Yield Enhancement of Biostimulants, Vitamin B12, and CoQ10 Compared to Inorganic Fertilizer in Radish
Abstract
1. Introduction
2. Materials and Methods
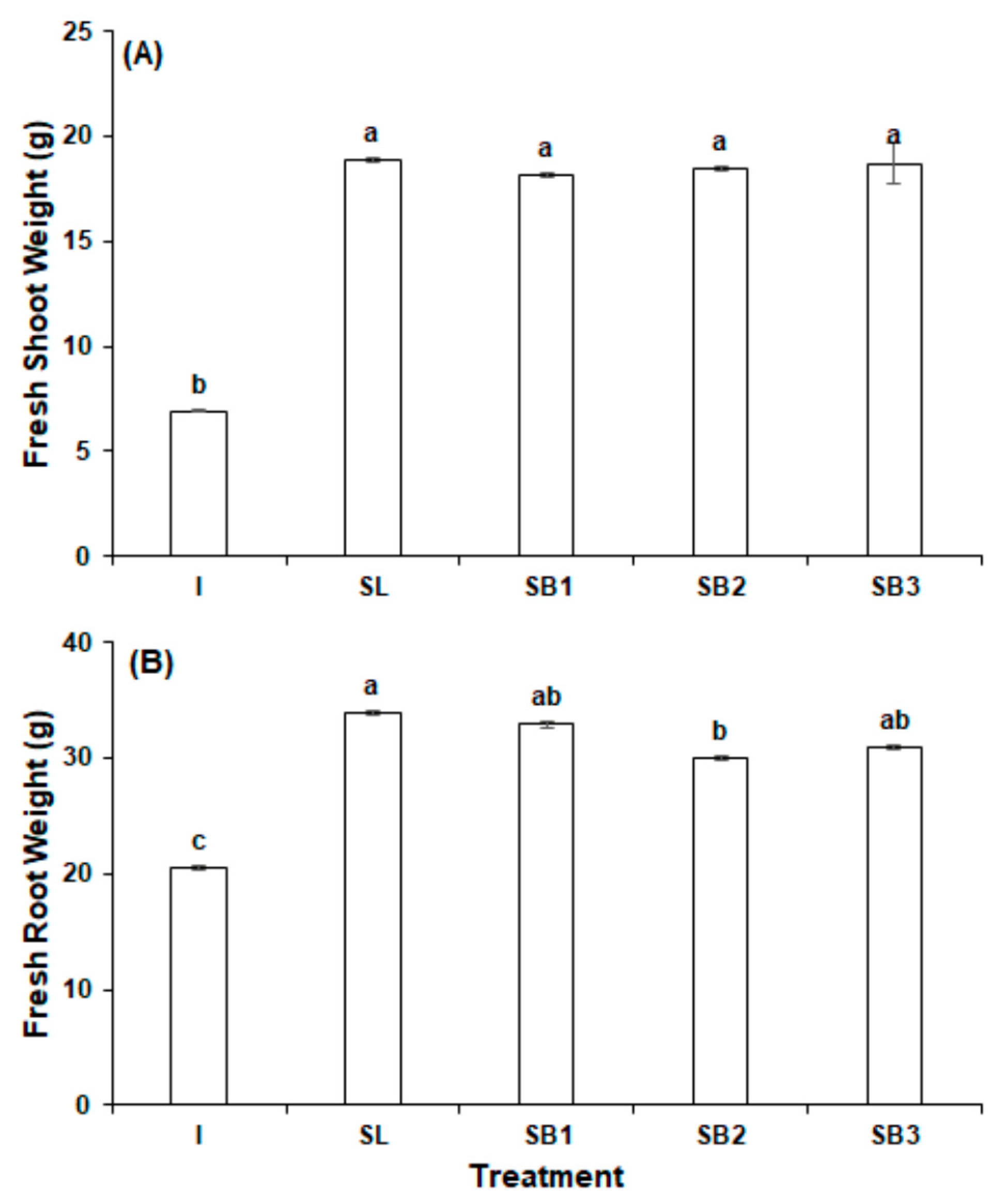
- Nutri-leaf inorganic fertilizer (1.0 g/L) (I)
- GPB Core (8.7 g/L) (SL)
- GPB Core (8.7 g/L) + 0.5 mg/L B12 (SB1)
- GPB Core (8.7 g/L) + 1.0 mg/L B12 (SB2)
- GPB Core (8.7 g/L) + 1.5 mg/L B12 (SB3)
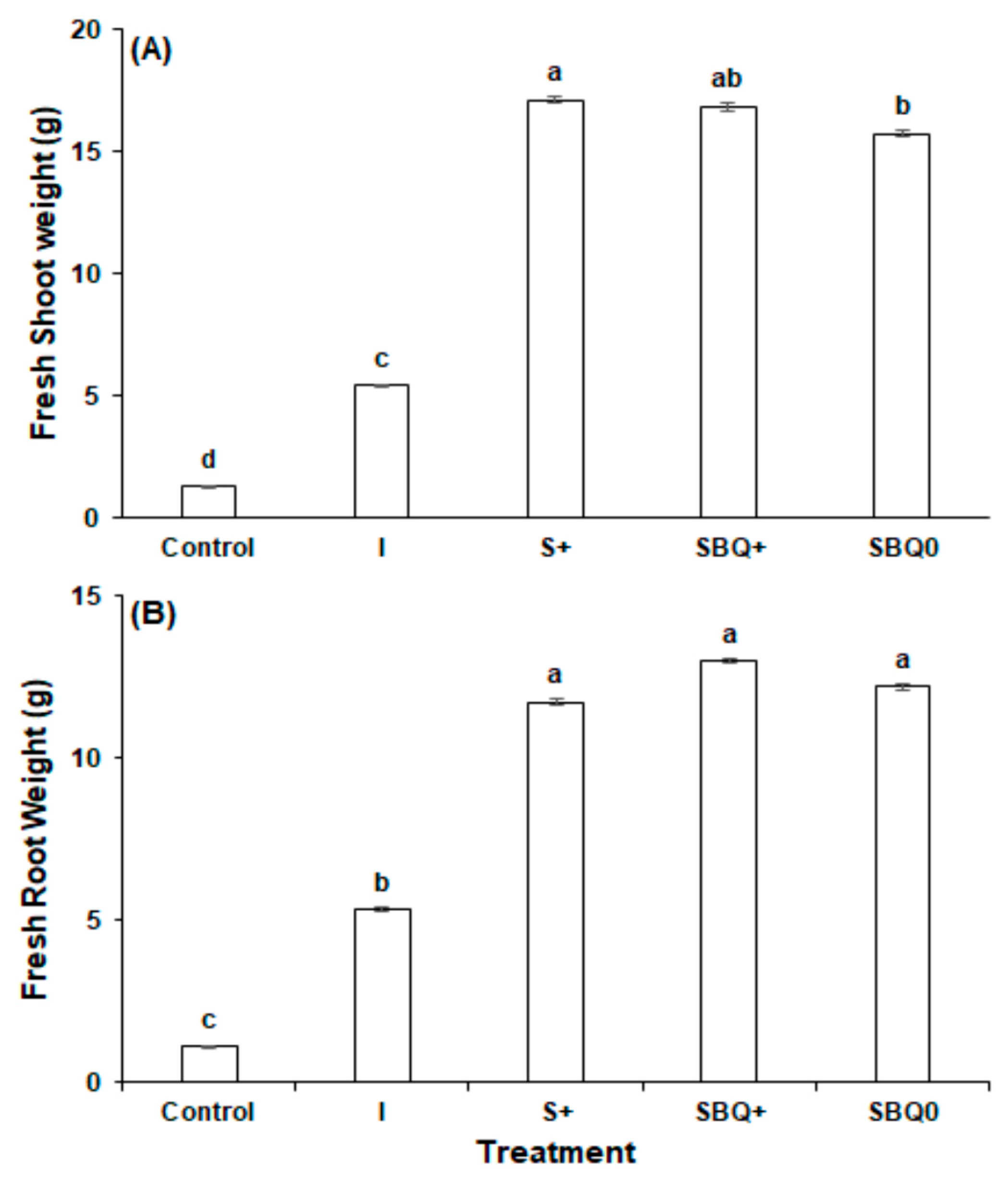
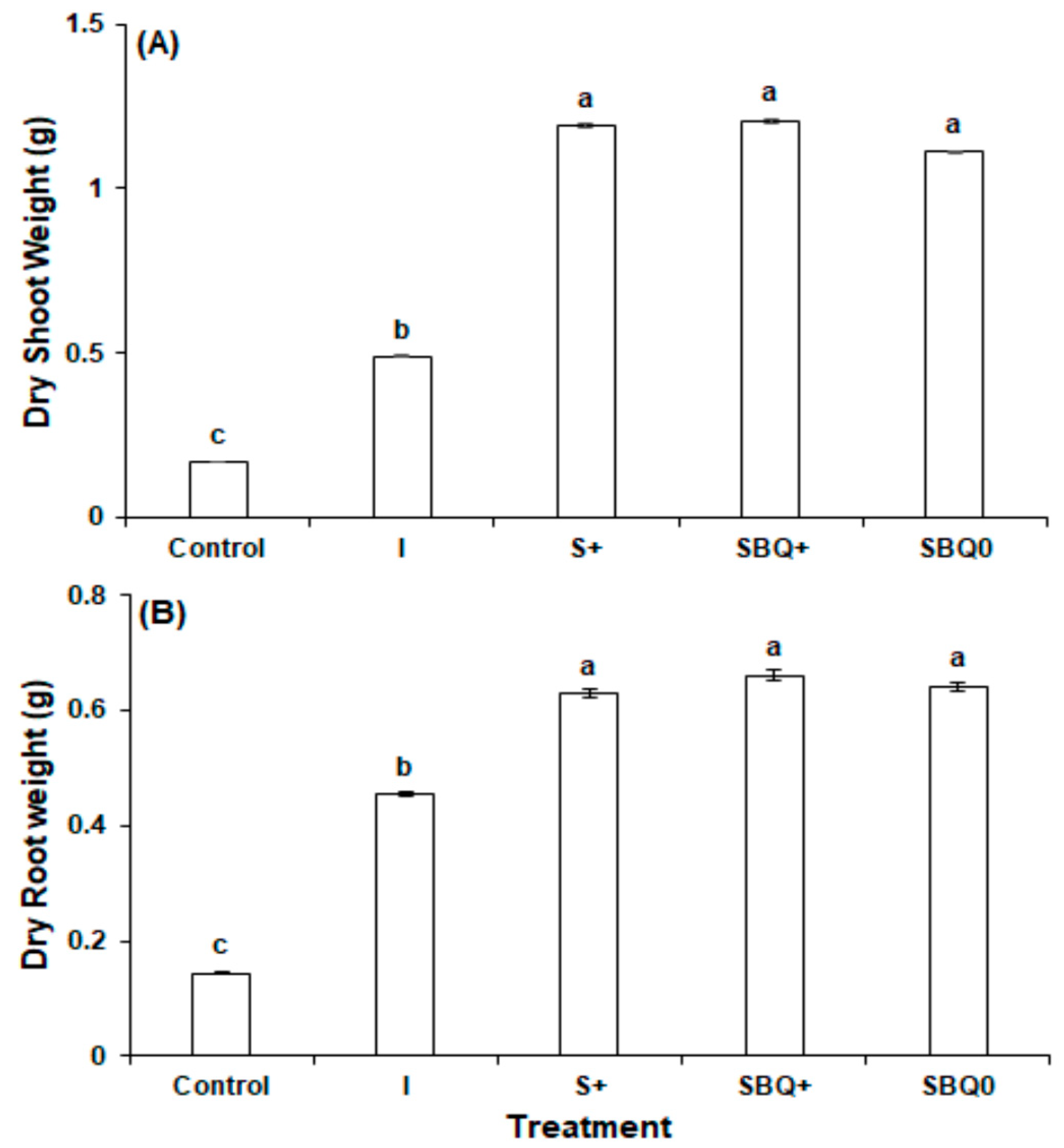
- Control
- Nutri-leaf inorganic fertilizer (1.0 g/L) (I)
- GPB Core (8.7 g/L) + 0.25 g/L Nutri-leaf (S+)
- GPB Core (8.7 g/L) + 1.5 mg/L B12 + 400 mg/L CoQ10 + 0.25 g/L Nutri-leaf (SBQ+)
- GPB Core (8.7 g/L) + 1.5 mg/L B12 + 400 mg/L CoQ10 (SBQ0)
3. Results
3.1. Experiment 1
3.2. Experiment 2
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Wojcieszek, J.; Jiménez-Lamana, J.; Bierła, K.; Ruzik, L.; Asztemborska, M.; Jarosz, M.; Szpunar, J. Uptake, translocation, size characterization and localization of cerium oxide nanoparticles in radish (Raphanus sativus L.). Sci. Total Environ. 2019, 683, 284–292. [Google Scholar] [CrossRef]
- Silvestre, W.P.; Pauletti, G.F.; Baldasso, C. Fodder radish (Raphanus sativus L.) seed cake as a feedstock for pyrolysis. Ind. Crops Prod. 2020, 154. [Google Scholar] [CrossRef]
- Musarika, S.; Atherton, C.E.; Gomersall, T.; Wells, M.J.; Kaduk, J.; Cumming, A.M.J.; Page, S.E.; Oechel, W.C.; Zona, D. Effect of water table management and elevated CO2 on radish productivity and on CH4 and CO2 fluxes from peatlands converted to agriculture. Sci. Total Environ. 2017, 584–585, 665–672. [Google Scholar] [CrossRef]
- Nishio, T. Economic and Academic Importance of Radish; Springer: Cham, Switzerland, 2017; pp. 1–10. [Google Scholar]
