Establishment of Critical Nitrogen Concentration Models in Winter Wheat under Different Irrigation Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Experiment Design
2.2. Analysis of N Content
2.3. Data Analysis
2.4. Model Building
2.4.1. Establishment of a Dilution Curve Model for the Critical Nitrogen Concentration
2.4.2. Construction of Wheat Nitrogen Nutrition Index Models
2.4.3. Construction of Wheat N Deficient Models
3. Results
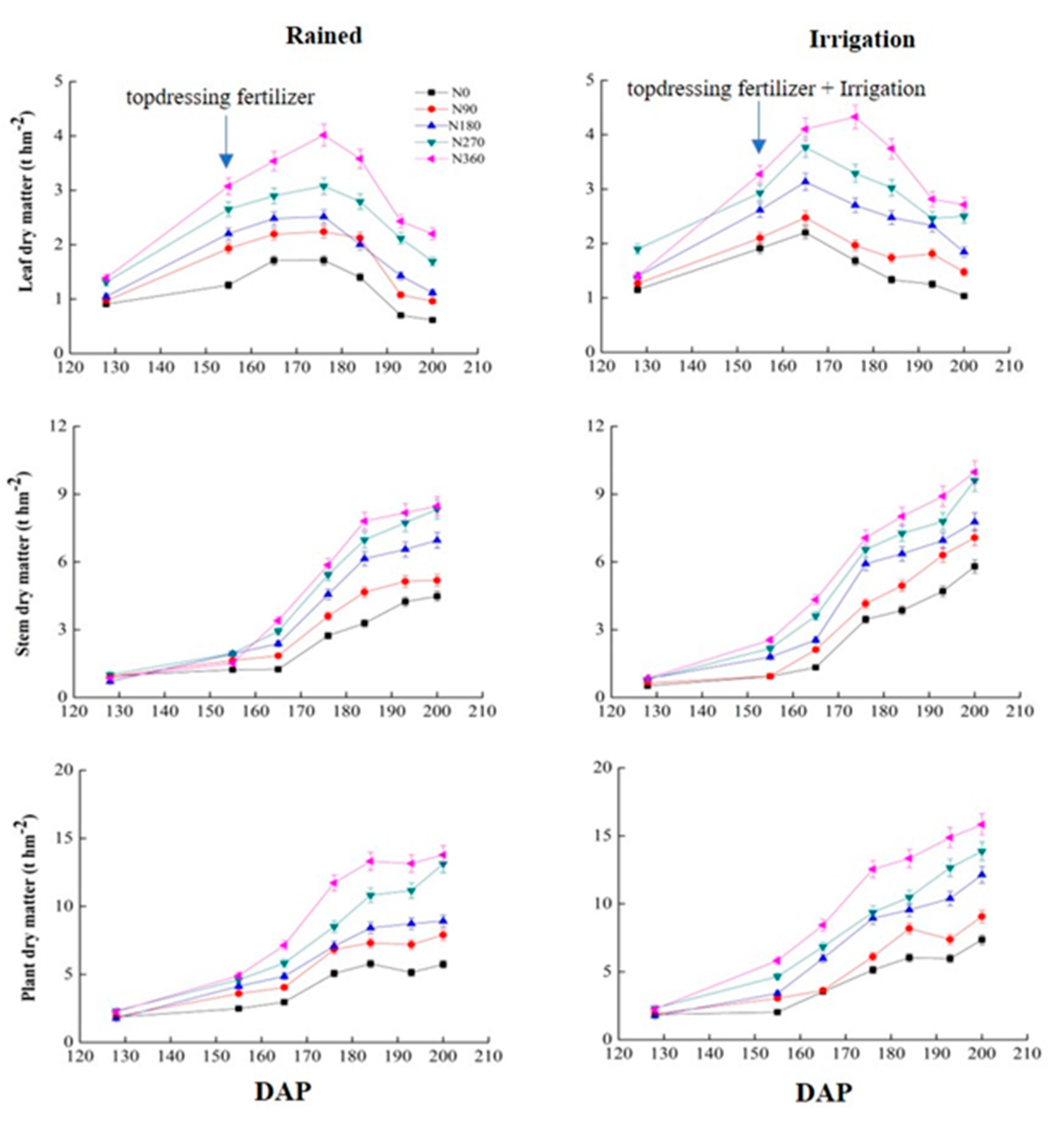
3.1. Effects of Different Irrigation Conditions on Wheat Biomass
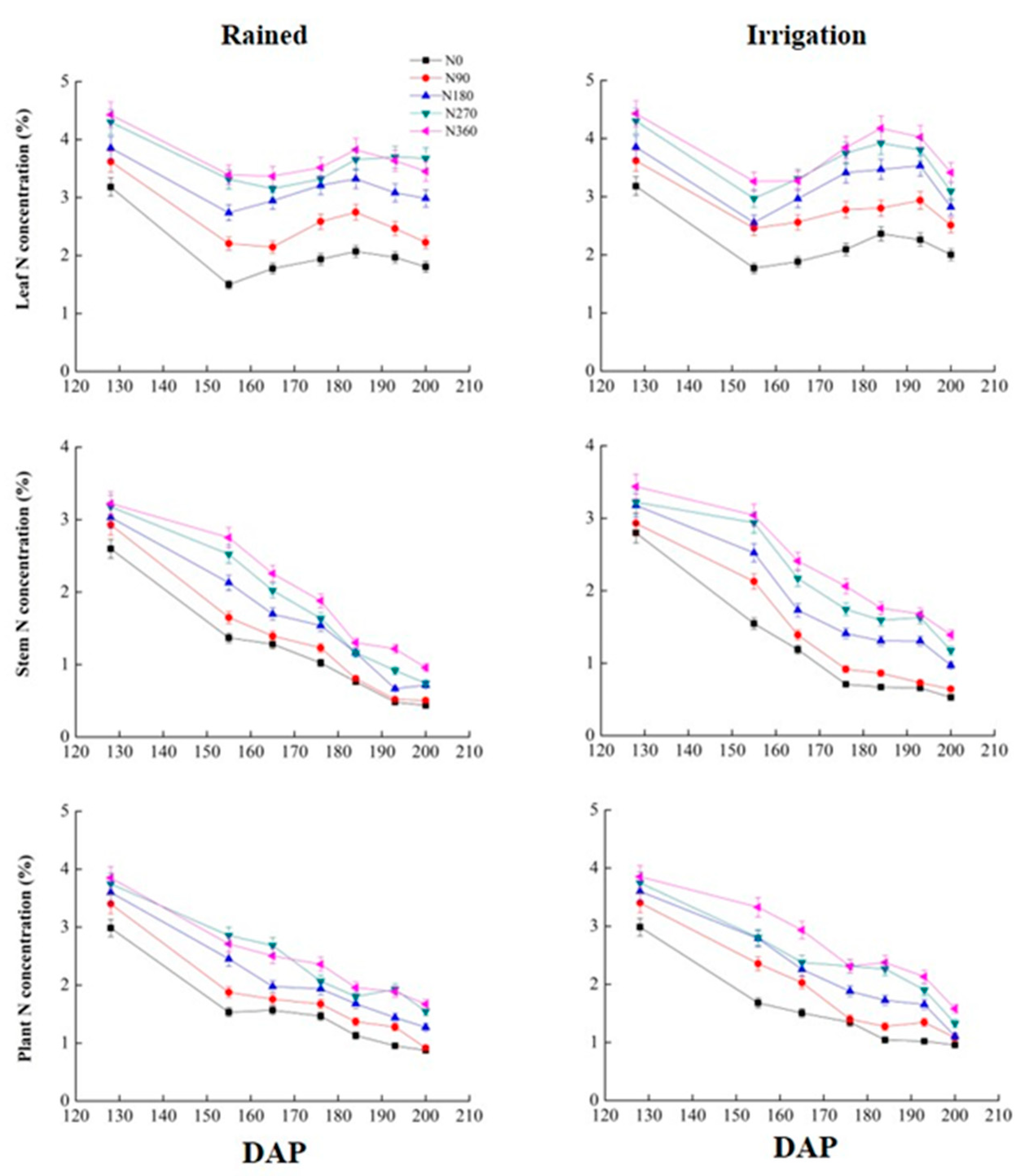
3.2. Effects of Different Irrigation Conditions on the N Content of Wheat
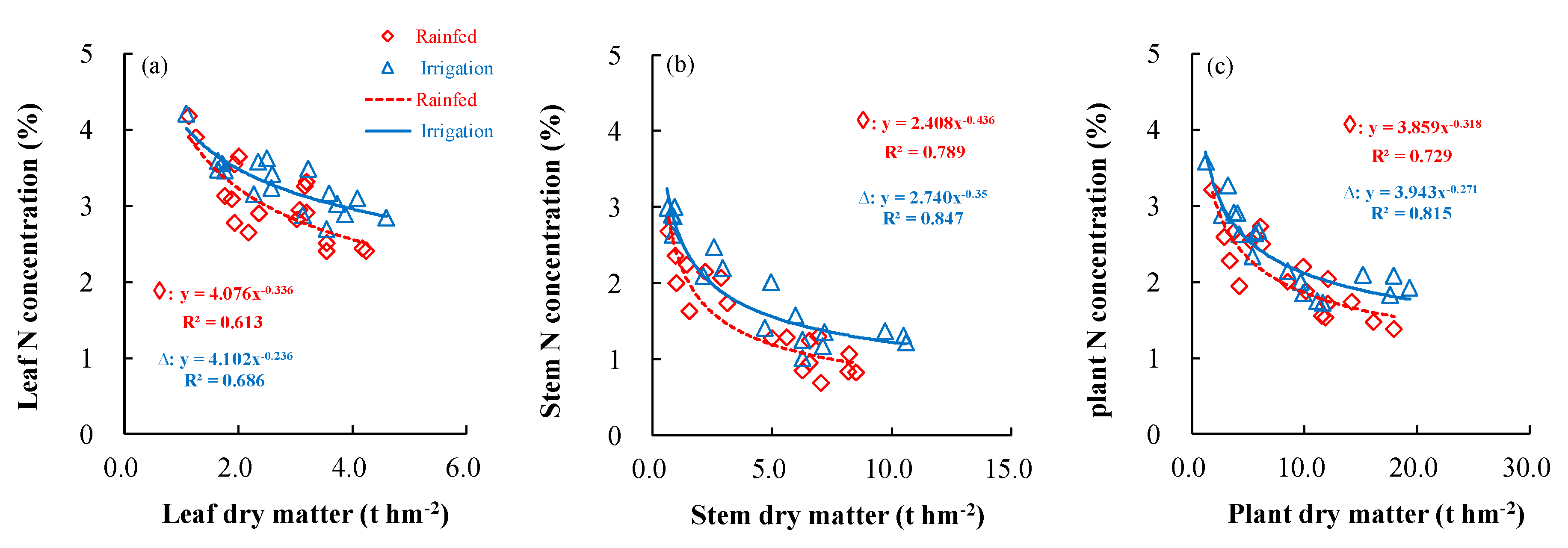
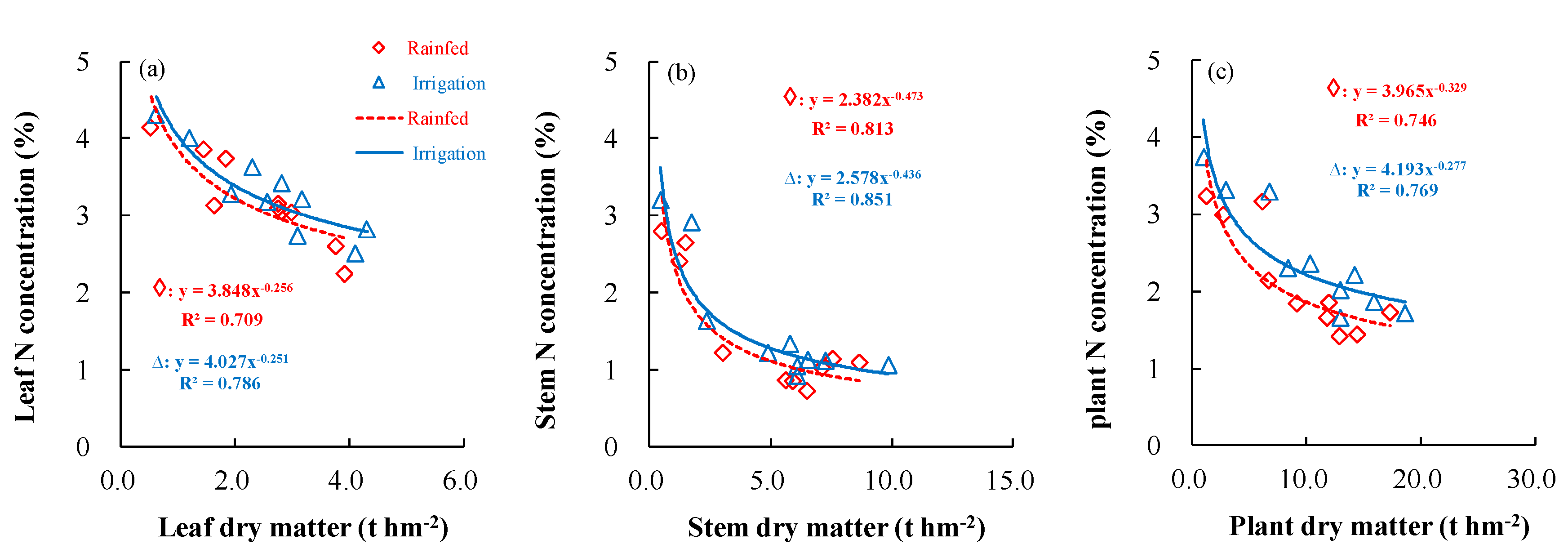
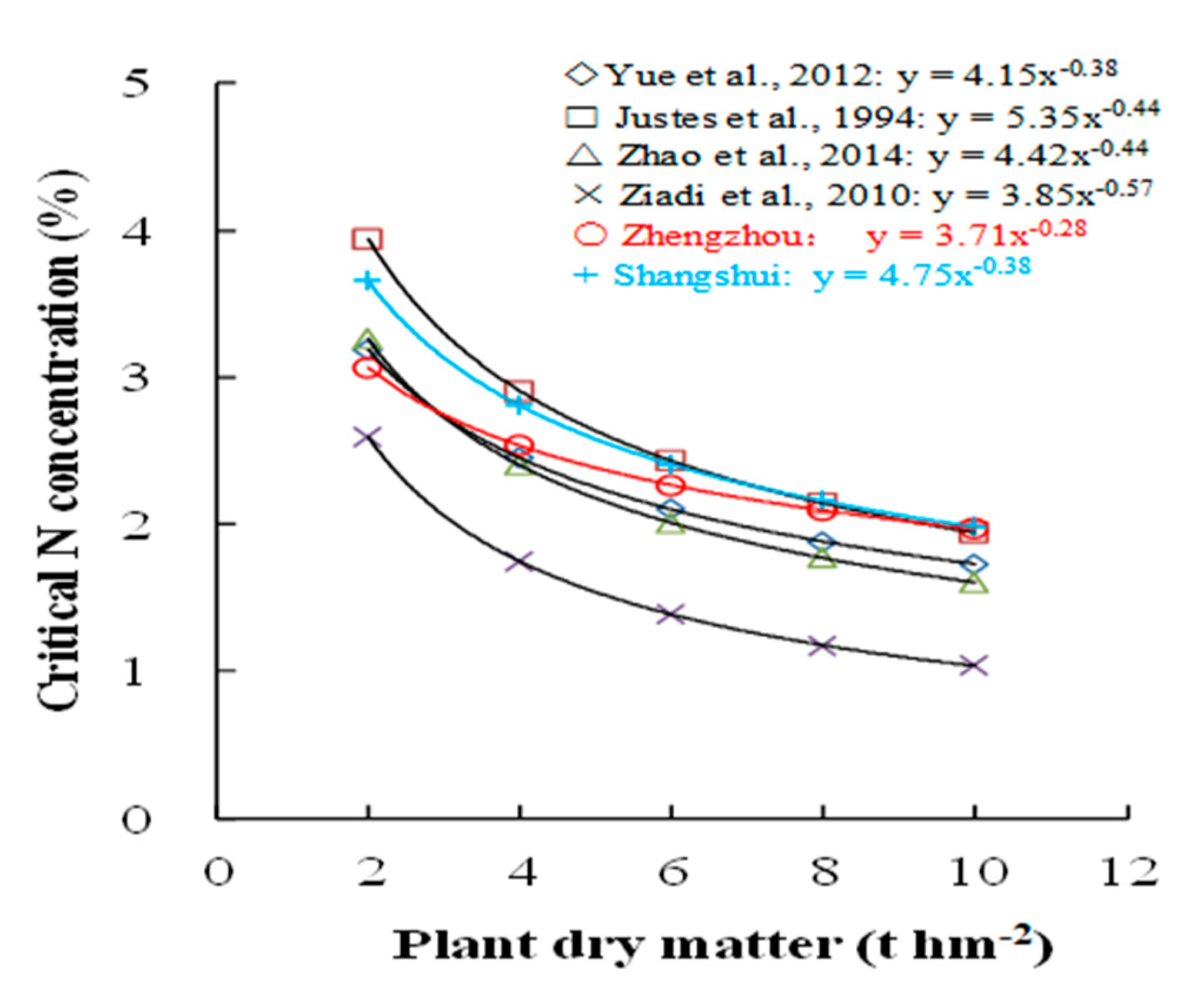
3.3. Effects of Different Irrigation Conditions on the Dilution Model of Critical N Concentrations in Wheat
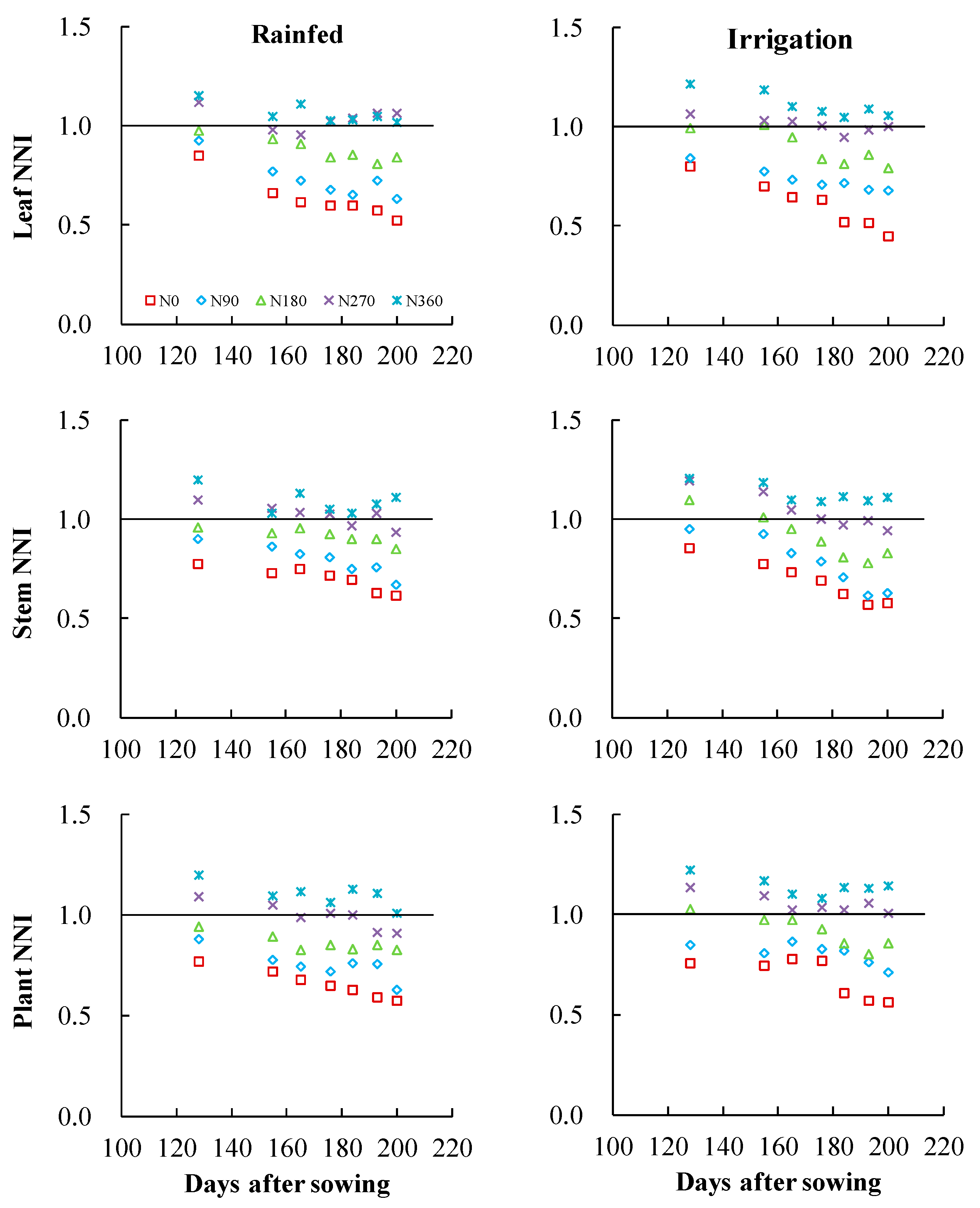
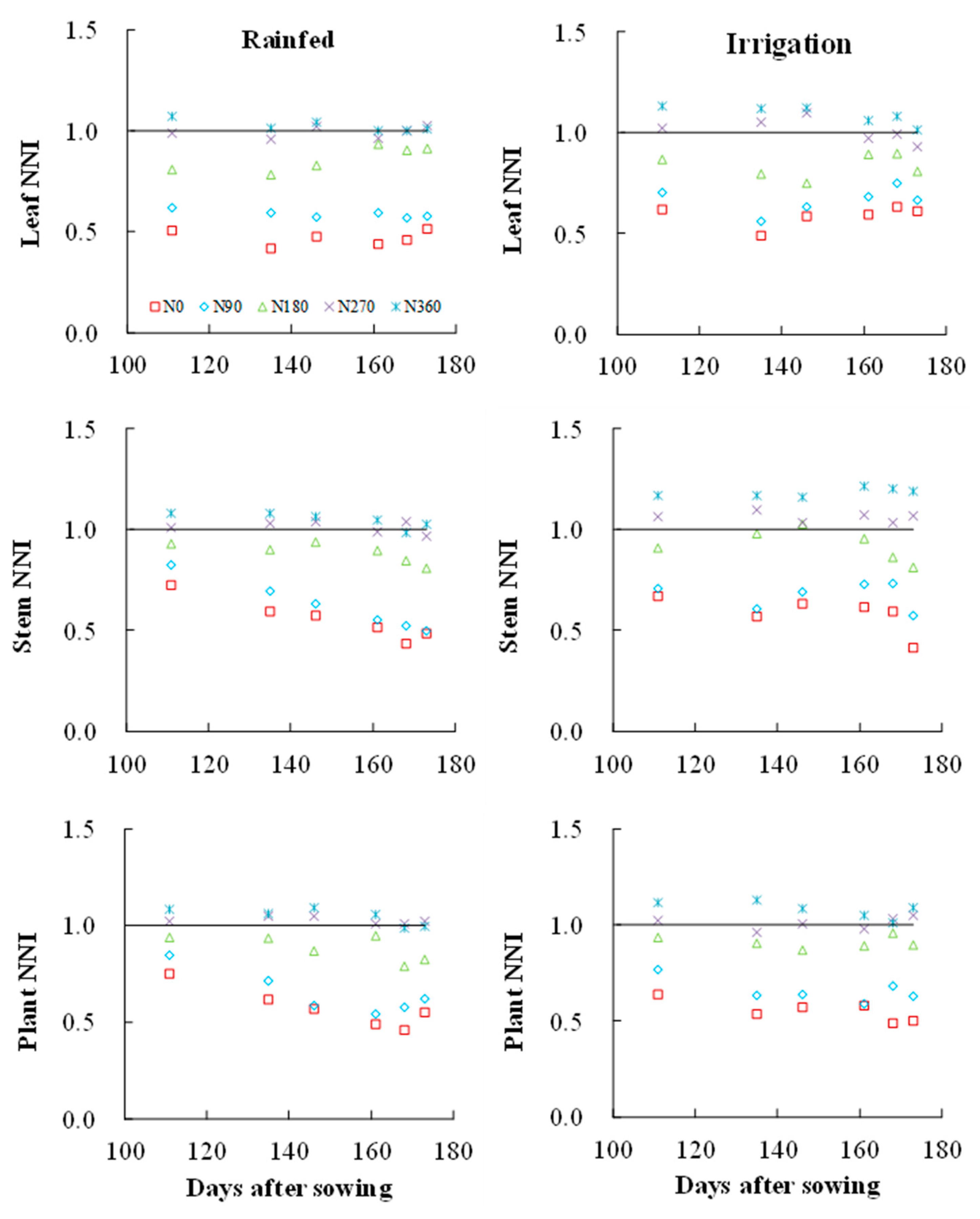
3.4. Effects of Different Irrigation Conditions on the N Nutrition Index of Wheat
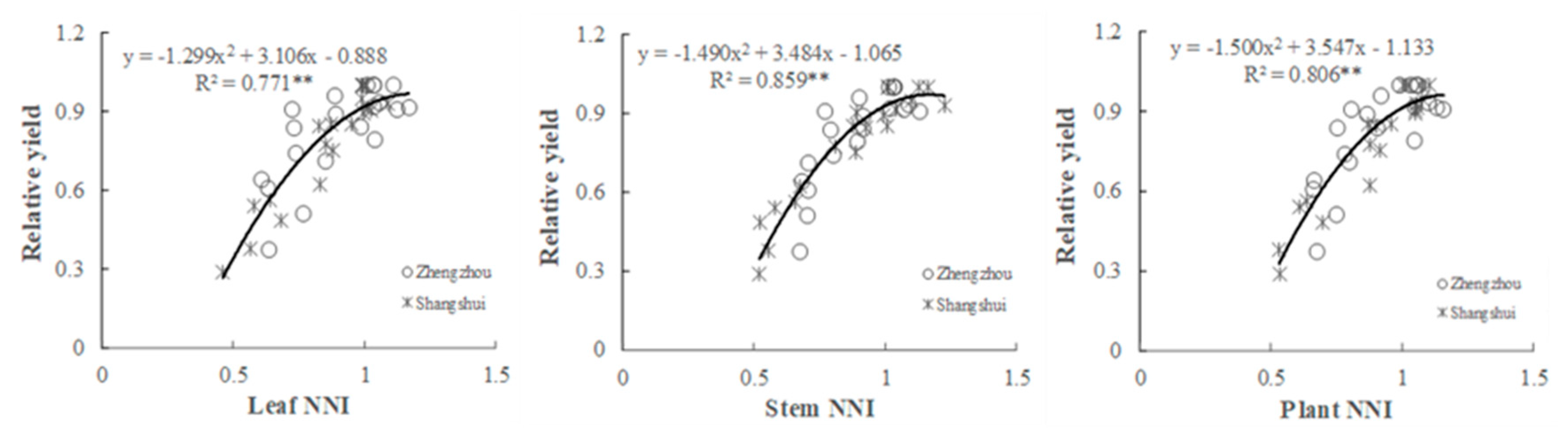
3.5. Relationship between the NNI and N Deficiency and Relative Yield in Wheat
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Hatfield, J.L.; Gitelson, A.A.; Schepers, J.S.; Walthall, C.L. Application of Spectral Remote Sensing for Agronomic Decisions. Agron. J. 2008, 100, 117–131. [Google Scholar] [CrossRef]
- Miao, Y.; Stewart, B.A.; Zhang, F. Long-term experiments for sustainable nutrient management in China. A review. Agron. Sustain. Dev. 2011, 31, 397–414. [Google Scholar] [CrossRef]
- Greenwood, D.J.; Lemaire, G.; Gosse, G.; Cruz, P.; Draycott, A.; Neeteson, J.J. Decline in Percentage N of C3 and C4 Crops with Increasing Plant Mass. Ann. Bot. 1990, 66, 425–436. [Google Scholar] [CrossRef]
- Plénet, D.; Lemaire, G. Relationships between dynamics of nitrogen uptake and dry matter accumulation in maize crops. Determination of critical N concentration. Plant Soil 1999, 216, 65–82. [Google Scholar]
- Lemaire, G.; Salette, J. Relation entre dynamique de croissance et dynamique de prélèvement d’azote pour un peuplement de graminées fourragères. I.–Etude de l’effet du milieu. Milieu. Agronomie 1984, 4, 423–430. [Google Scholar]
