Disease Resistant Citrus Breeding Using Newly Developed High Resolution Melting and CAPS Protocols for Alternaria Brown Spot Marker Assisted Selection
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Recovery of Hybrids
2.2. DNA Isolation
2.3. Sequencing of the SNP08 Locus
2.4. Bioinformatics Analysis
2.5. Nested-PCR
2.6. HRM Genotyping
2.7. Development of Cleaved Amplified Polymorphic Sequences (CAPS) Marker
2.8. KASPar SNP Genotyping
2.9. Statistical Analysis
2.10. Isolation and Production of A. alternata Inoculum
2.11. Inoculation and Evaluation of Disease Severity in Detached Leaves
3. Results and Discussion
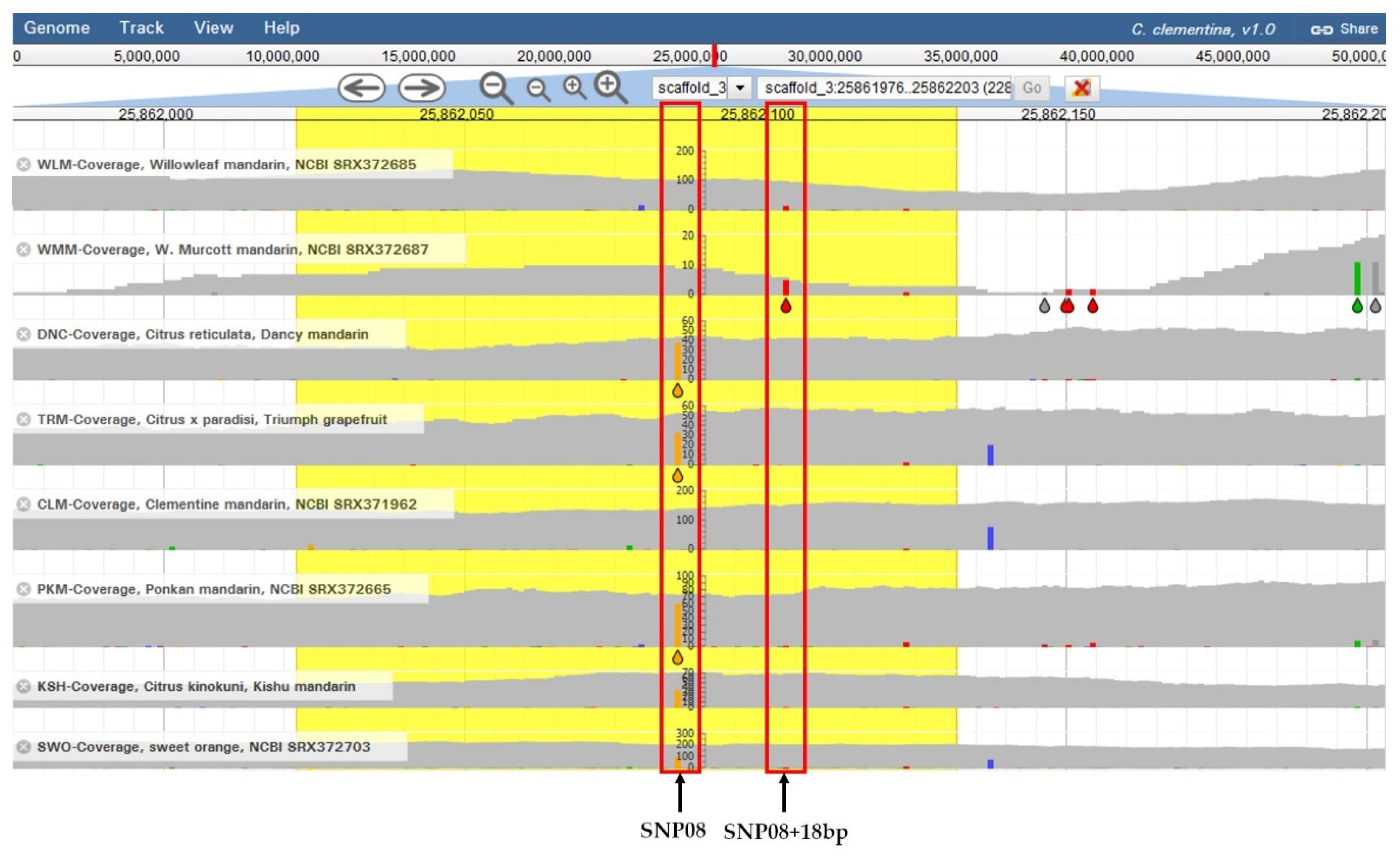
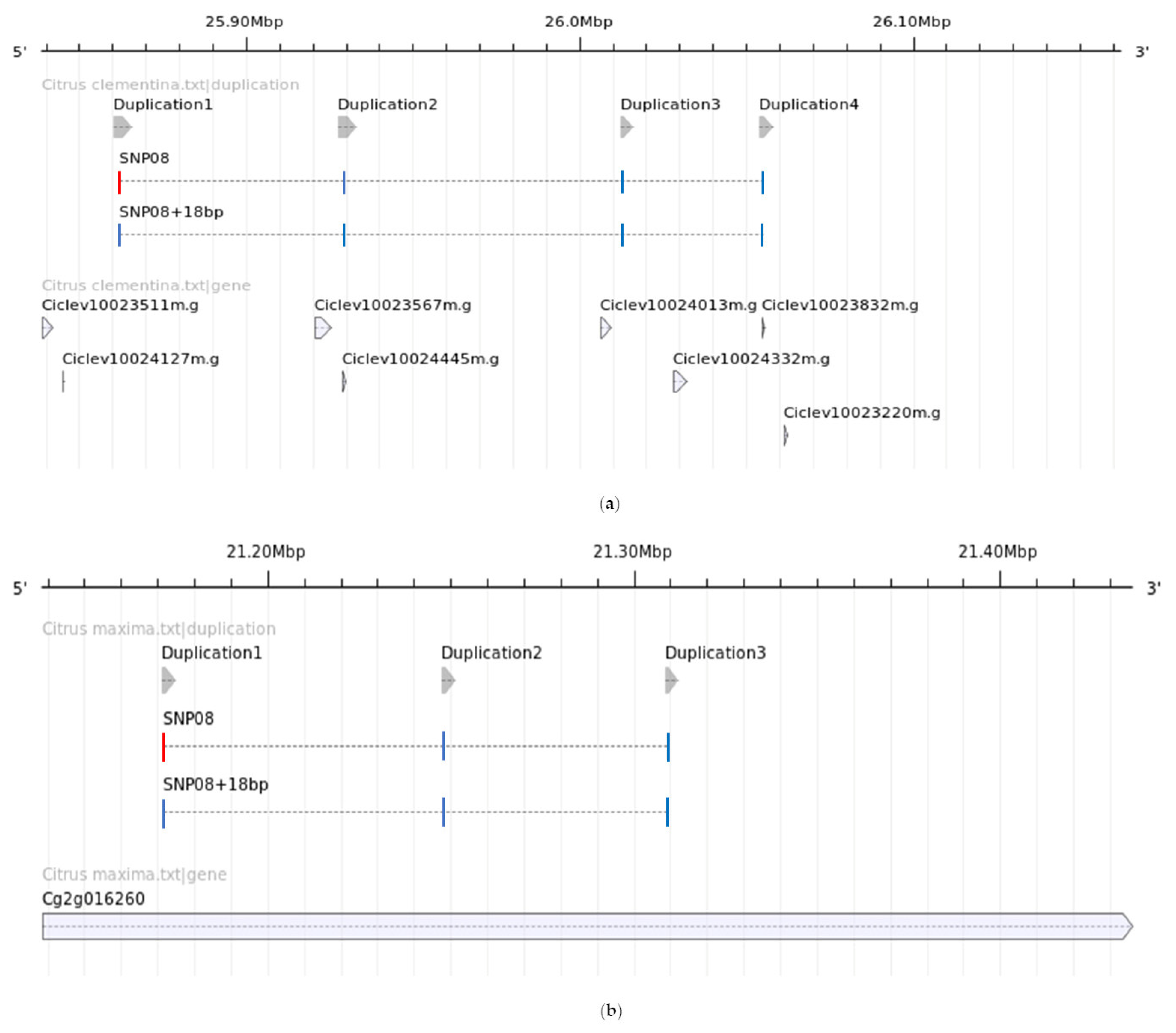
3.1. Sequencing and Analysis of the Locus Flanking SNP08 and Development of the HRM Protocol
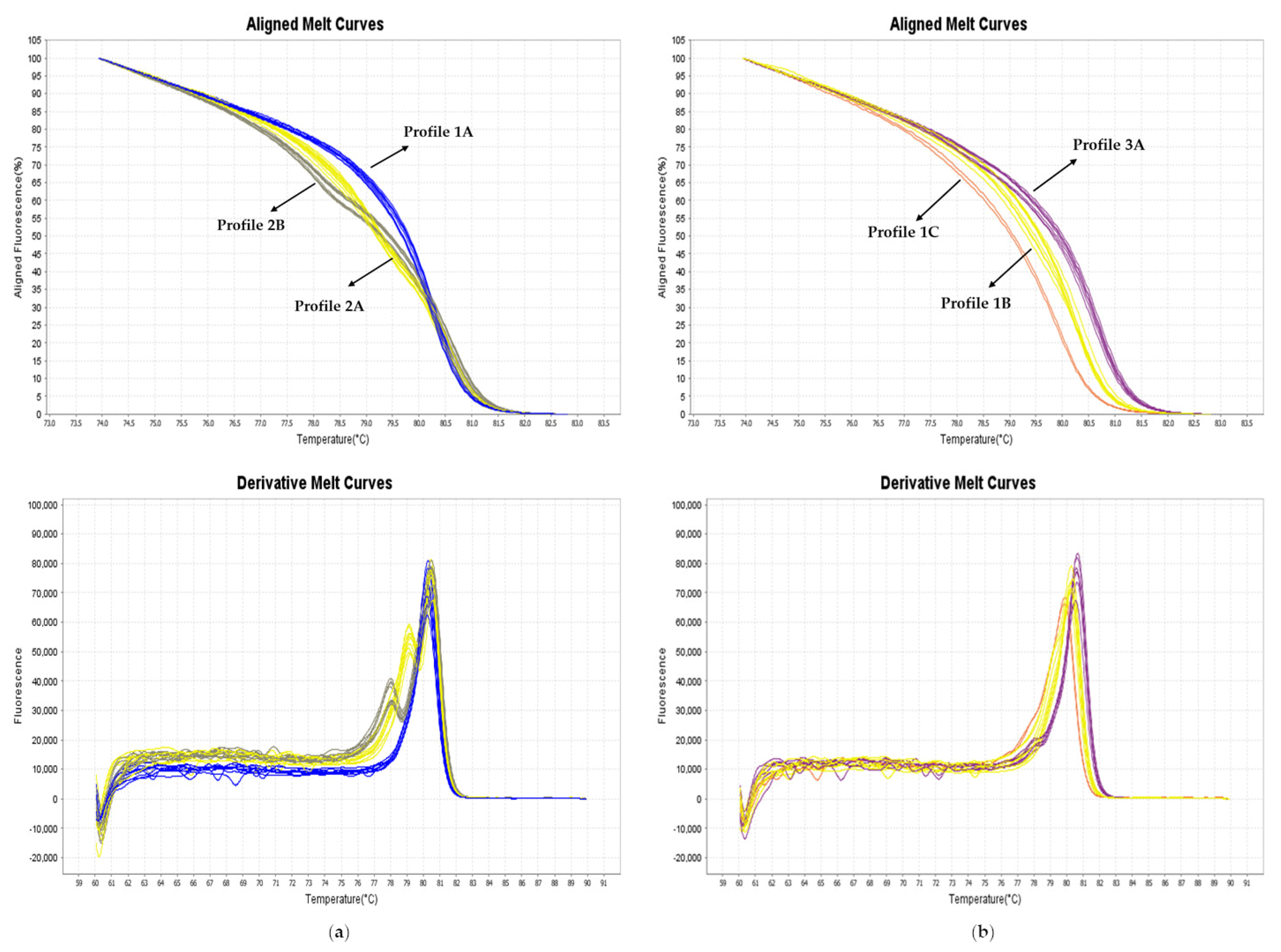
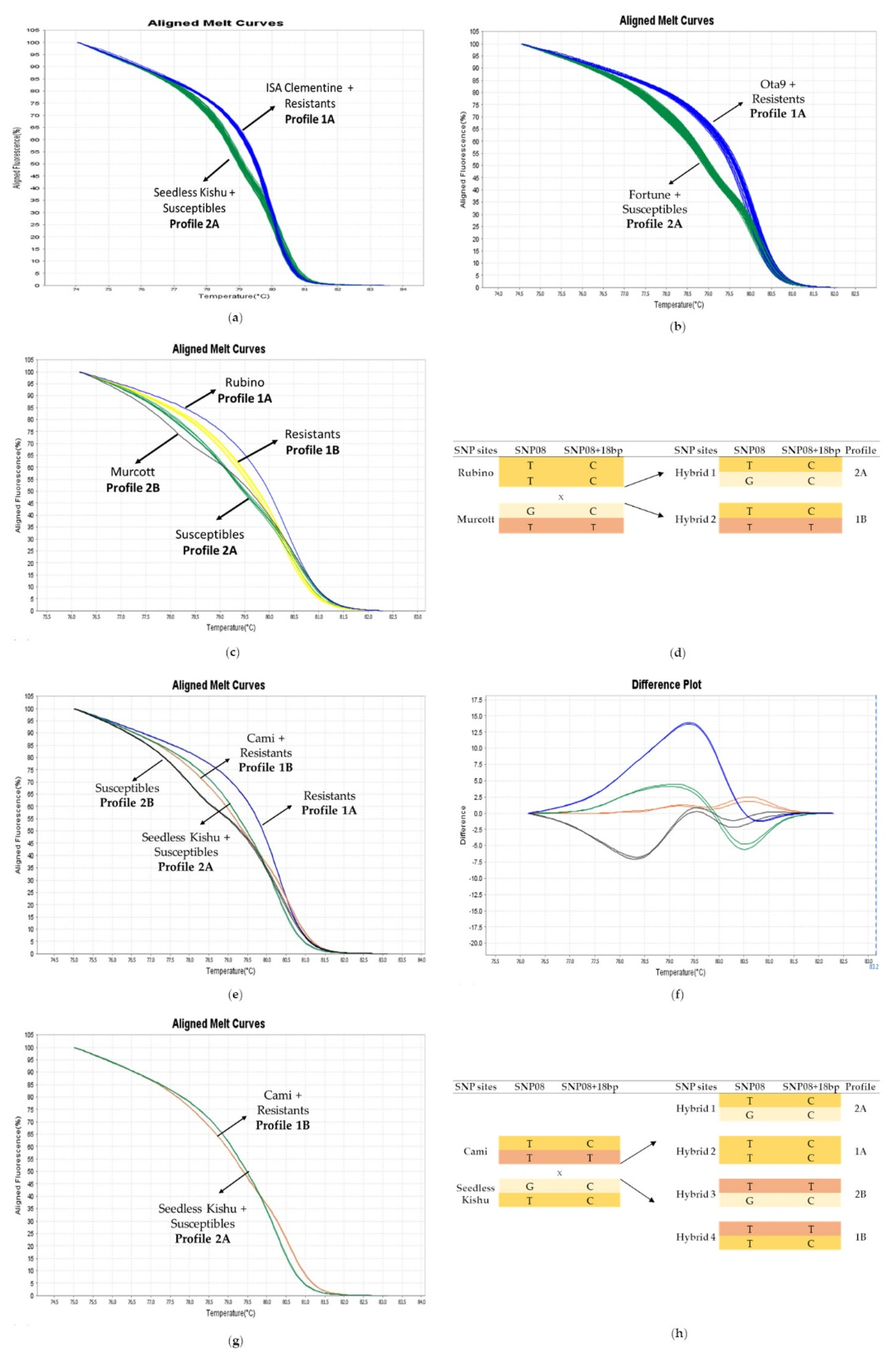
3.2. HRM Genotyping of the Germplasm Collection
3.3. HRM and KASPar Genotyping of the Hybrid Populations
3.4. CAPS Assay
3.5. In Vitro Phenotyping
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Caruso, M.; Smith, M.W.; Froelicher, Y.; Russo, G.; Gmitter, F.G. Traditional breeding. In The Genus Citrus; Talon, M., Caruso, M., Gmitter, F.G., Jr., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 129–148. [Google Scholar] [CrossRef]
- Shimizu, T. Genomic breeding. In The Genus Citrus; Talon, M., Caruso, M., Gmitter, F.G., Jr., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 149–169. ISBN 978-0-12-812163-4. [Google Scholar]
