Variation in Root System Architecture among the Founder Parents of Two 8-way MAGIC Wheat Populations for Selection in Breeding
Abstract
:1. Introduction
2. Materials and Methods
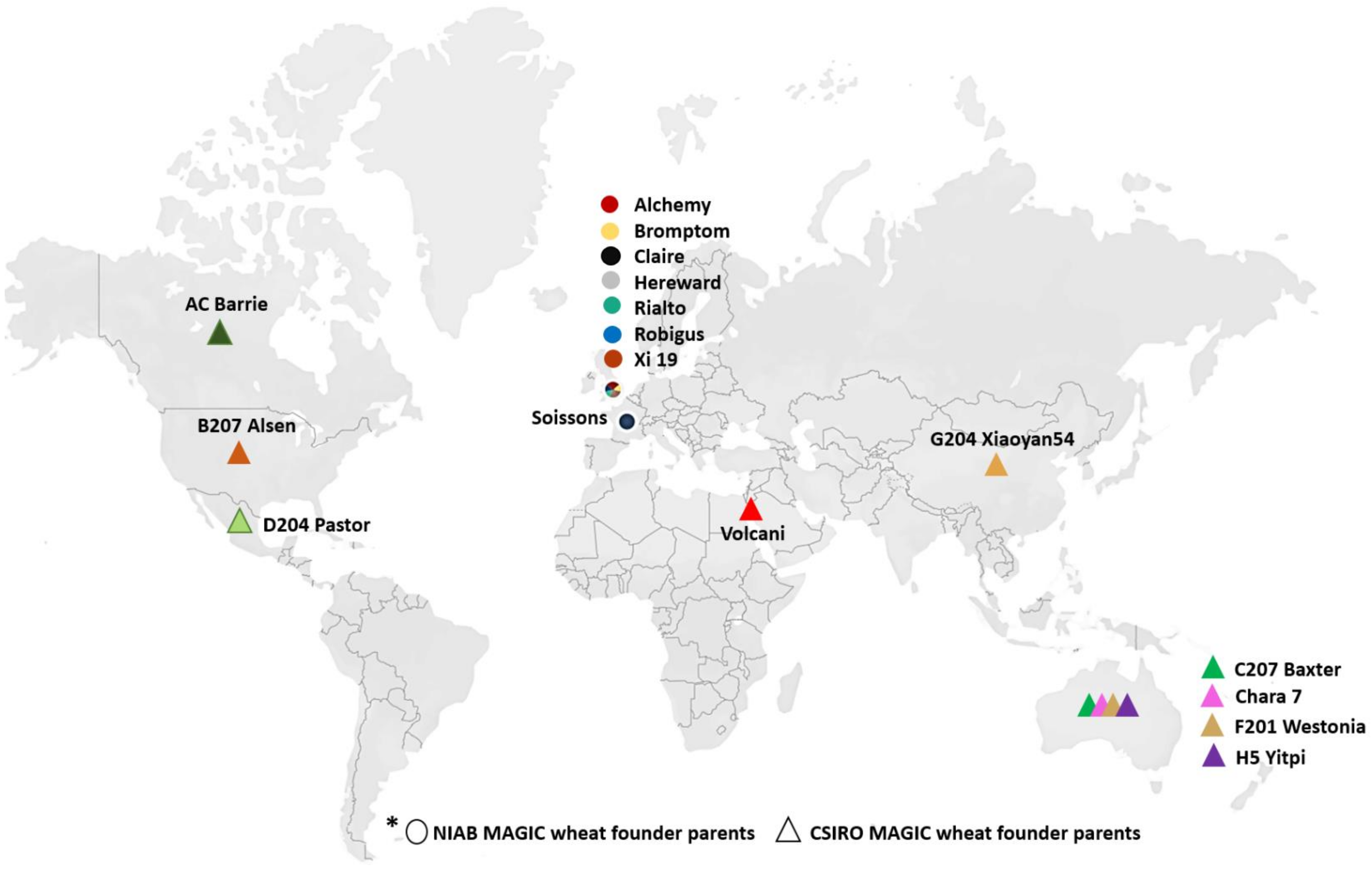
2.1. Plant Materials
2.2. Phenotyping System
2.3. Plant Cultivation and Experimental Design
2.4. Image Acquisition and Analysis
2.5. Statistical Analysis
3. Results
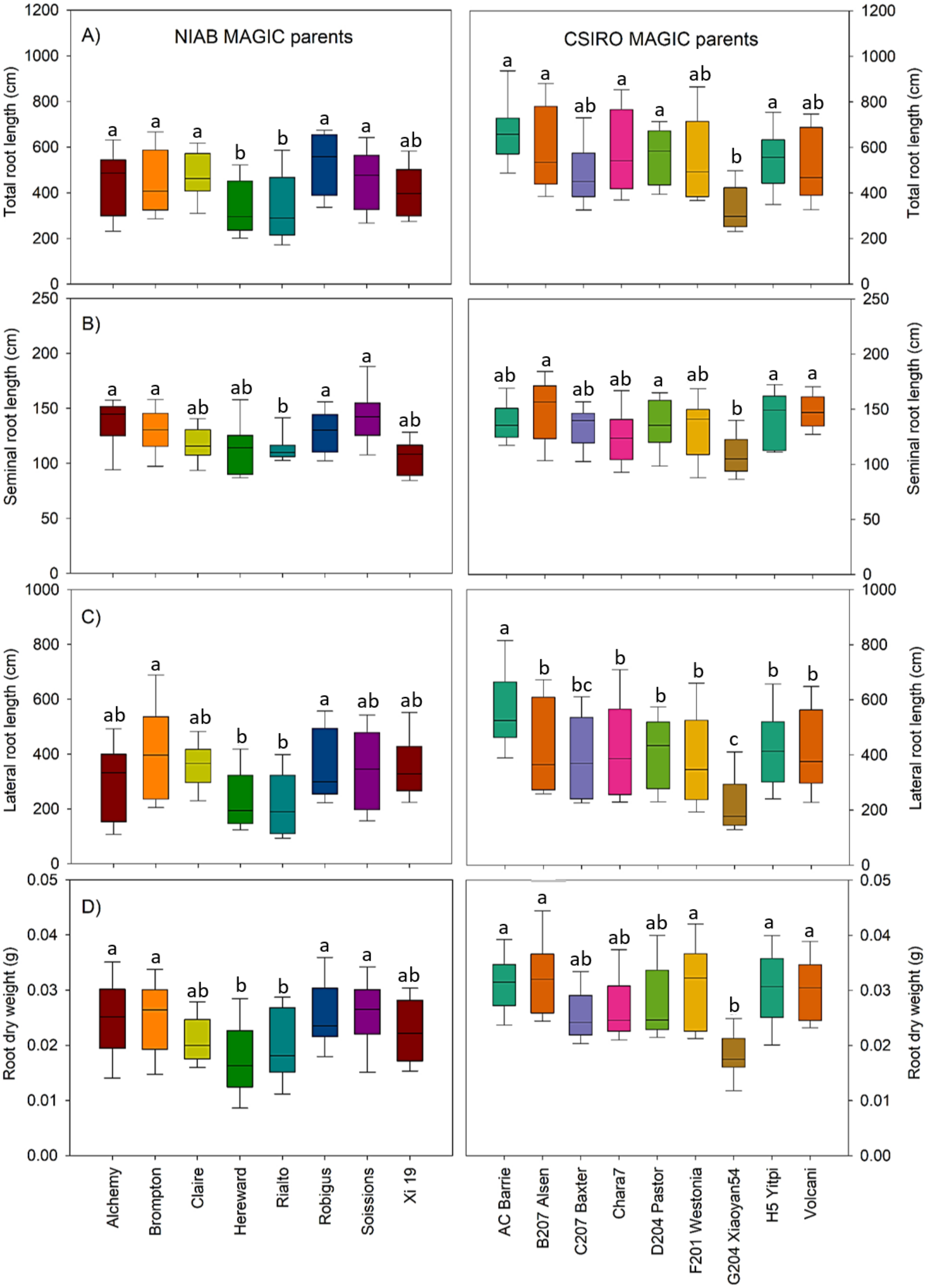
3.1. Variation in Root Lengths and Biomass Was Observed in Both Sets of MAGIC Parents
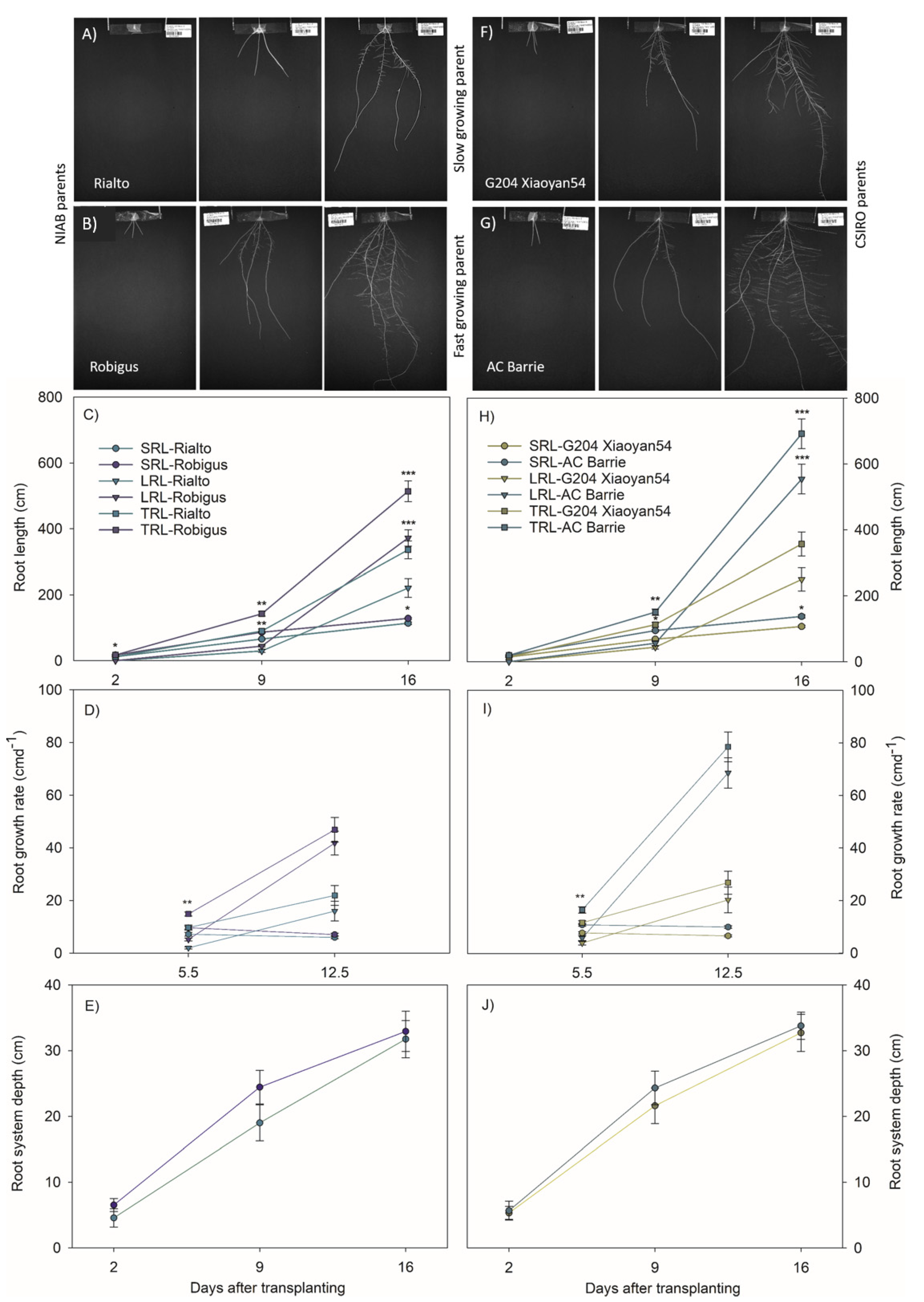
3.2. Slow and Fast Growing Founder Parents Differed Most through Elongation Rates of Lateral Roots
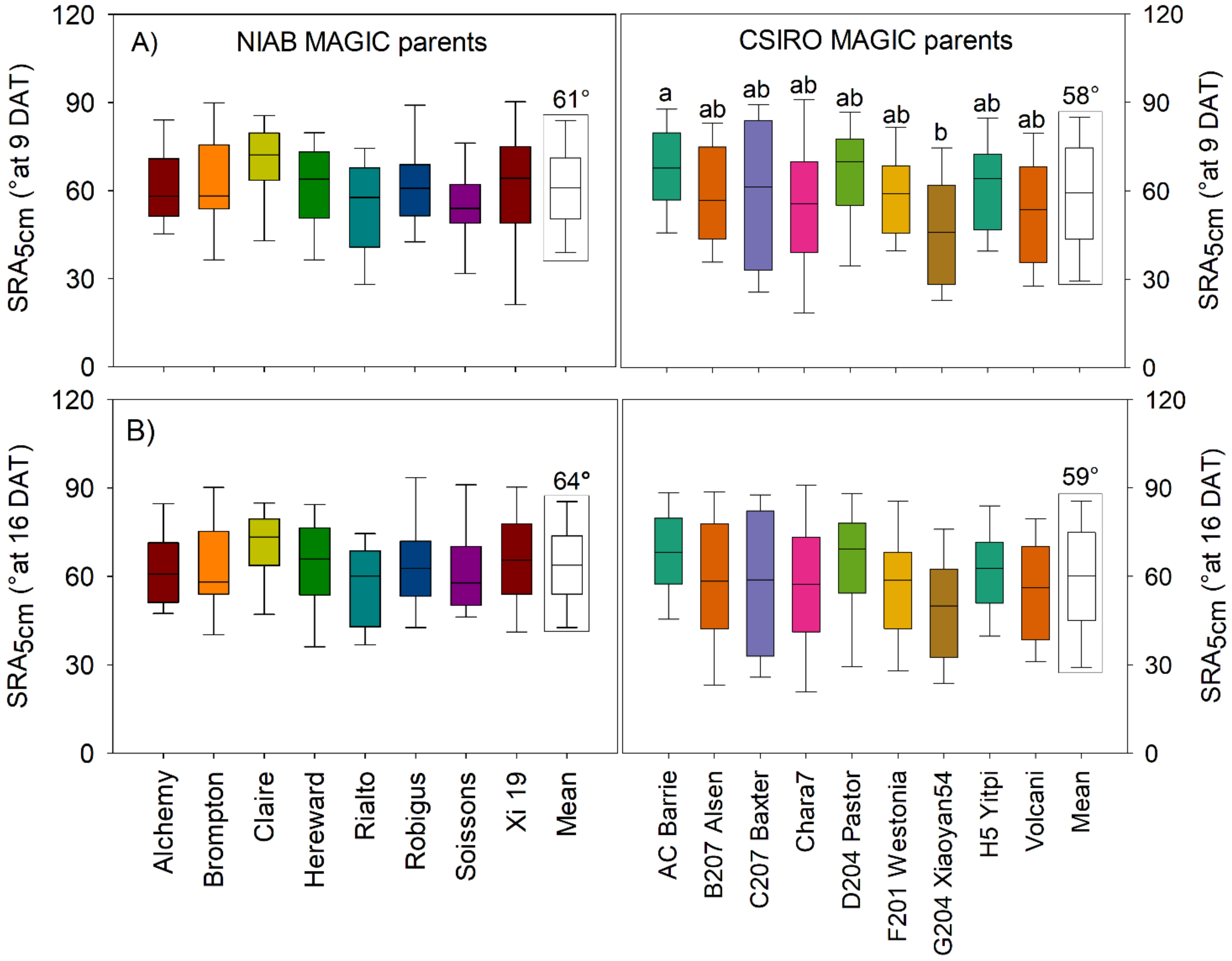
3.3. Phenotypic Variation for Seminal Root Angles Was Observed Only across CSIRO MAGIC Parents
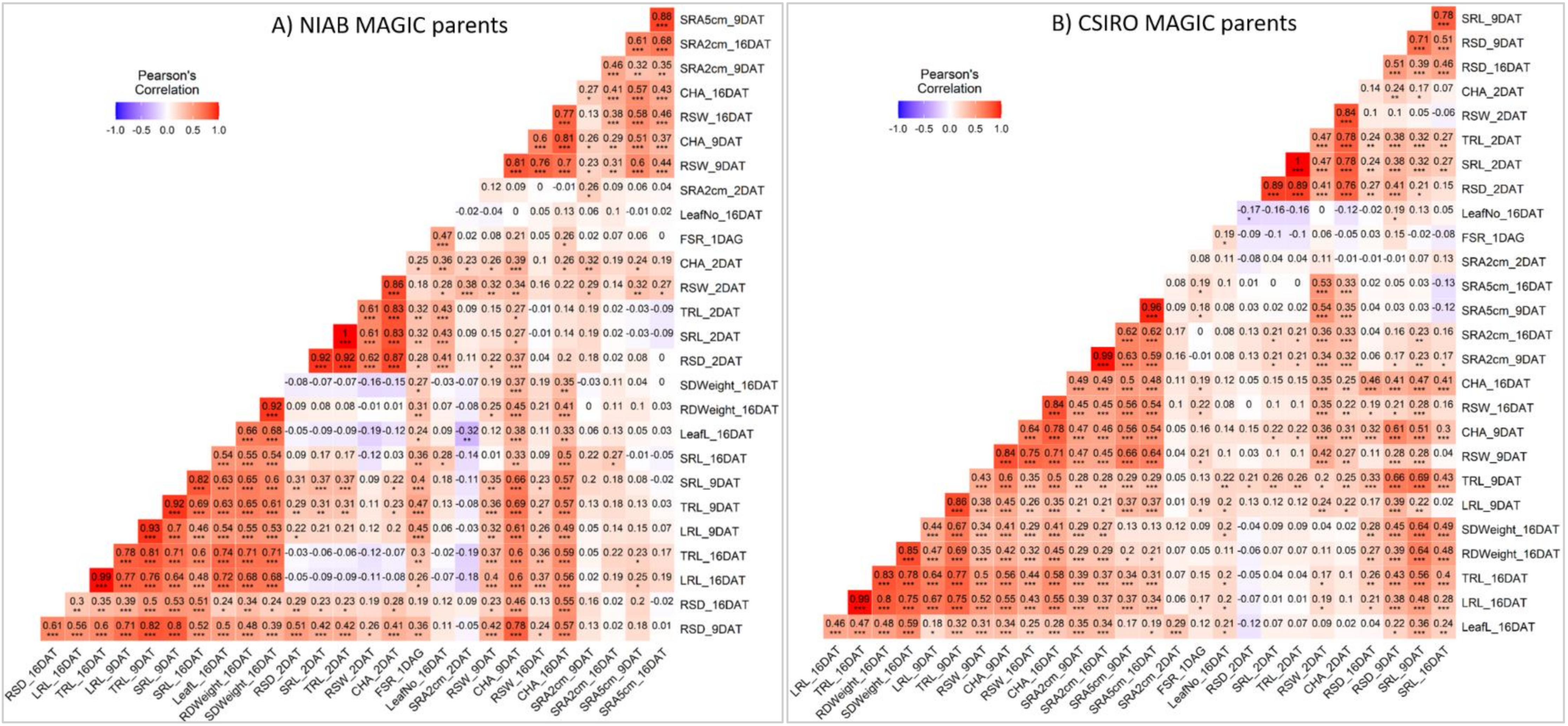
3.4. Traits with High Heritability, Repeatability and Correlations for Selection in Breeding
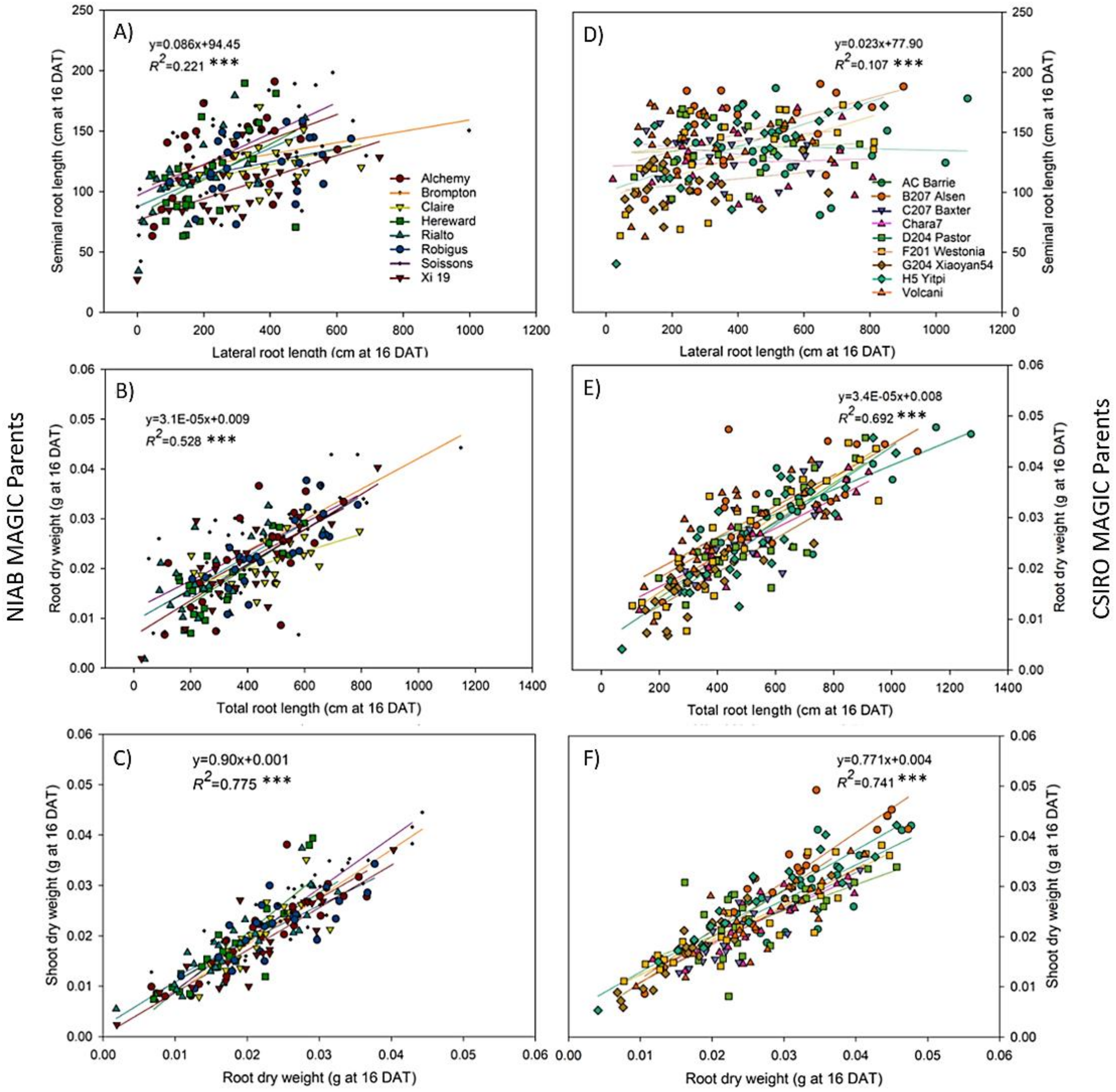
3.5. Fast Growing Parents Allocated More Resources to the Roots during Early Stages of Plant Development
4. Discussion
4.1. Key Traits for Potential Selection in Breeding
4.2. CSIRO MAGIC Parents Grew Faster Than NIAB MAGIC Parents
4.3. Seminal Root Angle Was Not Correlated to Root System Depth
4.4. Biomass Allocation Patterns Vary in Between Fast and Slow Growing Parents
4.5. Transfer of Results to Soil Conditions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shewry, P.R.; Hey, S.J. The contribution of wheat to human diet and health. Food Energy Secur. 2015, 4, 178–202. [Google Scholar] [CrossRef]
- Shewry, P.R. Wheat. J. Exp. Bot. 2009, 60, 1537–1553. [Google Scholar] [CrossRef] [PubMed]
- Dubcovsky, J.; Dvorak, J. Genome plasticity a key factor in the success of polyploid wheat under domestication. Science 2007, 316, 1862–1866. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Börner, A.; Röder, M.; Ganal, M. Assessing genetic diversity of wheat (Triticum aestivum L.) germplasm using microsatellite markers. Theor. Appl. Genet. 2002, 105, 699–707. [Google Scholar] [CrossRef]
- Matsuoka, Y. Evolution of polyploid Triticum wheats under cultivation: The role of domestication, natural hybridization and allopolyploid speciation in their diversification. Plant Cell Physiol. 2011, 52, 750–764. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Appels, R.; Eversole, K.; Feuille, C.; Keller, B.; Rogers, J.; Stein, N.; The International Wheat Genome Sequencing Consortium (IWGSC). Shifting the limits in wheat research and breeding using a fully annotated reference genome. Science 2018, 361, 1110–1126. [Google Scholar]