- Lemaire, G.; Gastal, F.; Cruz, P.; Greenwood, D.J.; Draycott, A. Relationships between plant-N, plant mass and relative growth rate for C3 and C4 crops. In the Proceedings of the First European Society of Agronomy Congress, Paris, France, 5–7 December 1990; pp. 1–5. [Google Scholar]
- Sheehy, J.E.; Dionora, M.J.A.; Mitchell, P.L.; Peng, S.; Cassman, K.G.; Lemaire, G.; Willisms, R.L. Critical nitrogen concentrations: Implications for high-yielding rice (Oryza sativa L.) cultivars in the tropics. Field Crops Res. 1998, 59, 31–41. [Google Scholar] [CrossRef]
- Colnenne, C.; Meynard, J.M.; Reau, R.; Justes, E.; Merrien, A. Determination of a Critical Nitrogen Dilution Curve for Winter Oilseed Rape. Ann. Bot. 1998, 81, 311–317. [Google Scholar] [CrossRef]
- Oosterom, E.J.V.; Carberry, P.S.; Muchow, R.C. Critical and minimum N contents for development and growth of grain sorghum. Field Crops Res. 2001, 70, 55–73. [Google Scholar] [CrossRef]
- Yang, H.; Cao, H.X.; Liu, M.Y.; Liu, S.H. Simulation of critical nitrogen concentration and nitrogen nutrition index of tomato under different water and nitrogen conditions. J. Plant Nutr. Fertil. 2015, 21, 1234–1242. [Google Scholar]
- Justes, E.; Mary, B.; Meynard, J.M.; Machet, J.M.; Thelier-Huche, L. Determination of a Critical Nitrogen Dilution Curve for Winter Wheat Crops. Ann. Bot. 1994, 74, 397–407. [Google Scholar] [CrossRef]
- Ziadi, N.; Bélanger, G.; Claessens, A.; Lefebvre, L.; Cambouris, A.N.; Tremblay, N.; Nolin, M.C.; Parent, L.É. Determination of a critical nitrogen dilution curve for spring wheat. Agron. J. 2010, 102, 241–250. [Google Scholar] [CrossRef]
- Zhao, B.; Yao, X.; Tian, Y.C.; Liu, X.J.; Cao, W.X.; Zhu, Y. Accumulative nitrogen deficit models of wheat aboveground part based on critical nitrogen concentration. Chin. J. Appl. Ecol. 2012, 23, 3141–3148. [Google Scholar]
- Yue, S.H.; Liu, C.Y.; Huang, Y.F.; Ye, Y.L. Simulating Critical Nitrogen Dilution Curve and Modeling N Nutrition Index in Winter Wheat in Central Henan Area. Acta Agronomica Sinica 2016, 42, 909–916. [Google Scholar] [CrossRef]
- Rahimi, A.; Sayadi, F.; Dashti, H.; Tajabadi, P. Effects of water and nitrogen supply on growth, water-use efficiency and mucilage yield of isabgol (Plantago ovata Forsk). J. Soil Sci. Plant Nut. 2013, 13, 341–354. [Google Scholar] [CrossRef]