- Butelli, E.; Licciardello, C.; Zhang, Y.; Liu, J.; Mackay, S.; Bailey, P.; Reforgiato-Recupero, G.; Martin, C. Retrotransposons control fruit-specific, cold-dependent accumulation of anthocyanins in blood oranges. Plant Cell 2012, 24, 1242–1255. [Google Scholar] [CrossRef]
- Wang, X.; Xu, Y.; Zhang, S.; Cao, L.; Huang, Y.; Cheng, J.; Wu, G.; Tian, S.; Chen, C.; Liu, Y.; et al. Genomic analyses of primitive, wild and cultivated citrus provide insights into asexual reproduction. Nat. Genet. 2017, 49, 765–772. [Google Scholar] [CrossRef]
- Cuenca, J.; Aleza, P.; Garcia-lor, A.; Ollitrault, P.; Navarro, L. Fine Mapping for Identification of Citrus Alternaria Brown Spot Candidate Resistance Genes and Development of New SNP Markers for Marker-Assisted Selection. Front. Plant Sci. 2016, 7, 1948. [Google Scholar] [CrossRef] [PubMed]
- Endo, T.; Fujii, H.; Omura, M.; Shimada, T. Fast-track breeding system to introduce CTV resistance of trifoliate orange into citrus germplasm, by integrating early flowering transgenic plants with marker- assisted selection. BMC Plant Biol. 2020, 20, 224. [Google Scholar] [CrossRef] [PubMed]
- Kohmoto, K.; Scheffer, R.P.; Whiteside, J. Host-Selective Toxins from Alternaria citri. Phytopathology 1979, 69, 667–671. [Google Scholar] [CrossRef]
- Pegg, K.G. Studies of a strain of Alternaria citri Pierce, the causal organism of brown spot of Emperor mandarin. Qld. J. Agric. Anim. Sci. 1966, 23, 15–28. [Google Scholar]