- Weil, R.; Kremen, A. Thinking across and beyond disciplines to make cover crops pay. J. Sci. Food Agric. 2007, 87, 551–557. [Google Scholar] [CrossRef]
- Rafiee, H.; Naghdi Badi, H.; Mehrafarin, A.; Qaderi, A.; Zarinpanjeh, N.; Sekara, A.; Zand, E. Application of plant biostimulants as new approach to improve the biological responses of medicinal plants-A critical review. J. Med. Plants 2016, 59, 3–39. [Google Scholar]
- Berlyn, G.P.; Russo, R.O. The use of organic biostimulants in nitrogen fixing trees. Nitrogen Fixing Tree Res. Rep. 1990, 8, 78–80. [Google Scholar]
- Russo, R.O.; Berlyn, G.P. The effect of an organic biostimulant (Roots TM) on the growth of loblolly pine (Pinus taeda) seedlings in greenhouse conditions. Agrociencia Ser. Recur. Nat. Renov. 1992, 2, 7–13. [Google Scholar]
- Russo, R.O.; Berlyn, G.P. Vitamin-Humic-Algal Root Biostimulant Increases Yield of Green Bean. HortScience 1992, 27, 847. [Google Scholar] [CrossRef]
- Bulgari, R.; Franzoni, G.; Ferrante, A. Biostimulants application in horticultural crops under abiotic stress conditions. Agronomy 2019, 9, 306. [Google Scholar] [CrossRef]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. Technol. Agric. 2017, 4, 5. [Google Scholar] [CrossRef]
- Kauffman, G.L.; Kneivel, D.P.; Watschke, T.L. Effects of a biostimulant on the heat tolerance associated with photosynthetic capacity, membrane thermostability, and polyphenol production of perennial ryegrass. Crop Sci. 2007, 47, 261–267. [Google Scholar] [CrossRef]
- Halpern, M.; Bar-Tal, A.; Ofek, M.; Minz, D.; Muller, T.; Yermiyahu, U. The Use of Biostimulants for Enhancing Nutrient Uptake. Adv. Agron. 2015, 130, 141–174. [Google Scholar] [CrossRef]
