Analysis of Pollination Services Provided by Wild and Managed Bees (Apoidea) in Wild Blueberry (Vaccinium angustifolium Aiton) Production in Maine, USA, with a Literature Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Fields
2.2. Fruit Set
2.3. Economic Modeling
2.4. Determining Proportions of Bee Groups
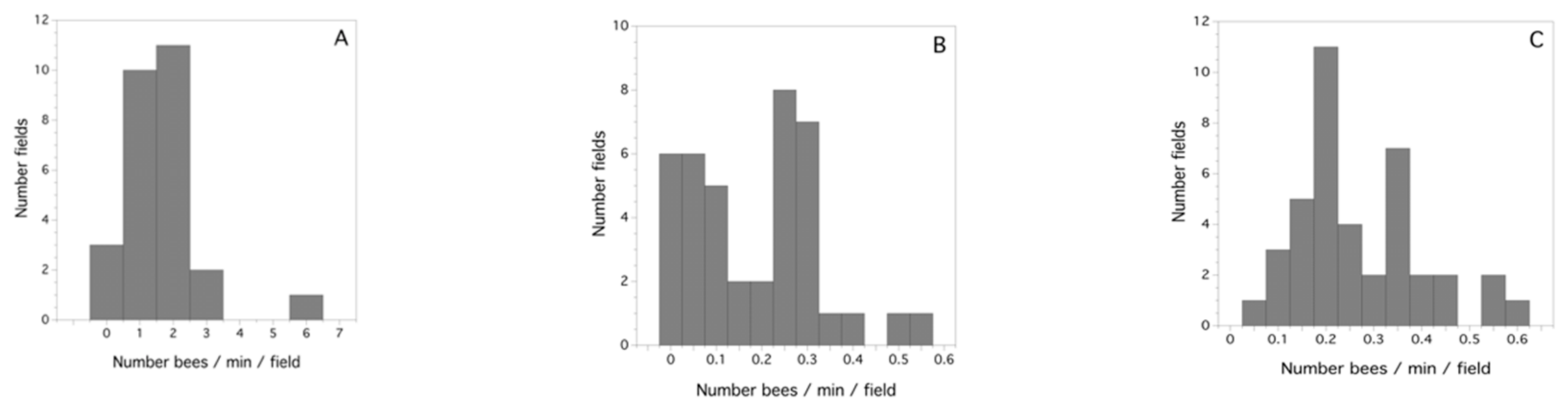
2.5. Determining Honey-Bee Activity and the Proportion of Honey Bees Foraging for 2010
2.6. Adjusting the Economic Value of the Bee Groups on the Basis of a Pollination Equivalency
2.7. Determining Species Richness
2.8. Statistical Analysis
3. Results
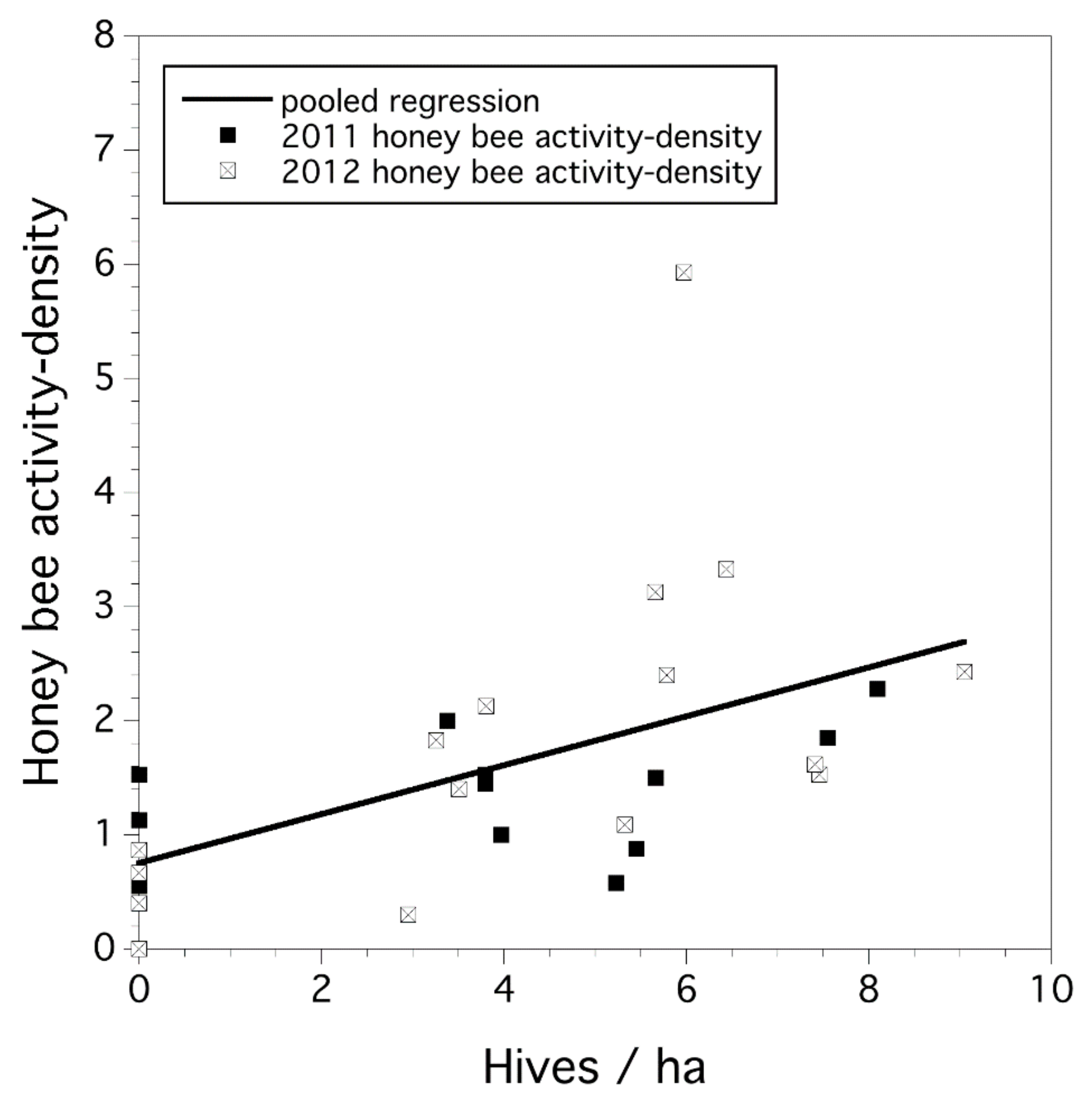
3.1. Honey-Bee Activity Density and Hive Stocking Rate
3.2. Study-Year Variation in Bee Activity Density
3.3. Species Richness and Bee Activity Density
3.4. Field Size
3.5. Fruit Set
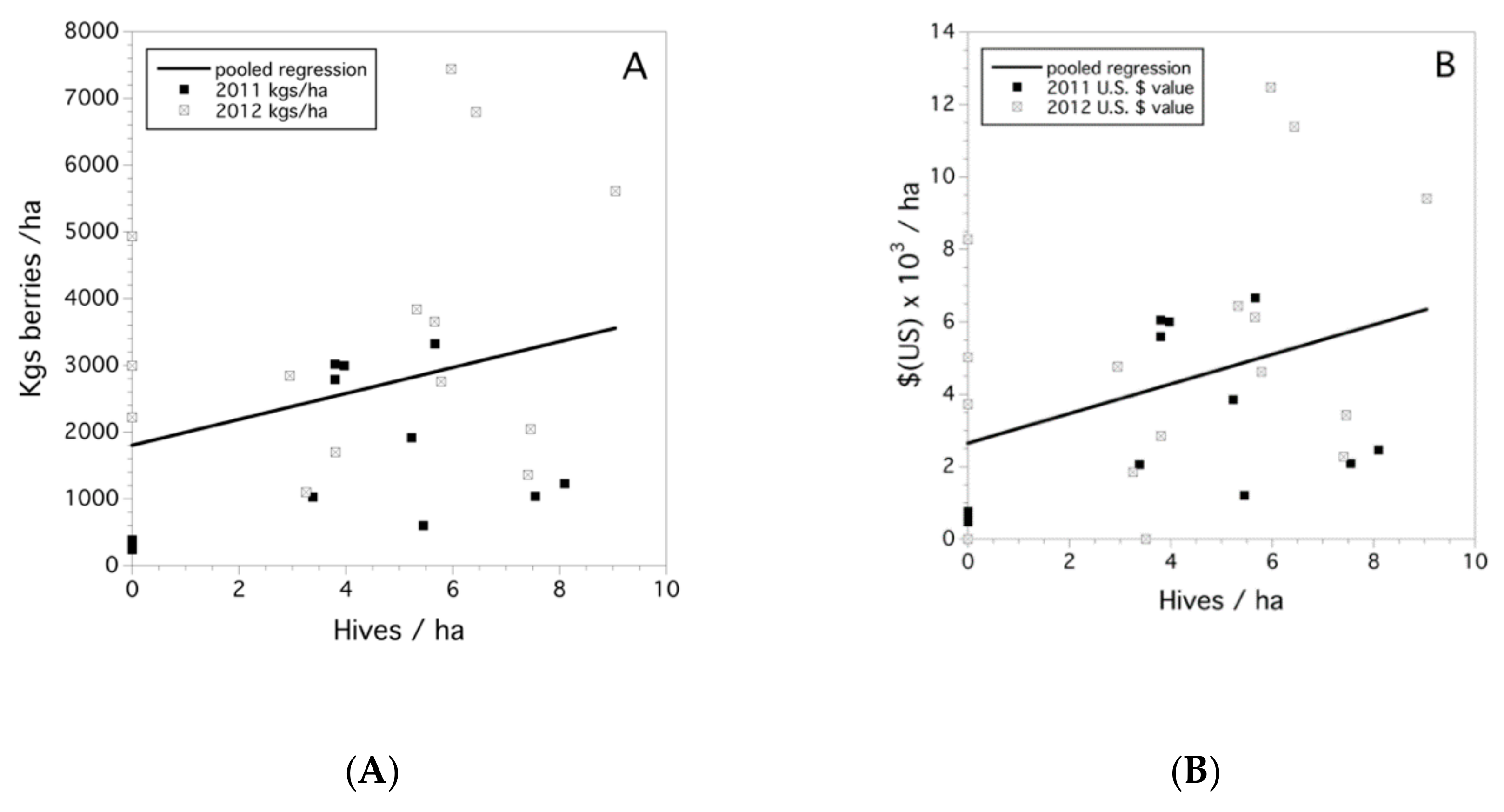
3.6. Yield and Crop Value
3.7. Economic Value of Wild Bees and Honey Bees
3.8. Pollination without Rental Honeybees
4. Discussion
4.1. Bee Abundance
4.2. Bee Abundance and Crop Fruit Set, Yield, and Crop Value
4.3. Bee Abundance and Field Size
4.4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Literature Review of Wild-Blueberry Reproductive Biology and Pollination
References
- Yarborough, D.E. Wild Blueberry Culture in Maine; Cooperative Extension: Maine Wild Blueberries Fact Sheet 220; The University of Maine: Orono, ME, USA, 2009; Available online: http://umaine.edu/blueberries/factsheets/production/wild-blueberry-culture-in-maine/ (accessed on 30 April 2020).
- Calderwood, L.; Yarborough, D.E.; Tooley, B. Maine Wild Blueberry Production Statistics; Cooperative Extension: Maine Wild Blueberries Fact Sheet; The University of Maine: Orono, ME, USA, 2019; Available online: https://extension.umaine.edu/blueberries/factsheets/statistics-2/crop-production-statistics-2019/ (accessed on 30 April 2020).
