Degradation of Doxycycline, Enrofloxacin, and Sulfamethoxypyridazine under Simulated Sunlight at Different pH Values and Chemical Environments
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemical Reagents and Quantification of the Three Antibiotics
2.2. Photodegradation Experiments
2.3. Modeling
3. Results and Discussion
3.1. Effect of pH on the Photodegradation of the Three Antibiotics
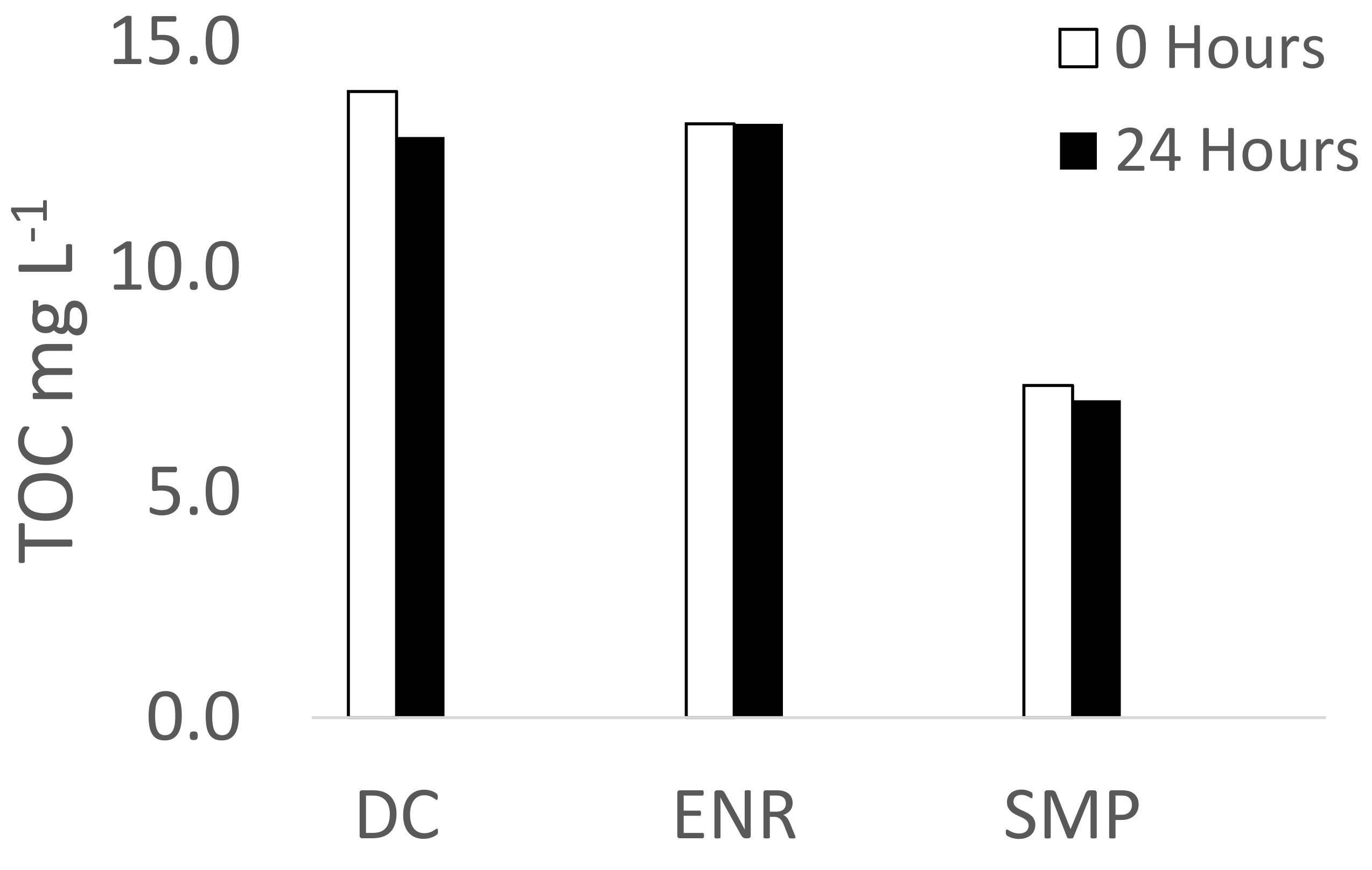
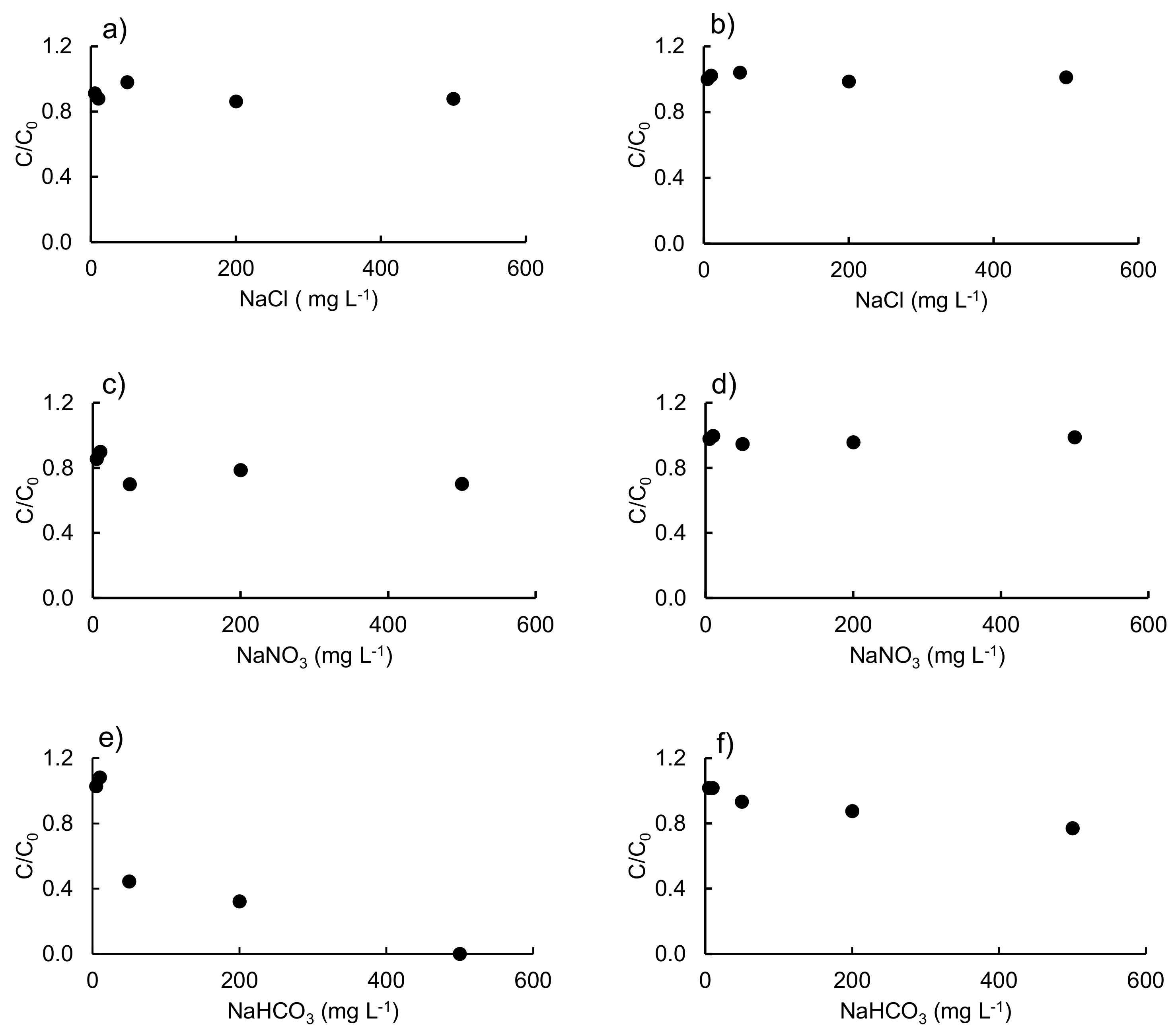
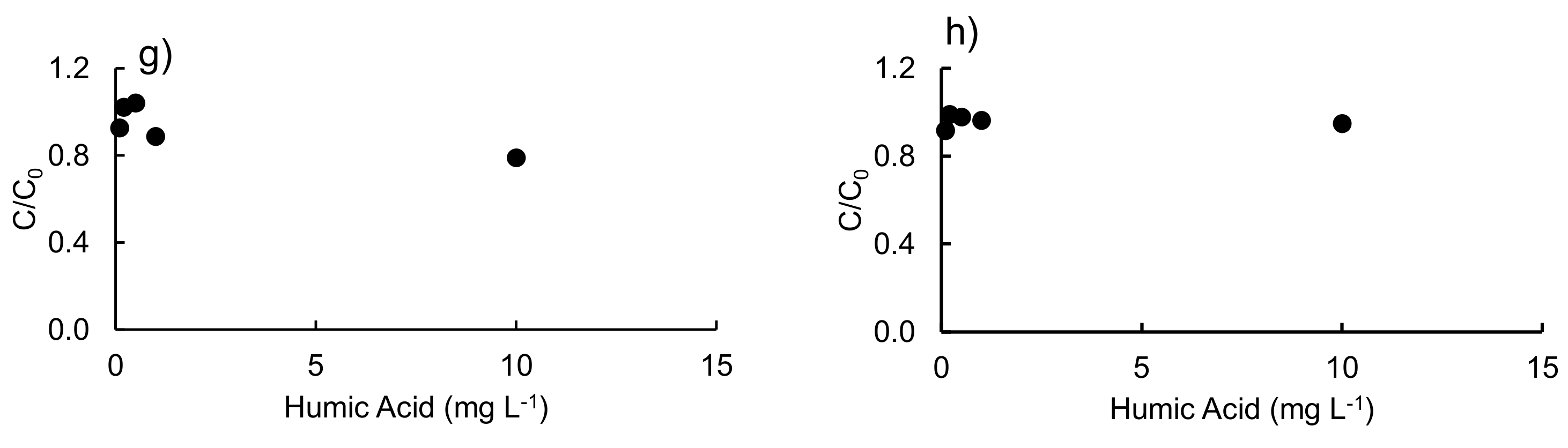
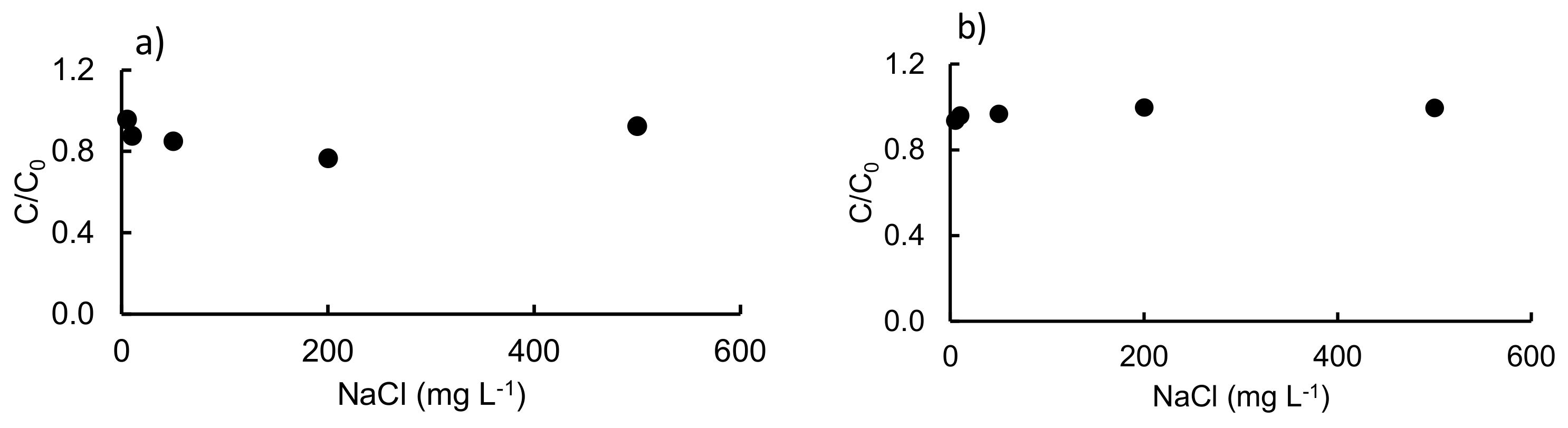
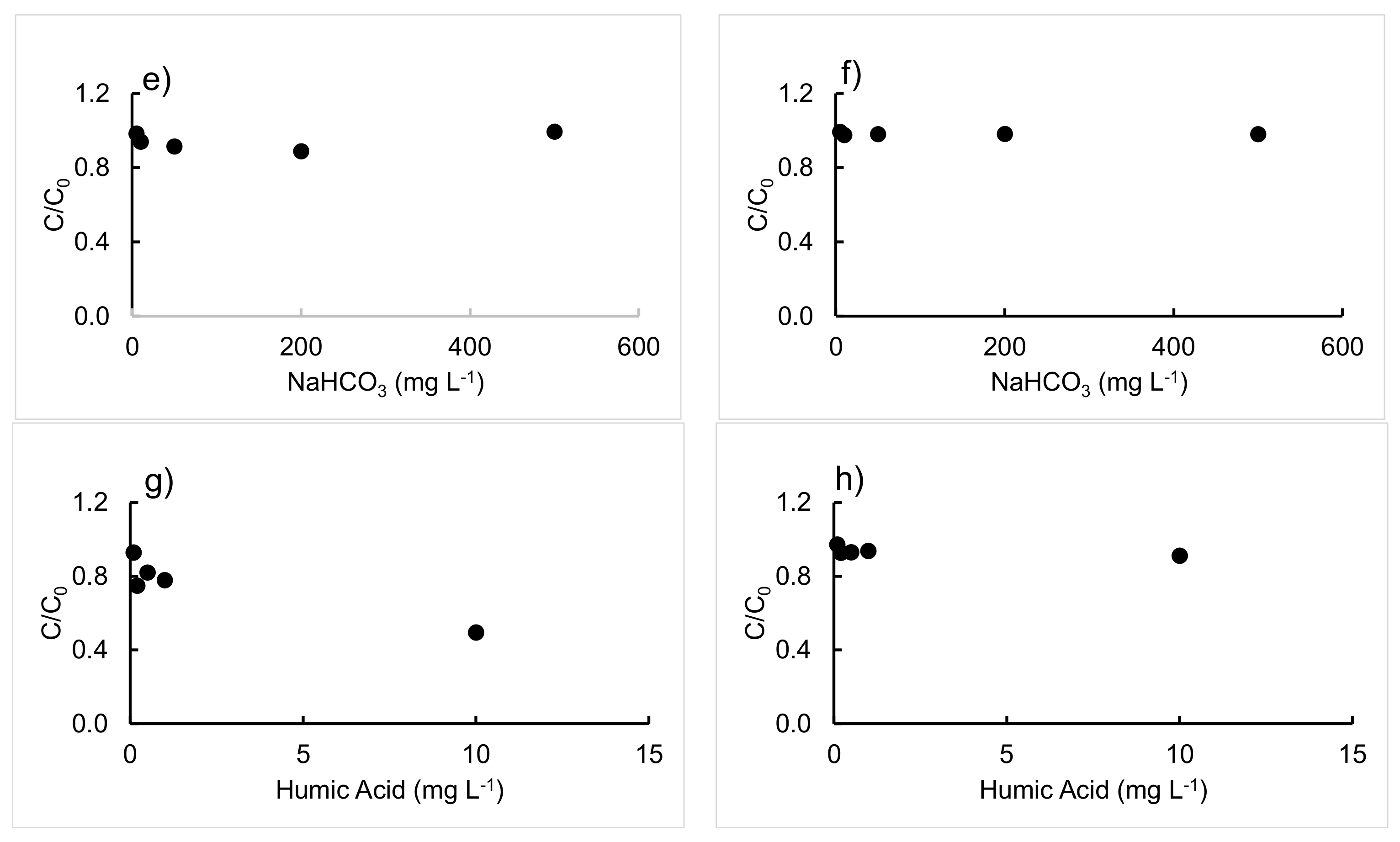
3.2. Effect of Different Salts and Humic Acids on the Photodegradation of the Three Antibiotics
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kümmerer, K. Antibiotics in the aquatic environment—A review—Part I. Chemosphere 2009, 75, 417–434. [Google Scholar] [CrossRef] [PubMed]
- Lyu, J.; Yang, L.; Zhang, L.; Ye, B.; Wang, L. Antibiotics in soil and water in China–A systematic review and source analysis. Environ. Pollut. 2020, 266, 115147. [Google Scholar] [CrossRef]
- Iglesias, A.; Nebot, C.; Miranda, J.M.; Vázquez, B.I.; Cepeda, A. Detection and quantitative analysis of 21 veterinary drugs in river water using high-pressure liquid chromatography tandem mass spectrometry. Environ. Sci. Pollut. Res. 2012, 19, 3235–3249. [Google Scholar] [CrossRef]
