Predisposing Factors for “Olive Quick Decline Syndrome” in Salento (Apulia, Italy)
Abstract
1. Introduction
2. Resilience of Olive Agro-Ecosystem of Salento
3. Current Characteristics of the Olive Groves of Salento
4. Occurrence of Olive Quick Decline Syndrome in the Salento Olive Agro-Ecosystem
5. Predisposing Factors for Olive Quick Decline Syndrome Acting on the Salento Olive Agro-Ecosystem
5.1. Cultivar Susceptibility
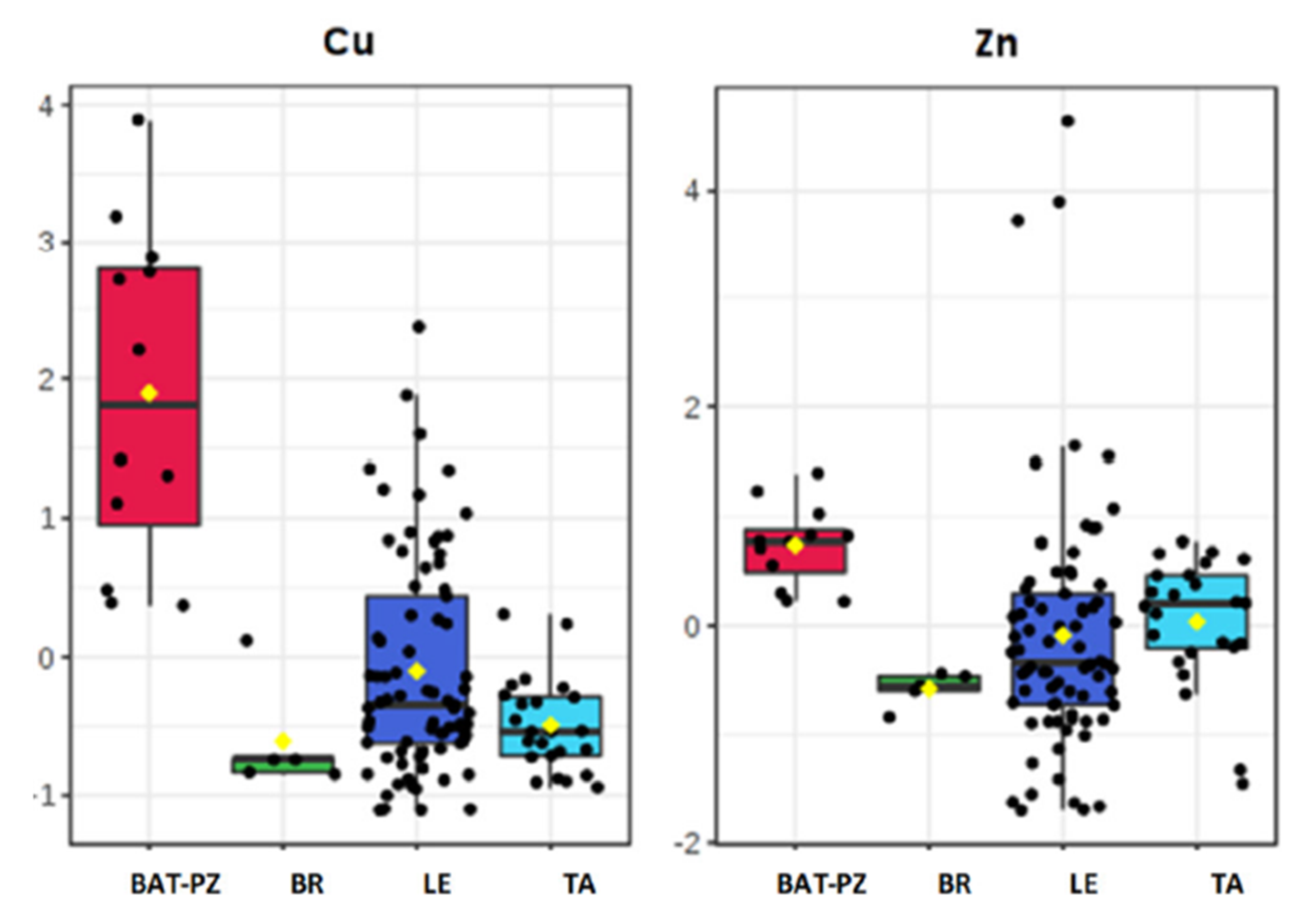
5.2. Soil Ionome Characteristics
5.3. Agronomical Practises
5.4. Tree Pruning
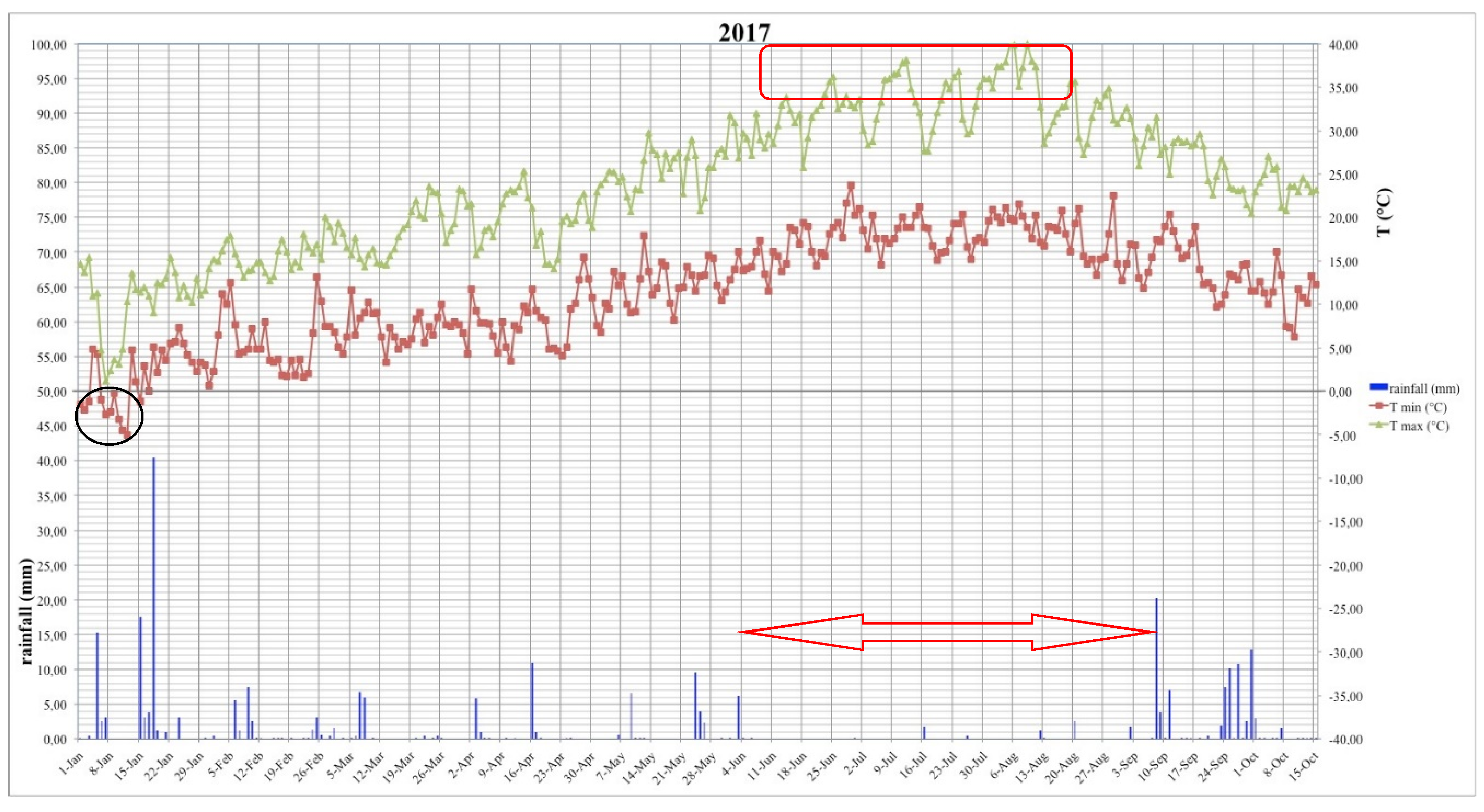
5.5. Climatic Changes
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Keane, P.J.; Kerr, A. Factors affecting disease development. In Plant Pathogens and Plant Disease; Brown, J.F., Ogle, H.G., Eds.; Rockvale Publications: Armidale, Australia, 1997; pp. 287–298. [Google Scholar]
- Scortichini, M. Epidemiology and predisposing factors of some major bacterial diseases of stone and fruit tree species. J. Plant Pathol. 2010, 92, S1.73–S1.78. [Google Scholar] [CrossRef]
- Saponari, M.; Boscia, D.; Nigro, F.; Martelli, G.P. Identification of DNA sequence related to Xylella fastidiosa in oleander, almond, and olive trees exhibiting leaf scorch symptoms in Apulia (Southern Italy). J. Plant Pathol. 2013, 93, 668. [Google Scholar] [CrossRef]
- Cariddi, C.; Saponari, M.; Boscia, D.; De Stradis, A.; Loconsole, G.; Nigro, F.; Porcelli, F.; Potere, O.; Martelli, G.P. Isolation of Xylella fastidiosa strain infecting olive and oleander in Apulia, Italy. J. Plant Pathol. 2014, 96, 425–429. [Google Scholar] [CrossRef]
- Martelli, G.P.; Boscia, D.; Porcelli, F.; Saponari, M. The olive quick decline syndrome in south-east Italy: A threatening phytosanitary emergency. Eur. J. Plant Pathol. 2016, 144, 235–243. [Google Scholar] [CrossRef]
- EFSA Panel on Plant Health (PLH). Update of the scientific opinion on the risk to plant health posed by Xylella fastidiosa in the EU territory. EFSA J. 2019, 17, 5665. [Google Scholar] [CrossRef]
- Fierro, A.; Liccardo, A.; Porcelli, F. A lattice model to manage the vector and the infection of the Xylella fastidiosa on olive trees. Sci. Rep. 2019, 9, 8273. [Google Scholar] [CrossRef]
- White, S.M.; Navas-Cortés, J.A.; Bullock, J.M.; Boscia, D.; Chapman, D.S. Estimating the epidemiology of emerging Xylella fastidiosa outbreaks in olives. Plant Pathol. 2020. [Google Scholar] [CrossRef]
