Insights into the Physiological and Biochemical Impacts of Salt Stress on Plant Growth and Development
Abstract
1. Introduction
1.1. Overview of Salinity
1.2. Salinity and Morphological Attributes
1.3. Salinity and Physiological Attributes
2. Salinity and Water Relations
3. Salinity and Biochemical Attributes
3.1. Salinity and Proline
3.2. Salinity and Polyamines
3.3. Salinity and Glycine-Betaine
4. Salinity and Enzymatic Attributes
5. Salinity and Phytohormones
Salinity and Growth Regulation
6. Salinity and Carbohydrate (Sugars) Metabolism in Plants
7. Salinity and Root Apoplastic Barriers
8. Salinity and Ionic Attributes
8.1. Salinity and Ionic Homeostasis
8.2. Ionic Influx and Efflux
9. Effects of Salinity on Potassium and Calcium
Salinity and Nitrogen
10. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shrivastava, P.; Kumar, R. Soil salinity: A serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J. Biol. Sci. 2015, 22, 123–131. [Google Scholar] [CrossRef]
- Rogel, J.A.; Ariza, F.A.; Silla, R.O. Soil salinity and moisture gradients and plant zonation in Mediterranean salt marshes of Southeast Spain. Wetlands 2000, 20, 357–372. [Google Scholar] [CrossRef]
- Flowers, T.J.; Garcia, A.; Koyama, M.; Yeo, A.R. Breeding for salt tolerance in crop plants—The role of molecular biology. Acta Physiol. Plant. 1997, 19, 427–433. [Google Scholar] [CrossRef]
- Rubio, J.S.; Garcia-Sanchez, F.; Rubio, F.; Martinez, V. Yield, blossom-end rot incidence and fruit quality in pepper plants under moderate salinity are affected by K+ and Ca+2 fertilization. Sci. Hortic. 2009, 119, 79–87. [Google Scholar] [CrossRef]
- Ghassemi, F.; Jakeman, A.J.; Nix, H.A. Salinization of Land and Water Resources; University of New south Wales Press Ltd.: Canberra, Australia, 1995. [Google Scholar]
- Sonowal, H.; Pal, P.B.; Shukla, K.; Ramana, K.V. Ramana, Aspalatone Prevents VEGF-Induced Lipid Peroxidation, Migration, Tube Formation, and Dysfunction of Human Aortic Endothelial Cells. Oxid. Med. Cell. Longev. 2017, 11, 2017. [Google Scholar] [CrossRef]
- Wang, H.; Tang, X.; Shao, C.; Shao, H.; Wang, L. Molecular cloning and bioinformatics analysis of a new plasma membrane Na+/H+ antiporter gene from the halophyte Kosteletzkya virginica. Sci. World J. 2014, 2014, 141675. [Google Scholar]
- Xiong, L.; Zhu, J.K. Molecular and genetic aspects of plant responses to osmotic stress. Plant Cell Environ. 2002, 25, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Zhou, J.; Sui, N. Mechanisms of salt tolerance in halophytes: Current understanding and recent advances. Open Llife Sci. 2018, 13, 149–154. [Google Scholar] [CrossRef]
- Ashraf, M.H.P.J.C.; Harris, P.J. Photosynthesis under stressful environments: An overview. Photosynthetica 2013, 51, 163–190. [Google Scholar] [CrossRef]
- Siew, R.; Klein, C.R. The effect of NaCl on some metabolic and fine structural changes during the greening of etiolated leaves. J. Cell Biol. 1969, 37, 590–596. [Google Scholar] [CrossRef]
- Slama, I.; Abdelly, C.; Bouchereau, A.; Flowers, T.; Savoure, A. Diversity, distribution and roles of osmoprotective compounds accumulated in halophytes under abiotic stress. Ann. Bot. 2015, 115, 433–447. [Google Scholar] [CrossRef] [PubMed]
- Cushman, J.C. Molecular cloning and expression of chloroplast NADP-malate dehydrogenase during crassulacean acid metabolism induction by salt stress. Photosynth. Res. 1990, 35, 15–27. [Google Scholar] [CrossRef]
- Sévin, D.C.; Stählin, J.N.; Pollak, G.R.; Kuehne, A.; Sauer, U. Global Metabolic Responses to Salt Stress in Fifteen Species. PLoS ONE 2016, 11, e0148888. [Google Scholar] [CrossRef] [PubMed]
- Dschida, W.J.; Platt-Aloia, K.A.; Thomson, W.W. Epidermal peels of Avicennia germinans (L.) Stearn: A useful system to study the function of salt glands. Ann. Bot. Lond. 1992, 70, 501–509. [Google Scholar] [CrossRef]
- Robert, I.L.; Frank, W.J.; Summy, K.R.; Hudson, D.Y.; Richard, S. The Biological Flora of Coastal Dunes and Wetlands. Avicennia Germinans J. Coast. Res. 2017, 33, 191–207. [Google Scholar]
- Tester, M.; Davenport, R. Na+ tolerance and Na+ transport in higher plants. Ann. Bot. 2003, 91, 503–527. [Google Scholar] [CrossRef] [PubMed]
- Shahid, M.A.; Balal, R.M.; Khan, N.; Rossi, L.; Rathinasabapathi, B.; Liu, G.; Khan, J.; Cámara-Zapata, J.M.; Martínez-Nicolas, J.J.; Garcia-Sanchez, F. Polyamines provide new insights into the biochemical basis of Cr-tolerance in Kinnow mandarin grafted on diploid and double-diploid rootstocks. Environ. Exp. Bot. 2018, 1, 248–260. [Google Scholar] [CrossRef]
- Zhu, J.; Hasegawa, P.M.; Bressan, R.A. Molecular aspects of osmotic stress in plants. Citri. Rev. Plant Sci. 1997, 16, 253–277. [Google Scholar] [CrossRef]
- Serrano, R.; Mulet, J.M.; Rios, G.; Marquez, J.A.; de Larrinoa, I.F.; Leube, M.P.; Mendizabal, I.; Pascual-Ahuir, A.; Proft, M.; Ros, R.; et al. A glimpse of the mechanism of ion homeostasis during salt stress. J. Exp. Bot. 1999, 50, 1023–1036. [Google Scholar] [CrossRef]
- Flowers, T.J.; Munns, R.; Colmer, T.D. Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann. Bot. 2015, 115, 419–431. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Babar, M.A. The stimulatory effects of plant growth promoting rhizobacteria and plant growth regulators on wheat physiology grown in sandy soil. Arch. Microbiol. 2019, 201, 769–785. [Google Scholar] [CrossRef] [PubMed]
- Price, A.; Hendry, G. Iron catalysed oxygen radical formation and its possible contribution to drought damage in nine native grasses and three cereals. Plant Cell Environ. 1991, 14, 477–484. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Effects of exogenously applied salicylic acid and putrescine alone and in combination with rhizobacteria on the phytoremediation of heavy metals and chickpea growth in sandy soil. Int. J. Phytoremed. 2018, 16, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Stevens, R.M.; Pech, J.M.; Grigson, G.J. A Short Non-Saline Sprinkling Increases the Tuber Weights of Saline Sprinkler Irrigated Potatoes. Agronomy 2017, 7, 4. [Google Scholar] [CrossRef]
- Nxele, X.; Klein, A.; Ndimba, B.K. Drought and salinity stress alters ROS accumulation, water retention, and osmolyte content in sorghum plants. Afr. J. Bot. 2017, 1, 261–266. [Google Scholar] [CrossRef]
- Golldack, D.; Li, C.; Mohan, H.; Probst, N. Tolerance to drought and salt stress in plants: Unraveling the signaling networks. Front. Plant Sci. 2014, 5, 151. [Google Scholar] [CrossRef]
- Naseem, H.; Ahsan, M.; Shahid, M.A.; Khan, N. Exopolysaccharides producing rhizobacteria and their role in plant growth and drought tolerance. J. Basic Microbiol. 2018, 58, 1009–1022. [Google Scholar] [CrossRef]
- Zekri, M.; Parsons, L.R. Calcium influnces growth and leaf mineral concentration of citrus under saline conditions. Hort. Sci. 1990, 25, 784–786. [Google Scholar] [CrossRef]
- Kumari, A.; Das, P.; Parida, A.K.; Agarwal, P.K. Proteomics, metabolomics, and ionomics perspectives of salinity tolerance in halophytes. Front. Plant Sci. 2015, 6, 537. [Google Scholar] [CrossRef]
- Saha, J.; Brauer, E.K.; Sengupta, A.; Popescu, S.C.; Gupta, K.; Gupta, B. Polyamines as redox homeostasis regulators during salt stress in plants. Front. Environ. Sci. 2015, 3, 21. [Google Scholar] [CrossRef]
- Mansour, M.M.F. Cell permeability under salt stress. In Strategies for Improving Salt Tolerance in Higher Plants; Jaiwal, P.K., Singh, R.P., Gulati, A., Eds.; Oxford and IBH: New Delhi, India, 1997; pp. 87–110. [Google Scholar]
- Ali, S.; Xie, L. Plant Growth Promoting and Stress mitigating abilities of Soil Born Microorganisms. Recent Pat. Food Nutr. Agric. 2019, 15. [Google Scholar] [CrossRef] [PubMed]
- Mansour, M.M.F.; Salama, K.H.A. Cellular basis of salinity tolerance in plants. Environ. Exp. Bot. 2004, 52, 113–122. [Google Scholar] [CrossRef]
- Le Gall, H.; Philippe, F.; Domon, J.M.; Gillet, F.; Pelloux, J.; Rayon, C. Cell wall metabolism in response to abiotic stress. Plants 2015, 4, 112–166. [Google Scholar] [CrossRef] [PubMed]
- Al-Amri, S.M. Improved growth, productivity and quality of tomato (Solanum lycopersicum L.) plants through application of shikimic acid. Saudi J. Biol. Sci. 2013, 1, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Mahlooji, M.; Seyed Sharifi, R.; Razmjoo, J.; Sabzalian, M.R.; Sedghi, M. Effect of salt stress on photosynthesis and physiological parameters of three contrasting barley genotypes. Photosynthetica 2017, 56, 549–556. [Google Scholar] [CrossRef]
- Shekari, F.; Abbasi, A.; Mustafavi, S.H. Effect of silicon and selenium on enzymatic changes and productivity of dill in saline condition. J. Saudi Soc. Agric. Sci. 2017, 1, 367–374. [Google Scholar] [CrossRef]
- Dolatabadian, A.; Sanavy, S.A.M.M.; Gholamhoseini, M.; Joghan, A.K.; Majdi, M.; Kashkooli, A. The role of calcium in improving photosynthesis and related physiological and biochemical attributes of spring wheat subjected to simulated acid rain. Physiol. Mol. Biol. Plants. 2013, 19, 189–198. [Google Scholar] [CrossRef]
- Farooq, S.; Azam, F. The use of cell membrane stability (CMS) technique to screen for salt tolerant wheat varieties. J. Plant Physiol. 2006, 163, 629–637. [Google Scholar] [CrossRef]
- Munns, R. Comparative physiology of salt and water stress. Plant Cell Environ. 2002, 25, 239–250. [Google Scholar] [CrossRef]
- Dolatabadi, N.; Toorchi, M. Rapeseed (Brassica napus L.) genotypes response to NaCl salinity. J. Biodiv. Environ. Sci. 2017, 10, 265–270. [Google Scholar]
- Wu, J.; Seliskar, D.M.; Gallagher, J.L. Stress tolerance in the march plant Spartina patens: Impact of NaCl on growth and root plasma membrane lipid composition. Physiol. Plant 1998, 102, 307–317. [Google Scholar] [CrossRef]