- Bu, Q.W.; Wang, B.; Huang, J.; Deng, S.B.; Yu, G. Pharmaceuticals and personal care products in the aquatic environment in China: A review. J. Hazard. Mater. 2013, 262, 189–211. [Google Scholar] [CrossRef]
- Isidori, M.; Lavorgna, M.; Nardelli, A.; Pascarella, A. Toxic and genotoxic evaluation of six antibiotics on non-targed organims. Sci. Total Environ. 2005, 346, 87–98. [Google Scholar] [CrossRef] [PubMed]
- Komijani, M.; Shamabadi, N.S.; Shahin, K.; Eghbalpour, F.; Tahsili, M.R.; Bahram, M. Heavy metal pollution promotes antibiotic resistance potential in the aquatic environment. Environ. Pollut. 2021, 274, 116569. [Google Scholar] [CrossRef]
- Samuelsen, O.B. Degradation of oxytetracycline in seawater at two different temperatures and light intensities, and the persistence of oxytetracycline in the sediment from a fish farm. Aquaculture 1989, 83, 7–16. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Fernández-Calviño, D.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Díaz-Raviña, M.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A.; Álvarez-Rodríguez, E. Biotic and abiotic dissipation of tetracyclines using simulated sunlight and in the dark. Sci. Total Environ. 2018, 635, 1520–1529. [Google Scholar] [CrossRef] [PubMed]
- Sanni, S.O.; Viljoen, E.L.; Ofomaja, A.E. Design of ordered Ag/AgBr nanostructures coupled activated carbon with enhanced charge carriers separation efficiency for photodegradation of tetracycline under visible light. J. Mol. Liq. 2020, 299, 112032. [Google Scholar] [CrossRef]