- Meena, M.; Gupta, S.K.; Swapnil, P.; Zehra, A.; Dubey, M.K.; Upadhyay, R.S. Alternaria Toxins: Potential Virulence Factors and Genes Related to Pathogenesis. Front. Microbiol. 2017, 8, 1451. [Google Scholar] [CrossRef] [PubMed]
- Batuman, O.; Ritenour, M.; Vicent, A.; Li, H.; Hyun, J.-W.; Catara, V.; Ma, H.; Cano, L.M. Diseases caused by fungi and oomycetes. In The Genus Citrus; Talon, M., Caruso, M., Gmitter, F.G., Jr., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 349–369. ISBN 978-0-12-812163-4. [Google Scholar]
- Solel, Z.; Kimchi, M. Susceptibility and Resistance of Citrus Genotypes to Alternaria alternata pv. citri. J. Phytopathol. 1997, 145, 389–391. [Google Scholar] [CrossRef]
- Vicent, A.; Badal, J.; Asensi, M.J.; Sanz, N.; Armengol, J.; Garc, J. Laboratory evaluation of citrus cultivars susceptibility and influence of fruit size on Fortune mandarin to infection by Alternaria alternata pv. citri. Eur. J. Plant Pathol. 2004, 110, 245–251. [Google Scholar] [CrossRef]
- Vicent, A.; Armengol, J.; García-Jiménez, J. Rain fastness and persistence of fungicides for control of alternaria brown spot of citrus. Plant Dis. 2007, 91, 393–399. [Google Scholar] [CrossRef]
- Vicent, A.; Armengol, J.; Sales, R.; García-Jiménez, J.; Alfaro-Lassala, F. First Report of Alternaria Brown Spot of Citrus in Spain. Plant Dis. 2000, 84, 1044. [Google Scholar] [CrossRef] [PubMed]
- Hardy, S.; Barkley, P.; Treeby, M.; Smith, M.; Sanderson, G. Australian Mandarin Production Manual; New South Wales Department of Primary Industries: Orange, Australia, 2020; 310p.
