|---|---|---|---|---|---|---|---|---|---|
| Sand | Silt | Clay | |||||||
| Coarse | Fine | ||||||||
| BL | 0.00156 | 15 cm | 1.84 | 1.36 | 48.78 | 15.30 | 16.07 | 19.85 | Coarse Sandy Loam |
| 45 cm | 1.52 | 1.40 | 53.54 | 17.05 | 11.65 | 17.76 | Coarse Sandy Loam | ||
| N-free | 0.00032 | 15 cm | 2.45 | 1.50 | 47.37 | 13.74 | 10.31 | 28.59 | Sandy Clay Loam |
| 45 cm | 1.32 | 1.34 | 53.57 | 14.74 | 16.58 | 15.10 | Coarse Sandy Loam | ||
| Urea | 0.00023 | 15 cm | 3.41 | 1.50 | 39.80 | 13.15 | 12.77 | 34.28 | Sandy Clay Loam |
| 45 cm | 2.90 | 1.36 | 39.47 | 12.39 | 14.75 | 33.39 | Sandy Clay Loam | ||
| CRF | 0.00034 | 15 cm | 2.61 | 1.60 | 45.44 | 13.29 | 14.99 | 26.28 | Sandy Clay Loam |
| 45 cm | 2.64 | 1.55 | 45.15 | 14.82 | 9.79 | 30.24 | Sandy Clay Loam | ||
| Treatment | Soil Depth | NHC (cmolc/kg) | pH | EC (mS/m) | P (mg/L) | Ex. Al (cmolc/kg) | ||
|---|---|---|---|---|---|---|---|---|
| (KCL) | (H2O) | (Bray1) | (Bray2) | |||||
| BL | 15 cm | 0.105 | 4.32 | 5.41 | 2.65 | 4.32 | 36.80 | 0.78 |
| 45 cm | 0.172 | 4.52 | 5.89 | 2.29 | 4.52 | 33.16 | 0.29 | |
| N-free | 15 cm | 0.051 | 4.72 | 6.21 | 2.69 | 4.72 | 24.05 | 0.17 |
| 45 cm | 0.054 | 4.31 | 5.83 | 1.58 | 4.31 | 68.75 | 0.59 | |
| Urea | 15 cm | 0.005 | 4.26 | 5.18 | 2.52 | 4.26 | 7.76 | 1.42 |
| 45 cm | 0.029 | 4.68 | 5.71 | 3.91 | 4.68 | 9.86 | 0.25 | |
| CRF | 15 cm | 0.008 | 4.59 | 5.64 | 3.45 | 4.59 | 19.35 | 0.29 |
| 45 cm | 0.006 | 4.64 | 5.95 | 2.76 | 4.64 | 24.19 | 0.21 | |
| Season | Fertilizer Treatment | WFproc,green (m3/t) | WFproc,grey (m3/t) | WFproc,sugarcane (m3/t) |
|---|---|---|---|---|
| Plant cane | CRF | 175.05 | 17.28 | 192.33 |
| Urea | 188.47 | 44.99 | 233.47 | |
| Ratoon | CRF | 185.12 | 5.79 | 190.47 |
| Urea | 234.66 | 2.93 | 237.59 |
| TCsoil of Lysimeters (g/m2) | TNsoil of Lysimeters (g/m2) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| BL | N-Free | Urea | CRF | BL | N-Free | Urea | CRF | ||
| First season: plant cane | 239 DAP | 2642 | 2682 | 2453 | 2390 | 252 | 239 | 221 | 207 |
| 329 DAP | 2498 | 2408 | 2268 | 2466 | 257 | 252 | 239 | 252 | |
| Second season: ratoon | 66 DAP | 2745 | 2088 | 2151 | 2426 | 252 | 171 | 194 | 230 |
| 127 DAP | 2318 | 1953 | 2381 | 2484 | 234 | 194 | 243 | 239 | |
| 184 DAP | 2313 | 2246 | 2516 | 2394 | 243 | 230 | 252 | 225 | |
| 253 DAP | 2250 | 2075 | 2412 | 2390 | 248 | 203 | 257 | 243 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rathnappriya, R.H.K.; Sakai, K.; Okamoto, K.; Kimura, S.; Haraguchi, T.; Nakandakari, T.; Setouchi, H.; Bandara, W.B.M.A.C. Examination of the Effectiveness of Controlled Release Fertilizer to Balance Sugarcane Yield and Reduce Nitrate Leaching to Groundwater. Agronomy 2022, 12, 695. https://doi.org/10.3390/agronomy12030695
Rathnappriya RHK, Sakai K, Okamoto K, Kimura S, Haraguchi T, Nakandakari T, Setouchi H, Bandara WBMAC. Examination of the Effectiveness of Controlled Release Fertilizer to Balance Sugarcane Yield and Reduce Nitrate Leaching to Groundwater. Agronomy. 2022; 12(3):695. https://doi.org/10.3390/agronomy12030695
Chicago/Turabian StyleRathnappriya, R. H. K., Kazuhito Sakai, Ken Okamoto, Sho Kimura, Tomokazu Haraguchi, Tamotsu Nakandakari, Hideki Setouchi, and W. B. M. A. C. Bandara. 2022. "Examination of the Effectiveness of Controlled Release Fertilizer to Balance Sugarcane Yield and Reduce Nitrate Leaching to Groundwater" Agronomy 12, no. 3: 695. https://doi.org/10.3390/agronomy12030695
APA StyleRathnappriya, R. H. K., Sakai, K., Okamoto, K., Kimura, S., Haraguchi, T., Nakandakari, T., Setouchi, H., & Bandara, W. B. M. A. C. (2022). Examination of the Effectiveness of Controlled Release Fertilizer to Balance Sugarcane Yield and Reduce Nitrate Leaching to Groundwater. Agronomy, 12(3), 695. https://doi.org/10.3390/agronomy12030695








