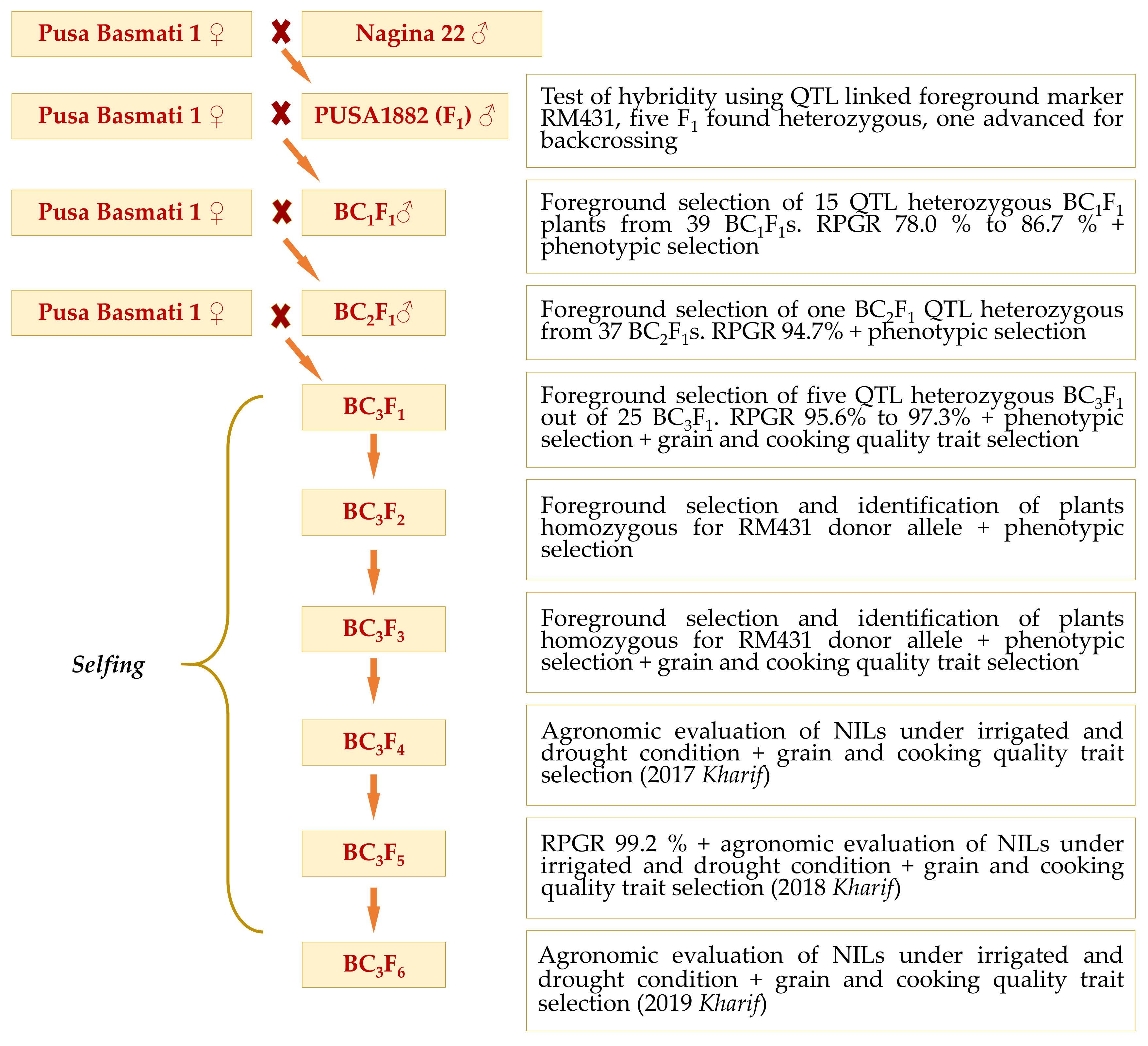
1. Introduction
India is gifted with a vast rice varietal diversity spread across diverse ecosystems. The region spanning from the Himalayan foothills traversing through north-western Indo-Gangetic plains is particularly bestowed with rices of incredible quality, popularly known as Basmati [
1]. Over the time, Basmati has become pride possession of Indian subcontinent, serving exquisite cuisine and thereby an export commodity for trade in the world market. Basmati export from India alone, has earned foreign exchange worth of INR 310,255.6 million (US
$ 4330.6 million) during 2019–2020 from an annual milled rice production of more than eight million tons [
2]. Besides this, almost an equal amount of as much as exported, is consumed in the domestic market. Since Basmati rice is confined to a specific geographic boundary earmarked as Basmati geographical indication (GI) area, their natural genetic diversity remains low rendering them sensitive to all major biotic and abiotic stress factors. With the ensuing threat of global climate change, rice is poised to suffer the onslaught of several stresses, particularly abiotic stresses. Among these, drought would be a major factor as global warming is recognized as one of the important causes of climate change [
3]. Global warming results from atmospheric increase in greenhouse gases propelling vagaries of temperature and rainfall [
4]. Furthermore, climate change envisages exacerbation of drought in the coming years [
5] and the strength and frequency of drought are expected to become worse [
6]. Rice requires about 3000 to 5000 L of water to yield one kilogram of rice [
7], and erratic rainfall can drive rice production ambivalent in rainfed regions. Estimates indicate that almost 34 million ha of rice in rainfed lowland areas in Asia alone recurrently suffers from water stress, in addition to an area of eight million ha under upland ecology [
8].
Rice is grown throughout the year in India depending on the geographical region, in different seasons such as
Kharif and
Rabi.
Kharif season coincides sowing in late summer, and is the principal rice growing season.
Rabi which coincides with the winter sowing, is however limited to peninsular India, where winter is mild.
Rabi, however, forms the second most important rice growing season with plantings restricted to southern and eastern India. Depending on geography, the weather parameters of
Kharif and
Rabi season vary, including intermittent spells of stress such as drought. Onset of drought can happen in all stages of rice crop. However, sensitivity to the drought stress depends on its duration and intensity. Although rice is affected badly in all phenological stages of growth, the stress occurring during the reproductive stage is particularly perilous, leading to significant reduction in grain yield [
