The Impact of Drought Stress on Soil Microbial Community, Enzyme Activities and Plants
Abstract
:1. Introduction
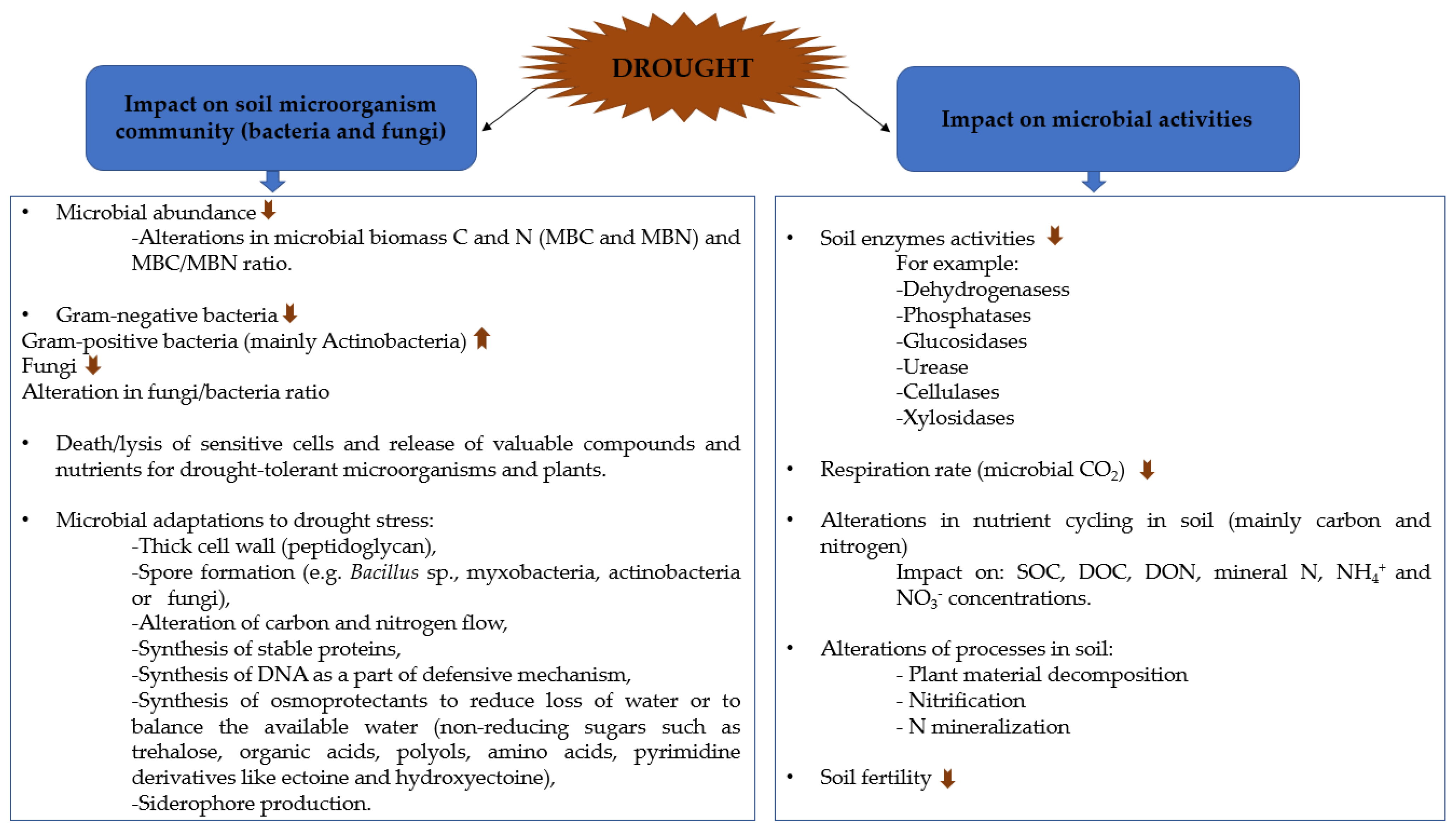
2. Impact of Drought Stress on Microbial Communities and Enzyme Activities
2.1. Effect of Drought on Microbial Communities
2.1.1. Microbial Adaptations to Drought Stress
2.1.2. Effect of Drought Stress on Microbial Community in Different Type of Soils or under Soil Modifications
2.2. Effect of Drought on Soil Enzyme Activities
2.3. Effect of Drought on Microbial Activity
2.4. Mitigation of Drought Effects on Microbial Activity by Soil Amendments
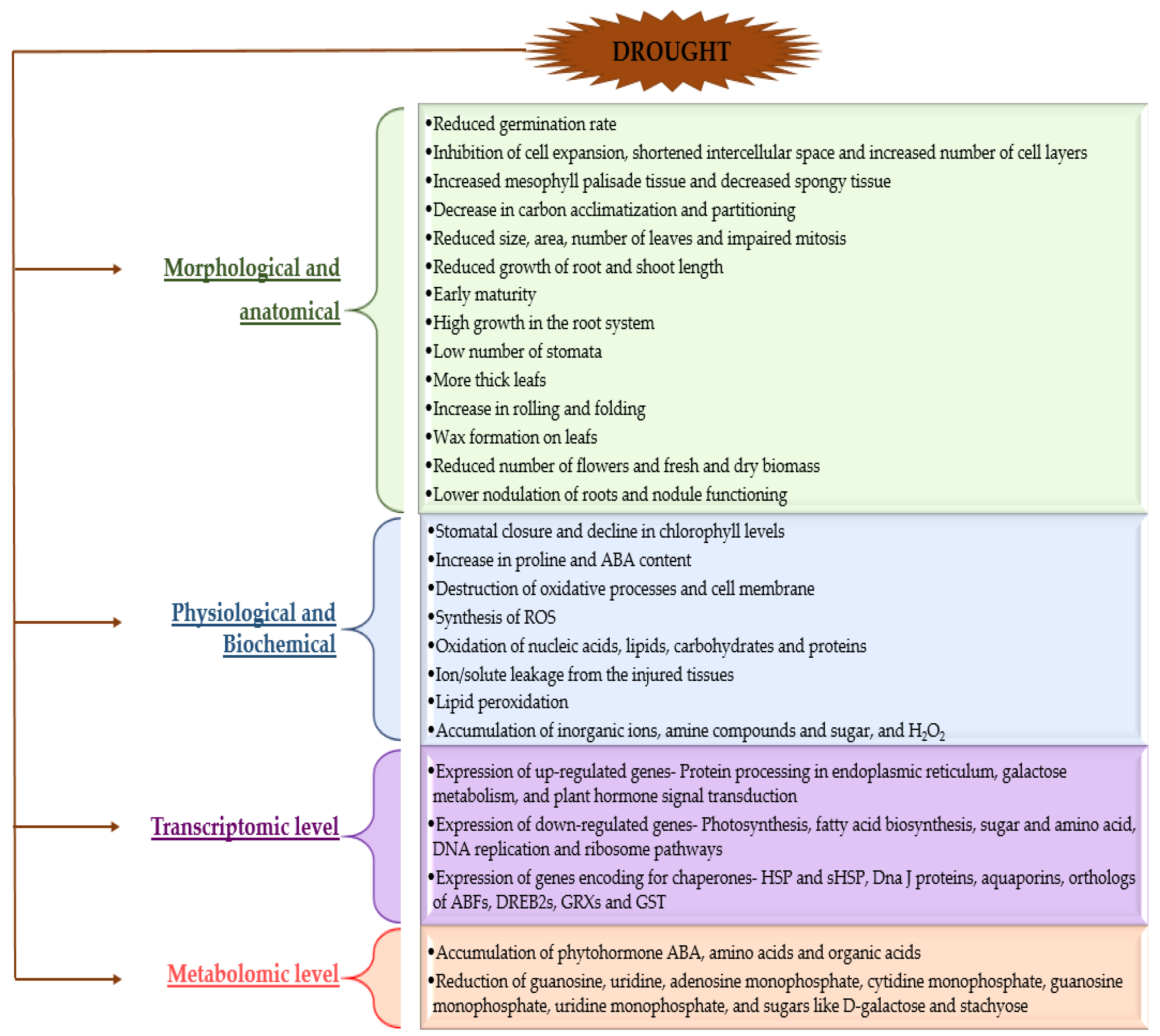
3. Plant Morphological, Physiological, Biochemical Responses towards Drought Stress
3.1. Morphological and Anatomical Changes in Plants in Response to Drought Stress
3.2. Physiological and Biochemical Changes in Plants in Response to Drought Stress
3.3. Transcriptomic and Metabolomic Changes in Plants under Drought Stress
4. Plant Growth Promotion and Protection by PGPB and PGPF under Drought Conditions
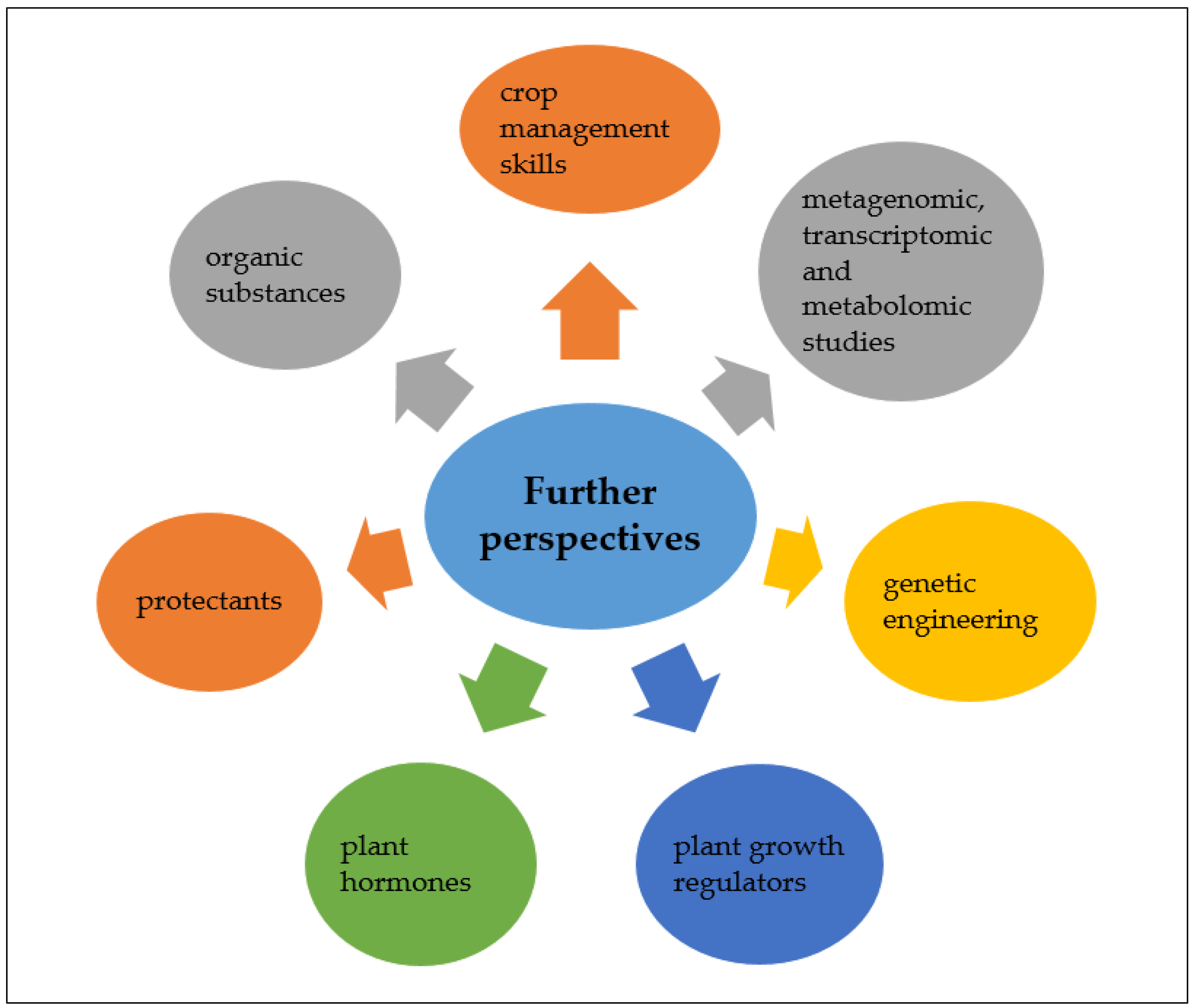
5. Conclusions and Further Perspectives
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lamaoui, M.; Jemo, M.; Datla, R.; Bekkaoui, F. Heat and drought stresses in crops and approaches for their mitigation. Front. Chem. 2018, 6, 1–14. [Google Scholar] [CrossRef]
- IPCC. Summary for Policymakers. In Climate Change 2021: The Physical Science Basis. Contribution of Working Group I to the Sixth Assessment Report of the Intergovernmental Panel on Climate Change; MassonDelmotte, V., Zhai, P., Pirani, A., Connors, S.L., Péan, C., Berger, S., Caud, N., Chen, Y., Goldfarb, L., Gomis, M.I., et al., Eds.; Cambridge University Press: Cambridge, UK, 2021; inpress. [Google Scholar]
- Kala, C.P. Environmental and socioeconomic impacts of drought in India: Lessons for drought management. Appl. Ecol. Environ. Res. 2017, 5, 43–48. [Google Scholar]
- Silva, E.C.D.; Albuquerque, M.B.D.; Neto, A.D.D.A.; Junior, C.D.D.S. Responses of Organisms to Water Stress. In Drought and Its Consequences to Plants- from Individual to Ecosystem, 1st ed.; Chapter 2; Akinci, S., Ed.; Şener Akıncı: London, UK, 2013; pp. 17–47. [Google Scholar]
- Abdul Rahman, N.S.N.; Abdul Hamid, N.W.; Nadarajah, K. Effects of Abiotic Stress on Soil Microbiome. Int. J. Mol. Sci. 2021, 22, 9036. [Google Scholar] [PubMed]
- Khan, R.; Gilani, H. Global drought monitoring with big geospatial datasets using Google Earth Engine. Environ. Sci. Pollut. Res. 2021, 28, 17244–17264. [Google Scholar] [CrossRef] [PubMed]
- Epule, E.T.; Peng, C.; Lepage, L.; Chen, Z. The causes, effects and challenges of Sahelian droughts: A critical review. Reg. Environ. Chang. 2014, 14, 145–156. [Google Scholar] [CrossRef]
- Wheaton, E.; Kulshreshtha, S.; Wittrock, V.; Koshida, G. Dry times: Hard lessons from the Canadian drought of 2001 and 2002. Can. Geograph. 2008, 52, 241–262. [Google Scholar] [CrossRef]
- Gornall, J.; Betts, R.; Burke, E.; Clark, R.; Camp, J.; Willett, K.; Wiltshire, A. Implications of climate change for agricultural productivity in the early twenty-first century. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 2973–2989. [Google Scholar] [CrossRef]
- Lesk, C.; Rowhani, P.; Ramankutty, N. Influence of extreme weather disasters on global crop production. Nature 2016, 529, 84–87. [Google Scholar] [CrossRef]
- Grillakis, M.G. Increase in severe and extreme soil moisture droughts for Europe under climate change. Sci. Total Environ. 2019, 660, 1245–1255. [Google Scholar] [CrossRef]
- Siebielec, S.; Siebielec, G.; Klimkowicz-Pawlas, A.; Gałązka, A.; Grządziel, J.; Stuczyński, T. Impact of water stress on microbial community and activity in sandy and loamy soils. Agronomy 2020, 10, 1429. [Google Scholar] [CrossRef]
- Ziernicka-Wojtaszek, A. Summer Drought in 2019 on Polish Territory-A Case Study. Atmosphere 2021, 12, 1475. [Google Scholar] [CrossRef]
- Kränzlein, M.; Geilfus, C.M.; Franzisky, B.L.; Zhang, X.; Wimmer, M.A.; Zörb, C. Physiological responses of contrasting maize (Zea mays L.) hybrids to repeated drought. J. Plant Growth Regul. 2021, 1–11. [Google Scholar] [CrossRef]
- Jamil, M.; Ahamd, M.; Anwar, F.; Zahir, Z.A.; Kharal, M.A.; Nazli, F. Inducing drought tolerance in wheat through combined use of L-tryptophan and Pseudomonas fluorescens. Pak. J. Agric. Sci. 2018, 55, 331–337. [Google Scholar]
- Yin, B.; Wang, Y.; Liu, P.; Hu, J.; Zhen, W. Effects of vesicular-arbuscular mycorrhiza on the protective system in strawberry leaves under drought stress. Front. Agric. China 2010, 4, 165–169. [Google Scholar] [CrossRef]
- Zapata, T.; Galindo, D.M.; Corrales-Ducuara, A.R.; Ocampo-Ibáñez, I.D. The diversity of culture-dependent gram-negative Rhizobacteria associated with Manihot esculenta crantz plants subjected to water-deficit stress. Diversity 2021, 13, 366. [Google Scholar] [CrossRef]
- Nguyen, L.T.T.; Osanai, Y.; Anderson, I.C.; Bange, M.P.; Tissue, D.T.; Singh, B.K. Flooding and prolonged drought have dierential legacy impacts on soil nitrogen cycling, microbial communities and plant productivity. Plant. Soil 2018, 431, 371–387. [Google Scholar] [CrossRef]
- Hanaka, A.; Ozimek, E.; Reszczy´ nska, E.; Jaroszuk-´Sciseł, J.; Stolarz, M. Plant tolerance to drought stress in the presence of supporting bacteria and fungi: An efficient strategy in horticulture. Horticulturae 2021, 7, 390. [Google Scholar] [CrossRef]
- Khan, M.N.; Zhang, J.; Luo, T.; Liu, J.; Ni, F.; Rizwan, M.; Fahad, S.; Hu, L. Morpho-physiological and biochemical responses of tolerant and sensitive rapeseed cultivars to drought stress during early seedling growth stage. Acta Physiol. Plant 2019, 41, 25. [Google Scholar] [CrossRef]
- You, J.; Zhang, Y.; Liu, A.; Li, D.; Wang, X.; Dossa, K.; Zhou, R.; Yu, J.; Zhang, Y.; Wang, L.; et al. Transcriptomic and metabolomic profiling of drought-tolerant and susceptible sesame genotypes in response to drought stress. BMC Plant Biol. 2019, 19, 267. [Google Scholar] [CrossRef] [Green Version]
- Breitkreuz, C.; Herzig, L.; Buscot, F.; Reitz, T.; Tarkka, M. Interactions between soil properties, agricultural management and cultivar type drive structural and functional adaptations of the wheat rhizosphere microbiome to drought. Environ. Microbiol. 2021, 23, 5866–5882. [Google Scholar] [CrossRef] [PubMed]
- Milošević, N.A.; Marinković, J.B.; Tintor, B.B. Mitigating abiotic stress in crop plants by microorganisms. Proc. Nat. Sci. Matica Serpska Novi. Sad 2012, 123, 17–26. [Google Scholar] [CrossRef]
- Pascual, J.A.; Hernandez, T.; Garcia, C.; Ayuso, M. Enzymatic activities in an arid soil amended with urban organic wastes: Laboratory experiment. Bioresour. Technol. 1998, 64, 131–138. [Google Scholar] [CrossRef]
