A Novel New Light Recipe Significantly Increases the Growth and Yield of Sweet Basil (Ocimum basilicum) Grown in a Plant Factory System
Abstract
1. Introduction
2. Materials and Methods
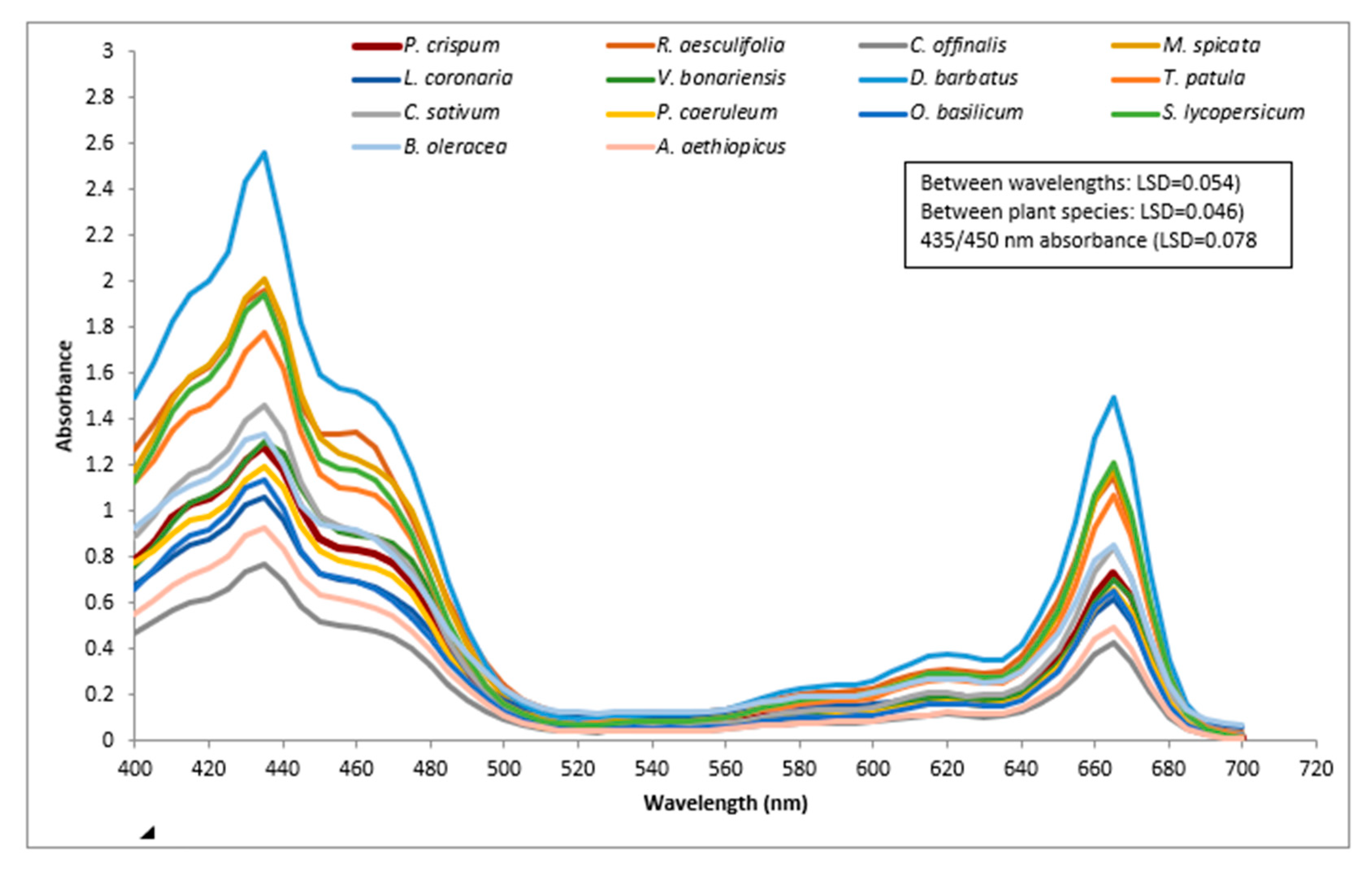
2.1. Absorption Spectra of 14 Plant Species between 400 and 700 nm
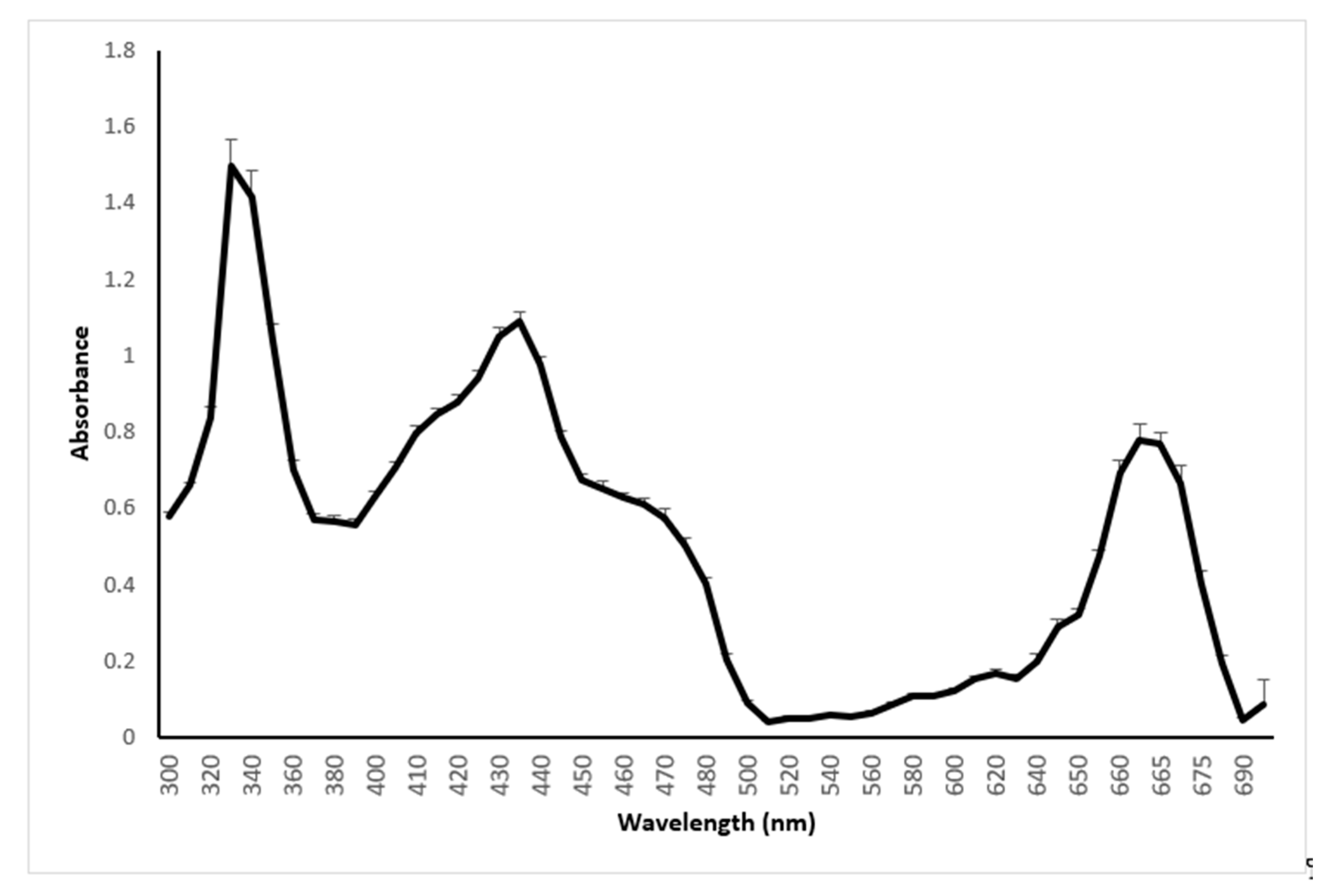
2.2. Absorption Spectrum of Sweet Basil between 300 and 700 nm
2.3. Growth of Sweet Basil under Different Lighting Regimes
2.4. Essential Oil Content
2.5. Light Use Efficiency (LUE)
2.6. Statistical Analysis
3. Results
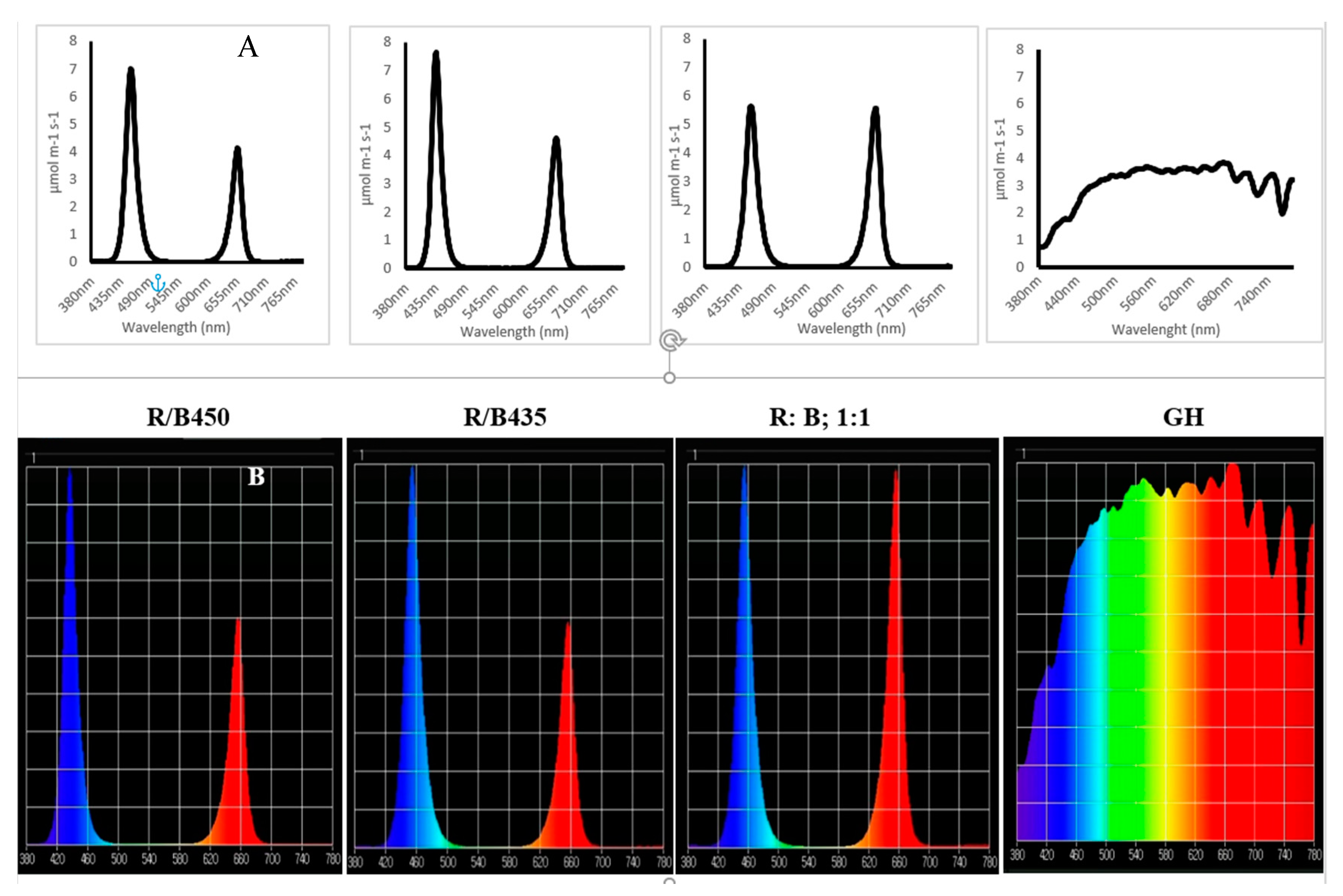
3.1. Light Spectrum Analyzed
3.1.1. Spectrum Analysis (between 400 and 700 nm) of 14 Plant Species
3.1.2. Spectrum Analysis (between 300 and 700 nm) of Sweet Basil (Ocimum basilicum)
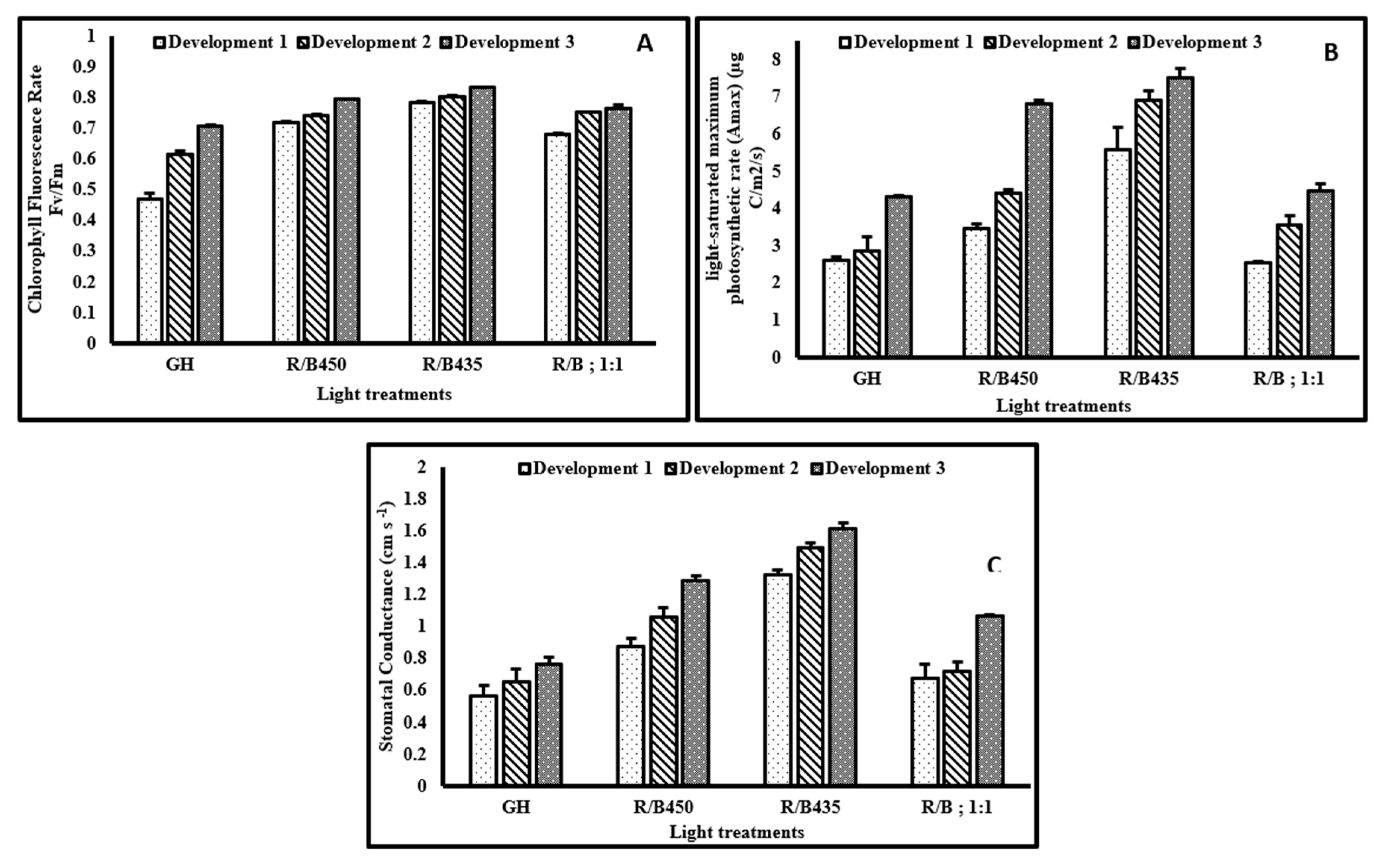
3.2. Physiological Responses
3.2.1. Chlorophyll Fluorescence Rate Fv/Fm
3.2.2. Light Saturated Maximum Photosynthetic Rate (Amax)
3.2.3. Stomatal Conductance (Gs)
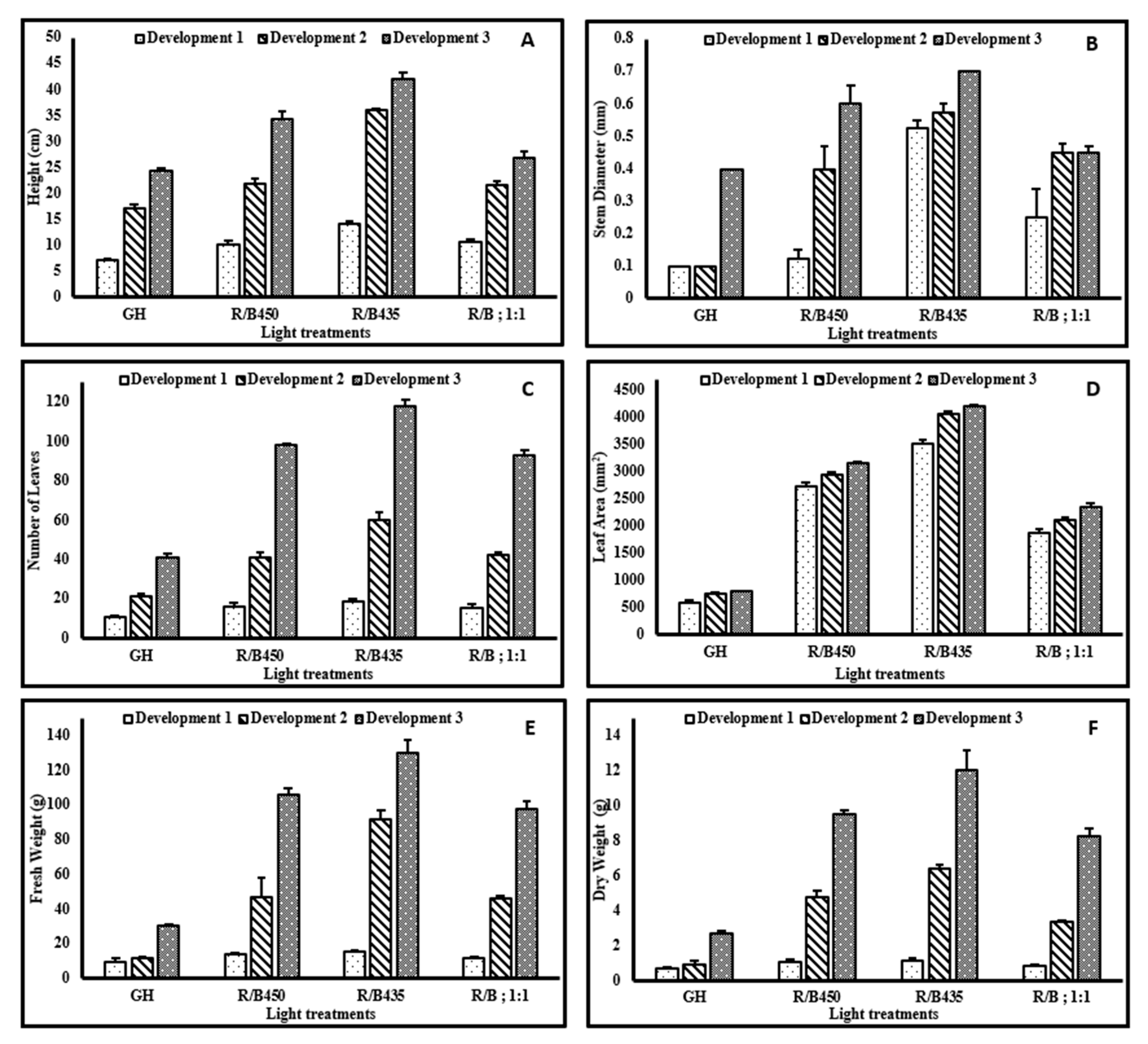
3.3. Growth Responses
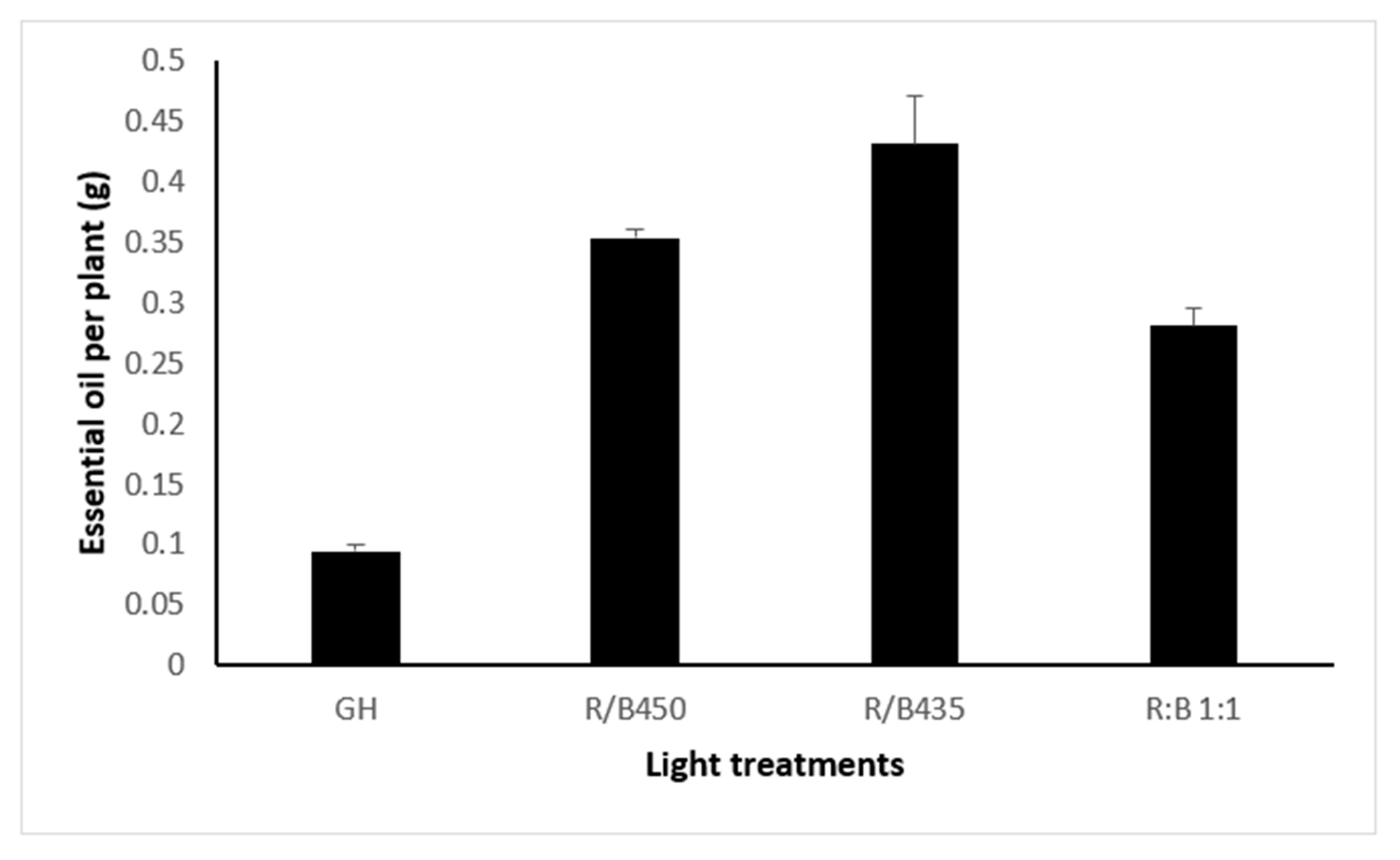
3.4. Essential Oil Yield
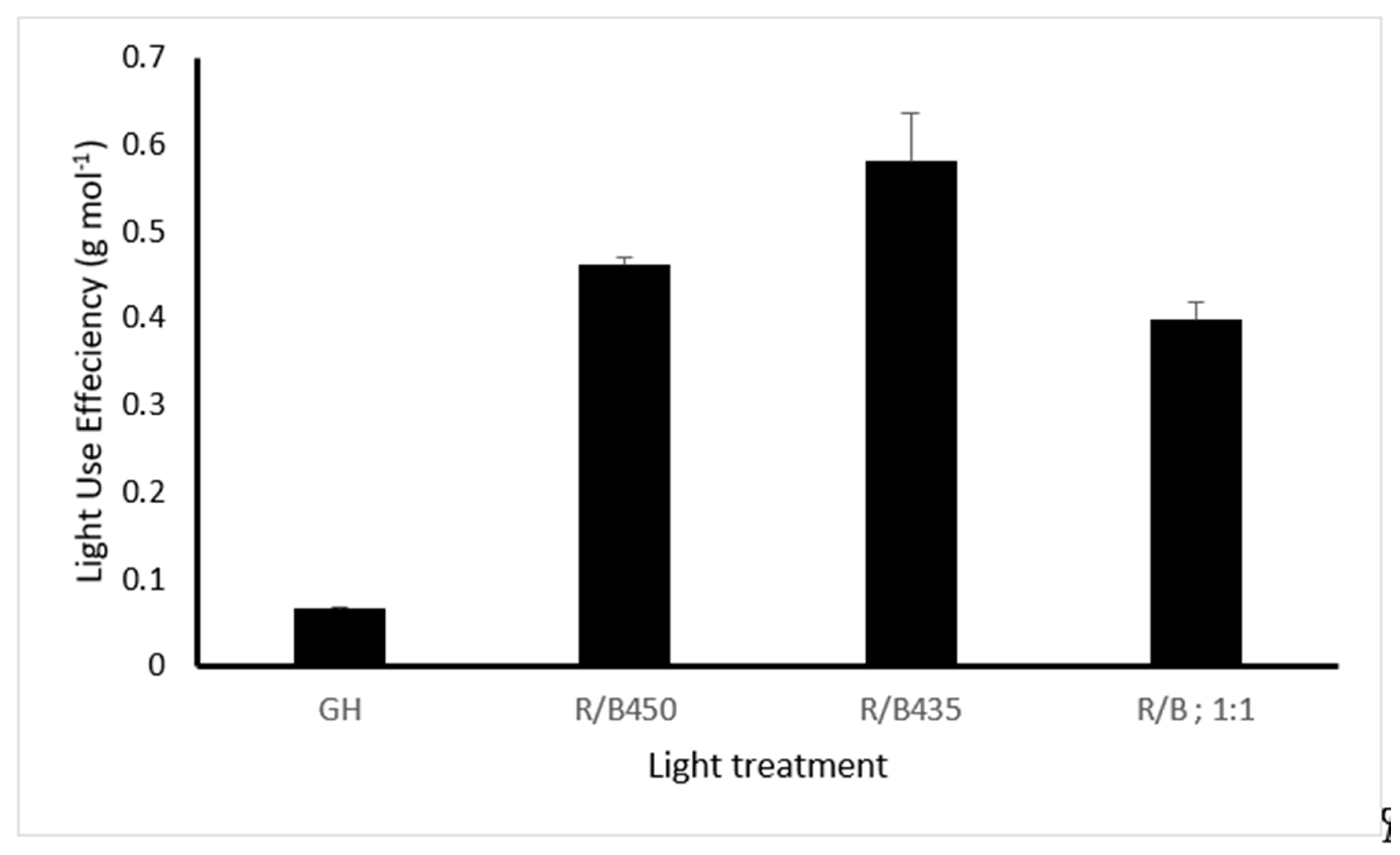
3.5. Light Use Efficiency (LUE)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Makri, O.; Kintzios, S. Ocimum sp. (Basil): Botany, Cultivation, Pharmaceutical Properties, and Biotechnology. J. Herbs Spices Med. Plants 2008, 13, 123–150. [Google Scholar] [CrossRef]
- Dube, S.; Upadhyay, P.; Tripathi, S. Antifungal, physicochemical, and insect-repelling activity of the essential oil of Ocimum basilicum. Can. J. Bot. 1989, 67, 2085–2087. [Google Scholar] [CrossRef]
- Hossain, M.A.; Kabir, M.; Salehuddin, S.; Rahman, S.M.; Das, A.; Singha, S.K.; Alam, M.K.; Rahman, A. Antibacterial properties of essential oils and methanol extracts of sweet basil Ocimum basilicum occurring in Bangladesh. Pharm. Biol. 2010, 48, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Simon, J.E.; Quinn, J.; Murray, R.G. Basil: A source of essential oils. Adv. New Crop. imber Press Portland OR (1990). 1990, 484–489. [Google Scholar]