- Balal, R.M.; Shahid, M.A.; Vincent, C.; Zotarelli, L.; Liu, G.; Mattson, N.S.; Garcia-Sanchez, F. (Kinnow mandarin plants grafted on tetraploid rootstocks are more tolerant to Cr-toxicity than those grafted on its diploids one. Environ. Exp. Bot. 2017, 140, 8–18. [Google Scholar] [CrossRef]
- Plant, A.L.; Bray, E.A. Regulation of Gene Expression by Abscissic Acid During Environmental Stresses; Marcel Dekker: New York, NY, USA, 1999; pp. 303–331. [Google Scholar]
- Delphine, S.; Alvino, A.; Zacchini, M.; Loreb, F. Consequences of salt stress on conductance to CO2 diffusion, Rubisco characteristics and anatomy of spinach leaves. Aust. J. Plant Physiol. 1998, 25, 395–402. [Google Scholar] [CrossRef]
- Mehmood, A.; Khan, N.; Irshad, M.; Hamayun, M. IAA Producing Endopytic Fungus Fusariun oxysporum wlw Colonize Maize Roots and Promoted Maize Growth Under Hydroponic Condition. Eur. Exp. Biol. 2018, 8, 24. [Google Scholar] [CrossRef]
- Romero-Aranda, R.; Moya, J.L.; Tadeo, F.R.; Legaz, F.; Primo-Millo, E.; Talón, M. Physiological and anatomical disturbances induced by chloride salts in sensitive and tolerant citrus: Beneficial and detrimental effects of cations. Plant Cell Environ. 1998, 21, 1243–1253. [Google Scholar] [CrossRef]
- Hasegawa, P.M.; Bressan, R.A.; Zhu, J.K.; Bohnert, H.J. Plant cellular and molecular responses to high salinity. Annu. Rev. Plant Physiol. 2000, 51, 463–499. [Google Scholar] [CrossRef]
- Blumwald, E.; Aharon, G.S.; Apse, M.P. Sodium transport in plant cells. Biochim. Biophys. Acta 2000, 1465, 140–151. [Google Scholar] [CrossRef]
- Maser, P.; Gierth, M.; Schroeder, J.I. Molecular mechanisms of potassium and sodium uptake in plants. Plant Soil. 2002, 247, 43–54. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Modulation of phytoremediation and plant growth by the treatment with PGPR, Ag nanoparticle and untreated municipal wastewater. Int. J. Phytoremed. 2016, 18, 1258–1269. [Google Scholar] [CrossRef]
- Zribi, L.; Fatma, G.; Fatma, R.; Salwa, R.; Hassan, N.; Nejib, R.M. Application of chlorophyll fluorescence for the diagnosis of salt stress in tomato “Solanum lycopersicum (variety Rio Grande)”. Sci. Hortic. 2009, 120, 367–372. [Google Scholar] [CrossRef]
- Orlovsky, N.; Japakova, U.; Zhang, H.; Volis, S. Effect of salinity on seed germination, growth and ion content in dimorphic seeds of Salicornia europaea L. (Chenopodiaceae). Plant Divers. 2016, 1, 183–189. [Google Scholar] [CrossRef] [PubMed]
- Shaterian, J.; Waterer, D.; De Jong, H.; Tanino, K.K. Differential stress responses to NaCl salt application in early and late maturing diploid potato (Solanum sp.) clones. Environ. Exp. Bot 2005, 54, 202–212. [Google Scholar] [CrossRef]
- Chartzoulakis, K.; Klapaki, G. Response of two greenhouse pepper hybrids to NaCl salinity during different growth stages. Sci. Hortic. 2000, 86, 247–260. [Google Scholar] [CrossRef]
- Kaya, C.; Tuna, A.L.; Ashrafa, M.; Altunlu, H. Improved salt tolerance of melon (Cucumis melo L.) by the addition of proline and potassium nitrate. Environ. Exp. Bot 2007, 60, 397–403. [Google Scholar] [CrossRef]
- Giuffrida, F.; Graziani, G.; Fogliano, V.; Scuderi, D.; Romano, D.; Leonardi, C. Effects of nutrient and NaCl salinity on growth, yield, quality and composition of pepper grown in soilless closed system. J. Plant Nutr. 2014, 37, 1455–1474. [Google Scholar] [CrossRef]
- Jiang, C.; Zu, C.; Lu, D.; Zheng, Q.; Shen, J.; Wang, H.; Li, D. Effect of exogenous selenium supply on photosynthesis, Na+ accumulation and antioxidative capacity of maize (Zea mays L.) under salinity stress. Sci. Rep. 2007, 7, 42039. [Google Scholar] [CrossRef]
- Savvas, D.; Lenz, F. Effect of NaCl or nutrient-induced salinity on growth, yield and composition of egg plants grown in rockwool. Sci. Hortic. 2000, 84, 37–47. [Google Scholar] [CrossRef]
- Abbas, W.; Ashraf, M.; Akram, N.A. Alleviation of salt-induced adverse effects in egg plant (Solanum melongena L.) by glycinebetaine and sugar beet extracts. Sci. Hortic. 2010, 125, 188–195. [Google Scholar] [CrossRef]
- Mahjoor, F.; Ghaemi, A.A.; Golabi, M.H. Interaction effects of water salinity and hydroponic growth medium on eggplant yield, water-use efficiency, and evapotranspiration. Int. Soil Water Conserv. 2016, 1, 99–107. [Google Scholar] [CrossRef]
- Balal, R.M.; Shahid, M.A.; Javaid, M.M.; Iqbal, Z.; Liu, G.D.; Zotarelli, L.; Khan, N. Chitosan alleviates phytotoxicity caused by boron through augmented polyamine metabolism and antioxidant activities and reduced boron concentration in Cucumis sativus L. Acta Physiol. Plant. 2017, 1, 31. [Google Scholar] [CrossRef]
- Tabatabaie, S.J.; Nazari, J. Influence of nutrient concentrations and NaCl salinity on the growth, photosynthesis, and essential oil content of peppermint and lemon verbena. Turk. J. Agric. For. 2007, 31, 245–253. [Google Scholar]
- Pardossi, A.; Bagnoli, G.; Malorgio, F.C.; Campiotti, A.; Tognoni, F. NaCl effects on celery (Apium graveolens L.) grown in NFT. Sci. Hortic 1999, 81, 229–242. [Google Scholar] [CrossRef]
- Zheng, Y.; Jia, A.; Ning, T.; Xu, J.; Li, Z.; Jiang, G. Potassium nitrate application alleviates sodium chloride stress in winter wheat cultivars differing in salt tolerance. J. Plant Physiol. 2008, 165, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Saini, S.; Sharma, I.; Pati, P.K. Versatile roles of brassinosteroid in plants in the context of its homoeostasis, signaling and crosstalks. Front. Plant Sci. 2015, 6, 950. [Google Scholar] [CrossRef] [PubMed]
- Kalhoro, N.A.; Rajpar, I.; Kalhoro, S.A.; Ali, A.; Raza, S.; Ahmed, M.; Kalhoro, F.A.; Ramzan, M.; Wahid, F. Effect of salts stress on the growth and yield of wheat (Triticum aestivum L.). Am. J. Plant Sci. 2016, 7, 2257. [Google Scholar] [CrossRef]
- Xie, Z.; Duan, L.; Tian, X.; Wang, B.; Eneji, A.E.; Li, Z. Coronatine alleviates salinity stress in cotton by improving the antioxidation defence system and radical-scavenging activity. J. Plant Physiol. 2008, 165, 375–384. [Google Scholar] [CrossRef]
- Sun, C.; Feng, D.; Mi, Z.; Li, C.; Zhang, J.; Gao, Y.; Sun, J. Impacts of Ridge-Furrow Planting on Salt Stress and Cotton Yield under Drip Irrigation. Water 2017, 9, 49. [Google Scholar] [CrossRef]
- Caines, A.M.; Shennan, C. Interactive effects of Ca+2 and NaCl salinity on the growth of two tomato genotypes differing in Ca+2 use efficiency. Plant Physiol. Biochem. 1999, 37, 569–576. [Google Scholar] [CrossRef]
- Evers, D.; Hemmer, K.; Hausman, J.F. Salt stress induced biometric and physiological changes in Solanum tuberosum L. cv. Bintje grown in vitro. Acta Physiol. Plant 1998, 20, 3–7. [Google Scholar] [CrossRef]
- Gao, Y.; Lu, Y.; Wu, M.; Liang, E.; Li, Y.; Zhang, D.; Yin, Z.; Ren, X.; Dai, Y.; Deng, D.; et al. Ability to Remove Na+ and Retain K+ Correlates with Salt Tolerance in Two Maize Inbred Lines Seedlings. Front. Plant Sci. 2016, 7, 1716. [Google Scholar] [CrossRef]
- Hu, Y.; Xia, S.; Su, Y.; Wang, H.; Luo, W.; Su, S.; Xiao, L. Brassinolide Increases Potato Root Growth In Vitro in a Dose-Dependent Way and Alleviates Salinity Stress. Biomed. Res. Int. 2016, 8231873. [Google Scholar] [CrossRef]
- Alom, R.; Hasan, M.A.; Islam, M.R.; Wang, Q.F. Germination characters and early seedling growth of wheat (Triticum aestivum L.) genotypes under salt stress conditions. JCSB 2016, 1, 383–392. [Google Scholar] [CrossRef]
- Tuna, A.L.; Kaya, C.; Ashraf, M.; Altunlu, H.; Yokas, I.; Yagmur, B. The effect of calcium sulphate on growth, membrane stability and nutrient uptake of tomato plants grown under salt stress. Environ. Exp. Bot. 2007, 59, 173–178. [Google Scholar] [CrossRef]
- De Pascale, S.; Barbieri, G. Effects of soil salinity and top removal on growth and yield of broad bean as a green vegetable. Sci. Hortic 1997, 71, 147–165. [Google Scholar] [CrossRef]
- Verma, S.; Mishra, S.N. Putrescine alleviation of growth in salt stressed Brassica juncea by inducing antioxidative defense system. J. Plant Physiol. 2005, 162, 669–677. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Schmidhalter, U. Drought and Salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil. Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Rahnama, H.; Ebrahimzadeh, H. The effect of NaCl on antioxidant activities in potato seedlings. Biol. Plant. 2005, 49, 93–97. [Google Scholar] [CrossRef]
- Korkmaz, A.; Şirikçi, R.; Kocaçınar, F.; Değer, Ö.; Demirkırıan, A.R. Alleviation of salt-induced adverse effects in pepper seedlings by seed application of glycinebetaine. Sci. Hortic. 2012, 4, 197–205. [Google Scholar] [CrossRef]
- Roy, T.S.; Chakraborty, R.; Parvez, M.N.; Mostofa, M.; Ferdous, J.; Ahmed, S. Yield, dry matter and specific gravity of exportable potato: Response to salt. Univ. J. Agric. Res. 2017, 5, 98–103. [Google Scholar]
- Mulholland, B.J.; Taylor, I.B.; Jackson, A.C.; Thornpson, A.J. Can ABA mediate responses of salinity have stressed tomato. Environ. Exp. Bot. 2003, 50, 17–28. [Google Scholar] [CrossRef]
- Mayak, S.; Tirosh, T.; Glick, B.R. Plant growth promoting bacteria confer resistance in tomato plants to salt stress. Plant Sci. Plant Sci. 2004, 42, 565–572. [Google Scholar]
- Smalle, J.; Van Der Straeten, D. Ethylene and vegetative development. Physiol. Plant. 1997, 100, 593–605. [Google Scholar] [CrossRef]
- Akhtar, S.S.; AkhtarAktas, H.; Abak, K.; Cakmak, I. Genotypic variation in the response of pepper to salinity. Sci. Hortic. 2006, 110, 260–266. [Google Scholar]
- Delgado, M.J.; Garrido, J.M.; Ligero, F.; Lluch, C. Nitrogen fixation and carbon metabolism by nodules and bacteroids of pea plants under sodium chloride stress. Physiol. Plant. 1993, 89, 824–829. [Google Scholar] [CrossRef]
- Borucki, W.; Sujkowska, M. The effects of sodium chloride-salinity upon growth, nodulation, and root nodule structure of pea (Pisum sativum L.) plants. Acta Physiol. Plant. 2008, 30, 293–301. [Google Scholar] [CrossRef]
- Andrzej, T.; Philip, P. Role of root microbiota in plant productivity. J. Exp. Bot. 2015, 66, 2167–2175. [Google Scholar]
