Introducing Perennial Grain in Grain Crops Rotation: The Role of Rooting Pattern in Soil Quality Management
Abstract
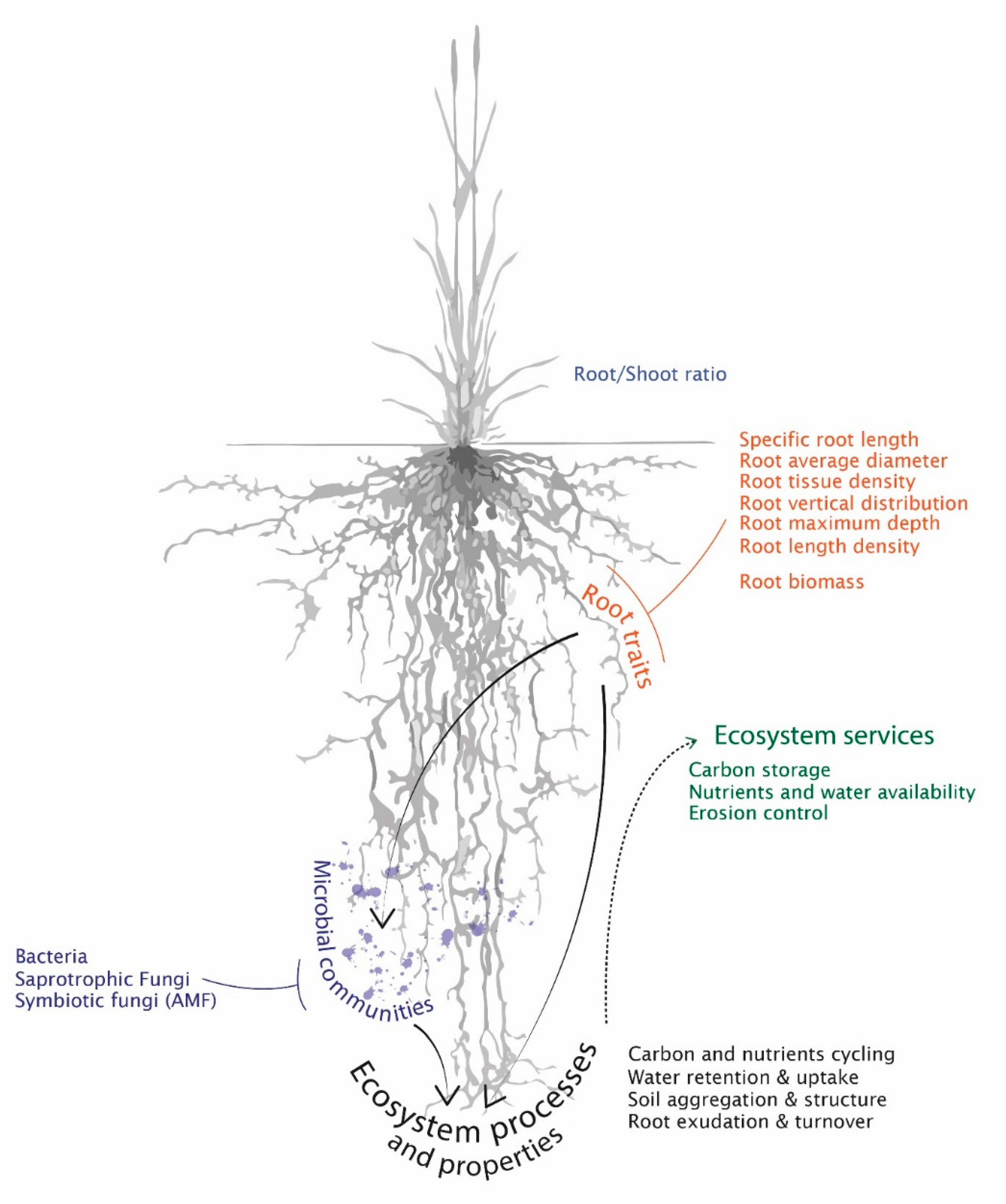
1. Introduction
2. Materials and Methods
2.1. Site, Soil and Sampling Description
2.2. Root Traits
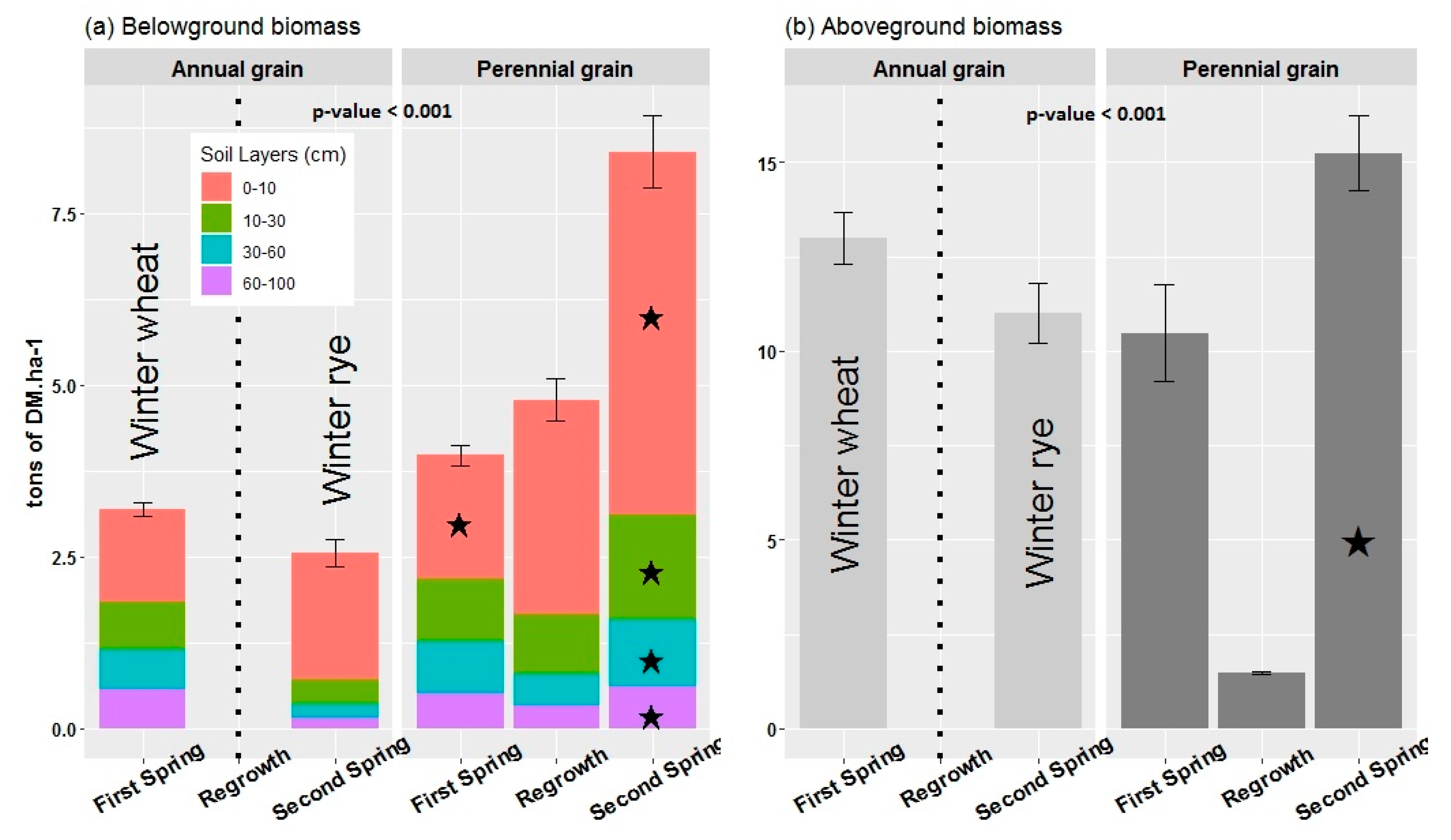
2.2.1. Root Biomass
2.2.2. Specific Root Length, Average Root Diameter, Root Tissue Density, and Root Length Density
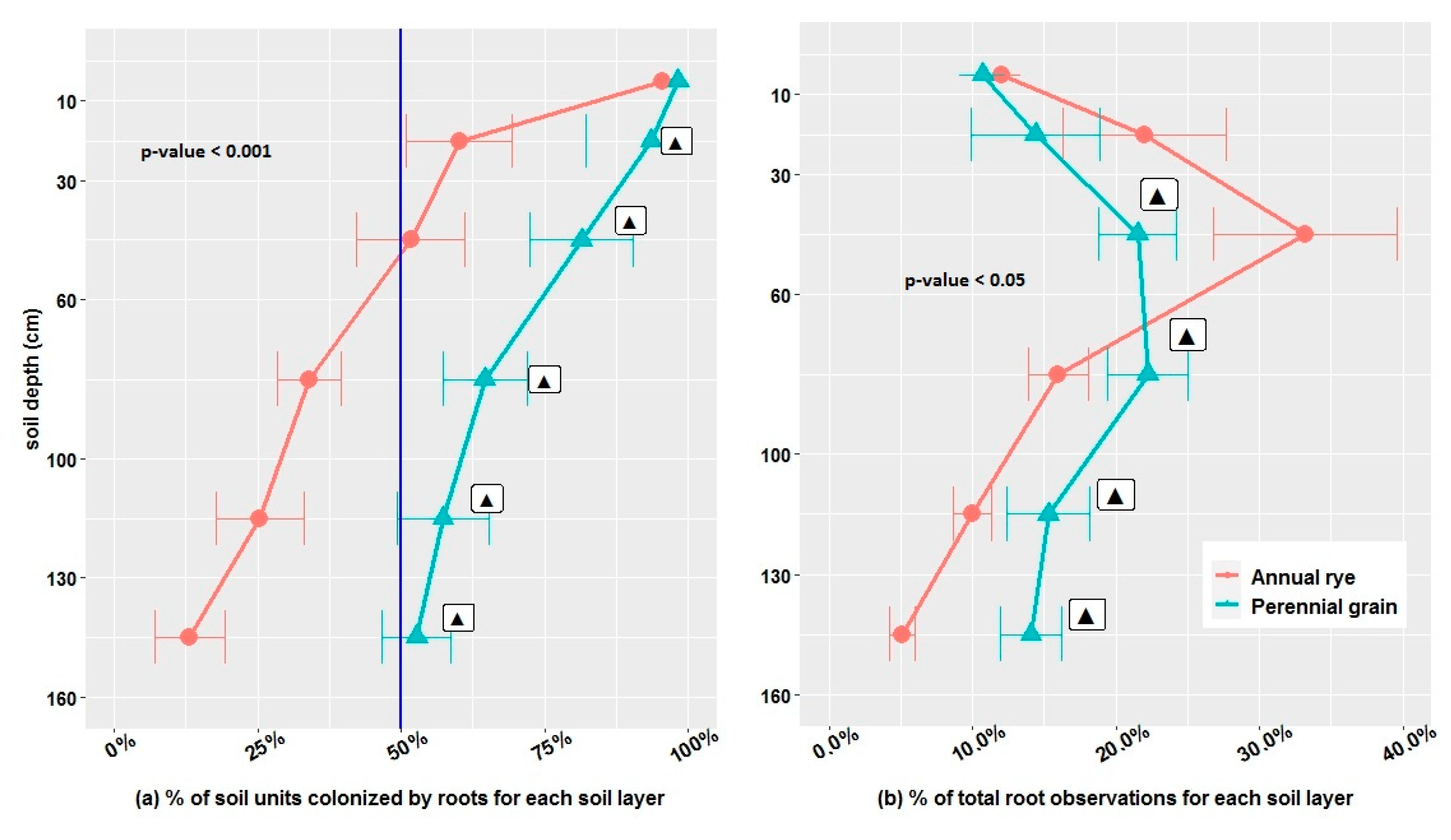
2.2.3. Maximum Rooting Depth, % of Soil Colonized by Roots, % of Total Root Observations
2.3. Aboveground Biomass
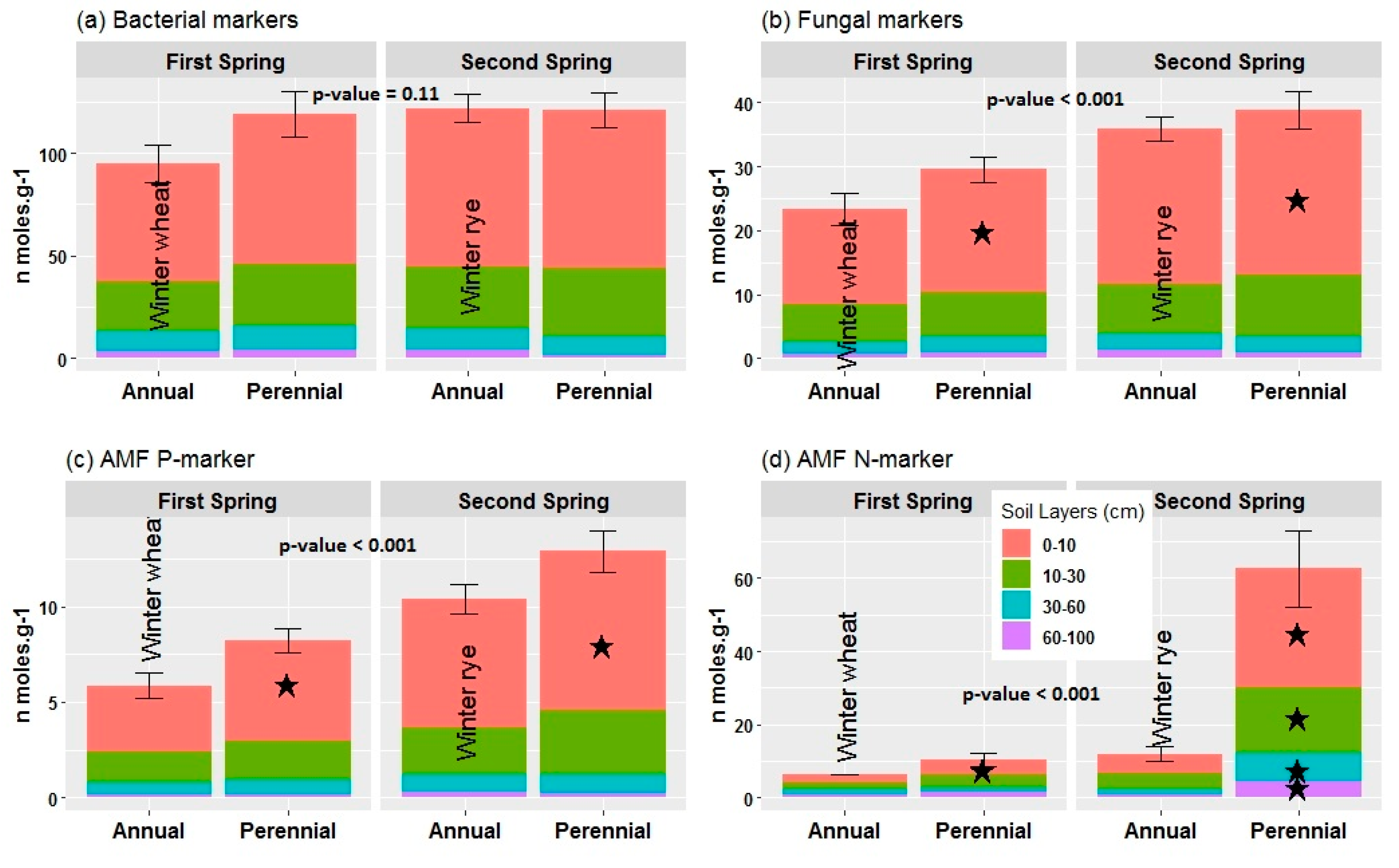
2.4. Soil Microbial Analysis
2.5. Statistical Analysis
3. Results
3.1. Above and Belowground Crops Biomass
3.2. Root Colonization of the Soil Profile
3.3. Specific Root Length, Diameter, Tissue Density, and Length Density
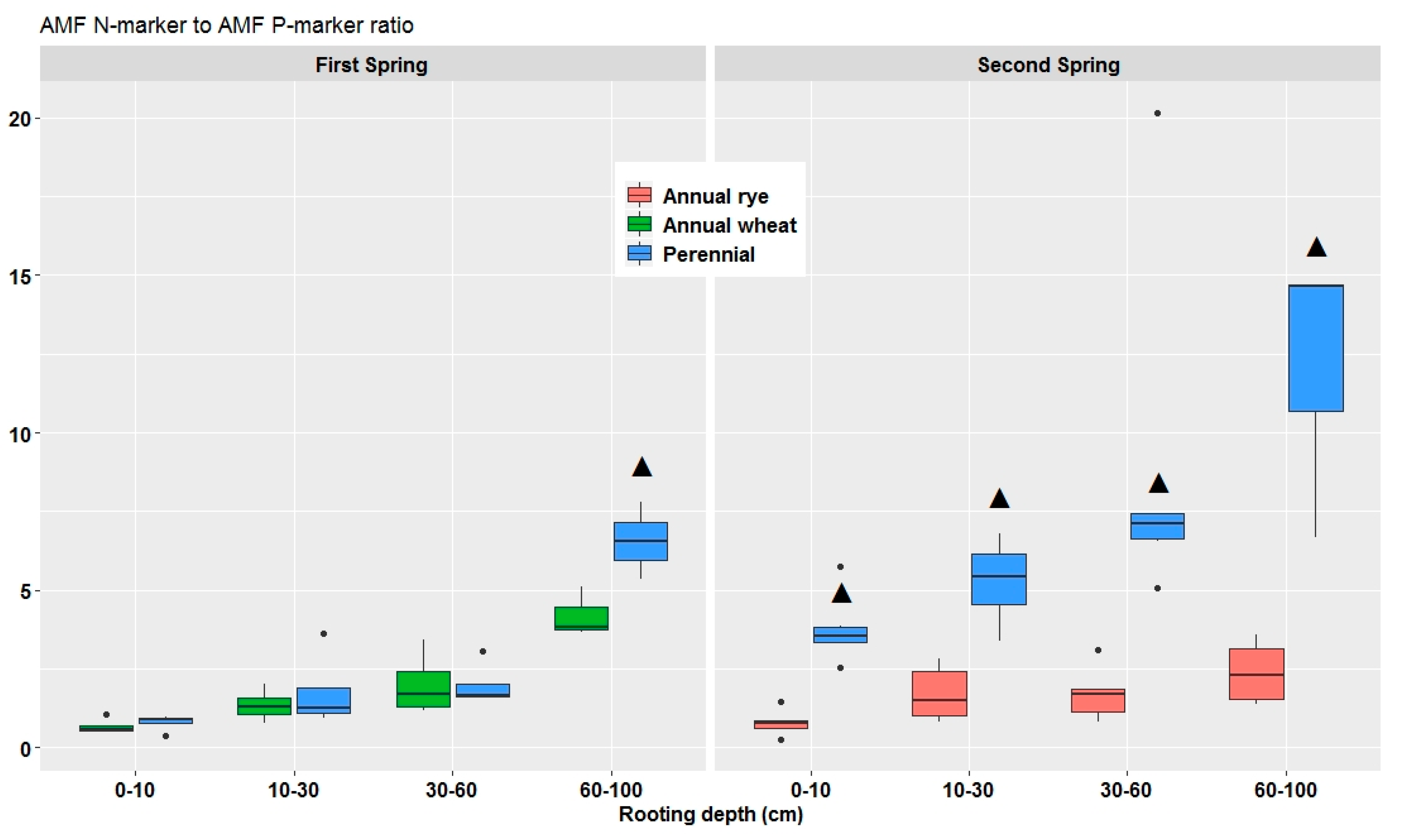
3.4. Soil Microbial PLFA and NLFA Markers
4. Discussion
4.1. Rapid Rooting and Microbial Pattern Changes
4.2. A Range of Influences on Soil Functioning and Services Supply
4.3. Are Two Years Enough to Consider Soil Gains and Services Supply?
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- DeHaan, L.; Christians, M.; Crain, J.; Poland, J. Development and Evolution of an Intermediate Wheatgrass Domestication Program. Sustainability 2018, 10, 1499. [Google Scholar] [CrossRef]
- Duchene, O.; Celette, F.; Ryan, M.R.; DeHaan, L.R.; Crews, T.E.; David, C. Integrating multipurpose perennial grains crops in Western European farming systems. Agric. Ecosyst. Environ. 2019, 284, 106591. [Google Scholar] [CrossRef]
- Ryan, M.R.; Crews, T.E.; Culman, S.W.; DeHaan, L.R.; Hayes, R.C.; Jungers, J.M.; Bakker, M.G. Managing for Multifunctionality in Perennial Grain Crops. BioScience 2018, 68, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Favre, J.R.; Castiblanco, T.M.; Combs, D.K.; Wattiaux, M.; Picasso, V.D. Forage nutritive value and predicted fiber digestibility of Kernza intermediate wheatgrass in monoculture and in mixture with red clover during the first production year. Anim. Feed. Sci. Technol. 2019, 258, 114298. [Google Scholar] [CrossRef]
- Jungers, J.M.; DeHaan, L.R.; Betts, K.J.; Sheaffer, C.C.; Wyse, D.L. Intermediate Wheatgrass Grain and Forage Yield Responses to Nitrogen Fertilization. Agron. J. 2017, 109, 462–472. [Google Scholar] [CrossRef]