- Kathirvel, P.; Ravi, S. Chemical composition of the essential oil from basil (Ocimum basilicum Linn.) and its in vitro cytotoxicity against HeLa and HEp-2 human cancer cell lines and NIH 3T3 mouse embryonic fibroblasts. Nat. Prod. Res. 2012, 26, 1112–1118. [Google Scholar] [CrossRef]
- Dasgupta, T.; Rao, A.; Yadava, P. Chemomodulatory efficacy of basil leaf (Ocimum basilicum) on drug metabolizing and antioxidant enzymes, and on carcinogen-induced skin and forestomach papillomagenesis. Phytomedicine 2004, 11, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Holm, Y. Bioactivity of Basil. In Basil; CRC Press: Boca Raton, FL, USA, 1999; pp. 117–139. [Google Scholar]
- Lee, S.-J.; Umano, K.; Shibamoto, T.; Lee, K.-G. Identification of volatile components in basil (Ocimum basilicum L.) and thyme leaves (Thymus vulgaris L.) and their antioxidant properties. Food Chem. 2005, 91, 131–137. [Google Scholar] [CrossRef]
- Murugan, K.; Murugan, P.; Noortheen, A. Larvicidal and repellent potential of Albizzia amara Boivin and Ocimum basilicum Linn against dengue vector, Aedes aegypti (Insecta: Diptera: Culicidae). Bioresour. Technol. 2007, 98, 198–201. [Google Scholar] [CrossRef] [PubMed]
- Sajjadi, S.E. Analysis of the essential oils of two cultivated basil (Ocimum basilicum L.) from Iran. DARU J. Pharm. Sci. 2006, 14, 128–130. [Google Scholar]
- Avetisyan, A.; Markosian, A.; Petrosyan, M.; Sahakyan, N.; Babayan, A.; Aloyan, S.; Trchounian, A. Chemical composition and some biological activities of the essential oils from basil Ocimum different cultivars. BMC Complement. Altern. Med. 2017, 17, 60. [Google Scholar] [CrossRef]
- Smith, R.; Adams, T.; Doull, J.; Feron, V.; Goodman, J.; Marnett, L.; Portoghese, P.; Waddell, W.; Wagner, B.; Rogers, A. Safety assessment of allylalkoxybenzene derivatives used as flavouring substances—Methyl eugenol and estragole. Food Chem. Toxicol. 2002, 40, 851–870. [Google Scholar] [CrossRef]
- Wallace, L.A.; Nelson, W.C.; Raymer, J.H.; Thomas, K.W. Identification of Polar Volatile Organic Compounds in Consumer Products and Common Microenvironments; US Environmental Protection Agency, Office of Research and Development: Washington, WA, USA, 1991.
