Larvicidal Activity of Geranylacetone Derivatives against Culex quinquefasciatus Larvae and Investigation of Environmental Toxicity and Non-Target Aquatic Species
Abstract
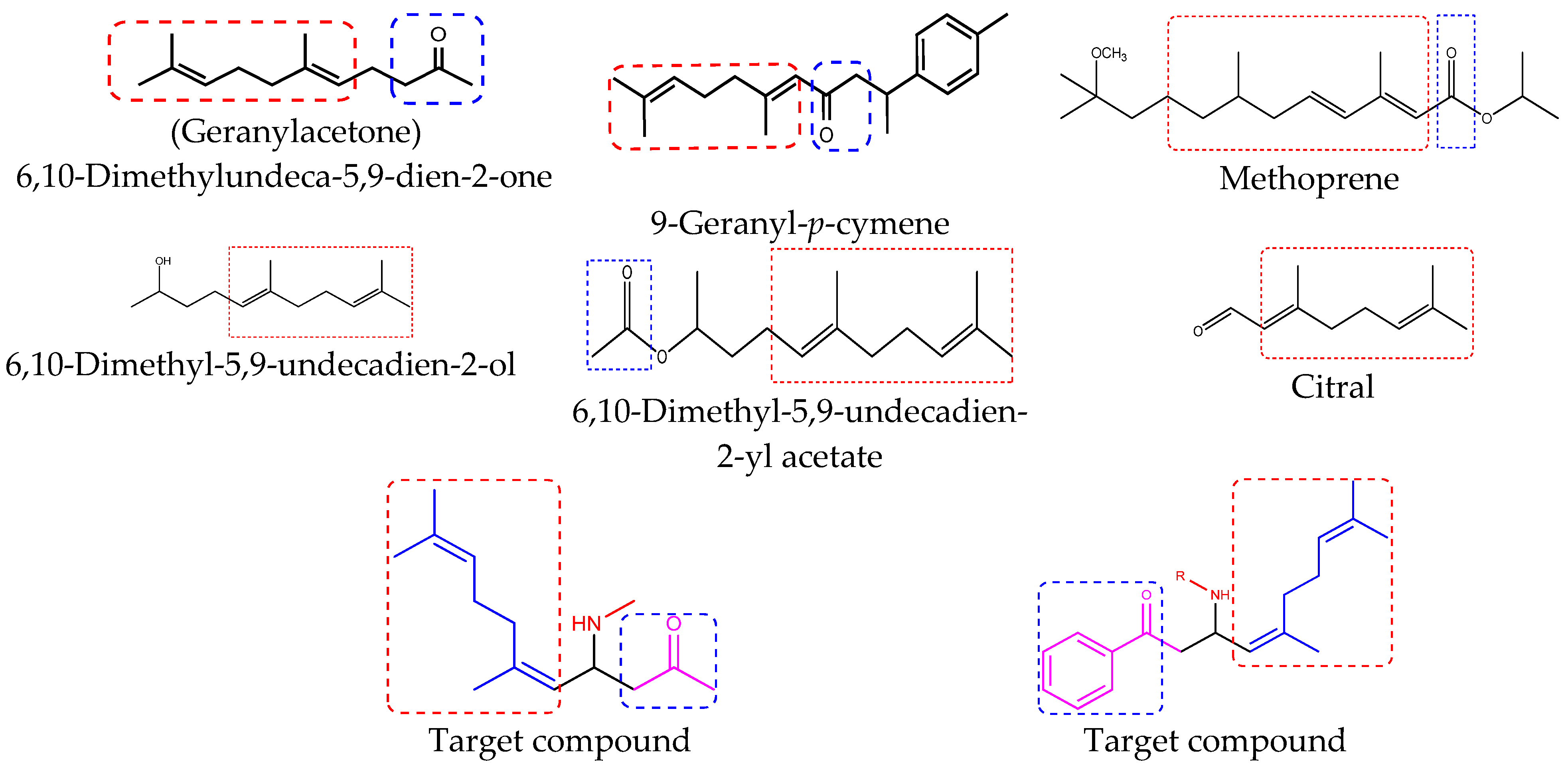
:1. Introduction
2. Materials and Method
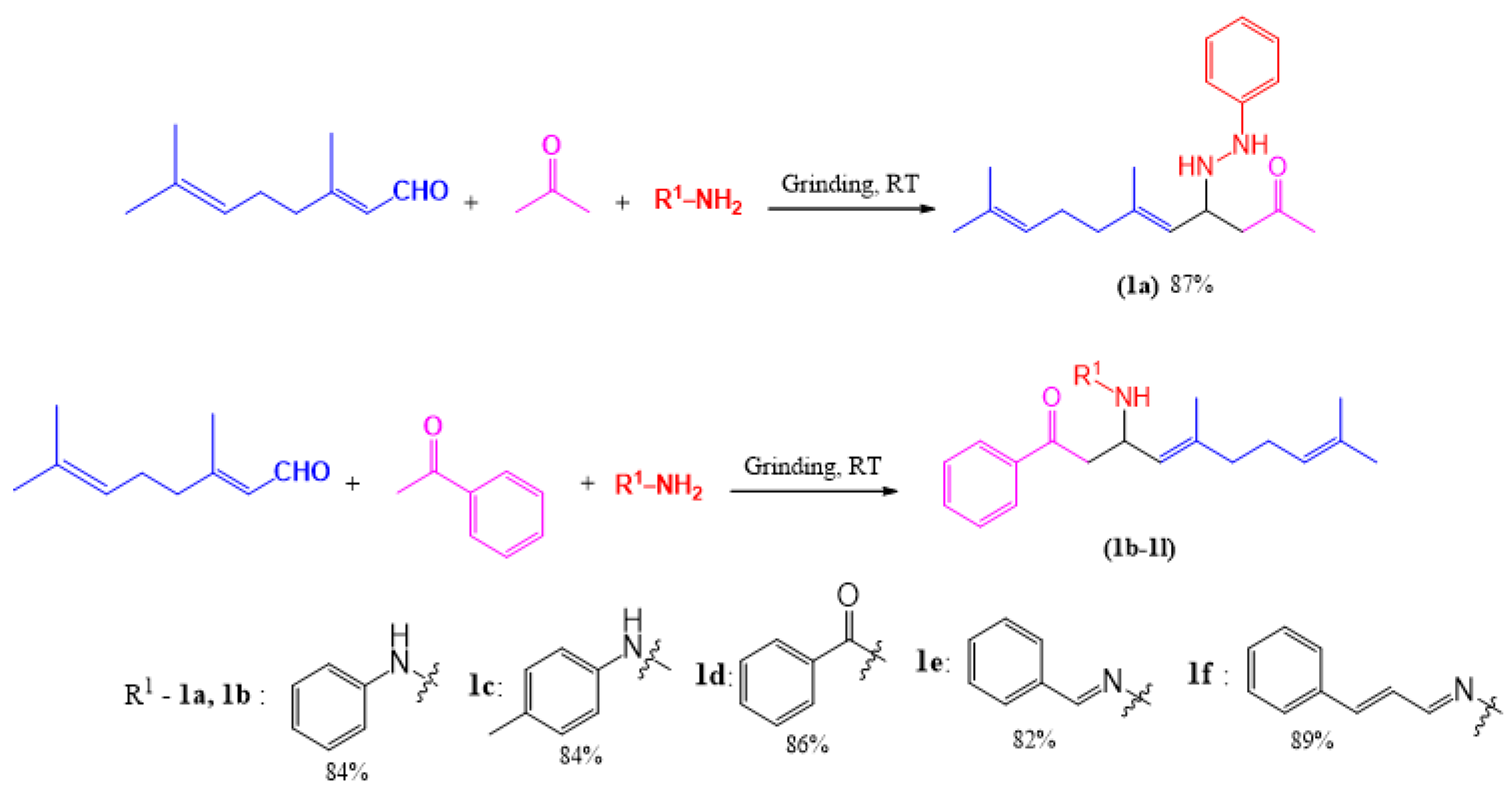
2.1. Synthesis of Compound 1a–f: General Procedure
2.1.1. 6,10-Dimethyl-4-(2-phenylhydrazinyl)undeca-5,9-dien-2-one (1a)
2.1.2. 5,9-Dimethyl-1-phenyl-3-(phenylamino)deca-4,8-dien-1-one (1b)
2.1.3. 5,9-Dimethyl-1-phenyl-3-(p-tolylamino)deca-4,8-dien-1-one (1c)
2.1.4. N-(5,9-Dimethyl-1-oxo-1-phenyldeca-4,8-dien-3-yl)benzamide (1d)
2.1.5. 3-(2-Benzylidenehydrazinyl)-5,9-dimethyl-1-phenyldeca-4,8-dien-1-one (1e)
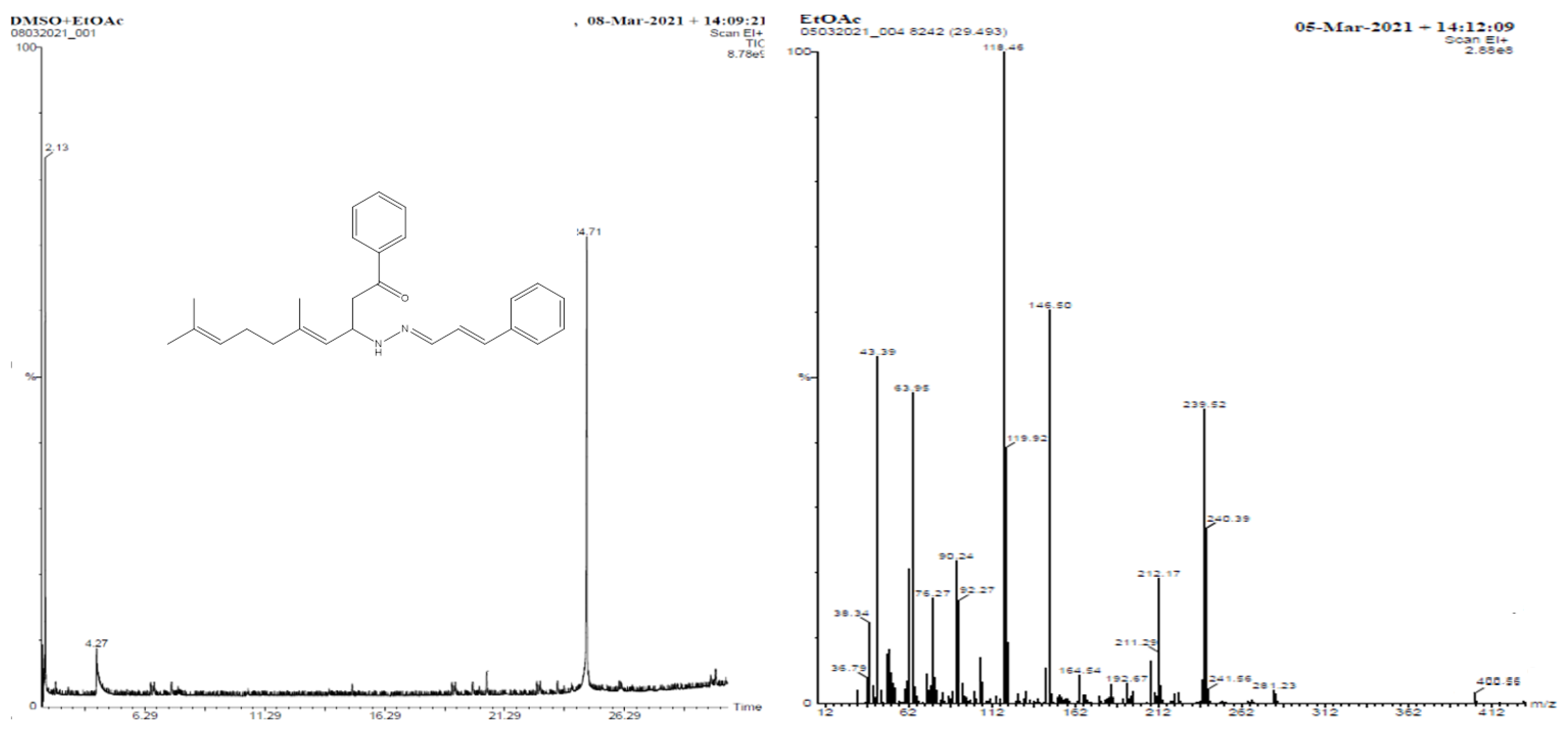
2.1.6. 5,9-Dimethyl-1-phenyl-3-(2-(3-phenylallylidene)hydrazinyl)deca-4,8-dien-1-one (1f)
2.2. Biological Activities
2.2.1. Larvicidal Activity
2.2.2. Antifeedant Activity
2.2.3. Statistical Analysis
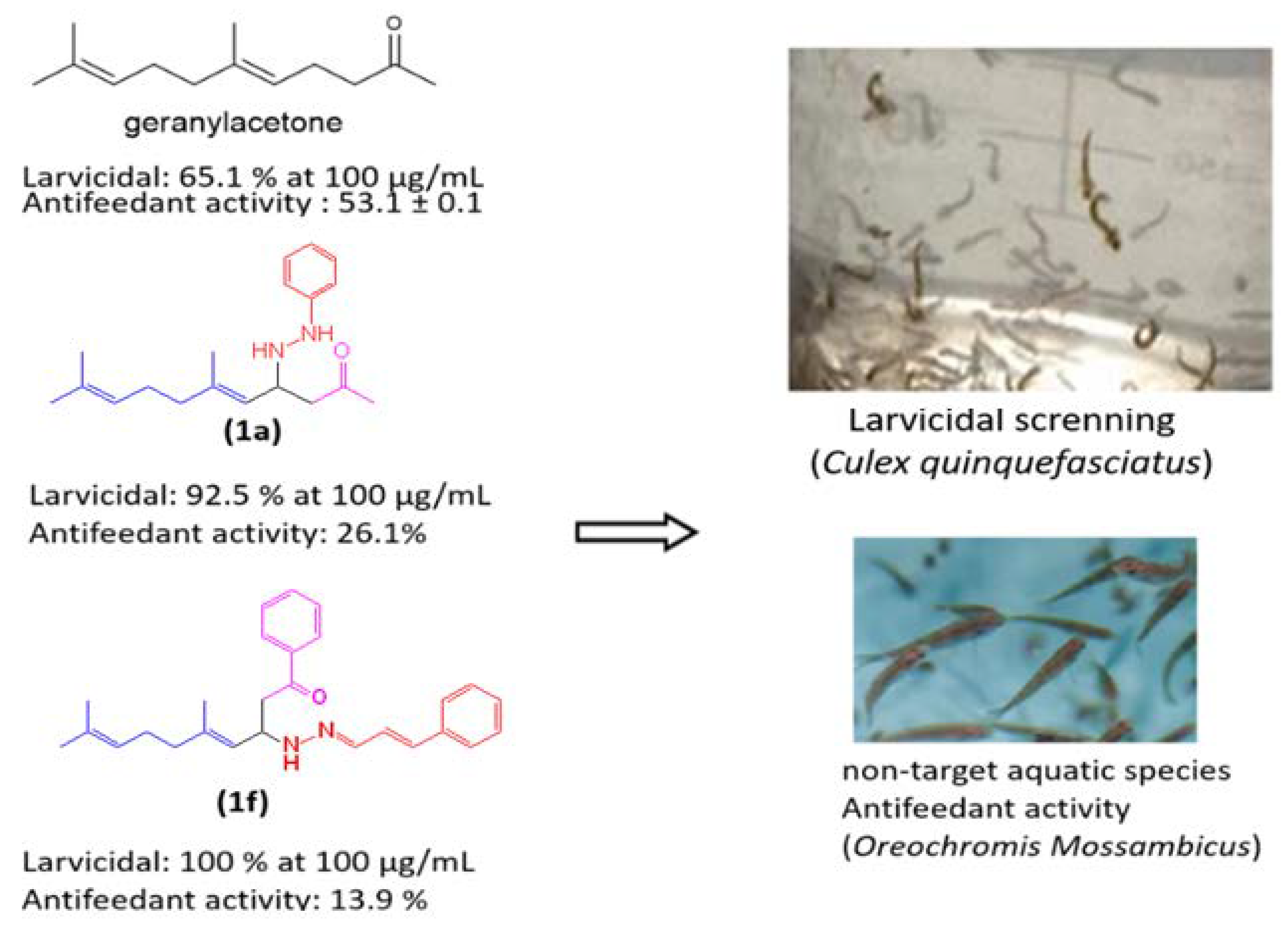
3. Results and Discussion
Biological Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Georges, K.; Jayaprakasam, B.; Dalavoy, S.S.; Nair, M.G. Pestmanaging activities of plant extracts and anthraquinones from Cassia nigricans from Burkina Faso. Bioresour. Technol. 2008, 99, 2037–2045. [Google Scholar] [CrossRef]
