Impact of the Legume Catch Crop Serradella on Subsequent Growth and P Mobilization under Barley in Different Fertilization Treatments
Abstract
:1. Introduction
2. Materials and Methods
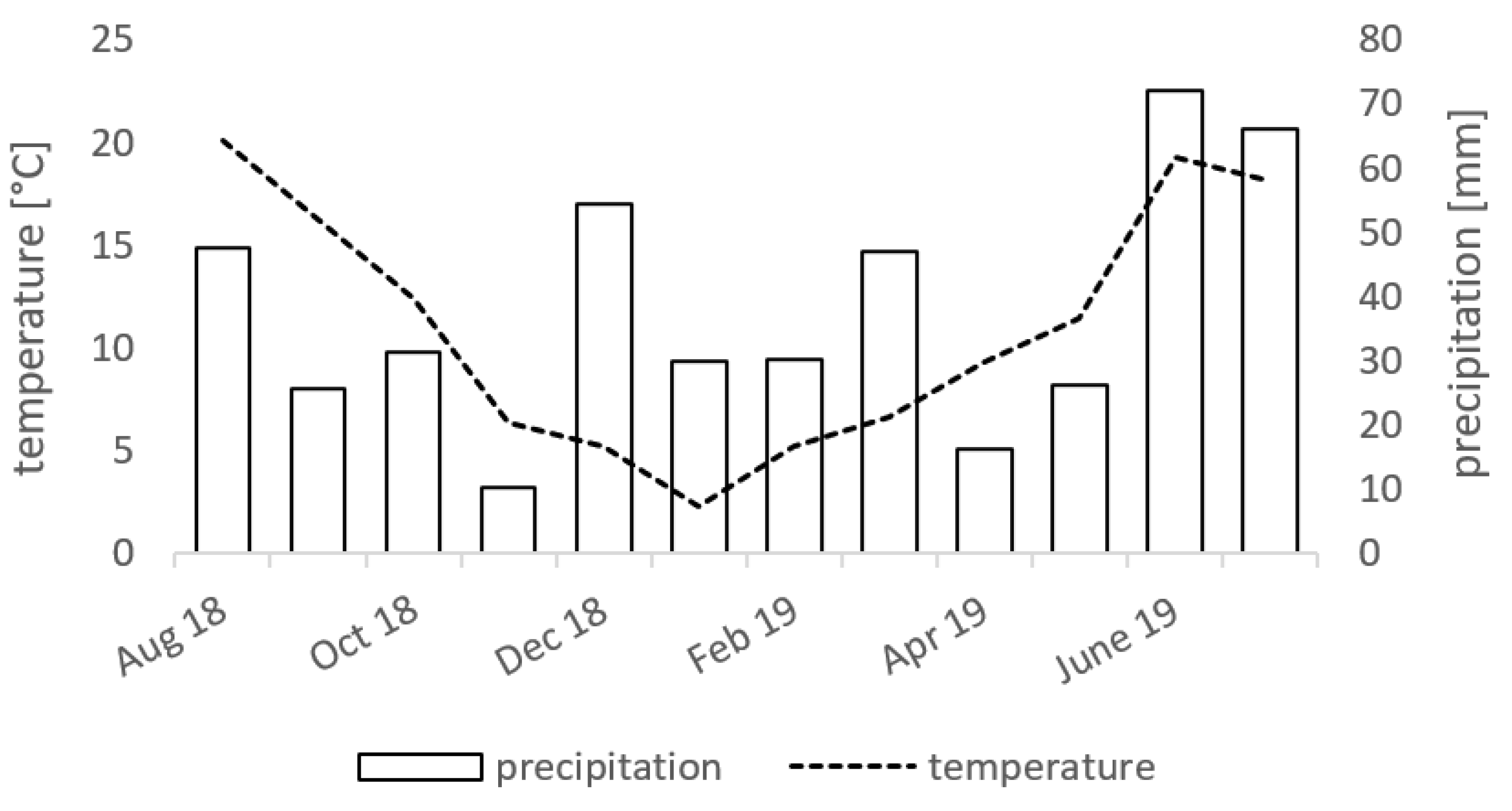
2.1. Experimental Design and Sampling
2.2. Analyses of Carbon, Nitrogen and Phosphorus in Plant and Soil
2.3. Analyses of Mycorrhizal Colonization and Soil Enzyme Activities
2.4. Statistical Analyses
3. Results
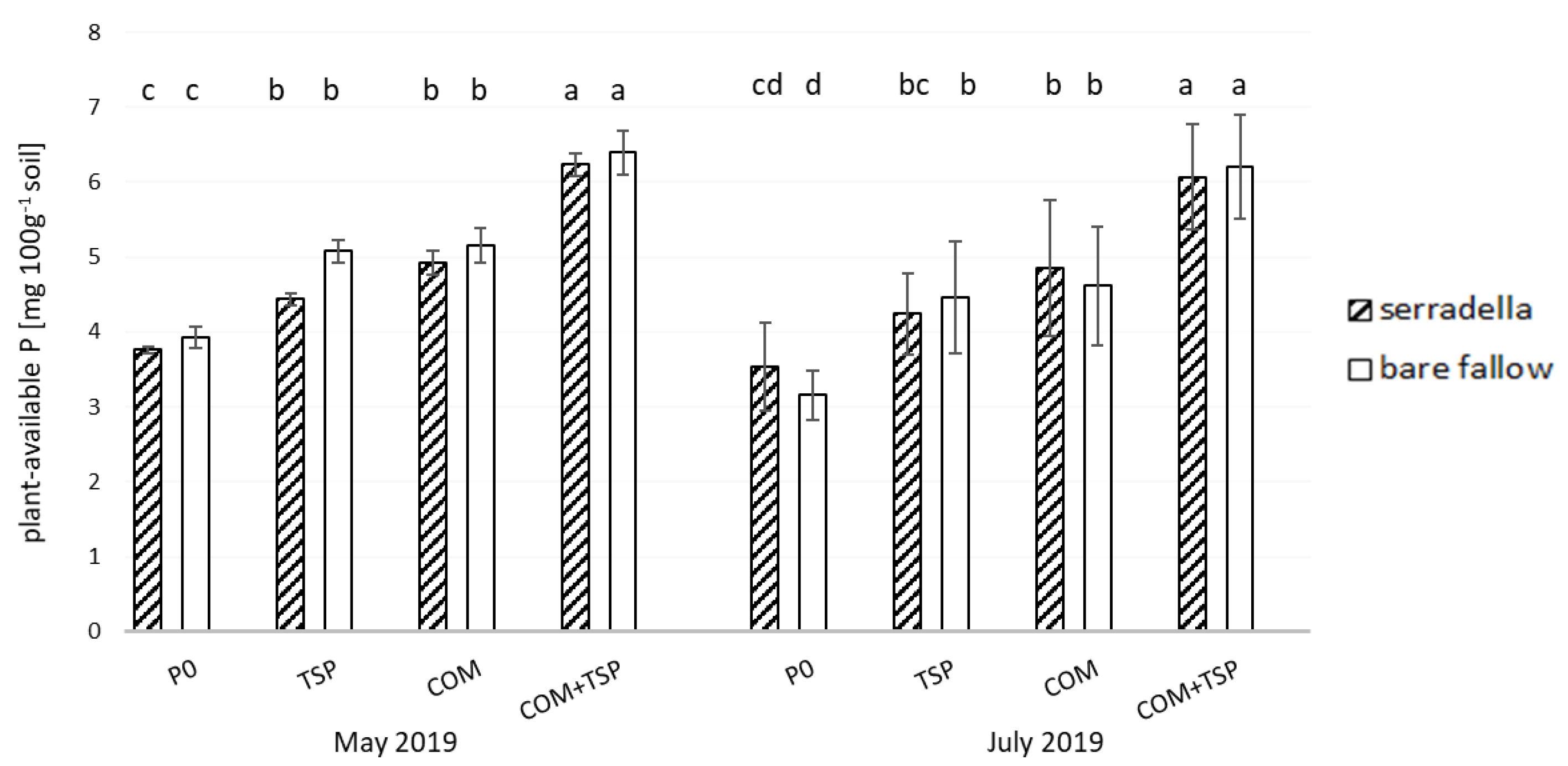
3.1. Plant-Available P Content in the Soil
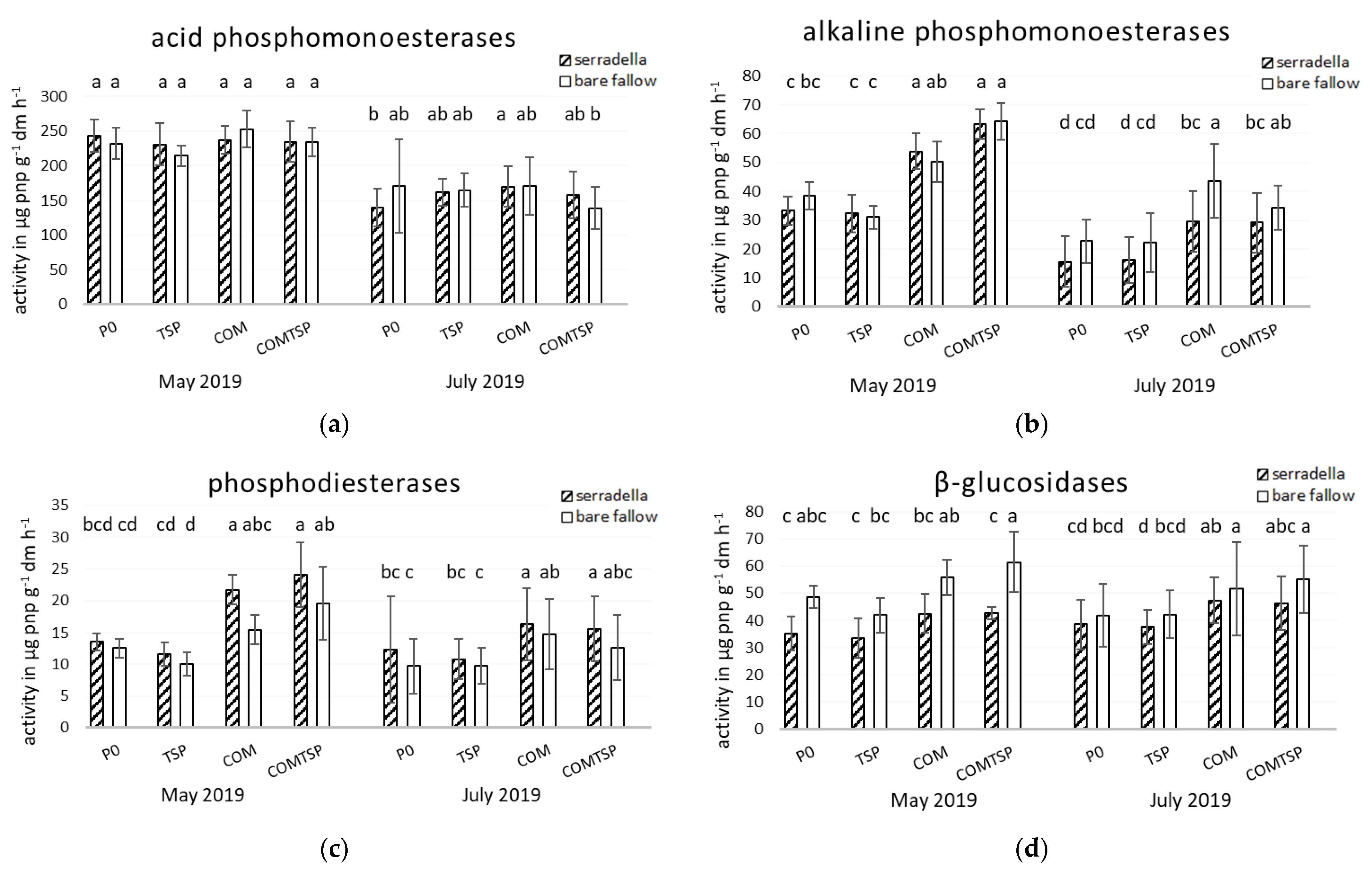
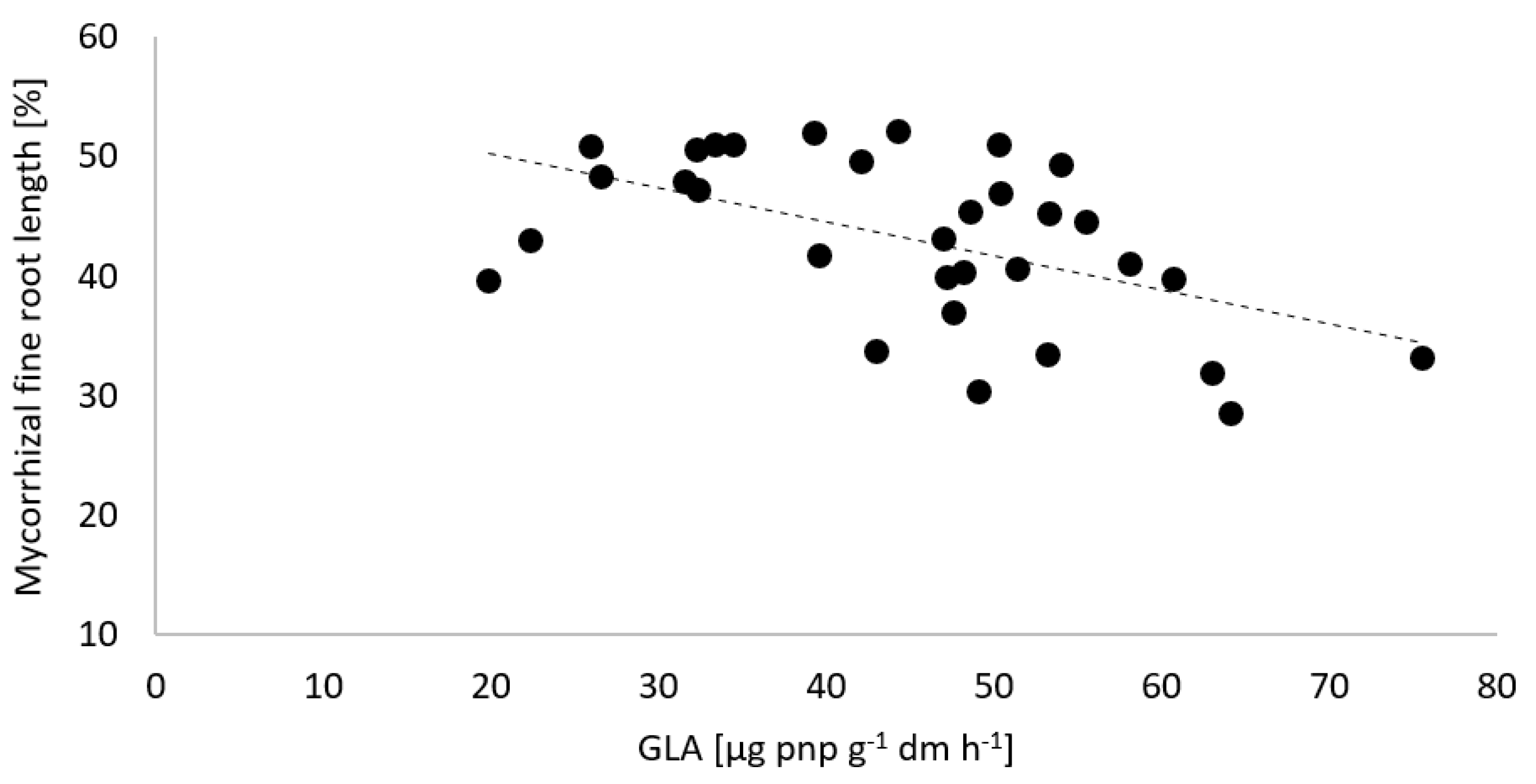
3.2. Potential Soil Enzyme Activities and Mycorrhiza Formation
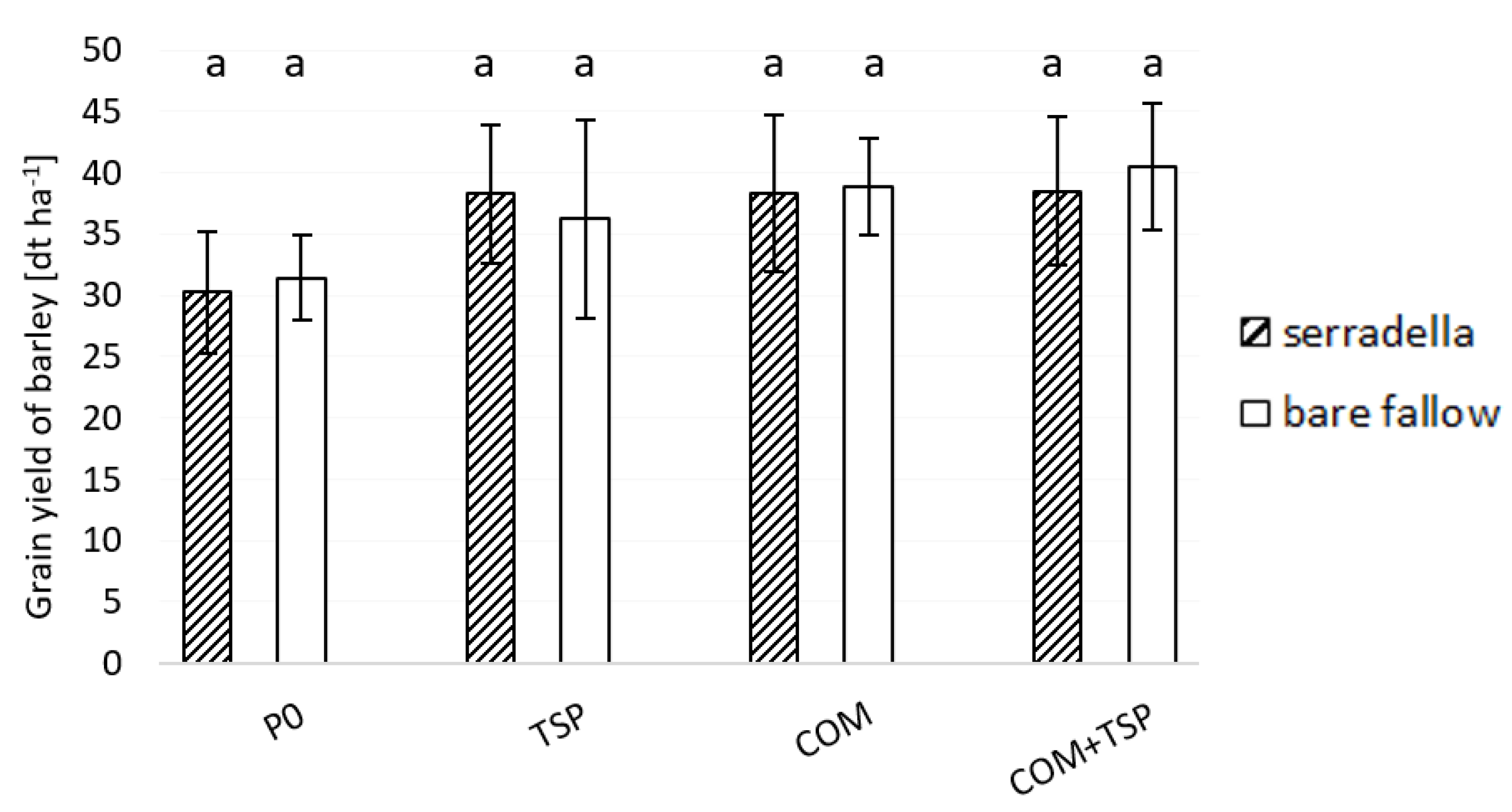
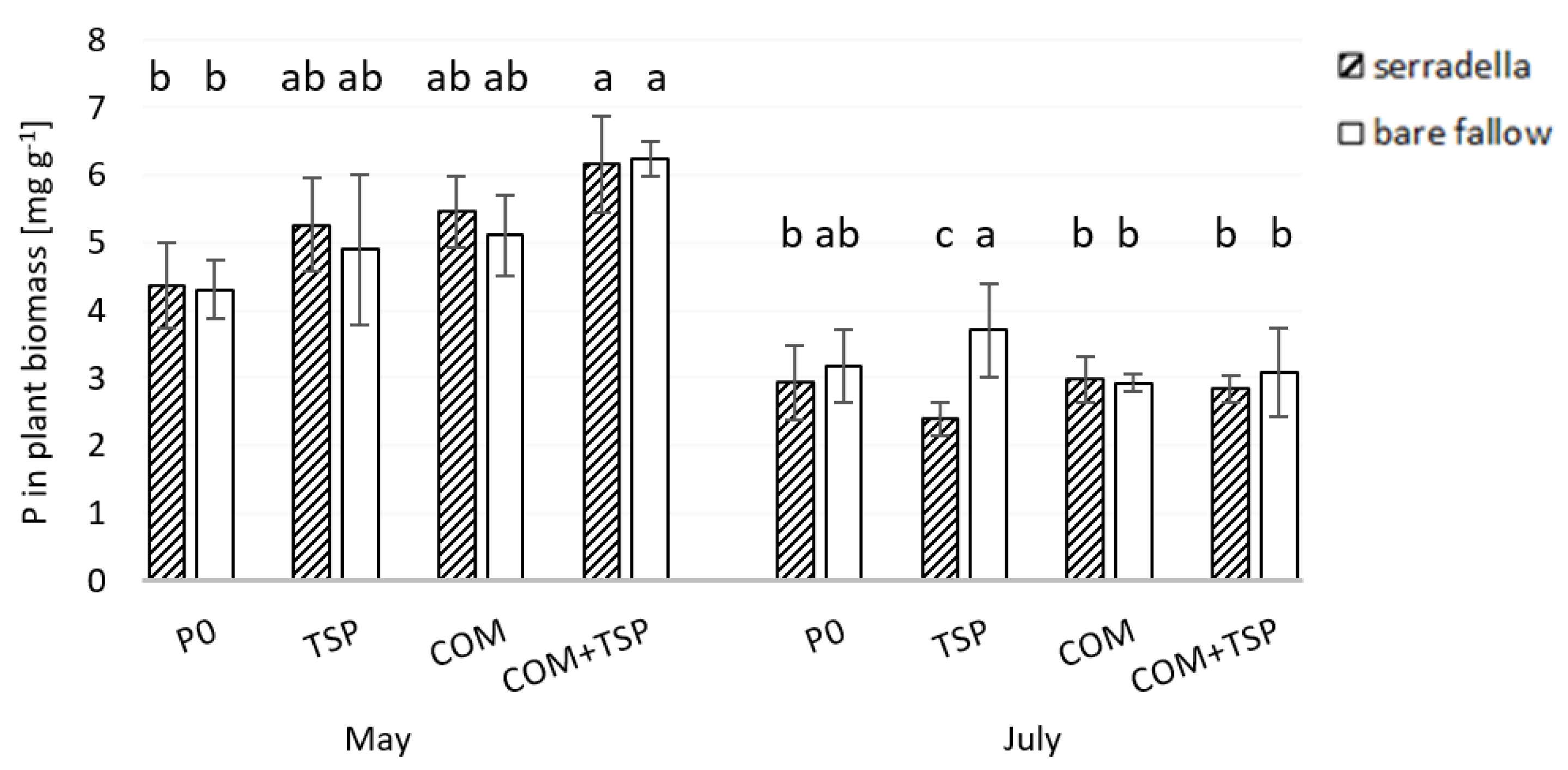
3.3. Grain Yield and P Content in the Shoot of Spring Barley
4. Discussion
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mengel, K.; Kirkby, E.A. Principles of Plant Nutrition: Phosphorus, 5th ed.; Kosegarten, H., Appel, T., Eds.; Kluwer Academic Publishers: Amsterdam, The Netherlands, 2001; pp. 453–479. [Google Scholar]
- Mengel, K. Agronomic measures for better utilization of soil and fertilizer phosphates. Eur. J. Agron. 1997, 7, 277–289. [Google Scholar] [CrossRef]
- Mar, S.S.; Okazaki, M. Investigation of Cd contents in several phosphate rocks used for the production of fertilizer. Microchem. J. 2012, 104, 17–21. [Google Scholar] [CrossRef]
- Kratz, S.; Schick, J.; Schnug, E. Trace elements in rock phosphates and P containing mineral and organo-mineral fertilizers sold in Germany. Sci. Total Environ. 2016, 542, 1013–1019. [Google Scholar] [CrossRef] [PubMed]
- Redecker, D.; Kodner, R.; Graham, L.E. Glomalean Fungi from the Ordovician. Sci. Total Environ. 2000, 289, 1920–1921. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Helgason, T.; Fitter, A.H. Natural selection and the evolutionary ecology of the arbuscular mycorrhizal fungi (Phylum Glomeromycota). J. Exp. Bot. 2009, 60, 2465–2480. [Google Scholar] [CrossRef] [Green Version]