- Sayer, E.J.; Crawford, J.A.; Edgerley, J.; Askew, A.P.; Hahn, C.Z.; Whitlock, R.; Dodd, I.C. Adaptation to chronic drought modifies soil microbial community responses to phytohormones. Commun. Biol. 2021, 4, 516. [Google Scholar] [CrossRef]
- Schimel, J.; Balser, T.C.; Wallenstein, M. Microbial stress-response physiology and its implications for ecosystem function. Ecology 2007, 88, 1386–1394. [Google Scholar] [CrossRef]
- Yerbury, J.J.; Stewart, E.M.; Wyatt, A.R.; Wilson, M.R. Quality control of protein folding in extracellular space. EMBO Rep. 2005, 6, 1131–1136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Csonka, L.N. Physiological and genetic responses of bacteria to osmotic stress. Microbiol. Rev. 1989, 53, 121–147. [Google Scholar] [CrossRef]
- LeBlanc, J.C.; Gonçalves, E.R.; Mohn, W.W. Global response to desiccation stress in the soil actinomycete Rhodococcus jostii RHA1. Appl. Environ. Microbiol. 2008, 74, 2627–2636. [Google Scholar] [CrossRef] [Green Version]
- Narváez-Reinaldo, J.J.; Barba, I.; González-López, J.; Tunnacliffe, A.; Manzanera, M. Rapid method for isolation of desiccation-tolerant strains and xeroprotectants. Appl. Environ. Microbiol. 2010, 76, 5254–5262. [Google Scholar] [CrossRef] [Green Version]
- Alvarez, H.M.; Silva, R.A.; Cesari, A.C.; Zamit, A.L.; Peressutti, S.R.; Reichelt, R.; Keller, U.; Malkus, U.; Rasch, C.; Maskow, T.; et al. Physiological and morphological responses of the soil bacterium Rhodococcus opacus strain PD630 to water stress. FEMS Microbiol. Ecol. 2004, 50, 75–86. [Google Scholar] [CrossRef] [Green Version]
- Palud, A.; Salem, K.; Cavin, J.F.; Beney, L.; Licandro, H. Identification and transcriptional profile of Lactobacillus paracasei genes involved in the response to desiccation and rehydration. Food Microbiol. 2020, 85, 103301. [Google Scholar] [CrossRef] [PubMed]
- Vílchez, J.I.; García-Fontana, C.; Román-Naranjo, D.; González-López, J.; Manzanera, M. Plant drought tolerance enhancement by trehalose production of desiccation-tolerant microorganisms. Front. Microbiol. 2016, 7, 1577. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manzanera, M. Dealing with water stress and microbial preservation. Environ. Microbiol. 2021, 23, 3351–3359. [Google Scholar] [CrossRef]
- Vílchez, J.I.; Niehaus, K.; Dowling, D.N.; González-López, J.; Manzanera, M. Protection of pepper plants from drought by Microbacterium sp. 3J1 by modulation of the plant’s glutamine and α-ketoglutarate content: A comparative metabolomics approach. Front. Microbiol. 2018, 9, 284. [Google Scholar] [CrossRef] [PubMed]
- Tanne, C.; Golovina, E.A.; Hoekstra, F.A.; Meffert, A.; Galinski, E.A. Glass-forming property of hydroxyectoine is the cause of its superior function as a desiccation protectant. Front. Microbiol. 2014, 5, 150. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Julca, I.; Alaminos, M.; González-López, J.; Manzanera, M. Xeroprotectants for the stabilization of biomaterials. Biotechnol. Adv. 2012, 30, 1641–1654. [Google Scholar] [CrossRef] [PubMed]
- SantaCruz-Calvo, L.; González-López, J.; Manzanera, M. Arthrobacter siccitolerans sp. nov., a highly desiccation-tolerant, xeroprotectant-producing strain isolated from dry soil. Int. J. Syst. Evol. Microbiol. 2013, 63, 4174–4180. [Google Scholar] [CrossRef] [PubMed]
- Da Costa, M.S.; Santos, H.; Galinski, E.A. An overview of the role and diversity of compatible solutes in Bacteria and Archaea. Adv. Biochem. Eng. Biotechnol. 1998, 61, 117–153. [Google Scholar] [PubMed]
- Crisp, A.; Boschetti, C.; Perry, M.; Tunnacliffe, A.; Micklem, G. Expression of multiple horizontally acquired genes is a hallmark of both vertebrate and invertebrate genomes. Genome Biol. 2015, 16, 50. [Google Scholar] [CrossRef] [Green Version]
- García-Fontana, C.; Narváez-Reinaldo, J.J.; Castillo, F.; González-López, J.; Luque, I.; Manzanera, M.A. New physiological role for the DNA molecule as a protector against drying Stress in desiccation-tolerant microorganisms. Front. Microbiol. 2016, 7, 2066. [Google Scholar] [CrossRef] [Green Version]
- Arzanesh, M.H.; Alikhani, H.A.; Khavazi, K.; Rahimian, H.A.; Miransari, M. Wheat (Triticum aestivum L.) growth enhancement by Azospirillum sp. under drought stress. World J. Microbiol. Biotechnol. 2011, 27, 197–205. [Google Scholar] [CrossRef]
- Hueso, S.; García, C.; Hernández, T. Severe drought conditions modify the microbial community structure, size and activity in amended and unamended soils. Soil Biol. Biochem. 2012, 50, 167–173. [Google Scholar] [CrossRef]
- Baldrian, P.; Merhautová, V.; Petránková, M.; Cajthaml, T.; Šnajdr, J. Distribution of microbial biomass and activity of extracellular enzymes in a hardwood forest soil reflect soil moisture content. Appl. Soil Ecol. 2010, 46, 177–182. [Google Scholar] [CrossRef]
- Bastida, F.; Torres, I.F.; Hernández, T.; García, C. The impacts of organic amendments: Do they confer stability against drought on the soil microbial community? Soil Biol. Biochem. 2017, 113, 173–183. [Google Scholar] [CrossRef]
- Veach, A.M.; Zeglin, L.H. Historical drought affects microbial population dynamics and activity during soil drying and re-wet. Microb. Ecol. 2020, 79, 662–674. [Google Scholar] [CrossRef]
- Castro, S.P.; Cleland, E.E.; Wagner, R.; Sawad, R.A.; Lipson, D.A. Soil microbial responses to drought and exotic plants shift carbon metabolism. ISME J. 2019, 13, 1776. [Google Scholar] [CrossRef] [PubMed]
- Schimel, J.P. Life in dry soils: Effects of drought on soil microbial communities and processes. Annu. Rev. Ecol. Evol. 2018, 49, 409–432. [Google Scholar] [CrossRef]
- Yuste, J.C.; Penuelas, J.; Estiarte, M.; Garcia-mas, J.; Mattana, S.; Ogaya, R.; Pujol, M.; Sardans, J. Drought-resistant fungi control soil organic matter decomposition and its response to temperature. Glob. Chang. Biol. 2011, 17, 1475–1486. [Google Scholar] [CrossRef]
- Naylor, D.; DeGraaf, S.; Purdom, E.; Coleman-Derr, D. Drought and host selection influence bacterial community dynamics in the grass root microbiome. ISME J. 2017, 11, 2691–2704. [Google Scholar] [CrossRef] [Green Version]
- Chernysheva, E.; Korobov, D.; Khomutova, T.; Fornasier, F.; Borisov, A. Soil microbiological properties in livestock corrals: An additional new line of evidence to identify livestock dung. J. Archaeol. Sci. Rep. 2021, 37, 103012. [Google Scholar] [CrossRef]
- Kompała-Bąba, A.; Bierza, W.; Sierka, E.; Błońska, A.; Besenyei, L.; Woźniak, G. The role of plants and soil properties in the enzyme activities of substrates on hard coal mine spoil heaps. Sci. Rep. 2021, 11, 5155. [Google Scholar] [CrossRef]
- Meena, A.; Rao, K.S. Assessment of soil microbial and enzyme activity in the rhizosphere zone under different land use/cover of a semiarid region, India. Ecol. Process. 2021, 10, 1–12. [Google Scholar] [CrossRef]
- Shukla, G.; Varma, A. (Eds.) Soil Enzymology; Springer: Berlin/Heidelberg, Germany, 2011; pp. 1–392. [Google Scholar]
