Evaluation of the Tolerance Ability of Wheat Genotypes to Drought Stress: Dissection through Culm-Reserves Contribution and Grain Filling Physiology
Abstract
1. Introduction
2. Materials and Methods
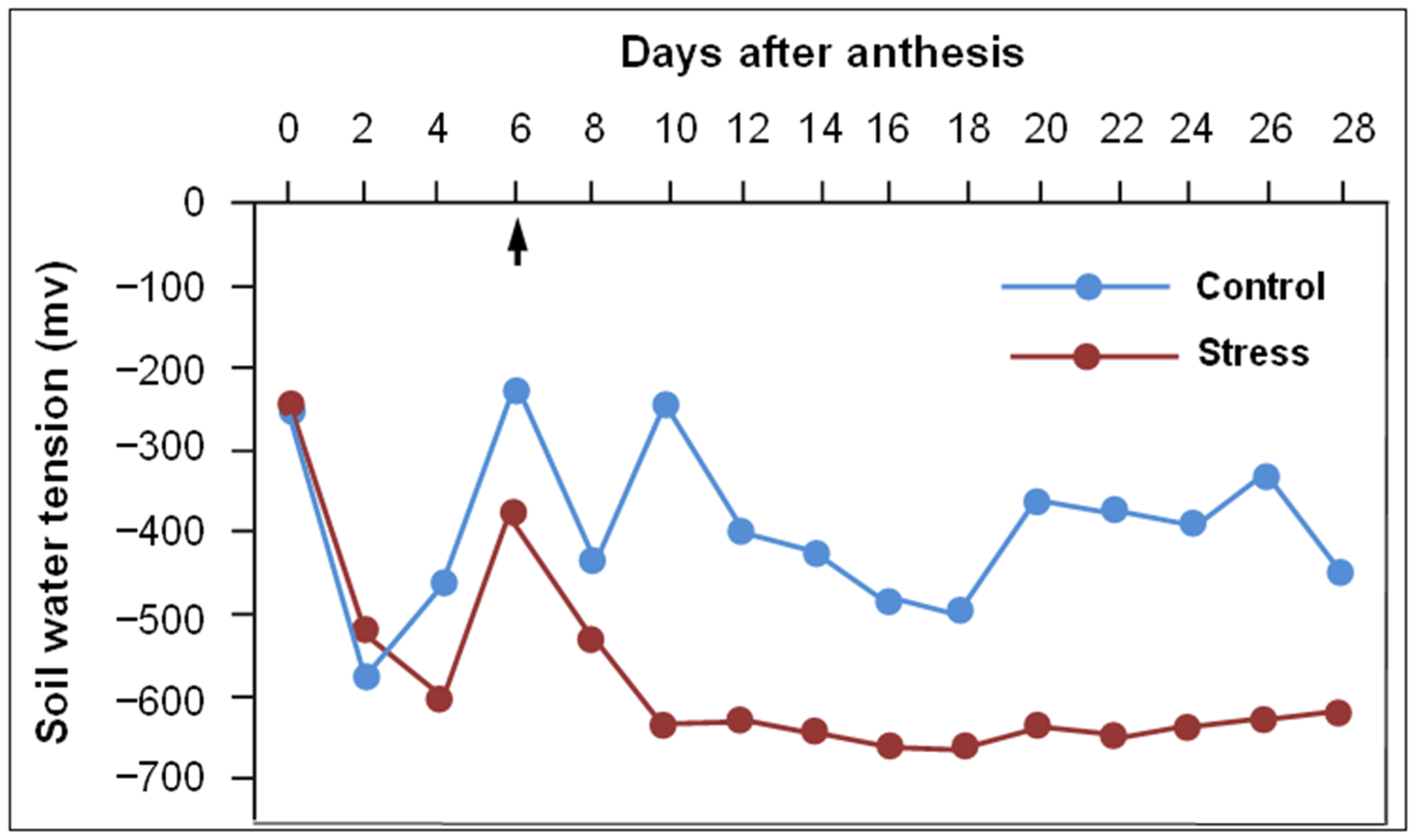
2.1. Experimentation
2.2. Sampling and Data Recoding
2.3. Estimation of Phenological Traits
2.4. Estimation of Culm WSCs
2.5. Statistical Analysis
3. Results
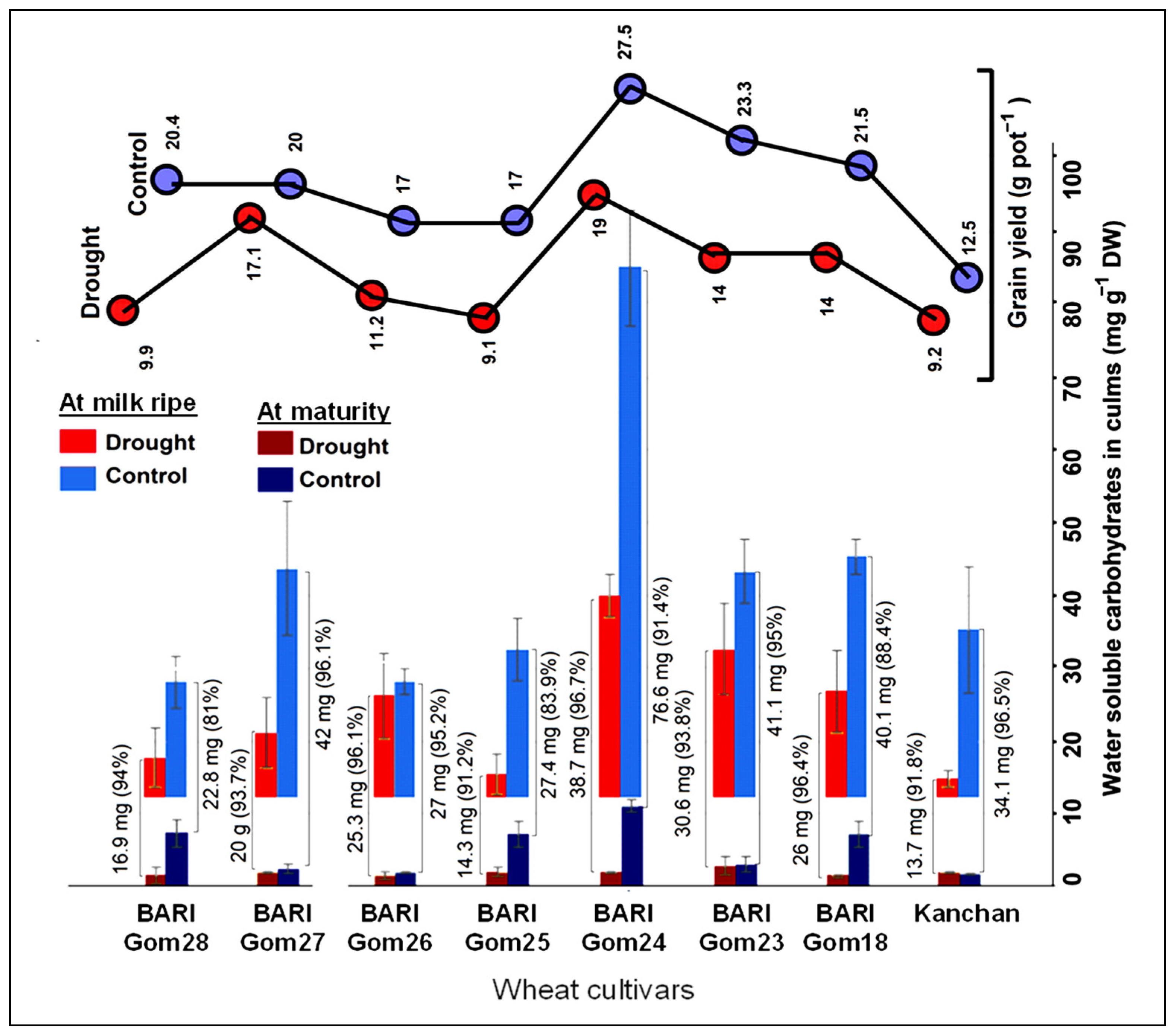
3.1. Yield and Its Components
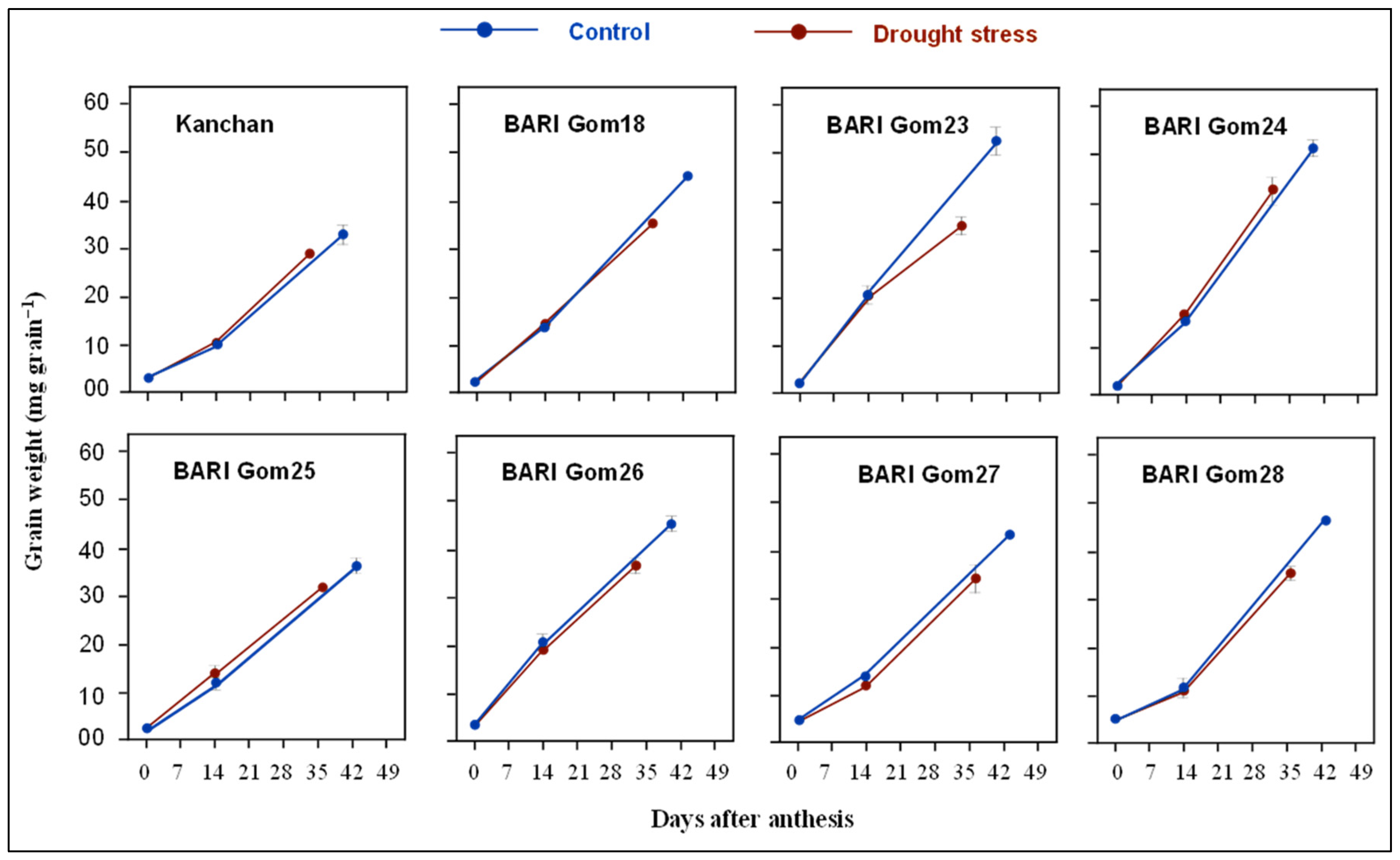
3.2. Grain Growth
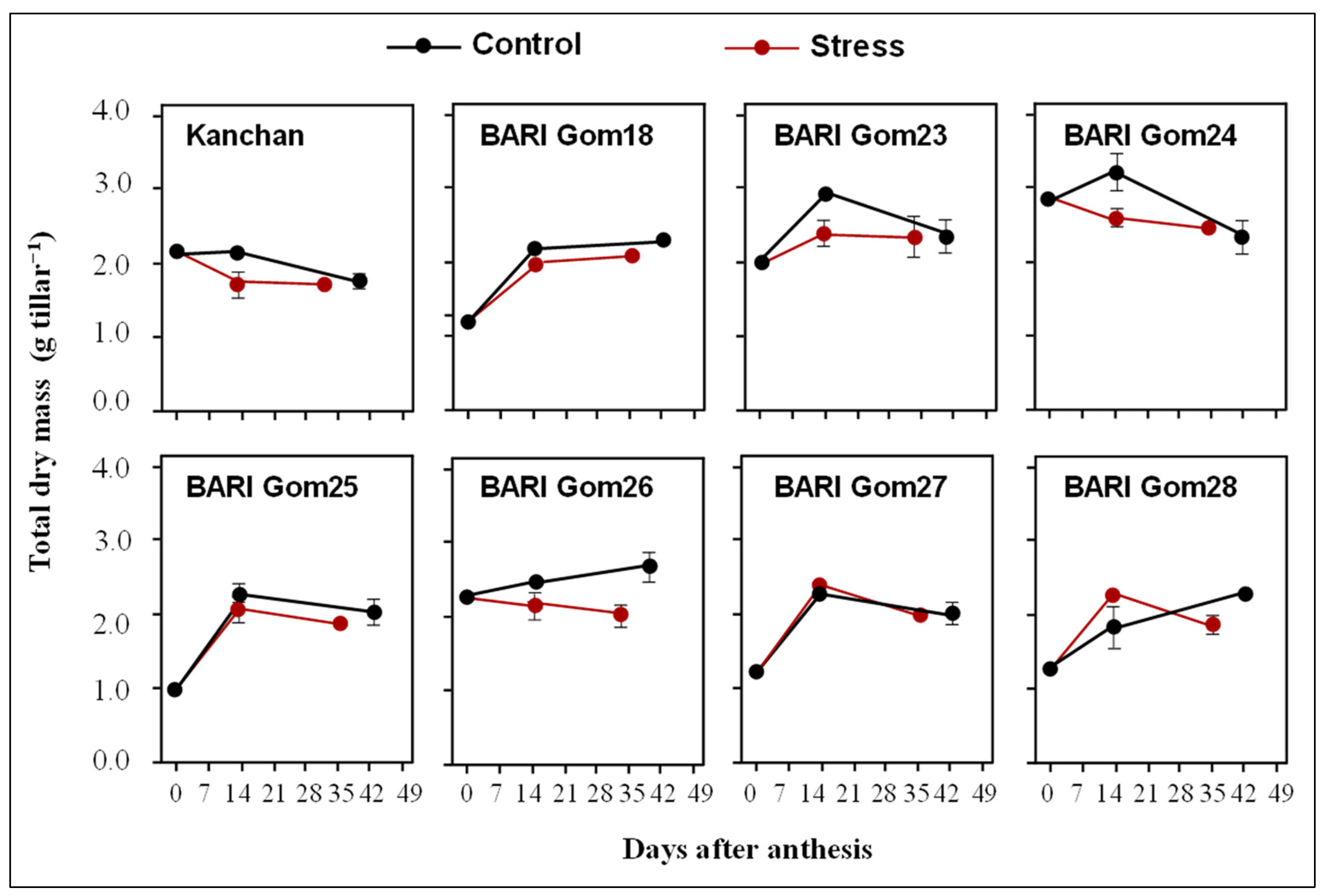
3.3. Changes in Total Dry Mass
3.4. Phenological Characters
3.5. Culm Reserves
3.6. Clustering of Wheat Cultivars into Different Drought Tolerance Groups
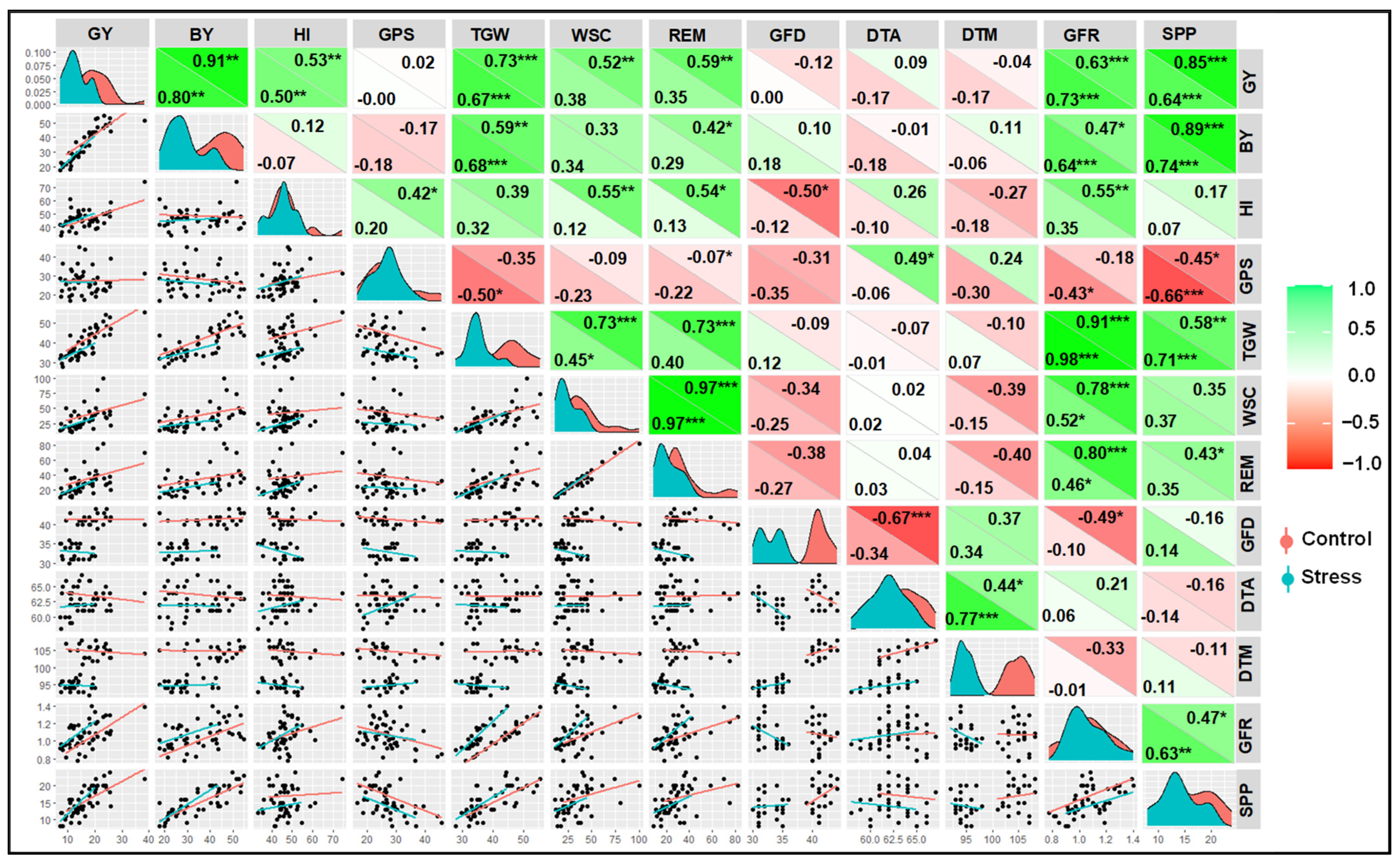
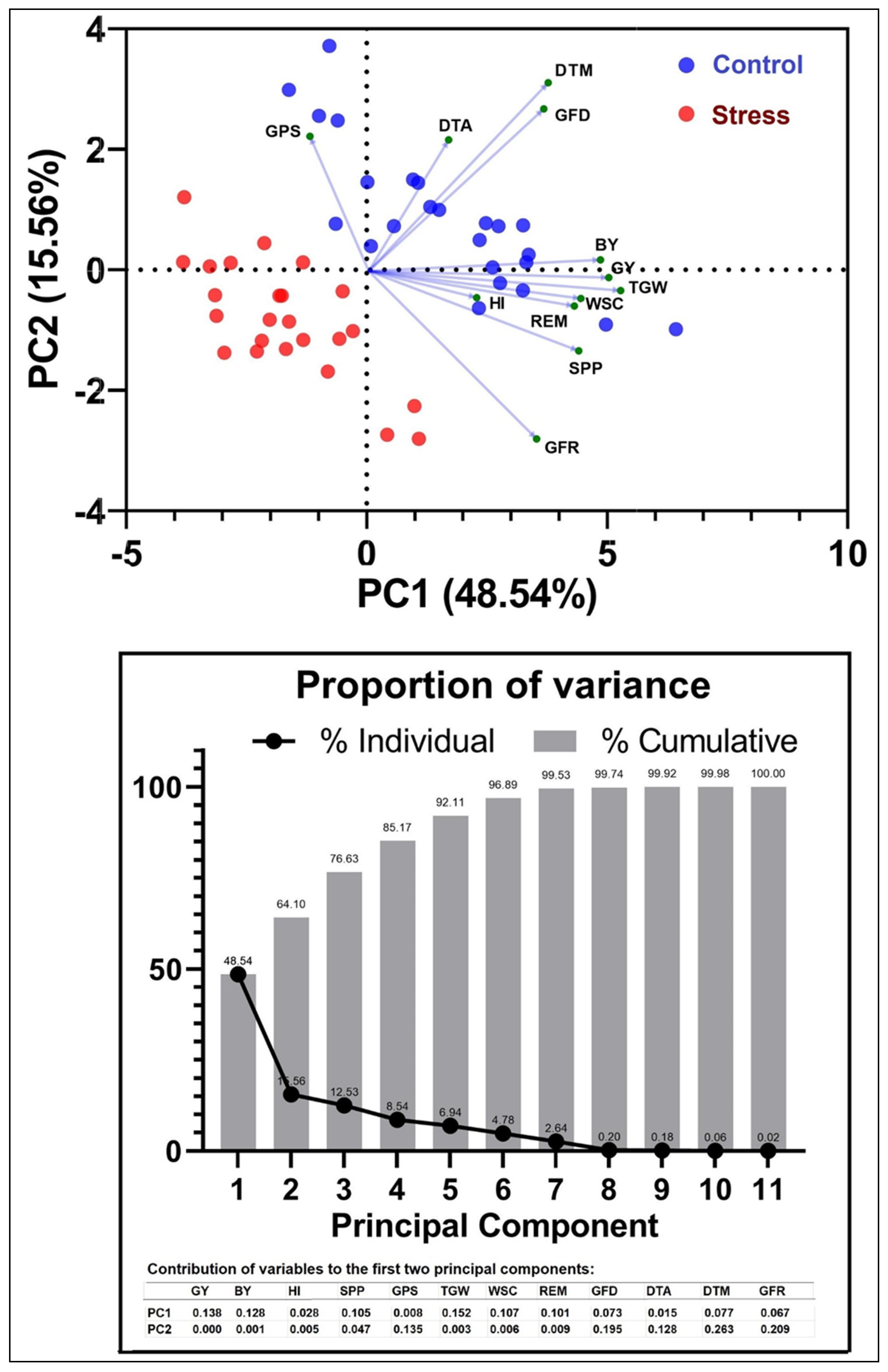
3.7. Correlation and PCA between Yield and Other Parameters
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BARI | Bangladesh Agricultural Research Institute |
| BY | Biomass yield |
| CRD | Completely randomized design |
| DAA | Days after anthesis |
| DTA | Days to anthesis |
| DTM | Days to maturity |
| DW | Dry weight |
| GFD | Grain filling duration |
| GFR | Grain filling rate |
| GPS | Grain per spike |
| GY | Grain yield |
| HI | Harvest index |
| IQR | Interquartile range |
| PCA | Principal component analysis |
| PC | Principal component |
| Q1 | First quartile (25th Percentile) |