- Arora, A.; Singh, V.P.; Mohan, J. Effect of Nitrogen and Water Stress on Photosynthesis and Nitrogen Content in Wheat. Biol. Plantarum 2001, 44, 153–155. [Google Scholar] [CrossRef]
- Li, S.X.; Wang, Z.H.; Malhi, S.S.; Li, S.Q.; Gao, Y.J.; Tian, X.H. Nutrient and Water Management Effects on Crop Production, and Nutrient and Water Use Efficiency in Dryland Areas of China. Adv. Agron. 2009, 102, 223–265. [Google Scholar]
- Ren, Z.S.; Qu, Z.Y.; Li, Z.; Liu, A.Q. Interactive Effects of Nitrogen Fertilization and Irrigation on Grain Yield, Water Use Efficiency and Nitrogen Use Efficiency of Mulched Drip-irrigated Maize in Hetao Irrigation District, China. J. Soil Water Conserv. 2016, 30, 149–155. [Google Scholar]
- Elazab, A.; Serret, M.D.; Araus, J.L. Interactive effect of water and nitrogen regimes on plant growth, root traits and water status of old and modern durum wheat genotypes. Planta 2016, 244, 125–144. [Google Scholar] [CrossRef]
- Pandey, R.K.; Maranville, J.W.; Admou, A. Deficit irrigation and nitrogen effects on maize in a Sahelian environment: I. Grain yield and yield components. Agric. Water Manag. 2000, 46, 1–13. [Google Scholar] [CrossRef]
- Li, Y.; Yin, Y.; Zhao, Q.; Wang, Z. Changes of Glutenin Subunits due to Water-Nitrogen Interaction Influence Size and Distribution of Glutenin Macropolymer Particles and Flour Quality. Crop Sci. 2011, 51, 2809–2819. [Google Scholar] [CrossRef]
- Araki, H.; Hamada, A.; Hossain, M.A.; Takahashi, T. Waterlogging at jointing and/or after anthesis in wheat induces early leaf senescence and impairs grain filling. Field Crops Res. 2012, 137, 27–36. [Google Scholar] [CrossRef]
- Wang, C.Y.; Liu, W.X.; Li, Q.X.; Ma, D.Y.; Lu, H.F.; Feng, W.; Xie, Y.X.; Zhu, Y.J.; Guo, T.C. Effects of different irrigation and nitrogen regimes on root growth and its correlation with above-ground plant parts in high-yielding wheat under field conditions. Field Crops Res. 2014, 165, 138–149. [Google Scholar] [CrossRef]
- Coelho, E.F.; Or, D. Root distribution and water uptake patterns of corn under surface and subsurface drip irrigation. Plant Soil 1998, 206, 123–136. [Google Scholar] [CrossRef]