- Miles, A.K.; Newman, T.K.; Gultzow, D.L.; Parfitt, S.C.; Drenth, A.; Smith, M.W. Commercial-scale Alternaria brown spot resistance screening as the first step in breeding new mandarins for Australia. Acta Hortic. 2015, 1065, 971–978. [Google Scholar] [CrossRef]
- Bassimba, D.D.M.; Mira, J.L.; Vicent, A. Evaluation of models for Alternaria brown spot of mandarin under Mediterranean conditions by partial receiver operating characteristic curve analysis. Phytopathology 2017, 107, 721–731. [Google Scholar] [CrossRef] [PubMed]
- De-Miguel, M.D.; Caballero, P.; Fernández-Zamudio, M.A. Varietal Change Dominates Adoption of Technology in Spanish Citrus Production. Agronomy 2019, 9, 631. [Google Scholar] [CrossRef]
- Cuenca, J.; Aleza, P.; Vicent, A.; Brunel, D.; Ollitrault, P.; Navarro, L. Genetically Based Location from Triploid Populations and Gene Ontology of a 3.3-Mb Genome Region Linked to Alternaria Brown Spot Resistance in Citrus Reveal Clusters of Resistance Genes. PLoS ONE 2013, 8, e76755. [Google Scholar] [CrossRef]
- Cuppen, E. Genotyping by allele-specific amplification (KASPar). CSH Protoc. 2007, 2007. [Google Scholar] [CrossRef]
- Semagn, K.; Babu, R.; Hearne, S.; Olsen, M. Single nucleotide polymorphism genotyping using Kompetitive Allele Specific PCR (KASP): Overview of the technology and its application in crop improvement. Mol. Breed. 2014, 33, 1–14. [Google Scholar] [CrossRef]
- Herrmann, M.G.; Durtschi, J.D.; Wittwer, C.T.; Voelkerding, K.V. Expanded Instrument Comparison of Amplicon DNA Melting Analysis for Mutation Scanning and Genotyping. Clin. Chem. 2007, 53, 1544–1548. [Google Scholar] [CrossRef]
- White, H.E.; Hall, V.J.; Cross, N.C.P. Methylation-Sensitive High-Resolution Melting-Curve Analysis of the SNRPN Gene as a Diagnostic Screen for Prader-Willi and Angelman Syndromes. Clin. Chem. 2007, 53, 1960–1962. [Google Scholar] [CrossRef]
- Distefano, G.; Caruso, M.; La Malfa, S.; Gentile, A.; Wu, S. High Resolution Melting Analysis Is a More Sensitive and Effective Alternative to Gel-Based Platforms in Analysis of SSR—An Example in Citrus. PLoS ONE 2012, 7, e44202. [Google Scholar] [CrossRef]
- Distefano, G.; La Malfa, S.; Gentile, A.; Wu, S.B. EST-SNP genotyping of citrus species using high-resolution melting curve analysis. Tree Genet. Genomes 2013, 9, 1271–1281. [Google Scholar] [CrossRef]
- Liew, M.; Pryor, R.; Palais, R.; Meadows, C.; Erali, M.; Lyon, E.; Wittwer, C. Genotyping of Single-Nucleotide Polymorphisms by High-Resolution Melting of Small Amplicons. Clin. Chem. 2004, 50, 1156–1164. [Google Scholar] [CrossRef]
- Distefano, G.; Lo Piero, A.R.; La Malfa, S.; Caruso, M.; Nicolosi, E.; Wu, S.; Gentile, A. Genotyping and Mutation Scanning by High Resolution Melting (HRM) Analysis of Citrus EST-SNPs and SSRs. Acta Hortic. 2015, 1065, 575–584. [Google Scholar] [CrossRef]
- Caruso, M.; Distefano, G.; Pietro Paolo, D.; La Malfa, S.; Russo, G.; Gentile, A.; Recupero, G.R. High resolution melting analysis for early identification of citrus hybrids: A reliable tool to overcome the limitations of morphological markers and assist rootstock breeding. Sci. Hortic. 2014, 180, 199–206. [Google Scholar] [CrossRef]
- Wu, S.B.; Wirthensohn, M.G.; Hunt, P.; Gibson, J.P.; Sedgley, M. High resolution melting analysis of almond SNPs derived from ESTs. Theor. Appl. Genet. 2008, 118, 1–14. [Google Scholar] [CrossRef]
- Shavrukov, Y.N. CAPS markers in plant biology. Russ. J. Genet. Appl. Res. 2016, 6, 279–287. [Google Scholar] [CrossRef]