- Hemkemeyer, M.; Schwalb, S.A.; Heinze, S.; Joergensen, R.G.; Wichern, F. Functions of elements in soil microorganisms. Microb. Res. 2021, 252, 126832. [Google Scholar] [CrossRef] [PubMed]
- Mulvaney, R.L.; Bremner, J.M. Use of p-benzoquinone and hydroquinone for retardation of urea hydrolysis in soils. Soil Biol. Biochem. 1978, 10, 297–302. [Google Scholar] [CrossRef]
- Hu, J.; Lin, X.; Wang, J.; Dai, J.; Chen, R.; Zhang, J.; Wong, M.H. Microbial functional diversity, metabolic quotient, and invertase activity of a sandy loam soil as affected by long-term application of organic amendment and mineral fertilizer. J. Soils Sediments 2011, 11, 271–280. [Google Scholar] [CrossRef]
- Bakshi, M.; Varma, A. Soil Enzyme: The State-of-Art. In Soil Enzymology; Shukla, G., Varma, A., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 22, pp. 1–23. [Google Scholar]
- Sardans, J.; Peñuelas, J. Drought decreases soil enzyme activity in a Mediterranean Quercus ilex L. forest. Soil Biol. Biochem. 2005, 37, 455–461. [Google Scholar] [CrossRef]
- Bååth, E.; Söderström, B. Seasonal and spatial variation in fungal biomass in a forest soil. Soil Biol. Biochem. 1982, 14, 353–358. [Google Scholar] [CrossRef]
- Larned, S.T.; Datry, T.; Robinson, C. Invertebrate and microbial responses to inundation in an ephemeral river reach in New Zealand: Effects of preceding dry periods. Aquat. Sci. 2007, 69, 554–567. [Google Scholar] [CrossRef]
- Pohlon, E.; Fandino, A.O.; Marxsen, J. Bacterial community composition and extracellular enzyme activity in temperate streambed sediment during drying and rewetting. PLoS ONE 2013, 8, e83365. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schreckinger, J.; Mutz, M.; Mendoza-Lera, C.; Frossard, A. Attributes of drying define the structure and functioning of microbial communities in temperate riverbed sediment. Front. Microbiol. 2021, 12, 676615. [Google Scholar] [CrossRef]
- Sirová, D.; Vrba, J.; Rejmánková, E. Extracellular enzyme activities in benthic cyanobacterial mats: Comparison between nutrient-enriched and control sites in marshes of northern Belize. Aquat. Microb. Ecol. 2006, 44, 11–20. [Google Scholar] [CrossRef]
- Criquet, S.; Farnet, A.M.; Tagger, S.; Le Petit, J. Annual variations of phenoloxidase activities in an evergreen oak litter: Influence of certain biotic and abiotic factors. Soil Biol. Biochem. 2000, 32, 1505–1513. [Google Scholar] [CrossRef]
- Criquet, S.; Ferre, E.; Farnet, A.M.; Le petit, J. Annual dynamics of phosphatase activities in an evergreen oak litter: Influence of biotic and abiotic factors. Soil Biol. Biochem. 2004, 36, 1111–1118. [Google Scholar] [CrossRef]
- Siebielec, G.; Siebielec, S.; Lipski, D. Long-term impact of sewage sludge, digestate and mineral fertilizers on plant yield and soil biological activity. J. Clean. Prod. 2018, 187, 372–379. [Google Scholar] [CrossRef]
- Weintraub, M.N.; Scott-Denton, L.E.; Schmidt, S.K.; Monson, R.K. The effects of tree rhizodeposition on soil exoenzyme activity, dissolved organic carbon, and nutrient availability in a subalpine forest ecosystem. Oecologia 2007, 154, 327–338. [Google Scholar] [CrossRef] [PubMed]
- Preece, C.; Verbruggen, E.; Liu, L.; Weedon, J.T.; Peñuelas, J. Effects of past and current drought on the composition and diversity of soil microbial communities. Soil Biol. Biochem. 2019, 131, 28–39. [Google Scholar] [CrossRef]
- Gleeson, D.B.; Herrmann, A.M.; Livesley, S.J.; Murphy, D.V. Influence of water potential on nitrification and structure of nitrifying bacterial communities in semiarid soils. Appl. Soil Ecol. 2008, 40, 189–194. [Google Scholar] [CrossRef]
- Fuchslueger, L.; Kastl, E.M.; Bauer, F.; Kienzl, S.; Hasibeder, R.; Ladreiter-Knauss, T.; Schmidt, M.; Bahn, M.; Schloter, M.; Richter, A.; et al. Effect of drought on nitrogen turnover and abundances of ammonia-oxidizers in mountain grassland. Biogeosciences 2014, 11, 6003–6015. [Google Scholar] [CrossRef] [Green Version]
- Deng, L.; Peng, C.; Kim, D.G.; Li, J.; Liu, Y.; Hai, X.; Liu, Q.; Huang, C.; Shangguan, Z.; Kuzyakov, Y. Drought effects on soil carbon and nitrogen dynamics in global natural ecosystems. Earth Sci. Rev. 2021, 214, 103501. [Google Scholar] [CrossRef]
- Hewedy, O.A.; Abdel Lateif, K.S.; Seleiman, M.F.; Shami, A.; Albarakaty, F.M.; El-Meihy, R.M. Phylogenetic diversity of Trichoderma strains and their antagonistic potential against soil-borne pathogens under stress conditions. Biology 2020, 9, 189. [Google Scholar] [CrossRef]
- Wang, W.X.; Vinocur, B.; Shoseyov, O.; Altman, A. Biotechnology of plant osmotic stress tolerance: Physiological and molecular considerations. Acta Hortic. 2001, 560, 285–292. [Google Scholar] [CrossRef]
- Wilmowicz, E.; Kućko, A.; Golińska, P.; Burchardt, S.; Przywieczerski, T.; Świdziński, M.; Brzozowska, P.; Kapuścińska, D. Abscisic acid and ethylene in the control of nodule-specific response on drought in yellow lupine. Environ. Exp. Bot. 2020, 169, 103900. [Google Scholar] [CrossRef]
- Ulrich, D.E.M.; Sevanto, S.; Ryan, M.; Albright, M.B.N.; Johansen, R.B.; Dunbar, J.M. Plant-microbe interactions before drought influence plant physiological responses to subsequent severe drought. Sci. Rep. 2019, 9, 249. [Google Scholar] [CrossRef] [Green Version]
- Jabbari, H.; Akbari, G.A.; Sima, N.A.K.K.; Rad, A.H.S.; Alahdadi, I.; Hamed, A.; Shariatpanahi, M.E. Relationships between seedling establishment and soil moisture content for winter and spring rapeseed genotypes. Ind. Crops Prod. 2013, 49, 177–187. [Google Scholar] [CrossRef]
- Yang, X.; Lu, M.; Wang, Y.; Wang, Y.; Liu, Z.; Chen, S. Response Mechanism of Plants to Drought Stress. Horticulturae 2021, 7, 50. [Google Scholar] [CrossRef]
- Sangtarash, M.H.; Qaderi, M.M.; Chinnappa, C.C.; Reid, D.M. Differential sensitivity of canola (Brassica napus L.) seedlings to ultraviolet-B radiation, water stress and abscisic acid. Environ. Exp. Bot. 2009, 66, 212–219. [Google Scholar] [CrossRef]
- Qaderi, M.M.; Kurepin, L.V.; Reid, D.M. Effects of temperature and watering regime on growth gas exchange and abscisic acid content of canola (Brassica napus L.) seedlings. Environ. Exp. Bot. 2012, 75, 107–113. [Google Scholar] [CrossRef]
- Liu, D.; Wu, L.; Naeem, M.S.; Liu, H.; Deng, X.; Xu, L.; Zhang, F.; Zhou, W. 5-Aminolevulinic acid enhances photosynthetic gas exchange, chlorophyll fluorescence and antioxidant system in oilseed rape under drought stress. Acta Physiol. Plant 2013, 35, 2747–2759. [Google Scholar] [CrossRef]
- Chartzoulakis, K.; Patakas, A.; Kofidis, G.; Bosabalidis, A.; Nastou, A. Water stress affects leaf anatomy, gas exchange, water relations and growth of two avocado cultivars. Sci. Hortic. 2002, 95, 39–50. [Google Scholar] [CrossRef]
- Furlan, A.; Llanes, A.; Luna, V.; Castro, S. Physiological and biochemical responses to drought stress and subsequent rehydration in the symbiotic association peanut-Bradyrhizobium sp. ISRN Agron. 2012, 2012, 890–895. [Google Scholar] [CrossRef] [Green Version]