- Xiang, Y.Z.; Zhang, F.C.; Fan, J.L.; Qiang, S.C.; Zou, H.Y.; Yan, S.C.; Wu, Y.; Tian, J.K. Nutrition diagnosis for N in bell pepper based on critical nitrogen model in solar greenhouse. Trans. Chin. Soc. Agric. Eng. 2016, 32, 89–97. [Google Scholar]
- Isaac, R.A.; Johnson, W.C. Determination of total nitrogen in plant tissue using a bloc digestor. Assoc. Off. Anal. Chem. 1976, 59, 98–100. [Google Scholar]
- Naud, C.; Makowski, D.; Jeuffroy, M.H. Is it useful to combine measurements taken during the growing season with a dynamic model to predict the nitrogen status of winter wheat? Eur. J. Agron. 2008, 28, 291–300. [Google Scholar] [CrossRef]
- Lemaire, G.; Jeuffroy, M.H.; Gastal, F. Diagnosis tool for plant and crop N status in vegetative stage: Theory and practices for crop N management. Eur. J. Agron. 2008, 28, 614–624. [Google Scholar] [CrossRef]
- Ma, X.M.; Wang, Z.Q.; Wang, X.C.; Wang, S.L. Effects of nitrogen forms on roots and N fertilizer efficiency of different wheat cultivars with specialized end-uses. Chin. J. Appl. Ecol. 2004, 15, 655–658. [Google Scholar]
- Zhu, X.K.; Guo, W.S.; Feng, C.N.; Peng, Y.X.; Ling, Q.H. Nitrogen absorption and utilization differences among wheat varieties for different end uses. Plant Nutr. Fertil. Sci. 2005, 11, 148–154. [Google Scholar]
- Tian, J.C. Studies on the Difference of Nitrogen Absorption, Transportation and Distribution in High and Low Protein Wheat Cultivars. Acta Agronomica Sinica 1994, 20, 76–83. [Google Scholar]
- Ata-Ul-Karim, S.T.; Zhu, Y.; Cao, Q.; Rehmani, M.I.A.; Cao, W.X.; Tang, L. In-season assessment of grain protein and amylose content in rice using critical nitrogen dilution curve. Eur. J. Agron. 2017, 90, 139–151. [Google Scholar] [CrossRef]
- Chen, P.F.; Haboudane, D.; Tremblay, N.; Wang, J.H.; Vigneault, P.; Li, B.G. New spectral indicator assessing the efficiency of crop nitrogen treatment in corn and wheat. Remote Sens. Environ. 2010, 114, 1987–1997. [Google Scholar] [CrossRef]
- Ata-Ul-Karim, S.T.; Yao, X.; Liu, X.J.; Cao, W.X.; Zhu, Y. Development of critical nitrogen dilution curve of Japonica rice in Yangtze River Reaches. Field Crops Res. 2013, 149, 149–158. [Google Scholar] [CrossRef]