- Li, J.; Liu, H.; Wang, H.; Luo, J.; Zhang, X.; Liu, Z.; Zhang, Y.; Zhai, L.; Lei, Q.; Ren, T.; et al. Managing irrigation and fertilization for the sustainable cultivation of greenhouse vegetables. Agric. Water Manag. 2018, 210, 354–363. [Google Scholar] [CrossRef]
- Bashir, M.A.; Liu, J.; Geng, Y.; Wang, H.; Pan, J.; Zhang, D.; Rehim, A.; Aon, M.; Liu, H. Co-culture of rice and aquatic animals: An integrated system to achieve production and environmental sustainability. J. Clean. Prod. 2020, 249, 119310. [Google Scholar] [CrossRef]
- Gouveia, N.A.; de Andrade, M.G.O.; de Ávila, J.; de Oliveira, T.R.; Simon, C.A.; de Lima, S.F. Evaluation of organic cotton residue and application of biostimulant in the production of radish (Raphanus sativus). Res. Soc. Dev. 2020, 9, e386974092. [Google Scholar] [CrossRef]
- Bulgari, R.; Cocetta, G.; Trivellini, A.; Vernieri, P.; Ferrante, A. Biostimulants and crop responses: A review. Biol. Agric. Hortic. 2015, 31, 1–17. [Google Scholar] [CrossRef]
- Lucini, L.; Rouphael, Y.; Cardarelli, M.; Canaguier, R.; Kumar, P.; Colla, G. The effect of a plant-derived biostimulant on metabolic profiling and crop performance of lettuce grown under saline conditions. Sci. Hortic. 2015, 182, 124–133. [Google Scholar] [CrossRef]
- Souri, M.K.; Hatamian, M. Aminochelates in plant nutrition: A review. J. Plant Nutr. 2019, 42, 67–78. [Google Scholar] [CrossRef]
- Petropoulos, S.A. Practical applications of plant biostimulants in greenhouse vegetable crop production. Agronomy 2020, 10, 1569. [Google Scholar] [CrossRef]
- Garcia, A.L.; Madrid, R.; Gimeno, V.; Rodriguez-Ortega, W.M.; Nicolas, N.; Garcia-Sanchez, F. The effects of amino acids fertilization incorporated to the nutrient solution on mineral composition and growth in tomato seedlings. Span. J. Agric. Res. 2011, 9, 852. [Google Scholar] [CrossRef]