- Cutler, S.R.; Rodriguez, P.L.; Finkelstein, R.R.; Abrams, S.R. Abscisic acid: Emergence of a core signaling network. Annu. Rev. Plant Biol. 2010, 61, 651–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calvo, O.C.; Franzaring, J.; Schmid, I.; Müller, M.; Brohon, N.; Fangmeier, A. Atmospheric CO2 enrichment and drought stress modify root exudation of barley. Glob. Chang. Biol. 2016, 23, 1292–1304. [Google Scholar] [CrossRef] [PubMed]
- Galeano, E.; Vasconcelos, T.S.; Novais de Oliveira, P.N.; Carrer, H. Physiological and molecular responses to drought stress in teak (Tectona grandis L.f.). PLoS ONE 2019, 14, e0221571. [Google Scholar] [CrossRef]
- Takahashi, F.; Kuromori, T.; Urano, K.; Yamaguchi-Shinozaki, K.; Shinozaki, K. Drought stress responses and resistance in plants: From cellular responses to long-distance intercellular communication. Front. Plant Sci. 2020, 11, 556972. [Google Scholar] [CrossRef] [PubMed]
- Gimenez, C.; Mitchell, V.J.; Lawlor, D.W. Regulation of photosynthetic rate of two sunflower hybrids under water stress. Plant Physiol. 1992, 98, 516–524. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, P.; Bai, T.H.; Ma, F.W. Effects of progressive drought on photosynthesis and partitioning of absorbed light in apple trees. J. Integr. Agric. 2015, 14, 681–690. [Google Scholar] [CrossRef] [Green Version]
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef]
- Feng, Y.L.; Cao, K.F. Photosynthesis and photo inhibition after night chilling in seedlings of two tropical tree species grown under three irradiances. Photosynthetica 2005, 43, 567–574. [Google Scholar] [CrossRef]
- Azzeme, A.M.; Abdullah, S.N.A.; Aziz, M.A.; Wahab, P.E.M. Oil palm leaves and roots differ in physiological response, antioxidant enzyme activities and expression of stress-responsive genes upon exposure to drought stress. Acta Physiol. Plant. 2016, 38, 1–12. [Google Scholar] [CrossRef]
- Sabra, A.; Daayf, F.; Renault, S. Differential physiological and biochemical responses of three Echinacea species to salinity stress. Scientia. Hort. 2012, 135, 23–31. [Google Scholar] [CrossRef]
- Tarazona, P.; Feussner, K.; Feussner, I. An enhanced plant lipidomics method based on multiplexed liquid chromatography-mass spectrometry reveals additional insights into cold- and drought-induced membrane remodeling. Plant J. 2015, 84, 621–633. [Google Scholar] [CrossRef]
- Dossa, K.; Li, D.; Wang, L.; Zheng, X.; Liu, A.; Yu, J.; Wei, X.; Zhou, R.; Fonceka, D.; Diouf, D.; et al. Transcriptomic, biochemical and physio-anatomical investigations shed more light on responses to drought stress in two contrasting sesame genotypes. Sci. Rep. 2017, 7, 8755. [Google Scholar] [CrossRef] [Green Version]
- Bao, G.; Tang, W.; An, Q.; Liu, Y.; Tian, J.; Zhao, N.; Zhu, S. Physiological effects of the combined stresses of freezing-thawing, acid precipitation and deicing salt on alfalfa seedlings. BMC Plant Biol. 2020, 20, 204. [Google Scholar] [CrossRef]
- Gupta, N.; Thind, S.K.; Bains, N.S. Glycine betaine application modifies biochemical attributes of osmotic adjustment in drought stressed wheat. Plant Growth Regul. 2014, 72, 221–228. [Google Scholar] [CrossRef]
- Abdi, G.; Shokrpour, M.; Karami, L.; Salami, S.A. Prolonged water deficit stress and methyl jasmonate-mediated changes in metabolite profile, flavonoid concentrations and antioxidant activity in peppermint (Mentha × piperita L.). Not. Bot. Horti. Agrobo. 2019, 47, 70–80. [Google Scholar] [CrossRef] [Green Version]
- Hou, P.; Wang, F.; Luo, B.; Li, A.; Wang, C.; Shabala, L.; Ahmed, H.A.I.; Deng, S.; Zhang, H.; Song, P.; et al. Antioxidant enzymatic activity and osmotic adjustment as components of the drought tolerance mechanism in Carex duriuscula. Plants 2021, 10, 436. [Google Scholar] [CrossRef] [PubMed]
- Ozturk, M.; Unal, B.T.; García-Caparrós, P.; Khursheed, A.; Gul, A.; Hasanuzzaman, M. Osmoregulation and its actions during the drought stress in plants. Physiol. Plant. 2021, 172, 1321–1335. [Google Scholar] [CrossRef] [PubMed]
- Khoyerdi, F.F.; Shamshiri, M.H.; Estaji, A. Changes in some physiological and osmotic parameters of several pistachio genotypes under drought stress. Sci. Hortic. 2016, 198, 44–51. [Google Scholar] [CrossRef]
- Liu, C.; Wang, Y.; Pan, K.; Li, W.; Zhang, L.; Shen, X.; Liu, L.; Deng, M. Responses of antioxidant defense system to drought stress in the leaves of Fargesia denudata seedlings, the staple food on the giant panda. Russ. J. Plant Physiol. 2014, 3, 374–383. [Google Scholar] [CrossRef] [Green Version]
- Moral, A.Z.; Martínez-Reyes, D.; Quinto, C.; Sanchez, F.; Díaz-Camino, C. Identification of small open reading frames (sORFs) associated with heat tolerance in nitrogen-fixing root nodules of Phaseolus vulgaris wild-type and cv BAT93. Aust. J. Crop Sci. 2021, 15, 28–37. [Google Scholar] [CrossRef]
- Ni, Z.; Liu, N.; Yu, Y.; Bi, C.; Chen, Q.; Qu, Y. The cotton 70-kDa heat shock protein GhHSP70-26 plays a positive role in the drought stress response. Environ. Exp. Bot. 2021, 191, 104628. [Google Scholar] [CrossRef]
- Feng, X.H.; Zhang, H.X.; Ali, M.; Gai, W.X.; Cheng, G.X.; Yu, Q.H.; Yang, S.B.; Li, X.X.; Gong, Z.H. A small heat shock protein CaHsp25. 9 positively regulates heat, salt, and drought stress tolerance in pepper (Capsicum annuum L.). Plant Physiol. Biochem. 2019, 142, 151–162. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.L.; Wang, Y.J.; Xie, S.L.; Wang, C.; Wang, W. Glycinebetaine application ameliorates negative effects of drought stress in tobacco. Russ. J. Plant Physiol. 2007, 54, 472. [Google Scholar] [CrossRef]
- Mignolet-Spruyt, L.; Xu, E.; Idänheimo, N.; Hoeberichts, F.A.; Mühlenbock, P.; Brosché, M.; Van Breusegem, F.; Kangasjärvi, J. Spreading the news: Subcellular and organellar reactive oxygen species production and signalling. J. Exp. Bot. 2016, 67, 3831–3844. [Google Scholar] [CrossRef] [Green Version]
- Dietz, K.-J. Thiol-based peroxidases and ascorbate peroxidases: Why plants rely on multiple peroxidase systems in the photosynthesizing chloroplast? Mol. Cells 2016, 39, 20–25. [Google Scholar] [PubMed] [Green Version]
- Dröse, S.; Brandt, U. Molecular mechanisms of superoxide production by the mitochondrial respiratory chain. Adv. Exp. Med. Biol. 2012, 748, 145–169. [Google Scholar]
- Pacher, P.; Beckman, J.S.; Liaudet, L. Nitric Oxide and Peroxynitrite in Health and Disease. Physiol. Rev. 2007, 87, 315–424. [Google Scholar] [CrossRef] [Green Version]
- Stadtman, E.R.; Moskovitz, J.; Levine, R.L. Oxidation of methionine residues of proteins: Biological consequences. Antioxid Redox Signal. 2003, 5, 577–582. [Google Scholar] [CrossRef]
- Marnett, L.J. Oxyradicals and DNA damage. Carcinogenesis 2000, 21, 361–370. [Google Scholar] [CrossRef] [Green Version]