- Cavanagh, C.; Morell, M.; Mackay, I.; Powell, W. From mutations to MAGIC: Resources for gene discovery, validation and delivery in crop plants. Curr. Opin. Plant Biol. 2008, 11, 215–221. [Google Scholar] [CrossRef]
- Huang, X.; Paulo, M.J.; Boer, M.; Effgen, S.; Keizer, P.; Koornneef, M.; van Eeuwijk, F.A. Analysis of natural allelic variation in Arabidopsis using a multiparent recombinant inbred line population. PNAS 2011, 108, 4488–4493. [Google Scholar] [CrossRef] [Green Version]
- Kover, P.X.; Valdar, W.; Trakalo, J.; Scarcelli, N.; Ehrenreich, I.M.; Purugganan, M.D.; Durrant, C.; Mott, R. A multiparent advanced generation inter–cross to fine–map quantitative traits in Arabidopsis thaliana. PLoS Genet. 2009, 5, e1000551. [Google Scholar] [CrossRef] [Green Version]
- Huang, B.E.; George, A.W.; Forrest, K.L.; Kilian, A.; Hayden, M.J.; Morell, M.K.; Cavanagh, C.R. A multiparent advanced generation inter-cross population for genetic analysis in wheat. Plant Biotechnol. J. 2012, 10, 826–839. [Google Scholar] [CrossRef] [PubMed]
- Mackay, I.J.; Bansept-Basler, P.; Barber, T.; Bentley, A.R.; Cockram, J.; Gosman, N.; Greenland, A.J.; Horsnell, R.; Howells, R.; O’Sullivan, D.M.; et al. An eight–parent multiparent advanced generation inter–cross population for winter–sown wheat: Creation, properties, and validation. G3: Genes Genom. Genet. 2011, 4, 1603–1610. [Google Scholar] [CrossRef] [Green Version]
- Fradgley, N.; Bentley, A.R.; Gardner, K.A.; Howell, P.; Mackay, I.J.; Scott, M.; Mott, R.; Cockram, J. A 16 Founder Wheat MAGIC Population for QTL Mapping. In Proceedings of the Plant and Animal Genome XXVI Conference, San Diego, CA, USA, 13–17 January 2018. [Google Scholar]
- Huang, B.E.; Verbyla, K.L.; Verbyla, A.P.; Raghavan, C.; Singh, V.K.; Gaur, P.; Leung, H.; Varshney, R.K.; Cavanagh, C.R. MAGIC populations in crops: Current status and future prospects. Theor. Appl. Genet. 2015, 128, 999–1017. [Google Scholar] [CrossRef] [PubMed]
- Stadlmeier, M.; Hartl, L.; Mohler, V. Usefulness of a Multiparent Advanced Generation Intercross Population with a greatly reduced mating design for genetic studies in winter wheat. Front. Plant Sci. 2018, 9, 1825. [Google Scholar] [CrossRef] [Green Version]
