1. Introduction
Potatoes play an important role in human diet worldwide and for that reason they rank fourth as one the most consumed vegetable crops in the world [
1]. The large per capita consumption can be easily justified because they are rich in carbohydrates, minerals but also contain a large amount of high quality of proteins, vitamin C, minerals and antioxidant activities [
2]. Potato is one of the major vegetable crops cultivated throughout the southern Mediterranean, with Spain and Italy being the main producers, with 2.0 and 1.3 million tons of annual production, respectively [
1]. However, during the last 10 years the farming sector has been facing a rapid decrease in total harvested area and total production in potato crop (36.7% and 19.5%, respectively), which has been partially compensated by the increasing yields due to the introduction of new high yielding genotypes and improved farming practices [
1].
Lately, there is an increased interest from potato growers in defining the optimal fertilization regimes in order to maximize total yield, while at the same time minimizing the production cost and maintaining high quality [
3,
4]. Optimizing the efficiency of nitrogen use has been suggested through various methods with the most common practice being the replacement of the single rate nitrogen fertilization through base dressing by multiple rates during the cultivation period through side dressings, depending on plant requirements and growth stage [
5,
6]. Moreover, nitrogen source (NH
4+ or NO
3−) can have a tremendous effect on potato growth and yield, while it can also affect nitrate content in tubers [
7] and nitrogen use efficiency of the crop [
8]. Other studies have shown that apart from nitrogen source, the time of application is equally important for tuber yield. In particular, NO
3− nitrogen may induce higher yields than NH
4+ nitrogen when applied before or during tuber formation, whereas after tuber formation NH
4+ nitrogen was more efficient in terms of tuber yield [
9]. Similarly, it has been suggested that application rates may also affect yield and according to many reports increased nitrogen rates could result in higher tubers yield [
10,
11], although tuber quality may also be affected [
12,
13], while excessive rates could induce late vegetation and decrease tuber bulking [
8].
Considering that nitrogen is usually applied based on empirical standards, the irrational use of fertilizers is very common aiming at higher yields without considering the environmental impact from fertilizer production and the groundwater contamination through leaching [
14,
15]. Tuber quality is also an issue, since mineral fertilization may have a strong impact on the quality of potatoes [
16], as for example nitrogen can present some serious effects on the dry matter, protein and starch content [
4].
Zeolites are hydrated alkaline aluminosilicate crystals providing numerous benefits not only to crops but also to the soil. Almost 50 types of zeolite have been recorded, among which clinoptilolite is the most common type of zeolite generally used in agricultural systems [
17]. Zeolites have a tendency to absorb NH
4+ and K
+, while they also contain some of the most fundamental macro- and micro-nutrients a crop needs [
18]. Likewise, zeolites present some serious positive effects to the crop by improving soil moisture and nitrogen availability and preventing nitrogen leaching, which leads to the prevention of quick nutrient loss and the facilitation of an adequate nutrient supply to crops [
19]. According to Ghannad et al. [
20] who studied the effect of different fertilizer programs on the quality and yield of potato, it was found that zeolite treatment as a fertilizer management practice increased the crop yield. Another suggested farming practice includes the combination of zeolite and compost which presents several advantages such as the improvement of water use efficiency and of soil properties, the increase of soil water holding capacity, and the reduction of soil and environmental pollution [
21,
22,
23].
Apart from the acquired knowledge regarding crop needs in terms of macro- and micro-nutrient fertilization, further information is needed for defining the optimal fertilizing regimes for the yield and quality of potato crop within the context of eco-friendly farming management. Taking into consideration the information referred above, it is essential to examine and exploit new fertilizing tools such as zeolite and manure which may replace and/or substitute conventional fertilizers in order to achieve higher yield without compromising the quality of the final product. Therefore, the aim of the present study was to evaluate the effect of different fertilization regimes on total yield, quality characteristics and chemical composition of two potato varieties cultivated in central Greece.