- Batchu, S.R.; Panditi, V.R.; Gardinali, P.R. Photodegradation of sulphonamide antibiotics in simulated and natural sunlight: Implications for their environmental fate. J. Environ. Sci. Health Part B 2014, 49, 200–211. [Google Scholar] [CrossRef]
- Conde-Cid, M.; Fernández-Calviño, D.; Nóvoa-Muñoz, J.C.; Arias-Estévez, M.; Díaz-Raviña, M.; Núñez-Delgado, A.; Fernández-Sanjurjo, M.J.; Álvarez-Rodríguez, E. Degradation of sulfadiazine, sulfachloropyridazine and sulfamethazine in aqueous media. J. Environ. Manag. 2018, 228, 239–248. [Google Scholar] [CrossRef]
- Dhiman, P.; Dhiman, N.; Kumar, A.; Sharma, G.; Naushad, M.; Ghfar, A.A. Solar active nano-Zn1−xMgxFe2O4 as a magnetically separable sustainable photocatalyst for degradation of sulfadiazine antibiotic. J. Mol. Liq. 2019, 294, 111574. [Google Scholar] [CrossRef]
- Fasani, E.; Rampi, M.; Albini, A. Photochemistry of some fluoroquinolones: Effect of pH and chloride ion. J. Chem. Soc. Perkin Trans. 1999, 2, 1901–1907. [Google Scholar] [CrossRef]
- Sciscenko, I.; Arques, A.; Varga, Z.; Bouchonnet, S.; Monfort, O.; Brigante, M.; Mailhot, G. Significant role of iron on the fate and photodegradation of enrofloxacin. Chemosphere 2021, 270, 129791. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Tian, J.; Gao, S.; Shao, P.; Qi, J.; Cui, F. Photodegradation of sulphonamides by g-C3N4 under visible light irradiation: Effectiveness, mechanism and pathways. Appl. Catal. B Environ. 2017, 210, 88–96. [Google Scholar] [CrossRef]