- Jiang, D.; Ye, Q.; Wang, F.; Cao, L. The Mining of Citrus EST-SNP and Its Application in Cultivar Discrimination. Agric. Sci. China 2010, 9, 179–190. [Google Scholar] [CrossRef]
- Amar, M.H.; Biswas, M.K.; Zhang, Z.; Guo, W.-W. Exploitation of SSR, SRAP and CAPS-SNP markers for genetic diversity of Citrus germplasm collection. Sci. Hortic. 2011, 128, 220–227. [Google Scholar] [CrossRef]
- Nonaka, K.; Fujii, H.; Kita, M.; Shimada, T.; Endo, T.; Yoshioka, T.; Omura, M. Identification and Parentage Analysis of Citrus Cultivars Developed in Japan by CAPS Markers. Hortic. J. 2017, 86, 208–221. [Google Scholar] [CrossRef]
- Shimada, T.; Fujii, H.; Endo, T.; Ueda, T.; Sugiyama, A.; Nakano, M.; Kita, M.; Yoshioka, T.; Shimizu, T.; Nesumi, H.; et al. Construction of a citrus framework genetic map anchored by 708 gene-based markers. Tree Genet. Genomes 2014, 10, 1001–1013. [Google Scholar] [CrossRef]
- Endo, T.; Fujii, H.; Yoshioka, T.; Omura, M.; Shimada, T. TaqMan-MGB SNP genotyping assay to identify 48 citrus cultivars distributed in the Japanese market. Breed. Sci. 2020, 70, 363–372. [Google Scholar] [CrossRef]
- Starrantino, A. Il mandarino “Cami”, un ibrido a maturazione medio-tardiva. Frutticoltura 1992, 54, 37–40. [Google Scholar]
- Nadori, E. Nadorcott mandarin: A promising new variety. Proc. Proc. Int. Soc. Citric. 2004, 1, 356–359. [Google Scholar]
- Barry, G.H.; Caruso, M.; Gmitter, F.G., Jr. Commercial scion varieties. In The Genus Citrus; Talon, M., Caruso, M., Gmitter, F.G., Jr., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 83–104. ISBN 978-0-12-812163-4. [Google Scholar]
- Hodgson, R.W. Horticultural Varieties of Citrus. In The Citrus Industry; Reuther, W., Webber, H.J., Batchelor, L.D., Eds.; University of California Press: Ruverside, CA, USA, 1967; pp. 431–591. ISBN 978-0-12-812163-4. [Google Scholar]
- Wutscher, H.K.; Maxwell, M.P.; Dube, D. Performance of young ‘Fairchild’and ‘Bower’mandarin hybrids on six rootstocks. Proc. Am. Soc. Hort. Sci. Trop. Reg. 1976, 24, 157–165. [Google Scholar]
- Nauer, E.M.; Roistacher, C.N.; Gumpf, D.J. Varietal update on the clonal protection program. Citrograph 1986, 71, 115–117. [Google Scholar]
- Caruso, M.; Ferlito, F.; Licciardello, C.; Allegra, M.; Strano, M.C.; Di Silvestro, S.; Russo, M.P.; Pietro Paolo, D.; Caruso, P.; Las Casas, G.; et al. Pomological Diversity of the Italian Blood Orange Germplasm. Sci. Hortic. 2016, 213, 331–339. [Google Scholar] [CrossRef]
- Furr, J.R. New tangerines for the desert. Calif. Citrog. 1964, 49, 266. [Google Scholar]
- Hearn, C.J. The ’Fallglo’ citrus hybrid in Florida. Proc. Fla. State Hortic. Soc. 1988, 100, 119–121. [Google Scholar]
- Barry, G.; Gmitter, F.; Chen, C.; Roose, M.; Federici, C.; McCollum, G.T. Investigating the parentage of ‘Orri’and ‘Fortune’mandarin hybrids. Acta Hortic. 2015, 1065, 449–456. [Google Scholar] [CrossRef]
- Russo, F. Il tangelo “Mapo” un nuovo e promettente ibrido di agrume. Ann. Ist. Sperim. Agrum. 1972, 5, 107–115. [Google Scholar]
- Reece, P.C.; Hearn, C.J.; Gardner, F.E. Nova tangelo--an early ripening hybrid. Proc. Fla. State Hortic. Soc. 1964, 11, 109–110. [Google Scholar]
- Russo, G.; Licciardello, C.; Caruso, P.; Russo, M.P.; Paolo, D.P.; Recupero, G.R.; Rapisarda, P.; Ballistreri, G.; Fabroni, S.; Caruso, M. New CREA citrus hybrids. Citrus Res. Technol. 2017, 37, 98–101. [Google Scholar] [CrossRef]