- Nigro, F.; Boscia, D.; Antelmi, I.; Ippolito, I. Fungal species associated with a severe decline of olive in Southern Italy. J. Plant Pathol. 2013, 93, 668. [Google Scholar] [CrossRef]
- Primavera, M.; D’Oronzo, C.; Muntoni, I.M.; Radina, F.; Fiorentino, G. Environment, crops and harvesting strategies during the II millenium B.C.: Resilience and adaptation in socio-economic systems of Bronze Age in Apulia (SE Italy). Q. Int. 2017, 436, 83–95. [Google Scholar] [CrossRef]
- Caliandro, A.; Stelluti, M. Ruolo dell’Olivicoltura nella Lotta alla Desertificazione; Monografia del Ministero dell’Ambiente e della Tutela del Territorio e del Mare; Ministero dell’Ambiente e della Tutela del Territorio e del Mare: Rome, Italy, 2005; 36p. [Google Scholar]
- Biasco, A. L’Olivicoltura Salentina Attraverso i Secoli; Tipografia degli agricoltori: Rome, Italy, 1937; 16p. [Google Scholar]
- Frisullo, S.; Camele, I.; Agosteo, G.E.; Boscia, D.; Martelli, G.P. Brief historical account of olive leaf scorch (“brusca”) in the Salento peninsula of Italy and state-of-the-art of the olive quick decline syndrome. J. Plant Pathol. 2014, 96, 441–449. [Google Scholar] [CrossRef]
- Petri, L. Ricerche sulla brusca dell’olivo. In Studi sulle Malattie dell’Olivo; Petri, L., Ed.; Memorie della Regia Stazione di Patologia Vegetale; Tipografia Nazionale G. Bertero: Rome, Italy, 1911; pp. 13–147. [Google Scholar]
- Petri, L. Le Malattie dell’Olivo. Manuale per l’Identificazione delle Malattie non Parassitarie e di Quelle Prodotte da Parassiti Vegetali con Speciale Riguardo ai Mezzi per Combatterle; Istituto Micrografico Italiano: Florence, Italy, 1915; 175p. [Google Scholar]
- Del Coco, L.; De Pascali, S.A.; Fanizzi, F.P. NMR-metabolomic study on monovarietal and blend Salento EVOOs including some from secular olive trees. Food Nutr. Sci. 2014, 5, 89–95. [Google Scholar]
- Cimato, A.; De Rinaldis, G.; Calogiuri, A.L.; Sani, G. Il Germoplasma Olivicolo in Provincia di Lecce; Camera di Commercio Industria, Artigianato e Agricoltura: Lecce, Italy, 2001; 89p. [Google Scholar]
- Luisi, N.; De Cicco, V. Danni da bromacile su olivi consociati ad agrumi in Puglia. Inf. Fitopatol. 1975, 25, 17–19. [Google Scholar]
- Santilli, E.; Lombardo, L.; Varlaro, M.E.; Nannelli, R.; Gagnarli, E.; Briccoli Bati, C. Effectiveness of the GAEC cross compliance standard maintenance of olive groves in good vegetative condition in avoiding the deteriorations of habitats and land abandonment. Ital. J. Agron. 2011, 6, e15. [Google Scholar] [CrossRef]