- NASS National Agricultural Statistics Service. Maine Agricultural Overview. 2019. Available online: http://www.nass.usda.gov/Statistics_by_State/Maine/ (accessed on 30 April 2020).
- Bell, D.J.; Rowland, L.J.; Smagula, J.; Drummond, F.A. Recent Advances in the Biology and Genetics of Lowbush Blueberry; Techical Bulletin 203; Maine Agricultural & Forest Experiment Station, University of Maine: Orono, ME, USA, 2009; p. 36. [Google Scholar]
- Bushmann, S.L.; Drummond, F.A. Abundance and Diversity of Wild Bees (Hymenoptera: Apoidea) Found in Lowbush Blueberry Growing Regions of Downeast Maine. Environ. Entomol. 2015, 44, 975–989. [Google Scholar] [CrossRef] [PubMed]
- Dibble, A.C.; Drummond, F.A.; Stubbs, C.; Veit, M.; Ascher, J.S. Bees of Maine, with a State Species Checklist. Northeast. Nat. 2017, 24, 1–48. [Google Scholar] [CrossRef]
- Javorek, S.K.; MacKenzie, K.E.; Kloet, S.P.V. Comparative Pollination Effectiveness among Bees (Hymenoptera: Apoidea) on Lowbush Blueberry (Ericaceae: Vaccinium angustifolium). Ann. Entomol. Soc. Am. 2002, 95, 345–351. [Google Scholar] [CrossRef]
- Drummond, F.A. Behavior of bees associated with the wild blueberry agro-ecosystem in the USA. Int. J. Entomol. Nematol. 2016, 2, 27–41. [Google Scholar]
- Lee, W.R. Pollination Studies on Low-Bush Blueberries. J. Econ. Entomol. 1958, 51, 544–545. [Google Scholar] [CrossRef]
- Rose, A.; Drummond, F.A.; Yarborough, D.E.; Asare, E. Maine Wild Blueberry Farmers: A 2010 Economic and Sociological Analysis of a Traditional Downeast Crop in Transition; Miscellaneous Report; Maine Agricultural & Forest Experiment Station, University of Maine: Orono, ME, USA, 2013; p. 445. [Google Scholar]
- Hanes, S.P.; Collum, K.; Hoshide, A.K.; Drummond, F.A.; Asare, E. Farmer perceptions of native pollinators and pollination strategies in the lowbush blueberry industry. Renew. Agric. Food Syst. 2013, 28, 1–8. [Google Scholar] [CrossRef]
- Hanes, S.; Collum, K.; Drummond, F.; Hoshide, A. Assessing Wild Pollinators in Conventional Agriculture: A Case Study from Maine’s Blueberry Industry. Hum. Ecol. Rev. 2018, 24, 97–113. [Google Scholar] [CrossRef]
- MacKenzie, K.; Rogers, D.; Javorek, S. The alfalfa leafcutting bee, Megachile rotundata Fabr.: An alternative managed pollinator of lowbush blueberry. Acta Hortic. 1997, 446, 87–90. [Google Scholar] [CrossRef]
- Stubbs, C.S.; Drummond, F.A. Management of the alfalfa leafcutting bee, Megachile rotundata (Hymenoptera: Megachilidae), for pollination of wild lowbush blueberry. J. Kans. Entomol. Soc. 1997, 70, 81–93. [Google Scholar]
- Whidden, T.L. The fidelity of commercially reared colonies of Bombus impatiens Cresson (Hymenoptera: Apidae) to lowbush blueberry in southern New Brunswick. Can. Entomol. 1996, 128, 957–958. [Google Scholar] [CrossRef]
- Stubbs, C.S.; Drummond, F.A. Bombus impatiens (Hymenoptera: Apidae): An alternative to Apis mellifera (Hymenoptera: Apidae) for lowbush blueberry production. J. Econ. Entomol. 2001, 94, 609–616. [Google Scholar] [CrossRef] [PubMed]
- Drummond, F.A. Commercial bumble bee pollination of lowbush blueberry. Int. J. Fruit Sci. 2012, 12, 54–64. [Google Scholar] [CrossRef]
- Asare, E.; Hoshide, A.K.; Drummond, F.A.; Chen, X.; Criner, G.K. Economic risk of bee pollination in Maine wild blueberry, Vaccinium angustifolium Aiton. J. Econ. Entomol. 2017, 110, 1980–1992. [Google Scholar] [CrossRef] [PubMed]
- Hoshide, A.K.; Drummond, F.A.; Stevens, T.H.; Venturini, E.M.; Hanes, S.P.; Sylvia, M.M.; Loftin, C.S.; Yarborough, D.E.; Averill, A.L. What is the value of wild bee pollination for wild blueberries and cranberries and who values it? Environments 2018, 5, 98. [Google Scholar] [CrossRef]
- Venturini, E.M.; Drummond, F.A.; Hoshide, A.K.; Dibble, A.C.; Stack, L.B. Pollination reservoirs for wild bee habitat enhancement: A review. Agroecol. Sustain. Food Syst. 2017, 41, 101–142. [Google Scholar] [CrossRef]
- Goulson, D.; Lye, G.C.; Darvill, B. Decline and conservation of bumble bees. Annu. Dev. Entomol. 2008, 53, 191–208. [Google Scholar] [CrossRef]
- Goulson, D.; Nicholls, E.; Botias, C.; Rotheray, E.L. Bee declines driven by combined stress from parasites, pesticides, and lack of flowers. Science 2015, 347, 1255957. [Google Scholar] [CrossRef]
- Allen-Wardwell, G.; Bernhardt, P.; Bitner, R.; Burquez, A.; Buchmann, S.; Cane, J.; Allen Cox, P.; Dalton, V.; Feinsinger, P.; Ingram, M.; et al. The potential consequences of pollinator declines on the conservation of biodiversity and stability of food crop yields. Conserv. Biol. 1998, 12, 8–17. [Google Scholar]
- Kevan, P.G. Pollinators as bioindicators of the state of the environment: Species, activity and diversity. Agric. Ecosys. Environ. 1999, 74, 373–393. [Google Scholar] [CrossRef]