- Govindarajan, M. Chemical composition and larvicidal activity of leaf essential oil from Clausenaanisata (Willd.) Hook. f. ex Benth (Rutaceae) against three mosquito species. Asian Pac. J. Trop. Med. 2010, 3, 874–887. [Google Scholar] [CrossRef] [Green Version]
- Sharon, P.L. Environmental safety review of methoprene and bacterially-derived pesticides commonly used for sustained mosquito control. Ecotoxicol. Environ. Saf. 2017, 139, 335–343. [Google Scholar] [CrossRef]
- Ostrand, K.G.; Thies, M.L.; Hall, D.D.; Carpenter, M. Gar ichthyootoxin: Its effect on natural predators and the toxin’s evolutionary function. Southwest. Nat. 1996, 41, 375–377. [Google Scholar]
- Blossom, H.E.; Rasmussen, S.A.; Andersen, N.G.; Larsen, T.O.; Nielsen, K.F.; Hansen, P.J. Prymnesium parvum revisited: Relationship between allelopathy, ichthyotoxicity, and chemical profiles in 5 strains. Aquat. Toxicol. 2014, 157, 159–166. [Google Scholar] [CrossRef]
- Jawad, L.A. Dangerous Fishes of the Eastern and Southern Arabian Peninsula; Springer: Cham, Germany, 2018. [Google Scholar]
- Bohlmann, F.; Zdero, C.; Hoffmann, E.; Mahanta, P.K.; Dorner, W. Neuediterpene und sesquiter peneaussudafrikanischen Helichrysum-Arten. Phytochemistry 1978, 17, 1917–1922. [Google Scholar] [CrossRef]