3. Results and Discussion
Total yield of each variety in relation to the fertilizer regimes is presented in
Table 1. There were statistically significant differences between the two varieties for all the tested fertilizer treatments. These differences could be attributed to seed (tuber) size, since it is well established that total yield is positively correlated with tuber size [
32,
33]. In our study, Kennebec variety seeds were larger than the Spunta ones, which explains the significant differences in yield. Regarding the varietal response to the fertilization regime, the treatment of slow release nitrogen fertilizer (T4) provided the highest yield for both varieties. Similarly to our study, Zareabyaneh and Bayatvarkeshi [
34] studied the effects of slow-release nitrogen fertilizers on nitrate leaching and its distribution in soil profile, N-use efficiency, and the tuber yield in potato crop, and they reported that slow release nitrogen fertilizers resulted in higher yield than the urea treatment. A possible explanation for this result could be the lower nitrate leaching and the higher nitrate availability in the case where slow release fertilizers were used compared to conventional ureic fertilizers. In addition, the application of standard fertilizer, with or without zeolite added, resulted in significantly lower yield than the slow release treatment and higher yield than manure (T3) and the control treatment (C) for both varieties. However, according to Zebarth et al. [
35] nitrogen accumulation and tuber yield are also associated with environmental factors such as the air temperature, the water availability, the growth stage and the intercepted irradiation and soil properties as well. Yield response to nitrogen fertilization is more related with tuber size which increases with increasing nitrogen rates and less with tuber number which shows varied response, while excessive nitrogen rates may have the opposite effect [
36,
37,
38,
39].
Apart from fertilizer rate, timing of nitrogen fertilization is equally important and appropriate recommendations for crop management are essential for achieving high tuber yields [
40]. However, nitrogen source is also very important for the achievement of high yields since the form of the applied nitrogen and its availability may affect plant biomass partitioning and allocation of assimilates in tubers [
41]. Mirdad [
42], who carried out a field study comparing the impact of organic (chicken manure) and mineral fertilizers on tuber yield of two potato cultivars (Nicola and Diamond), reported significant differences in tuber yield between the tested cultivars, although both of them showed the highest yield when moderate levels of organic (15 tons/ha) and mineral fertilization (300-150-150 kg/ha of N-P-K) were applied. Moreover, according to Wilkinson et al. [
43] the use of fertilizers containing stabilized urea-nitrogen may increase tuber yield through the increased plant growth and nitrogen use efficiency compared to non-stabilized and conventional fertilizers.
Dry matter contents (%) of leaves, shoots and tubers are presented in
Table 2. According to the results, statistically significant differences were observed between the fertilizer treatments and the studied varieties. Regarding the Kennebec variety, the highest amount of dry matter in leaves and tubers was achieved by control (C) and standard fertilizer treatment (T1), while the lowest one was occurred by treatments T1, T2 respectively. Different trends were observed in dry matter allocation for Spunta variety where the highest tuber DM content was observed for the T2 and T3 treatments, while T2 and T4 treatments resulted in the highest DM content in shoots and leaves, respectively. Overall, higher amounts of dry matter were observed in Spunta plant parts (leaves and shoots) regardless of the fertilization regime, whereas in tubers the highest dry matter content was observed in Spunta tubers and for T2 and T3 treatments, followed by T2 treatment.
These differences could be attributed to differences in tuber maturation earliness between the studied genotypes (recommendations of 105 DAP for harvest in the case Spunta and 110–130 DAP in the case of Kennebec) and the use of conventional nitrogen fertilizers (T1, T2 and T3 treatments) compared to slow release ones (T4), as well as to growing conditions that affected plant senescence (withering of aerial parts in the case of Spunta). According to Selladurai and Purakayastha [
44], dry matter content of potato plant tissues (shoots and tubers) may differ depending on the fertilizer regime and organic fertilizers could increase tuber dry matter, as it was observed in our study in the case of Spunta variety. Similarly, Mirdad [
42] reported a significant interaction of genotype and fertilization regime effects on tuber and foliage dry weight. Moreover, dry matter content of tubers may affect the end use of the product with tubers having low values (18%–20%) of dry matter being more suitable for cooking and less susceptible to mechanical bruising, whereas tubers with high dry matter content (>20%) are suitable for processing [
45]. Therefore, apart from the selection of the proper variety the adjustment of fertilization regime can be a cost-effective means to increase the high added of the final product through the regulation of tubers dry matter content [
13,
46].