- Crews, T.E. Closing the Gap between Grasslands and Grain Agriculture. Kans. J. Law Public Policy 2016, 26, 274. [Google Scholar]
- Crews, T.E.; Carton, W.; Olsson, L. Is the future of agriculture perennial? Imperatives and opportunities to reinvent agriculture by shifting from annual monocultures to perennial polycultures. Glob. Sustain. 2018, 1. [Google Scholar] [CrossRef]
- DeHaan, L.R.; Ismail, B.P. Perennial Cereals Provide Ecosystem Benefits. Cereal Foods World 2017, 62, 278–281. [Google Scholar] [CrossRef]
- Culman, S.W.; Snapp, S.S.; Ollenburger, M.; Basso, B.; DeHaan, L. Soil and Water Quality Rapidly Responds to the Perennial Grain Kernza Wheatgrass. Agron. J. 2013, 105, 735–744. [Google Scholar] [CrossRef]
- Thorup-Kristensen, K.; Halberg, N.; Nicolaisen, M.; Olesen, J.E.; Crews, T.E.; Hinsinger, P.; Kirkegaard, J.; Pierret, A.; Dresbøll, D.B. Digging Deeper for Agricultural Resources, the Value of Deep Rooting. Trends Plant Sci. 2020, 25, 406–417. [Google Scholar] [CrossRef]
- Vico, G.; Brunsell, N.A. Tradeoffs between water requirements and yield stability in annual vs. perennial crops. Adv. Water Resour. 2018, 112, 189–202. [Google Scholar] [CrossRef]
- Sainju, U.M.; Allen, B.L.; Lenssen, A.W.; Ghimire, R. Root biomass, root/shoot ratio, and soil water content under perennial grasses with different nitrogen rates. Field Crop. Res. 2017, 210, 183–191. [Google Scholar] [CrossRef]
- Dupont, S.T.; Beniston, J.; Glover, J.D.; Hodson, A.; Culman, S.W.; Lal, R.; Ferris, H. Root traits and soil properties in harvested perennial grassland, annual wheat, and never-tilled annual wheat. Plant Soil 2014, 381, 405–420. [Google Scholar] [CrossRef]
- Henneron, L.; Cros, C.; Picon-Cochard, C.; Rahimain, V.; Fontaine, S. Plant economic strategies of grassland species control soil carbon dynamics through rhizodeposition. J. Ecol. 2020, 108, 528–545. [Google Scholar] [CrossRef]
- Roumet, C.; Urcelay, C.; Díaz, S. Suites of root traits differ between annual and perennial species growing in the field. New Phytol. 2006, 170, 357–368. [Google Scholar] [CrossRef] [PubMed]
- Dignac, M.-F.; Derrien, D.; Barre, P.; Barot, S.; Cécillon, L.; Chenu, C.; Chevallier, T.; Freschet, G.T.; Garnier, P.; Guenet, B.; et al. Increasing soil carbon storage: Mechanisms, effects of agricultural practices and proxies. A review. Agron. Sustain. Dev. 2017, 37, 351. [Google Scholar] [CrossRef]