- Russo, F.; Starrantino, A.; Reforgiato Recupero, G. New promising mandarin and mandarin hybrids. Proc. Proc. Int. Soc. Citric. 1977, 2, 597–601. [Google Scholar]
- Tribulato, E.; La Rosa, G. Primosole e Simeto: Due nuovi ibridi di mandarino. Italus Hortus 1993, 1, 21–25. [Google Scholar]
- Starrantino, A.; Perri, F.; Terranova, G. II clementine Rubino. Frutticoltura 1994, 11, 61–62. [Google Scholar]
- Yamasaki, A.; Kitajima, A.; Ohara, N.; Tanaka, M.; Hasegawa, K. Histological study of expression of seedlessness in Citrus kinokuni ‘Mukaku Kishu’and its progenies. J. Am. Soc. Hortic. Sci. 2007, 132, 869–875. [Google Scholar] [CrossRef]
- Hensz, R.A. Mutation breeding and the development of the ‘Star Ruby’grapefruit. Proc. Int. Soc. Citric. 1977, 2, 582–585. [Google Scholar]
- Hearn, C.J. Performance of ‘Sunburst’, a new citrus hybrid. Proc. Fla. State Hort. Soc. 1979, 92, 1–3. [Google Scholar]
- Camacho, C.; Coulouris, G.; Avagyan, V.; Ma, N.; Papadopoulos, J.; Bealer, K.; Madden, T.L. BLAST+: Architecture and applications. BMC Bioinform. 2009, 10, 421. [Google Scholar] [CrossRef]
- De Souza, M.C.; Stuchi, E.S.; de Goes, A. Evaluation of tangerine hybrid resistance to Alternaria alternata. Sci. Hortic. 2009, 123, 1–4. [Google Scholar] [CrossRef]
- Treangen, T.J.; Salzberg, S.L. Repetitive DNA and next-generation sequencing: Computational challenges and solutions. Nat. Rev. Genet. 2012, 13, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Botticella, E.; Sestili, F.; Hernandez-lopez, A.; Phillips, A.; Lafiandra, D. High resolution melting analysis for the detection of EMS induced mutations in wheat SbeIIa genes. BMC Plant Biol. 2011, 11, 156. [Google Scholar] [CrossRef] [PubMed]
- Lochlainn, S.Ó.; Amoah, S.; Graham, N.S.; Alamer, K.; Rios, J.J.; Kurup, S.; Stoute, A.; Hammond, J.P.; Østergaard, L.; King, G.J.; et al. High Resolution Melt (HRM) analysis is an efficient tool to genotype EMS mutants in complex crop genomes. Plant Methods 2011, 7, 43. [Google Scholar] [CrossRef]
- Hong, Y.; Pandey, M.K.; Liu, Y.; Chen, X.; Liu, H. Identification and Evaluation of Single-Nucleotide Polymorphisms in Allotetraploid Peanut (Arachis hypogaea L.) Based on Amplicon Sequencing Combined with High Resolution Melting (HRM) Analysis. Front. Plant Sci. 2015, 6, 1068. [Google Scholar] [CrossRef]
- Elena, K. Alternaria brown spot of Minneola in Greece; evaluation of citrus species susceptibility. Eur. J. Plant Pathol. 2006, 115, 259–262. [Google Scholar] [CrossRef]
- Gardner, J.M.; Kono, Y.; Chandler, J.L. Bioassay and host-selectivity of Alternaria citri toxins affecting rough lemon and mandarins. Physiol. Mol. Plant Pathol. 1986, 29, 293–304. [Google Scholar] [CrossRef]
- Navarro, L.; Aleza, P.; Cuenca, J.; Juárez, J.; Pina, J.A.; Ortega, C.; Navarro, A.; Ortega, V. The mandarin triploid breeding program in Spain. Acta Hortic. 2015, 1065, 389–396. [Google Scholar] [CrossRef]
- Fishman, L.; McIntosh, M. Standard deviations: The biological bases of transmission ratio distortion. Annu. Rev. Genet. 2019, 53, 347–372. [Google Scholar] [CrossRef]
- Garavello, M.; Cuenca, J.; Dreissig, S.; Fuchs, J.; Navarro, L.; Houben, A.; Aleza, P. Analysis of Crossover Events and Allele Segregation Distortion in Interspecific Citrus Hybrids by Single Pollen Genotyping. Front. Plant Sci. 2020, 11, 615. [Google Scholar] [CrossRef]
- Liang, M.; Cao, Z.; Zhu, A.; Liu, Y.; Tao, M.; Yang, H.; Xu, Q.; Wang, S.; Liu, J.; Li, Y.; et al. Evolution of self-compatibility by a mutant Sm-RNase in citrus. Nat. Plants 2020, 6, 131–142. [Google Scholar] [CrossRef]

