Deciphering Substrate-Specific Methane Yields of Perennial Herbaceous Wild Plant Species
Abstract
1. Introduction
- By growing bioenergy crops, unused land can be returned to agricultural production and, if necessary, even protected from further degradation by adhering to best management practices.
- Bioenergy production enables a stable basis for the reliable provision of electricity and heat compared to wind and solar energy, which are subject to strong fluctuations.
2. Materials and Methods
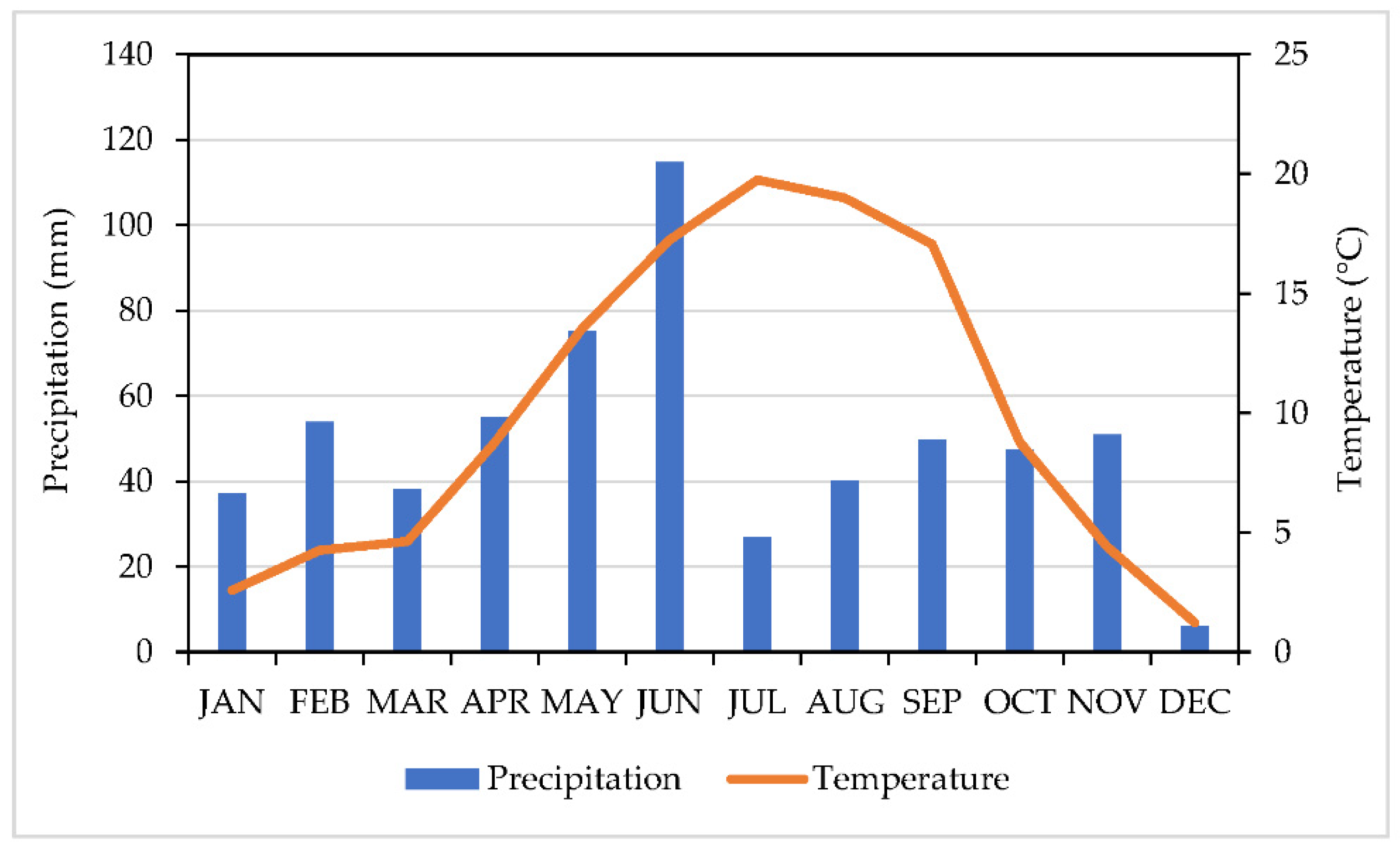
2.1. Origin and Harvest of Plant Material
2.2. Determination of C- and N-Content, Fibre Analyses
2.3. Biogas Batch Test
2.4. Statistical Analysis
3. Results and Discussion
3.1. Lignocellulosic Composition
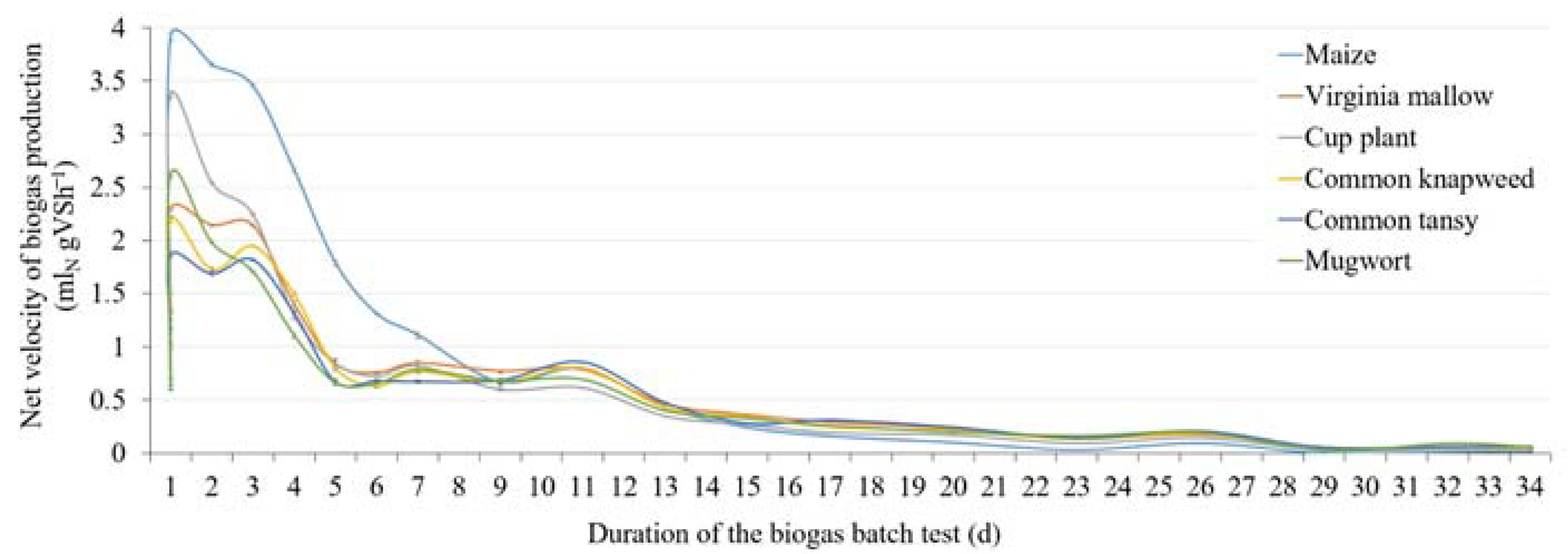
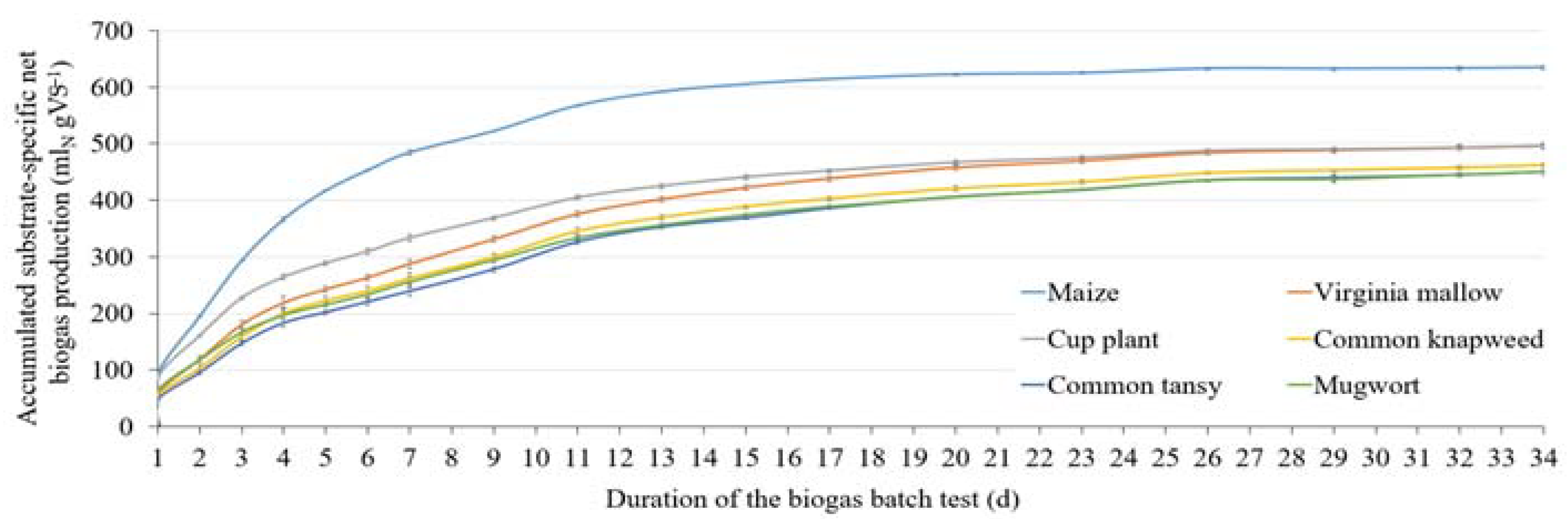
3.2. Methane Content and Substrate-Specific Methane Yield
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WBA. Global Bioenergy Statistics 2020; World Bioenergy Association: Stockholm, Sweden, 2020. [Google Scholar]
- Beale, C.V.; Long, S.P. Seasonal Dynamics of Nutrient Accumulation and Partitioning in the Perennial C4-Grasses Miscanthus x Giganteus and Spartina Cynosuroides. Biomass Bioenergy 1997, 12, 419–428. [Google Scholar] [CrossRef]
- Heaton, E.A.; Dohleman, F.G.; Long, S.P. Meeting US Biofuel Goals with Less Land: The Potential of Miscanthus. Glob. Chang. Biol. 2008, 14, 2000–2014. [Google Scholar] [CrossRef]
- Lewandowski, I.; Scurlock, J.M.; Lindvall, E.; Christou, M. The Development and Current Status of Perennial Rhizomatous Grasses as Energy Crops in the US and Europe. Biomass Bioenergy 2003, 25, 335–361. [Google Scholar] [CrossRef]
- Von Cossel, M.; Winkler, B.; Mangold, A.; Lask, J.; Wagner, M.; Lewandowski, I.; Elbersen, B.; Eupen, M.; Mantel, S.; Kiesel, A. Bridging the Gap Between Biofuels and Biodiversity Through Monetizing Environmental Services of Miscanthus Cultivation. Earth’s Future 2020, 8. [Google Scholar] [CrossRef]