9,
10]. The impact on rice plant is multifaceted, ranging from the reduction in dry matter accumulation to poor apportioning of metabolites from the source (stem and leaves) to sink (grain) [
11]. This results in reduced number of filled grains per panicle, reduced grain weight and, ultimately, the grain yield. Varietal diversity in rice embodies various mechanisms for drought tolerance particularly at the reproductive stage. Tolerance is established through manifestations such as reduction in the tiller number, leaf area, shortening and thickening of leaves, leaf rolling and promotion leaf senescence [
12,
13]. These mechanisms offer opportunities for varietal improvement targeting drought prone areas. Additionally, breeding of futuristic cultivars resilient to multiple stresses requires enhanced drought tolerance, especially at the reproductive stage. Drought stress at reproductive stage leads to delay in flowering which slows down the panicle development, impairs flowering and grain development leading to crop loss, that can be total at severe situations. Stress occurring at anthesis stage leads to sterile panicle, because the panicle that is still inside the leaf sheath, fails to develop. Failure of reproduction ultimately translates into significant yield decline. The reproductive stage drought stress (RSDS) tolerance in rice is considered as a complex and multifarious trait, governed by several genes, major and minor. These may include a large number of quantitative trait loci (QTLs) that may comprise of structural and functional genes as well as regulatory elements such as transcription factors, controlling a multitude of morpho-physiological and biochemical responses [
14].
Exploiting molecular markers of various kind, several studies have reported QTLs governing drought tolerance in rice, primarily using grain yield under drought stress (
DTY) as the surrogate trait. Among these,
qDTY1.1 is a major QTL for grain yield under drought stress (explaining about 12.6% to 16.9% of phenotypic variance), mapped on chromosome 1 independently from two tolerant cultivars such as Nagina 22 (N22) [
15] and Dhagaddeshi [
16].
qDTY1.1 was the first QTL reported to have consistent effect in different genetic backgrounds and was shown to affect many drought related traits like root length, relative water content, root thickness, osmotic adjustment and biomass [
16,
17]. Previous studies have demonstrated that
qDTY1.1 was flanked by markers RM431 on telomeric end and RM11943 on the centromeric end. Additionally, Bernier et al. [
17] identified a different major QTL on chromosome 12,
qDTY12.1, from the cross between Vandana and Way Rarem explaining approximately 51% of phenotypic variation. There are also other major QTLs reported which were demonstrated effective either under upland or lowland situations [
14,
18].