| Q3 | Third quartile (75th Percentile) |
| SPP | Spike per pot |
| TDM | Total dry mass |
| TGW | Thousand grain weight |
| WSCs | Water-soluble carbohydrates |
References
- Singh, N.P.; Pal, P.K.; Vaishali, S.K. Morpho-physiological characterization of Indian wheat genotypes and their evaluation under drought condition. Afr. J. Biotechnol. 2014, 13, 20. [Google Scholar]
- Bhusal, N.; Lee, M.; Han, A.R.; Kim, H.S. Responses to drought stress in Prunus sargentii and Larix kaempferi seedlings using morphological and physiological parameters. For. Ecol. Manag. 2020, 465, 118099. [Google Scholar] [CrossRef]
- Olivares-Villegas, J.J.; Reynolds, M.P.; McDonald, G.K. Drought-adaptive attributes in the Seri/Babax hexaploid wheat population. Funct. Plant Biol. 2007, 34, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Mohi-Ud-Din, M.; Hossain, M.A.; Rohman, M.M.; Uddin, M.N.; Haque, M.S.; Ahmed, J.U.; Hossain, A.; Hassan, M.M.; Mostofa, M.G. Multivariate Analysis of Morpho-Physiological Traits Reveals Differential Drought Tolerance Potential of Bread Wheat Genotypes at the Seedling Stage. Plants 2021, 10, 879. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Shiran, B.; Wan, J.; Lewis, D.C.; Jenkins, C.L.D.; Condon, A.; Richards, R.A.; Dolferus, R. Importance of pre-anthesis anther sink strength for maintenance of grain number during reproductive stage water stress in wheat. Plant Cell Environ. 2010, 33, 926–942. [Google Scholar] [CrossRef]