- Apel, K.H.; Hirt, H. Reactive oxygen species: Metabolism, oxidative stress, and signal transduction. Annu. Rev. Plant Biol. 2004, 55, 373–399. [Google Scholar] [CrossRef] [Green Version]
- Kaur, K.; Kaur, N.; Gupta, A.K.; Singh, I. Exploration of the antioxidative defense system to characterize chickpea genotypes showing differential response towards water deficit conditions. Plant Growth Regul. 2013, 70, 49–60. [Google Scholar] [CrossRef]
- Wang, W.B.; Kim, Y.H.; Lee, H.S.; Kim, K.Y.; Deng, X.P.; Kwak, S.S. Analysis of antioxidant enzyme activity during germination of alfalfa under salt and drought stresses. Plant Physiol. Biochem. 2009, 47, 570–577. [Google Scholar] [CrossRef]
- Kuźniak, E.K.; Skłodowska, M. Ascorbate, glutathione and related enzymes in chloroplasts of tomato leaves infected by Botrytis cinerea. Plant Sci. 2001, 160, 723–731. [Google Scholar] [CrossRef]
- Sun, Y.; Wang, C.; Chen, H.Y.H.; Ruan, H. Response of Plants to Water Stress: A Meta-Analysis. Front. Plant Sci. 2020, 11, 978. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Chen, J.; Huang, W.; Song, X.; Niu, J. Transcriptomics and metabolomics reveal purine and phenylpropanoid metabolism response to drought stress in Dendrobium sinense, an endemic orchid species in Hainan Island. Front. Genet. 2021, 12, 692702. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [Green Version]
- Jaiswal, S.; Antala, T.J.; Mandavia, M.K.; Chopra, M.; Jasrotia, R.S.; Tomar, R.S.; Kheni, J.; Angadi, U.B.; Iquebal, M.A.; Golakia, B.A.; et al. Transcriptomic signature of drought response in pearl millet (Pennisetum glaucum L.) and development of web-genomic resources. Sci. Rep. 2018, 8, 3382. [Google Scholar] [CrossRef]
- Egea, I.; Albaladejo, I.; Meco, V.; Morales, B.; Sevilla, A.; Bolarin, M.C.; Flores, F.B. The drought-tolerant Solanum pennellii regulates leaf water loss and induces genes involved in amino acid and ethylene/jasmonate metabolism under dehydration. Sci. Rep. 2018, 8, 2791. [Google Scholar] [CrossRef]
- You, J.; Wang, Y.; Zhang, Y.; Dossa, K.; Li, D.; Zhou, R.; Wang, L.; Zhang, X. Genome-wide identification and expression analyses of genes involved in raffinose accumulation in sesame. Sci. Rep. 2018, 8, 4331. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, P.; Yang, C.; Chen, H.; Song, C.; Zhang, X.; Wang, D. Transcriptomic basis for drought-resistance in Brassica napus L. Sci. Rep. 2017, 7, 40532. [Google Scholar] [CrossRef] [Green Version]
- Pires, M.V.; Júnior, A.A.P.; Medeiros, D.B.; Daloso, D.M.; Pham, P.A.; Barros, K.A.; Engqvist, M.K.; Florian, A.; Krahnert, I.; Maurino, V.G.; et al. The influence of alternative pathways of respiration that utilize branched-chain amino acids following water shortage in Arabidopsis. Plant Cell Environ. 2016, 39, 1304–1319. [Google Scholar] [CrossRef] [Green Version]
- Chmielewska, K.; Rodziewicz, P.; Swarcewicz, B.; Sawikowska, A.; Krajewski, P.; Marczak, L.; Ciesiolka, D.; Kuczynska, A.; Mikolajczak, K.; Ogrodowicz, P.; et al. Analysis of drought-induced proteomic and metabolomic changes in barley (Hordeum vulgare L.) leaves and roots unravels some aspects of biochemical mechanisms involved in drought tolerance. Front. Plant Sci. 2016, 7, 1108. [Google Scholar] [CrossRef] [PubMed]
- Bowne, J.B.; Erwin, T.A.; Juttner, J.; Schnurbusch, T.; Langridge, P.; Bacic, A.; Roessner, U. Drought responses of leaf tissues from wheat cultivars of differing drought tolerance at the metabolite level. Mol. Plant. 2012, 5, 418–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, W.; Vinocur, B.; Shoseyov, O.; Altman, A. Role of plant heat-shock proteins and molecular chaperones in the abiotic stress response. Trends Plant Sci. 2004, 9, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Howell, S.H. Endoplasmic reticulum stress responses in plants. Annu. Rev. Plant Biol. 2013, 64, 477–499. [Google Scholar] [CrossRef] [Green Version]
- Cho, E.K.; Hong, C.B. Over-expression of tobacco NtHSP70-1 contributes to drought-stress tolerance in plants. Plant Cell Rep. 2006, 25, 349–358. [Google Scholar] [CrossRef]
- Deshmukh, R.K.; Sonah, H.; Belanger, R.R. Plant aquaporins: Genome-wide identification, transcriptomics, proteomics, and advanced analytical tools. Front. Plant Sci. 2016, 7, 1896. [Google Scholar] [CrossRef] [Green Version]
- Hand, S.C.; Menze, M.A.; Toner, M.; Boswell, L.; Moore, D. LEA proteins during water stress: Not just for plants anymore. Annu. Rev. Physiol. 2011, 73, 115–134. [Google Scholar] [CrossRef] [Green Version]
- Prashanth, S.R.; Sadhasivam, V.; Parida, A. Over expression of cytosolic copper/zinc superoxide dismutase from a mangrove plant Avicennia marina in indica rice var Pusa Basmati-1 confers abiotic stress tolerance. Transgenic Res. 2008, 17, 281–291. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhang, Q.; Wu, J.; Zheng, X.; Zheng, S.; Sun, X.; Qiu, Q.; Lu, T. Gene knockout study reveals that cytosolic ascorbate peroxidase 2 (OsAPX2) plays a critical role in growth and reproduction in rice under drought, salt and cold stresses. PLoS ONE 2013, 8, e57472. [Google Scholar]
- Li, S. Redox modulation matters: Emerging functions for glutaredoxins in plant development and stress responses. Plants 2014, 3, 559–582. [Google Scholar] [CrossRef] [Green Version]
- Guo, Y.; Huang, C.; Xie, Y.; Song, F.; Zhou, X.A. tomato glutaredoxin gene SlGRX1 regulates plant responses to oxidative, drought and salt stresses. Planta 2010, 232, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Hu, Y.; Sprague, S.A.; Kakeshpour, T.; Park, J.; Nakata, P.A.; Cheng, N.; Hirschi, K.D.; White, F.F.; Park, S. Expression of a monothiol glutaredoxin, AtGRXS17, in tomato (Solanum lycopersicum) enhances drought tolerance. Biochem. Biophys. Res. Commun. 2017, 491, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Du, M.; Ding, G.; Cai, Q. The transcriptomic responses of Pinus massoniana to drought stress. Forests 2018, 9, 326. [Google Scholar] [CrossRef] [Green Version]
- Dong, B.; Wu, B.; Hong, W.; Li, X.; Li, Z.; Xue, L.; Huang, Y. Transcriptome analysis of the tea oil camellia (Camellia oleifera) reveals candidate drought stress genes. PLoS ONE 2017, 12, e0181835. [Google Scholar]
- Zhang, Q.; Liu, H.; Wu, X.; Wang, W. Identification of drought tolerant mechanisms in a drought-tolerant maize mutant based on physiological, biochemical and transcriptomic analyses. BMC Plant Biol. 2020, 20, 315. [Google Scholar] [CrossRef]
- Jiang, C.; Li, X.; Zou, J.; Ren, J.; Jin, C.; Zhang, H.; Yu, H.; Jin, H. Comparative transcriptome analysis of genes involved in the drought stress response of two peanut (Arachis hypogaea L.) varieties. BMC Plant Biol. 2021, 21, 64. [Google Scholar] [CrossRef]
- Tahmasebi, A.; Niazi, A. Comparison of Transcriptional Response of C3 and C4 Plants to Drought Stress Using Meta-Analysis and Systems Biology Approach. Front. Plant Sci. 2021, 12, 668736. [Google Scholar] [CrossRef]