- Kirimer, N.; Baser, K.H.C.; Demirci, B.; Duman, H. Essential oils of Sideritis species of Turkey belonging to the section Empedoclia. Chem. Nat. Compd. 2004, 40, 19–23. [Google Scholar] [CrossRef]
- Barrero, A.F.; Sanchez, J.F.; Altarejos, J.; Zafra, M.J. Homoditerpenes from the essential oil of Tanacetumannuum. Phytochemistry 1992, 31, 1727–1730. [Google Scholar] [CrossRef]
- Lopez Arze, J.B.; Collin, G.; Garneau, F.X.; Jean, F.I.; Gagnon, H. Essential oils from Bolivia. III. Asteraceae: Artemisia copa Philippi. J. Essent. Oil Res. 2004, 16, 554–557. [Google Scholar] [CrossRef]
- Vivekanandhan, P.; Usha-Raja-Nanthini, A.; Vallic, G.; Shivakumar, M.S. Comparative efficacy of Eucalyptus globulus (Labill) hydrodistilled essential oil andtemephos as mosquito larvicide. Nat. Prod. Res. 2019, 34, 1–4. [Google Scholar]
- Pino, J.; Mesa, J.; Muñoz, Y.; Martí, M.; Marbot, R. Volatile Components from Mango (Mangifera indica L.) Cultivars. J. Agric. Food Chem. 2005, 53, 2213–2223. [Google Scholar] [CrossRef] [PubMed]
- Fruekilde, P.; Hjorth, J.; Jensen, N.R.; Kotzias, D.; Larsen, B. OzonolysisatVegetationSurfaces. Atmos. Environ. 1998, 32, 1893–1902. [Google Scholar] [CrossRef]
- Bero, J.; Beaufay, C.; Hannaert, V.; Hérent, M.F.; Michels, P.; Quetin-Leclercq, J. Antitrypanosomal compounds from the essential oil and extracts of Keetia leucantha leaves with inhibitor activity on Trypanosoma brucei glyceraldehyde-3-phosphate dehydrogenase. Phytomedicine 2013, 203, 270–274. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.F.; He, J.; Li, Y.; Lu, Z.; Li, Z.; Liao, T. Diffusion coefficients and diffusion mechanisms of geranyl acetone in four adsorbents. Ion. Exch. Adsorpt. 2018, 4, 374–384. [Google Scholar]
- Bowman, L.R.; Donegan, S.; McCall, P.J. Is dengue vector control deficient in effectiveness or evidence? Systematic review and metaanalysis. PLoS Negl. Trop Dis. 2016, 10, e0004551-24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nkya, T.E.; Akhouayri, I.; Kisinza, W.; David, J.P. Impact of environment on mosquito response to pyrethroid insecticides: Facts, evidences and prospects. Insect Biochem. Mol. Biol. 2013, 43, 407–416. [Google Scholar] [CrossRef] [Green Version]
- Jayaraj, R.; Megha, P.; Sreedev, P. Review article. Organochlorine pesticides, their toxic effects on living organisms and their fate in the environment. Interdiscip. Toxicol. 2016, 9, 90–100. [Google Scholar] [CrossRef] [Green Version]
- Park, I.K.; Shin, S.C.; Kim, C.S.; Lee, H.J.; Choi, W.S.; Ahn, Y.J. Larvicidal activity of lignans identified in Phrymaleptostachya Var. asiatica roots against tree mosquito species. J. Agric. Food. Chem. 2005, 53, 969–972. [Google Scholar] [CrossRef]