- Wang, X.; Cai, J.; Liu, F.; Dai, T.; Cao, W.; Wollenweber, B.; Jiang, D. Multiple heat priming enhances thermo-tolerance to a later high temperature stress via improving subcellular antioxidant activities in wheat seedlings. Plant Physiol. Biochem. 2014, 74, 185–192. [Google Scholar] [CrossRef]
- Abid, M.; Ali, S.; Qi, L.K.; Zahoor, R.; Tian, Z.; Jiang, D.; Snider, J.L.; Dai, T. Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Sci. Rep. 2018, 8, 1–15. [Google Scholar] [CrossRef]
- Singh, B.; Jenner, C. Factors Controlling Endosperm Cell Number and Grain Dry Weight in Wheat: Effects of Shading on Intact Plants and of Variation in Nutritional Supply to Detached, Cultured Ears. Funct. Plant Biol. 1984, 11, 151–163. [Google Scholar] [CrossRef]
- Takahashi, T.; Tsuchihashi, N.; Nakaseko, K. Grain Filling Mechanisms in Spring Wheat. I. Grain filling phases according to the development of plant organs. Jpn. J. Crop. Sci. 1993, 62, 560–564. [Google Scholar] [CrossRef]
- Ehdaie, B.; Alloush, G.A.; Madore, M.A.; Waines, J.G. Genotypic Variation for Stem Reserves and Mobilization in Wheat: I. Postanthesis Changes in Internode Dry Matter. Crop. Sci. 2006, 46, 735–746. [Google Scholar] [CrossRef]
- Ovenden, B.; Milgate, A.; Lisle, C.; Wade, L.; Rebetzke, G.; Holland, J.B. Selection for water-soluble carbohydrate accumulation and investigation of genetic × environment interactions in an elite wheat breeding population. Theor. Appl. Genet. 2017, 130, 2445–2461. [Google Scholar] [CrossRef] [PubMed]