- Delhaize, E.; Rathjen, T.M.; Cavanagh, C.R. The genetics of rhizosheath size in a multiparent mapping population of wheat. J. Exp. Bot. 2015, 66, 4527–4536. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rebetzke, G.J.; Verbyla, A.P.; Verbyla, K.L.; Morell, M.K.; Cavanagh, C.R. Use of a large multiparent wheat mapping population in genomic dissection of coleoptile and seedling growth. Plant Biotechnol. J. 2014, 12, 219–230. [Google Scholar] [CrossRef] [PubMed]
- Barrero, J.M.; Cavanagh, C.; Verbyla, K.L.; Tibbits, J.F.; Verbyla, A.P.; Huang, B.E.; Rosewarne, G.M.; Stephen, S.; Wang, P.; Whan, A.; et al. Transcriptomic analysis of wheat near–isogenic lines identifies PM19–A1 and A2 as candidates for a major dormancy QTL. Genome Biol. 2015, 16, 1–18. [Google Scholar] [CrossRef] [Green Version]
- Boden, S.A.; Cavanagh, C.; Cullis, B.R.; Ramm, K.; Greenwood, J.; Finnegan, E.J.; Trevaskis, B.; Swain, S.M. Ppd–1 is a key regulator of inflorescence architecture and paired spikelet development in wheat. Nat. Plants 2015, 1, 1–6. [Google Scholar] [CrossRef]
- Richards, R.A.; Cavanagh, C.R.; Riffkin, P. Selection for erect canopy architecture can increase yield and biomass of spring wheat. Field Crop. Res. 2019, 244, 107649. [Google Scholar] [CrossRef]
- Camargo, A.V.; Mackay, I.; Mott, R.; Han, J.; Doonan, J.H.; Askew, K.; Corke, F.; Williams, K.; Bentley, A.R. Functional mapping of quantitative trait loci (QTLs) associated with plant performance in a wheat magic mapping population. Front. Plant Sci. 2018, 9, 887. [Google Scholar] [CrossRef] [PubMed]
- Riaz, A.; KockAppelgren, P.; Hehir, J.G.; Kang, J.; Meade, F.; Cockram, J.; Milbourne, D.; Spink, J.; Mullins, E.; Byrne, S. Genetic analysis using a multi–parent wheat population identifies novel sources of Septoria tritici blotch resistance. Genes 2020, 11, 887. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.; Corsi, B.; Ficke, A.; Tan, K.C.; Cockram, J.; Lillemo, M. Genetic mapping using a wheat multi–founder population reveals a locus on chromosome 2A controlling resistance to both leaf and glume blotch caused by the necrotrophic fungal pathogen Parastagonospora nodorum. Theor. Appl. Genet. 2020, 133, 785–808. [Google Scholar] [CrossRef] [Green Version]
- Gregory, P.J.; Eastham, J. Growth of shoots and roots, and interception of radiation by wheat and lupin crops on a shallow, duplex soil in response to time of sowing. Aust. J. Agric. Res. 1996, 47, 427–447. [Google Scholar] [CrossRef]