- Alexopoulou, E.; Zanetti, F.; Papazoglou, E.G.; Christou, M.; Papatheohari, Y.; Tsiotas, K.; Papamichael, I. Long-Term Studies on Switchgrass Grown on a Marginal Area in Greece under Different Varieties and Nitrogen Fertilization Rates. Ind. Crop. Prod. 2017, 107, 446–452. [Google Scholar] [CrossRef]
- David, K.; Ragauskas, A.J. Switchgrass as an Energy Crop for Biofuel Production: A Review of Its Ligno-Cellulosic Chemical Properties. Energy Environ. Sci. 2010, 3, 1182–1190. [Google Scholar] [CrossRef]
- Krzyżaniak, M.; Stolarski, M.J.; Szczukowski, S.; Tworkowski, J.; Bieniek, A.; Mleczek, M. Willow Biomass Obtained from Different Soils as a Feedstock for Energy. Ind. Crop. Prod. 2015, 75, 114–121. [Google Scholar] [CrossRef]
- McElroy, G.H.; Dawson, W.M. Biomass from Short-Rotation Coppice Willow on Marginal Land. Biomass 1986, 10, 225–240. [Google Scholar] [CrossRef]
- Stolarski, M.J.; Niksa, D.; Krzyżaniak, M.; Tworkowski, J.; Szczukowski, S. Willow Productivity from Small-and Large-Scale Experimental Plantations in Poland from 2000 to 2017. Renew. Sustain. Energy Rev. 2019, 101, 461–475. [Google Scholar] [CrossRef]
- Stolarski, M.J.; Szczukowski, S.; Tworkowski, J.; Klasa, A. Willow Biomass Production under Conditions of Low-Input Agriculture on Marginal Soils. For. Ecol. Manag. 2011, 262, 1558–1566. [Google Scholar] [CrossRef]
- Bufe, C.; Korevaar, H. Evaluation of Additional Crops for Dutch List of Ecological Focus Area: Evaluation of Miscanthus, Silphium Perfoliatum, Fallow Sown in with Melliferous Plants and Sunflowers in Seed Mixtures for Catch Crops; Wageningen Research Foundation (WR) Business Unit Agrosystems Research: Lelystad, The Netherlands, 2018. [Google Scholar]
- Franzaring, J.; Holz, I.; Kauf, Z.; Fangmeier, A. Responses of the Novel Bioenergy Plant Species Sida Hermaphrodita (L.) Rusby and Silphium Perfoliatum L. to CO2 Fertilization at Different Temperatures and Water Supply. Biomass Bioenergy 2015, 81, 574–583. [Google Scholar] [CrossRef]
