Identification and Characterisation of Stripe Rust Resistance Genes Yr66 and Yr67 in Wheat Cultivar VL Gehun 892
Abstract
:1. Introduction
2. Materials and Methods
2.1. Greenhouse Testing
2.2. DNA Extraction
2.3. Bulked Segregant Analysis (BSA)
2.4. Genetic Analysis and Linkage Mapping
3. Results
3.1. Inheritance of Stripe Rust Resistance
3.2. Molecular Mapping
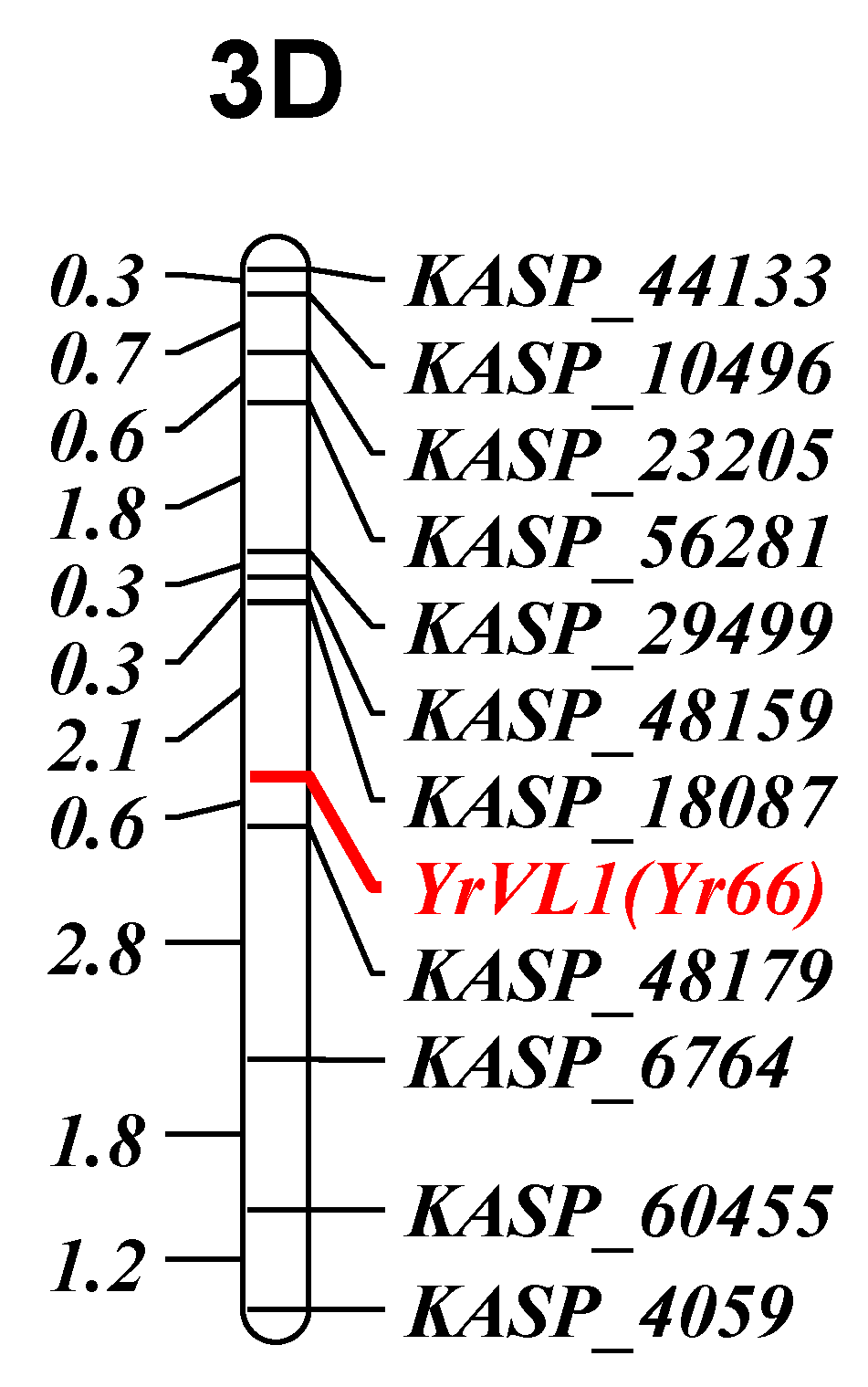
3.2.1. YrVL1
3.2.2. YrVL2
3.2.3. Polymorphism of YrVL1- and YrVL2-Linked Markers
3.3. Effectiveness of YrVL1 and YrVL2 against Different Pst Pathotypes
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dean, R.; Van Kan, J.A.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The top 10 fungal pathogens in molecular plant pathology. Mol. Plant. Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Murray, G.M.; Brennan, J.P. Estimating disease losses to the Australian wheat industry. Australas. Plant Pathol. 2009, 38, 558–570. [Google Scholar] [CrossRef]
- Bariana, H.S. Breeding for disease resistance. In Encyclopedia of Applied Plant Sciences; Thomas, B., Murphy, D.J., Murray, B.G., Eds.; Harcourt, Academic Press: London, UK, 2003; pp. 244–253. [Google Scholar]
- Oliver, R.P. A reassessment of the risk of rust fungi developing resistance to fungicides. Pest Manag. Sci. 2014, 70, 1641–1645. [Google Scholar] [CrossRef] [PubMed]
- Bariana, H.S.; Bansal, U.K. Breeding for disease resistance. In Encyclopedia of Applied Plant Sciences, 2nd ed.; Murray, B.G., Murphy, D.J., Eds.; Academic Press: Oxford, UK, 2017; pp. 69–76. [Google Scholar]
- Chen, X. Epidemiology and control of stripe rust Puccinia striiformis f. sp. tritici on wheat. Can. J. Plant Pathol. 2005, 27, 314–337. [Google Scholar] [CrossRef]
- Ellis, J.G.; Lagudah, E.S.; Spielmeyer, W.; Dodds, P.N. The past, present and future of breeding rust resistant wheat. Front. Plant. Sci. 2014, 5, 641. [Google Scholar] [CrossRef] [Green Version]
- Kolmer, J. Genetics of resistance to wheat leaf rust 1. Annu. Rev. Phytopathol. 1996, 34, 435–455. [Google Scholar] [CrossRef]
- Bariana, H.; Parry, N.; Barclay, I.R.; Loughman, R.; McLean, R.J.; Shankar, M.; Wilson, R.E.; Willey, N.J.; Francki, M. Identification and characterization of stripe rust resistance gene Yr34 in common wheat. Theor. Appl. Genet. 2005, 112, 1143–1148. [Google Scholar] [CrossRef]
- Sucher, J.; Boni, R.; Yang, P.; Rogowsky, P.; Büchner, H.; Kastner, C.; Kumlehn, J.; Krattinger, S.G.; Keller, B. The durable wheat disease resistance gene Lr34 confers common rust and northern corn leaf blight resistance in maize. Plant Biotechnol. J. 2017, 15, 489–496. [Google Scholar] [CrossRef]