- Ciervo, M. The olive quick decline syndrome (OQDS) diffusion in Apulia region: An apparent contradiction according to the agricultural model. Belgeo 2016, 4. [Google Scholar] [CrossRef]
- Martelli, G.P. The current status of the quick decline syndrome of olive in southern Italy. Phytoparasitica 2016, 44, 1–10. [Google Scholar] [CrossRef]
- Saponari, M.; Boscia, D.; Altamura, G.; Loconsole, G.; Zicca, S.; D’Attoma, G.; Morelli, M.; Palmisano, F.; Saponari, A.; Tavano, D.; et al. Isolation and pathogenicity of Xylella fastidiosa associated to the olive quick decline syndrome in southern Italy. Sci. Rep. 2017, 7, 17723. [Google Scholar] [CrossRef] [PubMed]
- Sosnowsky, M.R.; Fletcher, J.D.; Daly, A.M.; Rodoni, B.C.; Viljanen-Rollinson, S.L.H. Techniques for the treatment, removal and disposal of host material during programmes for plant pathogen eradication. Plant Pathol. 2009, 58, 621–635. [Google Scholar] [CrossRef]
- Saponari, M.; Boscia, D.; Loconsole, G.; Palmisano, F.; Savino, V.N.; Potere, O.; Martelli, G.P. New hosts of Xylella fastidiosa strain CoDiRO in Apulia. J. Plant Pathol. 2014, 96, 611. [Google Scholar] [CrossRef]
- Jeger, M.; Caffier, D.; Candresse, T.; Chatzivassiliou, E.; Dehnen-Schmutz, K.; Gilioli, G.; Grégoire, J.C.; Miret, J.A.J.; MacLeod, A.; Navarro, M.N.; et al. Updated pest categorization of Xylella fastidiosa. EFSA J. 2018, 16, 5357. [Google Scholar] [CrossRef]
- Nigro, F.; Antelmi, I.; Ippolito, A. Identification and characterization of fungal species associated with the quick decline of olive. J. Plant Pathol. 2014, 96, S4. [Google Scholar]
- Saponari, M.; Loconsole, G.; Cornara, D.; Yokomi, R.K.; De Stradis, A.; Boscia, D.; Bosco, D.; Martelli, G.P.; Krugner, R.; Porcelli, F. Infectivity and transmission of Xylella fastidiosa by Philaenus spumarius (Hemiptera Aphrophoridae) in Apulia, Italy. J. Econ. Entomol. 2014, 107, 1316–1319. [Google Scholar] [CrossRef] [PubMed]
- Strona, G.; Carstens, C.J.; Beck, P.S.A. Network analysis reveals why Xylella fastidiosa will persist in Europe. Sci. Rep. 2017, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- Marcelletti, S.; Scortichini, M. Xylella fastidiosa CoDiRO strain associated with olive quick decline syndrome in southern Italy belongs to a clonal complex of the subspecies pauca that evolved in Central America. Microbiology 2016, 162, 2087–2098. [Google Scholar] [CrossRef] [PubMed]
