Influence of SDHI Seed Treatment on the Physiological Conditions of Spring Barley Seedlings under Drought Stress
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growing Conditions
2.2. Assessment of Barley Seeds Quality
2.3. Physiological State of Plants
2.3.1. Plant Photosynthesis
2.3.2. Plant Chlorophyll Fluorescence
2.4. Statistical Analysis
3. Results
3.1. Effect of the Seed Treatments on Germination Energy and Germination Capacity
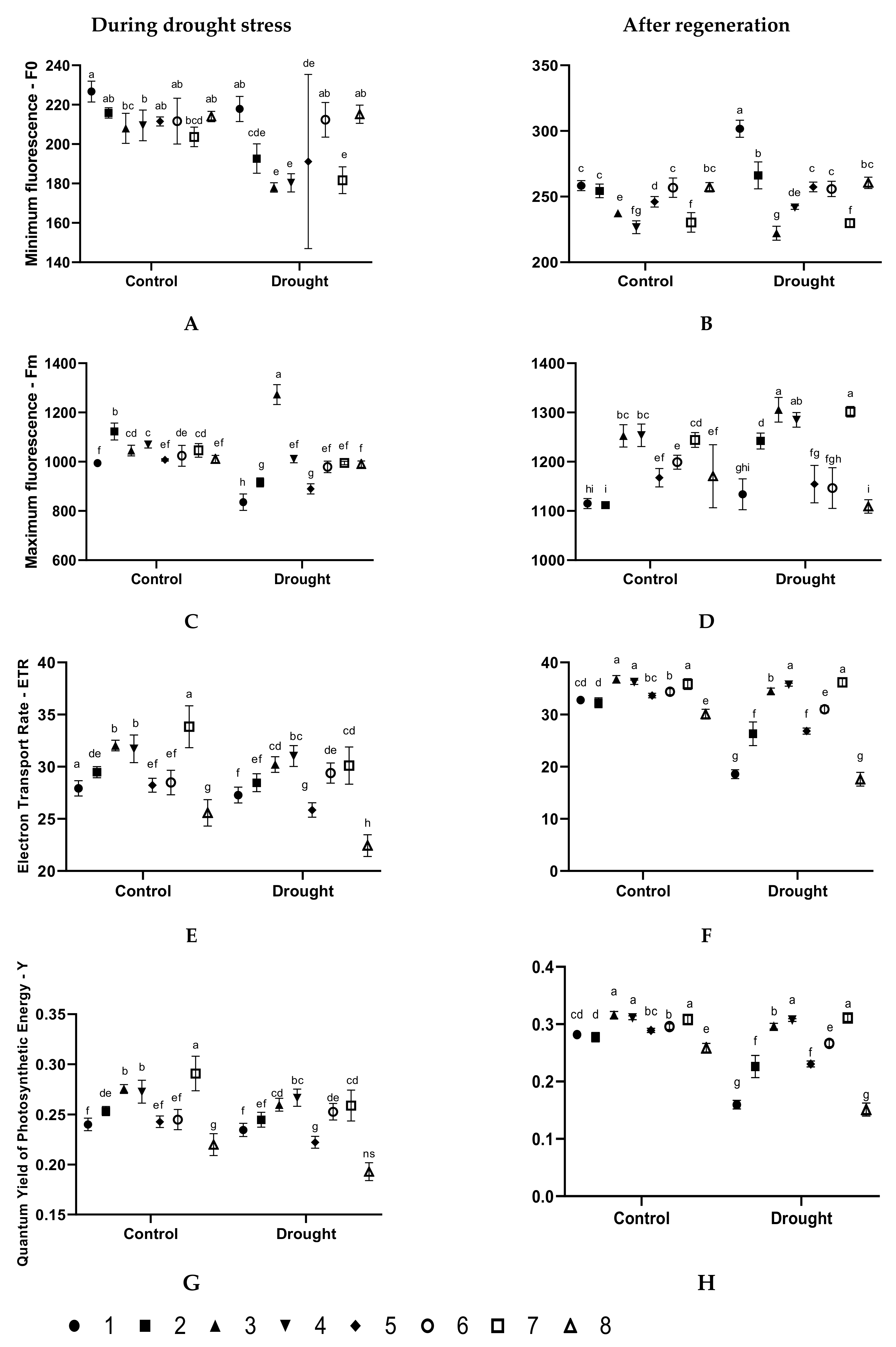
3.2. Effect of the Seed Treatments on the Physiological State of Plants During Drought Stress
3.3. Effect of the Seed Treatments on the Physiological State of Plants after After Regeneration
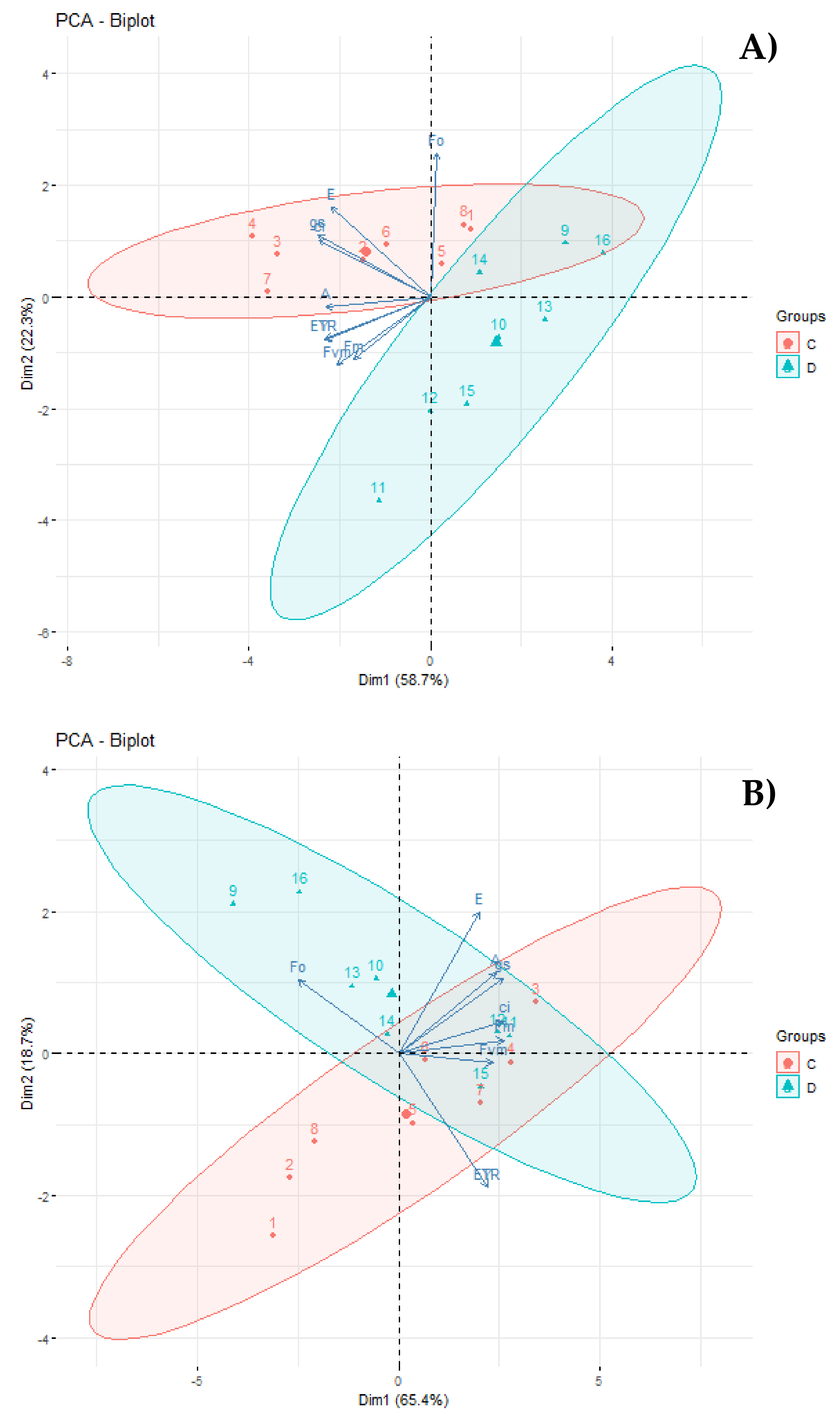
3.4. Principal Component Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Görtz, A.; Oerke, E.-C.; Puhl, T.; Steiner, U. Effect of environmental conditions on plant growth regulator activity of fungicidal seed treatments of barley. J. Appl. Bot. Food Qual. 2008, 82, 60–68. [Google Scholar]
- Kumar, S. Cultural Approaches for Plant Disease Management. Res. Rev. J. Agric. Sci. Technol. 2012, 1, 12–21. [Google Scholar]
- Montfort, F.; Klepper, B.L.; Smiley, R.W. Effects of Two Triazole Seed Treatments, Triticonazole and Triadimenol, on Growth and Development of Wheat. Pestic. Sci. 1996, 46, 315–322. [Google Scholar] [CrossRef]
- Sawinska, Z. The influence of seed treatment efficacy against leaf diseases in spring barley. Prog. Plant Prot. 2008, 48, 516–519. [Google Scholar]
- Sawinska, Z.; Khachatryan, K.; Sobiech, Ł.; Idziak, R.; Kosiada, T.; Skrzypczak, G.; Sobiech, L.; Idziak, R.; Kosiada, T.; Skrzypczak, G. Use of silver nanoparticles as a fungicide. Przem. Chem. 2014, 93, 1472–1474. [Google Scholar] [CrossRef]
- Deliopoulos, T.; Kettlewell, P.S.; Hare, M.C. Fungal disease suppression by inorganic salts: A review. Crop Prot. 2010, 29, 1059–1075. [Google Scholar] [CrossRef]
- Kuck, K.H.; Scheinpflug, H.; Pontzen, R. DMI fungicides. In Modern Selective Fungicides Properties, Applications, Mechanisms of Action; Lyr, H., Ed.; Gustav Fischer Verlag: New York, NY, USA, 1995; pp. 259–290. [Google Scholar]
- Förster, H.; Buchenauer, H.; Grossmann, F. Nebenwirkungen der systemischen Fungizide Triadimefon und Triadimenol auf Gersten-pflanzen. I. Beeinflussung von Wachstum und Ertrag. J. Plant Dis. Prot. 1980, 87, 473–492. [Google Scholar]
- Buchenauer, H.; Kutzner, B.; Kohts, T. Effect of various triazole fungicides on growth of cereal seedlings and tomato plants as well as on gibberellin contents and lipid metabolism in barley seedlings. J. Plant Dis. Prot. 1984, 91, 506–524. [Google Scholar]
