Vertical Farming: The Only Way Is Up?
Abstract
1. Vertical Farming: A Brief History
“A multilayer indoor plant production system in which all growth factors, such as light, temperature, humidity, carbon dioxide concentration ([CO2]), water, and nutrients, are precisely controlled to produce high quantities of high-quality fresh produce year-round, completely independent of solar light and other outdoor conditions.”
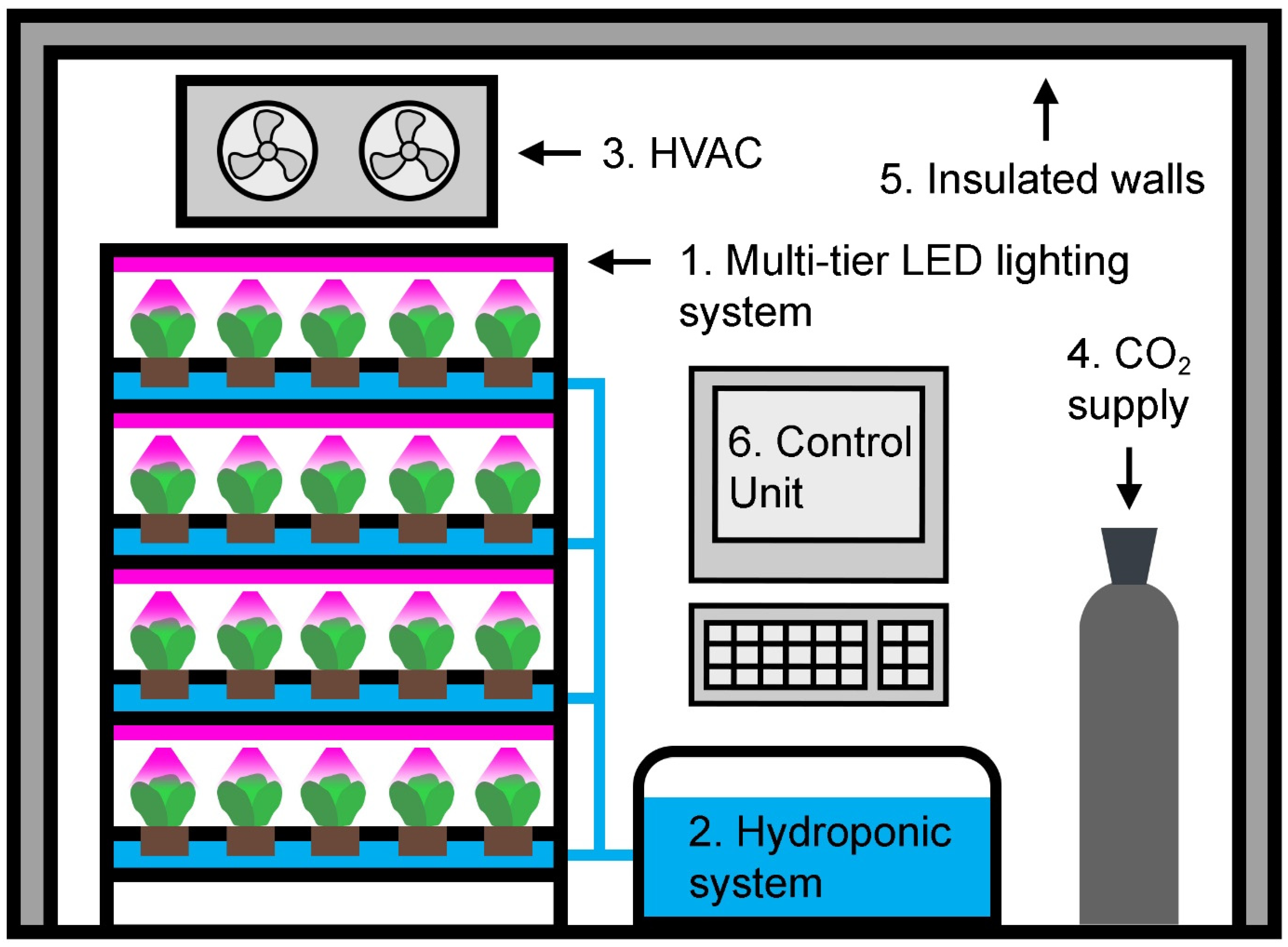
- Plant factory with artificial lighting (PFAL), an industrial-scale vertical farm located in a devoted building.