- Poirier, V.; Roumet, C.; Munson, A.D. The root of the matter: Linking root traits and soil organic matter stabilization processes. Soil Boil. Biochem. 2018, 120, 246–259. [Google Scholar] [CrossRef]
- De Vries, F.T.; Bardgett, R.D. Plant community controls on short-term ecosystem nitrogen retention. New Phytol. 2016, 210, 861–874. [Google Scholar] [CrossRef]
- Culman, S.W.; Dupont, S.; Glover, J.; Buckley, D.H.; Fick, G.; Ferris, H.; Crews, T. Long-term impacts of high-input annual cropping and unfertilized perennial grass production on soil properties and belowground food webs in Kansas, USA. Agric. Ecosyst. Environ. 2010, 137, 13–24. [Google Scholar] [CrossRef]
- Stokes, A.; Atger, C.; Bengough, A.G.; Fourcaud, T.; Sidle, R.C.; Bengough, A.G. Desirable plant root traits for protecting natural and engineered slopes against landslides. Plant Soil 2009, 324, 1–30. [Google Scholar] [CrossRef]
- Chantigny, M.H.; Angers, D.A.; Prévost, D.; Vézina, L.-P.; Chalifour, F.-P. Soil Aggregation and Fungal and Bacterial Biomass under Annual and Perennial Cropping Systems. Soil Sci. Soc. Am. J. 1997, 61, 262–267. [Google Scholar] [CrossRef]
- McKenna, T.P.; Crews, T.E.; Kemp, L.; Sikes, B.A. Community structure of soil fungi in a novel perennial crop monoculture, annual agriculture, and native prairie reconstruction. PLoS ONE 2020, 15, e0228202. [Google Scholar] [CrossRef] [PubMed]
- Sprunger, C.D.; Culman, S.W.; Robertson, G.P.; Snapp, S.S. How Does Nitrogen and Perenniality Influence Belowground Biomass and Nitrogen Use Efficiency in Small Grain Cereals? Crop. Sci. 2018, 58, 2110–2120. [Google Scholar] [CrossRef]
- Sainju, U.M.; Allen, B.L.; Lenssen, A.W.; Mikha, M. Root and soil total carbon and nitrogen under bioenergy perennial grasses with various nitrogen rates. Biomass Bioenergy 2017, 107, 326–334. [Google Scholar] [CrossRef]
- Sprunger, C.D.; Culman, S.W.; Peralta, A.L.; Dupont, S.T.; Lennon, J.T.; Snapp, S.S. Perennial grain crop roots and nitrogen management shape soil food webs and soil carbon dynamics. Soil Boil. Biochem. 2019, 137, 107573. [Google Scholar] [CrossRef]
- Martin, G.; Durand, J.-L.; Duru, M.; Gastal, F.; Julier, B.; Litrico, I.; Louarn, G.; Médiène, S.; Moreau, D.; Valentin-Morison, M.; et al. Role of ley pastures in tomorrow’s cropping systems. A review. Agron. Sustain. Dev. 2020, 40. [Google Scholar] [CrossRef]