- Mohammadipour, N.; Souri, M.K. Effects of different levels of glycine in the nutrient solution on the growth, nutrient composition, and antioxidant activity of coriander (Coriandrum sativum L.). Acta Agrobot. 2019, 72. [Google Scholar] [CrossRef]
- Zobiole, L.H.S.; de Oliveira, R.S.; Constantin, J.; Kremer, R.J.; Biffe, D.F. Amino acid application can be an alternative to prevent glyphosate injury in glyphosate-resistant soybeans. J. Plant Nutr. 2012, 35, 268–287. [Google Scholar] [CrossRef]
- Noroozlo, Y.A.; Souri, M.K.; Delshad, M. Stimulation Effects of Foliar Applied Glycine and Glutamine Amino Acids on Lettuce Growth. Open Agric. 2019, 4, 164–172. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Canaguier, R.; Svecova, E.; Cardarelli, M. Biostimulant action of a plant-derived protein hydrolysate produced through a hydrolysis. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef]
- Yan, B.X.; Sun, Q.Y. Glycine residues provide flexibility for enzyme active sites. J. Biol. Chem. 1997, 272, 3190–3194. [Google Scholar] [CrossRef] [PubMed]
- Adeva-Andany, M.M.; López-Maside, L.; Donapetry-García, C.; Fernández-Fernández, C.; Sixto-Leal, C. Enzymes involved in branched-chain amino acid metabolism in humans. Amino Acids 2017, 49, 1005–1028. [Google Scholar] [CrossRef]
- Tantawy, A.S.; Abdel-Mawgoud, A.M.R.; El-Nemr, M.A.; Chamoun, Y.G. Alleviation of salinity effects on tomato plants by application of amino acids and growth regulators. Eur. J. Sci. Res. 2009, 30, 484–494. [Google Scholar]
- Abdelgawad, K.F.; Mhmoud, A.A.; Mohamed, H.F.Y. Foliar spraying with some biostimulants improves growth, chemical constituents, and yield of head lettuce plant. Middle East J. Agric. Res. 2018, 7, 1268–1277. [Google Scholar]