- Heijden, M.G.A.V.D.; Bardgett, R.D.; Straalen, N.M.V. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar] [CrossRef] [PubMed]
- Cavicchioli, R.; William, J.R.; Timmis, K.N.; Azam, F.; Bakken, L.R.; Baylis, M.; Behrenfeld, M.J.; Boetius, A.; Boyd, P.W.; Classen, A.T.; et al. Scientists’ warning to humanity: Microorganisms and climate change. Nat. Rev. Microbiol. 2019, 17, 569–586. [Google Scholar] [CrossRef] [Green Version]
- Ullah, A.; Akbar, A.; Luo, Q.; Khan, A.H.; Manghwar, H.; Shaban, M.; Yang, X. Microbiome Diversity in Cotton Rhizosphere Under Normal and Drought Conditions. Microb. Ecol. 2019, 77, 429–439. [Google Scholar] [CrossRef]
- Nannipieri, P.; Ascher, J.; Ceccherini, M.T.; Landi, L.; Pietramellara, G.; Renella, G.; Valori, F. Microbial diversity and microbial activity in the rhizosphere. Cienc. Suelo 2007, 25, 89–97. [Google Scholar]
- Preece, C.; Peñuelas, J. Rhizodeposition under drought and consequences for soil communities and ecosystem resilience. Plant Soil 2016, 409, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Yang, Z.; Zhou, B.; Chen, Q.; Ge, X.; Shi, Y. Effects of drought on root architecture and non-structural carbohydrate of Cunninghamia lanceolata. Acta Ecol. Sin. 2018, 38, 6729–6740. [Google Scholar]
- Dai, L.; Zhang, G.; Yu, Z.; Ding, H.; Xu, Y.; Zhang, Z. Effect of drought stress and developmental stages on microbial community structure and diversity in peanut rhizosphere soil. Int. J. Mol. Sci. 2019, 20, 2265. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A.; Singh, S.; Gaurav, A.K.; Srivastava, S.; Verma, J.P. Plant growth-promoting bacteria: Biological tools for the mitigation of salinity stress in plants. Front. Microbiol. 2020, 11, 1216. [Google Scholar] [CrossRef]
- Khadka, R.B.; Uphoff, N. Effects of Trichoderma seedling treatment with system of rice intensification management and with conventional management of transplanted rice. PeerJ 2019, 7, 5877. [Google Scholar] [CrossRef] [Green Version]
- Arun, K.D.; Sabarinathan, K.G.; Gomathy, M.; Kannan, R.; Balachandar, D. Mitigation of drought stress in rice crop with plant growth-promoting abiotic stress-tolerant rice phyllosphere bacteria. J. Basic Microbiol. 2020, 60, 768–786. [Google Scholar] [CrossRef]
- Alamgir, A.N.M. Phytoconstituents-Active and Inert Constituents, Metabolic Pathways, Chemistry and Application of Phytoconstituents, Primary Metabolic Products, and Bioactive Compounds of Primary Metabolic Origin. In Therapeutic Use of Medicinal Plants and Their Extracts; Rainsford, K.D., Ed.; Progress in Drug Research; Springer: Cham, Switzerland, 2018; Volume 74, pp. 25–164. [Google Scholar]
- Shukla, N.; Awasthi, R.P.; Rawat, L.; Kumar, J. Biochemical and physiological responses of rice (Oryza sativa L.) as influenced by Trichoderma harzianum under drought stress. Plant Physiol. Biochem. 2012, 54, 78–88. [Google Scholar] [CrossRef]
- Etesami, H.; Maheshwari, D.K. Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: Action mechanisms and future prospects. Ecotoxicol. Environ. Saf. 2018, 156, 225–246. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Reckling, M.; Wirth, S. Biochar-based Bradyrhizobium inoculum improves growth of lupin (Lupinus angustifolius L.) under drought stress. Eur. J. Soil Biol. 2017, 78, 38–42. [Google Scholar] [CrossRef]
- Ngumbi, E.; Kloepper, J. Bacterial-mediated drought tolerance: Current and future prospects. Appl. Soil Ecol. 2016, 105, 109–125. [Google Scholar] [CrossRef]
- Enebe, M.C.; Babalola, O.O. The influence of plant growth-promoting rhizobacteria in plant tolerance to abiotic stress: A survival strategy. Appl. Microbiol. Biotechnol. 2018, 102, 7821–7835. [Google Scholar] [CrossRef] [Green Version]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S.; Boyce, A.N. Role of plant growth promoting rhizobacteria in agricultural sustainability-a review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef] [PubMed]
- Chandra, D.; Srivastava, R.; Gupta, V.V.S.R.; Franco, C.M.M.; Sharma, A.K. Evaluation of ACC-deaminase-producing rhizobacteria to alleviate water-stress impacts in wheat (Triticum aestivum L.) plants. Can. J. Microbiol. 2019, 65, 387–403. [Google Scholar] [CrossRef]
- Zafar-Ul-Hye, M.; Danish, S.; Abbas, M.; Ahmad, M.; Munir, T.M. ACC deaminase producing PGPR Bacillus amyloliquefaciens and Agrobacterium fabrum along with biochar improve wheat productivity under drought stress. Agronomy 2019, 9, 343. [Google Scholar] [CrossRef] [Green Version]
- Naseem, H.; Ahsan, M.; Shahid, M.A.; Khan, N. Exopolysaccharides producing rhizobacteria and their role in plant growth and drought tolerance. J. Basic Microbiol. 2018, 58, 1009–1022. [Google Scholar] [CrossRef]
- Sharifi, R.; Ryu, C.M. Revisiting bacterial volatile-mediated plant growth promotion: Lessons from the past and objectives for the future. Ann. Bot. 2018, 122, 349–358. [Google Scholar] [CrossRef] [Green Version]
- El-Daim, I.A.A.; Bejai, S.; Meijer, J. Bacillus velezensis 5113 induced metabolic and molecular reprogramming during abiotic stress tolerance in wheat. Sci. Rep. 2019, 9, 16282. [Google Scholar] [CrossRef] [Green Version]
- Goswami, M.; Deka, S. Plant growth-promoting rhizobacteria- alleviators of abiotic stresses in soil: A review. Pedosphere 2020, 30, 40–61. [Google Scholar] [CrossRef]
- Bano, Q.; Ilyas, N.; Bano, A.; Zafar, N.; Akram, A.; Hassan, F. Effect of Azospirillum inoculation on maize (Zea mays L.) under drought stress. Pak J. Bot. 2013, 45, 13–20. [Google Scholar]
- Vardharajula, S.; Zulfikar Ali, S.; Grover, M.; Reddy, G.; Bandi, V. Drought-tolerant plant growth promoting Bacillus spp.: Effect on growth, osmolytes, and antioxidant status of maize under drought stress. J. Plant Int. 2011, 6, 1–14. [Google Scholar]
- Sandhya, V.; Ali, S.K.Z.; Minakshi, G.; Reddy, G.; Venkateswarlu, B. Alleviation of drought stress effects in sunflower seedlings by the exopolysaccharides producing Pseudomonas putida strain GAP-P45. Biol. Fertil. Soils. 2009, 46, 17–26. [Google Scholar] [CrossRef]
- Lim, J.H.; Kim, S.D. Induction of drought stress resistance by multi-functional PGPR Bacillus licheniformis K11 in pepper. Plant Pathol. J. 2013, 29, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, M.V.; Burity, H.A.; Martínez, C.R.; Chanway, C.P. Alleviation of drought stress in the common bean (Phaseolus vulgaris L.) by co-inoculation with Paenibacillus polymyxa and Rhizobium tropici. Appl. Soil Ecol. 2008, 40, 182–188. [Google Scholar] [CrossRef]
- Staudinger, C.; Mehmeti-Tershani, V.; Gil-Quintana, E.; Gonzalez, E.M.; Hofhansl, F.; Bachmann, G.; Wienkoop, S. Evidence for a rhizobia-induced drought stress response strategy in Medicago truncatula. J. Proteom. 2016, 136, 202–213. [Google Scholar] [CrossRef] [Green Version]