- Hou, L.; Zhang, X.; Feng, G.; Zheng, L.; Zhang, Y.; Cao, N. Arbuscular mycorrhizal enhancement of phosphorus uptake and yields of maize under high planting density in the black soil region of China. Sci. Rep. 2021, 11, 1–11. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D. Mycorrhizal Symbiosis, 3rd ed.; Academic Press: San Diego, CA, USA, 2008; pp. 17–144. [Google Scholar]
- Miransari, M. Contribution of arbuscular mycorrhizal symbiosis to plant growth under different types of soil stress. Plant. Biol. 2010, 12, 563–569. [Google Scholar] [CrossRef]
- Smith, S.; Facelli, E.; Pope, S.; Andrew Smith, F. Plant performance in stressful environments; interpreting new and established knowledge of the roles of arbuscular mycorrhizas. Plant Soil 2010, 326, 3–20. [Google Scholar] [CrossRef]
- Smith, S.E.; Smith, F.A. Roles of arbuscular mycorrhizas in plant nutrition and growth: New paradigms from cellular to ecosystems scales. Annu. Rev. Plant Biol. 2011, 63, 227–250. [Google Scholar] [CrossRef] [Green Version]
- Łukowiak, R.; Grzebisz, W.; Sassenrathc, G.F. New insights into phosphorus management in agriculture—A crop rotation approach. Sci. Total Environ. 2016, 542, 1062–1077. [Google Scholar] [CrossRef] [Green Version]
- Bakhshandeh, S.; Corneo, P.E.; Mariotte, P.; Kertesz, M.A. Effect of crop rotation on mycorrhizal colonization and wheat yield under different fertilizer treatments. Agric. Ecosyst. Environ. 2017, 247, 130–136. [Google Scholar] [CrossRef]
- Eichler-Löbermann, B.; Köhne, S.; Kowalski, B.; Schnug, E. Effect of catch cropping on phosphorus bioavailability in comparison to organic and inorganic fertilization. J. Plant Nutr. 2008, 31, 659–676. [Google Scholar] [CrossRef]
- Talgre, L.; Lauringson, E.; Roostalu, H.; Astover, A.; Makke, A. Green manure as nutrient source for succeeding crops. Plant Soil Environ. 2012, 58, 275–281. [Google Scholar] [CrossRef] [Green Version]