- Buchenauer, H.; Röhner, E. Effect of triadimefon and triadimenol on growth of various plant species as well as on gibberellin content and sterol metabolism in shoots of barley seedlings. Pestic. Biochem. Physiol. 1981, 15, 58–70. [Google Scholar] [CrossRef]
- Anderson, H.M. Effect of triadimenol seed treatment on vegetative growth in winter wheat. Crop Res. 1989, 29, 29–36. [Google Scholar]
- Cavariani, C.; Velini, E.D.; Bicudo, S.J.; Nakagawa, J. Avaliação dos efeitos de doses de triadimenol e de tebuconazole sobre o crescimento do mesocótilo em plântulas de trigo. Pesq. Agropec. Bras. 1999, 29, 1035–1039. [Google Scholar]
- Avenot, H.; Morgan, D.P.; Michailides, T.J. Resistance to pyraclostrobin, boscalid and multiple resistance to Pristine® (pyraclostrobin + boscalid) fungicide in Alternaria alternata causing alternaria late blight of pistachios in California. Plant Pathol. 2008, 57, 135–140. [Google Scholar] [CrossRef]
- Kuhn, P.J. Mode of action of carboximides. In British Mycological Society Symposia Series; Elsevier: Amsterdam, The Netherlands, 1984; pp. 155–183. [Google Scholar]
- Stammler, G.; Brix, H.D.; Glattli, A.; Semar, M.; Schoefl, U. Biological properties of the carboxamide boscalid including recent studies on its mode of action. In Proceedings of the XVI Intonational Plant Protection Congress, Glasgow, KY, USA, 15–18 October 2007; pp. 40–45. [Google Scholar]
- Zhang, C.Q.; Liu, Y.H.; Ma, X.Y.; Feng, Z.; Ma, Z.H. Characterization of sensitivity of Rhizoctonia solani, causing rice sheath blight, to mepronil and boscalid. Crop Prot. 2009, 28, 381–386. [Google Scholar] [CrossRef]
- Avenot, H.F.; Michallides, T.J. Resistance to pyraclostrobin, boscalid and Pristine (pyraclostrobin plus boscalid) in Alternaria alternate isolates from California pistachio. Phytopathology 2007, 97, S5–S7. [Google Scholar]
- Sierotzki, H.; Scalliet, G. A Review of Current Knowledge of Resistance Aspects for the Next-Generation Succinate Dehydrogenase Inhibitor Fungicides. Phytopathology 2013, 103, 880–887. [Google Scholar] [CrossRef]
- Walter, H.; Tobler, H.; Ehrenfreund, J. O-Cyclopropyl-Carboxanilides and Their Use as Fungicides 2003. Patent EP1480955B1, 21 February 2020. [Google Scholar]
- Rheinheimer, J. Succinate dehydrogenase inhibitors. In Modern Crop Protection Compounds; Kraemer, W., Schirmer, U., Jeschke, P., Witschel, M., Eds.; Wiley-VCH: Weinheim, Germany, 2012; pp. 627–639. [Google Scholar]
- Zeun, R.; Scalliet, G.; Oostendorp, M. Biological activity of sedaxane—A novel broad-spectrum fungicide for seed treatment. Pest Manag. Sci. 2013, 69, 527–534. [Google Scholar] [CrossRef]
- Ajigboye, O.O.; Lu, C.; Murchie, E.H.; Schlatter, C.; Swart, G.; Ray, R.V. Altered gene expression by sedaxane increases PSII efficiency, photosynthesis and growth and improves tolerance to drought in wheat seedlings. Pestic. Biochem. Physiol. 2017, 137, 49–61. [Google Scholar] [CrossRef]