- Container farm, a modular vertical farm contained in a shipping container.
- In-store farm, a vertical farm located at the place of consumption or purchase (i.e., retail and restaurants).
- Appliance farm, a vertical farm appliance integrated into a home or office.
2. Closed-Loop Hydroponics: The Principal Circular Component of a Vertical Farm
3. Opportunities and Challenges: Is Growing Skywards a Fairytale?
3.1. Economic
3.2. Environmental
3.3. Social and Political
4. Plant-Growing Media in a Vertical Farm
5. The Vertical Farm Root-Associated Microbiome
5.1. The Role of Plant-Growing Media in Microbiome Functioning
5.2. The Plant Host: A Picky Landlord?
5.3. Plant Growth-Promoting Rhizobacteria Amendment to Improve Vertical Farm Circularity and Resilience
6. The Future Potential of Vertical Farming: The Need for Niche Expansion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Waldron, D. Evolution of Vertical Farms and the Development of a Simulation Methodology. WIT Trans. Ecol. Environ. 2018, 217, 975–986. [Google Scholar]
- SharathKumar, M.; Heuvelink, E.; Marcelis, L.F.M. Vertical Farming: Moving from Genetic to Environmental Modification. Trends Plant Sci. 2020, 25, 724–727. [Google Scholar] [CrossRef] [PubMed]
- Butturini, M.; Marcelis, L.F.M. Vertical farming in Europe. In Plant Factory; Elsevier: Amsterdam, The Netherlands, 2020; pp. 77–91. [Google Scholar]
- Al-Kodmany, K. The Vertical Farm: A Review of Developments and Implications for the Vertical City. Buildings 2018, 8, 24. [Google Scholar] [CrossRef]
- Walker, A.B. Cartoon in “Life” Magazine’s “Real Estate Number”of March. 1909. Available online: https://www.architakes.com/?p=1687 (accessed on 8 July 2021).
- Koolhaas, R. Delirious New York: A Retroactive Manifesto for Manhattan; The Monacelli Press: New York, NY, USA, 1997. [Google Scholar]
- Januszkiewicz, K.; Jarmusz, M. Envisioning Urban Farming for Food Security during the Climate Change Era. Vertical Farm within Highly Urbanized Areas. IOP Conf. Ser. Mater. Sci. Eng. 2017, 245, 052094. [Google Scholar] [CrossRef]
- Bailey, G.E. Vertical Farming; E.I. Du Pont De Nemours Powder Co.: Wilmington, DE, USA, 1915. [Google Scholar]
- Vandersteen, W. Op Het Eiland Amoras; Uitgeversmaatschappij N.V. Standaard-Boekhandel: Antwerp, Belgium, 1947. [Google Scholar]
- Noecker, N.L.; Gericke, W.F. The Complete Guide to Soilless Gardening. Am. Midl. Nat. 1940, 24, 766. [Google Scholar] [CrossRef]
- Despommier, D. Farming up the city: The rise of urban vertical farms. Trends Biotechnol. 2013, 31, 388–389. [Google Scholar] [CrossRef]
- Kozai, T.; Niu, G. Role of the plant factory with artificial lighting (PFAL) in urban areas. In Plant Factory; Elsevier: Amsterdam, The Netherlands, 2020; pp. 7–34. [Google Scholar]
- Kleszcz, J.; Kmiecik, P.; Świerzawski, J. Vegetable and Gardening Tower of Othmar Ruthner in the Voivodeship Park of Culture and Recreation in Chorzów—The First Example of Vertical Farming in Poland. Sustainability 2020, 12, 5378. [Google Scholar] [CrossRef]
- Despommier, D. The vertical farm: Controlled environment agriculture carried out in tall buildings would create greater food safety and security for large urban populations. J. Verbrauch. Leb. 2011, 6, 233–236. [Google Scholar] [CrossRef]
- Kozai, T.; Ohyama, K.; Chun, C. Commercialized closed systems with artificial lighting for plant production. Acta Hortic. 2006, 711, 61–70. [Google Scholar] [CrossRef]
- Association for Vertical Farming Urban Agriculture Integration Typology. Available online: https://vertical-farming.net/vertical-farming/integration-typology/ (accessed on 28 July 2021).