- Liang, X.G.; Zhang, J.T.; Zhou, L.L.; Li, X.H.; Zhou, S.L. Critical Nitrogen Dilution Curve and Nitrogen Nutrition Index for Summer Maize in North China Plain. Acta Agronomica Sinica 2013, 39, 292–299. [Google Scholar] [CrossRef]
| Exp. No. | Season and Site | Cultivar | Soil Characteristics | Treatments | Sampling Dates (Days after Planting (DAP)) |
|---|---|---|---|---|---|
| Exp. 1 | 2015−2016 Zhengzhou | Zhoumai 22 | Type: fluvo-aquic soil, OM①: 17.26 kg−1, Soil pH (CaCl2): 7.58, TN②: 1.04 g kg−1, AN③: 111.37 mg kg−1, AP④: 30.72 mg kg−1, AK⑤: 128.36 mg kg−1 | N rate (kg ha−1): N0, N90, N180, N270, N360, Irrigation regime (m3 hm−2): rainfed and irrigation (750) | 23-Feb (RS), 1-Mar, 8-Mar, 15-Mar (JS), 22-Mar, 29-Mar, 5-Apr, 12-Apr, 19-Apr (AS), 28-Apr (131, 138, 145, 152, 159, 166, 173, 180, 187, 196) |
| Exp. 2 | 2016−2017 Zhengzhou | Zhoumai 27 | Type: fluvo-aquic soil, OM: 22.47 g kg−1, Soil pH (CaCl2): 7.81, TN: 1.04 g kg−1, AN: 142.32 mg kg−1, AP: 75.90 mg kg−1, AK: 152.44 mg kg−1 | Same as Exp. 1 | 18-Feb (RS), 15-Mar (JS), 25-Mar, 6-Apr, 14-Apr, 23-Apr (AS), 30-Apr (128, 155, 165, 176, 184, 193, 200)_ |
| Exp. 3 | 2016−2017 Shangshui | Zhoumai 27 | Type: lime concretion black soil, OM: 21.33 g kg−1, Soil pH (CaCl2): 7.25, TN: 1.36 g kg−1, AN: 102.58 mg kg−1, AP: 66.35 mg kg−1, AK: 173.52 mg kg−1 | Same as Exp. 1 | 19-Feb (RS), 14-Mar (JS), 28-Mar, 18-Apr, 22-Apr (AS), 28-Apr (124, 152, 166, 187, 191, 197) |
| Exp. 4 | 2017−2018 Shangshui | Zhoumai 27 | Type: lime concretion black soil, OM: 20.91 g kg−1, Soil pH (CaCl2): 8.01, TN: 1.16 g kg−1, AN: 135.45 mg kg−1, AP: 40.21 mg kg−1, AK: 150.27 mg kg−1 | Same as Exp. 1 | 25-Feb (RS), 21-Mar (JS), 2-Apr, 14-Apr, 24-Apr (AS), 29-Apr (127, 151, 163, 175, 185, 190) |
| Source of Variation | df | Leaf Dry Matter (t hm−2) | Stem Dry Matter (t hm−2) | Plant Dry Matter (t hm−2) | |||
|---|---|---|---|---|---|---|---|
| Jointing | Anthesis | Jointing | Anthesis | Jointing | Anthesis | ||
| Site (S) | 1 | 2.692 | 5.033 * | 10.901 ** | 10.819 ** | 14.811 ** | 4.766 * |
| Water regime (W) | 1 | 4.895 * | 6.141 * | 10.548 ** | 44.992 ** | 18.093 ** | 49.878 ** |
| N rate (N) | 4 | 4.757 * | 4.684 * | 5.212 * | 25.228 ** | 17.064 ** | 33.142 ** |
| S*W | 1 | 4.926 * | 3.259 | 6.253 * | 3.209 | 8.854 * | 6.769 * |
| S*N | 4 | 2.327 | 1.886 | 3.764 * | 6.813 ** | 1.153 | 7.277 ** |