- Kasim, W.A.; Osman, M.E.; Omar, M.N.; Abd El-Daim, I.A.; Bejai, S.; Meijer, J. Control of drought stress in wheat using plant-growth-promoting bacteria. J. Plant Growth Regul. 2013, 32, 122–130. [Google Scholar] [CrossRef]
- Ma, Y.; Rajkumar, M.; Zhang, C.; Freitas, H. Inoculation of Brassica oxyrrhina with plant growth promoting bacteria for the improvement of heavy metal phytoremediation under drought conditions. J. Hazard. Mater. 2014, 320, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.; Lata, C.; Chauhan, P.S.; Nautiyal, C.S. Pseudomonas putida attunes morphophysiological, biochemical and molecular responses in Cicer arietinum L. during drought stress and recovery. Plant Physiol. Biochem. 2016, 99, 108–117. [Google Scholar] [CrossRef]
- Sherameti, I.; Tripathi, S.; Varma, A.; Oelmuller, R. The root-colonizing endophyte Pirifomospora indica confers drought tolerance in Arabidopsis by stimulating the expression of drought stress-related genes in leaves. Mol. Plant-Microbe Interact. 2008, 21, 799–807. [Google Scholar] [CrossRef] [Green Version]
- Hussain, M.B.; Zahir, Z.A.; Asghar, H.N.; Asgher, M. Can catalase and exopolysaccharides producing rhizobia ameliorate drought stress in wheat? Int. J. Agric. Biol. 2014, 16, 3–13. [Google Scholar]
- Behrooz, A.; Vahdati, K.; Rejali, F.; Lotfi, M.; Sarikhani, S.; Leslie, C. Arbuscular mycorrhiza and plant growth-promoting bacteria alleviate drought stress in walnut. HortScience 2019, 54, 1087–1092. [Google Scholar] [CrossRef] [Green Version]
- Morsy, M.; Cleckler, B.; Armuelles-Millican, H. Fungal endophytes promote tomato growth and enhance drought and salt tolerance. Plants 2020, 9, 877. [Google Scholar] [CrossRef] [PubMed]
- Liao, X.; Chen, J.; Guan, R.; Liu, J.; Sun, Q. Two arbuscular mycorrhizal fungi alleviates drought stress and improves plant growth in Cinnamomum migao seedlings. Mycobiology 2021, 49, 396–405. [Google Scholar] [CrossRef]
- Azad, K.; Kaminskyj, S.A. fungal endophyte strategy for mitigating the effect of salt and drought stress on plant growth. Symbiosis 2016, 68, 73–78. [Google Scholar] [CrossRef]
- Budak, H.; Kantar, M.; Yucebilgili Kurtoglu, K. Drought tolerance in modern and wild wheat. Sci. World. J. 2013, 4, 66–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oh, S.J.; Kim, Y.S.; Kwon, C.W.; Park, H.K.; Jeong, J.S.; Kim, J.K. Overexpression of the transcription factor AP37 in rice improves grain yield under drought conditions. Plant Physiol. 2013, 150, 1368–1379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valliyodan, B.; Nguyen, H.T. Understanding regulatory networks and engineering for enhanced drought tolerance in plants. Curr. Opin. Plant Biol. 2006, 9, 189–195. [Google Scholar]
- Trujillo, L.E.; Sotolongo, M.; Menendez, C.; Ochogavia, M.E.; Coll, Y.; Hernandez, I.; Hernandez, L. SodERF3, a novel sugarcane ethylene responsive factor (ERF), enhances salt and drought tolerance when over expressed in tobacco plants. Plant Cell Physiol. 2008, 49, 512–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Analyzed Parameters | Ecosystem Type | Mean Changes in All Ecosystems [%] | ||
|---|---|---|---|---|
| Forests | Grasslands | Shrublands | ||
| Litter and Root Biomass Response | ||||
| • Litter input | - | ↓ +++ | ↓ +++ | −8.7 |
| • Litter decomposition rate | ↓ +++ | - | ↓ +++ | −12.7 |
| • Litter C content | ↑ +++ | ↓ + | ↑ +++ | +23.4 |
| • Litter N content | ↑ +++ | ↓ + | - | +13.8 |
| • Root biomass | ↓ + | - | ↓ + | −6.7 |
| Soil pools and fluxes of C | ||||
| • SOC concentration | - | ↓ +++ | - | −3.3 |
| • DOC concentration | ↑ +++ | ↑ +++ | ↑ + | +59.2 |
| • Total CO2 efflux from soil | - | - | ↓ +++ | - |
| • Soil microbial respiration | ↑ +++ | ↑ +++ | - | +15.8 |
| Soil pool (concentration) and fluxes of N | ||||
| • Total N | - | - | ↑ +++ | - |
| • DON | ↑ + | - | ↑ + | +33.0 |
| • NH4+ | ↑ +++ | ↑ + | ↑ + | +52.5 |
| • NO3− | ↓ +++ | ↑ + | ↑ +++ | +16.0 |
| • SMN | ↑ +++ | ↑ + | ↑ + | +31.0 |
| • nitrification | ↓ + | - | ↓+++ | −13.8 |
| • N mineralization | ↓ + | - | ↓ + | −5.7 |
| • Soil microbial biomass | - | - | - | - |
| • MBC | - | - | - | +2.2 |
| • MBN | ↓ + | ↓ + | ↓ + | −10.4 |
| • MBC/MBN ratio | ↑ +++ | ↑ +++ | ↑ + | +29.7 |
| • F/B ratio | ↑ + | ↑ + | - | +15.6 |
| Soil enzyme activities | ||||
| • β-glucosidase | ↓ + | ↑ + | ↓ + | +3.5 |
| • urease | ↓ +++ | ↑ +++ | ↓ + | +12.7 |
| Drought Indices (Groups) | Average Values (%) | |
|---|---|---|
| Plasma membrane permeability (PMP) | ||
| • ROS | ↑ ++ | 65.7 |
| • MDA | ↑ + | 44.2 |
| • EL | ↑ ++ | 99.4 |
| Enzymatic antioxidants (EA) | ||
| • CAT | ↑ + | 28.8 |
| • POD | ↑ + | 28 |
| • SOD | ↑ + | 29.8 |
| • APX | ||
| • GR | — | |
| Non-enzymatic antioxidants (NEA) | ||
| • ABA | ↑ +++ | 126.6 |
| • AsA | ↑ + | 19.3 |
| • Proline | ↑ +++ | 136.8 |
| • Soluble sugar | ↑ +++ | 116.9 |
| • Car | — | |
| Plant growth (PG) | ||
| • Leaves | ↑ + | 17.1 |
| • Shoots | ↓ + | −20.5 |
| • Whole plants and roots | — | |
| • Dry weight | ↓ + | −28.8 |
| • Protein | — | |
| Photosynthesis (PS) | ||
| • Chl | ↓ + | −23.9 |
| • Fv/Fm | ↓ + | −13.1 |
| • qP | ↓ + | −26.4 |
| Variables | Sorghum | Barley |
|---|---|---|
| Total DEGs | 300 genes (2% orthologous with one of maize) | 2065 genes (7.2% orthologous with one of rice) |
| DEGs associated genes | Alkaloid biosynthesis, plant hormone signal transduction, MAPK signalling pathway, response to abiotic stimulus, and carbon metabolism. | |
| Transporter genes | Transmembrane transporter activity genes-SPX (Sb06g025950) and MS channel gene (Sb10g006710) | ABC transporter system- Contig18416_at and Contig13030_s_at |
| Shared DEGs | With maize- ASR protein | With rice- biosynthesis of secondary metabolites |
| Variables | Maize | Rice |
|---|---|---|
| Total DEGs | 4915 | 7291 |
| Upregulated genes | 2532 | 3491 |
| Downregulated genes | 2383 | 3800 |
| Highly upregulated identified DEGs | Probesets related to Cox family, fasciclin-like arabinogalactan proteins Three genes encoding for Di19 drought-induced 19 | RAB16B and RAB21 genes |
| Most highly downregulated identified DEGs | Histone H3-like proteins | PMEI-like and PEAMT2 genes |
| Stress tolerance genes | Heat shock proteins | LEA, HSP70, WSI76, and DREB1C |
| Predictive accuracy | 97.22% | 98.72% |
| 1. Gene Ontology Enrichment Analysis in Each Species: | ||
| DEGs | Small molecule metabolic process Response to chemical Carbohydrate metabolic process Organic acid metabolic process | Plant hormone signal transduction |
| Upregulated DEGs | Response to osmotic stress | Response to temperature stimulus Response to salt stress Response to osmotic stress |
| Downregulated DEGs | Photosynthesis Cofactor metabolic process | Photosynthesis Light reaction |
| GO terms: | ||
| Biological processes | 47 DEGs | Photosynthesis Small molecule metabolic process Oxidation-reduction process Response to abiotic stimulus 30 DEGs |
| Species-specific enriched biological processes | 34% | 25% |