- Yang, J.Y.; Cho, K.S.; Chung, N.H.; Kim, C.H.; Suh, J.W.; Lee, H.S. Constituents of volatile compounds derived from Melaleucaalternifolia leaf oil and acaricidal toxicities against house dust mites. J. Korean Soc. Appl. Biol. Chem. 2013, 56, 91–94. [Google Scholar] [CrossRef]
- Abdel-Fattah Mostafa, A.; Sathish Kumar, C.; Al-Askar, A.A.; Sayed, S.R.M.; Surendra Kumar, R.; Idhayadhulla, A. Synthesis of novel benzopyran-connected pyrimidine and pyrazole derivatives via a green method using Cu(II)-tyrosinase enzyme catalyst as potential larvicidal, antifeedant activities. RSC Adv. 2019, 9, 25533–25543. [Google Scholar] [CrossRef] [Green Version]
- Sujith, S.; Seghal Kiran, G.; Joseph Selvin, J.; Shakir, S.; Gandhimathi, R.; Panikkar, M.V.N.; Aseer, M. Biopotentials of seaweeds collected from southwest coast of india. J. Mar. Sci. Technol. 2009, 17, 67–73. [Google Scholar]
- Aboaba, S.A.; Ogunwande, I.A.; Walker, T.M.; Setzer, W.N.; Oladosu, I.A.; Ekundayo, O. Essential oil composition, antibacterial activity and toxicity of the leaves of Tetrapleura tetraptera (Schum. & Thonn.) taubert from Nigeria. Nat. Prod. Commun. 2009, 4, 287–290. [Google Scholar]
- Marumoto, S.; Okuno, Y.; Miyazawa, M. Inhibition of β-Secretase activity by monoterpenes, sesquiterpenes, and C13 norisoprenoids. J. Oleo Sci. 2017, 66, 851–855. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Compounds | % of Mortality | LD50 (µg/mL) a | ||