- Gansberger, M.; Montgomery, L.F.R.; Liebhard, P. Botanical Characteristics, Crop Management and Potential of Silphium Perfoliatum L. as a Renewable Resource for Biogas Production: A Review. Ind. Crop. Prod. 2015, 63, 362–372. [Google Scholar] [CrossRef]
- Hartmann, A.; Lunenberg, T. Yield Potential of Cup Plant under Bavarian Cultivation Conditions. J. Fur Kult. 2016, 68, 385–388. [Google Scholar] [CrossRef]
- Šiaudinis, G.; Jasinskas, A.; Šarauskis, E.; Steponavičius, D.; Karčauskienė, D.; Liaudanskienė, I. The Assessment of Virginia Mallow (Sida Hermaphrodita Rusby) and Cup Plant (Silphium Perfoliatum L.) Productivity, Physico–Mechanical Properties and Energy Expenses. Energy 2015, 93 Pt 1, 606–612. [Google Scholar] [CrossRef]
- Jablonowski, N.D.; Kollmann, T.; Meiller, M.; Dohrn, M.; Müller, M.; Nabel, M.; Zapp, P.; Schonhoff, A.; Schrey, S.D. Full Assessment of Sida (Sida Hermaphrodita) Biomass as a Solid Fuel. Gcb Bioenergy 2020, 12, 618–635. [Google Scholar] [CrossRef]
- Von Cossel, M.; Lewandowski, I.; Elbersen, B.; Staritsky, I.; Van Eupen, M.; Iqbal, Y.; Mantel, S.; Scordia, D.; Testa, G.; Cosentino, S.L.; et al. Marginal Agricultural Land Low-Input Systems for Biomass Production. Energies 2019, 12, 3123. [Google Scholar] [CrossRef]
- Vollrath, B.; Werner, A.; Degenbeck, M.; Illies, I.; Zeller, J.; Marzini, K. Energetische Verwertung von Kräuterreichen Ansaaten in der Agrarlandschaft und im Siedlungsbereich—eine Ökologische und Wirtschaftliche Alternative bei der Biogasproduktion; Energie aus Wildpflanzen; Bayerische Landesanstalt für Weinbau und Gartenbau: Veitshöchheim, France, 2012; p. 207. [Google Scholar]
- Emmerling, C. Impact of Land-Use Change towards Perennial Energy Crops on Earthworm Population. Appl. Soil Ecol. 2014, 84, 12–15. [Google Scholar] [CrossRef]
- Schmidt, A.; Lemaigre, S.; Delfosse, P.; von Francken-Welz, H.; Emmerling, C. Biochemical Methane Potential (BMP) of Six Perennial Energy Crops Cultivated at Three Different Locations in W-Germany. Biomass Conv. Bioref. 2018, 8, 873–888. [Google Scholar] [CrossRef]