- Tautges, N.E.; Jungers, J.M.; DeHaan, L.; Wyse, N.L.; Sheaffer, C.C. Maintaining grain yields of the perennial cereal intermediate wheatgrass in monoculture v. bi-culture with alfalfa in the Upper Midwestern USA. J. Agric. Sci. 2018, 156, 758–773. [Google Scholar] [CrossRef]
- Bardgett, R.D.; Mommer, L.; De Vries, F.T. Going underground: Root traits as drivers of ecosystem processes. Trends Ecol. Evol. 2014, 29, 692–699. [Google Scholar] [CrossRef]
- Craine, J.M.; Wedin, D.A.; Chapin, F.S.; Reich, P.B. The dependence of root system properties on root system biomass of 10 North American grassland species. Plant Soil 2003, 250, 39–47. [Google Scholar] [CrossRef]
- Craine, J.M.; Tilman, D.; Wedin, D.; Reich, P.B.; Tjoelker, M.G.; Knops, J. Functional traits, productivity and effects on nitrogen cycling of 33 grassland species. Funct. Ecol. 2002, 16, 563–574. [Google Scholar] [CrossRef]
- Craine, J.M.; Froehle, J.; Tilman, D.G.; Wedin, D.A.; Chapin, I. The relationships among root and leaf traits of 76 grassland species and relative abundance along fertility and disturbance gradients. Oikos 2001, 93, 274–285. [Google Scholar] [CrossRef]
- Freschet, G.T.; Roumet, C. Sampling roots to capture plant and soil functions. Funct. Ecol. 2017, 31, 1506–1518. [Google Scholar] [CrossRef]
- Bardgett, R.D.; McAlister, E. The measurement of soil fungal:bacterial biomass ratios as an indicator of ecosystem self-regulation in temperate meadow grasslands. Boil. Fertil. Soils 1999, 29, 282–290. [Google Scholar] [CrossRef]
- Degens, B. Decreases in organic C reserves in soils can reduce the catabolic diversity of soil microbial communities. Soil Boil. Biochem. 2000, 32, 189–196. [Google Scholar] [CrossRef]
- Fontaine, S.; Mariotti, A.; Abbadie, L. The priming effect of organic matter: A question of microbial competition? Soil Boil. Biochem. 2003, 35, 837–843. [Google Scholar] [CrossRef]
- Grigulis, K.; Lavorel, S.; Krainer, U.; Legay, N.; Baxendale, C.; Dumont, M.; Kastl, E.; Arnoldi, C.; Bardgett, R.D.; Poly, F.; et al. Relative contributions of plant traits and soil microbial properties to mountain grassland ecosystem services. J. Ecol. 2012, 101, 47–57. [Google Scholar] [CrossRef]
- Van Der Heijden, M.G.A.; Bardgett, R.D.; Van Straalen, N.M. The unseen majority: Soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol. Lett. 2008, 11, 296–310. [Google Scholar] [CrossRef]