Marker assisted introgression of major-effect QTLs could be a proficient and rapid approach for breeding rice varieties tolerance to drought stress [
17]. Consequent attempts to introgress/pyramid these QTLs by marker assisted selection, primarily into mega-varieties has found significant advancements towards breeding climate-adaptive cultivars [
19,
20], such as Sabitri (
qDTY3.2 and
qDTY12.1) [
21], IR 64 (
qDTY2.2 and
qDTY4.1), Vandana (
qDTY12.1) [
22] and Pusa 44 [
23]. In the past twelve years, there has been about sixty-six RSDS tolerant varieties released around the world, which involve several of these QTLs [
14]. Although important, there has not been a previous attempt to improve Basmati cultivars for RSDS tolerance. One of the major reasons for this hiatus was the lack of donors from the Basmati group. Use of donors from the non-Basmati backgrounds for Basmati improvement, present a major challenge of loss of grain quality of the Basmati parent, while introgression [
24]. However, marker assisted backcross breeding coupled with phenotypic selection, has been demonstrated to effectively address this problem [
25,
26,
27].
Developed by the Indian Council of Agricultural Research (ICAR)-Indian Agricultural Research Institute (ICAR-IARI), New Delhi and released for commercial cultivation during year 1989, Pusa Basmati 1 (PB1) is the world’s first semi-dwarf (105 cm) Basmati variety with high yielding potential. This variety, that showcased a tremendous level of improvement over the traditional Basmati cultivars that were low yielding (~2.3 tons/ha), photosensitive, tall (>150 cm), lodging with weaker stem and of long duration (>150 days), became popular among the farmers in no time. Besides the high yield (5.0 tons/ha), PB1 possesses insensitivity to photoperiodism, resistance to lodging, superior grain quality, semi-dwarf stature and a duration of 135–140 days. The grain quality of PB1 was adorned with strong aroma with explicit cooking qualities such as high kernel elongation ratio of 1.8, an average milled grain length of 7.4 mm and cooked kernel length of 13.7 mm [
19]. However, as that of traditional cultivars, PB1 too is highly sensitive to several stress factors such as pests and diseases as well as drought. This study forms the maiden attempt to improve any Basmati cultivar towards climate resilience, by introgressing RSDS tolerance into PB1. The augmented objectives were to recover the Basmati grain quality traits as well as the recovery of all the agronomic traits including yield in the near isogenic lines (NILs). Further, evaluation of the improved NILs across multiple locations and environments to ascertain the stability in yield and drought tolerance to deploy as commercial Basmati rice cultivar.
4. Discussion
Climate change is predicted to affect rice cultivation worldwide adversely, through various impacts, such as drought, excess rainfall, temperature fluctuations and also predisposing it to several biotic stresses. Any adverse effect on rice production would threaten world food security, because more than half of the world population is rice dependent. This is particularly important to rice consuming countries like India, where about 90% of the total rice production is internally consumed [
40]. Among these, biotic threats due to diseases and pests, as well as abiotic stresses such as drought, submergence and salinity are particularly relevant under climate change scenario. Predominantly in the rainfed environment of South and Southeast Asia, drought and submergence are more frequently encountered during the crop growing season [
41]. Most of the prominent and popular rice varieties are vulnerable to these abiotic stresses [
20]. Since climatic vagaries occur spontaneously and intermittently, growing tolerant rice varieties for wide range of stresses is the only economically viable option to manage abiotic stresses. This option is particularly important in the case of drought, because it remains as the most frequently occurring stress that bears the potential to fail a rice crop. Judging drought tolerance of genotypes and its transfer into elite backgrounds is quite laborious due to complex nature of tolerance. It is this complexity that renders the improvement for drought resilience through conventional breeding tardy. However, efforts can be remarkably hastened by leveraging molecular markers that are linked to various traits associated with drought tolerance. Moreover, use of markers can aid a relatively cost-effective and environment neutral selection, while improving the accuracy and reducing the turnover time. Molecular marker assisted introgression of traits provides various advantages such as easy recovery of an otherwise difficult phenotypic trait, easy selection, increased accuracy and shortening the breeding time. Therefore, marker assisted backcross breeding (MABB) was proven to be an efficient strategy for incorporation of desired trait associated genes/QTLs in numerous prominent rice varieties [
21,
42,
43]. MABB along with stringent phenotypic selection was successfully employed for Basmati rice improvement for different biotic stresses [
26,
30,
44,
45,
46,
47,
48]. MABB is also proven to a superior method for QTLs transfer into desired cultivar/variety for different abiotic stresses as well, such as drought. Out of the several QTLs reported for reproductive stage drought tolerance in rice,
qDTY1.1 [
15,
16],
qDTY2.1 [
33],
qDTY3.1 [
33,
49] and
qDTY12.1 [
50,
51], showed consistent grain yield under drought across different genetic backgrounds and has been used in breeding applications. Recently, Dwivedi et al. [
23] reported successful pyramiding of two QTLs,
qDTY2.1 and
qDTY3.1 into the megavariety, Pusa 44 significantly enhancing yield under drought. Despite the proven advantage, however, only few success stories in improving rice varieties for drought tolerance using MABB has been reported rice.