- Tahir, I.S.A.; Nakata, N. Remobilization of Nitrogen and Carbohydrate from Stems of Bread Wheat in Response to Heat Stress during Grain Filling. J. Agron. Crop. Sci. 2005, 191, 106–115. [Google Scholar] [CrossRef]
- Ehdaie, B.; Alloush, G.; Waines, J. Genotypic variation in linear rate of grain growth and contribution of stem reserves to grain yield in wheat. Field Crop. Res. 2008, 106, 34–43. [Google Scholar] [CrossRef]
- Hossain, M.A.; Araki, H.; Takahashi, T. Poor grain filling induced by water logging is similar to that in abnormal early ripening in wheat grown in Western Japan. Field Crops Res. 2011, 123, 100–108. [Google Scholar] [CrossRef]
- Wardlaw, I.F.; Willenbrink, J. Mobilization of fructan reserves and changes in enzyme activities in wheat stems correlate with water stress during kernel filling. New Phytol. 2000, 148, 413–422. [Google Scholar] [CrossRef]
- Rawson, H.; Evans, L. The contribution of stem reserves to grain development in a range of wheat cultivars of different height. Aust. J. Agric. Res. 1971, 22, 851–863. [Google Scholar] [CrossRef]
- Gent, M.P.N. Photosynthate Reserves during Grain Filling in Winter Wheat. Agron. J. 1994, 86, 159–167. [Google Scholar] [CrossRef]