The proximate composition and the energetic value of the studied potato varieties are presented in
Table 3 showing a significant effect of the fertilization treatment and the genotype on the tested parameters. Kennebec plants treated with the standard fertilizer (T1) had the highest fat content 3.1 g/kg fw). Protein content was beneficially affected by T4 and T1 treatments in the case of Spunta variety (26.0 and 25.9 g/kg fw, respectively), followed by T1 treatment for Kennebec (2.7 g/kg fw). Ash and carbohydrates content was the highest in T1 and T3 treatments (13 and 221.2 g/kg fw, respectively) for Spunta variety, with the latter treatment (T3 for Spunta variety) having also the highest calorific value (987 kcal/kg fw). According to Naz et al. [
47] who evaluated the effect of different levels of NPK fertilizers on the proximate composition of a potato crop grown at Abbottabad, fertilizer rates may have a significant effect on proximate composition of potato tubers, while similar results were reported by Ukom et al. [
48] about the effect of nitrogen fertilizers on sweet potato crop. In addition, Ahmed et al. [
49] reported a significant variation in carbohydrates and protein content under different nitrogen fertilization managements (different doses and sources of nitrogen), while the early study of Millard [
50] indicated the significant effect of nitrogen fertilizers nitrogen and dry matter content of potato tubers.
The free sugars composition results are listed in
Table 4. Sucrose, glucose and fructose were the only detected sugars in both varieties and for all the fertilizer treatments with great variation being detected. The same sugars were detected by Galdón [
51] who also indicated the variability in sugars composition among different potato cultivars. Standard fertilizer (T1) resulted in the highest content of glucose, fructose and total sugars content in the case of Spunta variety (3.3, 0.78 and 13.7 g/kg fw), while sucrose content was the highest in T3 treatment for the same variety (11.4 g/kg fw). On the other hand, the lowest contents of the detected sugars were observed in Kennebec variety and the control (0.3 and 0.06 g/kg fw of glucose and fructose, respectively) and T1 treatments (2.33 and 3.1 g/kg fw of sucrose and total sugars, respectively). Based on these results, the standard fertilizer treatment increased the reducing sugars content compared to the rest of the fertilizer treatments in Spunta potatoes suggesting a lower nitrogen availability, since according to De Wilde [
52] lower nitrogen availability resulted in higher content of reducing sugars (fructose and glucose) in three potato varieties, especially under nitrogen deprivation (no nitrogen). The fact that in our study the tested cultivars showed a different reducing sugars content in the control treatment where no nitrogen was added could be associated with tuber maturity at the day of harvest or tuber size [
53], since maturation is a complex process involving several parameters which usually do coincide e.g., senescence, sin set, dry matter accumulation and decrease of sucrose content [
54,
55,
56]. Sugars composition fluctuates during the tuber development with high levels of fructose being detected in immature tubers, then reducing sugars content reduces during development and increases again to moderate levels just before maturation [
57]. Sucrose content is an index of tuber maturity and the lower the content the more mature the tuber is [
58,
59]. Moreover, varietal differences in free sugars composition of tubers have been previously reported by Choi et al. [
60] and Silva et al. [
61]) which supports the observed variation in our study. In terms of quality, high free sugars content is not a desirable feature for potatoes destined for processing e.g., dehydrated or fried products, since it affects the taste of the final product, while high sucrose content at harvest is not suggested for storing tubers intended for processing [
62]. Moreover, high reducing sugars content is also a negative feature for potato processing, since they are correlated with acrylamide, a carcinogenic compound formed in the Maillard reaction during processing [
63,
64]. Therefore, it seems that the application of slow release fertilizers induced maturity of Spunta variety tubers as also indicated by the low dry matter content (see results in
Table 2), while it also enhanced their suitability for processing and storage.
The organic acids composition in relation to fertilizer regime and genotype is presented in
Table 5. The main detected organic acids were citric, oxalic and malic acid, followed by ascorbic acid and traces of fumaric acid, while total organic acids content ranged between 7.5 and 11.96 g/kg fw. Citric and malic acid were also reported as the main organic acids by Lisińska and Aniołowski [
65], while Wichrowska et al. [
66] and Bushway et al. [
67] detected tartaric, fumaric, oxalic, citric, ascorbic and malic acid with significant varietal differences in individual organic acids content. Significant differences in organic acids composition between various potato cultivars were observed by Galdón et al. [
51], who also highlighted the interrelation of organic acids and sugars content, since the former are converted to sugars during tuber maturation. Organic acids composition of tubers depends on maturity stage and the content of the main compounds such as citric and malic acid fluctuates and reduces with maturation, respectively [
57]. Moreover, a varied response of the tested varieties to fertilization regime was observed in the present study, with significant differences in individual and total organic acids content among the various treatments. The application of slow release nitrogen (T4) and standard (T1) fertilizers resulted in the highest oxalic and malic acid content for Spunta variety, respectively, while manure application (T3) increased ascorbic and citric acid content for the same variety. Likewise, manure treatment (T3) increased ascorbic acid content for Kennebec potatoes, while T1 treatment resulted in the highest citric and total organic acids content. According to Jadhav and Andrew [
68], nitrogen fertilization may affect organic acids content and acidity of tubers, as well as the quality of potato processing products, while Hamouz et al. [
69] suggested that increasing rates of nitrogen fertilization resulted in a decrease of ascorbic acid content. Therefore, high citric acids content is beneficial to potato quality due to the reduced discoloration after cooking through the chelation of free Fe cations [
70]. Moreover, Mozafar [
71] reported that excessive nitrogen rates may have a variable effect on the ascorbic acid content of food plants such as potato. On the other hand, Boydston et al. [
72] did not observe statistical differences in the ascorbic acid content under different nitrogen rate levels, but also noticed a significant year effect indicating the variable environmental effect.