- Von Cossel, M. Renewable Energy from Wildflowers—Perennial Wild Plant Mixtures as a Social-Ecologically Sustainable Biomass Supply System. Adv. Sustain. Syst. 2020, 4, 2000037. [Google Scholar] [CrossRef]
- Brauckmann, H.; Broll, G. Biogaserzeugung-Upscaling Der FuE-Ergebnisse Zu Neuen Kulturen Und Deren Implementierung; Universität Osnabrück: Osnabrück, Germany, 2016. [Google Scholar]
- Zuercher, A.; Stolzenburg, K.; Messner, J.; Wurth, W.; Löffler, C. Was Leisten Alternative Kulturen im Vergleich zu Energiemais? Landinfo. 9 January 2013, pp. 45–50. Available online: https://www.lfl.bayern.de/mam/cms07/ipz/dateien/aggf_2015_wurth_et_al.pdf (accessed on 2 February 2021).
- Vollrath, B.; Werner, A.; Degenbeck, M.; Marzini, K. Energetische Verwertung von Kräuterreichen Ansaaten in der Agrarlandschaft—eine Ökologische und Wirtschaftliche Alternative bei der Biogasproduktion (Phase II); Energie aus Wildpflanzen; Bayerische Landesanstalt für Weinbau und Gartenbau: Veitshöchheim, Germany, 2016; p. 241. [Google Scholar]
- Ruf, T.; Makselon, J.; Udelhoven, T.; Emmerling, C. Soil Quality Indicator Response to Land-Use Change from Annual to Perennial Bioenergy Cropping Systems in Germany. Gcb Bioenergy 2018, 10, 444–459. [Google Scholar] [CrossRef]
- Emmerling, C.; Schmidt, A.; Ruf, T.; von Francken-Welz, H.; Thielen, S. Impact of Newly Introduced Perennial Bioenergy Crops on Soil Quality Parameters at Three Different Locations in W-Germany. J. Plant Nutr. Soil Sci. 2017, 180, 759–767. [Google Scholar] [CrossRef]
- Friedrichs, J.C. Wirtschaftlichkeit des Anbaus von Wildpflanzenmischungen zur Energiegewinnung—Kalkulation der Erforderlichen Förderung zur Etablierung von Wildpflanzenmischungen. 2013. Available online: http://lebensraum-brache.de/wp-content/uploads/2014/04/Gutachten-32-13b-Wildpflanzenmischungen-zur-Energieerzeugung_Netzwerk-Lebensraum-Feldflur.pdf (accessed on 28 February 2021).