- Giampetruzzi, A.; Saponari, M.; Loconsole, G.; Boscia, D.; Savino, V.N.; Almeida, R.P.P.; Zicca, S.; Landa, B.B.; Chacon-Diaz, C.; Saldarelli, P. Genome-wide analysis provides evidence on the genetic relatedness of the emergent Xylella fastidiosa genotype in Italy to isolates from Central America. Phytopathology 2017, 107, 816–827. [Google Scholar] [CrossRef] [PubMed]
- Loconsole, G.; Saponari, M.; Boscia, D.; D’Attoma, G.; Morelli, M.; Martelli, G.P.; Almeida, R.P.P. Intercepted isolates of Xylella fastidiosa in Europe reveal novel genetic diversity. Eur. J. Plant Pathol. 2016, 146, 85–94. [Google Scholar] [CrossRef]
- Bergsma-Vlami, M.; Van de Bilt, J.L.J.; Tjou Tam Sin, N.N.A.; Helderman, C.M.; Gorkink Smits, P.P.M.A.; Landman, N.M.; Van Nieuwburg, J.G.W.; Van Veen, E.J.; Westenberg, M. Assessment of the genetic diversity of Xylella fastidiosa in imported ornamental Coffea arabica plants. Plant Pathol. 2017, 66, 1065–1074. [Google Scholar] [CrossRef]
- Jacques, M.-A.; Denancé, N.; Legendre, B.; Morel, E.; Briand, M.; Mississipi, S.; Durand, K.; Olivier, V.; Portier, P.; Poliakoff, F.; et al. New coffee plant-infecting Xylella fastidiosa variants derived via homologous recombination. Appl. Environ. Microbiol. 2015, 82, 1556–1568. [Google Scholar] [CrossRef]
- Bodino, N.; Cavalieri, V.; Dongiovanni, C.; Saladini, M.A.; Simonetto, A.; Volani, S.; Plazio, E.; Altamura, G.; Tauro, D.; Gilioli, G.; et al. Spittlebugs of Mediterranean olive groves: Host-plant exploitation throughout the year. Insects 2020, 11, 130. [Google Scholar] [CrossRef]
- Alves, E.; Marucci, C.R.; Lopes, J.R.S.; Leite, B. Leaf symptoms on plum, coffee and citrus and the relationship with the extent of xylem vessels colonized by Xylella fastidiosa. J. Phytopathol. 2004, 152, 291–297. [Google Scholar] [CrossRef]
- Pérez-Donoso, A.G.; Lenhof, J.J.; Pinney, K.; Labavitch, J.M. Vessel embolism and tyloses in early stages of Pierce’s disease. Aust. J. Grape Wine Res. 2015, 22, 81–86. [Google Scholar] [CrossRef]
- Fritsch, F.B.; Lin, H.; Walker, M.A. Xylella fastidiosa population dynamics in grapevine genotypes differing in susceptibility to Pierce’s disease. Am. J. Enol. Vitic. 2007, 58, 326–332. [Google Scholar] [CrossRef]
- De Benedictis, M.; De Caroli, M.; Baccelli, I.; Marchi, G.; Bleve, G.; Gallo, A.; Ranaldi, F.; Falco, V.; Pasquali, V.; Piro, G.; et al. Vessel occlusion in three cultivars of Olea europea naturally exposed to Xylella fastidiosa in open field. J. Phytopathol. 2017, 165, 589–594. [Google Scholar] [CrossRef]