- Zahran, H.H. Rhizobium-Legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol. Mol. Biol. R. 1999, 63, 968–989. [Google Scholar] [CrossRef]
- Batista-Santos, P.; Duro, N.; Rodrigues, A.P.; Semedo, J.N.; Alves, P.; da Costa, M.; Graça, I.; Pais, I.P.; Scotti-Campos, P.; Lidon, F.C.; et al. Is salt stress tolerance in Casuarina glauca Sieb. ex Spreng. associated with its nitrogen-fixing root-nodule symbiosis? An analysis at the photosynthetic level. Plant Physiol. Biochem. 2015, 1, 97–109. [Google Scholar] [CrossRef]
- Othman, Y.; Al-karaki, G.; Al-Tawaha, A.R.; Al-Horani, A. Variation in germination and ion uptake in barley genotypes under salinity conditions. World J. Agric. Res. 2006, 2, 11–15. [Google Scholar]
- Kandil, A.A.; Shareif, E.; Gad, M.A. Effect of Salinity on Germination and Seeding Parameters of Forage Cowpea Seed. Res. J. Seed Sci. 2017, 10, 17–26. [Google Scholar]
- Khan, M.A.; Rizvi, Y. Effect of salinity, temperature and growth regulators on the germination and early seedling growth of Atriplex griffithi Var. Stocksii. Can J. Bot. 1994, 72, 475–479. [Google Scholar] [CrossRef]
- Rajjou, L.; Duval, M.; Gallardo, K.; Catusse, J.; Bally, J.; Job, C.; Job, D. Seed germination and vigor. Annu. Rev. Plant Biol. 2012, 2, 507–533. [Google Scholar] [CrossRef]
- Meena, K.K.; Sorty, A.M.; Bitla, U.M.; Choudhary, K.; Gupta, P.; Pareek, A.; Minhas, P.S. Abiotic Stress Responses and Microbe-Mediated Mitigation in Plants: The Omics Strategies. Front. Plant Sci. 2017, 8, 172. [Google Scholar] [CrossRef]
- Stepien, P.; Klobus, G. Water relations and photosynthesis in Cucumus sativus L. leaves under salt stress. Biol. Plant. 2006, 50, 610–616. [Google Scholar] [CrossRef]
- Dobrota, C. Energy dependant plant stress acclimation. Rev. Environ. Sci. 2006, 5, 243–251. [Google Scholar] [CrossRef]
- Yeo, A.R.; Capron, S.J.M.; Flowers, T.J. The effect of salinity upon photosynthesis in rice (Oryza sativa L.) Gas exchange by individual leaves relation to their salt content. J. Exp. Bot. 1985, 36, 1240–1248. [Google Scholar] [CrossRef]
- Muradoglu, F.; Gundogdu, M.; Ercisli, S.; Encu, T.; Balta, F.; Jaafar, H.Z.; Zia-Ul-Haq, M. Cadmium toxicity affects chlorophyll a and b content, antioxidant enzyme activities and mineral nutrient accumulation in strawberry. Biol. Res. 2015, 48, 1–7. [Google Scholar] [CrossRef]
- El-Shintinawy, F.; El-Ansary, A. Differential Effect of Cd2+ and Ni2+ on Amino Acid Metabolism in Soybean Seedlings. Biol. Plant. 2000, 43, 79–84. [Google Scholar] [CrossRef]
- Puniran-Hartley, N.; Hartley, J.; Shabala, L.; Shabala, S. Salinity-induced accumulation of organic osmolytes in barley and wheat leaves correlates with increased oxidative stress tolerance: In planta evidence for cross-tolerance. Plant Physiol. Biochem. 2014, 1, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Legaz, M.F.; Ortiz, J.M.; Garcia-Lindon, A.; Cerda, A. Effect of salinity on growth, ion content and CO2 assimilation rate in lemon varieties on different rootstocks. Physiol. Plant. 1993, 89, 427–432. [Google Scholar] [CrossRef]
- Acosta-Motos, J.R.; Ortuño, M.F.; Bernal-Vicente, A.; Diaz-Vivancos, P.; Sanchez-Blanco, M.J.; Hernandez, J.A. Plant responses to salt stress: Adaptive mechanisms. Agronomy 2017, 7, 18. [Google Scholar] [CrossRef]
- Hanachi, S.; Van Labeke, M.C.; Mehouachi, T. Application of chlorophyll fluorescence to screen eggplant (Solanum melongena L.) cultivars for salt tolerance. Photosynthetica 2014, 52, 57–62. [Google Scholar] [CrossRef]
- Tiwari, J.K.; Munshi, A.D.; Kumar, R.; Pandey, R.N.; Arora, A.; Bhat, J.S.; Sureja, A.K. Effect of salt stress on cucumber: Na+–K+ ratio, osmolyte concentration, phenols and chlorophyll content. Acta Physiol. Plant. 2010, 1, 103–114. [Google Scholar] [CrossRef]
- Lycoskoufis, I.H.; Savvas, D.; Mavrogianopoulos, G. Growth, gas exchange, and nutrient status in pepper (Capsicum annuum L.) grown in recirculating nutrient solution as affected by salinity imposed to half of the root system. Sci. Hortic. 2005, 106, 147–161. [Google Scholar] [CrossRef]
- Zhang, T.; Gong, H.; Wen, X.; Lu, C. Salt stress induces a decrease in oxidation energy transfer from phycobilisomes to photosystem II but an increase to photosystem I in the cyanobacterium Spirulina platensis. J. Plant Physiol. 2010, 12, 20. [Google Scholar]
- Wu, X.; He, J.; Chen, J.; Yang, S.; Zha, D. Alleviation of exogenous 6-benzyladenine on two genotypes of eggplant (Solanum melongena Mill.) growth under salt stress. Protoplasma 2014, 251, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Yan, K.; Shao, H.; Shao, C.; Chen, P.; Zhao, S.; Brestic, M.; Chen, X. Physiological adaptive mechanisms of plants grown in saline soil and implications for sustainable saline agriculture in coastal zone. Acta Physiol. Plant. 2013, 1, 2867–2878. [Google Scholar] [CrossRef]
- Hayat, S.; Hasan, S.A.; Yusuf, M.; Hayat, Q.; Ahmad, A. Effect of 28- homobrassinolide on photosynthesis, fluorescence and antioxidant system in the presence or absence of salinity and temperature in Vigna radiata. Environ. Exp. Bot. 2010, 69, 105–112. [Google Scholar] [CrossRef]
- Misra, A.N.; Sahu, S.M.; Misra, M.; Singh, P.; Meera, I.; Das, N.; Kar, M.; Shau, P. Sodium chloride induced changes in leaf growth, and pigment and protein contents in two rice cultivars. Biol. Plant. 1997, 39, 257–262. [Google Scholar] [CrossRef]
- Neelesh, K.; Veena, P. Effect of Salt Stress on Growth Parameters, Moisture Content, Relative Water Content and Photosynthetic Pigments of Fenugreek Variety RMt-1. J. Plant Sci. 2015, 10, 210–221. [Google Scholar] [CrossRef]
- Bethke, P.C.; Drew, M.C. Stomatal and non-stomatal components to inhibition of photosynthesis in leaves of Capsicum annum during progressive exposure to NaCl salinity. Plant Physiol. 1992, 99, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Reddy, M.P.; Vora, A.B. Changes in pigment composition, Hill reaction activity and saccharides metabolism in Bajra (Pennisetum typhoides) leaves under NaCl salinity. Photosynthetica 1986, 20, 50–55. [Google Scholar]
- Gul, B.; Ansari, R.; Flowers, T.J.; Khan, M.A. Germination strategies of halophyte seeds under salinity. Environ. Exp. Bot. 2013, 1, 4–18. [Google Scholar] [CrossRef]
- Parida, A.K.; Das, A.B. Salt tolerance and salinity affects on plant. Ecotoxicol. Environ. 2005, 60, 324–349. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.Y.; Duan, H.; Yang, L.N.; Wang, Z.Q.; Zhou, S.C.; Yang, J.C. Effect of heat stress during meiosis on grain yield of rice cultivars differing in heat tolerance and its physiological mechanism. Acta Agron. Sin. 2008, 34, 2134–2142. [Google Scholar] [CrossRef]
- Elhamid, E.; Sadak, M.; Tawfik, M. Alleviation of Adverse Effects of Salt Stress in Wheat Cultivars by Foliar Treatment with Antioxidant 2—Changes in Some Biochemical Aspects, Lipid Peroxidation, Antioxidant Enzymes and Amino Acid Contents. Agric. Sci. 2014, 5, 1269–1280. [Google Scholar] [CrossRef]
- Khan, N.; Zandi, P.; Ali, S.; Mehmood, A.; Adnan Shahid, M.; Yang, J. Impact of salicylic acid and PGPR on the drought tolerance and phytoremediation potential of Helianthus annus. Front. Microbiol. 2018, 9, 2507. [Google Scholar] [CrossRef]
- Kartashov, A.V.; Radyukina, N.L.; Ivanov, Y.V.; Pashkovskii, P.P.; Shevyakova, N.I.; Kuznetsov, V.V. Role of antioxidants systems in wild plant adaptation to salt stress. Russ. J. Plant Physiol. 2008, 55, 463–468. [Google Scholar] [CrossRef]
- Katsumi, M. Physiological modes of brassinolide action in cucumber hypocotyl growth. In Brassinosteriods: Chemistry, Bioactivity and Applications; Cutler, H.G., Yokota, T., Adam, G., Eds.; American Chemical Society: Washington, DC, USA, 1991; pp. 246–254. [Google Scholar]
- Faried, H.N.; Ayyub, C.M.; Amjad, M.; Ahmed, R. Salinity impairs ionic, physiological and biochemical attributes in potato. Pak. J. Agric. Sci. 2016, 53, 17–25. [Google Scholar]
- Sofo, A.; Scopa, A.; Nuzzaci, M.; Vitti, A. Ascorbate Peroxidase and Catalase Activities and Their Genetic Regulation in Plants Subjected to Drought and Salinity Stresses. Int. J. Mol. Sci. 2015, 16, 13561–13578. [Google Scholar] [CrossRef] [PubMed]
- Gupta, B.; Huang, B. Mechanism of salinity tolerance in plants: Physiological, biochemical, and molecular characterization. Int. J. Genom. 2014, 2014. [Google Scholar] [CrossRef]
- Bhattacharyya, A.; Chattopadhyay, R.; Mitra, S.; Crowe, S.E. Oxidative Stress: An Essential Factor in the Pathogenesis of Gastrointestinal Mucosal Diseases. Physiol. Rev. 2014, 94, 329–354. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, M.; Bhatti, B. Physiological responses of some salt tolerance and salt sensitive lines of wheat (Crthamus tinctorius L.). Acta Physiol. Plant. 1999, 117, 23–28. [Google Scholar]
- Adnan, A.; Erkan, Y. Effect of salinity stress on chlorophyll, carotenoid content, and proline in Salicornia prostrata Pall. and Suaeda prostrata Pall. subsp. prostrata (Amaranthaceae). Braz. J. Bot. 2016, 39, 101–106. [Google Scholar]
- Kamal, A.; Qureshi, M.S.; Ashraf, M.Y.; Hussain, M. Salinity induced some changes in growth and physio-chemical aspects of two soybean [Glycine max (L.) merr] genotype. Pak. J. Bot. 2003, 35, 93–97. [Google Scholar]
- Parvin, K.; Haque, M.N. Protective Role of Salicylic Acid on Salt Affected Broccoli Plants. J. Agric. Ecol. 2017, 10, 1–10. [Google Scholar] [CrossRef]
- Stepien, P.; Klobus, G. Antioxidant defense in the leaves of C3 and C4 plants under salinity stress. Physiol. Plant. 2005, 125, 31–40. [Google Scholar] [CrossRef]
- Chandrasekaran, M.; Kim, K.; Krishnamoorthy, R.; Walitang, D.; Sundaram, S.; Joe, M.M.; Selvakumar, G.; Hu, S.; Oh, S.H.; Sa, T. Mycorrhizal symbiotic efficiency on C3 and C4 plants under salinity stress—A meta-analysis. Front. Microbiol. 2016, 11, 1246. [Google Scholar] [CrossRef] [PubMed]