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Nair, G.B.; Dhoble, S.J. A perspective perception on the applications of light-emitting diodes. Luminescence. 2015, 30, 1167–1175. [Google Scholar] [CrossRef]
- Green, M.A.; Zhao, J.; Wang, A.; Reece, P.J.; Gal, M. Efficient silicon light-emitting diodes. Nature 2001, 412, 805. [Google Scholar] [CrossRef]
- Van Ieperen, W.; Trouwborst, G. The application of LEDs as assimilation light source in greenhouse horticulture: A simulation study. In Proceedings of the International Symposium on High Technology for Greenhouse System Management, Naples, Italy, 4–6 October 2007; pp. 1407–1414. [Google Scholar]
- Tonzani, S. Lighting technology: Time to change the bulb. Nat. News 2009, 459, 312–314. [Google Scholar] [CrossRef]
- Singh, D.; Basu, C.; Meinhardt-Wollweber, M.; Roth, B. LEDs for energy efficient greenhouse lighting. Renew. Sustain. Energy Rev. 2015, 49, 139–147. [Google Scholar] [CrossRef]
- Bosma, R.H.; Lacambra, L.; Landstra, Y.; Perini, C.; Poulie, J.; Schwaner, M.J.; Yin, Y. The financial feasibility of producing fish and vegetables through aquaponics. Aquac. Eng. 2017, 78, 146–154. [Google Scholar] [CrossRef]
- Dickinson, J. Inventory of New York City Greenhouse Gas Emissions; DIANE Publishing: Collindale, PA, USA, 2007. [Google Scholar]
- Brown, C.S.; Schuerger, A.C.; Sager, J.C. Growth and photomorphogenesis of pepper plants under red light-emitting diodes with supplemental blue or far-red lighting. J. Am. Soc. Hortic. Sci. 1995, 120, 808–813. [Google Scholar] [CrossRef] [PubMed]
- Lin, K.-H.; Huang, M.-Y.; Huang, W.-D.; Hsu, M.-H.; Yang, Z.-W.; Yang, C.-M. The effects of red, blue, and white light-emitting diodes on the growth, development, and edible quality of hydroponically grown lettuce (Lactuca sativa L. var. capitata). Sci. Hortic. 2013, 150, 86–91. [Google Scholar] [CrossRef]
- Goins, G.D.; Yorio, N.; Sanwo, M.; Brown, C. Photomorphogenesis, photosynthesis, and seed yield of wheat plants grown under red light-emitting diodes (LEDs) with and without supplemental blue lighting. J. Exp. Bot. 1997, 48, 1407–1413. [Google Scholar] [CrossRef]