- Conclusion on the peer review of the pesticide risk assessment of the active substance fluxapyroxad (BAS 700 F). EFSA J. 2012, 10, 2522. [CrossRef]
- Cavell, P.; Blanchard, J.; Coquiller, M.; Fritz-Piou, S.; Gourmelin, P.; Morvan, Y.; Cousin, A. BAS 700 05 F: A new innovative fungicide solution from BASF to protect seed-, soil-borne diseases and more—Case study of barley protection. In Proceedings of the 10 Conférence Internationale sur les Maladies des Plantes, Tours, France, 3–5 December 2012; pp. 581–588. [Google Scholar]
- Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 2006, 11, 15–19. [Google Scholar] [CrossRef]
- Swędrzyńska, D.; Zielewicz, W.; Swędrzyński, A. Comparison of soil bioconditioners and standard fertilization in terms of the impact on yield and vitality of Lolium perenne and soil biological properties. Open Life Sci. 2019, 14, 666–680. [Google Scholar] [CrossRef]
- Bandurska, H.; Niedziela, J.; Pietrowska-Borek, M.; Nuc, K.; Chadzinikolau, T.; Radzikowska, D. Regulation of proline biosynthesis and resistance to drought stress in two barley (Hordeum vulgare L.) genotypes of different origin. Plant Physiol. Biochem. 2017, 118, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Weraduwage, S.M.; Chen, J.; Anozie, F.C.; Morales, A.; Weise, S.E.; Sharkey, T.D. The relationship between leaf area growth and biomass accumulation in Arabidopsis thaliana. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Mendoza, D.; Valdez-Salas, B.; Bernardo-Mazariegos, E.; Tzintzun-Camacho, O.; Gutiérrez-Miceli, F.; Ruíz-Valdiviezo, V.; Rodríguez-Hernández, L.; Sanchez-Viveros, G. Influence of monometallic and bimetallic phytonanoparticles on physiological status of mezquite. Open Life Sci. 2019, 14, 62–68. [Google Scholar] [CrossRef]
- Gupta, A.; Rico-Medina, A.; Caño-Delgado, A.I. The physiology of plant responses to drought. Science (80-.) 2020, 368, 266–269. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef]
- Baker, N.R. Chlorophyll Fluorescence: A Probe of Photosynthesis In Vivo. Annu. Rev. Plant Biol. 2008, 59, 89–113. [Google Scholar] [CrossRef] [PubMed]
- Bolhàr-Nordenkampf, H.R.; Öquist, G. Chlorophyll fluorescence as a tool in photosynthesis research. In Photosynthesis and Production in a Changing Environment; Springer: Dordrecht, The Netherlands, 1993; pp. 193–206. [Google Scholar]
- Waqas, M.A.; Kaya, C.; Riaz, A.; Farooq, M.; Nawaz, I.; Wilkes, A.; Li, Y. Potential Mechanisms of Abiotic Stress Tolerance in Crop Plants Induced by Thiourea. Front. Plant Sci. 2019, 10. [Google Scholar] [CrossRef]