- Botta, A. Enhancing plant tolerance to temperature stress with amino acids: An approach to their mode of action. Acta Hortic. 2012, 1009, 29–36. [Google Scholar] [CrossRef]
- Drobek, M.; Frąc, M.; Cybulska, J. Plant biostimulants: Importance of the quality and yield of horticultural crops and the improvement of plant tolerance to abiotic stress-a review. Agronomy 2019, 9, 335. [Google Scholar] [CrossRef]
- Giordano, M.; El-Nakhel, C.; Caruso, G.; Cozzolino, E.; De Pascale, S.; Kyriacou, M.C.; Colla, G.; Rouphael, Y. Stand-alone and combinatorial effects of plant-based biostimulants on the production and leaf quality of perennial wall rocket. Plants 2020, 9, 922. [Google Scholar] [CrossRef] [PubMed]
- Kocira, A.; Lamorska, J.; Kornas, R.; Nowosad, N.; Tomaszewska, M.; Leszczyńska, D.; Kozłowicz, K.; Tabor, S. Changes in Biochemistry and Yield in Response to Biostimulants Applied in Bean (Phaseolus vulgaris L.). Agronomy 2020, 10, 189. [Google Scholar] [CrossRef]
- Parađiković, N.; Vinković, T.; Vinković Vrček, I.; Žuntar, I.; Bojić, M.; Medić-Šarić, M. Effect of natural biostimulants on yield and nutritional quality: An example of sweet yellow pepper (Capsicum annuum L.) plants. J. Sci. Food Agric. 2011, 91, 2146–2152. [Google Scholar] [CrossRef]
- Kumaraswamy, R.V.; Kumari, S.; Choudhary, R.C.; Sharma, S.S.; Pal, A.; Raliya, R.; Biswas, P.; Saharan, V. Salicylic acid functionalized chitosan nanoparticle: A sustainable biostimulant for plant. Int. J. Biol. Macromol. 2019, 123, 59–69. [Google Scholar] [CrossRef] [PubMed]
- Milić, B.; Tarlanović, J.; Keserović, Z.; Magazin, N.; Miodragović, M.; Popara, G. Bioregulators can improve fruit size, yield and plant growth of northern highbush blueberry (Vaccinium corymbosum L.). Sci. Hortic. 2018, 235, 214–220. [Google Scholar] [CrossRef]