- Thorup-Kristensen, K.; Cortasa, M.S.; Loges, R. Winter wheat roots grow twice as deep as spring wheat roots, is this important for N uptake and N leaching losses? Plant Soil 2009, 322, 101–114. [Google Scholar] [CrossRef]
- Tracy, S.R.; Nagel, K.A.; Postma, J.A.; Fassbender, H.; Wasson, A.; Watt, M. Crop improvement from phenotyping roots: Highlights reveal expanding opportunities. Trends Plant Sci. 2020, 25, 105–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, J. Root architecture and plant productivity. Plant Physiol. 1995, 109, 7. [Google Scholar] [CrossRef]
- Rich, S.M.; Watt, M. Soil conditions and cereal root system architecture: Review and considerations for linking Darwin and Weaver. J. Exp. Bot. 2013, 64, 1193–1208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Severini, A.D.; Wasson, A.P.; Evans, J.R.; Richards, R.A.; Watt, M. Root phenotypes at maturity in diverse wheat and triticale genotypes grown in three field experiments: Relationships to shoot selection, biomass, grain yield, flowering time, and environment. Field Crop. Res. 2020, 255, 107870. [Google Scholar] [CrossRef]
- Asif, M.; Kamran, A. Plant breeding for water–limited environments. Crop. Sci. 2011, 51, 2911–2912. [Google Scholar] [CrossRef]
- Carvalho, P.; Azam-Ali, S.; Foulkes, M.J. Quantifying relationships between rooting traits and water uptake under drought in Mediterranean barley and durum wheat. J. Integr. Plant Biol. 2014, 56, 455–469. [Google Scholar] [CrossRef] [PubMed]
- King, J.; Gay, A.; Sylvester-Bradley, R.; Bingham, I.; Foulkes, J.; Gregory, P.; Robinson, D. Modelling cereal root systems for water and nitrogen capture: Towards an economic optimum. Ann. Bot. 2003, 91, 383–390. [Google Scholar] [CrossRef] [Green Version]
- Rich, S.M.; Christopher, J.; Richards, R.; Watt, M. Root phenotypes of young wheat plants grown in controlled environments show inconsistent correlation with mature root traits in the field. J. Exp. Bot. 2020, 71, 4751–4762. [Google Scholar] [CrossRef]
- Watt, M.; Magee, L.J.; McCully, M.E. Types, structure and potential for axial water flow in the deepest roots of field-grown cereals. New Phytol. 2008, 178, 135–146. [Google Scholar] [CrossRef]
- Xie, Q.; Fernando, K.M.; Mayes, S.; Sparkes, D.L. Identifying seedling root architectural traits associated with yield and yield components in wheat. Ann. Bot. 2017, 119, 1115–1129. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Zarebanadkouki, M.; Kaestner, A.; Carminati, A. Measurements of water uptake of maize roots: The key function of lateral roots. Plant Soil 2016, 398, 59–77. [Google Scholar] [CrossRef]