- Hahn, J.; De Mol, F.; Müller, J.; Knipping, M.; Minderlen, R.; Gerowitt, B. Wildpflanzen-Samen in Der Biogas-Prozesskette—Eintrags- Und Überlebensrisiko Unter Dem Einfluss von Prozessparametern; Universität Rostock: Rostock, Germany, 2018. [Google Scholar]
- Heiermann, M.; Plogsties, V. Wildpflanzen-Samen in Der Biogas-Prozesskette—Eintrags- Und Überlebensrisiko Unter Dem Einfluss von Prozessparametern—Teilprojekt 2; Leibniz-Institut für Agrartechnik und Bioökonomie e.V.: Potsdam, Germany, 2018. [Google Scholar]
- Von Cossel, M.; Lewandowski, I. Perennial Wild Plant Mixtures for Biomass Production: Impact of Species Composition Dynamics on Yield Performance over a Five-Year Cultivation Period in Southwest Germany. Eur. J. Agron. 2016, 79, 74–89. [Google Scholar] [CrossRef]
- Baum, G. Betriebswirtschaftliche Betrachtung der Wildpflanzennutzung für Biogasbetriebe 2019. Available online: https://baden-wuerttemberg.nabu.de/imperia/md/content/badenwuerttemberg/vortraege/baum_betriebswirtschaftl_wildpflanzen_f__r_biogas_ver__ffentlichung.pdf (accessed on 28 February 2021).
- Kuhn, W.; Zeller, J.; Bretschneider-Herrmann, N.; Drenckhahn, K. Energy from Wild Plants—Practical Tips for the Cultivation of Wild Plants to Create Biomass for Biogas Generation Plants. In The Field Habitat Network; Deutscher Jagdverband e.V. (DJV): Berlin, Germany, 2014; Volume 1, ISBN 978-3-936802-16-0. [Google Scholar]
- Kuhn, W. Interview Kuhn 2020. Available online: https://www.youtube.com/watch?v=K12eOkYxb_U (accessed on 28 February 2021).
- Frick, M.; Pfender, G. AG Wildpflanzen-Biogas Kißlegg 2019. Available online: https://baden-wuerttemberg.nabu.de/imperia/md/content/badenwuerttemberg/vortraege/frick_pr__sentation_hohenheim_12.03.2019_power_point.pdf (accessed on 28 February 2021).
- Von Cossel, M.; Steberl, K.; Hartung, J.; Agra Pereira, L.; Kiesel, A.; Lewandowski, I. Methane Yield and Species Diversity Dynamics of Perennial Wild Plant Mixtures Established Alone, under Cover Crop Maize (Zea Mays L.) and after Spring Barley (Hordeum Vulgare L.). Gcb Bioenergy 2019, 11, 1376–1391. [Google Scholar] [CrossRef]
- Mast, B.; Lemmer, A.; Oechsner, H.; Reinhardt-Hanisch, A.; Claupein, W.; Graeff-Hönninger, S. Methane Yield Potential of Novel Perennial Biogas Crops Influenced by Harvest Date. Ind. Crop. Prod. 2014, 58, 194–203. [Google Scholar] [CrossRef]
- Theuerl, S.; Herrmann, C.; Heiermann, M.; Grundmann, P.; Landwehr, N.; Kreidenweis, U.; Prochnow, A. The Future Agricultural Biogas Plant in Germany: A Vision. Energies 2019, 12, 396. [Google Scholar] [CrossRef]
- Weiland, P. Biogas Production: Current State and Perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef]
- Choong, Y.Y.; Norli, I.; Abdullah, A.Z.; Yhaya, M.F. Impacts of Trace Element Supplementation on the Performance of Anaerobic Digestion Process: A Critical Review. Bioresour. Technol. 2016, 209, 369–379. [Google Scholar] [CrossRef]
- Sauer, B.; Ruppert, H. Spurenelemente in Biogasanlagen: Eine Ausreichende Versorgung Durch Zufuhr Unterschiedlicher Energiepflanzenmischungen Oder Gülle Ist Möglich; Biogaskongress; Interdisziplinäres Zentrum für Nachhaltige Entwicklung der Universität Göttingen & Geowissenschaftliches Zentrum der Georg-August-Universität Göttingen: Göttingen, Germany, 2011. [Google Scholar]
- Schattauer, A.; Abdoun, E.; Weiland, P.; Plöchl, M.; Heiermann, M. Abundance of Trace Elements in Demonstration Biogas Plants. Biosyst. Eng. 2011, 108, 57–65. [Google Scholar] [CrossRef]
- Von Cossel, M.; Möhring, J.; Kiesel, A.; Lewandowski, I. Optimization of Specific Methane Yield Prediction Models for Biogas Crops Based on Lignocellulosic Components Using Non-Linear and Crop-Specific Configurations. Ind. Crop. Prod. 2018, 120, 330–342. [Google Scholar] [CrossRef]
- Siaudinis, G.; Jasinskas, A.; Slepetiene, A.; Karcauskiene, D. The Evaluation of Biomass and Energy Productivity of Common Mugwort (Artemisia Vulgaris L.) and Cup Plant (Silphium Perfoliatum L.) in Albeluvisol. Žemdirbystė (Agric.) 2012, 99, 357–362. [Google Scholar]
- Stolzenburg, K. Anbauerfahrungen und Erträge aus einem Dauerkulturen-Projekt des Landes BW 2019. Available online: https://baden-wuerttemberg.nabu.de/natur-und-landschaft/landwirtschaft/biogas/index.html (accessed on 28 February 2021).