- Averill, C.; Turner, B.L.; Finzi, A.C. Mycorrhiza-mediated competition between plants and decomposers drives soil carbon storage. Nat. 2014, 505, 543–545. [Google Scholar] [CrossRef]
- Gianinazzi, S.; Gollotte, A.; Binet, M.-N.; Van Tuinen, D.; Redecker, D.; Wipf, D. Agroecology: The key role of arbuscular mycorrhizas in ecosystem services. Mycorrhiza 2010, 20, 519–530. [Google Scholar] [CrossRef]
- Phillips, R.P.; Brzostek, E.R.; Midgley, M.G. The mycorrhizal-associated nutrient economy: A new framework for predicting carbon-nutrient couplings in temperate forests. New Phytol. 2013, 199, 41–51. [Google Scholar] [CrossRef]
- FAO. World Reference Base for Soil Resources 2014: International Soil Classification System for Naming Soils and Creating Legends for Soil Maps; FAO: Rome, Italy, 2014. [Google Scholar]
- Frostegård, A.; Bath, E. The use of phospholipid fatty acid analysis to estimate bacterial and fungal biomass in soil. Biol. Fertil. Soils 1996, 22, 59–65. [Google Scholar] [CrossRef]
- Frostegard, A.; Tunlid, A.; Bååth, E. Use and misuse of PLFA measurements in soils. Soil Boil. Biochem. 2011, 43, 1621–1625. [Google Scholar] [CrossRef]
- Zelles, L. Fatty acid patterns of phospholipids and lipopolysaccharides in the characterisation of microbial communities in soil: A review. Boil. Fertil. Soils 1999, 29, 111–129. [Google Scholar] [CrossRef]
- Olsson, P.A.; Bååth, E.; Jakobsen, I.; Söderström, B. The use of phospholipid and neutral lipid fatty acids to estimate biomass of arbuscular mycorrhizal fungi in soil. Mycol. Res. 1995, 99, 623–629. [Google Scholar] [CrossRef]
- Olsson, P.A.; Thingstrup, I.; Jakobsen, I. Estimation of the biomass of arbuscular mycorrhizal fungi in a linseed field. Soil Biol. Biochem. 1999, 9, 1879–1887. [Google Scholar] [CrossRef]
- Sharma, M.P.; Buyer, J.S. Comparison of biochemical and microscopic methods for quantification of arbuscular mycorrhizal fungi in soil and roots. Appl. Soil Ecol. 2015, 95, 86–89. [Google Scholar] [CrossRef]
- Vestberg, M.; Palojärvi, A.; Pitkänen, T.; Kaipainen, S.; Puolakka, E.; Keskitalo, M. Neutral lipid fatty acid analysis is a sensitive marker for quantitative estimation of arbuscular mycorrhizal fungi in agricultural soil with crops of different mycotrophy. Agric. Food Sci. 2012, 21, 12–27. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing (Version 3.1.2). 2019. Available online: https://www.r-project.org/index.html (accessed on 21 July 2020).