- Bariana, H.S.; McIntosh, R.A. Cytogenetic studies in wheat. XV. Location of rustresistance genes in VPM1 and their genetic linkage with other disease resistance genes in chromosome 2A. Genome 1993, 36, 476–482. [Google Scholar] [CrossRef]
- Bansal, U.; Bariana, H.; Wong, D.; Randhawa, M.; Wicker, T.; Hayden, M.; Keller, B. Molecular mapping of an adult plant stem rust resistance gene Sr56 in winter wheat cultivar Arina. Theor. Appl. Genet. 2014, 127, 1441–1448. [Google Scholar] [CrossRef]
- Nsabiyera, V.; Qureshi, N.; Bariana, H.S.; Wong, D.; Forrest, K.L.; Hayden, M.J.; Bansal, U.K. Molecular markers for adult plant leaf rust resistance gene Lr48 in wheat. Mol. Breed. 2016, 36, 65. [Google Scholar] [CrossRef]
- Manly, K.F.; Cudmore, R.H., Jr.; Meer, J.M. Map Manager QTX, cross-platform software for genetic mapping. Mamm. Genome 2001, 12, 930–932. [Google Scholar] [CrossRef] [PubMed]
- Kosambi, D.D. The estimation of map distances from recombination values. Ann. Eugen. 1943, 12, 172–175. [Google Scholar] [CrossRef]
- Voorrips, R. MapChart: Software for the graphical presentation of linkage maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef] [Green Version]
- Muleta, K.T.; Bulli, P.; Rynearson, S.; Chen, X.; Pumphrey, M. Loci associated with resistance to stripe rust (Puccinia striiformis f. sp. tritici) in a core collection of spring wheat (Triticum aestivum). PLoS ONE 2017, 12, e0179087. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Wong, D.; Forrest, K.; Allen, A.; Chao, S.; Huang, B.E.; Maccaferri, M.; Salvi, S.; Milner, S.G.; Cattivelli, L. Characterization of polyploid wheat genomic diversity using a high-density 90,000 single nucleotide polymorphism array. Plant Biotechnol. J. 2014, 12, 787–796. [Google Scholar] [CrossRef] [Green Version]
- Somers, D.J.; Isaac, P.; Edwards, K. A high-density microsatellite consensus map for bread wheat (Triticum aestivum L.). Theor. Appl. Genet. 2004, 109, 1105–1114. [Google Scholar] [CrossRef]
- Li, Y.; Niu, Y.; Chen, X. Mapping a stripe rust resistance gene YrC591 in wheat variety C591 with SSR and AFLP markers. Theor. Appl. Genet. 2009, 118, 339–346. [Google Scholar] [CrossRef]
- Macer, R.C.F. The formal and monosomic genetic analysis of stripe rust (Puccinia striiformis) reistance in wheat. In Proceedings of the 2nd International Wheat Genetics Symposium, Lund, Sweden, 19–24 August 1963. [Google Scholar]