|---|---|---|---|---|
| 25 µg/mL | 50 µg/mL | 100 µg/mL | ||
| Geranylacetone | 29.8 ± 0.6 | 42.6 ± 0.2 | 65.1 ± 0.4 | 67.2 |
| 1a | 32.7 ± 0.6 | 62.3 ± 0.4 | 92.5 ± 0.8 | 42.0 |
| 1b | 37.6 ± 0.8 | 67.3 ± 0.13 | 97.3 ± 0.1 | 35.6 |
| 1c | 46.2 ± 0.1 | 82.9 ± 0.2 | 100 ± 0.0 | 18.6 |
| 1d | 40.1 ± 0.3 | 81.3 ± 0.1 | 99.1 ± 0.2 | 25.9 |
| 1e | 38.8 ± 0.9 | 68.6 ± 0.12 | 98.7 ± 0.3 | 34.2 |
| 1f | 48.0 ± 0.3 | 86.1 ± 0.3 | 100 ± 0.0 | 14.1 |
| Negative control | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
| Compounds | % of Mortality at 25 µg/mL | % of Mortality at 50 µg/mL | % of Mortality at 100 µg/mL | LD50 (µg/mL) a |
|---|---|---|---|---|
| Geranylacetone | 20.0 ± 0.0 | 32.5 ± 0.2 | 53.1 ± 0.1 | 92.2 |
| 1a | 9.2 ± 0.5 | 15.2 ± 0.1 | 26.1 ± 0.1 | >100 |
| 1b | 8.2 ± 0.2 | 15.1 ± 0.6 | 33.4± 0.32 | >100 |
| 1c | 5.5 ± 0.3 | 11.1 ± 0.7 | 23.1 ± 0.4 | >100 |
| 1d | 7.2 ± 0.7 | 14.2 ± 0.9 | 26.2 ± 0.1 | >100 |
| 1e | 8.2 ± 0.2 | 15.1 ± 0.6 | 33.4± 0.32 | >100 |
| 1f | 3.0 ± 0.1 | 6.21 ± 0.1 | 13.9 ± 0.2 | >100 |
| Negative control | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 | 0.0 ± 0.