In the present study, MABB was utilized to introgress a RSDS tolerance QTL
qDTY1.1 from N22 into PB1, one of the popular Basmati rice varieties of India. Popular for its high yield, better-quality grain, and excellent cooking quality and pleasing aroma, PB1 is the first ever semi-dwarf Basmati rice variety developed. Bred through conventional convergent breeding procedures, PB1 was derived from a cross between Pusa 150/Karnal local. Pusa 150 has Basmati 370 in its lineage, while Karnal local was a landrace with better aroma and cooking quality. Karnal local was a selection from Haryana Basmati collection 19 (HBC19), that was later released as Taraori Basmati [
52]. Despite its excellent yield gain over the conventional Basmati cultivars, PB1 was semi-dwarf with excellent plant architecture, photo insensitivity, high yielding and with unparalleled grain quality [
53]. However, like its congeners, PB1 is also sensitive to drought as well as to many biotic stresses. Released during 1989, PB1 remains popular even today among the Basmati farmers of India, despite above limitations. Furthermore, till date there is no report on improvement any Basmati rice variety for drought tolerance. Therefore, the present was carried out with the objective to incorporate drought tolerance in PB1. The donor parent, N22 is a tall upland rice variety belonging to
aus group. N22 is a pure line selection from a landrace, Rajbhog [
54] that possesses deeper root system, shorter duration and tolerant to heat and drought [
55]. This variety has short bold non-aromatic grains with very less elongation on cooking.
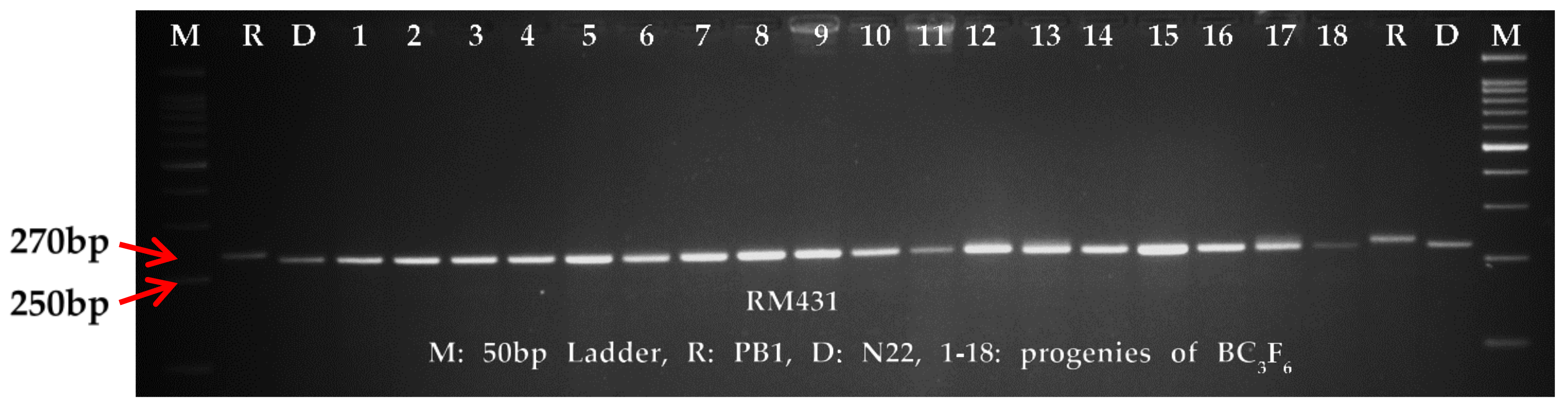
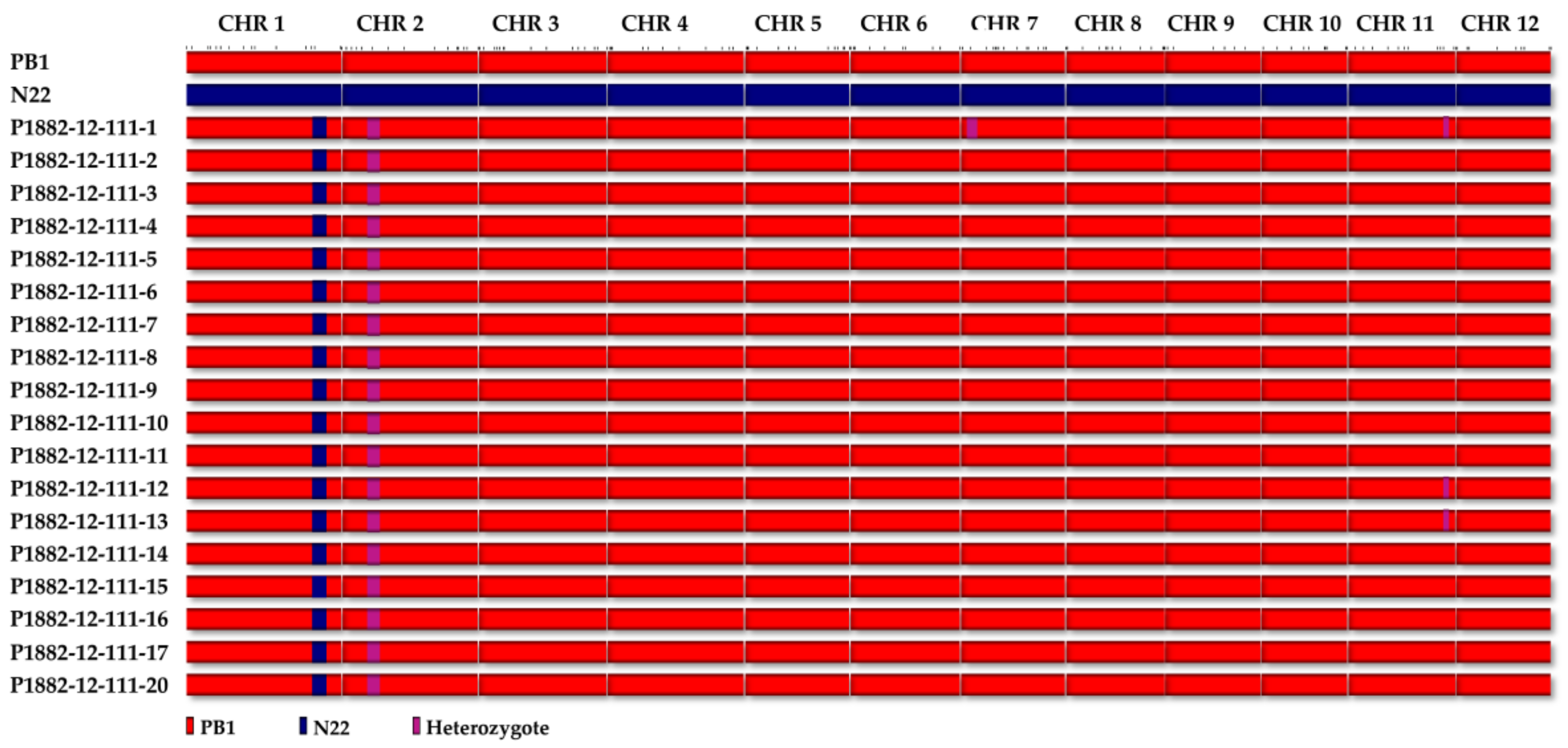
The genome wide average polymorphism between the parents, PB1 and N22 was 17.35%, which was more than that of the carrier chromosome of
qDTY1.1. The molecular analyses on the background recovery has revealed maximum recovery of chromosome 1, thereby eliminating the chances for linkage drag. The fact that maximum background markers (101 markers) were surveyed on chromosome 1, substantiate our claim for the elimination of linkage drag. Furthermore, the agronomic recovery of the NILs in general suggests that no undesirable trait has been incorporated by chance from other donor fragments, that might have escaped marker-based background selection. In this study,