- Zürcher, A. Permanent Crops as an Alternative to Maize—Wild Plant Mixtures, Jerusalem Artichoke, Cup Plant, Virginia Mallow and Szarvasi 2014. Available online: https://docplayer.org/53901435-Dauerkulturen-als-alternativen-zu-mais-wildartenmischungen-topinambur-durchwachsene-silphie-virginiamalve-und-riesenweizengras.html (accessed on 28 February 2021).
- Seppälä, M.; Laine, A.; Rintala, J. Screening Novel Plants for Biogas Production in Northern Conditions. Bioresour. Technol. 2013, 139, 355–362. [Google Scholar] [CrossRef]
- Kiesel, A.; Lewandowski, I. Miscanthus as Biogas Substrate—Cutting Tolerance and Potential for Anaerobic Digestion. Gcb Bioenergy 2017, 9, 153–167. [Google Scholar] [CrossRef]
- Naumann, C.; Bassler, R. VDLUFA Methodenbuch: Die Chemische Untersuchung von Futtermitteln; VDLUFA-Verlag: Darmstadt, Germany, 2006. [Google Scholar]
- von Cossel, M.; Mangold, A.; Iqbal, Y.; Lewandowski, I. Methane Yield Potential of Miscanthus (Miscanthus × Giganteus (Greef et Deuter)) Established under Maize (Zea Mays L.). Energies 2019, 12, 4680. [Google Scholar] [CrossRef]
- Von Cossel, M.; Möhring, J.; Kiesel, A.; Lewandowski, I. Methane Yield Performance of Amaranth (Amaranthus Hypochondriacus L.) and Its Suitability for Legume Intercropping in Comparison to Maize (Zea Mays L.). Ind. Crop. Prod. 2017, 103, 107–121. [Google Scholar] [CrossRef]
- VDI. VDI 4630: Fermentation of Organic Materials—Characterization of the Substrate, Sampling, Collection of Material Data, Fermentation Tests; Verein Deutscher Ingenieure e.V.—Gesellschaft Energie und Umwelt: Düsseldorf, Germany, 2016; Available online: https://infostore.saiglobal.com/en-us/Standards/VDI-4630-2016-1115305_SAIG_VDI_VDI_2590568/ (accessed on 28 February 2021).
- Piepho, H.-P. An Algorithm for a Letter-Based Representation of All-Pairwise Comparisons. J. Comput. Graph. Stat. 2004, 13, 456–466. [Google Scholar] [CrossRef]
- Wolfinger, R. Covariance Structure Selection in General Mixed Models. Commun. Stat.-Simul. Comput. 1993, 22, 1079–1106. [Google Scholar] [CrossRef]
- Kenward, M.G.; Roger, J.H. Small Sample Inference for Fixed Effects from Restricted Maximum Likelihood. Biometrics 1997, 53, 983–997. [Google Scholar] [CrossRef]
- Koch, K.; Post, M.; Auer, M.; Lehuhn, M. Einsatzstoffspezifische Besonderheiten in Der Prozessführung; Biogas Forum Bayern; Arbeitsgemeinschaft Landtechnik und landwirtschaftliches Bauwesen in Bayern e.V.: Freising, Germany, 2017. [Google Scholar]
- Ohly, N. Verfahrenstechnische Untersuchungen Zur Optimierung Der Biogas-Gewinnung Aus Nachwachsenden Rohstoffen. Ph.D. Thesis, Technische Universität Bergakademie Freiberg, Freiberg, Germany, 2006. [Google Scholar]
- Krimmer, E.; Marzini, K.; Heidinger, I. Wild Plant Mixtures for Biogas: Promoting Biodiversity in a Production-Integrated Manner—Practical Trials for Ecological Enhancement of the Landscape. Nat. Und Landsch. 2021, 53, 12–21. [Google Scholar] [CrossRef]
- Amon, T.; Amon, B.; Kryvoruchko, V.; Machmüller, A.; Hopfner-Sixt, K.; Bodiroza, V.; Hrbek, R.; Friedel, J.; Pötsch, E.; Wagentristl, H.; et al. Methane Production through Anaerobic Digestion of Various Energy Crops Grown in Sustainable Crop Rotations. Bioresour. Technol. 2007, 98, 3204–3212. [Google Scholar] [CrossRef]