- Adamek, E.; Baran, W.; Sobczak, A. Assessment the biodegradability of selected sulfa drugs in two polluted rivers in Poland: Effects of seasonal variations, accidental contamination, turbidity and salinity. J. Hazard. Mater. 2016, 313, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Sukul, P.; Lamshöft, M.; Sebastian Zúhlke, S.; Spiteller, M. Photolysis of 14C-sulfadiazine in water and manure. Chemosphere 2008, 71, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Biošić, M.; Mitrevski, M.; Babić, S. Environmental behavior of sulfadiazine, sulfamethazine, and their metabolites. Environ. Sci. Pollut. Res. 2017, 24, 9802–9812. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Esmorís, C.; Conde-Cid, M.; Fernández-Sanjurjo, M.J.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Arias-Estévez, M. Environmental relevance of adsorption of doxycycline, enrofloxacin, and sulfamethoxypyridazine before and after the removal of organic matter from soils. J. Environ. Manag. 2021, 287, 112354. [Google Scholar] [CrossRef]
- Arias, M.; Barral, M.T.; Díaz-Fierros, F. Effects of associations between humic acids and iron or aluminium on the flocculation and aggregation of kaolin and quartz. Eur. J. Soil Sci. 1996, 41, 335–343. [Google Scholar] [CrossRef]