- Munné-Bosch, S.; Alegre, L. Cross-stress tolerance and stress “memory” in plants: An integrated view. Environ. Exp. Bot. 2013, 94, 1–2. [Google Scholar] [CrossRef]
- Li, Y.; Ye, W.; Wang, M.; Yan, X. Climate change and drought: A risk assessment of crop-yield impacts. Clim. Res. 2009, 39, 31–46. [Google Scholar] [CrossRef]
- Hatzig, S.V.; Nuppenau, J.-N.; Snowdon, R.J.; Schießl, S.V. Drought stress has transgenerational effects on seeds and seedlings in winter oilseed rape (Brassica napus L.). BMC Plant Biol. 2018, 18, 297. [Google Scholar] [CrossRef]
- You, J.; Chan, Z. ROS Regulation During Abiotic Stress Responses in Crop Plants. Front. Plant Sci. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanpour, M.; Mohammadi, H.; Kariman, K. Nanosilicon-based recovery of barley ( Hordeum vulgare ) plants subjected to drought stress. Environ. Sci. Nano 2020, 7, 443–461. [Google Scholar] [CrossRef]
- Kobus-Cisowska, J.; Szulc, P.; Szczepaniak, O.; Dziedziński, M.; Szymanowska, D.; Szymandera-Buszka, K.; Goryńska-Goldmann, E.; Gazdecki, M.; Telichowska, A.; Ligaj, M. Variability of Hordeum vulgare L. Cultivars in Yield, Antioxidant Potential, and Cholinesterase Inhibitory Activity. Sustainability 2020, 12, 1938. [Google Scholar] [CrossRef]
- Hellal, F.A.; El-Shabrawi, H.M.; Abd El-Hady, M.; Khatab, I.A.; El-Sayed, S.A.A.; Abdelly, C. Influence of PEG induced drought stress on molecular and biochemical constituents and seedling growth of Egyptian barley cultivars. J. Genet. Eng. Biotechnol. 2018, 16, 203–212. [Google Scholar] [CrossRef]
- Ceccarelli, S.; Grando, S.; Baum, M. Participatory plant breeding in water-limited environments. Exp. Agric. 2007, 43, 411–435. [Google Scholar] [CrossRef]
- Nosalewicz, A.; Siecińska, J.; Śmiech, M.; Nosalewicz, M.; Wiącek, D.; Pecio, A.; Wach, D. Transgenerational effects of temporal drought stress on spring barley morphology and functioning. Environ. Exp. Bot. 2016, 131, 120–127. [Google Scholar] [CrossRef]
- Dąbrowska, B.; Pokojska, H.; Suchorska-Tropiło, T. Metody Laboratoryjnej Oceny Materiału Siewnego; Wydawnictwo SGGW: Warsaw, Poland, 2000. [Google Scholar]
- ISTA. 2011. Available online: https://www.seedtest.org/en/technical-committees-documents-_content--−1--3496.html (accessed on 20 April 2020).
- Kowalczewski, P.Ł.; Radzikowska, D.; Ivanišová, E.; Szwengiel, A.; Kačániová, M.; Sawinska, Z. Influence of Abiotic Stress Factors on the Antioxidant Properties and Polyphenols Profile Composition of Green Barley (Hordeum vulgare L.). Int. J. Mol. Sci. 2020, 21, 397. [Google Scholar] [CrossRef]
- Michael, G. Biplots in Practice; Books; Fundacion BBVA/BBVA Foundation: Bilbao, Spain, 2010. [Google Scholar]
- Ludlow, M.M.; Muchow, R.C. A Critical Evaluation of Traits for Improving Crop Yields in Water-Limited Environments. Adv. Agron. 1990, 43, 107–153. [Google Scholar]