- Varney, G.; Canny, M. Rates of water uptake into the mature root system of maize plants. New Phytol. 1993, 123, 775–786. [Google Scholar] [CrossRef]
- Gioia, T.; Galinski, A.; Lenz, H.; Müller, C.; Lentz, J.; Muller, C.; Lentz, J.; Heinz, K.; Briese, C.; Putz, A.; et al. GrowScreen–PaGe, a non–invasive, high–throughput phenotyping system based on germination paper to quantify crop phenotypic diversity and plasticity of root traits under varying nutrient supply. Funct. Plant Biol. 2016, 44, 76–93. [Google Scholar] [CrossRef]
- Richards, R.A.; Rebetzke, G.J.; Watt, M.; Condon, A.T.; Spielmeyer, W.; Dolferus, R. Breeding for improved water productivity in temperate cereals: Phenotyping, quantitative trait loci, markers and the selection environment. Funct. Plant Biol. 2010, 37, 85–97. [Google Scholar] [CrossRef]
- Wissuwa, M.; Kretzschmar, T.; Rose, T.J. From promise to application: Root traits for enhanced nutrient capture in rice breeding. J. Exp. Bot. 2016, 67, 3605–3615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watt, M.; Fiorani, F.; Usadel, B.; Rascher, U.; Muller, O.; Schurr, U. Phenotyping: New Windows into the Plant for Breeders. Annu. Rev. Plant Biol. 2020, 71, 689–712. [Google Scholar] [CrossRef] [PubMed]
- Wasson, A.P.; Nagel, K.A.; Tracy, S.; Watt, M. Beyond Digging: Noninvasive Root and Rhizosphere Phenotyping. Trends Plant Sci. 2020, 25, 119–120. [Google Scholar] [CrossRef] [Green Version]
- Hoagland, D.R.; Arnon, D.I. The water-culture method for growing plants without soil. In Circular, 2nd ed.; California Agricultural Experiment Station: St. Thomas, VI, USA, 1950; Volume 347, pp. 1–32. [Google Scholar]
- Falconer, D.; Mackay, T. Introduction to Quantitative Genetics; Harlow, Ed.; Longmans Green: Essex, UK, 1996. [Google Scholar]
- Holland, J.B.; Nyquist, W.E.; Cervantes-Martínez, C.T. Estimating and interpreting heritability for plant breeding: An update. Plant Breed. 2003, 22, 9–112. [Google Scholar]
- Stoffel, M.A.; Nakagawa, S.; Schielzeth, H. rptR: Repeatability estimation and variance decomposition by generalized linear mixed-effects models. Methods Ecol. Evol. 2017, 11, 1639–1644. [Google Scholar] [CrossRef] [Green Version]
- RCore, Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016; Available online: https://www.r-project.org/ (accessed on 14 February 2019).