- Jiao, S.; Zheng, S.; Yin, D.; Wang, L.; Chen, L. Aqueous oxytetracycline degradation and the toxicity change of degradation compounds in photoirradiation process. J. Environ. Sci. 2008, 20, 806–813. [Google Scholar] [CrossRef]
- Zhao, C.; Pelaez, M.; Duan, X.; Deng, H.; O’Shea, K.; Fatta-kassinos, D.; Dionysiou, D.D. Role of pH on photolytic and photocatalytic degradation of antibiotic oxytetracycline in aqueous solution under visible/solar light. Kinetics and mechanism studies. Appl. Catal. B Environ. 2013, 134–135, 83–92. [Google Scholar] [CrossRef]
- Prabhakaran, D.; Sukul, P.; Lamshöft, M.; Maheswari, M.A.; Zühlke, S.; Spiteller, M. Photolysis of difloxacin and saafloxacin in aqueous systems. Chemosphere 2009, 77, 739–746. [Google Scholar] [CrossRef] [PubMed]
- Bian, X.; Zhang, J. Photodegradation of sulfadiazine in aqueous solution and the affeting factors. J. Chem. 2016, 2016, 1–5. [Google Scholar] [CrossRef] [Green Version]
- López-Peñalver, J.J.; Sánchez-Polo, M.; Gómez-Pacheco, C.V.; Rivera-Utrilla, J. Photodegradation of tetracyclines in aqueous solution by using UV and UV/H2O2 oxidation processes. J. Chem. Technol. Biotechnol. 2010, 85, 1325–1333. [Google Scholar] [CrossRef]
- Rodríguez-López, L.; Cela-Dablanca, R.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Fernández-Calviño, D.; Arias-Estévez, M. Photodegradation of Ciprofloxacin, Clarithromycin and Trimethoprin: Influence of pH and Humic acids. Molecules 2021, 26, 3080. [Google Scholar] [CrossRef]