- Reynolds, M.P.; Mujeeb-Kazi, A.; Sawkins, M. Prospects for utilising plant-adaptive mechanisms to improve wheat and other crops in drought- and salinity-prone environments. Ann. Appl. Biol. 2005, 146, 239–259. [Google Scholar] [CrossRef]
- Palta, J.A.; Chen, X.; Milroy, S.P.; Rebetzke, G.J.; Dreccer, M.F.; Watt, M. Large root systems: Are they useful in adapting wheat to dry environments? Funct. Plant Biol. 2011, 38, 347. [Google Scholar] [CrossRef]
- Daszkowska-Golec, A.; Collin, A.; Sitko, K.; Janiak, A.; Kalaji, H.M.; Szarejko, I. Genetic and Physiological Dissection of Photosynthesis in Barley Exposed to Drought Stress. Int. J. Mol. Sci. 2019, 20, 6341. [Google Scholar] [CrossRef] [PubMed]
- Witkowska-Banaszczak, E.; Radzikowska, D.; Ratajczak, K. Chemical profile and antioxidant activity of Trollius europaeus under the influence of feeding aphids. Open Life Sci. 2018, 13, 312–318. [Google Scholar] [CrossRef]
- Habash, D.; Percival, M.P.; Baker, N.R. Rapid chlorophyll fluorescence technique for the study of penetration of photosynthetically active herbicides into leaf tissue. Weed Res. 1985, 25, 389–395. [Google Scholar] [CrossRef]
- Berdugo, C.A.; Mahlein, A.K.; Steiner, U.; Oerke, E.C.; Dehne, H.W. Use of non-invasive sensors to detect beneficial effects of fungicides on wheat physiology. In Proceedings of the 11th International Conference on Precision Agriculture (ICPA), Indianapolis, IN, USA, 15–18 July 2012. [Google Scholar]
- Matiu, M.; Ankerst, D.P.; Menzel, A. Interactions between temperature and drought in global and regional crop yield variability during 1961−2014. PLoS ONE 2017, 12, e0178339. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Hu, F.; Ma, L. Phosphate-solubilizing bacteria from safflower rhizosphere and their effect on seedling growth. Open Life Sci. 2019, 14, 246–254. [Google Scholar] [CrossRef]
- Boyer, J.S. Plant Productivity and Environment. Science 1982, 218, 443–448. [Google Scholar] [CrossRef]
- Baker, N.R. Applications of chlorophyll fluorescence can improve crop production strategies: An examination of future possibilities. J. Exp. Bot. 2004, 55, 1607–1621. [Google Scholar] [CrossRef]
- Genty, B.; Briantais, J.-M.; Baker, N.R. The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim. Biophys. Acta Gen. Subj. 1989, 990, 87–92. [Google Scholar] [CrossRef]
- Zlatev, Z. Drought-Induced Changes in Chlorophyll Fluorescence of Young Wheat Plants. Biotechnol. Biotechnol. Equip. 2009, 23, 438–441. [Google Scholar] [CrossRef]
- Ajigboye, O.O.; Murchie, E.; Ray, R.V. Foliar application of isopyrazam and epoxiconazole improves photosystem II efficiency, biomass and yield in winter wheat. Pestic. Biochem. Physiol. 2014, 114, 52–60. [Google Scholar] [CrossRef]