The antioxidant properties of the studied samples were evaluated with four different assays and the results are presented in
Table 6. The highest antioxidant activity for all the assays was recorded in extracts from the Spunta variety, except for the DPPH assay where extracts from Kennebec tubers (T3 treatment) had the lowest EC
50 values (5.43 mg/mL). Similarly, for the reducing power assay the highest antioxidant activity was detected for Spunta varieties that received T2 treatment, while the extracts from the control, T2 and T4 treatments showed the highest activity for the β-carotene/linoleate and the lipid peroxidation (TBARS) assays. According to the literature, fertilization rates may affect antioxidant properties of colored-flesh potato tubers through the increase of polyphenols content such as chlorogenic acid and anthocyanins [
16]. In contrast, Boydston et al. [
72] estimated the antioxidant capacity of two potato varieties without recording any significant differences for the different tested nitrogen rates. Moreover, Seijo-Rodríguez et al. [
73] evaluated the antioxidant activity and total phenols and flavonoids content in tubers of 35 potato varieties with varied skin and flesh color and reported a great variation in the tested parameters among the tested varieties, as well a strong correlation of antioxidant activities with polyphenols content. Similarly, Andre et al. [
74] found a strong correlation between the total phenolics content and the hydrophilic antioxidant capacity of 74 Andean potato cultivars. However, several other bioactive compounds such as carotenoids and ascorbic acid may have a synergistic effect on the overall antioxidant activity of potato tubers making it difficult to unravel the impact of fertilizers [
16,
75].
The composition of total carotenoids content and chlorophylls in the tested potatoes varieties is presented in
Table 7. Manure treatment (T3) benefited total carotenoids content in Kennebec and Spunta tubers. Regarding chlorophylls content, the control treatment resulted in the highest content of chlorophyll a and b for the Kennebec and Spunta variety, respectively.
Like in our study, the application of N-P-K fertilizers did not show any significant effects on carotenoids content of nine potato varieties, whereas a significant variation was observed among the tested varieties [
76]. Similar results were reported by Tierno et al. [
77] who also observed significant differences in total carotenoids content (1.12 to 11.9 μg lutein equivalents per g dw) of six potato cultivars and ten breeding lines, while Burmeister et al. [
78] suggested a similar range of carotenoids for color-fleshed potatoes (2.57 to 14.77 μg of β-carotene equivalents per g dw). In contrast, Ukom et al. [
48] evaluated the carotenoids content of selected sweet potato varieties as influenced by different levels of nitrogen fertilizer and they observed an increasing trend for β-carotene with increasing nitrogen levels, although a varied response among the tested cultivars was reported. Regarding chlorophyll content, most of the studies refer to chlorophyll in leaves which increased with increasing nitrogen fertilizer and manure rates, while a genotype effect was also observed [
79,
80].
However, according to Griffiths et al. [
81], chlorophyll content in tubers is closely associated with the physiological maturity of tubers and the genotype with significant variation being observed.