- Labrum, K.E. The location of Yr2 and Yr6 genes conferring resistance to yellow rust. In Proceedings of the Fifth European and Mediterranean Cereal Rusts Conference, Bari and Rome, Italy, 28 May–4 June 1980. [Google Scholar]
- Lin, F.; Chen, X.M. Genetics and molecular mapping of genes for race-specific all-stage resistance and non-race-specific high-temperature adult-plant resistance to stripe rust in spring wheat cultivar Alpowa. Theor. Appl. Genet. 2007, 114, 1277–1287. [Google Scholar] [CrossRef]
- Ren, R.S.; Wang, M.N.; Chen, X.M.; Zhang, Z.J. Characterization and molecular mapping of Yr52 for high-temperature adult-plant resistance to stripe rust in spring wheat germplasm PI 183527. Theor. Appl. Genet. 2012, 125, 847–857. [Google Scholar] [CrossRef]
- Zhou, X.L.; Wang, M.N.; Chen, X.M.; Lu, Y.; Kang, Z.S.; Jing, J.X. Identification of Yr59 conferring high-temperature adult-plant resistance to stripe rust in wheat germplasm PI 178759. Theor. Appl. Genet. 2014, 127, 935–945. [Google Scholar] [CrossRef] [PubMed]
- Randhawa, M.; Bansal, U.; Valarik, M.; Klocova, B.; Dolezel, J.; Bariana, H. Molecular mapping of stripe rust resistance gene Yr51 in chromosome 4AL of wheat. Theor. Appl. Genet. 2014, 127, 317–324. [Google Scholar] [CrossRef]
- Randhawa, M.; Bariana, H.; Mago, R.; Bansal, U. Mapping of a new stripe rust resistance locus Yr57 on chromosome 3BS of wheat. Mol. Breed. 2015, 35, 65. [Google Scholar] [CrossRef]
- Gessese, M.; Bariana, H.; Wong, D.; Hayden, M.; Bansal, U. Molecular mapping of stripe rust resistance gene Yr81 in a common wheat landrace Aus27430. Plant Dis. 2019, 103, 1166–1171. [Google Scholar] [CrossRef] [PubMed]
- Pakeerathan, K.; Bariana, H.; Qureshi, N.; Wong, D.; Hayden, M.; Bansal, U. Dentification of a new source of stripe rust resistance Yr82 in wheat. Theor. Appl. Genet. 2019, 132, 3169–3176. [Google Scholar] [CrossRef] [PubMed]
- Lagudah, E.S.; McFadden, H.; Singh, R.P.; Huerta-Espino, J.; Bariana, H.S.; Spielmeyer, W. Molecular genetic characterization of the Lr34/Yr18 slow rusting resistance gene region in wheat. Theor. Appl. Genet. 2006, 114, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Uauy, C.; Brevis, J.C.; Chen, X.; Khan, I.; Jackson, L.; Chicaiza, O.; Distelfeld, A.; Fahima, T.; Dubcovsky, J. High-temperature adult-plant (HTAP) stripe rust resistance gene Yr36 from Triticum turgidum ssp. dicoccoides is closely linked to the grain protein content locus Gpc-B1. Theor. Appl. Genet. 2005, 112, 97–105. [Google Scholar]