- Ruggiero, A.; Punzo, P.; Landi, S.; Costa, A.; Van Oosten, M.; Grillo, S. Improving Plant Water Use Efficiency through Molecular Genetics. Horticulturae 2017, 3, 31. [Google Scholar] [CrossRef]
- Ohashi, Y.; Nakayama, N.; Saneoka, H.; Fujita, K. Effects of drought stress on photosynthetic gas exchange, chlorophyll fluorescence and stem diameter of soybean plants. Biol. Plant. 2006, 50, 138–141. [Google Scholar] [CrossRef]
- Araújo, W.L.; Nunes-Nesi, A.; Osorio, S.; Usadel, B.; Fuentes, D.; Nagy, R.; Balbo, I.; Lehmann, M.; Studart-Witkowski, C.; Tohge, T.; et al. Antisense Inhibition of the Iron-Sulphur Subunit of Succinate Dehydrogenase Enhances Photosynthesis and Growth in Tomato via an Organic Acid–Mediated Effect on Stomatal Aperture. Plant Cell 2011, 23, 600–627. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, D.; Meneses, M.; Nunes-Nesi, A.; Araújo, W.L.; Tapia, R.; Gómez, I.; Holuigue, L.; Gutiérrez, R.A.; Fernie, A.R.; Jordana, X. A Deficiency in the Flavoprotein of Arabidopsis Mitochondrial Complex II Results in Elevated Photosynthesis and Better Growth in Nitrogen-Limiting Conditions. Plant Physiol. 2011, 157, 1114–1127. [Google Scholar] [CrossRef]
| Treatment ID | Active Substances of Seed Treatments | Trade Name of Treatments |
|---|---|---|
| 1 | Control – untreated | |
| 2 | Fludioxonil 25 g/L | Maxim® 025 FS/ Celest® 025 FS |
| 3 | Fludioxonil 25 g/L + sedaxane 25 g/L | Vibrance® Duo / Maxim® Power |
| 4 | Fludioxonil 25 g/L + sedaxane 25 g/L + triticonazole 20 g/L | Vibrance® Star |
| 5 | Triticonazole 50 g/L | Triter 050 FS |
| 6 | Prothioconazole 150 g/L + tebuconazole 20 g/L | Redigo® Pro 170 FS |
| 7 | Fludioxonil 33.3 g/L + fluxapyroxad 33.3 g/L + triticonazole 33.3 g/L | Kinto® Plus |
| 8 | Fludioxonil 50 g/L | Madron 50 FS |
| Treatment | Germination Energy | Germination Capacity | Vigor Index | Root Length | Shoot Length |
|---|---|---|---|---|---|
| 1. Control – untreated | 92.0 ± 0.1a | 92.7 ± 1.2bc | 5263 ± 544.8a | 60.4 ± 3.5ab | 57.2 ± 6.4ab |
| 2. Fludioxonil | 92.0 ± 0.1a | 95.3 ± 2.3abc | 4796 ± 154.5a | 51.0 ± 3.8b | 52.1 ± 2.2ab |
| 3. Fludioxonil + sedaxane | 88.0 ± 4.0a | 95.3 ± 2.3abc | 5832 ± 480.8a | 59.6 ± 6.9ab | 66.3 ± 5.0a |
| 4. Fludioxonil + sedaxane + triticonazole | 93.3 ± 4.6a | 94.0 ± 3.5abc | 5497 ± 436.4a | 70.1 ± 8.7a | 58.9 ± 4.1ab |
| 5. Triticonazole | 92.0 ± 6.9a | 97.3 ± 2.3a | 4211 ± 496.5ab | 56.4 ± 2.7ab | 44.8 ± 2.5b |
| 6. Prothioconazole + tebuconazole | 80.0 ± 4.0b | 91.3 ± 2.3c | 3192 ± 165.4b | 57.4 ± 4.4ab | 40.0 ± 3.2b |
| 7. Fludioxonil + fluxapyroxad + triticonazole | 92.0 ± 6.0a | 94.7 ± 4.2abc | 5005 ± 652.2a | 67.2 ± 1.4a | 54.2 ± 15.97ab |