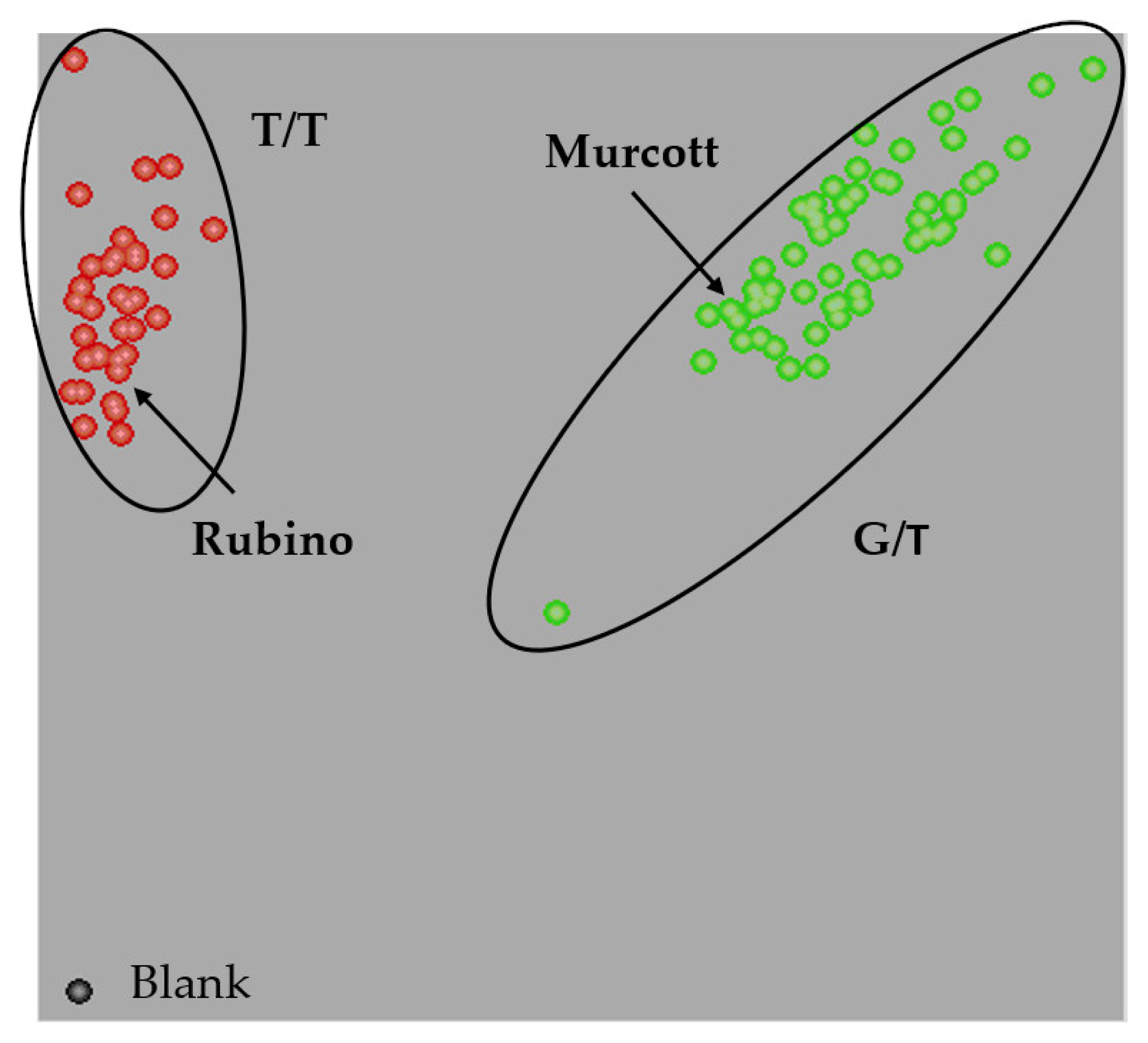

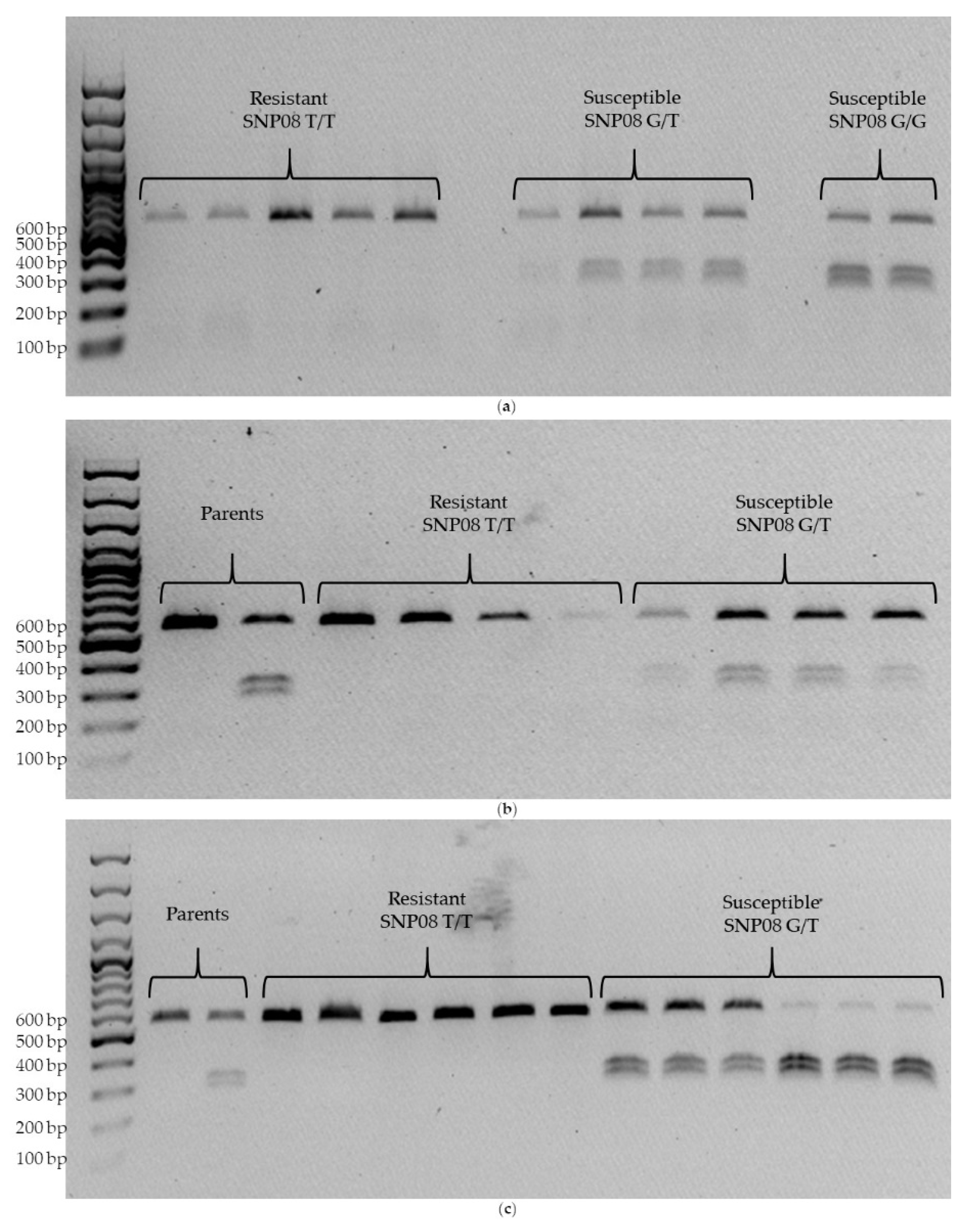

| Accession | Description | References |
|---|---|---|
| 50-15A-6 | ‘Clementine’ mandarin × ‘Avana’ mandarin/Unreleased parent of the CREA breeding program | [36] |
| Afourer | Possibly ‘Murcott’ tangor × ‘Mandalina’ mandarin | [37,38] |
| Avana (Willowleaf) | Old mandarin selection | [39] |
| Bower | ‘Clementine’ mandarin × ‘Orlando’ tangelo | [40] |
| Cami | ‘50-15A-6′ mandarin × ‘Mapo’ tangelo | [36] |
| Carvalhais | Mandarin of unknown parentage | [39] |
| Clemapo | ‘Clementine’ mandarin × ‘Mapo’ tangelo/early-maturing hybrid released in the 1990′s by a private breeding program | - |
| Daisy | ‘Fortune’ mandarin × ‘Fremont’ mandarin | [41] |
| Dancy | Seedling of Moragne tangierine | [39] |
| Doppio Sanguigno | Sweet orange clonal selection | [42] |
| Ellendale | Tangor of unknown parentage | [39] |
| Emperor | ‘Ponkan’ mandarin selection | [38,39] |
| Encore | ‘King’ mandarin × ‘Willowleaf’ mandarin | [39] |
| Fairchild | ‘Clementine’ mandarin × ‘Orlando’ tangelo | [43] |
| Fallglo | ‘Bower’ mandarin ×’ Temple’ tangor | [44] |
| Foma107 | ‘Fortune’ mandarin × ‘Mapo’ tangelo/Unreleased parent of the CREA breeding program | - |
| Fortune | ‘Clementine’ mandarin × ‘Orlando’ tangelo | [45] |
| Fremont | ‘Clementine’ mandarin × ‘Ponkan’ mandarin | [39] |
| ISA | Old clementine clone of Italian origin | - |
| Kara | ‘King’ mandarin × ‘Owari’ satsuma | [39] |
| King | Natural tangor of Asian origin | [39] |
| Kinnow | ‘King’ mandarin × ‘Willowleaf’ mandarin | [39] |
| Malvasio | Mandarin of unknown parentage | [39] |
| Mapo | ‘Avana’ mandarin × ‘Duncan’ grapefruit | [46] |
| Michal | ‘Clementine’ mandarin × ‘Dancy’ mandarin | [38] |
| Minneola | ‘Duncan’ grapefruit × ‘Dancy’ mandarin | [39] |
| Murcott | Tangor of unknown parentage | [39] |
| Nova | ‘Clementine’ mandarin × ‘Orlando’ tangelo | [47] |
| Okitsu | Satsuma clonal selection | [39] |
| Ortanique | Tangor of unknown parentage | [39] |
| OTA9 | ‘Oroval’ clementine × ‘Moro’ sweet orange | [48] |
| Page | ‘Minneola’ tangelo × ‘Clementine’ mandarin | [39] |
| Palazzelli | ‘Clementine’ mandarin × ‘King’ mandarin | [49] |
| Ponkan | Old mandarin selection | [39] |
| Primosole | ‘Miho’ satsuma × ‘Carvalhais’ mandarin | [50] |
| Rubino | Late clementine clone | [51] |
| Seedless Kishu | Seedless mutation of ‘Kishu’ mandarin | [52] |
| Simeto | ‘Miho’ satsuma × ‘Avana’ mandarin | [50] |
| Star Ruby | Grapefruit clonal selection | [53] |
| Sunburst | ‘Robinson’ mandarin × ‘Osceola’ mandarin | [54] |
| Wilking | ‘King’ tangor × ‘Willowleaf ‘ mandarin | [39] |
| Primer Name | Primer Sequence (5′-3′) | Ta (°C) | Product Size (bp) | Position in Chromosome 3 of the Clementine Genome |
|---|---|---|---|---|
| SNP08_nested PCR | F: AACTCACAAACATGTCTTCAACAA | 57 | 572 | 25861812…25861844 |
| R: ATTGTCAATTGGTGGGAAGC | 25862364…25862383 | |||
| SNP08_HRM | F: AGCGAATAAATTTGATGCTGAGC | 60 | 110 | 25862022…25862044 |
| R: TCTTGTAGGACTAAAATTTCACTTGGA | 25862105…25862131 |
| Genotypes | SNP08 | SNP08+18bp | Genotype Code |
|---|---|---|---|
| Clemapo | TT | CC | 1A |
| Fallglo | TT | CC | 1A |
| Foma107 | TT | CC | 1A |
| ISA | TT | CC | 1A |
| Kara | TT | CC | 1A |
| Kinnow | TT | CC | 1A |
| Mapo | TT | CC | 1A |
| Okitsu | TT | CC | 1A |
| OTA9 | TT | CC | 1A |
| Rubino | TT | CC | 1A |
| Simeto | TT | CC | 1A |
| Wilking | TT | CC | 1A |