- Winfree, R. Pollinator-dependent crops: An increasingly risky business. Curr. Biol. 2008, 18, R968–R969. [Google Scholar] [CrossRef]
- Borns, H.W. Changing paradigms of surficial geology in Maine: From the biblical flood to glaciation. In Deglacial History and Relative Sea-level Changes, Northern New England and Adjacent Canada; Special Paper 351; Geological Society of America: Boulder, CO, USA, 2001; Volume 351, pp. 1–7. [Google Scholar]
- Drummond, F.A.; Smagula, J.; Annis, S.; Yarborough, D.E. Organic Wild Blueberry Production; Techincal Bulletin 852; Maine Agricultural & Forest Experiment Station, University of Maine: Orono, ME, USA, 2009; p. 43. [Google Scholar]
- Jones, M.S.; Vanhanen, H.; Peltola, R.; Drummond, F.A. A global review of arthropod-mediated ecosystem-services in Vaccinium berry agroecosystems. Terr. Arthropod Rev. 2014, 7, 41–78. [Google Scholar] [CrossRef]
- Gawler, S.; Cutko, A. Natural Landscapes of Maine: A Guide to Natural Communities and Ecosystems; Maine Natural Areas Program: Augusta, ME, USA, 2010; p. 347. [Google Scholar]
- ArcGIS; Version 10.0; Environmental Systems Research Institute Inc.: Redlands, CA, USA, 2011.
- Qu, H.; Drummond, F.A. Simulation-based modeling of wild blueberry pollination. Comput. Electron. Agric. 2018, 144, 94–101. [Google Scholar] [CrossRef]
- Drummond, F.A. Wild blueberry fruit drop: A consequence of seed set? Agronomy 2020, 10, 939. [Google Scholar] [CrossRef]
- Garibaldi, L.A.; Steffan-Dewenter, I.; Winfree, R.; Aizen, M.A.; Bommarco, R.; Cunningham, S.A.; Kremen, C.; Carvalheiro, L.G.; Harder, L.D.; Afik, O.; et al. Wild Pollinators Enhance Fruit Set of Crops Regardless of Honey Bee Abundance. Science 2013, 339, 1608–1611. [Google Scholar] [CrossRef] [PubMed]
- SAS Institute. JMP®; Version 14; SAS Institute Inc.: Cary, NC, USA, 2017. [Google Scholar]
- Losey, J.E.; Vaughan, M. The economic value of ecological services provided by insects. BioScience 2006, 56, 311–323. [Google Scholar] [CrossRef]
- Cane, J.H. Exotic non-social bees (Hymenoptera: Apoidea) in North America: Ecological implications. In For Nonnative Crops, Whence Pollinator for the Future? Strickler, K., Cane, J.H., Eds.; Thomas Say Publications in Entomology: Lanham, MD, USA, 2003. [Google Scholar]
- Stubbs, C.S.; Drummond, F.A.; Ginsberg, H. Effects of Invasive Plant Species on Pollinator Service and Reproduction in Native Plants at Acadia National Park; Technical Report NPS/NER/NRTR- 2007-096; National Park Service: Boston, MA, USA, 2007.
- Cutler, G.C.; Reeh, K.W.; Sproule, J.M.; Ramanaidu, K. Berry unexpected: Nocturnal pollination of lowbush blueberry. Can. J. Plant Sci. 2012, 92, 707–711. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.V.; Christensen, R.H.B. Lmertest Package: Tests in linear mixed effects models. J. Stat. Software 2017, 88, 1–26. [Google Scholar] [CrossRef]
- Yarborough, D.E. Improving Your Wild Blueberry Yields; Cooperative Extension: Maine Wild Blueberries Fact Sheet; The University of Maine: Orono, ME, USA, 2013; Available online: https://extension.umaine.edu/blueberries/factsheets/wild-blueberry-production/ (accessed on 12 July 2020).
- Eaton, L.J.; Murray, J.E. Relationships of pollinator numbers in blueberry fields to fruit development and yields. Acta Hortic. 1997, 446, 181–188. [Google Scholar] [CrossRef]
- Eaton, L.J.; Nams, V.O. Honeybee stocking numbers and wild blueberry production in Nova Scotia. Can. J. Plant Sci. 2012, 92, 1305–1310. [Google Scholar] [CrossRef]
- Boulanger, L.W. Blueberry pollination and solitary bees. Maine Farm Res. 1964, 12, 5–11. [Google Scholar]
- Desjardins, E.; de Oliveira, D. Commercial bumble bee Bombus impatiens (Hymenoptera: Apidae) as a pollinator in lowbush blueberry (Ericale: Ericaceae) fields. J. Econ. Entomol. 2006, 99, 443–449. [Google Scholar] [CrossRef] [PubMed]
- Smagula, J.M.; Litten, W.; Chen, Y.; Dunham, S. Variation of fruit set and fruit characteristics of wild lowbush blueberries (Vaccinium angustifolium Ait.) in a managed field. Acta Hortic. 1997, 446, 109–115. [Google Scholar] [CrossRef]
- Schut, L.; Tyedmers, P.; Cutler, G.C.; Melathopoulos, A. Is early pollination to lowbush blueberry an ecosystem service or disservice? Agric. Ecosys. Environ. 2017, 239, 368–375. [Google Scholar] [CrossRef]
- Wood, G.W.; Wood, F.A. Nectar production and its relation to fruitset in the lowbush blueberry. Can. J. Bot. 1963, 41, 1675–1679. [Google Scholar] [CrossRef]
- Yarborough, D. Blueberry Enterprise Budget; Cooperative Extension: Maine Wild Blueberries Fact Sheet 260; The University of Maine: Orono, ME, USA, 2016; Available online: https://extension.umaine.edu/blueberries/factsheets/marketing-and-business-management/260-blueberry-enterprise-budget/ (accessed on 11 July 2020).