- Akbar, S.; Sultan, S. Soil bacteria showing a potential of chlorpyrifos degradation and plant growth enhancement. Braz. J. Microbiol. 2016, 47, 563–570. [Google Scholar] [CrossRef]
- Bhantana, P.; Lazarovitch, N. Evapotranspiration, Crop coefficient and growth of two young pomegranate (Punica granatum L.) varieties under salt stress. Agric. Water Manag. 2010, 97, 715–722. [Google Scholar] [CrossRef]
- Gorai, M.; Ennajeh, M.; Khemira, H.; Neffati, M. Combined effect of NaCl-salinity and hypoxia on growth, photosynthesis, water relations and solute accumulation in Phragmite saustralis plants. Flora 2010, 205, 462–470. [Google Scholar] [CrossRef]
- Baby, T.; Collins, C.; Tyerman, S.D.; Gilliham, M. Salinity negatively affects pollen tube growth and fruit set in grapevines and is not mitigated by silicon. Am. J. Enol. Vitic. 2016, 67, 218–228. [Google Scholar] [CrossRef]
- Van Zelm, E.; Zhang, Y.; Testerink, C. Salt tolerance mechanisms of plants. Ann. Rev. Plant Biol. 2020, 71, 403–433. [Google Scholar] [CrossRef] [PubMed]
- Aldesuquy, H.; Baka, Z.; Mickky, B. Kinetin and spermine mediated induction of salt tolerance in wheat plants: Leaf area, photosynthesis and chloroplast ultrastructure of flag leaf at ear emergence. Egypt. J. Basic Appl. Sci. 2014, 1, 77–87. [Google Scholar] [CrossRef]
- Szalai, G.; Janda, K.; Darkó, É.; Janda, T.; Peeva, V.; Pál, M. Comparative analysis of polyamine metabolism in wheat and maize plants. Plant Physiol. Biochem. 2017, 1, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Syvertsen, J.; Levy, Y. Salinity Interactions with Other Abiotic and Biotic Stresses in Citrus; University of Florida, Indian River Research and Education Center: Ft. Pierce, FL, USA, 2005. [Google Scholar]
- Negrão, S.; Schmöckel, S.M.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Moir-Barnetson, L.; Veneklaas, E.J.; Colmer, T.D. Salinity tolerances of three succulent halophytes (Tecticornia spp.) differentially distributed along a salinity gradient. Funct. Plant Biol. 2016, 4, 739–750. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Babar, M.A. The root growth of wheat plants, the water conservation and fertility status of sandy soils influenced by plant growth promoting rhizobacteria. Symbiosis 2017, 72, 195–205. [Google Scholar] [CrossRef]
- Bohnert, H.J.; Shen, B. Transformation and compatible solutes. Sci. Hortic. 1999, 78, 237–260. [Google Scholar] [CrossRef]
- Shuyskaya, E.V.; Rakhamkulova, Z.F.; Lebedeva, M.P.; Kolesnikov, A.V.; Safarova, A.; Borisochkina, T.I.; Toderich, K.N. Different mechanisms of ion homeostasis are dominant in the recretohalophyte Tamarix ramosissima under different soil salinity. Acta Physiol. Plant. 2017, 1, 81. [Google Scholar] [CrossRef]
- Hernandez, J.A.; Almansa, M.S. Short term effects of salt stress on antioxidant systems and leaf water relations of pea leaves. Physiol. Plant. 2002, 115, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Ali, S.; Zandi, P.; Mehmood, A.; Ullah, S.; Ikram, M.; Ismail, M.; Babar, M.A. Role of sugars, amino acids and organic acids in improving plant abiotic stress tolerance. Pak. J. Bot. 1997, 52, 2. [Google Scholar] [CrossRef]
- Hong, B.; Barg, R.; Ho, T.D. Developmental and organ specific expression of an ABA and stress induced protein in barley. Plant Mol. Biol. 1992, 18, 663–674. [Google Scholar] [CrossRef]
- Hare, P.D.; Cress, W.A.; Staden, J.V. Dissecting the roles of osmolyte accumulation during stress. Plant Cell Environ. 1998, 21, 535–554. [Google Scholar] [CrossRef]
- Galinski, E.A. Compatible solutes of halophilic eubacteria-eubacteria-molecular principles, water-solute interactions, stress protection. Cell Mol. Life Sci. 1993, 49, 487–496. [Google Scholar] [CrossRef]
- Lutts, S.; Lefèvre, I. How can we take advantage of halophyte properties to cope with heavy metal toxicity in salt-affected areas? Ann. Bot. 2015, 1, 509–528. [Google Scholar] [CrossRef]
- Brown, A.D. Microbial Water Stress Physiology, Principles and Perspectives; Wiley: Hoboken, NJ, USA, 1990. [Google Scholar]
- Solomon, A.; Beer, S.; Waisel, Y.; Jones, G.P.; Paleg, L.G. Effects of NaCl on the Carboxylating activity of Rubisco from Tamarix jordanis in the presence and absence of proline-related compatible solutes. Plant Physiol. 1994, 90, 198–204. [Google Scholar] [CrossRef]
- Papageorgiou, G.; Murata, N. The unusually strong stability effects of glycine betaine on the structure and function of the oxygen evolving photosystem II complex. Photosynth. Res. 1995, 44, 243–252. [Google Scholar] [CrossRef]
- Bose, J.; Rodrigo-Moreno, A.; Shabala, S. ROS homeostasis in halophytes in the context of salinity stress tolerance. J. Exp. Bot. 2014, 1, 1241–1257. [Google Scholar] [CrossRef]
- Stoop, J.M.H.; Williamson, J.D.; Pharr, D.M. Mannitol metabolism in plants: A method for coping with stress. Trends Plant Sci. 1996, 1, 139–144. [Google Scholar] [CrossRef]
- Moran, J.F.; Becana, M.; Iturbe-Ormaetxe, I.; Frechilla, S.; Klucas, R.V.; Aparicio-Tejo, P. Drought induces oxidative stress in pea plants. Planta 1994, 94, 346–352. [Google Scholar] [CrossRef]
- Managbanag, J.R.; Torzilli, A.P. An analysis of trehalose, glycerol, and mannitol accumulation during heat and salt stress in a salt marsh isolate of Aureobasidium pullulans. Mycologia 2002, 1, 384–391. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Zandi, P. Effects of exogenously applied plant growth regulators in combination with PGPR on the physiology and root growth of chickpea (Cicer arietinum) and their role in drought tolerance. J. Plant Interact. 2018, 1, 239–247. [Google Scholar] [CrossRef]
- Tipirdamaz, R.; Gagneul, D.; Duhazé, C.; Aïnouche, A.; Monnier, C.; Özkum, D.; Larher, F. Clustering of halophytes from an inland salt marsh in Turkey according to their ability to accumulate sodium and nitrogenous osmolytes. Environ. Exp. Bot. 2006, 57, 139–153. [Google Scholar] [CrossRef]
- Ashraf, M.; Harris, P.J.C. Potential biochemical indicators of salinity tolerance in plants. Plant Sci. 2004, 166, 3–16. [Google Scholar] [CrossRef]
- Sandhu, D.; Cornacchione, M.V.; Ferreira, J.F.; Suarez, D.L. Variable salinity responses of 12 alfalfa genotypes and comparative expression analyses of salt-response genes. Sci. Rep. 2017, 22, 42958. [Google Scholar] [CrossRef]
- Liang, X.; Zhang, L.; Natarajan, S.K.; Becker, D.F. Proline Mechanisms of Stress Survival. Antioxid. Redox Signal. 2013, 19, 998–1011. [Google Scholar] [CrossRef]
- Dar, M.I.; Naikoo, M.I.; Rehman, F.; Naushin, F.; Khan, F.A. Proline accumulation in plants: Roles in stress tolerance and plant development. In Osmolytes and Plants Acclimation to Changing Environment: Emerging Omics Technologies; Springer: New Delhi, India, 2016; pp. 155–166. [Google Scholar]
- Ali, B.; Hayat, S.; Ahmad, A. 28-homobrassinolide ameliorates the saline stress in Cicer arietinum L. Environ. Exp. Bot. 2007, 59, 217–223. [Google Scholar] [CrossRef]
- Mantilla, B.S.; Marchese, L.; Casas-Sánchez, A.; Dyer, N.A.; Ejeh, N.; Biran, M.; Silber, A.M. Proline Metabolism is Essential for Trypanosoma brucei brucei Survival in the Tsetse Vector. PLoS Pathog. 2017, 13, e1006158. [Google Scholar] [CrossRef]
- Kahlaoui, B.; Hachicha, M.; Misle, E.; Fidalgo, F.; Teixeira, J. Physiological and biochemical responses to the exogenous application of proline of tomato plants irrigated with saline water. J. Saudi Soc. Agric. Sci. 2018, 1, 17–23. [Google Scholar] [CrossRef]
- Nemoto, Y.; Sasakuma, T. Differential stress responses of early salt-stress responding genes in common wheat. Phytochemistry 2002, 61, 129–133. [Google Scholar] [CrossRef]
- Jaarsma, R.; de Vries, R.S.; de Boer, A.H. Effect of salt stress on growth, Na+ accumulation and proline metabolism in potato (Solanum tuberosum) cultivars. PLoS ONE 2013, 8, e60183. [Google Scholar] [CrossRef] [PubMed]
- Joseph, E.A.; Radhakrishnan, V.V.; Mohanan, K.V. A Study on the Accumulation of Proline—An Osmoprotectant Amino Acid under Salt Stress in Some Native Rice Cultivars of North Kerala, India. Univ. J. Agric. Res. 2015, 3, 15–22. [Google Scholar] [CrossRef]
- Knight, H.; Anthony, J.; Knight, M.R. Calcium signaling in Arabidopsis thaliana responding to drought and salinity. Plant J. 1997, 12, 1067–1078. [Google Scholar] [CrossRef]
- Misra, N.; Gupta, A.K. Interactive effects of sodium and calcium on proline metabolism in salt tolerant green gram cultivar. Am. J. Plant Physiol. 2006, 1, 1–2. [Google Scholar]
- Ashraf, M.; Foolad, M.R. Roles of glycinebetaine and proline in improving plant abiotic stress resistance. Environ. Exp. Bot. 2007, 59, 206–216. [Google Scholar] [CrossRef]
- Huang, Z.; Zhao, L.; Chen, D.; Liang, M.; Liu, Z.; Shao, H.; Long, X. Salt Stress Encourages Proline Accumulation by Regulating Proline Biosynthesis and Degradation in Jerusalem Artichoke Plantlets. PLoS ONE 2013, 8, e62085. [Google Scholar] [CrossRef]
- Lutts, S.; Majerus, V.; Kinet, J.M. NaCl effects on proline metabolism in rice (Oryza sativa) seedlings. Int. Rice Res. 1999, 25, 39–40. [Google Scholar] [CrossRef]
- Devnarain, N.; Crampton, B.G.; Chikwamba, R.K.; Becker, J.V.W.; O’Kennedy, M.M. Physiological responses of selected African sorghum landraces to progressive water stress and re-watering. S. Afr. J. 2016, 103, 61–69. [Google Scholar] [CrossRef]
- Yan, H.; Gang, L.Z.; Zhao, C.Y.; Guo, W.Y. Effects of exogenous proline on the physiology of soybean plantlets regenerated from embryo in vitro and on the ultra-structure of their mitochondria under NaCl stress. Soybean Sci. 2000, 19, 314–319. [Google Scholar]
- Hare, P.D.; Cress, W.A.; Staden, J.V. Disruptive effects of exogenous proline on chloroplast and mitochondrial ultrastructure in Arabidopsis leaves. S. Afr. J. 2002, 68, 393–396. [Google Scholar] [CrossRef]