- Allegaert, S.D. The Vertical Farm Industry: Exploratory Research of a Wicked Situation; Wageningen University and Research: Wageningen, The Netherlands, 2020. [Google Scholar]
- Kozai, T.; Niu, G. Plant factory as a resource-efficient closed plant production system. In Plant Factory; Elsevier: Amsterdam, The Netherlands, 2020; pp. 93–115. [Google Scholar]
- Jones, J.B., Jr. Hydroponics: A Practical Guide for the Soilless Grower; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Son, J.E.; Kim, H.J.; Ahn, T.I. Hydroponic Systems. In Plant Factory; Elsevier: Amsterdam, The Netherlands, 2016; pp. 213–221. [Google Scholar]
- Verhagen, J.B.G.M. Stability of growing media from a physical, chemical and biological perspective. Acta Hortic. 2009, 819, 135–142. [Google Scholar] [CrossRef]
- Sambo, P.; Nicoletto, C.; Giro, A.; Pii, Y.; Valentinuzzi, F.; Mimmo, T.; Lugli, P.; Orzes, G.; Mazzetto, F.; Astolfi, S.; et al. Hydroponic Solutions for Soilless Production Systems: Issues and Opportunities in a Smart Agriculture Perspective. Front. Plant Sci. 2019, 10, 923. [Google Scholar] [CrossRef]
- De Rijck, G.; Schrevens, E. Elemental bioavailability in nutrient solutions in relation to complexation reactions. J. Plant Nutr. 1998, 21, 849–859. [Google Scholar] [CrossRef]
- Tomasi, N.; De Nobili, M.; Gottardi, S.; Zanin, L.; Mimmo, T.; Varanini, Z.; Römheld, V.; Pinton, R.; Cesco, S. Physiological and molecular characterization of Fe acquisition by tomato plants from natural Fe complexes. Biol. Fertil. Soils 2013, 49, 187–200. [Google Scholar] [CrossRef]
- Lee, J.G.; Lee, B.Y.; Lee, H.J. Accumulation of Phytotoxic Organic Acids in Reused Nutrient Solution During Hydroponic Cultivation of Lettuce (Lactuca sativa L.). Sci. Hortic. 2006, 110, 119–128. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Verheust, Y.; Bonarrigo, G.; Van Hulle, S. Closed hydroponic systems: Operational parameters, root exudates occurrence and related water treatment. Rev. Environ. Sci. Bio/Technol. 2017, 16, 59–79. [Google Scholar] [CrossRef]
- Lau, V.; Mattson, N. Effects of Hydrogen Peroxide on Organically Fertilized Hydroponic Lettuce (Lactuca sativa L.). Horticulturae 2021, 7, 106. [Google Scholar] [CrossRef]
- Benis, K.; Ferrão, P. Commercial farming within the urban built environment—Taking stock of an evolving field in northern countries. Glob. Food Sec. 2018, 17, 30–37. [Google Scholar] [CrossRef]
- Benke, K.; Tomkins, B. Future food-production systems: Vertical farming and controlled-environment agriculture. Sustain. Sci. Pract. Policy 2017, 13, 13–26. [Google Scholar] [CrossRef]
- Graamans, L.; Baeza, E.; van den Dobbelsteen, A.; Tsafaras, I.; Stanghellini, C. Plant factories versus greenhouses: Comparison of resource use efficiency. Agric. Syst. 2018, 160, 31–43. [Google Scholar] [CrossRef]
- Kikuchi, Y.; Kanematsu, Y.; Yoshikawa, N.; Okubo, T.; Takagaki, M. Environmental and resource use analysis of plant factories with energy technology options: A case study in Japan. J. Clean. Prod. 2018, 186, 703–717. [Google Scholar] [CrossRef]
- Tuomisto, H.L. Vertical Farming and Cultured Meat: Immature Technologies for Urgent Problems. One Earth 2019, 1, 275–277. [Google Scholar] [CrossRef]