- Jiang, D.; Fan, X.; Dai, T.; Cao, W. Nitrogen fertiliser rate and post-anthesis waterlogging effects on carbohydrate and nitrogen dynamics in wheat. Plant Soil 2008, 304, 301–314. [Google Scholar] [CrossRef]
- Hossain, A.; Takahashi, T.; Jinno, H.; Senju, K.; Kawata, Y.; Zhan, L.; Araki, H. Grain Filling Mechanisms in Two Wheat Cultivars, Haruyutaka and Daichinominori, grown in Western Japan and in Hokkaido. Plant Prod. Sci. 2010, 13, 156–163. [Google Scholar] [CrossRef]
- Hossain, M.A.; Takahashi, T.; Araki, H. Mechanisms and Causes of Poor Grain Filling in Wheat; Lambert Academic Publishing: Saarbrücken, Germany, 2012; p. 90. [Google Scholar]
- Araki, H.; Hamada, A.; Hossain, A.; Takahashi, T. Waterlogging at jointing and/or after anthesis in wheat induces early leaf senescence and impairs grain filling. Field Crop. Res. 2012, 137, 27–36. [Google Scholar] [CrossRef]
- Ruuska, S.A.; Rebetzke, G.J.; van Herwaarden, A.F.; Richards, R.A.; Fettell, N.A.; Tabe, L.; Jenkins, C.L.D. Genotypic varia-tion in water-soluble carbohydrate accumulation in wheat. Funct. Plant Biol. 2006, 33, 799–809. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, P.; Li, M.; Chang, L.; Cheng, H.; Chai, S.; Yang, D. Dynamic responses of accumulation and remobilization of water soluble carbohydrates in wheat stem to drought stress. Plant Physiol. Biochem. 2020, 155, 262–270. [Google Scholar] [CrossRef] [PubMed]