- Pang, J.; Palta, J.A.; Rebetzke, G.J.; Milroy, S.P. Wheat genotypes with high early vigour accumulate more nitrogen and have higher photosynthetic nitrogen use efficiency during early growth. Funct. Plant Biol. 2013, 41, 215–222. [Google Scholar] [CrossRef]
- Watt, M.; Kirkegaard, J.A.; Rebetzke, G.J. A wheat genotype developed for rapid leaf growth copes well with the physical and biological constraints of unploughed soil. Funct. Plant Biol. 2005, 32, 695–706. [Google Scholar] [CrossRef]
- Richard, O.; Pineau, C.; Loubet, S.; Chalies, C.; Vile, D.; Marquès, L.; Berthomieu, P. Diversity analysis of the response to Zn within the Arabidopsis thaliana species revealed a low contribution of Zn translocation to Zn tolerance and a new role for Zn in lateral root development. Plant Cell Environ. 2011, 34, 1065–1078. [Google Scholar] [CrossRef]
- Dhanda, S.; Sethi, G.; Behl, R. Indices of drought tolerance in wheat genotypes at early stages of plant growth. J. Agron. Crop. Sci. 2004, 190, 6–12. [Google Scholar] [CrossRef]
- Laperche, A.; Devienne-Barret, F.; Maury, O.; Le Gouis, J.; Ney, B. A simplified conceptual model of carbon/nitrogen functioning for QTL analysis of winter wheat adaptation to nitrogen deficiency. Theor. Appl. Genet. 2006, 113, 1131–1146. [Google Scholar] [CrossRef] [PubMed]
- MacMillan, K.; Emrich, K.; Piepho, H.P.; Mullins, C.; Price, A. Assessing the importance of genotype x environment interaction for root traits in rice using a mapping population. I: A soil–filled box screen. Theor. Appl. Genet. 2006, 113, 977–986. [Google Scholar] [CrossRef]
- Ao, J.; Fu, J.; Tian, J.; Yan, X.; Liao, H. Genetic variability for root morph–architecture traits and root growth dynamics as related to phosphorus efficiency in soybean. Funct. Plant Biol. 2010, 37, 304–312. [Google Scholar] [CrossRef]
- Rebetzke, G.J.; Richards, R.A. Genetic improvement of early vigour in wheat. Aust. J. Agric. Res. 1999, 50, 291–301. [Google Scholar] [CrossRef]
- Tsegaye, D.; Dessalegn, T.; Dessalegn, Y.; Share, G. Genetic variability, correlation and path analysis in durum wheat germplasm (Triticum durum Desf). Agric. Res. Rev. 2012, 1, 107–112. [Google Scholar]
- Palta, J.A.; Watt, M. Vigorous crop root systems: Form and function for improving the capture of water and nutrients. In Applications for Genetic Improvement and Agronomy; Sadras, V., Calderini, D., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; pp. 309–325. [Google Scholar]
- Fradgley, N.; Evans, G.; Biernaskie, J.M.; Cockram, J.S.; Marr, E.C.; Oliver, A.G.; Ober, E.; Jones, H. Effects of breeding history and crop management on the root architecture of wheat. Plant Soil 2020, 452, 587–600. [Google Scholar] [CrossRef]
- Camargo, A.V.; Mott, R.; Gardner, K.A.; Mackay, I.J.; Corke, F.; Doonan, J.H.; Kim, J.T.; Bentley, A.R. Determining phenological patterns associated with the onset of senescence in a wheat MAGIC mapping population. Front. Plant Sci. 2016, 7, 1540. [Google Scholar] [CrossRef] [Green Version]
- Subbiah, B.; Katyal, J.; Narasimham, R.; Dakshinamurti, C. Preliminary investigations on root distribution of high yielding wheat varieties. Int. J. Appl. Radiat. Isotopes 1968, 19, 385–390. [Google Scholar] [CrossRef]
- Botwright, T.; Rebetzke, G.; Condon, A.; Richards, R. Influence of the gibberellin–sensitive Rht8 semi–dwarf gene on leaf anatomy and seedling vigour in wheat (Triticum aestivum L.). Ann. Bot. 2005, 95, 631–639. [Google Scholar] [CrossRef]
- Ellis, M.H.; Rebetzke, G.J.; Chandler, P.; Bonnett, D.; Spielmeyer, W.; Richards, R.A. The effect of different height reducing genes on the early growth of wheat. Funct. Plant Biol. 2004, 31, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Rebetzke, G.J.; Ellis, M.H.; Bonnett, D.G.; Richards, R.A. Molecular mapping of genes for coleoptile growth in bread wheat (Triticum aestivum L.). Theor. Appl. Genet. 2007, 114, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Maydup, M.L.; Graciano, C.; Guiamet, J.J.; Tambussi, E.A. Analysis of early vigour in twenty modern cultivars of bread wheat (Triticum aestivum L.). Crop. Pasture Sci. 2012, 63, 987–996. [Google Scholar] [CrossRef]
- Moore, C.; Rebetzke, G. Genomic regions for embryo size and early vigour in multiple wheat (Triticum aestivum L.) populations. Agronomy 2015, 5, 152–179. [Google Scholar] [CrossRef] [Green Version]
- Von Wangenheim, D.; Banda, J.; Schmitz, A.; Boland, J.; Bishopp, A.; Maizel, A.; Stelzer, E.H.K.; Bennett, M. Early developmental plasticity of lateral roots in response to asymmetric water availability. Nat. Plants 2020, 6, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Pflugfelder, D.; Kochs, J.; Koller, R.; Jahnke, S.; Mohl, C.; Pariyar, S.R.; Fassbender, H.; Nagel, K.A.; Watt, M.; van Dusschoten, D. The root system architecture of wheat establishing in soil is associated with varying elongation rates of seminal roots. J. Exp. Bot. (under review).