- Greenleaf, S.; Williams, N.; Winfree, R.; Kremen, C. Bee foraging ranges and their relationship to body size. Oecologia 2007, 153, 589–596. [Google Scholar] [CrossRef]
- Groff, S.C.; Loftin, C.S.; Drummond, F.A.; Bushmann, S.; McGill, B. Spatial prediction of lowbush blueberry native bee pollinators in Maine, USA. Environ. Model. Soft. 2016, 79, 1–9. [Google Scholar] [CrossRef]
- Drummond, F.A.; Stubbs, C.S. Sampling bee populations in lowbush blueberry in Maine. Acta Hortic. 1997, 446, 101–108. [Google Scholar] [CrossRef]
- Cutler, G.C.; Nams, V.O.; Craig, P.; Sproule, J.M.; Sheffield, C.S. Wild bee pollinator communities of lowbush blueberry fields: Spatial and temporal trends. Basic Appl. Ecol. 2014, 16, 73–85. [Google Scholar] [CrossRef]
- Stubbs, C.S.; Jacobson, H.A.; Osgood, E.A.; Drummond, F.A. Alternative forage plants for native (wild) bees associated with lowbush blueberry, Vaccinium spp., in Maine; Technical Bulletin 148; Maine Agricultural & Forest Experiment Station, University of Maine: Orono, ME, USA, 1992. [Google Scholar]
- Isaacs, R.; Kirk, A.K. Pollination services provided to small and large highbush blueberry fields by wild and managed bees. J. Appl. Ecol. 2010, 47, 841–849. [Google Scholar] [CrossRef]
- Aras, P.; De Oliveira, D.; Savoie, L. Effect of a honey bee (Hymenoptera: Apidae) gradient on the pollination and yield of lowbush blueberry. J. Econ. Entomol. 1996, 89, 1080–1083. [Google Scholar] [CrossRef]
- Venturini, E.M.; Drummond, F.A.; Ballman, E. Native Andrena response to burning in the wild blueberry agroecosystem. J. Kans. Entomol. Soc. 2017, 90, 131–145. [Google Scholar] [CrossRef]
- Venturini, E.M.; Drummond, F.A.; Hoshide, A.K.; Dibble, A.C.; Stack, L.B. Pollination Reservoirs in Lowbush Blueberry (Ericales: Ericaceae). J. Econ. Entomol. 2017, 110, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Gallai, N.; Salles, J.; Settele, J.; Vaissiere, B.E. Economic valuation of the vulnerability of world agriculture confronted with pollinator decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Aizen, M.A.; Garibaldi, L.A.; Cunningham, S.A.; Klein, A.M. Long-term global trends in crop yield and production reveal no current pollination shortage but increasing pollinator dependency. Curr. Biol. 2008, 18, 1572–1575. [Google Scholar] [CrossRef]
- Calderone, N.W. Insect pollinated crops, insect pollinators and US agriculture: Trend analysis of aggregate data for the period 1992–2009. PLoS ONE 2012, 7, e37235. [Google Scholar] [CrossRef]
- McCauley, D.J. Selling out on nature. Nature 2006, 443, 27–28. [Google Scholar] [CrossRef]
- Winfree, R. The conservation and restoration of wild bees. Ann. N. Y. Acad. Sci. 2010, 1195, 169–197. [Google Scholar] [CrossRef]
- Klein, A.; Vaissiere, B.E.; Cane, J.H.; Steffan-Dewenter, I.; Cunningham, S.A.; Kremen, C.; Tscharntke, T. Importance of pollinators in changing landscapes for world crops. Proc. R. Soc. B 2007, 274, 303–313. [Google Scholar] [CrossRef]
- Vander Kloet, S.P. The Genus Vaccinium in North America; Agriculture and Agri-Food Canada: Ottawa, ON, Canada, 1988; Volume 1828.
- Beers, L.; Rowland, L.J.; Drummond, F.A. Genetic diversity of lowbush blueberry throughout its U.S. native range in managed and non-managed populations. Agriculture 2019, 9, 113. [Google Scholar] [CrossRef]
- Bell, D.J.; Rowland, L.J.; Polashcock, J.J.; Drummond, F.A. Suitability of EST-PCR markers developed in highbush blueberry for genetic fingerprinting and relationship studies in lowbush blueberry and related species. J. Am. Soc. Hortic. Sci. 2008, 133, 1–7. [Google Scholar] [CrossRef]
- DeGomez, T. Pruning Lowbush Blueberry Fields; Cooperative Extension: Maine Wild Blueberries Fact Sheet 229 UMaine Extension Number 2168; The University of Maine: Orono, ME, USA, 1988; Available online: https://extension.umaine.edu/blueberries/factsheets/production/pruning-lowbush-blueberry-fields/ (accessed on 30 April 2020).