- Zeyner, A.; Romanowski, K.; Vernunft, A.; Harris, P.; Müller, A.M.; Wolf, C.; Kienzle, E. Effects of Different Oral Doses of Sodium Chloride on the Basal Acid-Base and Mineral Status of Exercising Horses Fed Low Amounts of Hay. PLoS ONE 2017, 12, e0168325. [Google Scholar] [CrossRef] [PubMed]
- Jimenez-Bremont, J.F.; Becerra-Flora, A.; Hernandez-Lucero, E.; Rodriguez-Kessler, M.; Acosta-Gallegos, J.A.; Ramirez-Pimental, J.G. Proline accumulation in two bean cultivars under salt stress and the effect of polyamines and ornithine. Biol. Plant. 2006, 50, 763–766. [Google Scholar] [CrossRef]
- Gill, S.S.; Tuteja, N. Polyamines and abiotic stress tolerance in plants. Plant Signal Behav. 2010, 5, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Griffith, S.M.; Banowetz, G.M. Nitrogen nutrition and flowering. In Nitrogen Nutrition in Higher Plants; Srivastava, H.S., Singh, R.P., Eds.; Associated Publ. Co.: New Delhi, India, 1995; pp. 385–400. [Google Scholar]
- Kumar, R.; Khurana, A.; Sharma, A.K. Role of plant hormones and their interplay in development and ripening of fleshy fruits. J. Exp. Bot. 2013, 65, 4561–4575. [Google Scholar] [CrossRef]
- Diao, Q.; Song, Y.; Shi, D.; Qi, H. Interaction of Polyamines, Abscisic Acid, Nitric Oxide, and Hydrogen Peroxide under Chilling Stress in Tomato (Lycopersicon esculentum Mill.) Seedlings. Front. Plant Sci. 2017, 8, 203. [Google Scholar] [CrossRef] [PubMed]
- Shabala, S.; Cuin, T.A.; Pottosin, I. Polyamines prevent NaCl-induced K+ efflux from pea mesophyll by blocking non-selective cation channels. FEBS Lett. 2007, 581, 1993–1999. [Google Scholar] [CrossRef]
- Rodríguez, A.A.; Maiale, S.J.; Menéndez, A.B.; Ruiz, O.A. Polyamine oxidase activity contributes to sustain maize leaf elongation under saline stress. J. Exp. Bot. 2009, 1, 4249–4262. [Google Scholar] [CrossRef]
- Mishra, S.N.; Sharma, I. Putrescine as a growth inducer and as a source of nitrogen for mustard seedlings under sodium chloride salinity. Indian J. Exp. Biol. 1994, 32, 916–918. [Google Scholar]
- Lakra, N.; Tomar, P.C.; Mishra, S.N. Growth response modulation by putrescine in Indian mustard Brassica juncea L. under multiple stress. Indian J. Exp. Biol. 2016, 54, 262–270. [Google Scholar]
- Maruri-López, I.; Jiménez-Bremont, J. Hetero- and homodimerization of Arabidopsis thaliana arginine decarboxylase AtADC1 and AtADC2. Biochem. Biophys. Res. Commun. 2017, 11, 508–513. [Google Scholar] [CrossRef] [PubMed]
- Wani, S.H.; Singh, N.B.; Haribhushan, A.; Mir, J.I. Compatible solute engineering in plants for abiotic stress tolerance-role of glycine betaine. Curr. Genom. 2013, 14, 157. [Google Scholar] [CrossRef] [PubMed]
- Baloda, A.; Madanpotra, S.; Jaiwal, P.K. Transformation of mung bean plants for abiotic stress tolerance by introducing codA gene, for an osmoprotectant glycine betaine. J. Plant Stress Physiol. 2017, 3, 5–11. [Google Scholar] [CrossRef]
- Wei, D.; Zhang, W.; Wang, C.; Meng, Q.; Li, G.; Chen, T.H.; Yang, X. Genetic engineering of the biosynthesis of glycinebetaine leads to alleviate salt-induced potassium efflux and enhances salt tolerance in tomato plants. Plant Sci. 2017, 257, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Park, E.J.; Jeknic, Z.; Chen, T.H.H. Exogenous application of glycine betaine increases chilling tolerance in tomato plants. Plant Cell Physiol. 2006, 47, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Kim, W.Y.; Yun, D.J. A New Insight of Salt Stress Signaling in Plant. Mol. Cells 2016, 39, 447–459. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Bano, A. Rhizobacteria and Abiotic Stress Management. In Plant Growth Promoting Rhizobacteria for Sustainable Stress Management; Springer: Singapore, 2019; pp. 65–80. [Google Scholar]
- Mohammad, N.; Naeem, K.; Zakia, A.; Abdul, G. Genetic diversity and disease response of rust in bread wheat collected from Waziristan Agency, Pakistan. Int. J. Biodivers. Conserv. 2011, 3, 10–18. [Google Scholar]
- Shanker, A.; Venkateswarlu, B. Abiotic Stress in Plants: Mechanisms and Adaptations. BoD–Books on Demand; IntechOpen: Norderstedt, Germany, 2011; p. 22. [Google Scholar]
- Nawaz, K.; Ashraf, M. Exogenous application of glycine betaine modulates activities of antioxidants in maize plants subjected to salt stress. J. Agron. Crop Sci. 2009, 196, 28–37. [Google Scholar] [CrossRef]
- Annunziata, M.G.; Ciarmiello, L.F.; Woodrow, P.; Maximova, E.; Fuggi, A.; Carillo, P. Durum Wheat Roots Adapt to Salinity Remodeling the Cellular Content of Nitrogen Metabolites and Sucrose. Front. Plant Sci. 2016, 7, 2035. [Google Scholar] [CrossRef]
- Wyn-Jones, R.G.; Gorham, J.; McDonnell, E. Organic and inorganic solute contents as selection criteria for salt tolerance in the Triticeae. In Salinity Tolerance in Plants: Strategies for Crop Improvement; Staples, R., Toennissen, G.H., Eds.; Wiley and Sons: New York, NY, USA, 1984; pp. 189–203. [Google Scholar]
- Woodrow, P.; Ciarmiello, L.F.; Annunziata, M.G.; Pacifico, S.; Iannuzzi, F.; Mirto, A.; Carillo, P. Durum wheat seedling responses to simultaneous high light and salinity involve a fine reconfiguration of amino acids and carbohydrate metabolism. Plant Physiol. 2016. [Google Scholar] [CrossRef]
- Majeed, K.; Al-Hamzawi, A. Effect of Calcium Nitrate, Potassium Nitrate and Anfaton on Growth and Storability of Plastic Houses Cucumber (Cucumis sativus L. cv. Al-Hytham). Am. J. Plant Sci. 2010, 5, 278–290. [Google Scholar]
- Kong, X.; Luo, Z.; Dong, H.; Eneji, A.E.; Li, W. Effects of non-uniform root zone salinity on water use, Na+ recirculation, and Na+ and H+ flux in cotton. J. Exp. Bot. 2012, 63, 2105–2116. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Shabala, L.; Liu, X.; Azzarello, E.; Zhou, M.; Pandolfi, C.; Shabala, S. Linking salinity stress tolerance with tissue-specific Na+ sequestration in wheat roots. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef]
- Maleka, P.; Kontturi, M.; Pehu, B.; Somersalo, S. Photosynthesis response of drought and salt stressed tomato and turnip plants to foliar applied glycinebetaine. Physiol. Plant. 1999, 105, 45–50. [Google Scholar]
- Athar, H.U.R.; Zafar, Z.U.; Ashraf, M. Glycinebetaine Improved Photosynthesis in Canola under Salt Stress: Evaluation of Chlorophyll Fluorescence Parameters as Potential Indicators. J. Agron. Crop Sci. 2015, 201, 428–442. [Google Scholar] [CrossRef]
- Luo, J.Y.; Zhang, S.; Peng, J.; Zhu, X.Z.; Lv, L.M.; Wang, C.Y.; Cui, J.J. Effects of Soil Salinity on the Expression of Bt Toxin (Cry1Ac) and the Control Efficiency of Helicoverpa armigera in Field- Grown Transgenic Bt Cotton. PLoS ONE 2017, 12, e0170379. [Google Scholar] [CrossRef]
- Raza, M.A.S.; Shahid, A.M.; Saleem, M.F.; Khan, I.H.; Ahmad, S.; Ali, M.; Iqbal, R. Effects and management strategies to mitigate drought stress in oilseed rape (Brassica napus L.): A review. Zemdirbyste-Agriculture 2017, 104, 85–94. [Google Scholar] [CrossRef]
- Akram, R.; Fahad, S.; Hashmi, M.Z.; Wahid, A.; Adnan, M.; Mubeen, M.; Khan, N.; Rehmani, M.I.; Awais, M.; Abbas, M.; et al. Trends of electronic waste pollution and its impact on the global environment and ecosystem. Environ. Sci. Pollut. Res. Int. 2019, 1, 1–6. [Google Scholar] [CrossRef]
- Bhuiyan, M.S.; Maynard, G.; Raman, A.; Hodgkins, D.; Mitchell, D.; Nicol, H. Salt effects on proline and glycine betaine levels and photosynthetic performance in Melilotus siculus, Tecticornia pergranulata and Thinopyrum ponticum measured in simulated saline conditions. Funct. Plant Biol. 2016, 4, 254–265. [Google Scholar] [CrossRef]
- Demiral, T.; Türkan, I. Does exogenous glycinebetaine affect antioxidative system of rice seedlings under NaCl treatment? J. Plant Physiol. 2004, 18, 1089–1100. [Google Scholar] [CrossRef] [PubMed]
- Talaat, N.B.; Shawky, B.T. Influence of arbuscular mycorrhizae on yield, nutrients, organic solutes, and antioxidant enzymes of two wheat cultivars under salt stress. J. Soil Sci. Plant Nutr. 2011, 174, 283–291. [Google Scholar] [CrossRef]
- Ruiz-Lozano, J.M.; Porcel, R.; Azcón, C.; Aroca, R. Regulation by arbuscular mycorrhizae of the integrated physiological response to salinity in plants: New challenges in physiological and molecular studies. J. Exp. Bot. 2012, 28, 4033–4044. [Google Scholar] [CrossRef]
- Ghosh, B.; Ali, M.N.; Saikat, G. Response of Rice under Salinity Stress: A Review Update. J. Res. Rice 2016, 4, 1–8. [Google Scholar] [CrossRef]
- Mack, G.; Hoffmann, C.M.; Marlander, B. Nitrogen compounds in organs of two sugar beet genotypes (Beta vulgaris L.) during the season. Field Crop. Res. 2007, 102, 210–218. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Rahman, M.A.; Guo, J.; Kang, Z.; Babar, M.A. Comparative physiological and metabolic analysis reveals a complex mechanism involved in drought tolerance in chickpea (Cicer arietinum L.) induced by PGPR and PGRs. Sci. Rep. 2019, 9, 1–19. [Google Scholar] [CrossRef]
- Ashraf, M. Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol. Adv. 2009, 27, 84–93. [Google Scholar] [CrossRef] [PubMed]
- Turan, S.; Tripathy, B.C. Salt and genotype impact on antioxidative enzymes and lipid peroxidation in two rice cultivars during de-etiolation. Protoplasma 2013, 1, 209–222. [Google Scholar] [CrossRef]
- Das, M.K.; Roychoudhury, A. ROS and responses of antioxidant as ROS-scavengers during environmental stress in plants. Front. Environ. Sci. 2014, 2, 1–3. [Google Scholar] [CrossRef]
- Elstner, E.F. Mechanisms of oxygen activation in different compartments of plant cell. In Active Oxygen/Oxidative Stress and Plant Metabolism; Pell, E.J., Stefen, K.L., Eds.; American society of Plant Physiol: Rockville, MD, USA, 1991; pp. 13–25. [Google Scholar]
- Vardhini, B.V.; Anjum, N.A. Brassinosteroids make plant life easier under abiotic stresses mainly by modulating major components of antioxidant defense system. Front. Environ. Sci. 2015, 12, 67. [Google Scholar] [CrossRef]