qDTY1.1 was found providing increased level of drought tolerance in the NILs vis-à-vis PB1. It is also known that QTLs from a particular genetic background usually show minor effects or may remain completely silent in diverse genetic backgrounds [
56]. In the case of drought, interaction between QTL × genetic background has been a major bottleneck limiting the use of QTLs for MABB in rice [
50,
57,
58]. Nevertheless,
qDTY1.1 derived from N22 has been demonstrated effective in multiple non-Basmati backgrounds such as MTU1010, IR64 and Swarna, which showed its role as a major effective QTL governing grain yield under reproductive stage water stress [
15]. This justifies the use of
qDTY1.1 as a most desirable candidate for MABB to improve prominent high-yielding varieties with augmented drought tolerance.
Notwithstanding, drought has a significant role in reducing the yield under drought relative to irrigated cultivation. Under drought, yield reduction was found majorly influenced by spikelet fertility. In comparative assessment, there was no significant yield reduction noticed under unstressed condition among the NILs as a consequence of introgressing the
qDTY1.1. In an earlier report on introgression of another QTL,
qDTY3.1, a negative effect on grain yield was encountered by Venuprasad et al. [
33] under unstressed treatment. The NILs evolved under this program, showed significant higher gain for grain yield over PB1, with P1882-12-111-2, P1882-12-111-3, P1882-12-111-5, P1882-12-111-6, P1882-12-111-7, P1882-12-111-10, P1882-12-111-12, P1882-12-111-15 and P1882-12-111-17 having clear advantage under both stressed and unstressed treatments. This also indicated that negative effect of QTL × background interaction was not observed under PB1 background. P1882-12-111-17 had the maximum grain yield among the NILs followed by P1882-12-111-3, P1882-12-111-5, P1882-12-111-6, P1882-12-111-7 and P1882-12-111-12 under both stressed, and unstressed conditions during all the years. This signified the effectiveness of