- Moore, J.W.; Herrera-Foessel, S.; Lan, C.; Schnippenkoetter, W.; Ayliffe, M.; Huerta-Espino, J.; Lillemo, M.; Viccars, L.; Milne, R.; Periyannan, S.; et al. A recently evolved hexose transporter variant confers resistance to multiple pathogens in wheat. Nat. Genet. 2015, 47, 1494–1498. [Google Scholar] [CrossRef]


| Generation | Population | Rust Response * | No of Lines | χ² (1:1) | |
|---|---|---|---|---|---|
| Observed | Expected | ||||
| RIL | VL/Wes#1 | HR | 33 | 40.0 | 1.225 |
| HS | 47 | 40.0 | 1.225 | ||
| Total | 80 | 80.0 | 2.450 | ||
| VL/Wes#4 | HR | 38 | 38.5 | 0.006 | |
| HS | 39 | 38.5 | 0.006 | ||
| Total | 77 | 77.0 | 0.013 | ||
| Marker Name | Physical Position (bp) | Primer Sequence | ||
|---|---|---|---|---|
| Allele Specific Primer A1 a | Allele Specific Primer A2 b | Common Primer | ||
| YrVL1 | ||||
| KASP_44133 | 2,242,884 | agaagaaagggtgccgccatga | gaagaaagggtgccgccatgc | ggcagccgatcgactacatgttatt |
| KASP_10496 | 2,400,568 | agtggaggtcgaagcatgcagt | gtggaggtcgaagcatgcagc | gctgcccaggctggctcgta |
| KASP_23205 | 2,593,371 | gacagcattgtCgataaacact | gacagcattgtCgataaacacg | ggtgccaccacaagttctta |
| KASP_56281 | 2,626,318 | catcctcaaacttggtattatcagaagta | cctcaaacttggtattatcagaagtg | tcataaaggcactctacaacttgcttgat |
| KASP_29499 | 2,981,592 | ccggaagaccgctgaccca | cggaagaccgctgacccg | ggttaatgttgccacttccatagtcaaa |
| KASP_48159 | 3,237,190 | ccctgttcaagccagaggt | ccctgttcaagccagaggc | acctgcttgaaagagcggc |
| KASP_18087 | 3,549,840 | aaagaaggcctcagtttgacgatatt | aagaaggcctcagtttgacgatatc | ggtcattgtcaagcgcttccttgta |
| KASP_48179 | Not known | agtacatatccccagctgctt | agtacatatccccagctgctc | agctggacatggtgctgttt |
| KASP_6764 | Not known | gttcataataaagaggaggctggt | gttcataataaagaggaggctggc | ttcagcgggaactcgaacat |
| KASP_60455 | 3,301,402 | gttctctttgaccgccttaca | gttctctttgaccgccttacg | gCtCatctcagcgagatctca |
| KASP_4059 | 2,976,737 | ggagataccgggtaaagaaatca | ggagataccgggtaaagaaatcg | aattaccaatattttgccatgtgc |
| YrVL2 | ||||
| KASP_13220 | 712,082,664 | gaattttactgctgttattgtggtgttt | gaattttactgctgttattgtggtgttg | gccttcagcattacagcaaatgaaatgaa |
| KASP_62475 | 716,963,586 | gttgaatgtctactttcatccgcca | gaatgtctactttcatccgccg | caggataatactgtgcaaaccagcctt |
| KASP_37096 | 716,966,240 | gagcgccaacgcttggttcca | gcgccaacgcttggttccc | cttgacaagacacagtagggtatacatat |
| KASP_2239 | 721,207,696 | ctctagggtggcgaggca | ctctagggtggcgaggcg | catacagctaaccgggcgag |