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82. [Google Scholar] [CrossRef]
- Bergmann, J.; Weigelt, A.; Van Der Plas, F.; Laughlin, D.C.; Kuyper, T.W.; Guerrero-Ramírez, N.R.; Valverde-Barrantes, O.J.; Bruelheide, H.; Freschet, G.T.; Iversen, C.M.; et al. The fungal collaboration gradient dominates the root economics space in plants. Sci. Adv. 2020, 6, eaba3756. [Google Scholar] [CrossRef]
- Roumet, C.; Birouste, M.; Picon-Cochard, C.; Ghestem, M.; Osman, N.; Vrignon-Brenas, S.; Cao, K.-F.; Stokes, A. Root structure-function relationships in 74 species: Evidence of a root economics spectrum related to carbon economy. New Phytol. 2016, 210, 815–826. [Google Scholar] [CrossRef]
- Garnier, E.; Navas, M.-L. Diversité Fonctionnelle des Plantes: Traits des Organismes, Structure des Communautés, Propriétés des Ecosystèmes: Cours; De Boeck: Bruxelles, Belgium, 2013. [Google Scholar]
- Freschet, G.T.; Kichenin, E.; Wardle, D.A. Explaining within-community variation in plant biomass allocation: A balance between organ biomass and morphology above vs below ground? J. Veg. Sci. 2015, 26, 431–440. [Google Scholar] [CrossRef]
- Comas, L.H.; Mueller, K.E.; Taylor, L.; Midford, P.E.; Callahan, H.S.; Beerling, D.J. Evolutionary Patterns and Biogeochemical Significance of Angiosperm Root Traits. Int. J. Plant Sci. 2012, 173, 584–595. [Google Scholar] [CrossRef]
- Li, Y.; Bezemer, T.M.; Yang, J.; Lü, X.; Li, X.; Wen-Ju, L.; Han, X.; Li, Q. Changes in litter quality induced by N deposition alter soil microbial communities. Soil Boil. Biochem. 2019, 130, 33–42. [Google Scholar] [CrossRef]
- Neely, C.L.; Beare, M.; Hargrove, W.L.; Coleman, D.C. Relationships between fungal and bacterial substrate-induced respiration, biomass and plant residue decomposition. Soil Boil. Biochem. 1991, 23, 947–954. [Google Scholar] [CrossRef]
- Sauvadet, M.; Chauvat, M.; Brunet, N.; Bertrand, I. Can changes in litter quality drive soil fauna structure and functions? Soil Boil. Biochem. 2017, 107, 94–103. [Google Scholar] [CrossRef]
- Grayston, S.; Griffith, G.; Mawdsley, J.; Campbell, C.D.; Bardgett, R. Accounting for variability in soil microbial communities of temperate upland grassland ecosystems. Soil Boil. Biochem. 2001, 33, 533–551. [Google Scholar] [CrossRef]
- Davison, J.; Öpik, M.; Daniell, T.J.; Moora, M.; Zobel, M. Arbuscular mycorrhizal fungal communities in plant roots are not random assemblages. FEMS Microbiol. Ecol. 2011, 78, 103–115. [Google Scholar] [CrossRef]
- Boerner, R.E.J. Plant life span and response to inoculation with vesicular-arbuscular mycorrhizal fungi. Mycorrhiza 1992, 1, 153–161. [Google Scholar] [CrossRef]
- Liu, W. Correlation between specific fine root length and mycorrhizal colonization of maize in different soil types. Front. Agric. China 2009, 3, 13–15. [Google Scholar] [CrossRef]
- Montgomery, D.R. Soil erosion and agricultural sustainability. PANS 2007, 104, 13268–13272. [Google Scholar] [CrossRef]
- Gyssels, G.; Poesen, J.; Bochet, E.; Li, Y. Impact of plant roots on the resistance of soils to erosion by water: A review. Prog. Phys. Geogr. Earth Environ. 2005, 29, 189–217. [Google Scholar] [CrossRef]
- Jastrow, J.; Miller, R. Soil Aggregate Stabilization and Carbon Sequestration: Feedbacks through Organomineral Associations. In Soil Processes and the Carbon Cycle; CRC Press: Boca Raton, FL, USA, 2018; pp. 207–223. [Google Scholar]
- Sanaullah, M.; Chabbi, A.; Leifeld, J.; Bardoux, G.; Billou, D.; Rumpel, C. Decomposition and stabilization of root litter in top- and subsoil horizons: What is the difference? Plant Soil 2010, 338, 127–141. [Google Scholar] [CrossRef]
- Wright, A.J.; Schnitzer, S.A.; A Dickie, I.; Gunderson, A.R.; Pinter, G.A.; Mangan, S.A.; Reich, P.B. Complex facilitation and competition in a temperate grassland: Loss of plant diversity and elevated CO2 have divergent and opposite effects on oak establishment. Oecologia 2012, 171, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Noguchi, S.; Nik, A.R.; Kasran, B.; Tani, M.; Sammori, T.; Morisada, K. Soil physical properties and preferential flow pathways in tropical rain forest, Bukit Tarek, Peninsular Malaysia. J. For. Res. 1997, 2, 115–120. [Google Scholar] [CrossRef]
- De Baets, S.; Poesen, J.; Knapen, A.; Galindo, P. Impact of root architecture on the erosion-reducing potential of roots during concentrated flow. Earth Surf. Process. Landforms 2007, 32, 1323–1345. [Google Scholar] [CrossRef]
- Le Bissonnais, Y.; Prieto, I.; Roumet, C.; Nespoulous, J.; Metayer, J.; Huon, S.; Villatoro, M.; Stokes, A. Soil aggregate stability in Mediterranean and tropical agro-ecosystems: Effect of plant roots and soil characteristics. Plant Soil 2017, 424, 303–317. [Google Scholar] [CrossRef]