| W*N | 4 | 3.561 * | 3.445 * | 4.319 * | 4.255 * | 4.568 * | 5.031 * |
| S*W*N | 4 | 2.016 | 3.384 * | 3.357 * | 4.125 * | 1.854 | 6.011 ** |
| Tissue Dry Matter (t hm−2) | Water | DAP (Days) | Zhengzhou (2016–2017) | Shangshui (2016−2017) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N0 | N90 | N180 | N270 | N360 | N0 | N90 | N180 | N270 | N360 | |||
| Leaf | Rained | 155Z(152)S | 1.257e | 1.898d | 2.202c | 2.645b | 3.053a | 1.729d | 2.282c | 2.699b | 2.849a | 2.833a |
| 193(191) | 0.702e | 1.055d | 1.426c | 2.118b | 2.427a | 1.172d | 1.914c | 2.371b | 2.693a | 2.814a | ||
| Irrigation | 155(152) | 1.909c | 2.101c | 2.615b | 2.931b | 3.276a | 1.683d | 2.162c | 2.658b | 2.867b | 3.317a | |
| 193(191) | 1.251d | 1.811c | 2.333b | 2.462b | 2.815a | 1.501c | 1.923b | 2.441a | 2.643a | 2.785a | ||
| Stem | Rained | 155(152) | 1.229c | 1.648b | 1.931a | 1.944a | 1.521b | 2.079e | 2.987d | 3.159c | 3.473b | 3.886a |
| 193(191) | 4.240e | 5.138d | 6.556c | 7.734b | 8.176a | 4.687d | 5.428c | 6.371a | 6.426a | 6.027b | ||
| Irrigation | 155(152) | 1.869b | 1.907b | 1.755b | 2.324a | 2.251a | 1.742e | 2.974c | 2.177d | 3.393b | 4.517a | |
| 193(191) | 4.702e | 6.300d | 6.945c | 7.793b | 8.913a | 4.891d | 5.897c | 5.884c | 6.759b | 7.318a | ||
| Plant | Rained | 155(152) | 2.486e | 3.580d | 4.133c | 4.597b | 4.897a | 3.808c | 5.270b | 5.858b | 6.32a | 6.720a |
| 193(191) | 5.134e | 7.192d | 8.727c | 11.164b | 13.152a | 7.199c | 10.569b | 11.171ab | 11.811a | 11.508a | ||
| Irrigation | 155(152) | 2.029e | 3.042d | 3.406c | 4.605b | 5.819a | 3.425d | 4.935c | 5.137c | 6.261b | 7.835a | |
| 193(191) | 5.968e | 7.379d | 10.389c | 12.652b | 14.872a | 9.191e | 10.231d | 11.381c | 12.015b | 13.892a | ||
| Source of Variation | df | Leaf N Concentration (%) | Stem N Concentration (%) | Plant N Concentration (%) | |||
|---|---|---|---|---|---|---|---|
| Jointing | Anthesis | Jointing | Anthesis | Jointing | Anthesis | ||
| Site (S) | 1 | 16.734 ** | 7.248 * | 30.832 ** | 5.618 * | 6.584 * | 5.721 * |
| Water regime (W) | 1 | 9.186 * | 9.891 * | 13.929 ** | 14.601 ** | 10.944 ** | 5.347 * |
| N rate (N) | 4 | 32.488 ** | 42.921 ** | 22.942 ** | 8.235 ** | 28.455 ** | 10.758 ** |