- Meissner, R.; Rupp, H.; Seeger, J.; Schonert, P. Influence of mineral fertilizers and different soils types on nutrient leaching: Results of lysimeter studies in East Germany. Land Degrad. Rehabil. 1995, 6, 163–170. [Google Scholar] [CrossRef]
- Orzech, K. Spring Barley in Pure Sowing and Witch Catch Crops in Crop Rotations. Ph.D. Thesis, University of Warmia and Mazury, Olsztyn, Poland, 2013. [Google Scholar]
- Liu, J.; Bergkvist, G.; Ulen, B. Biomass production and phosphorus retention by catch crops on clayey soils in southern and central Sweden. Field Crop. Res. 2015, 171, 130–137. [Google Scholar] [CrossRef] [Green Version]
- Kling, C.; Doering, T. Manifold green manures—Part V: French Serradella and subterranean clover. Org. Grow. 2014, 26, 32–33. [Google Scholar]
- Stepkowski, T.; Zak, M.; Moulin, L.; Kroliczak, J.; Golinksa, B.; Narozna, D.; Safronova, V.I.; Madrzak, C.J. Bradyrhizobium canariense and Bradyrhizobium japonicum are the two dominant rhizobium species in root nodules of lupin and Serradella plants growing in Europe. Syst. Appl. Microbiol. 2011, 34, 368–375. [Google Scholar] [CrossRef]
- Vargas, L.K.; Volpiano, C.G.; Lisboa, B.B.; Giongo, A.; Beneduzi, A.; Passaglia, L.M.P. Microbes for Legume Improvement, 2nd ed.; Zaidi, A., Khan, M.S., Musarrat, J., Eds.; Springer International Publishing AG: Cham, Switzerland, 2017; pp. 153–174. [Google Scholar] [CrossRef]
- Kidd, D.R.; Ryan, M.H.; Hailing, R.E.; Lambers, H.; Sandral, G.A.; Yang, Z.; Culvenor, R.A.; Cawthray, G.; Stefanski, A.; Simpson, R.J. Rhizosphere carboxylates and morphological root traits in pasture legumes and grasses. Plant Soil 2016, 402, 77–89. [Google Scholar] [CrossRef]
- Hajiboland, R.; Sadeghzadeh, N.; Moradtalab, N.; Aliasgharzad, N.; Schweikert, K.; Poschenrieder, C. The arbuscular mycorrhizal mycelium from barley differentially influences various defense parameters in the non-host sugar beet under co-cultivation. Mycorrhiza 2020, 30, 647–661. [Google Scholar] [CrossRef]