- Kim, H.-H.; Goins, G.D.; Wheeler, R.M.; Sager, J.C. Green-light supplementation for enhanced lettuce growth under red-and blue-light-emitting diodes. HortScience 2004, 39, 1617–1622. [Google Scholar] [CrossRef]
- Ohashi-Kaneko, K.; Takase, M.; Kon, N.; Fujiwara, K.; Kurata, K. Effect of light quality on growth and vegetable quality in leaf lettuce, spinach and komatsuna. Environ. Control Biol. 2007, 45, 189–198. [Google Scholar] [CrossRef]
- Kook, K.K.H.-S.; Kim, H.-S.K.K. The Effect of Blue-light-emitting Diodes on Antioxidant Properties and Resistance to Botrytis cinerea in Tomato. J. Plant Pathol. Microbiol. 2013, 4. [Google Scholar] [CrossRef]
- Wu, M.-C.; Hou, C.-Y.; Jiang, C.-M.; Wang, Y.-T.; Wang, C.-Y.; Chen, H.-H.; Chang, H.-M. A novel approach of LED light radiation improves the antioxidant activity of pea seedlings. Food Chem. 2007, 101, 1753–1758. [Google Scholar] [CrossRef]
- McCree, K.J. Test of current definitions of photosynthetically active radiation against leaf photosynthesis data. Agric. Meteorol. 1972, 10, 443–453. [Google Scholar] [CrossRef]
- Gallo, K.; Daughtry, C. Techniques for Measuring Intercepted and Absorbed Photosynthetically Active Radiation in Corn Canopies 1. Agron. J. 1986, 78, 752–756. [Google Scholar] [CrossRef]
- Britton, C.; Dodd, J. Relationships of photosynthetically active radiation and shortwave irradiance. Agric. Meteorol. 1976, 17, 1–7. [Google Scholar] [CrossRef]
- Massa, G.D.; Kim, H.-H.; Wheeler, R.M.; Mitchell, C.A. Plant productivity in response to LED lighting. HortScience 2008, 43, 1951–1956. [Google Scholar] [CrossRef]
- Carvalho, S.D.; Schwieterman, M.L.; Abrahan, C.E.; Colquhoun, T.A.; Folta, K.M. Light quality dependent changes in morphology, antioxidant capacity, and volatile production in sweet basil (Ocimum basilicum). Front. Plant Sci. 2016, 7, 1328. [Google Scholar] [CrossRef]
- Goto, E. Plant production in a closed plant factory with artificial lighting. In Proceedings of the VII International Symposium on Light in Horticultural Systems 956, Wageningen, The Netherlands, 14 October 2012; pp. 37–49. [Google Scholar]