- Volaire, F.; Lelievre, F. Production, persistence, and water-soluble carbohydrate accumulation in 21 contrasting populations of Dactylis glomerata L. subjected to severe drought in the south of France. Aust. J. Agric. Res. 1997, 48, 933. [Google Scholar] [CrossRef]
- Foulkes, J.; Sylvester-Bradley, R.; Weightman, R.; Snape, J. Identifying physiological traits associated with improved drought resistance in winter wheat. Field Crop. Res. 2007, 103, 11–24. [Google Scholar] [CrossRef]
- Rana, M.R.; Karim, M.M.; Hassan, M.J.; Hossain, M.A.; Haque, M.A. Grain filling patterns of barley as affected by high temperature stress. J. Bangladesh Agric. Univ. 2017, 15, 174–181. [Google Scholar] [CrossRef]
- Karim, M.M.; Islam, M.A.; Rana, M.R.; Hossain, M.A.; Kader, M.A. Screening of barley genotypes for drought tolerance based on culm reserves contribution to grain yield. J. Bangladesh Agric. Univ. 2018, 16, 62–66. [Google Scholar] [CrossRef]
- Shekhar, I.; Paul, S. Rice and Wheat Crop Productivity in the Indo-Gangetic Plains of India: Changing Pattern of Growth and Future Strategies. Ind. J. Agric. Econ. 2012, 67, 238–252. [Google Scholar]
- Rokonujjman, M.; Kader, M.A.; Begum, S.A.; Sarker, A. Evaluating manurial value of bio-slurry for tomato cultivation in sub-tropical floodplain soil. J. South Pacific Agric. 2019, 22, 23–29. [Google Scholar]
- BMD (Bangladesh Meteorological Department). 2020. Available online: http://www.bmd.gov.bd/Document/climateofbangladesh.doc (accessed on 27 August 2020).