| S*W | 1 | 8.366 * | 3.172 | 11.660 ** | 5.071 * | 15.526 ** | 1.077 |
| S*N | 4 | 5.211 * | 6.105 ** | 8.618 ** | 3.053 * | 7.316 ** | 1.951 |
| W*N | 4 | 5.920 ** | 7.175 ** | 6.106 ** | 3.477 * | 6.8765 ** | 1.703 |
| S*W*N | 4 | 3.378 * | 8.082 ** | 3.025 * | 3.144 * | 5.029 * | 2.145 |
| Tissue N Concentration (%) | Water | DAP (Days) | Zhengzhou (2016–2017) | Shangshui (2016–2017) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N0 | N90 | N180 | N270 | N360 | N0 | N90 | N180 | N270 | N360 | |||
| Leaf | Rained | 155(152) | 1.497d | 2.206c | 2.738b | 3.316a | 3.392a | 2.247d | 2.972c | 3.379b | 3.692a | 3.331b |
| 193(191) | 1.969d | 2.465c | 3.082b | 3.699a | 3.636a | 2.132d | 2.768c | 3.507b | 3.783ab | 3.843a | ||
| Irrigation | 155(152) | 1.773d | 2.460c | 2.556c | 2.969b | 3.263a | 2.571d | 3.042c | 3.430b | 3.516b | 3.764a | |
| 193(191) | 2.262e | 2.937d | 3.533c | 3.809b | 4.025a | 2.480c | 3.578b | 4.083a | 4.168a | 4.185a | ||
| Stem | Rained | 155(152) | 1.369e | 1.649d | 2.129c | 2.523b | 2.752a | 0.695b | 0.783b | 1.112a | 1.213a | 1.073a |
| 193(191) | 0.483c | 0.519c | 0.667c | 0.917b | 1.217a | 0.474b | 0.568b | 1.061a | 1.069a | 1.004a | ||
| Irrigation | 155(152) | 1.548d | 2.127c | 2.521b | 2.940a | 3.041a | 0.726e | 1.156d | 1.433c | 2.136a | 1.844b | |
| 193(191) | 0.658c | 0.729c | 1.305b | 1.628a | 1.678a | 0.641c | 1.072b | 1.148b | 1.227b | 1.482a | ||
| Plant | Rained | 155(152) | 1.534d | 1.877c | 2.453b | 2.856a | 2.712a | 1.935d | 2.603c | 3.053ab | 3.231a | 2.952b |
| 193(191) | 0.952c | 1.277b | 1.440b | 1.927a | 1.886a | 0.944c | 1.391b | 1.73a | 1.883a | 1.868a | ||
| Irrigation | 155(152) | 1.682d | 2.357c | 2.789b | 2.803b | 3.328a | 2.106d | 2.965c | 3.129bc | 3.181b | 3.398a | |
| 193(191) | 1.021e | 1.346d | 1.654c | 1.904b | 2.134a | 1.620d | 1.835c | 1.916bc | 2.073ab | 2.129a | ||
| Location | Irrigation Condition | Component | Regression Equation | R2 |
|---|---|---|---|---|
| Zhengzhou | Rainfed | Leaf | y = −27.81x + 28.24 | 0.808 |
| Stem | y = −26.87x + 28.63 | 0.708 | ||
| Plant | y = −27.25x + 28.46 | 0.812 | ||
| Irrigation | Leaf | y = −32.75x + 33.33 | 0.732 | |
| Stem | y = −32.40x + 32.94 | 0.679 | ||
| Plant | y = −36.32x + 37.57 | 0.752 | ||