- Eye Hortilux. The McCree Curve | EYE Hortilux. Available online: https://eyehortilux.com/grower-education/how-light-affects-plants/the-mccree-curve/ (accessed on 4 October 2019).
- Ögren, E.; Evans, J. Photosynthetic light-response curves. Planta 1993, 189, 182–190. [Google Scholar] [CrossRef]
- Gross, L.J. Photosynthetic dynamics in varying light environments: A model and its application to whole leaf carbon gain. Ecology 1982, 63, 84–93. [Google Scholar] [CrossRef]
- Terashima, I.; Fujita, T.; Inoue, T.; Chow, W.S.; Oguchi, R. Green light drives leaf photosynthesis more efficiently than red light in strong white light: Revisiting the enigmatic question of why leaves are green. Plant Cell Physiol. 2009, 50, 684–697. [Google Scholar] [CrossRef] [PubMed]
- Fan, X.-X.; Xu, Z.-G.; Liu, X.-Y.; Tang, C.-M.; Wang, L.-W.; Han, X.-l. Effects of light intensity on the growth and leaf development of young tomato plants grown under a combination of red and blue light. Sci. Hortic. 2013, 153, 50–55. [Google Scholar] [CrossRef]
- Lichtenthaler, H.; Buschmann, C.; Döll, M.; Fietz, H.-J.; Bach, T.; Kozel, U.; Meier, D.; Rahmsdorf, U. Photosynthetic activity, chloroplast ultrastructure, and leaf characteristics of high-light and low-light plants and of sun and shade leaves. Photosynth. Res. 1981, 2, 115–141. [Google Scholar] [CrossRef] [PubMed]
- Aldarkazali, M.; Rihan, H.Z.; Carne, D.; Fuller, M.P. The Growth and Development of Sweet Basil (Ocimum basilicum) and Bush Basil (Ocimum minimum) Grown under Three Light Regimes in a Controlled Environment. Agronomy 2019, 9, 743. [Google Scholar] [CrossRef]
- Saha, S.; Monroe, A.; Day, M.R. Growth, yield, plant quality and nutrition of basil (Ocimum basilicum L.) under soilless agricultural systems. Ann. Agric. Sci. 2016, 61, 181–186. [Google Scholar] [CrossRef]
- Burbott, A.J.; Loomis, W.D. Effects of light and temperature on the monoterpenes of peppermint. Plant Physiol. 1967, 42, 20–28. [Google Scholar] [CrossRef]