qDTY1.1 in enhancing yield under drought stress. However, the effect of the introgressed QTL among the PB1 NILs was more conspicuous in the 2019 than in the previous years. In the recent report from World Meteorological Organization [
59], it has been reported that climate change is accelerating by the last five-year period between 2015–2019. Although 2016, has been identified as the most erratic year, 2019 also showed significantly high incidence of drought than 2017 and 2018. Therefore, 2019 season data were more explicit to showcase the drought tolerance response of the NILs. This was evident from performance of NILs vis-à-vis PB1, which showed significant deviation for traits such as SF, GY, DF and PH particularly during 2019 and 2017 seasons. In all the seasons, however, the donor (N22) and the positive control (IR86918-B-B-305) remained the best performers under drought possibly indicating the necessity to transfer additional QTLs from multiple donors to further push the tolerance threshold of PB1. Similar observations were made by Vikram et al. [
15] that
qDTY1.1 had a considerable effect on enhancing the GY under both stressed and unstressed conditions. Similar effect of
qDTY1.1 on improving the yield was earlier reported, conditioned through several adventitious drought-linked traits such as root characteristics, relative water content, biomass, and osmotic adjustment [
17,
60,
61]. Meta QTLs have also been documented for maximum root length and GY within the
qDTY1.1 region [
62,
63]. In the PB1 background,
qDTY1.1 seems to enhance the yield under drought indirectly by modulating spikelet fertility as inferred from stepwise regression analysis. This implies on the importance of maintaining higher spikelet filling in achieving higher yields. Therefore, we hypothesize that grain filling could be the putative trait,
qDTY1.1 is associated with, that require further validation.