- Manschadi, A.M.; Hammer, G.L.; Christopher, J.T.; Devoil, P. Genotypic variation in seedling root architectural traits and implications for drought adaptation in wheat (Triticum aestivum L.). Plant Soil 2008, 303, 115–129. [Google Scholar] [CrossRef]
- Nakamoto, T.; Oyanagi, A. The direction of growth of seminal roots of Triticum aestivum L. and experimental modification thereof. Ann. Bot. 1994, 73, 363–367. [Google Scholar] [CrossRef]
- Oyanagi, A. Gravitropic response growth angle and vertical distribution of roots of wheat (Triticum aestivum L.). Plant Soil 1994, 165, 323–326. [Google Scholar] [CrossRef]
- Watt, M.; Silk, W.K.; Passioura, J.B. Rates of root and organism growth, soil conditions, and temporal and spatial development of the rhizosphere. Ann. Bot. 2006, 97, 839–855. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watt, M.; McCully, M.E.; Kirkegaard, J.A. Soil strength and rate of root elongation alter the accumulation of Pseudomonas spp. and other bacteria in the rhizosphere of wheat. Funct. Plant Biol. 2003, 30, 483–491. [Google Scholar] [CrossRef]
- Watt, M.; Moosavi, S.; Cunningham, S.C.; Kirkegaard, J.A.; Rebetzke, G.J.; Richards, R.A. A rapid, controlled–environment seedling root screen for wheat correlates well with rooting depths at vegetative, but not reproductive, stages at two field sites. Ann. Bot. 2013, 112, 447–455. [Google Scholar] [CrossRef] [Green Version]
- Rambla, C.; Van Der Meer, S.; Voss-Fels, K.P.; Makhoul, M.; Obermeier, C.; Snowdon, R.; Ober, E.S.; Watt, M.; Alahmad, S.; Hickley, L.T. A Toolkit to Rapidly Modify Root Systems Through Single Plant Selection. Ann. Bot. (accepted; in press).
- Palta, J.; Gregory, P. Drought affects the fluxes of carbon to roots and soil in 13C pulse–labelled plants of wheat. Soil Biol. Biochem. 1997, 29, 1395–1403. [Google Scholar] [CrossRef]
- Palta, J.A.; Chen, X.; Milroy, S.P.; Rebetzke, G.J.; Dreccer, M.F.; Watt, M. Large root systems: Are they useful in adapting wheat to dry environments? Funct. Plant Biol. 2011, 38, 347–354. [Google Scholar] [CrossRef] [Green Version]
- Rahnama, A.; Munns, R.; Poustini, K.; Watt, M. A screening method to identify genetic variation in root growth response to a salinity gradient. J. Exp. Bot. 2011, 62, 69–77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schillaci, M.; Arsova, B.; Walker, R.; Smith, P.M.; Nagel, K.A.; Roessner, U.; Watt, M. Time–resolution of the shoot and root growth of the model cereal Brachypodium in response to inoculation with Azospirillum bacteria at low phosphorus and temperature. Plant Growth Regul. 2021, 93, 149–162. [Google Scholar] [CrossRef]
- Volkmar, K.M. Water stressed nodal roots of wheat: Effects on leaf growth. Funct. Plant Biol. 1997, 24, 49–56. [Google Scholar] [CrossRef]
- Chochois, V.; Vogel, J.P.; Rebetzke, G.J.; Watt, M. Variation in adult plant phenotypes and partitioning among seed and stem–borne roots across Brachypodium distachyon accessions to exploit in breeding cereals for well–watered and drought environments. Plant Physiol. 2015, 168, 953–967. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rostamza, M.; Richards, R.A.; Watt, M. Response of millet and sorghum to a varying water supply around the primary and nodal roots. Ann. Bot. 2013, 112, 439–446. [Google Scholar] [CrossRef] [PubMed]
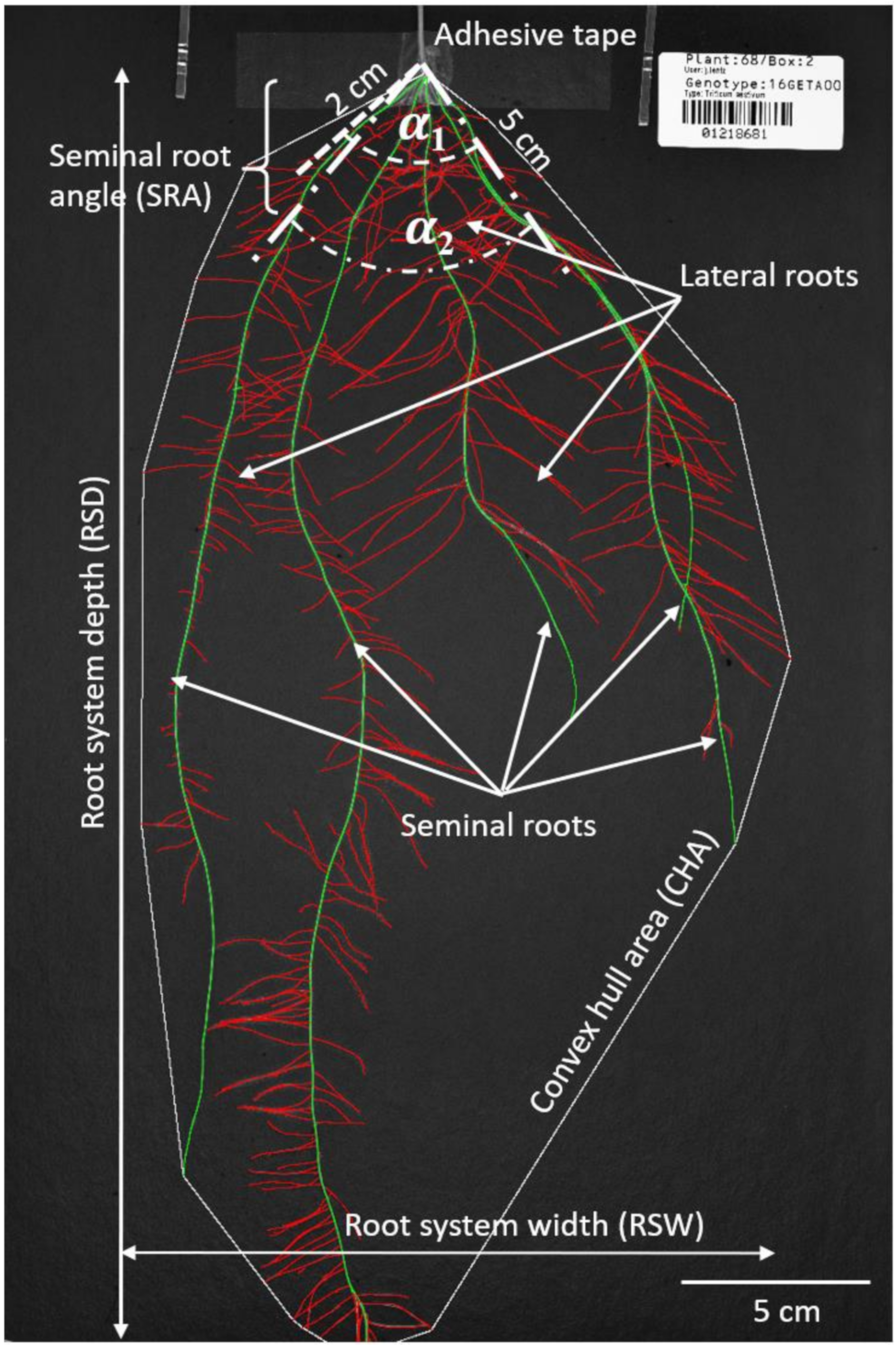
| Trait | Description | Unit |
|---|---|---|
| First seminal root (FSR) | Length of radicle (measured 1 day after germination) | mm |
| Seminal root length (SRL) | Length of all seminal roots | cm |
| Lateral root length (LRL) | Length of all roots branched from seminal roots | cm |
| Total root length (TRL) | Total sum of seminal and lateral root length | cm |
| Root system depth (RSD) | Maximal vertical depth of a root system | cm |
| Root system width (RSW) | Maximal horizontal distribution of a root system | cm |
| Convex hull area (CHA) | Area of the convex hull that encompasses the root system | cm2 |
| Seminal root angle (SRA) | Angle between the outermost left and right seminal roots measured at 2 or 5 cm distance from seed respectively | ° |
| Leaf length (LeafL) | Length between the seed and tip of the longest leaf | cm |
| Number of leaves (LeafNo) | Number of leaves (without cotyledons) | - |