- Zhao, L. Steam Alternating Solvent Process. SPE Reserv. Eval. Eng. 2007, 10, 185–190. [Google Scholar] [CrossRef]
- Medlyn, B.E. Physiological basis of the light use efficiency model. Tree Physiol. 1998, 18, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Kozai, T. Resource use efficiency of closed plant production system with artificial light: Concept, estimation and application to plant factory. Proc. Jpn. Acad. Ser. B 2013, 89, 447–461. [Google Scholar] [CrossRef] [PubMed]
- Inada, K. Action spectra for photosynthesis in higher plants. Plant Cell Physiol. 1976, 17, 355–365. [Google Scholar]
- Mitchell, C.; Stutte, G. Sole-source lighting for controlled-environment agriculture. In Lighting up Profits Understanding Greenhouse Lighting, 2nd ed.; Meister Media Worldwide: Willoughby, OH, USA, 2004. [Google Scholar]
- Ustin, S.L.; Gitelson, A.A.; Jacquemoud, S.; Schaepman, M.; Asner, G.P.; Gamon, J.A.; Zarco-Tejada, P. Retrieval of foliar information about plant pigment systems from high resolution spectroscopy. Remote Sens. Environ. 2009, 113, S67–S77. [Google Scholar] [CrossRef]
- Kozai, T. Smart Plant Factory: The Next Generation Indoor Vertical Farms; Springer: Berlin, Germany, 2018. [Google Scholar]
- Kim, H.-H.; Wheeler, R.M.; Sager, J.C.; Yorio, N.C.; Goins, G.D. Light-emitting diodes as an illumination source for plants: A review of research at Kennedy Space Center. Habitation 2005, 10, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, G.; Blasioli, S.; Cellini, A.; Maia, L.; Crepaldi, A.; Braschi, I.; Spinelli, F.; Nicola, S.; Fernandez, J.A.; Stanghellini, C. Unravelling the role of red: Blue LED lights on resource use efficiency and nutritional properties of indoor grown sweet basil. Front. Plant Sci. 2019, 10, 305. [Google Scholar] [CrossRef] [PubMed]
- Schwend, T.; Prucker, D.; Peisl, S.; Nitsopoulos, A.; Mempel, H. The rosmarinic acid content of basil and borage correlates with the ratio of red and far-red light. Eur. J. Hortic. Sci. 2016, 81, 243–247. [Google Scholar] [CrossRef]