- Rich, P.R.; Bonner, W.D., Jr. The sites of superoxide anion generation in higher plant mitochondria. Arch Biochem. Biophys. 1978, 188, 206–213. [Google Scholar] [CrossRef]
- Song, X.S.; Hu, W.H.; Mao, W.H.; Ogweno, J.O.; Zhou, Y.H.; Yu, J.Q. Response of ascorbate peroxidase isoenzymes and ascorbate regeneration system to abiotic stresses in Cucumis sativus L. Plant Physiol. Biochem. 2005, 1, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Mehmood, A.; Hussain, A.; Irshad, M.; Hamayun, M.; Iqbal, A.; Khan, N. In vitro production of IAA by endophytic fungus Aspergillus awamori and its growth promoting activities in Zea mays. Symbiosis 2019, 77, 225–235. [Google Scholar] [CrossRef]
- Xia, X.J.; Gao, C.J.; Song, L.X.; Zhou, Y.H.; Shi, K.A.I.; Yu, J.Q. Role of H2O2 dynamics in brassinosteroid-induced stomatal closure and opening in S olanum lycopersicum. Plant Cell Environ. 2014, 37, 2036–2050. [Google Scholar] [CrossRef] [PubMed]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 79–194. [Google Scholar] [CrossRef]
- Das, S.K.; Patra, J.K.; Thatoi, H. Antioxidative response to abiotic and biotic stresses in mangrove plants: A review. Intern. Rev. Hydrobiol. 2016, 101, 3–19. [Google Scholar] [CrossRef]
- Sairam, R.K.; Tyagi, A. Physiology and molecular biology of salinity stress tolerance in plants. Curr. Sci. 2004, 86, 407–421. [Google Scholar]
- Zhang, M.; Fang, Y.M.; Ji, Y.H.; Jiang, Z.P.; Wang, L. Effects of salt stress on ion content, antioxidant enzymes and protein profile in different tissues of Broussonetia Papyrifera. S. Afr. J. Bot. 2013, 85, 1–9. [Google Scholar] [CrossRef]
- Wahid, A.; Parveen, M.; Gelani, S.; Basra, S.M.A. Pretreatment of seed with H2O2 improves salt tolerance of wheat seedlings by alleviation of oxidative damage and expression of stress proteins. J. Plant Physiol. 2007, 164, 283–294. [Google Scholar] [CrossRef]
- Shalata, A.; Neumann, P.M. Exogenous ascorbic acid increases resistance to salt stress and reduces lipid peroxidation. J. Exp. Bot. 2001, 52, 2207–2211. [Google Scholar] [CrossRef]
- Khan, N.; Ali, S.; Shahid, M.A.; Kharabian-Masouleh, A. Advances in detection of stress tolerance in plants through metabolomics approaches. Plant Omics 2017, 10, 153. [Google Scholar] [CrossRef]
- Aliniaeifard, S.; Hajilou, J.; Tabatabaei, S.J.; Sifi-Kalhor, M. Effects of Ascorbic Acid and Reduced Glutathione on the Alleviation of Salinity Stress in Olive Plants. Int. J. Fruit Sci. 2016, 16, 395–409. [Google Scholar] [CrossRef]
- Ding, X.; Jiang, Y.; He, L.; Zhou, Q.; Yu, J.; Hui, D.; Huang, D. Exogenous glutathione improves high root-zone temperature tolerance by modulating photosynthesis, antioxidant and osmolytes systems in cucumber seedlings. Sci. Rep. 2016, 6, 35424. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, B. Interactive Effects of Silicon and Potassium Nitrate in Improving Salt Tolerance of Wheat. J. Integr. Agric. 2013. [Google Scholar] [CrossRef]
- Parihar, P.; Singh, S.; Singh, R.; Singh, V.P.; Prasad, S.M. Effect of salinity stress on plants and its tolerance strategies: A review. Environ. Sci. Pollut. Res. 2015, 22, 4056–4075. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.; Qi, H.; Qiao, W.; Shi, J.; Xu, Q.; Zhou, H.; Yan, G.; Huang, Q. Cotton (Gossypium hirsutum L.) genotypes with contrasting K+/Na+ ion homeostasis: Implications for salinity tolerance. Acta Physiol. Plant. 2017, 1, 77. [Google Scholar] [CrossRef]
- Walia, H.; Wilson, C.; Condamine, P.; Liu, X.; Ismail, A.M.; Close, T.J. Large scale expression profiling and physiological characterization of Jasmonic acid-mediated adaptation of barley to salinity stress. Plant Cell Environ. 2007, 30, 410–421. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Z.; Guo, J.; Zhu, A.; Zhang, L.; Zhang, M. Exogenous jasmonic acid can enhance tolerance of wheat seedlings to salt stress. Ecotoxicol. Environ. Saf. 2014, 1, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, P.; Rasool, S.; Gul, A.; Sheikh, S.A.; Akram, N.A.; Ashraf, M.; Kazi, A.M.; Gucel, S. Jasmonates: Multifunctional Roles in Stress Tolerance. Front. Plant Sci. 2016, 7, 813. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Babar, M.A. Metabolic and physiological changes induced by plant growth regulators and plant growth promoting rhizobacteria and their impact on drought tolerance in Cicer arietinum L. PLoS ONE 2019, 14, e0213040. [Google Scholar] [CrossRef] [PubMed]
- Wani, S.H.; Kumar, V.; Shriram, V.; Sah, S.K. Phytohormones and their metabolic engineering for abiotic stress tolerance in crop plants. Crop J. 2016, 4, 162–176. [Google Scholar] [CrossRef]
- Seif El-Yazal, S.A.; Seif El-Yazal, M.A.; Dwidar, E.F.; Rady, M.M. Phytohormone crosstalk research: Cytokinin and its crosstalk with other phytohormones. Curr. Protein Pept. Sci. 2015, 16, 395–405. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Bano, A.; Ali, S.; Babar, M.A. Crosstalk amongst phytohormones from planta and PGPR under biotic and abiotic stresses. Plant Growth Regul. 2020, 90, 189–203. [Google Scholar] [CrossRef]
- Nafisi, M.; Fimognari, L.; Sakuragi, Y. Interplays between the cell wall and phytohormones in interaction between plants and necrotrophic pathogens. Phytochemistry 2015, 1, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Aldesuquy, H.S.; Gaber, A.M. Effect of growth regulators on leaf area, pigment content and photosynthetic activity on Vicia faba plants irrigated by sea water. Biol. Plant. 1993, 35, 519–527. [Google Scholar] [CrossRef]
- Babar, S.; Siddiqi, E.H.; Hussain, I.; Hayat Bhatti, K.; Rasheed, R. Mitigating the effects of salinity by foliar application of salicylic acid in fenugreek. Physiol. J. 2014, 2014. [Google Scholar] [CrossRef]
- Vishwakarma, K.; Upadhyay, N.; Kumar, N.; Yadav, G.; Singh, J.; Mishra, R.K.; Sharma, S. Abscisic Acid Signaling and Abiotic Stress Tolerance in Plants: A Review on Current Knowledge and Future Prospects. Front. Plant Sci. 2017, 8, 161. [Google Scholar] [CrossRef]
- Mehmood, A.; Hussain, A.; Irshad, M.; Khan, N.; Hamayun, M.; Ismail; Afridi, S.G.; Lee, I.J. IAA and flavonoids modulates the association between maize roots and phytostimulant endophytic Aspergillus fumigatus greenish. J. Plant Interact. 2018, 1, 532–542. [Google Scholar] [CrossRef]
- Barnawal, D.; Bharti, N.; Tripathi, A.; Pandey, S.S.; Chanotiya, C.S.; Kalra, A. ACC-deaminase-producing endophyte Brachybacterium paraconglomeratum strain SMR20 ameliorates Chlorophytum salinity stress via altering phytohormone generation. J. Plant Growth Reg. 2016, 35, 553–564. [Google Scholar] [CrossRef]
- Beaudoin, N.; Serizet, C.; Gosti, F.; Giraudat, J. Interaction between abscisic acid and ethylene signalling cascades. Plant Cell. 2000, 2, 1103–1115. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A.; Rahman, M.A.; Rathinasabapathi, B.; Babar, M.A. UPLC-HRMS-based untargeted metabolic profiling reveals changes in chickpea (Cicer arietinum) metabolome following long-term drought stress. Plant Cell Environ. 2019, 42, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Anuradha, S.; Rao, S.S.R. Effect of brassinosteroids on salinity stress induced inhibition of seed germination and seedling growth of rice (Oryza sativa L.). Plant Growth Regul. 2001, 33, 151–153. [Google Scholar] [CrossRef]
- Sharma, I.; Ching, E.; Saini, S.; Bhardwaj, R.; Pati, P.K. Exogenous application of brassinosteroid offers tolerance to salinity by altering stress responses in rice variety Pusa Basmati-1. Plant Physiol. Biochem. 2013, 69, 17–26. [Google Scholar] [CrossRef]
- Anuradha, S.; Rao, S.S.R. Application of brassinosteroids to rice seeds (Oryza sativa L.) reduced the impact of salt stress on growth, prevented photosynthetic pigments loss and increased nitrate reductase activity. Plant Growth Regul. 2003, 40, 29–32. [Google Scholar] [CrossRef]
- Shahid, M.A.; Balal, R.M.; Khan, N.; Simón-Grao, S.; Alfosea-Simón, M.; Cámara-Zapata, J.M.; Mattson, N.S.; Garcia-Sanchez, F. Rootstocks influence the salt tolerance of Kinnow mandarin trees by altering the antioxidant defense system, osmolyte concentration, and toxic ion accumulation. Sci. Hortic. 2019, 10, 1. [Google Scholar] [CrossRef]
- Shahid, M.A.; Balal, R.M.; Khan, N.; Zotarelli, L.; Liu, G.D.; Sarkhosh, A.; Fernández-Zapata, J.C.; Nicolás, J.J.; Garcia-Sanchez, F. Selenium impedes cadmium and arsenic toxicity in potato by modulating carbohydrate and nitrogen metabolism. Ecotoxicol. Environ. Saf. 2019, 30, 588–599. [Google Scholar] [CrossRef] [PubMed]
- Houimli, S.I.M.; Denden, M.; El-Hadj, S.B. Induction of salt tolerance in pepper (Capsicum annum) by 24-epibrassinolide. Eurasian J. Biosci. 2008, 2, 83–90. [Google Scholar]
- Thussagunpanit, J.; Jutamanee, K.; Sonjaroon, W.; Kaveeta, L.; Chai-Arree, W.; Pankean, P.; Suksamrarn, A. Effects of brassinosteroid and brassinosteroid mimic on photosynthetic efficiency and rice yield under heat stress. Photosynth 2015, 1, 312–320. [Google Scholar] [CrossRef]
- Qayyum, B.; Shabbaz, M.; Akram, N.A. Interachive effect of foliar application of 24—Epibrassinolide and root zone salinity on morpho – physiological attributes of wheat (triticum aestivum L.). Int. J. Agric. Biol. 2007, 9, 584–589. [Google Scholar]
- Hairat, S.; Khurana, P. Improving Photosynthetic Responses during Recovery from Heat Treatments with Brassinosteroid and Calcium Chloride in Indian Bread Wheat Cultivars. Am. J. Plant Sci. 2015, 6, 1827–1849. [Google Scholar] [CrossRef]
- Amzallag, G.N. Brassinosteroids as metahormones evidences from specific influence during critical period in sorghum development. Plant Biol. 2002, 4, 656–663. [Google Scholar] [CrossRef]
- Gruszka, D.; Janeczko, A.; Dziurka, M.; Pociecha, E.; Oklestkova, J.; Szarejko, I. Barley Brassinosteroid Mutants Provide an Insight into Phytohormonal Homeostasis in Plant Reaction to Drought Stress. Front. Plant Sci. 2016, 7, 1824. [Google Scholar] [CrossRef]