- Herrmann, C.; Idler, C.; Heiermann, M. Biogas Crops Grown in Energy Crop Rotations: Linking Chemical Composition and Methane Production Characteristics. Bioresour. Technol. 2016, 206, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Herrmann, C.; Plogsties, V.; Willms, M.; Hengelhaupt, F.; Eberl, V.; Eckner, J.; Strauß, C.; Idler, C.; Heiermann, M. Methane Production Potential of Various Crop Species Grown in Energy Crop Rotations. Landtechnik 2016, 71, 194–209. [Google Scholar]
- Ohl, S.; Hartung, E. Comparative Assessment of Different Methods to Determine the Biogas Yield. In Proceedings of the International Conference on Agricultural Engineering-AgEng 2010: Towards Environmental Technologies, Clermont-Ferrand, France, 6–8 September 2010. [Google Scholar]
- Mangold, A.; Lewandowski, I.; Hartung, J.; Kiesel, A. Miscanthus for Biogas Production: Influence of Harvest Date and Ensiling on Digestibility and Methane Hectare Yield. Gcb Bioenergy 2019, 11, 50–62. [Google Scholar] [CrossRef]
- Dandikas, V.; Heuwinkel, H.; Lichti, F.; Drewes, J.E.; Koch, K. Correlation between Biogas Yield and Chemical Composition of Energy Crops. Bioresour. Technol. 2014, 174, 316–320. [Google Scholar] [CrossRef]
- Triolo, J.M.; Sommer, S.G.; Møller, H.B.; Weisbjerg, M.R.; Jiang, X.Y. A New Algorithm to Characterize Biodegradability of Biomass during Anaerobic Digestion: Influence of Lignin Concentration on Methane Production Potential. Bioresour. Technol. 2011, 102, 9395–9402. [Google Scholar] [CrossRef] [PubMed]
- Thomsen, S.T.; Spliid, H.; Østergård, H. Statistical Prediction of Biomethane Potentials Based on the Composition of Lignocellulosic Biomass. Bioresour. Technol. 2014, 154, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Niu, W.; Han, L.; Liu, X.; Huang, G.; Chen, L.; Xiao, W.; Yang, Z. Twenty-Two Compositional Characterizations and Theoretical Energy Potentials of Extensively Diversified China’s Crop Residues. Energy 2016, 100, 238–250. [Google Scholar] [CrossRef]
- FNR. Erhöhung Des Ertragspotentials Heimischer Wildpflanzenmischungen Unter Berücksichtigung von Biodiversität Und Wasserschutz. Available online: https://www.fnr.de/index.php?id=11150&fkz=2219NR215 (accessed on 21 January 2021).
- Knapkon. Energie Für Die Biogasanlagen; Knapkon: Frickenhausen, Germany, 2020. [Google Scholar]
- Iqbal, Y.; Kiesel, A.; Wagner, M.; Nunn, C.; Kalinina, O.; Hastings, A.F.S.J.; Clifton-Brown, J.C.; Lewandowski, I. Harvest Time Optimization for Combustion Quality of Different Miscanthus Genotypes across Europe. Front. Plant Sci. 2017, 8, 727. [Google Scholar] [CrossRef] [PubMed]
- Christian, D.G.; Riche, A.B.; Yates, N.E. Growth, Yield and Mineral Content of Miscanthus x Giganteus Grown as a Biofuel for 14 Successive Harvests. Ind. Crop. Prod. 2008, 28, 320–327. [Google Scholar] [CrossRef]

| Trivial Name | Botanical Name | Life Cycle | Origin |
|---|---|---|---|
| Common knapweed | Centaurea nigra L. | Perennial | Temperate Europe |
| Common tansy | Tanacetum vulgare L. | Perennial | Temperate Europe and Asia |
| Cup plant | Silphium perfoliatum L. | Perennial | Northern America |
| Maize | Zea mays L. | Annual | Central America |
| Mugwort | Artemisia vulgaris L. | Perennial | Temperate Europe, Alaska, Northern Africa and Asia |
| Virginia mallow | Sida hermaphrodita L. Rusby | Perennial | Northern America |