- Hallikainen, E. Life Cycle Assessment on Vertical Farming; Aalto University: Aalto, Finland, 2018. [Google Scholar]
- Wildeman, R. Vertical Farming: A Future Perspective or a Mere conceptual Idea? University of Twente: Enschede, The Netherlands, 2020. [Google Scholar]
- Pinstrup-Andersen, P. Is it time to take vertical indoor farming seriously? Glob. Food Sec. 2018, 17, 233–235. [Google Scholar] [CrossRef]
- Beacham, A.M.; Vickers, L.H.; Monaghan, J.M. Vertical farming: A summary of approaches to growing skywards. J. Hortic. Sci. Biotechnol. 2019, 94, 277–283. [Google Scholar] [CrossRef]
- Pattison, P.M.; Tsao, J.Y.; Brainard, G.C.; Bugbee, B. LEDs for photons, physiology and food. Nature 2018, 563, 493–500. [Google Scholar] [CrossRef]
- Kozai, T.; Niu, G. Challenges for the Next-Generation PFALs; Elsevier Inc.: Amsterdam, The Netherlands, 2020. [Google Scholar]
- Gruda, N. Increasing Sustainability of Growing Media Constituents and Stand-Alone Substrates in Soilless Culture Systems. Agronomy 2019, 9, 298. [Google Scholar] [CrossRef]
- Bol, R.; Gruau, G.; Mellander, P.E.; Dupas, R.; Bechmann, M.; Skarbøvik, E.; Bieroza, M.; Djodjic, F.; Glendell, M.; Jordan, P.; et al. Challenges of reducing phosphorus based water eutrophication in the agricultural landscapes of Northwest Europe. Front. Mar. Sci. 2018, 5, 276. [Google Scholar] [CrossRef]
- Hilton, S.P.; Keoleian, G.A.; Daigger, G.T.; Zhou, B.; Love, N.G. Life Cycle Assessment of Urine Diversion and Conversion to Fertilizer Products at the City Scale. Environ. Sci. Technol. 2021, 55, 593–603. [Google Scholar] [CrossRef]
- Lam, L.; Kurisu, K.; Hanaki, K. Comparative environmental impacts of source-separation systems for domestic wastewater management in rural China. J. Clean. Prod. 2015, 104, 185–198. [Google Scholar] [CrossRef]
- El-Nakhel, C.; Geelen, D.; De Paepe, J.; Clauwaert, P.; De Pascale, S.; Rouphael, Y. An Appraisal of Urine Derivatives Integrated in the Nitrogen and Phosphorus Inputs of a Lettuce Soilless Cultivation System. Sustainability 2021, 13, 4218. [Google Scholar] [CrossRef]
- Magwaza, S.T.; Magwaza, L.S.; Odindo, A.O.; Mashilo, J.; Mditshwa, A.; Buckley, C. Evaluating the feasibility of human excreta-derived material for the production of hydroponically grown tomato plants—Part I: Photosynthetic efficiency, leaf gas exchange and tissue mineral content. Agric. Water Manag. 2020, 234, 106114. [Google Scholar] [CrossRef]
- Halbert-Howard, A.; Häfner, F.; Karlowsky, S.; Schwarz, D.; Krause, A. Evaluating recycling fertilizers for tomato cultivation in hydroponics, and their impact on greenhouse gas emissions. Environ. Sci. Pollut. Res. 2020, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Specht, K.; Weith, T.; Swoboda, K.; Siebert, R. Socially acceptable urban agriculture businesses. Agron. Sustain. Dev. 2016, 36, 17. [Google Scholar] [CrossRef]
- Al-Chalabi, M. Vertical farming: Skyscraper sustainability? Sustain. Cities Soc. 2015, 18, 74–77. [Google Scholar] [CrossRef]
- Liu, T.; Yang, M.; Han, Z.; Ow, D.W. Rooftop production of leafy vegetables can be profitable and less contaminated than farm-grown vegetables. Agron. Sustain. Dev. 2016, 36, 41. [Google Scholar] [CrossRef]