- Zadoks, J.C.; Chang, T.T.; Konzak, C.F. A decimal code for the growth stages of cereals. Weed Res. 1974, 14, 415–421. [Google Scholar] [CrossRef]
- Dias, A.S.; Lidon, F.C. Evaluation of grain filling rate and duration in bread and durum wheat under heat stress after anthesis. J. Agron. Crop Sci. 2009, 195, 137–147. [Google Scholar] [CrossRef]
- Yemm, E.W.; Willis, A.J. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J. 1954, 57, 508–514. [Google Scholar] [CrossRef]
- Asada, K. Production and scavenging of reactive oxygen species in chloro- plasts and their functions. Plant Physiol. 2006, 141, 391–396. [Google Scholar] [CrossRef]
- Altenbach, S.B. New insights into the effects of high temperature, drought and post-anthesis fertilizer on wheat grain development. J. Cereal Sci. 2012, 56, 39–50. [Google Scholar] [CrossRef]
- Yang, W.; Peng, S.; Dionisio-Sese, M.L.; Laza, R.C.; Visperas, R.M. Grain filling duration, a crucial determinant of genotypic variation of grain yield in field-grown tropical irrigated rice. Field Crop. Res. 2008, 105, 221–227. [Google Scholar] [CrossRef]
- Austin, R.B.; Morgan, C.L.; Ford, M.A.; Blackwell, R.D. Contributions to grain yield from pre-anthesis assimilation in tall and dwarf barley genotypes in two contrasting seasons. Ann. Bot. 1980, 45, 309–316. [Google Scholar] [CrossRef]
- Hossain, A.; Takahashi, T.; Zhang, L.; Nakatsukasa, M.; Kimura, K.; Kurashige, H.; Hirata, T.; Ariyoshi, M. Physiological Mechanisms of Poor Grain Growth in Abnormally Early Ripening Wheat Grown in West Japan. Plant Prod. Sci. 2009, 12, 278–284. [Google Scholar] [CrossRef]
- Islam, M.A.; Hossain, M.A.; Atikuzzamman, M.; Islam, M.S.; Razzak, M.A.; Sathi, M.A. Assessment of wheat genotypes based on culm reserves contribution to grain yield. Int. J. Adv. Res. Technol. 2018, 7, 1–6. [Google Scholar]
- Islam, M.A.; Fakir, M.S.A.; Hossain, M.A.; Sathi, M.A. Genotypic variation of wheat (Triticum aestivum L.) in grain filling and contribution of culm reserves to yield. Bangladesh J. Bot. 2021, 50, 51–59. [Google Scholar] [CrossRef]
- Zhang, J.; Chen, W.; Dell, B.; Vergauwen, R.; Zhang, X.; Mayer, J.E.; Ende, W.V.D. Wheat genotypic variation in dynamic fluxes of WSC components in different stem segments under drought during grain filling. Front. Plant Sci. 2015, 6, 624. [Google Scholar] [CrossRef]
- Goggin, D.E.; Setter, T.L. Fructosyltransferase activity and fructan accumulation during development in wheat exposed to terminal drought. Funct Plant Biol. 2004, 31, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.-L.; Jing, R.-L.; Chang, X.-P.; Li, W. Identification of Quantitative Trait loci and Environmental Interactions for Accumulation and Remobilization of Water-Soluble Carbohydrates in Wheat (Triticum aestivum L.) Stems. Genetics 2007, 176, 571–584. [Google Scholar] [CrossRef]
- Li, W.; Zhang, B.; Li, R.; Chang, X.; Jing, R. Favorable Alleles for Stem Water-Soluble Carbohydrates Identified by Association Analysis Contribute to Grain Weight under Drought Stress Conditions in Wheat. PLoS ONE 2015, 10, e0119438. [Google Scholar] [CrossRef] [PubMed]
- Nadia, K.; Chang, X.; Jing, R. Genetic Dissection of Stem Water-Soluble Carbohydrates and Agronomic Traits in Wheat under Different Water Regimes. J. Agric. Sci. 2017, 9, 42. [Google Scholar] [CrossRef]
- Khaleghi, A.; Naderi, R.; Brunetti, C.; Maserti, B.; Salami, S.A.; Babalar, M. Morphological, physiochemical and antioxidant responses of Maclura pomifera to drought stress. Sci. Rep. 2019, 9, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bhusal, N.; Lee, M.; Lee, H.; Adhikari, A.; Han, A.R.; Kim, H.S. Evaluation of morphological, physiological, and biochemical traits for assessing drought resistance in eleven tree species. Sci. Total. Environ. 2021, 779, 146466. [Google Scholar] [CrossRef] [PubMed]


| Cultivars | Drought Stress | Grain Yield (g pot−1) | Biomass Yield (g pot−1) | Harvest Index (%) | Spikes Pot −1 | Grains Spike−1 | 1000-Grain Weight (g) |
|---|---|---|---|---|---|---|---|
| Kanchan | Control | 12.46 | 26.70 d–f | 46.43 ab | 10.67 | 35.95 | 33.12 f |
| Stressed | 9.16 | 22.23 f | 41.00 ab | 10.33 | 30.52 | 29.76 f | |
| BARI Gom 18 | Control | 21.52 | 53.53 a | 40.17 ab | 21.67 | 21.79 | 45.78 a–c |