| 8. Fludioxonil | 93.3 ± 4.6a | 96.0 ± 2.0ab | 5285 ± 897.1a | 56.9 ± 7.8ab | 56.5 ± 8.1ab |
| LSD | 7.3 | 4.6 | 1138 | 15.1 | 20.7 |
| Treatment | During Drought Stress | After Regeneration | ||
|---|---|---|---|---|
| Control | Drought | Control | Drought | |
| 1. Control – untreated | 6.440 ± 0.290 j | 6.767 ± 0.416 ij | 9.627 ± 0.340 h | 10.553 ± 0.125 g |
| 2. Fludioxonil | 8.930 ± 0.061 d | 8.643 ± 0.174 def | 8.005 ± 0.065 i | 12.180 ± 0.100 cd |
| 3. Fludioxonil + sedaxane | 10.580 ± 0.370 b | 8.803 ± 0.280 de | 13.797 ± 0.237 a | 13.387 ± 0.270 b |
| 4. Fludioxonil + sedaxane + triticonazole | 11.760 ± 0.340 a | 8.350 ± 0.165 efg | 13.077 ± 0.112 b | 13.807 ± 0.329 a |
| 5. Triticonazole | 7.210 ± 0.288 hi | 8.060 ± 0.017 g | 9.970 ± 0.05 h | 11.013 ± 0.012 f |
| 6. Prothioconazole + tebuconazole | 9.390 ± 0.250 c | 7.387 ± 0.352 h | 13.107 ± 0.122 b | 11.880 ± 0.180 de |
| 7. Fludioxonil + fluxapyroxad + triticonazole | 11.307 ± 0.124 a | 8.197 ± 0.196 fg | 12.527 ± 0.072 c | 11.553 ± 0.278 e |
| 8. Fludioxonil | 6.560 ± 0.110 j | 7.570 ± 0.208 h | 9.623 ± 0.035 h | 10.640 ± 0.288 g |
| LSD | 0.454 | 0.350 | ||
| Treatment | During Drought Stress | After Regeneration | ||
|---|---|---|---|---|
| Control | Drought | Control | Drought | |
| 1. Control – untreated | 1.903 ± 0.046 e | 1.550 ± 0.010 f | 1.990 ± 0.010 j | 3.020 ± 0.010 gh |
| 2. Fludioxonil | 2.205 ± 0.085 d | 1.523 ± 0.040 f | 2.567 ± 0.227 i | 3.217 ± 0.091 fg |
| 3. Fludioxonil + sedaxane | 2.717 ± 0.115 b | 1.350 ± 0.040 gh | 3.793 ± 0.114 a | 3.673 ± 0.051 ab |
| 4. Fludioxonil + sedaxane + triticonazole | 3.110 ± 0.040 a | 1.463 ± 0.105 fg | 3.403 ± 0.086 cdef | 3.617 ± 0.110 abc |
| 5. Triticonazole | 1.983 ± 0.178 e | 1.523 ± 0.143 f | 2.923 ± 0.118 h | 3.523 ± 0.021 bcd |
| 6. Prothioconazole + tebuconazole | 2.190 ± 0.085 d | 1.953 ± 0.064 e | 3.247 ± 0.168 efg | 3.370 ± 0.392 cdef |
| 7. Fludioxonil + fluxapyroxad + triticonazole | 2.523 ± 0.131 c | 1.280 ± 0.066 h | 3.013 ± 0.038 gh | 3.493 ± 0.095 bcde |
| 8. Fludioxonil | 2.110 ± 0.098 d | 1.390 ± 0.066 fgh | 2.497 ± 0.129 i | 3.347 ± 0.093 def |
| LSD | 0.166 | 0.256 | ||
| Treatment | During Drought Stress | After Regeneration | ||
|---|---|---|---|---|
| Control | Drought | Control | Drought | |
| 1. Control – untreated | 0.100 ± 0.001 de | 0.067 ± 0.006 gh | 0.130 ± 0.010 g | 0.180 ± 0.010 e |
| 2. Fludioxonil | 0.130 ± 0.010 bc | 0.073 ± 0.006 fgh | 0.157 ± 0.015 f | 0.193 ± 0.015 e |
| 3. Fludioxonil + sedaxane | 0.170 ± 0.020 a | 0.083 ± 0.006 efg | 0.267 ± 0.015 a | 0.227 ± 0.006 bc |
| 4. Fludioxonil + sedaxane + triticonazole | 0.167 ± 0.021 a | 0.087 ± 0.006 ef | 0.243 ± 0.006 b | 0.223 ± 0.015 c |