| KASP_71995 | 721,207,752 | catggtggcgctctgatccgt | ggtggcgctctgatccgc | catacagtctgcttattaacatacagctaa |
| KASP_61786 | 750,083,910 | cgcagttttgacacctcgt | cgcagttttgacacctcgc | caattcggagttgtagaattcttca |
| Cultivar/Stock. | KASP_2239 | KASP_18087 | KASP_48179 |
|---|---|---|---|
| YrVL1 (Yr66) | - | C:C | T:T |
| Yrvl1 (yr66) | - | T:T | C:C |
| YrVL2 (Yr67) | G:G | - | - |
| Yrvl2 (yr67) | A:A | ||
| - | - | ||
| Correll, Espada, Cobra, AGT Katana, Baxter, Calingiri, Carnamah, Catalina, Chara, Chief CL Plus, Coolah, Corack, Crusader, Dart, Derrimut, DS Faraday, EGA Bonnie Rock, EGA Burke, EGA Gregory, EGA Wedgetail, EGA Wylie, Emu Rock, Fortune, Gauntlet, Gazelle, GBA Sapphire, Giles, Gladius, Grenade CL Plus, Impala, Impose CL Plus, Janz, Justica CL Plus, Lancer, Lang, Livingston, LRPB Arrow, LRPB Flanker, LRPB Kittyhawk, LRPB Reliant, Magenta, Mansfield, Merinda, Naparoo, Preston, SF Scenario, Shield, Strzelecki, Sunco, Sunguard, Sunmax, Sunvale, Trojan, Waagan, Wallup, Westonia, Wyalkatchem, Wylah, Yandanooka | A:A | T:T | C:C |
| Axe, EGA Bounty, Lincoln, Sentinel, SF Adagio, Spitfire, Suntop, Sunzell, Ventura, Beaufort | G:G | T:T | C:C |
| Kunjin, Merlin | G:G | C:C | C:C |
| Elmore CL PLus, King Rock, Kord CL Plus, Mace, Mackellar, Ninja, Young | A:A | C:C | C:C |
| Orion, Phantom, SQP Revenue, | A:A | C:C | T:T |
| Bolac, Envoy, Forrest, Harper, Scout, Wedin, Yitpi | A:A | T:T | T:T |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bariana, H.; Kant, L.; Qureshi, N.; Forrest, K.; Miah, H.; Bansal, U. Identification and Characterisation of Stripe Rust Resistance Genes Yr66 and Yr67 in Wheat Cultivar VL Gehun 892. Agronomy 2022, 12, 318. https://doi.org/10.3390/agronomy12020318
Bariana H, Kant L, Qureshi N, Forrest K, Miah H, Bansal U. Identification and Characterisation of Stripe Rust Resistance Genes Yr66 and Yr67 in Wheat Cultivar VL Gehun 892. Agronomy. 2022; 12(2):318. https://doi.org/10.3390/agronomy12020318
Chicago/Turabian StyleBariana, Harbans, Lakshmi Kant, Naeela Qureshi, Kerrie Forrest, Hanif Miah, and Urmil Bansal. 2022. "Identification and Characterisation of Stripe Rust Resistance Genes Yr66 and Yr67 in Wheat Cultivar VL Gehun 892" Agronomy 12, no. 2: 318. https://doi.org/10.3390/agronomy12020318
APA StyleBariana, H., Kant, L., Qureshi, N., Forrest, K., Miah, H., & Bansal, U. (2022). Identification and Characterisation of Stripe Rust Resistance Genes Yr66 and Yr67 in Wheat Cultivar VL Gehun 892. Agronomy, 12(2), 318. https://doi.org/10.3390/agronomy12020318