- Cardinale, M.; Luvisi, A.; Meyer, J.B.; Sabella, E.; De Bellis, L.; Cruz, A.C.; Ampatzidis, Y.; Cherubini, P. Specific Fluorescence in Situ Hybridization (FISH) Test to Highlight Colonization of Xylem Vessels by Xylella fastidiosa in Naturally Infected Olive Trees (Olea europaea L.). Front. Plant Sci. 2018, 9, 431. [Google Scholar] [CrossRef]
- Moschettini, C. Della Rogna degli Ulivi; Stamperia Aniello De Dominicis: Naples, Italy, 1790. [Google Scholar]
- Buonaurio, R.; Moretti, C.; Passos da Silva, D.; Cortese, C.; Ramos, C.; Venturi, V. The olive knot disease as a model to study the role of interspecies bacterial communities in plant disease. Front. Plant Sci. 2015, 6, 434. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Minaya, J.; Conesa, D.; López-Quílez, A.; Saponari, M.; Vicent, A. Insights into the spatio-temporal spread of Xylella fastidiosa in south-eastern Italy. In Proceedings of the European Conference on Xylella fastidiosa: Finding Answers to a Global Problem, Palma de Mallorca, Spain, 13–15 November 2017. [Google Scholar] [CrossRef]
- Bini, C. Geology and Geomorphology. In The soils of Italy; Costantini, E.A.C., Dazzi, C., Eds.; Springer: Dordrecht, The Netherlands, 2013; pp. 39–56. [Google Scholar] [CrossRef]
- Ghorbani, R.; Wilcockson, S.; Koochekl, A.; Leifert, C. Soil management for sustainable crop disease control: A review. In Organic Farming, Pest Control and Remediation of Soil Pollutants; Lichtfouse, E., Ed.; Springer: San Francisco, CA, USA, 2009; pp. 177–227. [Google Scholar]
- Del Coco, L.; Migoni, D.; Girelli, C.R.; Angilé, F.; Scortichini, M.; Fanizzi, F.P. Soil and leaf ionome heterogeneity in Xylella fastidiosa subsp. pauca-infected, non infected and treated olive groves in Apulia, Italy. Plants 2020, 9, 760. [Google Scholar] [CrossRef] [PubMed]
- Eide, D.J. The oxidative stress in zinc deficiency. Metallomics 2011, 3, 1124–1129. [Google Scholar] [CrossRef]
- La Fontaine, S.; Quinn, J.M.; Nakamoto, S.S.; Page, M.D.; Göhre, V.; Moseley, J.L.; Kropat, J.; Merchant, S. Copper-dependent iron assimilation pathway in the model photosynthetic eukaryote Chlamydomonas reinhardtii. Eukaryot. Cell 2002, 1, 736–757. [Google Scholar] [CrossRef]
- Cabot, C.; Martos, S.; Llugany, M.; Gallego, B.; Toirà, R.; Poschenrieder, C. A role for zinc in plant defense against pathogens and herbivores. Front. Plant Sci. 2019, 10, 1171. [Google Scholar] [CrossRef]
- Kim, S.H.; Hong, J.K.; Lee, S.C.; Sohn, K.H.; Jung, H.W.; Hwang, B.K. CAZFP1, Cys2/His2-type zinc finger transcription factor gene functions as a pathogen-induced early-defense gene in Capsicuum annuum. Plant Mol. Biol. 2004, 55, 883–904. [Google Scholar] [CrossRef]