- Kamuro, Y.; Takatsuto, S. Practical application of brassinosteroids in agricultural fields. In Brassinosteroids: Steroidal Plant Hormones; Sakurai, A., Yokota, T., Clouse, S.D., Eds.; Springer: Tokyo, Japan, 1999; pp. 223–241. [Google Scholar]
- Ali, B.; Hasan, S.A.; Hayat, S.; Hayat, Q.; Yadav, S.; Fariduddin, Q.; Ahmad, A. A role for brassinosteroids in the amelioration of aluminium stress through antioxidant system in mung bean (Vigna radiata L. Wilczek). Environ. Exp. Bot. 2008, 62, 153–159. [Google Scholar] [CrossRef]
- Hasan, S.A.; Hayat, S.; Ali, B.B.; Ahmad, A. 28-Homobrassinolide protects chickpea (Cicer arietinum) from cadmium toxicity by stimulating antioxidants. Environ. Pollut. 2008, 151, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Rady, M.M.; Osman, A.S. Response of growth and antioxidant system of heavymetal-contaminated tomato plants to 24-epibrassinolide. Afr. J. Agric. Res. 2012, 7, 3249–3254. [Google Scholar]
- Jain, M.; Mathur, G.; Koul, S.; Sarin, N. Ameliorative effects of proline on salt induced lipid peroxidation in Cell lines of groundnut (Arachis hypogea L.). Plant Cell Rep. 2001, 20, 463–468. [Google Scholar] [CrossRef]
- Abdullahi, B.A.; Gu, X.; Gan, Q.; Yang, Y. Brassinolide amelioration of aluminum toxicity in mung bean seedling growth. J. Plant Nutr. 2003, 26, 1725–1734. [Google Scholar] [CrossRef]
- Martínez-Cuenca, M.R.; Primo-Capella, A.; Forner-Giner, M.A. Influence of Rootstock on Citrus Tree Growth: Effects on Photosynthesis and Carbohydrate Distribution, Plant Size, Yield, Fruit Quality, and Dwarfing Genotypes. Plant Growth 2016, 16, 107. [Google Scholar]
- Khelil, A.; Menu, T.; Ricard, B. Adaptive response to salt involving carbohydrate metabolism in leaves of a salt-sensitive tomato cultivars. Plant Physiol. Biochem. 2007, 45, 551–559. [Google Scholar] [CrossRef]
- Bartels, D.; Sunkar, R. Drought and salt tolerance in plants. Crit. Rev. Plant Sci. 2005, 24, 23–58. [Google Scholar] [CrossRef]
- Hoekstra, F.A.; Golovina, E.A.; Buitkink, J. Mechanisms of plant desiccation tolerance. Trends Plant Sci. 2001, 6, 431–438. [Google Scholar] [CrossRef]
- Pierik, R.; Christa, T. The Art of Being Flexible: How to Escape from Shade, Salt, and Drought. Top. Rev. Plast. 2014, 166, 5–22. [Google Scholar] [CrossRef] [PubMed]
- Libal-Wekseler, J.; Fernak, B.; Michael, S.T.; Zhu, J.K. Carbohydrate metabolism is affected in response to salt stress. Horticulture 1997, 20, 1043–1045. [Google Scholar]
- Khan, N.; Bano, A. Role of plant growth promoting rhizobacteria and Ag-nano particle in the bioremediation of heavy metals and maize growth under municipal wastewater irrigation. Int. J. Phytorem. 2016, 18, 211–221. [Google Scholar] [CrossRef]
- Schreiber, L. Transport barriers made of cutin, suberin and associated waxes. Trends Plant Sci. 2010, 15, 546–553. [Google Scholar] [CrossRef] [PubMed]
- Lux, A.; Šottníková, A.; Opatrná, J.; Greger, M. Differences in structure of adventitious roots in Salix clones with contrasting characteristics of cadmium accumulation and sensitivity. Physiol. Plant. 2004, 120, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Shabala, S. Learning from halophytes: Physiological basis and strategies to improve abiotic stress tolerance in crops. Ann. Bot. 2013, 112, 1209–1221. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Broadley, M.R. Chloride in soil and its uptake and movement within the plant. A review. Ann. Bot. 2001, 88, 967–988. [Google Scholar] [CrossRef]
- Grattan, S.R.; Grieve, C.M. Mineral nutrient acquisition and response by plants grown in saline environments. In Hand Book of Plant and Crop Stress; Pessarakli, M., Ed.; Marcel Dekker: New York, NY, USA, 1999; pp. 203–299. [Google Scholar]
- Ikram, M.; Ali, N.; Jan, G.; Guljan, F.; Khan, N. Endophytic fungal diversity and their interaction with plants for agriculture sustainability under stressful condition. Recent Pat. Food Nutr. Agric. 2019, 12. [Google Scholar] [CrossRef]
- Marzec, M.; Muszynska, A.; Gruszka, D. The Role of Strigolactones in Nutrient-Stress Responses in Plants. Int. J. Mol. Sci. 2013, 14, 9286–9304. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.; Khan, N.; Nouroz, F.; Erum, S.; Nasim, W.; Shahid, M.A. In vitro effects of GA 3 on morphogenesis of CIP potato explants and acclimatization of plantlets in field. In Vitro Cell. Dev. Biol. 2018, 54, 104–111. [Google Scholar] [CrossRef]
- Wang, M.; Zheng, Q.; Shen, Q.; Guo, S. The critical role of potassium in plant stress response. Int. J. Mol. Sci. 2013, 14, 7370–7390. [Google Scholar] [CrossRef]
- Ali, S.; Khan, N.; Nouroz, F.; Erum, S.; Nasim, W.A. Effects of sucrose and growth regulators on the microtuberization of cip potato (Solanum tuberosum) germplasm. Pak. J. Bot. 2018, 1, 763–768. [Google Scholar]
- Maathuis, F.J.M.; Ahmad, I.; Patishtan, J. Regulation of Na+ fluxes in plants. Front. Plant Sci. 2014, 5, 467. [Google Scholar] [CrossRef] [PubMed]
- Conde, A.; Chaves, M.M.; Gerós, H. Membrane transport, sensing and signaling in plant adaptation to environmental stress. Plant Cell Physiol. 2011, 1, 1583–1602. [Google Scholar] [CrossRef]
- Wyn Jones, R.G.; Pollard, A. Proteins, enzymes and inorganic ions. In Encyclopedia of Plant Physiology; New Series; Lauchli, A., Person, A., Eds.; Springer: New York, NY, USA, 1983; Volume 15B, pp. 528–562. [Google Scholar]
- Wang, H.; Wu, Z.; Chen, Y.; Yang, C.; Shi, D. Effects of salt and alkali stresses on growth and ion balance in rice (Oryza sativa L.). Plant Soil Environ. 2011, 8, 286–294. [Google Scholar] [CrossRef]
- Rossi, L.; Francini, A.; Minnocci, A.; Sebastiani, L. Salt stress modifies apoplastic barriers in olive (Olea europaea L.): A comparison between a salt-tolerant and a salt-sensitive cultivar. Sci. Hortic. 2015, 192, 38–46. [Google Scholar] [CrossRef]
- Krishnamurthy, P.; Ranathunge, K.; Nayak, S.; Schreiber, L.; Mathew, M.K. Root apoplastic barriers block Na+ transport to shoots in rice (Oryza sativa L.). J. Exp. Bot. 2011, 62, 4215–4228. [Google Scholar] [CrossRef]
- Kosová, K.; Vítámvás, P.; Urban, M.O.; Prášil, I.T. Plant proteome responses to salinity stress–comparison of glycophytes and halophytes. Funct. Plant Biol. 2013, 23, 775–786. [Google Scholar] [CrossRef] [PubMed]
- Moya, J.L.; Primo-Millo, E.; Talon, M. Morphological factors determining salt tolerance in citrus seedlings: The shoot to root ratio modulates passive root uptake of chloride ions and their accumulation in leaves. Plant Cell Environ. 1999, 22, 1425–1433. [Google Scholar] [CrossRef]
- Hussain, M.; Park, H.W.; Farooq, M.; Jabran, K.; Lee, D.J. Morphological and physiological basis of salt resistance in different rice genotypes. Int. J. Agric. Biol. 2013, 15, 113–118. [Google Scholar]
- Carden, D.E.; Walker, J.D.; Flowers, T.J.; Miller, A.J. Single cell measurement of the contributions of cytosolic Na+ and K+ to salt tolerance. Plant Physiol. 2003, 131, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Mahmud, S.; Sharmin, S.; Chowdhury, B.L.; Hossain, M.A. Research Article Effect of Salinity and Alleviating Role of Methyl Jasmonate in Some Rice Varieties. Asian J. Plant Sci. 2017, 16, 87–93. [Google Scholar] [CrossRef]
- Manaa, A.; Ben Ahmed, H.; Valot, B.; Bouchet, J.P.; Aschi-Smiti, S.; Causse, M.; Faurobert, M. Salt and genotype impact on plant physiology and root proteome variations in tomato. J. Exp. Bot. 2011, 62, 2797–2813. [Google Scholar] [CrossRef] [PubMed]
- Akca, Y.; Samsunlu, E. The effect of salt stress on growth, chlorophyll content, proline and nutrient accumulation and K/Na ratio in walnut. Pak. J. Bot. 2012, 44, 1513–1520. [Google Scholar]
- Hakim, M.A.; Juraimi, A.S.; Hanafi, M.M.; Ismail, M.R.; Rafii, M.Y.; Islam, M.M.; Selamat, A. The effect of salinity on growth, ion accumulation and yield of rice varieties. J. Anim. Plant Sci. 2014, 24, 874–885. [Google Scholar]
- Deinlein, U.; Stephan, A.B.; Horie, T.; Luo, W.; Xu, G.; Schroeder, J.I. Plant salt-tolerance mechanisms. Trends Plant. Sci. 2014, 19, 371–379. [Google Scholar] [CrossRef]
- Adabnejad, H.; Kavousi, H.R.; Hamidi, H.; Tavassolian, I. Assessment of the vacuolar Na+/H+ antiporter (NHX1) transcriptional changes in Leptochloa fusca L. in response to salt and cadmium stresses. Mol. Biol. Res. Commun. 2015, 4, 133–142. [Google Scholar] [PubMed]
- Guyton, A.C.; Hall, J.E. Textbook of Medical Physiology; Saunders: Philadelphia, PA, USA, 1986; p. 20. [Google Scholar]
- Kumari, P.H.; Kumar, S.A.; Sivan, P.; Katam, R.; Suravajhala, P.; Rao, K.S.; Kishor, P.B.K. Overexpression of a Plasma Membrane Bound Na+/H+ Antiporter-Like Protein (SbNHXLP) Confers Salt Tolerance and Improves Fruit Yield in Tomato by Maintaining Ion Homeostasis. Front. Plant Sci. 2016, 7, 2027. [Google Scholar] [CrossRef] [PubMed]
- Volkov, V. Salinity tolerance in plants. Quantitative approach to ion transport starting from halophytes and stepping to genetic and protein engineering for manipulating ion fluxes. Front. Plant Sci. 2015, 6, 873. [Google Scholar] [CrossRef]
- Veraplakorn, V.; Nanakorn, M.; Kaveeta, L.; Srisom, S.; Bennett, I.J. Variation in ion accumulation as a measure of salt tolerance in seedling and callus of Stylosanthes guianensis. Exp. Plant Physiol. 2013, 25, 106–115. [Google Scholar] [CrossRef][Green Version]
- Mukhtar, I.; Shahid, M.A.; Khan, M.W.; Balal, R.M.; Iqbal, M.M.; Naz, T.; Zubair, M.; Ali, H.H. Improving salinity tolerance in chili by exogenous application of calcium and sulphur. Soil Environ. 2016, 1, 35. [Google Scholar]
- Bao, A.; Zhao, Z.; Ding, G.; Shi, L.; Xu, F.; Cai, H. The Stable Level of Glutamine synthetase 2 Plays an Important Role in Rice Growth and in Carbon-Nitrogen Metabolic Balance. Int. J. Mol. Sci. 2015, 16, 12713–12736. [Google Scholar] [CrossRef]
- Bohnert, H.J.; Sheveleva, E. Plant stress adaptations-making metabolism move. Curr. Opin. Biol. 1998, 1, 267–274. [Google Scholar] [CrossRef]