| Crop | NDF (% of DM) | ADF (% of DM) | ADL (% of DM) | Cellulose (% of DM) | Hemicellulose (% of DM) |
|---|---|---|---|---|---|
| Common knapweed | 57.6 + 1.9 ab | 47.3 + 1.9 a | 9.7 + 0.7 b | 37.6 + 1.3 a | 10.3 + 0.6 b |
| Common tansy | 62.4 + 1.9 a | 50.9 + 1.9 a | 12.8 + 0.7 a | 38.1 + 1.3 a | 11.5 + 0.6 b |
| Cup plant | 52.0 + 2.4 b | 44.6 + 2.3 a | 6.7 + 0.9 c | 37.9 + 1.6 a | 7.4 + 0.7 c |
| Maize | 52.7 + 1.9 b | 29.0 + 1.9 b | 3.3 + 0.7 d | 25.8 + 1.3 b | 23.7 + 0.6 a |
| Mugwort | 61.9 + 3.4 a | 52.0 + 3.3 a | 12.6 + 1.3 ab | 39.4 + 2.3 a | 9.9 + 1.0 bc |
| Virginia | 58.7 + 1.9 ab | 47.8 + 1.9 a | 7.0 + 0.7 c | 40.8 + 1.3 a | 10.9 + 0.6 b |
| Crop | NT (% of DM) | CT (% of DM) | CT:NT Ratio | Ash (% of DM) | DMCDS (%) |
|---|---|---|---|---|---|
| Common knapweed | 0.7 + 0.1 bc | 46.1 + 0.3 bc | 68.3 + 4.2 bc | 6.4 + 0.3 b | 93.6 + 0.3 c |
| Common tansy | 0.6 + 0.1 bd | 47.3 + 0.3 a | 75.5 + 4.2 b | 6.1 + 0.3 bc | 93.9 + 0.3 bc |
| Cup plant | 0.6 + 0.1 cd | 44.0 + 0.3 d | 77.9 + 5.2 b | 9.2 + 0.3 a | 90.8 + 0.3 d |
| Maize | 0.8 + 0.1 b | 45.4 + 0.3 c | 57.2 + 4.2 c | 4.1 + 0.3 d | 95.9 + 0.3 a |
| Mugwort | 0.4 + 0.1 d | 46.8 + 0.4 ab | 127.1 + 7.3 a | 5.2 + 0.4 cd | 94.8 + 0.4 ab |
| Virginia | 1.2 + 0.1 a | 45.7 + 0.3 bc | 38.0 + 4.2 d | 6.7 + 0.3 b | 93.3 + 0.3 c |
| Crop | CH4 (%) | SMY (lN kgVS−1) |
|---|---|---|
| Common knapweed | 53.7 + 0.2 ab | 248.5 + 4.1 c |
| Common tansy | 54.2 + 0.2 a | 243.2 + 4.1 c |
| Cup plant | 53.3 + 0.3 bc | 264.7 + 5.0 b |
| Maize | 52.9 + 0.2 c | 337.5 + 4.1 a |
| Mugwort | 53.5 + 0.4 ac | 241.5 + 7.0 c |
| Virginia | 54.1 + 0.2 ab | 267.2 + 4.1 b |
| NDF | ADF | ADL | CEL | HC | Ash | NT | CT | CNR | SMY | |
|---|---|---|---|---|---|---|---|---|---|---|
| ADF | 0.78 ** | |||||||||
| ADL | 0.83 *** | 0.87 *** | ||||||||
| CEL | 0.65 * | n.r. | n.r. | |||||||
| HC | n.s. | n.r. | −0.61 * | −0.89 *** | ||||||
| Ash | n.s. | n.s. | n.s. | n.s. | −0.80 ** | |||||
| NT | n.s. | −0.15 * | −0.41 * | 0.03 * | n.s. | n.s. | ||||
| CT | 0.67 ** | n.s. | 0.70 ** | n.s. | n.s. | n.s. | n.s. | |||
| CNR | n.s. | 0.33 * | 0.54 * | n.s. | n.s. | n.s. | n.r. | n.s. | ||
| SMY | −0.66 * | −0.96 *** | −0.88 *** | −0.89 *** | 0.90 *** | n.s. | 0.26 * | n.s. | −0.39 * | |
| CH4 | n.s. | 0.69 ** | 0.59 * | 0.66 ** | −0.51 ** | 0.32 * | n.s. | n.s. | n.s. | −0.64 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
von Cossel, M.; Pereira, L.A.; Lewandowski, I. Deciphering Substrate-Specific Methane Yields of Perennial Herbaceous Wild Plant Species. Agronomy 2021, 11, 451. https://doi.org/10.3390/agronomy11030451
von Cossel M, Pereira LA, Lewandowski I. Deciphering Substrate-Specific Methane Yields of Perennial Herbaceous Wild Plant Species. Agronomy. 2021; 11(3):451. https://doi.org/10.3390/agronomy11030451
Chicago/Turabian Stylevon Cossel, Moritz, Lorena Agra Pereira, and Iris Lewandowski. 2021. "Deciphering Substrate-Specific Methane Yields of Perennial Herbaceous Wild Plant Species" Agronomy 11, no. 3: 451. https://doi.org/10.3390/agronomy11030451
APA Stylevon Cossel, M., Pereira, L. A., & Lewandowski, I. (2021). Deciphering Substrate-Specific Methane Yields of Perennial Herbaceous Wild Plant Species. Agronomy, 11(3), 451. https://doi.org/10.3390/agronomy11030451