- Gupta, S.K.; Rai, A.K.; Kanwar, S.S.; Sharma, T.R. Comparative analysis of zinc finger proteins involved in plant disease resistance. PLoS ONE 2012, 7, e42578. [Google Scholar] [CrossRef]
- Poschenrieder, C.; Toirà, R.; Barcelo, J. Can metal defend plants against biotic stress? Trends Plant Sci. 2006, 11, 288–295. [Google Scholar] [CrossRef] [PubMed]
- Fones, H.; Preston, G.M. The impact of transition metals on bacterial plant disease. FEMS Microbiol. Rev. 2012, 37, 495–519. [Google Scholar] [CrossRef]
- Rodriguez, F.I.; Esch, J.J.; Hall, A.E.; Binder, B.M.; Schaller, G.E.; Bleecker, A.B. A copper cofactor for the ethylene receptor ETR1 from Arabidopsis. Science 1999, 283, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Chmielowska, J.; Deckert, J.; Diaz, J. Activity of peroxidases and phenylalanine ammonia-lyase in lupine and soybean seedlings treated with copper and an ethylene inhibitor. Biol. Lett. 2008, 45, 59–67. [Google Scholar]
- Liu, M.; Pirrello, J.; Chervin, C.; Roustan, J.-P.; Bouzayen, M. Ethylene Control of Fruit Ripening: Revisiting the Complex Network of Transcriptional Regulation. Plant Physiol. 2015, 169, 2380–2390. [Google Scholar] [CrossRef] [PubMed]
- Farzadfar, S.; Zarinkamar, F.; Hojati, M. Magnesium and manganese affect photosynthesis, essential oil composition and phenolic compounds of Tanacetum parthenium. Plant Physiol. Biochem. 2017, 112, 207–217. [Google Scholar] [CrossRef] [PubMed]
- Cobine, P.A.; Cruz, L.F.; Navarrete, F.; Duncan, D.; Tygart, M.; De La Fuente, L. Xylella fastidiosa differentially accumulates mineral elements in biofilm and planktonic cells. PLoS ONE 2013, 8, e54936. [Google Scholar] [CrossRef]
- Navarrete, F.; De La Fuente, L. Zinc detoxification is required for full virulence and modification of the host leaf ionome by Xylella fastidiosa. Mol. Plant Microbe Interact. 2015, 28, 497–507. [Google Scholar] [CrossRef]
- Scortichini, M.; Migoni, D.; Angilè, F.; Del Coco, L.; Girelli, C.R.; Zampella, L.; Mastrobuoni, F.; Fanizzi, F.P. Xylella fastidiosa subsp. pauca on olive in Salento (southern Italy): Infected trees have low in planta micronutrient content. Phytopath. Medit. 2019, 58, 39–48. [Google Scholar] [CrossRef]
- Collavo, A.; Resistenza al Glifosate nelle Colture Arboree. Veneto Agricoltura, Forum Fitoiatrico 19 Gennaio, 2012, Legnaro (PD), Italy. Available online: https://www.venetoagricoltura.org/upload/pubblicazioni/Forum%20Fito%20023/COLLAVO.pdf (accessed on 25 July 2020).