- Kim, H.-S.; Kim, K.-R.; Lim, G.-H.; Kim, J.-W.; Kim, K.-H. Influence of airborne dust on the metal concentrations in crop plants cultivated in a rooftop garden in Seoul. Soil Sci. Plant Nutr. 2015, 61, 88–97. [Google Scholar] [CrossRef][Green Version]
- Carlile, W.R.; Cattivello, C.; Zaccheo, P. Organic Growing Media: Constituents and Properties. Vadose Zone J. 2015, 14, vzj2014.09.0125. [Google Scholar] [CrossRef]
- Alexander, P.D.; Bragg, N.C.; Meade, R.; Padelopoulos, G.; Watts, O. Peat in horticulture and conservation: The UK response to a changing world. Mires Peat 2008, 3, 1–10. [Google Scholar]
- Gruda, N. Current and future perspective of growing media in europe. Acta Hortic. 2012, 960, 37–43. [Google Scholar] [CrossRef]
- Blok, C.; Eveleens, B.; van Winkel, A. Growing media for food and quality of life in the period 2020–2050. Acta Hortic. 2021, 1305, 341–356. [Google Scholar] [CrossRef]
- Gruda, N.; Qaryouti, M.M.; Leonardi, C. Growing media. In Good Agricultural Practices for Greenhouse Vegetable Crops; FAO: Rome, Italy, 2013; pp. 271–301. [Google Scholar]
- Carlile, B.; Coules, A. Towards sustainability in growing media. Acta Hortic. 2013, 1013, 341–349. [Google Scholar] [CrossRef]
- Barrett, G.E.; Alexander, P.D.; Robinson, J.S.; Bragg, N.C. Achieving environmentally sustainable growing media for soilless plant cultivation systems—A review. Sci. Hortic. 2016, 212, 220–234. [Google Scholar] [CrossRef]
- Schmilewski, G. The role of peat in assuring the quality of growing media. Mires Peat 2008, 3, 1–8. [Google Scholar]
- Atzori, G.; Pane, C.; Zaccardelli, M.; Cacini, S.; Massa, D. The Role of Peat-Free Organic Substrates in the Sustainable Management of Soilless Cultivations. Agronomy 2021, 11, 1236. [Google Scholar] [CrossRef]
- Grunert, O.; Reheul, D.; Van Labeke, M.-C.; Perneel, M.; Hernandez-Sanabria, E.; Vlaeminck, S.E.; Boon, N. Growing media constituents determine the microbial nitrogen conversions in organic growing media for horticulture. Microb. Biotechnol. 2016, 9, 389–399. [Google Scholar] [CrossRef]
- Van Gerrewey, T.; Ameloot, N.; Navarrete, O.; Vandecruys, M.; Perneel, M.; Boon, N.; Geelen, D. Microbial activity in peat-reduced plant growing media: Identifying influential growing medium constituents and physicochemical properties using fractional factorial design of experiments. J. Clean. Prod. 2020, 256, 120323. [Google Scholar] [CrossRef]
- Grunert, O.; Hernandez-Sanabria, E.; Vilchez-Vargas, R.; Jauregui, R.; Pieper, D.H.; Perneel, M.; Van Labeke, M.-C.; Reheul, D.; Boon, N. Mineral and organic growing media have distinct community structure, stability and functionality in soilless culture systems. Sci. Rep. 2016, 6, 18837. [Google Scholar] [CrossRef] [PubMed]
- Montagne, V.; Capiaux, H.; Barret, M.; Cannavo, P.; Charpentier, S.; Grosbellet, C.; Lebeau, T. Bacterial and fungal communities vary with the type of organic substrate: Implications for biocontrol of soilless crops. Environ. Chem. Lett. 2017, 15, 537–545. [Google Scholar] [CrossRef]
- Regeling Handelspotgronden RHP the European Knowledge Centre for Growing Media Since 1963. Available online: https://www.rhp.nl/ (accessed on 9 August 2021).