- Chen, Y.; Hu, C.; Qu, J.; Yang, M. Photodegradation of tetracycline and formation of reactive oxygen species in aqueous tetracycline solution under simulated sunlight irradiation. J. Photochem. Photobiol. A Chem. 2008, 197, 81–87. [Google Scholar] [CrossRef]
- Boreen, A.L.; Arnold, W.A.; McNeill, K. Triplet-sensitized photodegradation of sulfa drugs containing six-membered heterocyclic groups: Identification of an SO2 extrusion photoproduct. Environ. Sci. Technol. 2005, 39, 3630–3638. [Google Scholar] [CrossRef]
- Bahnmüller, S.; von Gunten, U.; Canonica, S. Sunlight-induced transformation of sulfadiazine and sulfamethoxazole in surface waters and wastewater effluents. Water Res. 2014, 57, 183–192. [Google Scholar] [CrossRef]
- Bahnmüller, S.; Loi, C.H.; Linge, K.L.; von Gunten, U.; Canonica, S. Degradation rates of benzotriazoles and benzothiazoles under UV-C irradiation and the advanced oxidation process UV/H2O2. Water Res. 2015, 74, 143–154. [Google Scholar] [CrossRef]
- Khan, M.A.; Musarrat, J. Tetracycline-Cu(II) photo-induced fragmentation of serum albumin. Comp. Biochem. Physiol. Part C 2002, 131, 439–446. [Google Scholar] [CrossRef]
- Liu, Y.; He, X.; Duan, X.; Fu, Y.; Dioysiou, D.D. Photochemical Degradation of oxytetracycline: Influence of pH and role of carbonate radical. Chem. Eng. J. 2015, 276, 113–121. [Google Scholar] [CrossRef]