- Njeru, E.M.; Avio, L.; Sbrana, C.; Turrini, A.; Bocci, G.; Barberi, P.; Giovanetti, M. First evidence for a major cover crop effect on arbuscular mycorrhizal fungi and organic maize growth. Agron. Sustain. Dev. 2013, 34, 841–849. [Google Scholar] [CrossRef] [Green Version]
- IUSS Working Group WRB. World Reference Base for Soil Resources 2014, Update 2015 International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; World Soil Resources Reports No. 106; FAO: Rome, Italy, 2015. [Google Scholar]
- Riehm, H. Arbeitsvorschrift zur Bestimmung der Phosphorsäure und des Kaliums nach Lactatverfahren. Z Pflanzenernähr Düng Bodenkd 1948, 40, 152–156. [Google Scholar]
- VDLUFA. Methode A 6.2.1.2—Bestimmung von Phosphor und Kalium im Doppellactat (DL)-Auszug, in Verband Deutscher Landwirtschaftlicher Untersuchungs- und Forschungsanstalten (ed.): VDLUFA-Methodenbuch, Band I, Die Untersuchung von Boden, 4th ed.; VDLUFA-Verlag: Darmstadt, Germany, 2002. [Google Scholar]
- Schachtschabel, P.P.; Beyme, B. Löslichkeit des anorganischen Bodenphosphors und Phosphatdüngung. J. Plant Nutr. Soil Sci. 1980, 143, 306–316. [Google Scholar] [CrossRef]
- Kerschberger, M.; Hege, L.; Jungk, A. Phosphordüngung nach Bodenuntersuchung und Pflanzenbedarf; VDLUFA Standpunkt, VDLUFA Verlag: Darmstadt, Germany, 1997. [Google Scholar]
- Brundrett, M.C.; Piché, Y.; Peterson, R.L. A new method for observing the morphology of vesicular-arbuscular mycorrhizae. Can. J. Bot. 1983, 62, 2128–2134. [Google Scholar] [CrossRef]
- McGonigle, T.P.; Miller, M.H.; Evans, D.G.; Fairchild, G.L.; Swan, J.A. A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol. 1990, 115, 495–501. [Google Scholar] [CrossRef]
- Tabatabai, M.A.; Bremner, J.M. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol. Biochem. 1969, 1, 301–307. [Google Scholar] [CrossRef]