| Shangshui | Rainfed | Leaf | y = −29.79x + 29.42 | 0.808 |
| Stem | y = −22.85x + 23.85 | 0.708 | ||
| Plant | y = −26.78x + 28.04 | 0.826 | ||
| Irrigation | Leaf | y = −36.85x + 38.65 | 0.788 | |
| Stem | y = −23.66x + 27.14 | 0.612 | ||
| Plant | y = −34.43x + 36.59 | 0.773 | ||
| All | Leaf | y = −30.87x + 31.49 | 0.751 | |
| Stem | y = −26.41x + 28.02 | 0.652 | ||
| Plant | y = −30.81x + 32.23 | 0.763 |
| Treatment | Zhengzhou | Shangshui | ||
|---|---|---|---|---|
| 2015−2016 | 2016−2017 | 2016−2017 | 2017−2018 | |
| Rainfed | ||||
| N0 | 2675.1 d | 4091.8 d | 3650.2 d | 2011.7 d |
| N90 | 5312.5 c | 5658.6 c | 5850.4 c | 3758.5 c |
| N180 | 6025.0 b | 6000.3 b | 6733.7b | 5900.2 b |
| N270 | 7175.1 a | 6750.3 a | 7542.1 a | 6928.7 a |
| N360 | 6578.0 ab | 6308.6 ab | 6842.1 b | 6343.6 ab |
| Irrigation | ||||
| N0 | 4187.5 d | 5100.3 d | 4775.3 c | 3408.5 d |
| N90 | 5812.5 c | 7242.0 c | 5767.1 bc | 4041.8 c |
| N180 | 6475.1 b | 7650.3 b | 6525.4 ab | 6041.9 b |
| N270 | 8175.0 a | 7975.3 a | 7658.7 a | 7157.8 a |
| N360 | 7525.1 a | 7233.6 c | 6933.7 ab | 6654.1 a |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, B.-B.; Zhao, X.-H.; Meng, Y.; Liu, M.-R.; Duan, J.-Z.; He, L.; Jiao, N.-Y.; Feng, W.; Zhu, Y.-J. Establishment of Critical Nitrogen Concentration Models in Winter Wheat under Different Irrigation Levels. Agronomy 2020, 10, 556. https://doi.org/10.3390/agronomy10040556
Guo B-B, Zhao X-H, Meng Y, Liu M-R, Duan J-Z, He L, Jiao N-Y, Feng W, Zhu Y-J. Establishment of Critical Nitrogen Concentration Models in Winter Wheat under Different Irrigation Levels. Agronomy. 2020; 10(4):556. https://doi.org/10.3390/agronomy10040556
Chicago/Turabian StyleGuo, Bin-Bin, Xiao-Hui Zhao, Yu Meng, Meng-Ran Liu, Jian-Zhao Duan, Li He, Nian-Yuan Jiao, Wei Feng, and Yun-Ji Zhu. 2020. "Establishment of Critical Nitrogen Concentration Models in Winter Wheat under Different Irrigation Levels" Agronomy 10, no. 4: 556. https://doi.org/10.3390/agronomy10040556
APA StyleGuo, B.-B., Zhao, X.-H., Meng, Y., Liu, M.-R., Duan, J.-Z., He, L., Jiao, N.-Y., Feng, W., & Zhu, Y.-J. (2020). Establishment of Critical Nitrogen Concentration Models in Winter Wheat under Different Irrigation Levels. Agronomy, 10(4), 556. https://doi.org/10.3390/agronomy10040556