- Ferreira, I.Q.; Arrobas, M.; Claro, A.M.; Rodrigues, M.A. Soil management in rainfed olive orchards may result in conflicting effects on olive production and soil fertility. Span. J. Agric. Res. 2013, 2, 472–480. [Google Scholar] [CrossRef]
- Kremer, R.J.; Means, N.E.; Kim, S. Glyphosate affects soybean root exudation and rhizosphere microorganisms. Intern. J. Anal. Chem. 2005, 85, 1165–1174. [Google Scholar] [CrossRef]
- Kremer, R.J.; Means, N.E. Glyphosate and glyphosate-resistant crop interactions with rhizosphere microorganisms. Europ. J. Agron. 2009, 31, 153–161. [Google Scholar] [CrossRef]
- Serra, A.P.; Marchetti, M.E.; Da Silva Candido, A.C.; Ribero Dias, A.C.; Christoffoleti, P.J. Glyphosate influence on nitrogen, manganese, iron, copper and zinc nutritional efficiency in glyphosate resistant soybean. Cienc. Rural 2011, 41, 77–84. [Google Scholar] [CrossRef]
- Johal, G.S.; Huber, D.M. Glyphosate effects on diseases of plants. Eur. J. Agron. 2009, 31, 144–152. [Google Scholar] [CrossRef]
- Aristide, L.; Reed, R.L.; Wilkes, R.A.; Youngster, T.; Kukurugya, M.A.; Katz, V.; Sasaki, C.R.S. Glyphosate-induced specific and widespread perturbations in the metabolome of soil Pseudomonas species. Front. Environ. Sci. 2017, 5, 34. [Google Scholar] [CrossRef]
- Martinez, D.A.; loaning, U.E.; Graham, M.C. Impacts of glyphosate-based herbicides on disease resistance and health of crops: A review. Environ. Sci. Eur. 2018, 30, 2. [Google Scholar] [CrossRef]
- Felline, S.; Del Coco, L.; Kaleb, S.; Guarnieri, G.; Fraschetti, S.; Terlizzi, A.; Fanizzi, F.P.; Falace, A. The response of the algae Fucus virsoides (Fucales, Ochrophyta) to Roundup® solution exposure: A metabolomics approach. Environ. Pollut. 2019, 254, 112977. [Google Scholar] [CrossRef]
- Morales, A.; Hurtado, M.I.; Fischer, T. Influence of pruning intensity of olive trees on the oil characteristics (“Coratina”). Acta Hortic. 2014, 1057, 717–723. [Google Scholar] [CrossRef]
- Sibbett, G.S. Pruning mature bearing olive trees. In Olive Production Manual, 2nd ed.; Sibbett, G.S., Ferguson, L., Eds.; University of California, Division of Agriculture and Natural Resource: Oakland, CA, USA, 2005; pp. 55–59. [Google Scholar]
- Rodrigues, M.A.; Lopes, J.I.; Ferreira, I.Q.; Arrobas, M. Olive tree response to the severity of pruning. Turk. J. Agric. For. 2018, 42, 103–113. [Google Scholar] [CrossRef]
- Tombesi, A. Advances in harvesting and pruning of olive trees. J. Food Sci. Nutr. 2013, 42, 97–103. [Google Scholar]
- Polade, S.D.; Gershunov, A.; Cayan, D.R.; Dettinger, M.D.; Pierce, D.W. Precipitation in a warming world: Assessing projected hydro-climate changes in California and other Mediterranean climate regions. Sci. Rep. 2017, 7, 10783. [Google Scholar] [CrossRef]
- Parent, C.; Capelli, N.; Berger, A.; Crèvecoeur, M. An overview of plant responses to waterlogging. Plant Stress 2008, 2, 20–27. [Google Scholar]
- Navarro, C.; Parra, M.A. Plantacíon. In El Cultivo del Olivo; Barranco, D., Fernández-Escobar, R., Rallo, L., Eds.; Junta de Andalucía y Mundi-Prensa: Madrid, Spain, 2008; pp. 163–195. [Google Scholar]
- Pezeshki, S.R.; Pardue, J.H.; De Laune, R.D. Leaf gas exchange and growth of flood-tolerant and flood-sensitive tree species under low soil redox condition. Tree Physiol. 1996, 16, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Sanzani, S.M.; Schena, L.; Nigro, F.; Sergeeva, V.; Ippolito, A.; Salerno, M.G. Abiotic diseases of olive. J. Plant Pathol. 2012, 94, 469–491. [Google Scholar] [CrossRef]