- Browman, M.G.; Tabatabai, M.A. Phosphodiesterase activity of soils. Soil Sci. Soc. Am. J. 1978, 42, 284–290. [Google Scholar] [CrossRef]
- Tabatabai, M.A. Soil Enzymes. In Methods of Soil Analysis. Part 2; Page, A.L., Miller, R.H., Keeney, D.R., Eds.; ASA: Madison, WI, USA, 1982; pp. 903–948. [Google Scholar]
- Eivazi, F.; Tabatabai, M.A. Phosphatases in soils. Soil Biol. Biochem. 1977, 9, 167–172. [Google Scholar] [CrossRef]
- Nannipieri, P.; Johnson, R.L.; Paul, E.A. Criteria for measurement of microbial growth and activity in soil. Soil Biol. Biochem. 1978, 10, 223–229. [Google Scholar] [CrossRef]
- Falkowski, P.G.; Fenchel, T.; Delong, E.F. The microbial engines that drive earth’s biogeochemical cycles. Science 2008, 320, 1034–1039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peine, M.; Vitow, N.; Grafe, M.; Baum, C.; Zicker, T.; Eichler-Löbermann, B.; Schulz, S.; Schloter, M.; Leinweber, P. Effect of triple superphosphate and biowaste compost on mycorrhizal colonization and enzymatic P mobilization under maize in a long-termin field experiment. J. Plant Nutr. Soil Sci. 2019, 182, 167–174. [Google Scholar] [CrossRef]
- Castillo, C.G.; Puccio, F.; Morales, D.; Borie, F.; Sieverding, E. Early arbuscular mycorrhiza colonization of wheat, barley and oats in Andosols of southern Chile. J. Soil. Sci. Plant Nutr. 2012, 12, 511–524. [Google Scholar] [CrossRef]
- Hetrick, B.A.D.; Wilson, G.W.T.; Todd, T.C. Mycorrhizal response in wheat cultivars: Relationship to phosphorus. Can. J. Bot. 1996, 74, 19–25. [Google Scholar] [CrossRef]
- Grace, E.J.; Cotsaftis, O.; Tester, M.; Smith, F.A.; Smith, S.E. Arbuscular mycorrhizal inhibition of growth in barley cannot be attributed to extent of colonization, fungal phosphorus uptake or effects on expression of plant phosphate transporter genes. New Phytol. 2009, 181, 938–949. [Google Scholar] [CrossRef]