- Soppelsa, S.; Kelderer, M.; Casera, C.; Bassi, M.; Robatscher, P.; Matteazzi, A.; Andreotti, C. Foliar applications of biostimulants promote growth, yield and fruit quality of strawberry plants grown under nutrient limitation. Agronomy 2019, 9, 483. [Google Scholar] [CrossRef]

| Experiment | Treatments | Differences |
|---|---|---|
| Experiment 1 |
| Experiment 1 is to identify the role of biostimulant B12 in combination to the GBP Core formula tested against chemical fertilization. Experiment 2 is the combination of GBP Core formula with various inputs (i.e., chemical fertilizer, B12, and co-enzyme CoQ10. |
| Experiment 2 |
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rehim, A.; Amjad Bashir, M.; Raza, Q.-U.-A.; Gallagher, K.; Berlyn, G.P. Yield Enhancement of Biostimulants, Vitamin B12, and CoQ10 Compared to Inorganic Fertilizer in Radish. Agronomy 2021, 11, 697. https://doi.org/10.3390/agronomy11040697
Rehim A, Amjad Bashir M, Raza Q-U-A, Gallagher K, Berlyn GP. Yield Enhancement of Biostimulants, Vitamin B12, and CoQ10 Compared to Inorganic Fertilizer in Radish. Agronomy. 2021; 11(4):697. https://doi.org/10.3390/agronomy11040697
Chicago/Turabian StyleRehim, Abdur, Muhammad Amjad Bashir, Qurat-Ul-Ain Raza, Kevin Gallagher, and Graeme P. Berlyn. 2021. "Yield Enhancement of Biostimulants, Vitamin B12, and CoQ10 Compared to Inorganic Fertilizer in Radish" Agronomy 11, no. 4: 697. https://doi.org/10.3390/agronomy11040697
APA StyleRehim, A., Amjad Bashir, M., Raza, Q.-U.-A., Gallagher, K., & Berlyn, G. P. (2021). Yield Enhancement of Biostimulants, Vitamin B12, and CoQ10 Compared to Inorganic Fertilizer in Radish. Agronomy, 11(4), 697. https://doi.org/10.3390/agronomy11040697