- Scortichini, M.; Chen, J.; De Caroli, M.; Dalessandro, G.; Pucci, N.; Modesti, V.; L’Aurora, A.; Petriccione, M.; Zampella, L.; Mastrobuoni, F.; et al. A zinc-copper-citric acid biocomplex shows promise for control of Xylella fastidiosa subsp. pauca in olive trees in Apulia region (southern Italy). Phytopath. Medit. 2018, 57, 48–72. [Google Scholar] [CrossRef]
- Ruiz, N.; Barranco, D.; Rapoport, H.F. Anatomical response of olive (Olea europea L.) to freezing temperatures. J. Hortic. Sci. Biotech. 2006, 81, 783–790. [Google Scholar] [CrossRef]
- Brito, C.; Dinis, L.T.; Moutinho-Pereira, J.; Correia, C.M. Drought stress effects and olive tree acclimation under a changing climate. Plants 2019, 8, 232. [Google Scholar] [CrossRef]
- Taiz, L.; Zeiger, E. Plant Physiology, 4th ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2006. [Google Scholar]
- Fernández, J.E.; Diaz-Espejo, A.; Infante, J.M.; Durán, P.; Palomo, M.J.; Chamorro, V.; Girón, I.F.; Villagarcía, L. Water relations and gas exchange in olive trees under regulated deficit irrigation and partial rootzone drying. Plant Soil 2006, 84, 273–291. [Google Scholar] [CrossRef]
- Stefanoudaki, E.; Chartzoulakis, K.; Koutsaftakis, A.; Kotsifaki, F. Effect of drought stress on qualitative characteristics of olive oil of cv Koroneiki. Grasas Aceites 2001, 52, 202–206. [Google Scholar] [CrossRef]
- Tognetti, R.; D’Andria, R.; Sacchi, R.; Lavini, A.; Morelli, G.; Alvino, A. Deficit irrigation affects seasonal changes in leaf physiology and oil quality of Olea europaea (cultivars Frantoio and Leccino). Ann. Appl. Biol. 2007, 150, 169–186. [Google Scholar] [CrossRef]
- Gomez-Cadenas, A.; Tadeo, F.R.; Talon, M.; Primo Millo, E. Leaf abscission induced by ethylene in water-stressed intact seedlings of Cleopatra mandarin requires previous abscisic acid accumulation in roots. Plant Physiol. 1996, 112, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Scortichini, M.; Cesari, G.L. An evaluation of monitoring surveys of the quarantine bacterium Xylella fastidiosa performed in containment and buffer areas of Apulia, southern Italy. Appl. Biosaf. 2019, 24, 96–99. [Google Scholar] [CrossRef]
- Scortichini, M. The multi-millenial olive agro-ecosystem of Salento (Apulia, Italy) threatened by Xylella fastidiosa subsp. pauca: A working possibility of restoration. Sustainability 2020, 12, 6700. [Google Scholar] [CrossRef]





© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scortichini, M. Predisposing Factors for “Olive Quick Decline Syndrome” in Salento (Apulia, Italy). Agronomy 2020, 10, 1445. https://doi.org/10.3390/agronomy10091445
Scortichini M. Predisposing Factors for “Olive Quick Decline Syndrome” in Salento (Apulia, Italy). Agronomy. 2020; 10(9):1445. https://doi.org/10.3390/agronomy10091445
Chicago/Turabian StyleScortichini, Marco. 2020. "Predisposing Factors for “Olive Quick Decline Syndrome” in Salento (Apulia, Italy)" Agronomy 10, no. 9: 1445. https://doi.org/10.3390/agronomy10091445
APA StyleScortichini, M. (2020). Predisposing Factors for “Olive Quick Decline Syndrome” in Salento (Apulia, Italy). Agronomy, 10(9), 1445. https://doi.org/10.3390/agronomy10091445