- Myra, M.; MacKenzie, K.; Vander Kloet, S.P. Investigation of a possible sexual function specialization in the lowbush blueberry (Vaccinium angustifolium Aiton. Ericaceae). Small Fruits Rev. 2004, 3, 313–324. [Google Scholar] [CrossRef]
- Bell, D.J.; Rowland, L.J.; Drummond, F.A. Fine-scale spatial genetic structure associated with Vaccinium angustifolium Aiton (Ericaceae). Int. J. Bot. 2012, 2, 72–82. [Google Scholar] [CrossRef]
- Drummond, F. Reproductive Biology of Wild Blueberry (Vaccinium angustifolium Aiton). Agriculture 2019, 9, 69. [Google Scholar] [CrossRef]
- White, S.N.; Boyd, N.S.; van Acker, R.C. Growing degree-day models for predicting lowbush blueberry (Vaccinium angustifolium Ait.) ramet emergence, tip dieback, and flowering in Nova Scotia, Canada. HortScience 2012, 47, 1014–1021. [Google Scholar] [CrossRef]
- Bell, D.J.; Drummond, F.A.; Rowland, J.L. Evidence of functional gender polymorphisms in a population of the hermaphroditic lowbush blueberry (Vaccinium angustifolium Ait.). Botany 2012, 90, 393–399. [Google Scholar] [CrossRef]
- Aalders, L.E.; Hall, I.V. Pollen incompatibility and fruit set in lowbush blueberries. Can. J. Genet. Cytol. 1961, 3, 300–307. [Google Scholar] [CrossRef]
- Usui, M.; Kevan, P.G.; Obbard, M. 2005. Pollination and breeding system of lowbush blueberries, Vaccinium angustifolium Ait. and V. myrtilloides Michx (Ericacaeae), in the boreal forest. Can. Field Nat. 2005, 119, 48–57. [Google Scholar] [CrossRef]
- Bell, D.J.; Rowland, L.J.; Stommel, J.; Drummond, F.A. Yield variation among clones of lowbush blueberry as a function of kinship and self-compatibility. J. Am. Soc. Hortic. Sci. 2010, 135, 1–12. [Google Scholar]
- Drummond, F.A.; Dibble, A.C.; Stubbs, C.; Bushmann, S.; Ascher, J.; Ryan, J. A natural history of change in native bees associated with lowbush blueberry in Maine. Northeast. Nat. 2017, 24, 49–68. [Google Scholar] [CrossRef]
- Du Clos, B.; Loftin, C.; Drummond, F.A. Non-crop habitat use by wild bees in a mixed-use agricultural landscape. Environ. Entomol. 2020, 49, 502–515. [Google Scholar] [CrossRef]
- Drummond, F.A.; Ballman, E.; Collins, J. Are They Weeds or a Life Force? Or Sustainability on the Edge. Spire Maine J. Conserv. Sustain. 2017. Available online: https://umaine.edu/spire/2017/05/04/drummond-et-al/ (accessed on 10 July 2020).
- Moisan-DeSerres, J.; Chagnon, M.; Fournier, V. Influence of windbreaks and forest borders on abundance and species richness of native pollinators in lowbush blueberry fields in Québec, Canada. Can. Entomol. 2014, 147, 432–442. [Google Scholar] [CrossRef]
- McKechnie, I.M.; Thomsen, C.J.; Sargent, R.D. Forested field edges support a greater diversity of wild pollinators in lowbush blueberry (Vaccinium angustifolium). Agric. Ecosys. Environ. 2017, 237, 154–161. [Google Scholar] [CrossRef]
- Dibble, A.C.; Berg Stack, L.; Drummond, F.A. Bee visitation on flowers in Maine, United States, reveals the relative attractiveness of plants through space and time: Part I. Environ. Entomol. 2020, 49, 726–737. [Google Scholar] [CrossRef]
- Dibble, A.C.; Berg Stack, L.; Drummond, F.A. Plant origin and other attributes impact bee forage patterns in a common garden study in Maine, United States; Part, II. Environ. Entomol. 2020, 49, 738–752. [Google Scholar] [CrossRef]
- Venturini, E.; Berg-Stack, L.; Dibble, A.C.; Drummond, F.A.; Hoshide, A.K. Enhancing Native Bees for Wild Lowbush Blueberry Crop Pollination: Bee Pasture; Cooperative Extension: Maine Wild Blueberries Fact Sheet; The University of Maine: Orono, ME, USA, 2015; p. 9. Available online: http://umaine.edu/blueberries/files/2010/05/2015-Bee-Pasture-Fact-Sheet.pdf (accessed on 10 July 2020).
- Venturini, E.M.; Drummond, F.A.; Hoshide, A.K. Organic establishment of pollination reservoirs in the lowbush blueberry (Ericales: Ericaceae) agroecosystem. Open Agric. 2018, 3, 393–403. [Google Scholar] [CrossRef]
- Moisan-Deserres, J.; Girard, M.; Chagnon, M.; Fournier, V. Pollen loads and specificity of native pollinators of lowbush blueberry. J. Econ. Entomol. 2014, 107, 1156–1162. [Google Scholar] [CrossRef]
- Stubbs, C.S.; Drummond, F.A. Blueberry and cranberry (Vaccinium spp.) pollination: A comparison of managed and native bee foraging behavior. Acta Hortic. 1997, 437, 341–344. [Google Scholar] [CrossRef]
- Rowland, L.J.; Ogden, E.L.; Bell, D.J.; Drummond, F.A. Pollen-mediated gene flow in managed fields of lowbush blueberry. Can. J. Plant Sci. 2019, 100, 95–102. [Google Scholar] [CrossRef]
- Bajcz, A.; Hiebeler, D.; Drummond, F.A. Grid-Set-Match, an agent-based simulation model, predicts fruit set for the Maine lowbush blueberry (Vaccinium angustifolium) agroecosystem. Ecol. Model. 2017, 361, 80–94. [Google Scholar] [CrossRef]
- Yarborough, D.E.; Drummond, F.A.; Annis, S.; D’Appollonio, J. Maine Wild blueberry systems analysis. Acta Hortic. 2017, 1180, 151–160. [Google Scholar] [CrossRef]
- Obsie, E.Y.; Qu, H.; Drummond, F. Wild blueberry yield prediction using a combination of computer simulation and machine learning algorithms. Comput. Electron. Agric. 2020, 178, 105778. [Google Scholar] [CrossRef]
- Boulanger, L.W.; Wood, G.W.; Osgood, E.A.; Dirks, C.O. Native Bees Associated with the Lowbush Blueberry in Maine and Eastern Canada; Technical Bulletin 26; Maine Agricultural & Forest Experiment Station, University of Maine: Orono, ME, USA, 1967; p. 22. [Google Scholar]
- Osgood, E.A. Soil Characteristics of Nesting Sites of Solitary Bees Associated with the Low-Bush Blueberry in Maine; Technical Bulletin 59; Maine Agricultural & Forest Experiment Station, University of Maine: Orono, ME, USA, 1972. [Google Scholar]
- Drummond, F.A. Honeybees and Blueberry Pollination; Cooperative Extension: Maine Wild Blueberries Fact Sheet 629; The University of Maine: Orono, ME, USA, 2002; Available online: http://umaine.edu/blueberries/factsheets/bees/629-honey-bees-and-blueberry-pollination/ (accessed on 30 April 2020).