| 5. Triticonazole | 0.110 ± 0.010 d | 0.070 ± 0.001 fgh | 0.217 ± 0.006 c | 0.193 ± 0.006 e |
| 6. Prothioconazole + tebuconazole | 0.140 ± 0.010 b | 0.077 ± 0.006 fgh | 0.213 ± 0.006 cd | 0.196 ± 0.004 de |
| 7. Fludioxonil + fluxapyroxad + triticonazole | 0.163 ± 0.015 a | 0.077 ± 0.006 fgh | 0.233 ± 0.006 b | 0.227 ± 0.012 bc |
| 8. Fludioxonil | 0.117 ± 0.015 cd | 0.063 ± 0.006 h | 0.147 ± 0.015 fg | 0.197 ± 0.006 de |
| LSD | 0.019 | 0.028 | ||
| Treatment | During Drought Stress | After Regeneration | ||
|---|---|---|---|---|
| Control | Drought | Control | Drought | |
| 1. Control – untreated | 212.7 ± 3.786 f | 171.0 ± 4.583 i | 221.7 ± 2.517 h | 228.0 ± 8.660 gh |
| 2. Fludioxonil | 242.3 ± 9.019 bc | 188.3 ± 3.055 gh | 236.7 ± 1.528 fg | 238.0 ± 1.000 f |
| 3. Fludioxonil + sedaxane | 253.0 ± 4.359 ab | 196.7 ± 6.658 g | 262.3 ± 3.512 a | 247.6 ± 7.767 cde |
| 4. Fludioxonil + sedaxane + triticonazole | 256.0 ± 5.000 a | 196.7 ± 5.686 g | 260.0 ±4.359 ab | 252.0 ± 2.646 bcd |
| 5. Triticonazole | 220.7 ± 10.017 ef | 179.0 ± 2.646 hi | 239.0 ± 2.646 ef | 239.3 ± 2.082 ef |
| 6. Prothioconazole + tebuconazole | 232.3 ± 10.970 cd | 193.7 ± 9.292 g | 237.3 ± 12.423 f | 238.6 ± 1.155 f |
| 7. Fludioxonil + fluxapyroxad + triticonazole | 245.3 ± 4.509 ab | 190.3 ± 3.512 gh | 253.0 ± 0.000 bc | 243.7 ± 3.215 def |
| 8. Fludioxonil | 230.0 ± 4.582 de | 170.0 ± 7.810 i | 237.0 ± 5.568 f | 237.7 ± 2.309 f |
| LSD | 11.628 | 8.915 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Radzikowska, D.; Grzanka, M.; Kowalczewski, P.Ł.; Głowicka-Wołoszyn, R.; Blecharczyk, A.; Nowicki, M.; Sawinska, Z. Influence of SDHI Seed Treatment on the Physiological Conditions of Spring Barley Seedlings under Drought Stress. Agronomy 2020, 10, 731. https://doi.org/10.3390/agronomy10050731
Radzikowska D, Grzanka M, Kowalczewski PŁ, Głowicka-Wołoszyn R, Blecharczyk A, Nowicki M, Sawinska Z. Influence of SDHI Seed Treatment on the Physiological Conditions of Spring Barley Seedlings under Drought Stress. Agronomy. 2020; 10(5):731. https://doi.org/10.3390/agronomy10050731
Chicago/Turabian StyleRadzikowska, Dominika, Monika Grzanka, Przemysław Łukasz Kowalczewski, Romana Głowicka-Wołoszyn, Andrzej Blecharczyk, Marcin Nowicki, and Zuzanna Sawinska. 2020. "Influence of SDHI Seed Treatment on the Physiological Conditions of Spring Barley Seedlings under Drought Stress" Agronomy 10, no. 5: 731. https://doi.org/10.3390/agronomy10050731
APA StyleRadzikowska, D., Grzanka, M., Kowalczewski, P. Ł., Głowicka-Wołoszyn, R., Blecharczyk, A., Nowicki, M., & Sawinska, Z. (2020). Influence of SDHI Seed Treatment on the Physiological Conditions of Spring Barley Seedlings under Drought Stress. Agronomy, 10(5), 731. https://doi.org/10.3390/agronomy10050731