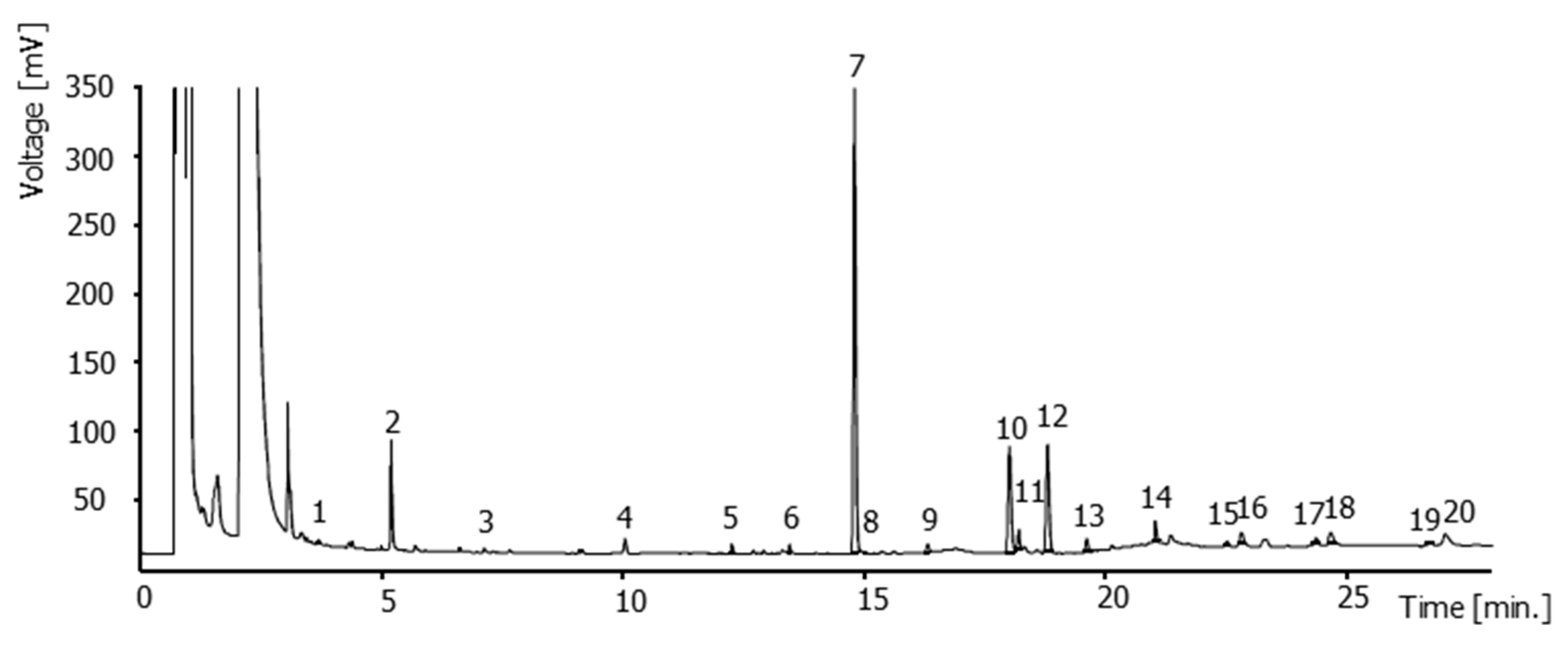
Fatty acids composition in relation to the fertilization regime and the studies variety is presented in
Table 8. Twenty fatty acids were detected in all the tested samples (
Figure 1), with palmitic acid being the most abundant one (43.67%–52.94%), followed by stearic (11.88%–16.0%), linoleic (9.96%–20.29%), oleic (2.21%–7.56%), behemic (2.30%–3.74%), and arachidic acid (1.96–2.90%). Saturated fatty acids was the most abundant class of fatty acid (69.8%–78.1%), followed by polyunsaturated (15.80%–26.16%) and monounsaturated fatty acids (3.88–9.27%). Similar results were reported by Yang and Bernards [
82] who also suggested that new fatty acids can be formed after wounding and the resulting suberization, while Uri et al. [
83] detected palmitic and stearic as the main fatty acids in six potato cultivars. In contrast, Camire et al. [
84] reported that polyunsaturated fatty acids were the most abundant class followed by saturated and monounsaturated ones (0.058, 0.035 and 0.003 g/100 g fw, respectively). Moreover, Galliard [
85] identified linoleic as the main fatty acid, while they suggested the impact of genotype and maturation stage on fatty acid composition of tubers [
86]. Therefore, the contrasting results could be attributed to differences in the studied genotypes and tubers maturity, as well as to extraction protocols. Regarding the effect of fertilization regime, a varied response was observed between the two varieties without specific trends being noticed.