- Frąszczak, B.; Golcz, A.; Zawirska-Wojtasiak, R.; Janowska, B. Growth rate of sweet basil and lemon balm plants grown under fluorescent lamps and LED modules. Acta Sci. Pol. Hortorum Cultus 2014, 13, 3–13. [Google Scholar]
- Glowacka, B. Wpływ składu spektralnego światłana wzrost rozsady bazylii pospolitej (Ocimum basilicum L.), melisy lekarskiej (Melissa officinalis L.) i ogórecznika lekarskiego (Borago officinalis L.). Zesz. Probl. Postępów Nauk Rol. 2008, 527, 131–138. [Google Scholar]
- Jensen, N.B.; Clausen, M.R.; Kjaer, K.H. Spectral quality of supplemental LED grow light permanently alters stomatal functioning and chilling tolerance in basil (Ocimum basilicum L.). Sci. Hortic. 2018, 227, 38–47. [Google Scholar] [CrossRef]
- Kang, C.-Y.; Lian, H.-L.; Wang, F.-F.; Huang, J.-R.; Yang, H.-Q. Cryptochromes, phytochromes, and COP1 regulate light-controlled stomatal development in Arabidopsis. Plant Cell 2009, 21, 2624–2641. [Google Scholar] [CrossRef] [PubMed]
- Darko, E.; Heydarizadeh, P.; Schoefs, B.; Sabzalian, M.R. Photosynthesis under artificial light: The shift in primary and secondary metabolism. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130243. [Google Scholar] [CrossRef] [PubMed]
- Assmann, S.M.; Shimazaki, K. The multisensory guard cell. Stomatal responses to blue light and abscisic acid. Plant Physiol. 1999, 119, 809–816. [Google Scholar] [CrossRef] [PubMed]
- Doi, M.; Kitagawa, Y.; Shimazaki, K.-i. Stomatal blue light response is present in early vascular plants. Plant Physiol. 2015, 169, 1205–1213. [Google Scholar] [CrossRef]
- Inoue, S.-I.; Kinoshita, T. Blue Light Regulation of Stomatal Opening and the Plasma Membrane H+-ATPase. Plant Physiol. 2017, 174, 531–538. [Google Scholar] [CrossRef]
- Lurie, S. The effect of wavelength of light on stomatal opening. Planta 1978, 140, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Sharkey, T.D.; Raschke, K. Effect of light quality on stomatal opening in leaves of Xanthium strumarium L. Plant Physiol. 1981, 68, 1170–1174. [Google Scholar] [CrossRef] [PubMed]
- Lamb, J.J.; Røkke, G.; Hohmann-Marriott, M.F. Chlorophyll fluorescence emission spectroscopy of oxygenic organisms at 77 K. Photosynthetica 2018, 56, 105–124. [Google Scholar] [CrossRef]