- Wood, G.W. The influence of honey bee pollination on fruit set of the lowbush blueberry. Can. J. Plant Sci. 1961, 41, 332–335. [Google Scholar] [CrossRef]
- Wood, G.W. Evidence of increased fruit set in lowbush blueberry by using honey bees. HortScience 1969, 4, 211–212. [Google Scholar]
- Lomond, D.; Larson, D.J. Honey bees, Apis mellifera (Hymenoptera: Apidae), as pollinators of lowbush blueberry, Vaccinium angustifolium, on Newfoundland coastal barrens. Can. Entomol. 1983, 115, 1647–1651. [Google Scholar] [CrossRef]
- Stubbs, C.S.; Drummond, F.A. Pollination of lowbush blueberry by Anthophora pallipes villosula and Bombus impatiens (Hymenoptera: Anthophoridae and Apidae). J. Kans. Entomol. Soc. 2000, 72, 330–333. [Google Scholar]
- Stubbs, C.S.; Drummond, F.A.; Osgood, E.A. Osmia ribifloris biedermannii and Megachile rotundata (Hymenoptera: Megachilidae) introduced into the lowbush blueberry agroecosystem in Maine. J. Kans. Entomol. Soc. 1994, 67, 173–185. [Google Scholar]
- Fulton, M.; Jesson, L.K.; Bobiwash, K.; Schoen, D.J. Mitigation of pollen limitation in the lowbush blueberry agroecosystem: Effect of augmenting natural pollinators. Ecosphere 2015, 6, 1–19. [Google Scholar] [CrossRef]
- Stubbs, C.S.; Drummond, F.A.; Allard, S.L. Bee conservation and increasing Osmia spp. in Maine lowbush blueberry fields. Northeast. Nat. 1997, 4, 133–144. [Google Scholar] [CrossRef]
- Calderwood, L.; Drummond, F. IPM Tactics to Reduce Pesticide Exposure to Honey and Native Bees; Cooperative Extension: Maine Wild Blueberries Fact Sheet; The University of Maine: Orono, ME, USA, 2019; Available online: https://extension.umaine.edu/blueberries/wp-content/uploads/sites/41/2019/01/2009e.pdf (accessed on 16 August 2020).
- Drummond, F.A. Estimating Fruit Set and Pollinator Force in Wild Blueberry; Cooperative Extension: Maine Wild Blueberries Fact Sheet 2019; The University of Maine: Orono, ME, USA, 2020; Available online: https://extension.umaine.edu/blueberries/estimating-the-strength-of-your-pollinator-force-in-wild-blueberry/ (accessed on 12 July 2020).
- Drummond, F.A.; Yarborough, D. Commercial Bumble Bee Management (Bombus impatiens) for Wild Blueberry Pollination; Cooperative Extension: Maine Wild Blueberries Fact Sheet 302 UMaine Extension Number 2421; The University of Maine: Orono, ME, USA, 2002; Available online: https://extension.umaine.edu/blueberries/factsheets/bees/302-commercial-bumble-bee-bombus-impatiens-management-for-wild-blueberry-pollination/ (accessed on 12 July 2020).
- Stubbs, C.S.; Drummond, F.A. Pollination of wild lowbush blueberry, Vaccinium angustifolium by the alfalfa leafcutting bee, Megachile rotundata. Acta Hortic. 1997, 446, 189–196. [Google Scholar] [CrossRef]
- Sheffield, C.S. Summer bees for spring crops? Potential problems with Megachile rotundata (Fab.) (Hymenoptera: Megachilidae) as a pollinator of lowbush blueberry (Ericaceae). J. Kans. Entomol. Soc. 2008, 81, 276–287. [Google Scholar] [CrossRef]
- Drummond, F.A. Factors that affect yield in wild blueberry, Vaccinium angustifolium Aiton. Agric. Res. Tech. 2019, 22, 556212. [Google Scholar] [CrossRef]
- Bell, H.P. The development of the blueberry seed. Can. J. Botany 1957, 35, 139–153. [Google Scholar] [CrossRef]
- Stevens, T.; Hoshide, A.K.; Drummond, F.A. Willingness to pay for native pollination of blueberries: A conjoint analysis. Int. J. Agric. Market. 2015, 2, 68–77. [Google Scholar]
- Jesson, L.; Schoen, D.; Cutler, C.; Bates, S. Pollination in Lowbush Blueberry; A summary of research findings from the Canadian Pollination Initiative; NSERC-CANPOLIN School of Environmental Sciences, University of Guelph: Guelph, ON, Canada, 2014. [Google Scholar]
- Drummond, F.A.; Stubbs, C.S. Wild Bee Conservation for Wild Blueberry Fields; Cooperative Extension: Maine Wild Blueberries Fact Sheet 630; The University of Maine: Orono, ME, USA, 2003; p. 12. Available online: https://extension.umaine.edu/blueberries/factsheets/bees/630-wild-bee-conservation-for-wild-blueberry-fields/ (accessed on 12 July 2020).