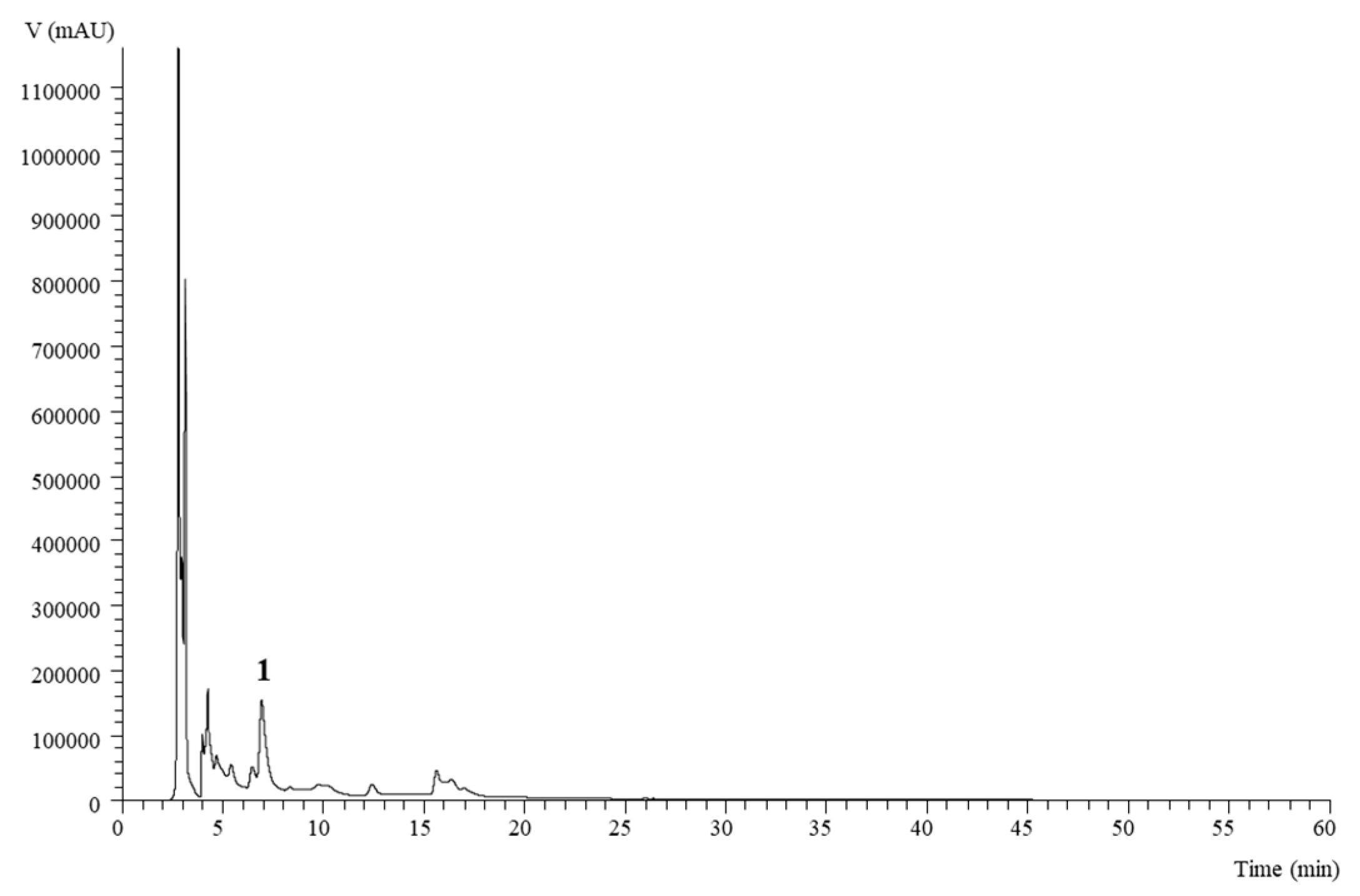
The analysis of phenolic compounds showed the presence of a single polyphenol, namely 5-O-caffeoylquinic acid or chlorogenic acid (
Table 9;
Figure 2).
This compound was identified taking into account the chromatographic characterization with the commercial standard. Chlorogenic acid was the main or the only polyphenol in tuber flesh detected in variable amounts depending on the genotype and flesh color [
74,
77,
87,
88]. The highest amount of chlorogenic acid were detected in extracts obtained from Spunta tubers subjected to the control and T1 treatment. The reports in the literature suggest a negative correlation of phenolic compounds content and nitrogen fertilization [
16,
89,
90], while no differences were observed when different farming systems (conventional and organic cultivation) were compared [
91]. In contrast, Lugasi et al. [
88] did not observe any effect of nitrogen fertilization on phenolic compounds content, while they reported chlorogenic acid content within the range of 6.0 and 22.3 mg/kg fw. Similarly, Hamouz et al. [
92] suggested that increasing rates of Mg and K may result in a decreased content of phenolic compounds of two potato varieties.