| 50-15A-6 | TT | CT | 1B |
| Avana | TT | CT | 1B |
| Cami | TT | CT | 1B |
| Carvalhais | TT | CT | 1B |
| Encore | TT | CT | 1B |
| Fremont | TT | CT | 1B |
| King | TT | CT | 1B |
| Palazzelli | TT | CT | 1B |
| Afourer | TT | TT | 1C |
| Bower | GT | CC | 2A |
| Daisy | GT | CC | 2A |
| Doppio Sanguigno | GT | CC | 2A |
| Fairchild | GT | CC | 2A |
| Fortune | GT | CC | 2A |
| Michal | GT | CC | 2A |
| Nova | GT | CC | 2A |
| Page | GT | CC | 2A |
| Primosole | GT | CC | 2A |
| Seedless Kishu | GT | CC | 2A |
| Star Ruby | GT | CC | 2A |
| Sunburst | GT | CC | 2A |
| Ellendale | GT | CT | 2B |
| Emperor | GT | CT | 2B |
| Murcott | GT | CT | 2B |
| Ortanique | GT | CT | 2B |
| Ponkan | GT | CT | 2B |
| Dancy | GG | CC | 3A |
| Malvasio | GG | CC | 3A |
| Minneola | GG | CC | 3A |
| Parent Combinations | Hybrids Analysed | Resistants | % | Susceptibles | % | Chi-Square | p Value |
|---|---|---|---|---|---|---|---|
| ISA × Seedless Kishu | 351 | 186 | 53.0 | 165 | 47.0 | 1.256 | 0.262 |
| Foma107 × Seedless Kishu | 162 | 74 | 45.7 | 88 | 54.3 | 1.210 | 0.271 |
| Cami × Seedless Kishu | 84 | 39 | 46.4 | 45 | 53.6 | 0.429 | 0.513 |
| Rubino × Murcott | 148 | 54 | 36.5 | 94 | 63.5 | 10.811 | 0.001 |
| Fortune × Ota9 | 117 | 60 | 51.3 | 57 | 48.7 | 0.077 | 0.782 |
| Total | 862 | 413 | 47.9 | 449 | 52.1 | 1.503 | 0.220 |
| Parent Combinations | Hybrids Analysed | Resistants | % | Susceptibles | % | Chi-Square | p Value |
|---|---|---|---|---|---|---|---|
| ISA × Seedless Kishu | 14 | 10 | 71.4 | 4 | 28.6 | 2.571 | 0.109 |
| Foma107 × Seedless Kishu | 20 | 10 | 50.0 | 10 | 50.0 | 0.000 | 1.000 |
| Cami × Seedless Kishu | 20 | 12 | 60.0 | 8 | 40.0 | 0.800 | 0.371 |
| Rubino × Murcott | 26 | 8 | 30.8 | 18 | 69.2 | 3.846 | 0.050 |
| Fortune × Ota9 | 21 | 13 | 61.9 | 8 | 38.1 | 1.190 | 0.275 |
| Total | 101 | 53 | 52.5 | 48 | 47.5 | 0.248 | 0.619 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arlotta, C.; Ciacciulli, A.; Strano, M.C.; Cafaro, V.; Salonia, F.; Caruso, P.; Licciardello, C.; Russo, G.; Smith, M.W.; Cuenca, J.; et al. Disease Resistant Citrus Breeding Using Newly Developed High Resolution Melting and CAPS Protocols for Alternaria Brown Spot Marker Assisted Selection. Agronomy 2020, 10, 1368. https://doi.org/10.3390/agronomy10091368
Arlotta C, Ciacciulli A, Strano MC, Cafaro V, Salonia F, Caruso P, Licciardello C, Russo G, Smith MW, Cuenca J, et al. Disease Resistant Citrus Breeding Using Newly Developed High Resolution Melting and CAPS Protocols for Alternaria Brown Spot Marker Assisted Selection. Agronomy. 2020; 10(9):1368. https://doi.org/10.3390/agronomy10091368
Chicago/Turabian StyleArlotta, Carmen, Angelo Ciacciulli, Maria Concetta Strano, Valeria Cafaro, Fabrizio Salonia, Paola Caruso, Concetta Licciardello, Giuseppe Russo, Malcolm Wesley Smith, Jose Cuenca, and et al. 2020. "Disease Resistant Citrus Breeding Using Newly Developed High Resolution Melting and CAPS Protocols for Alternaria Brown Spot Marker Assisted Selection" Agronomy 10, no. 9: 1368. https://doi.org/10.3390/agronomy10091368
APA StyleArlotta, C., Ciacciulli, A., Strano, M. C., Cafaro, V., Salonia, F., Caruso, P., Licciardello, C., Russo, G., Smith, M. W., Cuenca, J., Aleza, P., & Caruso, M. (2020). Disease Resistant Citrus Breeding Using Newly Developed High Resolution Melting and CAPS Protocols for Alternaria Brown Spot Marker Assisted Selection. Agronomy, 10(9), 1368. https://doi.org/10.3390/agronomy10091368