- Carlile, W.R.; Wilson, D.P. Microbial activity in growing media—A brief review. Acta Hortic. 1991, 294, 197–206. [Google Scholar] [CrossRef]
- Pot, S.; De Tender, C.; Ommeslag, S.; Delcour, I.; Ceusters, J.; Gorrens, E.; Debode, J.; Vandecasteele, B.; Vancampenhout, K. Understanding the Shift in the Microbiome of Composts That Are Optimized for a Better Fit-for-Purpose in Growing Media. Front. Microbiol. 2021, 12, 757. [Google Scholar] [CrossRef]
- de la Fuente Cantó, C.; Simonin, M.; King, E.; Moulin, L.; Bennett, M.J.; Castrillo, G.; Laplaze, L. An extended root phenotype: The rhizosphere, its formation and impacts on plant fitness. Plant J. 2020, 103, 951–964. [Google Scholar] [CrossRef] [PubMed]
- Hacquard, S.; Garrido-Oter, R.; González, A.; Spaepen, S.; Ackermann, G.; Lebeis, S.; McHardy, A.C.; Dangl, J.L.; Knight, R.; Ley, R.; et al. Microbiota and Host Nutrition across Plant and Animal Kingdoms. Cell Host Microbe 2015, 17, 603–616. [Google Scholar] [CrossRef]
- York, L.M.; Carminati, A.; Mooney, S.J.; Ritz, K.; Bennett, M.J. The holistic rhizosphere: Integrating zones, processes, and semantics in the soil influenced by roots. J. Exp. Bot. 2016, 67, 3629–3643. [Google Scholar] [CrossRef]
- Vandenkoornhuyse, P.; Quaiser, A.; Duhamel, M.; Le Van, A.; Dufresne, A. The importance of the microbiome of the plant holobiont. New Phytol. 2015, 206, 1196–1206. [Google Scholar] [CrossRef]
- Wagg, C.; Schlaeppi, K.; Banerjee, S.; Kuramae, E.E.; van der Heijden, M.G.A. Fungal-bacterial diversity and microbiome complexity predict ecosystem functioning. Nat. Commun. 2019, 10, 4841. [Google Scholar] [CrossRef] [PubMed]
- Berendsen, R.L.; Pieterse, C.M.J.; Bakker, P.A.H.M. The rhizosphere microbiome and plant health. Trends Plant Sci. 2012, 17, 478–486. [Google Scholar] [CrossRef]
- Fierer, N. Embracing the unknown: Disentangling the complexities of the soil microbiome. Nat. Rev. Microbiol. 2017, 15, 579–590. [Google Scholar] [CrossRef] [PubMed]
- Pershina, E.; Valkonen, J.; Kurki, P.; Ivanova, E.; Chirak, E.; Korvigo, I.; Provorov, N.; Andronov, E. Comparative Analysis of Prokaryotic Communities Associated with Organic and Conventional Farming Systems. PLoS ONE 2015, 10, e0145072. [Google Scholar] [CrossRef] [PubMed]
- Raynaud, X.; Nunan, N. Spatial Ecology of Bacteria at the Microscale in Soil. PLoS ONE 2014, 9, e87217. [Google Scholar] [CrossRef]
- Torsvik, V. Prokaryotic Diversity--Magnitude, Dynamics, and Controlling Factors. Science 2002, 296, 1064–1066. [Google Scholar] [CrossRef]
- Ramos, C.; Mølbak, L.; Molin, S. Bacterial Activity in the Rhizosphere Analyzed at the Single-Cell Level by Monitoring Ribosome Contents and Synthesis Rates. Appl. Environ. Microbiol. 2000, 66, 801–809. [Google Scholar] [CrossRef]
- Bulgarelli, D.; Schlaeppi, K.; Spaepen, S.; van Themaat, E.V.L.; Schulze-Lefert, P. Structure and Functions of the Bacterial Microbiota of Plants. Annu. Rev. Plant Biol. 2013, 64, 807–838. [Google Scholar] [CrossRef] [PubMed]