| Stressed | 14.02 | 28.69 c–f | 48.96 ab | 13.67 | 28.25 | 36.48 ef | |
| BARI Gom 23 | Control | 23.33 | 48.53 ab | 48.02 ab | 19.00 | 23.47 | 52.40 a |
| Stressed | 14.05 | 28.05 c–f | 49.99 ab | 14.67 | 27.45 | 35.16 ef | |
| BARI Gom 24 | Control | 27.46 | 48.34 ab | 56.20 a | 19.00 | 27.72 | 51.48 ab |
| Stressed | 19 | 39.73 a–e | 48.10 ab | 19.00 | 23.79 | 42.24 c–e | |
| BARI Gom 25 | Control | 17.02 | 37.29 a–f | 45.81 ab | 12.67 | 36.89 | 36.98 d–f |
| Stressed | 9.15 | 20.83 f | 43.72 ab | 10.33 | 27.89 | 31.99 f | |
| BARI Gom 26 | Control | 17.07 | 39.31 a–e | 43.53 ab | 16.00 | 23.98 | 44.98 bc |
| Stressed | 11.2 | 23.27 e–f | 48.46 ab | 11.33 | 28.33 | 35.67 ef | |
| BARI Gom 27 | Control | 19.96 | 44.21 a–c | 44.70 ab | 20.33 | 22.52 | 43.82 cd |
| Stressed | 17.14 | 40.11 a–d | 42.45 ab | 17.67 | 28.54 | 34.17 f | |
| BARI Gom 28 | Control | 20.36 | 36.81 b–f | 56.63 a | 16.00 | 27.44 | 46.42 a-c |
| Stressed | 9.88 | 26.94 d–f | 36.44 b | 14.67 | 19.28 | 34.75 f | |
| Significance | Cultivar (C) | 6.22 ** | 10.13 ** | 1.68 NS | 0.46 NS | 2.72 * | 24.78 ** |
| (F value) | Stress (S) | 39.25 ** | 69.62 ** | 3.01 NS | 0.43 NS | 0.21 NS | 181.4 ** |
| C × S | 0.77 NS | 2.88 * | 3.81 ** | 0.14 NS | 2.23 NS | 4.5 ** |
| Cultivars | Drought Stress | Grain Filling Duration (d) | Days to Anthesis (d) | Days to Maturity (d) | Grain Filling Rate (mg grain−1 d−1) | Grain Weight (mg Grain−1) |
|---|---|---|---|---|---|---|
| Kanchan | Control | 40.0 | 65.0 | 108.0 | 0.84 | 33.0 |
| Stressed | 31.3 | 64.3 | 99.0 | 0.93 | 29.24 | |
| BARI Gom 18 | Control | 43.3 | 63.0 | 107.3 | 1.36 | 56.61 |
| Stressed | 34.3 | 62.0 | 99.0 | 1.13 | 38.6 | |
| BARI Gom 23 | Control | 41.3 | 64.3 | 106.3 | 1.37 | 52.79 |
| Stressed | 31.3 | 62.0 | 100.6 | 1.12 | 35.13 | |
| BARI Gom 24 | Control | 39.6 | 63.6 | 109.3 | 1.29 | 51.23 |
| Stressed | 31.6 | 61.6 | 100.3 | 1.36 | 42.6 | |
| BARI Gom 25 | Control | 40.6 | 61.3 | 107.0 | 0.86 | 35.54 |
| Stressed | 34.3 | 59.6 | 101.0 | 0.92 | 31.5 | |
| BARI Gom 26 | Control | 40.3 | 66.3 | 110.0 | 1.16 | 45.15 |
| Stressed | 30.6 | 64.0 | 102.6 | 1.21 | 36.94 | |
| BARI Gom 27 | Control | 41.6 | 61.3 | 106.3 | 0.93 | 38.34 |
| Stressed | 34.6 | 62.0 | 99.3 | 0.99 | 34.32 | |
| BARI Gom 28 | Control | 43.0 | 61.3 | 107.6 | 1.15 | 46.21 |
| Stressed | 35.3 | 59.0 | 101.3 | 1.01 | 35.63 | |
| Significance | Cultivar (C) | 2.83 * | 6.67 ** | 1.86 NS | 5.8 ** | 5.4 ** |
| (F value) | Stress (S) | 172.18 ** | 8.88 ** | 58.04 ** | 0.51 NS | 25.77 ** |
| C × S | 0.5 NS | 0.5 NS | 0.64 NS | 1.01 NS | 1.23 NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Islam, M.A.; De, R.K.; Hossain, M.A.; Haque, M.S.; Uddin, M.N.; Fakir, M.S.A.; Kader, M.A.; Dessoky, E.S.; Attia, A.O.; El-Hallous, E.I.; et al. Evaluation of the Tolerance Ability of Wheat Genotypes to Drought Stress: Dissection through Culm-Reserves Contribution and Grain Filling Physiology. Agronomy 2021, 11, 1252. https://doi.org/10.3390/agronomy11061252
Islam MA, De RK, Hossain MA, Haque MS, Uddin MN, Fakir MSA, Kader MA, Dessoky ES, Attia AO, El-Hallous EI, et al. Evaluation of the Tolerance Ability of Wheat Genotypes to Drought Stress: Dissection through Culm-Reserves Contribution and Grain Filling Physiology. Agronomy. 2021; 11(6):1252. https://doi.org/10.3390/agronomy11061252
Chicago/Turabian StyleIslam, Md. Amirul, Rajib Kumar De, Md. Alamgir Hossain, Md. Sabibul Haque, Md. Nesar Uddin, Md. Solaiman Ali Fakir, Md. Abdul Kader, Eldessoky S. Dessoky, Attia O. Attia, Ehab I. El-Hallous, and et al. 2021. "Evaluation of the Tolerance Ability of Wheat Genotypes to Drought Stress: Dissection through Culm-Reserves Contribution and Grain Filling Physiology" Agronomy 11, no. 6: 1252. https://doi.org/10.3390/agronomy11061252
APA StyleIslam, M. A., De, R. K., Hossain, M. A., Haque, M. S., Uddin, M. N., Fakir, M. S. A., Kader, M. A., Dessoky, E. S., Attia, A. O., El-Hallous, E. I., & Hossain, A. (2021). Evaluation of the Tolerance Ability of Wheat Genotypes to Drought Stress: Dissection through Culm-Reserves Contribution and Grain Filling Physiology. Agronomy, 11(6), 1252. https://doi.org/10.3390/agronomy11061252