- Fornara, D.A.; Tilman, D. Plant functional composition influences rates of soil carbon and nitrogen accumulation. J. Ecol. 2008, 96, 314–322. [Google Scholar] [CrossRef]
- Kätterer, T.; Bolinder, M.A.; Andrén, O.; Kirchmann, H.; Menichetti, L. Roots contribute more to refractory soil organic matter than above-ground crop residues, as revealed by a long-term field experiment. Agric. Ecosyst. Environ. 2011, 141, 184–192. [Google Scholar] [CrossRef]
- Rasse, D.P.; Rumpel, C.; Dignac, M.-F. Is soil carbon mostly root carbon? Mechanisms for a specific stabilisation. Plant Soil 2005, 269, 341–356. [Google Scholar] [CrossRef]
- Freschet, G.T.; Violle, C.; Roumet, C.; Garnier, E. Interactions between Soil and Vegetation: Structure of Plant Communities and Soil Functioning. In Soils as a Key Component of the Critical Zone 6: Ecology; Wiley: Hoboken, NJ, USA, 2018; pp. 83–104. [Google Scholar]
- Philippot, L.; Hallin, S.; Börjesson, G.; Baggs, E.M. Biochemical cycling in the rhizosphere having an impact on global change. Plant Soil 2008, 321, 61–81. [Google Scholar] [CrossRef]
- Orwin, K.H.; Buckland, S.M.; Johnson, D.; Turner, B.L.; Smart, S.M.; Oakley, S.; Bardgett, R.D. Linkages of plant traits to soil properties and the functioning of temperate grassland. J. Ecol. 2010, 98, 1074–1083. [Google Scholar] [CrossRef]
- Abalos, D.; De Deyn, G.B.; Kuyper, T.W.; Van Groenigen, J.W. Plant species identity surpasses species richness as a key driver of N2O emissions from grassland. Glob. Chang. Boil. 2013, 20, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Necpalova, M.; Anex, R.P.; Kravchenko, A.N.; Abendroth, L.J.; Del Grosso, S.J.; Dick, W.A.; Helmers, M.J.; Herzmann, D.; Lauer, J.G.; Nafziger, E.D.; et al. What does it take to detect a change in soil carbon stock? A regional comparison of minimum detectable difference and experiment duration in the north central United States. J. Soil Water Conserv. 2014, 69, 517–531. [Google Scholar] [CrossRef]
- Jungers, J.M.; DeHaan, L.H.; Mulla, D.J.; Sheaffer, C.C.; Wyse, N.L. Reduced nitrate leaching in a perennial grain crop compared to maize in the Upper Midwest, USA. Agric. Ecosyst. Environ. 2019, 272, 63–73. [Google Scholar] [CrossRef]
- Sutherlin, C.E.; Brunsell, N.; De Oliveira, G.; Crews, T.E.; DeHaan, L.; Vico, G. Contrasting Physiological and Environmental Controls of Evapotranspiration over Kernza Perennial Crop, Annual Crops, and C4 and Mixed C3/C4 Grasslands. Sustainability 2019, 11, 1640. [Google Scholar] [CrossRef]
- De Oliveira, G.; Brunsell, N.A.; Sutherlin, C.E.; Crews, T.E.; DeHaan, L.R. Energy, water and carbon exchange over a perennial Kernza wheatgrass crop. Agric. For. Meteorol. 2018, 249, 120–137. [Google Scholar] [CrossRef]
- Poirier, V.; Angers, D.A.; Whalen, J.K. Formation of millimetric-scale aggregates and associated retention of 13C–15N-labelled residues are greater in subsoil than topsoil. Soil Boil. Biochem. 2014, 75, 45–53. [Google Scholar] [CrossRef]
- Prieto, I.; Stokes, A.; Roumet, C. Root functional parameters predict fine root decomposability at the community level. J. Ecol. 2016, 104, 725–733. [Google Scholar] [CrossRef]
- Freschet, G.T.; Aerts, R.; Cornelissen, J.H.C. A plant economics spectrum of litter decomposability: Afterlife effects of the plant economics spectrum. Funct. Ecol. 2012, 26, 56–65. [Google Scholar] [CrossRef]
- Guyonnet, J.P.; Cantarel, A.; Simon, L.; Haichar, F.E.Z. Root exudation rate as functional trait involved in plant nutrient-use strategy classification. Ecol. Evol. 2018, 8, 8573–8581. [Google Scholar] [CrossRef]
- Poorter, H.; Niklas, K.J.; Reich, P.B.; Oleksyn, J.; Poot, P.; Mommer, L. Biomass allocation to leaves, stems and roots: Meta-analyses of interspecific variation and environmental control: Tansley review. New Phytol. 2012, 193, 30–50. [Google Scholar] [CrossRef]
- Huo, C.; Luo, Y.; Cheng, W. Rhizosphere priming effect: A meta-analysis. Soil Boil. Biochem. 2017, 111, 78–84. [Google Scholar] [CrossRef]
- Barea, J.M.; Azcón, R.; Azcón-Aguilar, C. Mycorrhizosphere interactions to improve plant fitness and soil quality. Antonie van Leeuwenhoek 2002, 81, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Johansson, J.F.; Paul, L.R.; Finlay, R.D. Microbial interactions in the mycorrhizosphere and their significance for sustainable agriculture. FEMS Microbiol. Ecol. 2004, 48, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Rillig, M.C.; Wright, S.F.; Eviner, V.T. The role of arbuscular mycorrhizal fungi and glomalin in soil aggregation: Comparing effects of five plant species. Plant Soil 2002, 238, 325–333. [Google Scholar] [CrossRef]