- Graham, J.T.; Eissenstat, D.M.; Drouillard, D.L. On the relationship between a plant’s mycorrhizal dependency and rate of vesicular mycorrhizal colonization. Funct. Ecol. 1991, 5, 773–779. [Google Scholar] [CrossRef]
- Poveda, J.; Hermosa, R.; Monte, E.; Nicolás, C. Trichoderma harzianum favours the access of arbuscular mycorrhizal fungi to non-host Brassicaceae roots and increases plant productivity. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hallama, M.; Pekrun, C.; Lambers, H.; Kandeler, E. Hidden miners—the roles of cover crops and soil microorganisms in phosphorus cycling through agroecosystems. Plant Soil 2019, 434, 7–45. [Google Scholar] [CrossRef] [Green Version]
- Hause, B.; Maier, W.; Miersch, O.; Kramell, R.; Strack, D. Induction of jasmonate biosynthesis in arbuscular mycorrhizal barley roots. Plant Physiol. 2002, 130, 1213–1220. [Google Scholar] [CrossRef] [Green Version]
- Kaiser, C.; Kilburn, M.R.; Clode, P.L.; Fuchslueger, L.; Koranda, M.; Cliff, J.B.; Solaiman, Z.M.; Murphy, D.V. Exploring the transfer of recent plant photosynthates to soil microbes: Mycorrhizal pathway vs direct root exudation. New Phytol. 2015, 205, 1537–1551. [Google Scholar] [CrossRef] [PubMed]
- Loeppmann, S.; Breidenbach, A.; Spielvogel, S.; Dippold, M.A.; Blagodatskaya, E. Organic Nutrients Induced Coupled C- and P-Cycling Enzyme Activities During Microbial Growth in Forest Soils. Front. For. Glob. Change 2020, 3–100. [Google Scholar] [CrossRef]
- Trinchera, A.; Ciaccia, C.; Testani, E.; Baratella, V.; Campanelli, G.; Leteo, F.; Canali, S. Mycorrhiza-mediated interference between cover crop and weed in organic winter cereal agroecosystems: The mycorrhizal colonization intensity indicator. Ecol. Evol. 2019, 9, 1–12. [Google Scholar] [CrossRef]
- Dindar, E.; Şağban, F.O.; Başkaya, H.S. Evaluation of soil enzyme activities as soil quality indicators in sludge-amended soils. J. Environ. Biol. 2015, 36, 919–926. [Google Scholar] [PubMed]