| Molecular function | Cation binding Metal ion binding Antioxidant activity 8 DEGs | Oxidoreductase activity Catalytic activity 8 DEGs |
| Cellular component terms | DNA packaging complex Nucleosome Thylakoid 28 DEGs | Chloroplast Plastid 31 DEGs |
| Common biological processes | Metabolic process and upregulated response to stress (41% common) | |
| 2. Pathway Enrichment: | Enriched with metabolic pathways Carbon metabolism-related terms Photosynthesis Biosynthesis of secondary metabolites pathways | 13 KEGG pathways Downregulated photosynthesis pathway Carbon fixation in photosynthesis genes Expression of hormone signal transduction |
| Upregulated metabolic pathways and genes | Asparagine synthetase, acyl-CoA oxidase and peroxidases, with 1 and 9 unique pathways in maize and rice. | |
| 3. Identification of Consensus Modules: | ||
| Genes related to response to water deprivation and small molecule metabolic process | BP-10, KEGG-5 | |
| Cell wall organization and cell cycle | BP- 18, KEGG- 3 | |
| Photosynthesis | BP- 6, KEGG- 6 | |
| Biogenesis and biosynthesis | BP- 6, KEGG- 2 | |
| 4. Identification of Hub Genes: | Mainly enriched in pyrimidine metabolism | |
| Cell wall organization and cell cycle | FOR1 and PV72 | |
| Biogenesis and biosynthesis | PDHE1-A and HyPRP18 | |
| Genes related to response to water deprivation and small molecule metabolic process | Protein of unknown function DUF676 and PDX1 | |
| Photosynthesis | Ankyrin-like protein and UBC37 | |
| 5. Identification of Differential Co-expression Modules: | ||
| Photosynthesis and response to cytokinin | MF- 5, BP- 8, KEGG- 3 | |
| Organic acid catabolic process | MF- 2, BP- 17, KEGG- 6 | |
| Response to stress | MF- 2, BP- 9, KEGG- 1 | |
| Cell wall organization | MF- 8, BP- 30, KEGG- 3 | |
| Alanine, aspartate, glutamate metabolism (KEGG: 00250) enriched pathways among genes | MF- 1, KEGG- 1 | |
| 6. Co-localization of DEGs with QTL Intervals: | ||
| DEGs localized within QTLs regions | 801 DEGs | 1724 DEGs |
| Drought tolerance | 141 (2.8%) | 122 (1.6%) |
| Photosynthetic gas exchange | 444 (10.5%) | 139 (1.9%) |
| d13C | 59 (1.2%) | 105 (1.4%) |
| Root characteristics traits | 157 (3.19%) | 1358 (18.6%) |
| Microorganisms | Plants | Protection/Effect/Mechanisms | References |
|---|---|---|---|
| Azospirillum lipoferum | Maize (Zea mays. L) | Improves growth of the plant, increase in amino acid contents, sugar accumulation, and Pro production. | [165] |
| Bacillus spp. | Maize (Zea mays. L) | Improves intake of soluble sugar, amino acids, and Pro reduces loss of electrolytes and activity of catalase and glutathione PER enzymes. | [166] |
| Pseudomonas putida strain GAP-P45 | Sunflower (var. Sunbred Helianthus annuus L.) | Improved uptake of nutrients in plant and increased growth of the plant. | [167] |
| Bacillus licheformis strain K11 | Pepper (Capsicum annum) | Promotes growth of pepper, produces ACC deaminase, and prevents phytophthora blight. | [168] |
| Rhizobium tropici and Paenibacillus polymyxa | Bean (Phaseolus vulgaris) | Improved growth, nodulation, and nitrogen content. | [169] |
| Sinorhizobium medicae | Barrel medic (Medicago truncatula) | Delay in drought-induced leaf senescence, increase in potassium, drought-responsive proteins, and osmolyte production. | [170] |
| Bacillus amyloliquefaciens 5113 and Azospirillum brasilense NO40 | Wheat (Triticum aestivum L.) | Improves plant growth, enhances enzyme activities (mono-dehydroascorbate reductase (MDHAR), glutathione reductase (GR), and dehydroascorbate reductase (DHAR) and APX), lower antioxidant enzyme activities, and increases photosynthesis. | [171] |
| Pseudomonas libanensis TR1 and Pseudomonas reactans Ph3R3 | Smooth-stem turnip (Brassica oxyrrhina) | Enhances plant growth, leaf relative water content (RWC), resistance to heavy metals and antibiotics, increased chlorophyll content, and decrease in malondialdehyde content. | [172] |
| Pseudomonas putida MTCC5279 (RA) | Chickpea (Cicer arietinum L.) | Improves plant growth, nodule formation, low antioxidant enzymes, and increases biochemical responses. | [173] |
| Piriformospora indica | Thale cress (Arabidopsis thaliana) | Expression of stress-related genes. | [174] |
| Rhizobium leguminosarum (LR-30), Mesorhizobium ciceri (CR-30 and CR39), and Rhizobium phaseoli (MR-2) | Wheat (Triticum aestivum L.) | Improved root colonization, nutrient or water holding capacity of the rhizosphere, Improved drought tolerance index of the wheat seedlings, enhanced the root or shoot lengths and fresh or dry biomass of the seedlings, production of phytohormones (IAA), exopolysaccharides or catalase, osmolytes and antioxidants in the rhizosphere. | [175] |
| Azospirillum sp. | Wheat (Triticum aestivum L.) | Plant growth enhancement. | [42] |
| Trichoderma harzianum | Rice (Oryza sativa L.) | Delay to wilt, drought-induced changes (stomatal conductance, photosynthesis, and leaf discoloration), promote plant growth, increase in Pro content, reduction in H2O2 content, and increase in total phenolics. | [153] |
| Glomus etunicatum | English walnut (Juglans regia) | Improve height, fresh weight, and the number of leaves of the walnut plant increased the biosynthesis of some metabolites, including soluble sugar and Pro, total phenolic content, peroxidase activity, and starch content as well as peroxidase enzyme activity. | [176] |
| Ampelomyces sp. | Tomato (Solanum lycopersicum var. Better Boy) | Enhancement of plant growth, fruit yield, drought tolerance and resistance to pathogens. | [177] |
| Glomus lamellosum and Glomus etunicatum | Cinnamon (Cinnamomum migao) | Improvement in seedling growth, higher POD and CAT activity, decrease in sugar and osmoreceptor content, reduction of accumulation of MDA, and enhancement of water-use efficiency in the plant. | [178] |
| Alternaria sp. and Trichoderma harzianum | Tomato (Solanum lycopersicum var. Rutger) | Improvement in root and shoot biomass, enhancement of water-use efficiency, and better photosynthetic efficiency. | [179] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bogati, K.; Walczak, M. The Impact of Drought Stress on Soil Microbial Community, Enzyme Activities and Plants. Agronomy 2022, 12, 189. https://doi.org/10.3390/agronomy12010189
Bogati K, Walczak M. The Impact of Drought Stress on Soil Microbial Community, Enzyme Activities and Plants. Agronomy. 2022; 12(1):189. https://doi.org/10.3390/agronomy12010189
Chicago/Turabian StyleBogati, Kalisa, and Maciej Walczak. 2022. "The Impact of Drought Stress on Soil Microbial Community, Enzyme Activities and Plants" Agronomy 12, no. 1: 189. https://doi.org/10.3390/agronomy12010189
APA StyleBogati, K., & Walczak, M. (2022). The Impact of Drought Stress on Soil Microbial Community, Enzyme Activities and Plants. Agronomy, 12(1), 189. https://doi.org/10.3390/agronomy12010189