- Seto, Y.; Ochi, M.; Onoue, S.; Yamada, S. High-throughput screening strategy for photogenotoxic potential of pharmaceutical substances using fluorescent intercalating dye. J. Pharm. Biomed. Anal. 2010, 52, 781–786. [Google Scholar] [CrossRef] [PubMed]
- Thiele-Bruhn, S. Pharmaceutical antibiotic compounds in soils—A Review. J. Plant Nutr. Soil Sci. 2003, 166, 145–167. [Google Scholar] [CrossRef]
- Xuan, R.; Arisi, L.; Wang, Q.; Yates, S.R.; Biswas, K.C. Hydrolysis and photolysis of oxytetracycline in aqueous solution. J. Environ. Sci. Health Part B 2010, 45, 73–81. [Google Scholar] [CrossRef]
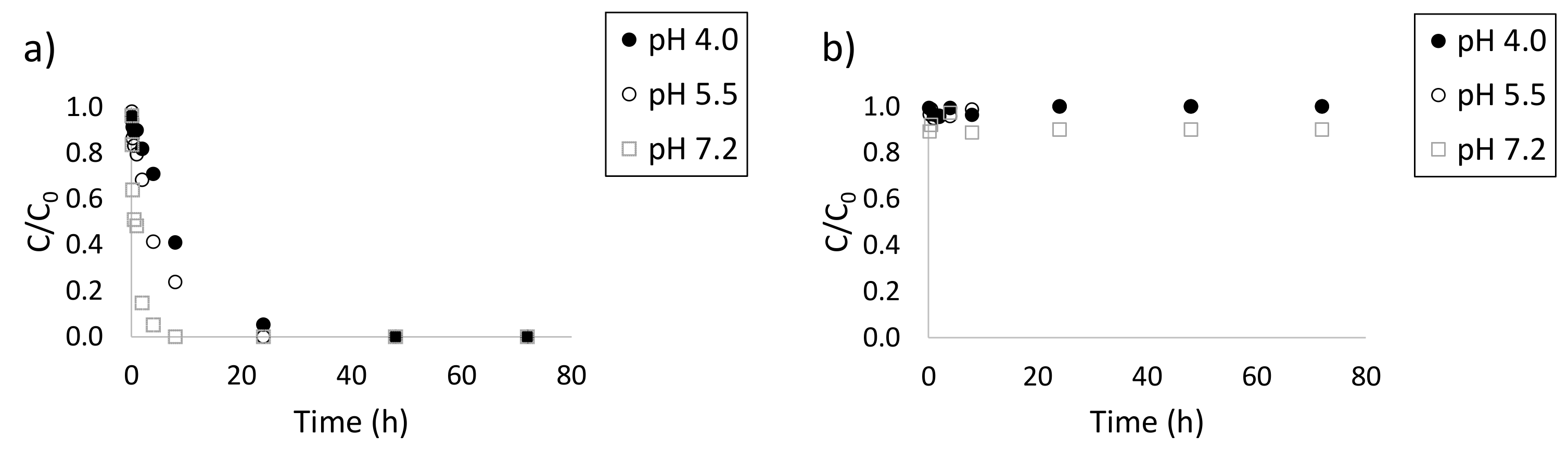

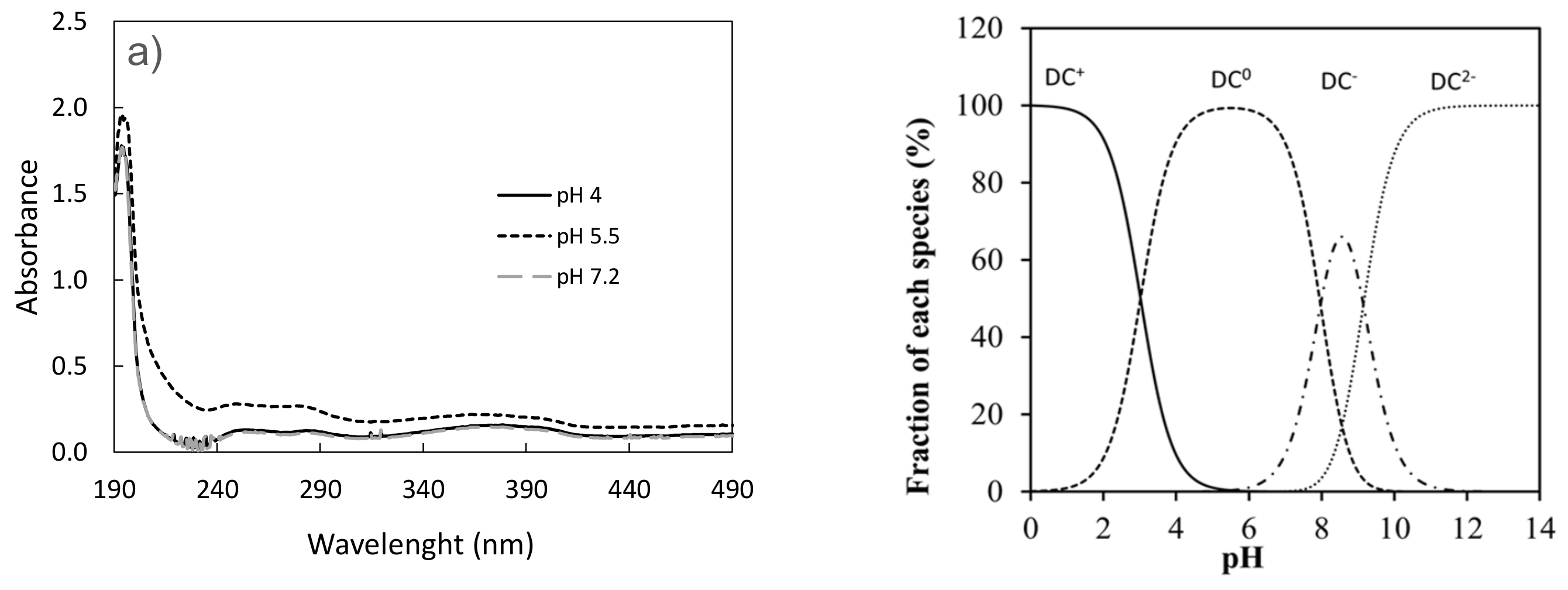
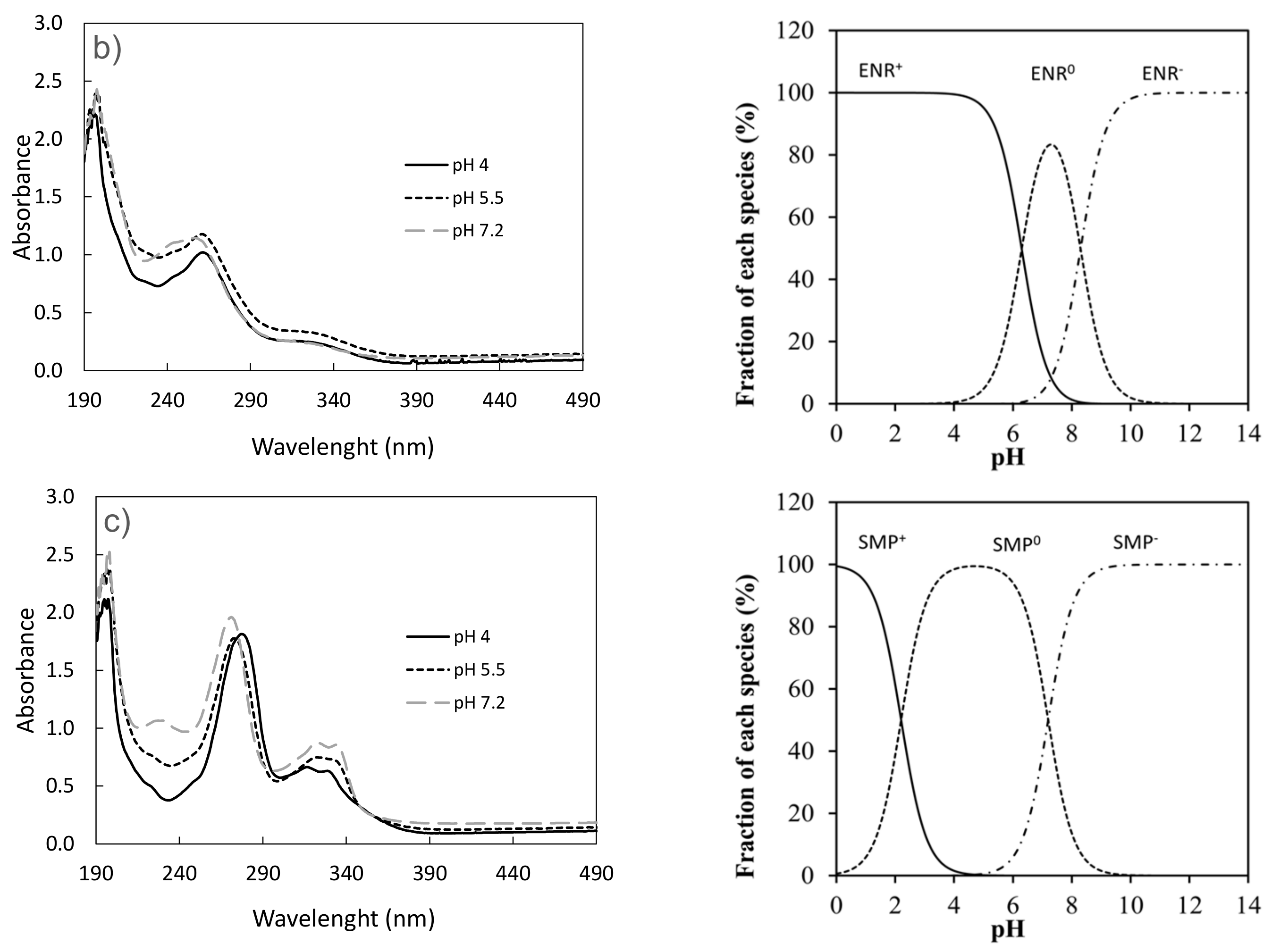
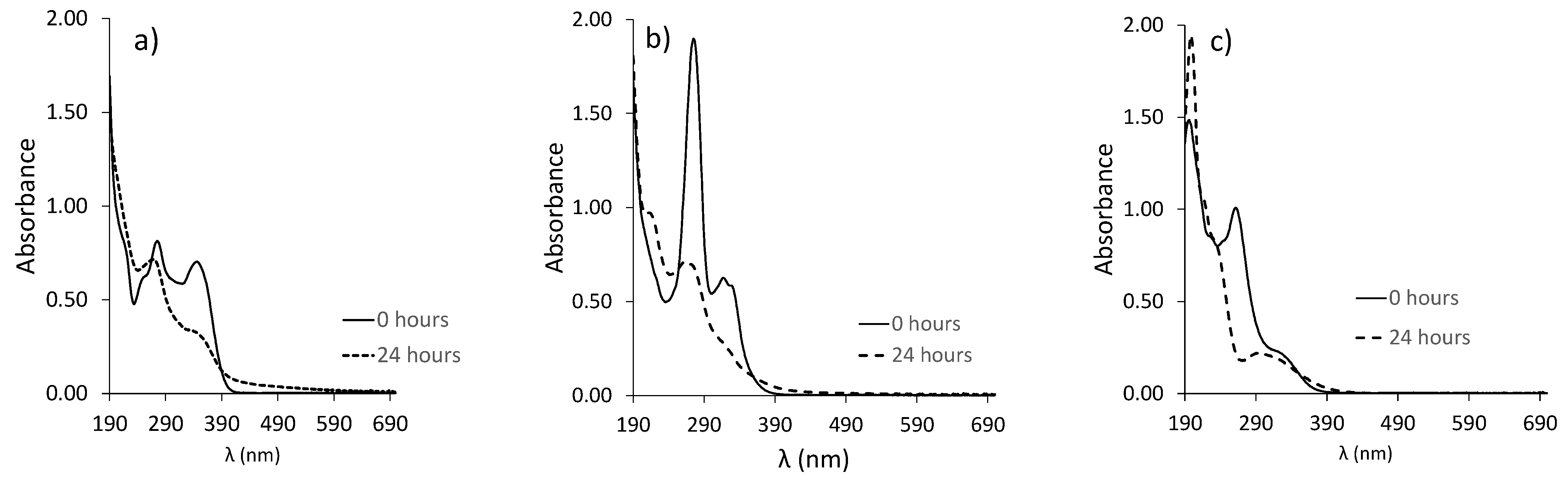


| Doxycycline | k (h−1) | t1/2 (h) | R2 |
|---|---|---|---|
| pH 4.0 | 0.106 ± 0.007 | 6.5 ± 0.4 | 0.969 |
| pH 5.5 | 0.208 ± 0.013 | 3.3 ± 0.2 | 0.984 |
| pH 7.2 | 0.988 ± 0.097 | 0.7 ± 0.1 | 0.956 |
| Enrofloxacin | |||
| pH 4.0 | 0.472 ± 0.051 | 1.5 ± 0.2 | 0.988 |
| pH 5.5 | 0.844 ± 0.107 | 0.8 ± 0.1 | 0.984 |
| pH 7.2 | 3.833 ± 0.254 | 0.2 ± 0.1 | 0.986 |
| Sulfamethoxypyridazine | |||
| pH 4.0 | 0.122 ± 0.025 | 5.7 ± 1.4 | 0.917 |
| pH 5.5 | 0.111 ± 0.012 | 6.2 ± 0.7 | 0.976 |