Stability of yield performance under stress situations across the seasons is also important for a NIL to be considered for varietal evaluation prior to release as a cultivar across different rice growing areas of the country. The AMMI stability analysis carried out towards assessing the stable yielding potential, revealed that the NILs showed differential stability pattern. Two concomitant stability indices AST and YSI were used for assessing the yield stability, while AST alone was used for SF. Considering the stability indices and mean performance, one of the NILs, P1882-12-111-12 was adjudged stable for both GY and SF. This line can be a potential candidate for varietal evaluation pipeline in the future.
Among the agronomic traits, it was found that there was a delay in DF found across the NILs under stressed conditions. Delay in the flowering time under drought was also reported earlier in rice [
16,
64,
65], which is due to delay in flower development and slow rate of panicle elongation under stress. The delay was however conspicuous in IR64, the short duration high yielding check variety used in the study. In short duration varieties with little drought adaptation, flower development delay occurs more significantly than longer duration cultivars. Similarly, there was a general decrease in PH among all the lines under stress, the degree of decrease was slightly reduced among the NILs. However, there was no increase in height observed among the NILs, although they have all possessed
qDTY1.1, attributable to the tight linkage of
qDTY1.1 with
SD1 gene, the gene responsible for tallness in N22 [
66]. In the present study, we surmise that linkage between the
qDTY1.1 and
sd1 allele might have broken resulting in semi-dwarf NILs with height similar to that of PB1.
Tolerance indices are better yardsticks for judging the tolerance of genotypes than the yield
per se under stress condition. Three popular stress indices were utilized in the present study to identify best performing NILs. DYI is based on the mixed model which accounts for the genotype × stress level interaction across different environments. For drought prone areas, the combination of DYI with deviations in genotype performance under irrigated conditions may facilitate breeders to select genotypes with no yield reduction under favorable environment in comparison to currently cultivated varieties [
37]. STI sorts out the entries which perform well under nonstress and fairly well under stress condition, high value of STI for a genotype infers higher tolerance level to the drought stress [
39]. Fisher and Maurer [
39] proposed SSI that estimates the yield reduction due to unfavorable environment in comparison to favorable environment. Lower the SSI value lesser the yield difference between stress and non-stress condition and hence greater is the tolerance of the genotype [
67]. Thus, DYI, STI and SSI favored P1882-12-111-17 as best genotype for drought situation due to its highest grain yield under both stressed and unstressed conditions followed by P1882-12-111-3, P1882-12-111-6, P1882-12-111-7, P1882-12-111-12, P1882-12-111-15 and P1882-12-111-20. Further, P1882-12-111-17 out yielded majority of the remaining NILs. Among various traits measured in the present study, SF stood out as the most significant trait influencing grain yield under drought stress. Successful reproduction and efficient grain filling are the important determinants of ultimate grain yield under stress which is reflected as higher spikelet fertility. Thus, the NILs showed superior yields due to the maintenance of spikelet fertility under drought stress.
PB1 being a Basmati rice variety having specialty grain characteristics, transfer of drought tolerance from a non-aromatic variety with totally different grain architecture was a great challenge. Since the QTL transfer was into a Basmati background from a non-Basmati donor, a critical comprehensive quality check was done under both non-stress and stress situations in 2019. The NILs generated, possessed all the grain and cooking quality attributes of PB1 along with drought tolerance (
Figure 4).
The recovery of quality in the NILs was achieved by stringent phenotypic selection integrated to the marker-based selection for the target trait, RSDS. NILs under well-watered treatment has grain quality similar to that of PB1, while an inconspicuous quality variation was noticed under drought situation. In spite of the successful recovery of PB1 genome component governing grain quality among the NILs, there was a marginal reduction in head rice recovery noticed under severe stress, although statistically insignificant, together with a slight increase in chalkiness. It is well known that drought at grain filling interferes with starch packaging in the grains, leading to chalkiness. Besides, the grain quality under stressed treatment was not significantly different from that under unstressed, indicating that drought had little effect of grain quality in the NILs. Grain quality recovery using MABB with augmented phenotypic selection, particularly in Basmati cultivars, was earlier reported from several studies [
27,
30,
68].