| Root dry weight (RDWeight) | Root dry weight | g |
| Shoot dry weight (SDWeight) | Shoot dry weight | g |
| Root mass ratio (RMR) | Dry mass of root divided by the total dry mass of entire plant | g/g |
| Shoot mass ratio (SMR) | Dry mass of shoot divided by the total dry mass of entire plant | g/g |
| Trait | Heritability (H2) | Repeatability (r) | |
|---|---|---|---|
| MAGIC Founder Parents | MAGIC Founder Parents | ||
| NIAB | CSIRO | ||
| First seminal root (1 DAG) | 0.43 | 0.64 | 0.36 |
| Seminal root length (2 DAT) | 0.29 | 0.32 | 0.58 |
| Seminal root length (9 DAT) | 0.20 | 0.44 | 0.54 |
| Seminal root length (16 DAT) | 0.23 | 0.35 | 0.44 |
| Lateral root length (9 DAT) | 0.40 | 0.22 | 0.34 |
| Lateral root length (16 DAT) | 0.36 | 0.38 | 0.65 |
| Total root length (2 DAT) | 0.29 | 0.32 | 0.58 |
| Total root length (9 DAT) | 0.36 | 0.19 | 0.44 |
| Total root length (16 DAT) | 0.35 | 0.40 | 0.67 |
| Root system depth (2 DAT) | 0.38 | 0.07 | 0.30 |
| Root system depth (9 DAT) | 0.30 | 0.19 | 0.42 |
| Root system depth (16 DAT) | 0.12 | - | 0.10 |
| Root system width (2 DAT) | 0.25 | 0.12 | 0.24 |
| Root system width (9 DAT) | 0.24 | 0.30 | 0.34 |
| Root system width (16 DAT) | 0.16 | 0.29 | 0.23 |
| Seminal root angle 2cm (2 DAT) | - | 0.06 | 0.23 |
| Seminal root angle 2cm (9 DAT) | 0.06 | 0.10 | 0.14 |
| Seminal root angle 2cm (16 DAT) | - | 0.22 | 0.06 |
| Seminal root angle 5cm (9 DAT) | 0.41 | 0.10 | 0.08 |
| Seminal root angle 5cm (16 DAT) | 0.30 | 0.20 | 0.21 |
| Convex hull area (2 DAT) | 0.39 | 0.13 | 0.33 |
| Convex hull area (9 DAT) | 0.23 | 0.24 | 0.32 |
| Convex hull area (16 DAT) | 0.12 | 0.26 | 0.19 |
| Leaf length (16 DAT) | 0.60 | 0.84 | 0.58 |
| Leaf number (16 DAT) | 0.67 | 0.61 | 0.42 |
| Root dry weight (16 DAT) | 0.25 | 0.50 | 0.34 |
| Shoot dry weight (16 DAT) | 0.37 | 0.50 | 0.38 |
| MAGIC Wheat | Founder Parents | Root Dry Weight (g) | Shoot Dry Weight (g) | Root Mass Ratio | Shoot Mass Ratio |
|---|---|---|---|---|---|
| NIAB | Alchemy | 0.022a | 0.021ab | 0.513 | 0.487 |
| Brompton | 0.027a | 0.024a | 0.525 | 0.475 | |
| Claire | 0.021ab | 0.020ab | 0.516 | 0.484 | |
| Hereward | 0.017b | 0.018b | 0.493 | 0.507 | |
| Rialto | 0.019b | 0.019b | 0.492 | 0.508 | |
| Robigus | 0.024a | 0.022ab | 0.516 | 0.484 | |
| Soissons | 0.024a | 0.023a | 0.511 | 0.489 | |
| Xi-19 | 0.021ab | 0.018b | 0.529 | 0.471 | |
| CSIRO | AC Barrie | 0.033a | 0.029a | 0.529 | 0.471 |
| B207 Alsen | 0.030a | 0.031a | 0.493 | 0.507 | |
| C207 Baxter | 0.024ab | 0.021b | 0.53 | 0.47 | |
| Chara7 | 0.026ab | 0.023ab | 0.533 | 0.467 | |
| D204 Pastor | 0.027ab | 0.023ab | 0.526 | 0.474 | |
| F201 Westonia | 0.026ab | 0.024ab | 0.524 | 0.476 | |
| G204 Xiaoyan54 | 0.018b | 0.018b | 0.497 | 0.503 | |
| H5 Yitpi | 0.026ab | 0.026ab | 0.501 | 0.499 | |
| Volcani | 0.028a | 0.024ab | 0.533 | 0.467 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pariyar, S.R.; Nagel, K.A.; Lentz, J.; Galinski, A.; Wilhelm, J.; Putz, A.; Adels, S.; Heinz, K.; Frohberg, C.; Watt, M. Variation in Root System Architecture among the Founder Parents of Two 8-way MAGIC Wheat Populations for Selection in Breeding. Agronomy 2021, 11, 2452. https://doi.org/10.3390/agronomy11122452
Pariyar SR, Nagel KA, Lentz J, Galinski A, Wilhelm J, Putz A, Adels S, Heinz K, Frohberg C, Watt M. Variation in Root System Architecture among the Founder Parents of Two 8-way MAGIC Wheat Populations for Selection in Breeding. Agronomy. 2021; 11(12):2452. https://doi.org/10.3390/agronomy11122452
Chicago/Turabian StylePariyar, Shree R., Kerstin A. Nagel, Jonas Lentz, Anna Galinski, Jens Wilhelm, Alexander Putz, Sascha Adels, Kathrin Heinz, Claus Frohberg, and Michelle Watt. 2021. "Variation in Root System Architecture among the Founder Parents of Two 8-way MAGIC Wheat Populations for Selection in Breeding" Agronomy 11, no. 12: 2452. https://doi.org/10.3390/agronomy11122452
APA StylePariyar, S. R., Nagel, K. A., Lentz, J., Galinski, A., Wilhelm, J., Putz, A., Adels, S., Heinz, K., Frohberg, C., & Watt, M. (2021). Variation in Root System Architecture among the Founder Parents of Two 8-way MAGIC Wheat Populations for Selection in Breeding. Agronomy, 11(12), 2452. https://doi.org/10.3390/agronomy11122452