- Gianfreda, L.; Ruggiero, P. Enzyme activities in soil. In Nucleic Acids and Proteins in Soil; Nannipieri, P., Smalla, K., Eds.; Springer: Berlin/Heidelberg, Germany, 2006; Volume 8, pp. 257–311. [Google Scholar] [CrossRef]
- Nannipieri, P.; Giagnoni, L.; Landi, L.; Renella, G. Role of phosphatase enzymes in soil. In Phosphorus in Action: Biological Processes in Soil Phosphorus Cycling; Bunemann, E., Oberson, A., Frossard, E., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 26, pp. 251–244. [Google Scholar]
- Bindraban, P.S.; Dimkpa, C.O.; Pandey, R. Exploring phosphorus fertilizers and fertilzation strategies for improved human and environmental health. Biol. Fertil. Soils 2020, 56, 299–317. [Google Scholar] [CrossRef] [Green Version]
- Cadot, S.; Bélanger, G.; Ziadi, N.; Morel, C.; Sinaj, S. Critical plant and soil phosphorus for wheat, maize, and rapeseed after 44 years of P fertilizer. Nutr. Cycl. Agroecosyst. 2018, 112, 417–433. [Google Scholar] [CrossRef] [Green Version]
- Gao, Y.; Cooper, D.J.; Ma, X. Phosphorus addition have no impact on plant biomass or soil nitrogen in an alpine meadow on the Qinghai-Tibetan Plateau, China. Appl. Soil Ecol. 2016, 106, 18–23. [Google Scholar] [CrossRef]


| Fertilization | Ctot (g kg−1) | Ntot (g kg−1) | Ptot (g kg−1) |
|---|---|---|---|
| P0 | 7.7 ± 0.0 a | 0.9 ± 0.0 a | 0.5 ± 0.0 c |
| TSP | 7.9 ± 0.0 a | 1.0 ± 0.0 a | 0.5 ± 0.0 bc |
| COM | 9.9 ± 0.0 b | 1.1 ± 0.0 a | 0.5 ± 0.0 ab |
| COM + TSP | 10.3 ± 0.0 b | 1.0 ± 0.0 a | 0.6 ± 0.0 a |
| Sampling | Parameter | Treatment | Fertilization | Interaction | |
|---|---|---|---|---|---|
| May 2019 | ACP | p | 0.828 | 0.507 | 0.931 |
| F | 0.048 | 0.854 | 0.209 | ||
| ALP | p | 0.965 | 0.025 | 0.971 | |
| F | 0.002 | 3.445 | 0.127 | ||
| PDE | p | 0.229 | 0.062 | 0.899 | |
| F | 1.534 | 2.630 | 0.263 | ||
| GLA | p | 0.002 | 0.097 | 0.906 | |
| F | 12.660 | 2.243 | 0.251 | ||
| P-DL | p | 0.980 | <0.001 | 0.998 | |
| F | <0.001 | 9.540 | 0.032 | ||
| AMF | p | <0.001 | 0.679 | 0.287 | |