| pH 7.2 | 0.205 ± 0.020 | 3.4 ± 0.3 | 0.980 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Álvarez-Esmorís, C.; Rodríguez-López, L.; Fernández-Calviño, D.; Núñez-Delgado, A.; Álvarez-Rodríguez, E.; Arias-Estévez, M. Degradation of Doxycycline, Enrofloxacin, and Sulfamethoxypyridazine under Simulated Sunlight at Different pH Values and Chemical Environments. Agronomy 2022, 12, 260. https://doi.org/10.3390/agronomy12020260
Álvarez-Esmorís C, Rodríguez-López L, Fernández-Calviño D, Núñez-Delgado A, Álvarez-Rodríguez E, Arias-Estévez M. Degradation of Doxycycline, Enrofloxacin, and Sulfamethoxypyridazine under Simulated Sunlight at Different pH Values and Chemical Environments. Agronomy. 2022; 12(2):260. https://doi.org/10.3390/agronomy12020260
Chicago/Turabian StyleÁlvarez-Esmorís, Cristina, Lucia Rodríguez-López, David Fernández-Calviño, Avelino Núñez-Delgado, Esperanza Álvarez-Rodríguez, and Manuel Arias-Estévez. 2022. "Degradation of Doxycycline, Enrofloxacin, and Sulfamethoxypyridazine under Simulated Sunlight at Different pH Values and Chemical Environments" Agronomy 12, no. 2: 260. https://doi.org/10.3390/agronomy12020260
APA StyleÁlvarez-Esmorís, C., Rodríguez-López, L., Fernández-Calviño, D., Núñez-Delgado, A., Álvarez-Rodríguez, E., & Arias-Estévez, M. (2022). Degradation of Doxycycline, Enrofloxacin, and Sulfamethoxypyridazine under Simulated Sunlight at Different pH Values and Chemical Environments. Agronomy, 12(2), 260. https://doi.org/10.3390/agronomy12020260