- De Vries, F.T.; Hoffland, E.; Van Eekeren, N.; Brussaard, L.; Bloem, J. Fungal/bacterial ratios in grasslands with contrasting nitrogen management. Soil Boil. Biochem. 2006, 38, 2092–2103. [Google Scholar] [CrossRef]
- Bardgett, R.D.; Hobbs, P.J.; Frostegård, Å. Changes in soil fungal: Bacterial biomass ratios following reductions in the intensity of management of an upland grassland. Biol. Fert. Soils 1996, 22, 261–264. [Google Scholar] [CrossRef]
- Clemmensen, K.E.; Bahr, A.; Ovaskainen, O.; Dahlberg, A.; Ekblad, A.; Wallander, H.; Stenlid, J.; Finlay, R.; Wardle, D.A.; Lindahl, B.D. Roots and Associated Fungi Drive Long-Term Carbon Sequestration in Boreal Forest. Science 2013, 339, 1615–1618. [Google Scholar] [CrossRef]
- Six, J.; Frey, S.D.; Thiet, R.K.; Batten, K.M. Bacterial and Fungal Contributions to Carbon Sequestration in Agroecosystems. Soil Sci. Soc. Am. J. 2006, 70, 555–569. [Google Scholar] [CrossRef]
- Cotrufo, M.F.; Soong, J.L.; Horton, A.J.; Campbell, E.E.; Haddix, M.L.; Wall, D.H.; Parton, W.J. Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat. Geosci. 2015, 8, 776–779. [Google Scholar] [CrossRef]
- Haddix, M.L.; Paul, E.A.; Cotrufo, M.F. Dual, differential isotope labeling shows the preferential movement of labile plant constituents into mineral-bonded soil organic matter. Glob. Chang. Boil. 2016, 22, 2301–2312. [Google Scholar] [CrossRef] [PubMed]
- Lemaire, G.; Gastal, F.; Franzluebbers, A.J.; Chabbi, A. Grassland–Cropping Rotations: An Avenue for Agricultural Diversification to Reconcile High Production with Environmental Quality. Environ. Manag. 2015, 56, 1065–1077. [Google Scholar] [CrossRef] [PubMed]
- Grover, K.; Karsten, H.D.; Roth, G.W. Corn Grain Yields and Yield Stability in Four Long-Term Cropping Systems. Agron. J. 2009, 101, 940–946. [Google Scholar] [CrossRef]
- Franzluebbers, A.J.; Sawchik, J.; Taboada, M.A. Agronomic and environmental impacts of pasture–crop rotations in temperate North and South America. Agric. Ecosyst. Environ. 2014, 190, 18–26. [Google Scholar] [CrossRef]
- King, A.E.; Blesh, J. Crop rotations for increased soil carbon: Perenniality as a guiding principle. Ecol. Appl. 2017, 28, 249–261. [Google Scholar] [CrossRef] [PubMed]
- Soussana, J.-F.; Lemaire, G. Coupling carbon and nitrogen cycles for environmentally sustainable intensification of grasslands and crop-livestock systems. Agric. Ecosyst. Environ. 2014, 190, 9–17. [Google Scholar] [CrossRef]
- Crews, T.E.; Peoples, M.B. Can the Synchrony of Nitrogen Supply and Crop Demand be Improved in Legume and Fertilizer-based Agroecosystems? A Review. Nutr. Cycl. Agroecosystems 2005, 72, 101–120. [Google Scholar] [CrossRef]

| Soil Depth Layer (cm) | SOIL Texture | Crop Treatment | Organic Matter | Total Nitrogen | Phosphorus | C:N Ratio | pH | Biological Activity Traces | Macro-Porosity |
|---|---|---|---|---|---|---|---|---|---|
| (g kg−1) | |||||||||
| 0–30 | Sandy loam | Annual | 23.8 (1.5) | 1.4 (0.5) | 0.04 (0.006) | 8.3 (0.3) | 6.7 (0.04) | Strong | High |
| Perennial | 24.6 (0.9) NS | 1.9 (0.4) NS | 0.03 (0.01) NS | 9.7 (0.4) NS | 6.8 (0.04) NS | ||||
| 30–60 | Sandy clay loam | Annual | 9.7 (0.9) | 0.7 (0.1) | 0.01 (0.003) | 8.4 (0.05) | 6.7 (0.02) | Strong | High |
| Perennial | 9.9 (0.6) NS | 0.6 (0.2) NS | 0.01 (0.002) NS | 8.2 (0.08) NS | 7.0 (0.04) NS | ||||
| 60–100 | Silty clay loam | Annual | 5.7 (0.6) | 0.4 (0.08) | 0.009 (0.002) | 6.0 (0.09) | 7.2 (0.03) | Moderate | Moderate |
| Perennial | 6.0 (0.7) NS | 0.4 (0.1) NS | 0.01 (0.004) NS | 5.4 (0.04) NS | 6.8 (0.02) NS | ||||
| 100–160 | Clay loam | - | - | - | - | - | Low | Low–temporary hydromorphy | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Duchene, O.; Celette, F.; Barreiro, A.; Dimitrova Mårtensson, L.-M.; Freschet, G.T.; David, C. Introducing Perennial Grain in Grain Crops Rotation: The Role of Rooting Pattern in Soil Quality Management. Agronomy 2020, 10, 1254. https://doi.org/10.3390/agronomy10091254
Duchene O, Celette F, Barreiro A, Dimitrova Mårtensson L-M, Freschet GT, David C. Introducing Perennial Grain in Grain Crops Rotation: The Role of Rooting Pattern in Soil Quality Management. Agronomy. 2020; 10(9):1254. https://doi.org/10.3390/agronomy10091254
Chicago/Turabian StyleDuchene, Olivier, Florian Celette, Ana Barreiro, Linda-Maria Dimitrova Mårtensson, Grégoire T. Freschet, and Christophe David. 2020. "Introducing Perennial Grain in Grain Crops Rotation: The Role of Rooting Pattern in Soil Quality Management" Agronomy 10, no. 9: 1254. https://doi.org/10.3390/agronomy10091254
APA StyleDuchene, O., Celette, F., Barreiro, A., Dimitrova Mårtensson, L.-M., Freschet, G. T., & David, C. (2020). Introducing Perennial Grain in Grain Crops Rotation: The Role of Rooting Pattern in Soil Quality Management. Agronomy, 10(9), 1254. https://doi.org/10.3390/agronomy10091254