| F | 28.930 | 0.583 | 1.339 | ||
| P-plant | p | 0.549 | 0.002 | 0.973 | |
| F | 0.370 | 5.894 | 0.123 | ||
| C/N plant | p | 0,268 | 0.416 | 0.497 | |
| F | 1.293 | 1.026 | 0.871 | ||
| July 2019 | ACP | p | 0.894 | 0.546 | 0.631 |
| F | 0.018 | 0.786 | 0.653 | ||
| ALP | p | 0.053 | 0.160 | 0.434 | |
| F | 4.172 | 1.823 | 0.989 | ||
| PDE | p | 0.240 | 0.630 | 0.831 | |
| F | 1.460 | 0.654 | 0.364 | ||
| GLA | p | 0.157 | 0.527 | 0.622 | |
| F | 2.150 | 0.818 | 0.667 | ||
| P-DL | p | 0.815 | 0.002 | 0.857 | |
| F | 0.036 | 6.161 | 0.327 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vitow, N.; Zicker, T.; Chiba, A.; Zacher, A.; Eichler-Löbermann, B.; Schulz, S.; Schloter, M.; Baum, C.; Leinweber, P. Impact of the Legume Catch Crop Serradella on Subsequent Growth and P Mobilization under Barley in Different Fertilization Treatments. Agronomy 2021, 11, 2437. https://doi.org/10.3390/agronomy11122437
Vitow N, Zicker T, Chiba A, Zacher A, Eichler-Löbermann B, Schulz S, Schloter M, Baum C, Leinweber P. Impact of the Legume Catch Crop Serradella on Subsequent Growth and P Mobilization under Barley in Different Fertilization Treatments. Agronomy. 2021; 11(12):2437. https://doi.org/10.3390/agronomy11122437
Chicago/Turabian StyleVitow, Nora, Theresa Zicker, Akane Chiba, Anika Zacher, Bettina Eichler-Löbermann, Stefanie Schulz, Michael Schloter, Christel Baum, and Peter Leinweber. 2021. "Impact of the Legume Catch Crop Serradella on Subsequent Growth and P Mobilization under Barley in Different Fertilization Treatments" Agronomy 11, no. 12: 2437. https://doi.org/10.3390/agronomy11122437
APA StyleVitow, N., Zicker, T., Chiba, A., Zacher, A., Eichler-Löbermann, B., Schulz, S., Schloter, M., Baum, C., & Leinweber, P. (2021). Impact of the Legume Catch Crop Serradella on Subsequent Growth and P Mobilization under Barley in Different Fertilization Treatments. Agronomy, 11(12), 2437. https://doi.org/10.3390/agronomy11122437