- Bulgarelli, D.; Rott, M.; Schlaeppi, K.; Ver Loren van Themaat, E.; Ahmadinejad, N.; Assenza, F.; Rauf, P.; Huettel, B.; Reinhardt, R.; Schmelzer, E.; et al. Revealing structure and assembly cues for Arabidopsis root-inhabiting bacterial microbiota. Nature 2012, 488, 91–95. [Google Scholar] [CrossRef]
- Bakker, P.A.H.M.; Berendsen, R.L.; Doornbos, R.F.; Wintermans, P.C.A.; Pieterse, C.M.J. The rhizosphere revisited: Root microbiomics. Front. Plant Sci. 2013, 4, 165. [Google Scholar] [CrossRef]
- Korenblum, E.; Dong, Y.; Szymanski, J.; Panda, S.; Jozwiak, A.; Massalha, H.; Meir, S.; Rogachev, I.; Aharoni, A. Rhizosphere microbiome mediates systemic root metabolite exudation by root-to-root signaling. Proc. Natl. Acad. Sci. USA 2020, 117, 3874–3883. [Google Scholar] [CrossRef]
- Luu, V.T.; Weinhold, A.; Ullah, C.; Dressel, S.; Schoettner, M.; Gase, K.; Gaquerel, E.; Xu, S.; Baldwin, I.T. O- Acyl Sugars Protect a Wild Tobacco from Both Native Fungal Pathogens and a Specialist Herbivore. Plant Physiol. 2017, 174, 370–386. [Google Scholar] [CrossRef] [PubMed]
- Bulgarelli, D.; Garrido-Oter, R.; Münch, P.C.; Weiman, A.; Dröge, J.; Pan, Y.; McHardy, A.C.; Schulze-Lefert, P. Structure and Function of the Bacterial Root Microbiota in Wild and Domesticated Barley. Cell Host Microbe 2015, 17, 392–403. [Google Scholar] [CrossRef]
- Schlaeppi, K.; Dombrowski, N.; Oter, R.G.; Ver Loren van Themaat, E.; Schulze-Lefert, P. Quantitative divergence of the bacterial root microbiota in Arabidopsis thaliana relatives. Proc. Natl. Acad. Sci. USA 2014, 111, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Terán, A.; Navarro-Díaz, M.; Benítez, M.; Lira, R.; Wegier, A.; Escalante, A.E. Host genotype explains rhizospheric microbial community composition: The case of wild cotton metapopulations (Gossypium hirsutum L.) in Mexico. FEMS Microbiol. Ecol. 2020, 96, fiaa109. [Google Scholar] [CrossRef]
- Peiffer, J.A.; Spor, A.; Koren, O.; Jin, Z.; Tringe, S.G.; Dangl, J.L.; Buckler, E.S.; Ley, R.E. Diversity and heritability of the maize rhizosphere microbiome under field conditions. Proc. Natl. Acad. Sci. USA 2013, 110, 6548–6553. [Google Scholar] [CrossRef]
- Liu, F.; Hewezi, T.; Lebeis, S.L.; Pantalone, V.; Grewal, P.S.; Staton, M.E. Soil indigenous microbiome and plant genotypes cooperatively modify soybean rhizosphere microbiome assembly. BMC Microbiol. 2019, 19, 201. [Google Scholar] [CrossRef] [PubMed]
- Schulze-Lefert, P.; Panstruga, R. A molecular evolutionary concept connecting nonhost resistance, pathogen host range, and pathogen speciation. Trends Plant Sci. 2011, 16, 117–125. [Google Scholar] [CrossRef]
- Toju, H.; Peay, K.G.; Yamamichi, M.; Narisawa, K.; Hiruma, K.; Naito, K.; Fukuda, S.; Ushio, M.; Nakaoka, S.; Onoda, Y.; et al. Core microbiomes for sustainable agroecosystems. Nat. Plants 2018, 4, 247–257. [Google Scholar] [CrossRef]