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Zharani, M.; Al-Eissa, M.S.; Rudayni, H.A.; Ali, D.; Alarifi, S.; Surendrakumar, R.; Idhayadhulla, A. Larvicidal Activity of Geranylacetone Derivatives against Culex quinquefasciatus Larvae and Investigation of Environmental Toxicity and Non-Target Aquatic Species. Agronomy 2021, 11, 2342. https://doi.org/10.3390/agronomy11112342
Al-Zharani M, Al-Eissa MS, Rudayni HA, Ali D, Alarifi S, Surendrakumar R, Idhayadhulla A. Larvicidal Activity of Geranylacetone Derivatives against Culex quinquefasciatus Larvae and Investigation of Environmental Toxicity and Non-Target Aquatic Species. Agronomy. 2021; 11(11):2342. https://doi.org/10.3390/agronomy11112342
Chicago/Turabian StyleAl-Zharani, Mohammed, Mohammed S. Al-Eissa, Hassan A. Rudayni, Daoud Ali, Saud Alarifi, Radhakrishnan Surendrakumar, and Akbar Idhayadhulla. 2021. "Larvicidal Activity of Geranylacetone Derivatives against Culex quinquefasciatus Larvae and Investigation of Environmental Toxicity and Non-Target Aquatic Species" Agronomy 11, no. 11: 2342. https://doi.org/10.3390/agronomy11112342
APA StyleAl-Zharani, M., Al-Eissa, M. S., Rudayni, H. A., Ali, D., Alarifi, S., Surendrakumar, R., & Idhayadhulla, A. (2021). Larvicidal Activity of Geranylacetone Derivatives against Culex quinquefasciatus Larvae and Investigation of Environmental Toxicity and Non-Target Aquatic Species. Agronomy, 11(11), 2342. https://doi.org/10.3390/agronomy11112342