- Stubbs, C.S.; Drummond, F.A.; Yarborough, D. Field Conservation Management of Native Leafcutting and Mason Osmia Bees; Cooperative Extension: Maine Wild Blueberries Fact Sheet 301 UMaine Extension No. 2420; The University of Maine: Orono, ME, USA, 2000; p. 6. Available online: https://extension.umaine.edu/blueberries/factsheets/bees/301-field-conservation-management-of-native-leafcutting-and-mason-osmia-bees/ (accessed on 12 July 2020).
- Skinner, J.; Wilson, M.; Asare, E.; Bajcz, A.; Bickerman, K.; Chapin, S.; Dibble, A.C.; du Clos, B.; Collum, K.; Hanes, S. How to Estimate Native Bee Abundance in the Field; The University of Maine: Orono, ME, USA, 2014; Available online: https://extension.umaine.edu/blueberries/factsheets/pollination/ (accessed on 12 July 2020).
- Du Clos, B.; Loftin, C.; Drummond, F.A. BeeMapper Tool Quick Reference Guide. 2017. Available online: https://umaine.edu/beemapper/wp-content/uploads/sites/353/2017/07/BeeMapper-Quick-Guide-for-Web.pdf (accessed on 5 May 2020).
| Fruit Set Model | Predictors | Degrees of Freedom | F Ratio | Probability 1 | Coefficient |
|---|---|---|---|---|---|
| Late fruit set HB, WB, Richness | |||||
| Fixed effects Marginal r2: 0.336 | HB WB Richness | 1, 24 1, 24 1, 24 | 6.604 6.416 4.046 | 0.017 0.018 0.056 | 5.18 ± 2.02 28.56 ± 11.28 −0.75 ± 0.37 |
| Random effects | Year Year × HB Year × WB Year × Richness | <0.001 0.164 0.133 0.376 | |||
| Late fruit set HB, BB, OB, Richness | |||||
| Fixed effects Marginal r2: 0.398 | HB BB OB Richness | 1, 23 1, 23 1, 23 1, 23 | 5.982 18.523 2.098 3.029 | 0.023 <0.001 0.161 0.095 | 5.14 ± 2.10 28.45 ± 6.61 29.37 ± 20.28 −0.77 ± 0.44 |
| Random effects | Year Year × HB Year × BB Year × OB Year × Richness | <0.001 0.210 0.311 0.402 0.719 | |||
| CV of fruit set HB, WB, Richness | |||||
| Fixed effects Marginal r2: 0.203 | HB WB Richness | 1, 24 1, 24 1, 24 | 3.328 4.493 1.115 | 0.086 0.045 0.301 | −6.25 ± 3.43 − 40.62 ± 19.16 0.67 ± 0.64 |
| Random effects | Year Year × HB Year × WB Year × Richness | <0.001 0.095 0.115 0.299 | |||
| CV of fruit set HB, BB, OB, Richness | |||||
| Fixed effects Marginal r2: 0.278 | HB BB OB Richness | 1, 22 1, 22 1, 22 1, 22 | 159.366 2731.374 0.119 0.102 | <0.001 <0.001 0.733 0.752 | −8.15 ± 0.65 −52.26 ± 5.98 −31.22 ± 90.50 0.32 ± 0.35 |
| Random effects | Year Year × HB Year × BB Year × OB Year × Richness | <0.001 0.177 0.226 0.326 0.564 | |||
| Fruit Set Model | Predictors | Degrees of Freedom | F Ratio | Probability 1 | Coefficient |
|---|---|---|---|---|---|
| Yield (kg/ha) Late fruit set, field size (ha), field management | |||||
| Fixed effects Marginal r2: 0.238 | Late fruit set Field size Management | 1, 22040 1, 100771 1, 385689 | 6.555 4.023 0.887 | 0.011 0.049 0.346 | 48.72 ± 19.03 −549.46 ± 198.08 −301.89 ± 320.56 |
| Random effects | Year Year × Late fruit set Year × Field size (ha) Year × Management | <0.001 0.764 0.589 0.677 |
| Bee Group | Mean Value of Pollination (USD) | Mean Value of Pollination (Percentage of Gross in USD) |
|---|---|---|
| A: bee groups considered as equivalent pollinators | ||
| HB | $19,370.55 | 64% |
| BB | $2033.85 | 14% |
| OB | $4239.15 | 22% |
| B: bee group activity density adjusted by pollination equivalencies | ||
| HB | $15,024.61 | 43% |
| BB | $5614.49 | 33% |
| OB | $5016.96 | 24% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bushmann, S.L.; Drummond, F.A. Analysis of Pollination Services Provided by Wild and Managed Bees (Apoidea) in Wild Blueberry (Vaccinium angustifolium Aiton) Production in Maine, USA, with a Literature Review. Agronomy 2020, 10, 1413. https://doi.org/10.3390/agronomy10091413
Bushmann SL, Drummond FA. Analysis of Pollination Services Provided by Wild and Managed Bees (Apoidea) in Wild Blueberry (Vaccinium angustifolium Aiton) Production in Maine, USA, with a Literature Review. Agronomy. 2020; 10(9):1413. https://doi.org/10.3390/agronomy10091413
Chicago/Turabian StyleBushmann, Sara L., and Francis A. Drummond. 2020. "Analysis of Pollination Services Provided by Wild and Managed Bees (Apoidea) in Wild Blueberry (Vaccinium angustifolium Aiton) Production in Maine, USA, with a Literature Review" Agronomy 10, no. 9: 1413. https://doi.org/10.3390/agronomy10091413
APA StyleBushmann, S. L., & Drummond, F. A. (2020). Analysis of Pollination Services Provided by Wild and Managed Bees (Apoidea) in Wild Blueberry (Vaccinium angustifolium Aiton) Production in Maine, USA, with a Literature Review. Agronomy, 10(9), 1413. https://doi.org/10.3390/agronomy10091413