- De Zutter, N.; Ameye, M.; Debode, J.; De Tender, C.; Ommeslag, S.; Verwaeren, J.; Vermeir, P.; Audenaert, K.; De Gelder, L. Shifts in the rhizobiome during consecutive in planta enrichment for phosphate-solubilizing bacteria differentially affect maize P status. Microb. Biotechnol. 2021, 14, 1594–1612. [Google Scholar] [CrossRef]
- Chauhan, H.; Bagyaraj, D.J.; Selvakumar, G.; Sundaram, S.P. Novel plant growth promoting rhizobacteria—Prospects and potential. Appl. Soil Ecol. 2015, 95, 38–53. [Google Scholar] [CrossRef]
- Kim, S.-W.; Suda, W.; Kim, S.; Oshima, K.; Fukuda, S.; Ohno, H.; Morita, H.; Hattori, M. Robustness of Gut Microbiota of Healthy Adults in Response to Probiotic Intervention Revealed by High-Throughput Pyrosequencing. DNA Res. 2013, 20, 241–253. [Google Scholar] [CrossRef]
- Azizoglu, U.; Yilmaz, N.; Simsek, O.; Ibal, J.C.; Tagele, S.B.; Shin, J.-H. The fate of plant growth-promoting rhizobacteria in soilless agriculture: Future perspectives. 3 Biotech 2021, 11, 382. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Lee, J. Beneficial bacteria and fungi in hydroponic systems: Types and characteristics of hydroponic food production methods. Sci. Hortic. 2015, 195, 206–215. [Google Scholar] [CrossRef]
- Moncada, A.; Vetrano, F.; Miceli, A. Alleviation of Salt Stress by Plant Growth-Promoting Bacteria in Hydroponic Leaf Lettuce. Agronomy 2020, 10, 1523. [Google Scholar] [CrossRef]
- Utkhede, R.S.; Lévesque, C.A.; Dinh, D. Pythium aphanidermatum root rot in hydroponically grown lettuce and the effect of chemical and biological agents on its control. Can. J. Plant Pathol. 2000, 22, 138–144. [Google Scholar] [CrossRef]
- Sheridan, C.; Depuydt, P.; De Ro, M.; Petit, C.; Van Gysegem, E.; Delaere, P.; Dixon, M.; Stasiak, M.; Aciksöz, S.B.; Frossard, E.; et al. Microbial Community Dynamics and Response to Plant Growth-Promoting Microorganisms in the Rhizosphere of Four Common Food Crops Cultivated in Hydroponics. Microb. Ecol. 2017, 73, 378–393. [Google Scholar] [CrossRef]
- Van Gerrewey, T.; Vandecruys, M.; Ameloot, N.; Perneel, M.; Van Labeke, M.-C.; Boon, N.; Geelen, D. Microbe-Plant Growing Media Interactions Modulate the Effectiveness of Bacterial Amendments on Lettuce Performance Inside a Plant Factory with Artificial Lighting. Agronomy 2020, 10, 1456. [Google Scholar] [CrossRef]
- Kusuma, P.; Pattison, P.M.; Bugbee, B. From physics to fixtures to food: Current and potential LED efficacy. Hortic. Res. 2020, 7, 56. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Van Gerrewey, T.; Boon, N.; Geelen, D. Vertical Farming: The Only Way Is Up? Agronomy 2022, 12, 2. https://doi.org/10.3390/agronomy12010002
Van Gerrewey T, Boon N, Geelen D. Vertical Farming: The Only Way Is Up? Agronomy. 2022; 12(1):2. https://doi.org/10.3390/agronomy12010002
Chicago/Turabian StyleVan Gerrewey, Thijs, Nico Boon, and Danny Geelen. 2022. "Vertical Farming: The Only Way Is Up?" Agronomy 12, no. 1: 2. https://doi.org/10.3390/agronomy12010002
APA StyleVan Gerrewey, T., Boon, N., & Geelen, D. (2022). Vertical Farming: The Only Way Is Up? Agronomy, 12(1), 2. https://doi.org/10.3390/agronomy12010002