- Bonales-Alatorre, E.; Pottosin, I.; Shabala, L.; Chen, Z.H.; Zeng, F.; Jacobsen, S.E.; Shabala, S. Differential Activity of Plasma and Vacuolar Membrane Transporters Contributes to Genotypic Differences in Salinity Tolerance in a Halophyte Species, Chenopodium quinoa. Int. J. Mol. Sci. 2013, 14, 9267–9285. [Google Scholar] [CrossRef] [PubMed]
- Cherel, L. Regulation of K+ channel activities in plants: From physiological to molecular aspects. J. Exp. Bot. 2004, 55, 337–351. [Google Scholar] [CrossRef] [PubMed]
- Shabala, S.; Pottosin, I. Regulation of potassium transport in plants under hostile conditions: Implications for abiotic and biotic stress tolerance. Physiol. Plant. 2014, 151, 257–279. [Google Scholar] [CrossRef] [PubMed]
- Humphries, E.S.A.; Dart, C. Neuronal and Cardiovascular Potassium Channels as Therapeutic Drug Targets: Promise and Pitfalls. J. Biomol. Screen. 2015, 20, 1055–1073. [Google Scholar] [CrossRef] [PubMed]
- Machnicka, B.; Czogalla, A.; Hryniewicz-Jankowska, A.; Bogusławska, D.M.; Grochowalska, R.; Heger, E.; Sikorski, A.F. Spectrins: A structural platform for stabilization and activation of membrane channels, receptors and transporters. Biochim. Biophys. Acta 2014, 1, 620–634. [Google Scholar] [CrossRef]
- Tavakoli, M.; Poustini, K.; Besharati, H.; Ali, S. Variable salinity responses of 25 alfalfa genotypes and comparative salt-response ion distribution. Russ. J. Plant Physiol. 2019, 66, 231–239. [Google Scholar] [CrossRef]
- Carillo, P.; Parisi, D.; Woodrow, P.; Pontecorvo, G.; Massaro, G.; Annunziata, M.G.; Fuggi, A.; Sulpice, R. Salt-induced accumulation of glycine betaine is inhibited by high light in durum wheat. Funct. Plant Biol. 2011, 22, 139–150. [Google Scholar] [CrossRef]
- Awada, S.; Campbell, W.F.L.; Dudle, M.; Jurinak, J.J. Interactive effects of sodium chloride, sodium sulfate, calcium sulfate and calcium chloride on snapbean growth, photosynthesis and ion uptake. J. Plant Nutr. 1995, 18, 889–900. [Google Scholar] [CrossRef]
- Multari, S.; Stewart, D.; Russell, W.R. Potential of Fava Bean as Future Protein Supply to Partially Replace Meat Intake in the Human Diet. Compr. Rev. Food Sci. 2015, 14, 511–522. [Google Scholar] [CrossRef]
- Hura, T.; Szewczyk-Taranek, B.; Hura, K.; Nowak, K.; Pawłowska, B. Physiological Responses of Rosa rubiginosa to Saline Environment. Water Air Soil Pollut. 2017, 228, 81. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gowayed, M.H.; Al-Zahrani, H.S.; Metwali, E.M. Improving the salinity tolerance in potato (Solanum tuberosum) by exogenous application of silicon dioxide nanoparticles. Int. J. Agric. Biol. 2017, 1, 19. [Google Scholar]
- Pessarakli, M.; Haghighi, M.; Sheibanirad, A. Plant Responses under Environmental Stress Conditions. Adv. Plants Agric. Res. 2015, 2, 00073. [Google Scholar] [CrossRef]
- Debouba, M.; Gouia, A.; Suzuki, M.H.; Ghorbel, M. NaCl stress effects on enzymes involved in nitrogen assimilation pathway in tomato “Lycopersicon esculentum” seedlings. J. Plant Physiol. 2006, 163, 1247–1258. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, M.; Guo, R.; Shi, D.; Liu, B.; Lin, X.; Yang, C. Effects of slat stress on ion balance and nitrogen metabolism of old and young leaves in rice (Oryza sativa L.). BMC Plant Biol. 2012, 12, 194. [Google Scholar] [CrossRef]
- Pandey, M.; Srivastava, A.K.; D’Souza, S.F.; Penna, S. Thiourea, a ROS Scavenger, Regulates Source-to-Sink Relationship to Enhance Crop Yield and Oil Content in Brassica juncea (L.). PLoS ONE 2013, 8, e73921. [Google Scholar] [CrossRef]
- Koksal, N.; Alkan-Torun, A.; Kulahlioglu, I.; Ertargin, E.; Karalar, E. Ion uptake of marigold under saline growth conditions. Springer Plus. 2016, 5, 139. [Google Scholar] [CrossRef]
- Khan, N.; Bano, A. Role of PGPR in the Phytoremediation of Heavy Metals and Crop Growth under Municipal Wastewater Irrigation. In Phytoremediation; Springer: Cham, Switzerland, 2018; pp. 135–149. [Google Scholar]
- Pravin, V.; Rosazlin, A.; Tumirah, K.; Salmah, I.; Amru, N.B. Role of Plant Growth Promoting Rhizobacteria in Agricultural Sustainability—A Review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef]
- Pal, D.; Khozin-Goldberg, I.; Cohen, Z.; Boussiba, S. The effect of light, salinity, and nitrogen availability on lipid production by Nannochloropsis sp. Appl. Microbiol. Biotech. 2011, 90, 1429–1441. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Hou, Z.; Wu, L.; Liang, Y.; Wei, C. Effects of salinity and nitrogen on cotton growth in arid environment. Plant Soil. 2010, 326, 61–73. [Google Scholar] [CrossRef]
- Seemann, J.R.; Thomas, D.S. Salinity and nitrogen effects on photosynthesis, ribulose-1, 5-bisphosphate carboxylase and metabolite pool sizes in Phaseolus vulgaris L. Plant Physiol. 1986, 82, 555–560. [Google Scholar] [CrossRef] [PubMed]
- Giblin, A.E.; Weston, N.B.; Banta, G.T.; Tucker, J.; Hopkinson, C.S. The effects of salinity on nitrogen losses from an oligohaline estuarine sediment. Estuaries Coasts 2010, 33, 1054–1068. [Google Scholar] [CrossRef]
- Laura, R.D. Salinity and nitrogen mineralization in soil. Soil Biol. Bioch. 1977, 9, 333–336. [Google Scholar] [CrossRef]
- Villa-Castorena, M.; Ulery, A.L.; Catalán-Valencia, E.A.; Remmenga, M.D. Salinity and nitrogen rate effects on the growth and yield of chile pepper plants. Soil Sci. Soc. Am. J. 2003, 67, 1781–1789. [Google Scholar] [CrossRef]
- Kahn, A.E.; Durako, M.J. Thalassia testudinum seedling responses to changes in salinity and nitrogen levels. J. Exp. Mar. Biol. Ecol. 2006, 335, 1–12. [Google Scholar] [CrossRef]
- Zhang, D.; Li, W.; Xin, C.; Tang, W.; Eneji, A.E.; Dong, H. Lint yield and nitrogen use efficiency of field-grown cotton vary with soil salinity and nitrogen application rate. Field Crops Res. 2012, 138, 63–70. [Google Scholar] [CrossRef]
- Al-Harbi, A.R.; Wahb-Allah, M.A.; Abu-Muriefah, S.S. Salinity and nitrogen level affects germination, emergence, and seedling growth of tomato. Int. J. Veg. Sci. 2008, 14, 380–392. [Google Scholar] [CrossRef]
- Akhtar, M.; Hussain, F.; Ashraf, M.Y.; Qureshi, T.M.; Akhter, J.; Awan, A.R. Influence of salinity on nitrogen transformations in soil. Commun. Soil Sci. Plant Anal. 2012, 43, 1674–1683. [Google Scholar] [CrossRef]
- Esmaili, E.; Kapourchal, S.A.; Malakouti, M.J.; Homaee, M. Interactive effect of salinity and two nitrogen fertilizers on growth and composition of sorghum. Plant Soil Environ. 2008, 54, 537–546. [Google Scholar] [CrossRef]
- Azizian, A.; Sepaskhah, A.R. Maize response to different water, salinity and nitrogen levels: Agronomic behavior. Int. J. Plant Prod. 2014, 8, 107–130. [Google Scholar]
- Tang, Z.; Liu, Y.; Guo, X.; Zu, Y. The combined effects of salinity and nitrogen forms on Catharanthus roseus: The role of internal ammonium and free amino acids during salt stress. J. Plant Nutr. Soil Sci. 2011, 174, 135–144. [Google Scholar] [CrossRef]
- Garg, N.; Chandel, S. The effects of salinity on nitrogen fixation and trehalose metabolism in mycorrhizal Cajanus cajan (L.) millsp. plants. J. Plant Growth Reg. 2011, 30, 490–503. [Google Scholar] [CrossRef]
- Navarro, J.M.; Botella, M.A.; Cerda, A.; Martinez, V. Phosphorus uptake and translocation in salt stressed melon plants. J. Plant Physiol. 2001, 158, 375–381. [Google Scholar] [CrossRef]
- Zakery-Asl, M.A.; Bolandnazar, S.; Oustan, S. Effect of salinity and nitrogen on growth, sodium, potassium accumulation, and osmotic adjustment of halophyte Suaeda aegyptiaca (Hasselq.) Zoh. Arch. Agron. Soil Sci. 2014, 60, 785–792. [Google Scholar] [CrossRef]
- Zeng, W.Z.; Xu, C.; Wu, J.W.; Huang, J.S.; Zhao, Q.; Wu, M.S. Impacts of salinity and nitrogen on the photosynthetic rate and growth of sunflowers (Helianthus annuus L.). Pedosphere 2014, 24, 635–644. [Google Scholar] [CrossRef]
- Cardeñosa, V.; Medrano, E.; Lorenzo, P.; Sánchez-Guerrero, M.C.; Cuevas, F.; Pradas, I.; Moreno-Rojas, J.M. Effects of salinity and nitrogen supply on the quality and health-related compounds of strawberry fruits (Fragaria× ananassa cv. Primoris). J. Sci. Food Agric. 2015, 95, 2924–2930. [Google Scholar] [CrossRef] [PubMed]
- Min, W.; Guo, H.; Zhang, W.; Zhou, G.; Ma, L.; Ye, J.; Liang, Y.; Hou, Z. Response of soil microbial community and diversity to increasing water salinity and nitrogen fertilization rate in an arid soil. Acta Agric. Scand. Sect. B Soil Plant. Sci. 2016, 66, 117–126. [Google Scholar] [CrossRef]
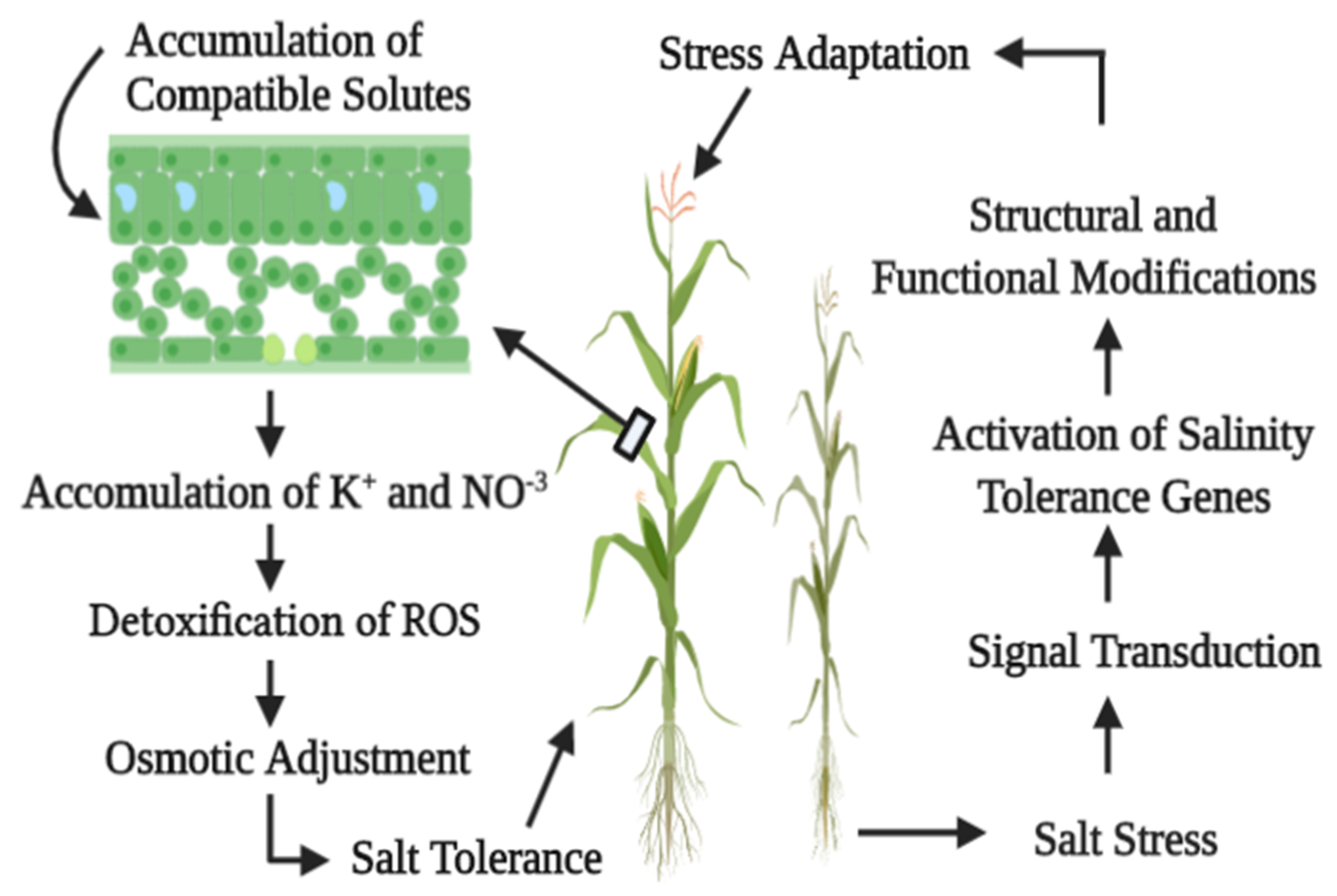
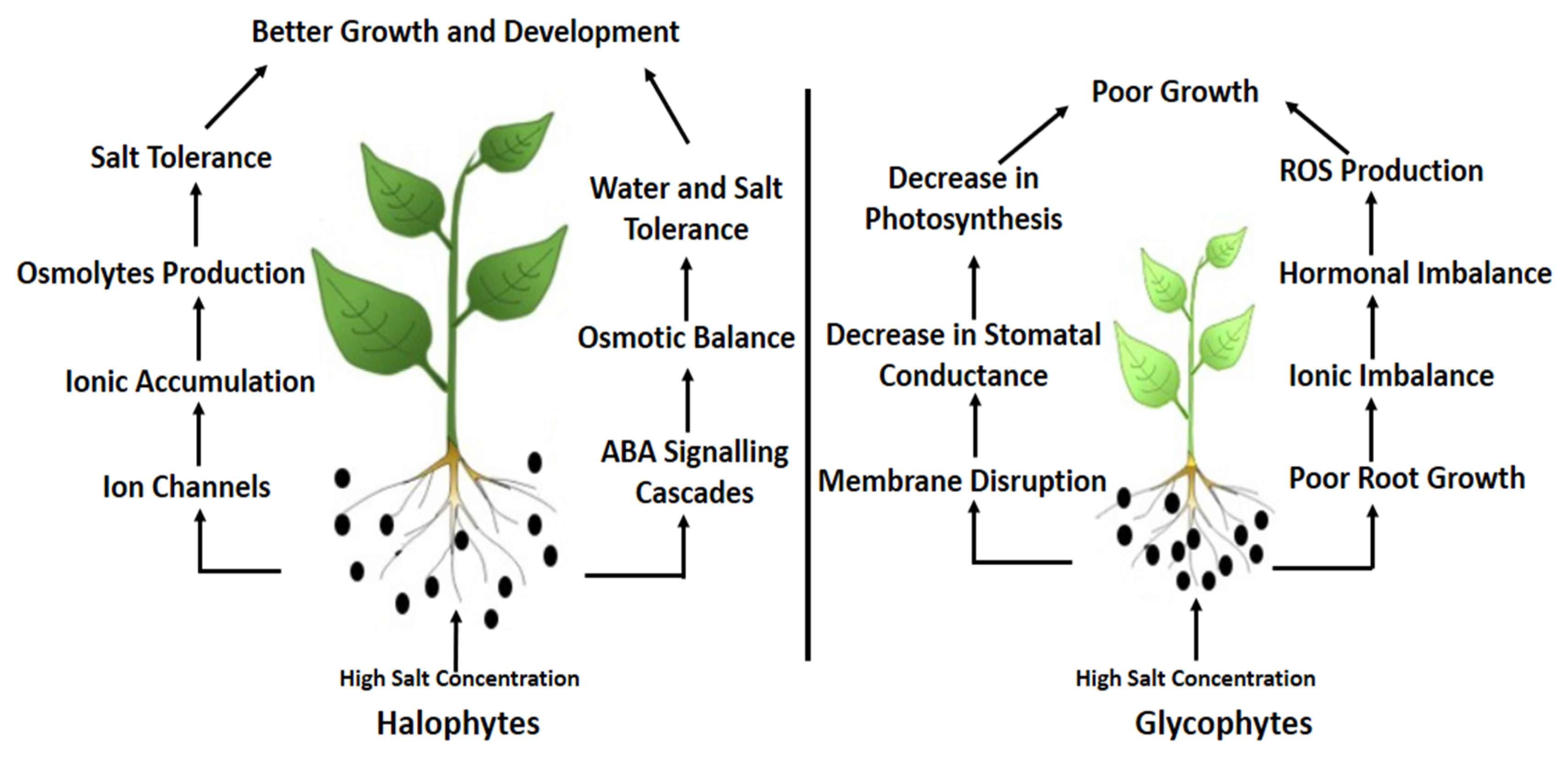

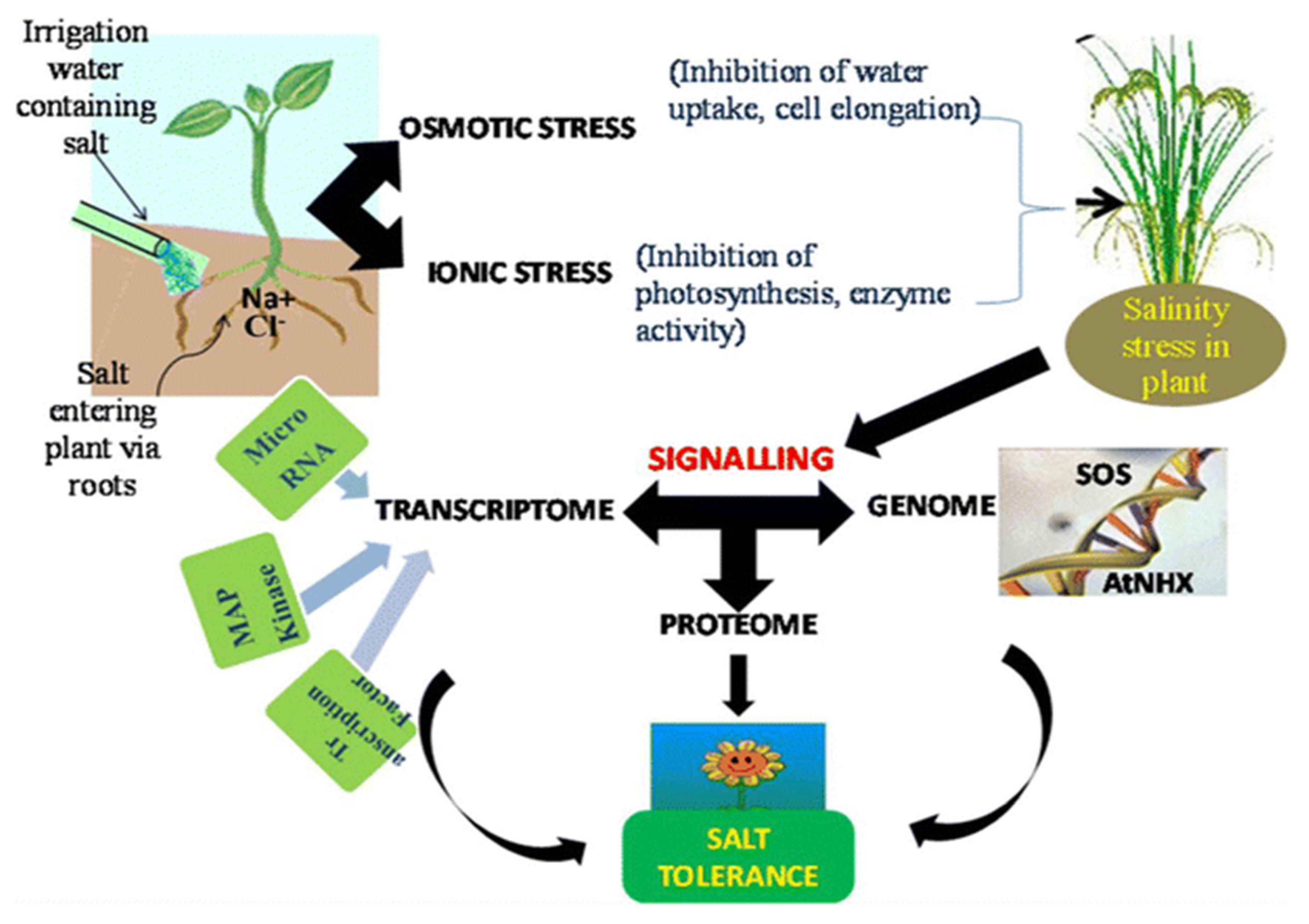

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahid, M.A.; Sarkhosh, A.; Khan, N.; Balal, R.M.; Ali, S.; Rossi, L.; Gómez, C.; Mattson, N.; Nasim, W.; Garcia-Sanchez, F. Insights into the Physiological and Biochemical Impacts of Salt Stress on Plant Growth and Development. Agronomy 2020, 10, 938. https://doi.org/10.3390/agronomy10070938
Shahid MA, Sarkhosh A, Khan N, Balal RM, Ali S, Rossi L, Gómez C, Mattson N, Nasim W, Garcia-Sanchez F. Insights into the Physiological and Biochemical Impacts of Salt Stress on Plant Growth and Development. Agronomy. 2020; 10(7):938. https://doi.org/10.3390/agronomy10070938
Chicago/Turabian StyleShahid, Muhammad Adnan, Ali Sarkhosh, Naeem Khan, Rashad Mukhtar Balal, Shahid Ali, Lorenzo Rossi, Celina Gómez, Neil Mattson, Wajid Nasim, and Francisco Garcia-Sanchez. 2020. "Insights into the Physiological and Biochemical Impacts of Salt Stress on Plant Growth and Development" Agronomy 10, no. 7: 938. https://doi.org/10.3390/agronomy10070938
APA StyleShahid, M. A., Sarkhosh, A., Khan, N., Balal, R. M., Ali, S., Rossi, L., Gómez, C., Mattson, N., Nasim, W., & Garcia-Sanchez, F. (2020). Insights into the Physiological and Biochemical Impacts of Salt Stress on Plant Growth and Development. Agronomy, 10(7), 938. https://doi.org/10.3390/agronomy10070938