- Balegh, S.; Biddulph, O. The photosynthetic action spectrum of the bean plant. Plant Physiol. 1970, 46, 1–5. [Google Scholar] [CrossRef]
- Roshchina, V.a.V. Fluorescing World of Plant Secreting Cells; Science Publishers: Rawalpindi, Pakistan, 2008. [Google Scholar]
- Hoffmann, A.M.; Noga, G.; Hunsche, M. High blue light improves acclimation and photosynthetic recovery of pepper plants exposed to UV stress. Environ. Exp. Bot. 2015, 109, 254–263. [Google Scholar] [CrossRef]
- Taulavuori, K.; Julkunen-Tiitto, R.; Hyöky, V.; Taulavuori, E. Blue Mood for Superfood. Nat. Prod. Commun. 2013, 8. [Google Scholar] [CrossRef]
- Amaki, W.; Yamazaki, N.; Ichimura, M.; Watanabe, H. Effects of Light Quality on the Growth and Essential Oil Content in Sweet Basil. Acta Hortic. 2011, 907, 91–94. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rihan, H.Z.; Aldarkazali, M.; Mohamed, S.J.; McMulkin, N.B.; Jbara, M.H.; Fuller, M.P. A Novel New Light Recipe Significantly Increases the Growth and Yield of Sweet Basil (Ocimum basilicum) Grown in a Plant Factory System. Agronomy 2020, 10, 934. https://doi.org/10.3390/agronomy10070934
Rihan HZ, Aldarkazali M, Mohamed SJ, McMulkin NB, Jbara MH, Fuller MP. A Novel New Light Recipe Significantly Increases the Growth and Yield of Sweet Basil (Ocimum basilicum) Grown in a Plant Factory System. Agronomy. 2020; 10(7):934. https://doi.org/10.3390/agronomy10070934
Chicago/Turabian StyleRihan, Hail Z., Mohammed Aldarkazali, Shiren J. Mohamed, Nancy B. McMulkin, Marwa H. Jbara, and Michael P. Fuller. 2020. "A Novel New Light Recipe Significantly Increases the Growth and Yield of Sweet Basil (Ocimum basilicum) Grown in a Plant Factory System" Agronomy 10, no. 7: 934. https://doi.org/10.3390/agronomy10070934
APA StyleRihan, H. Z., Aldarkazali, M., Mohamed, S. J., McMulkin, N. B., Jbara, M. H., & Fuller, M. P. (2020). A Novel New Light Recipe Significantly Increases the Growth and Yield of Sweet Basil (Ocimum basilicum) Grown in a Plant Factory System. Agronomy, 10(7), 934. https://doi.org/10.3390/agronomy10070934






