An Overview of the Oil Palm Industry: Challenges and Some Emerging Opportunities for Nanotechnology Development
Abstract
1. Introduction
2. Oil Palm Industry Challenges
2.1. Production and Nutritional Value of Palm Oil
2.2. Current Problems Faced by the Oil Palm Industry
2.3. Basal Stem Rot/Ganoderma Disease
2.4. Ganoderma Symptoms, Mode of Infection and Epidemiology
2.5. The Current Approach in Ganoderma Disease Management
- Clean clearing – the remaining fragments within the infected palm area were removed by digging a pit with 1.5 m2 and 60 cm depth [34].
- Sanitation – infected palm and stump were excavated by digging out a large hole (2 m × 2 m × 1 m) and then shredded it into smaller pieces [17].
- Digging trenches – the infected palm or stump was isolated by a digging trench with a dimension of 2 m × 2 m area (0.5 m wide and 1 m depth) to avoid the contact of infected-root and healthy-root [35].
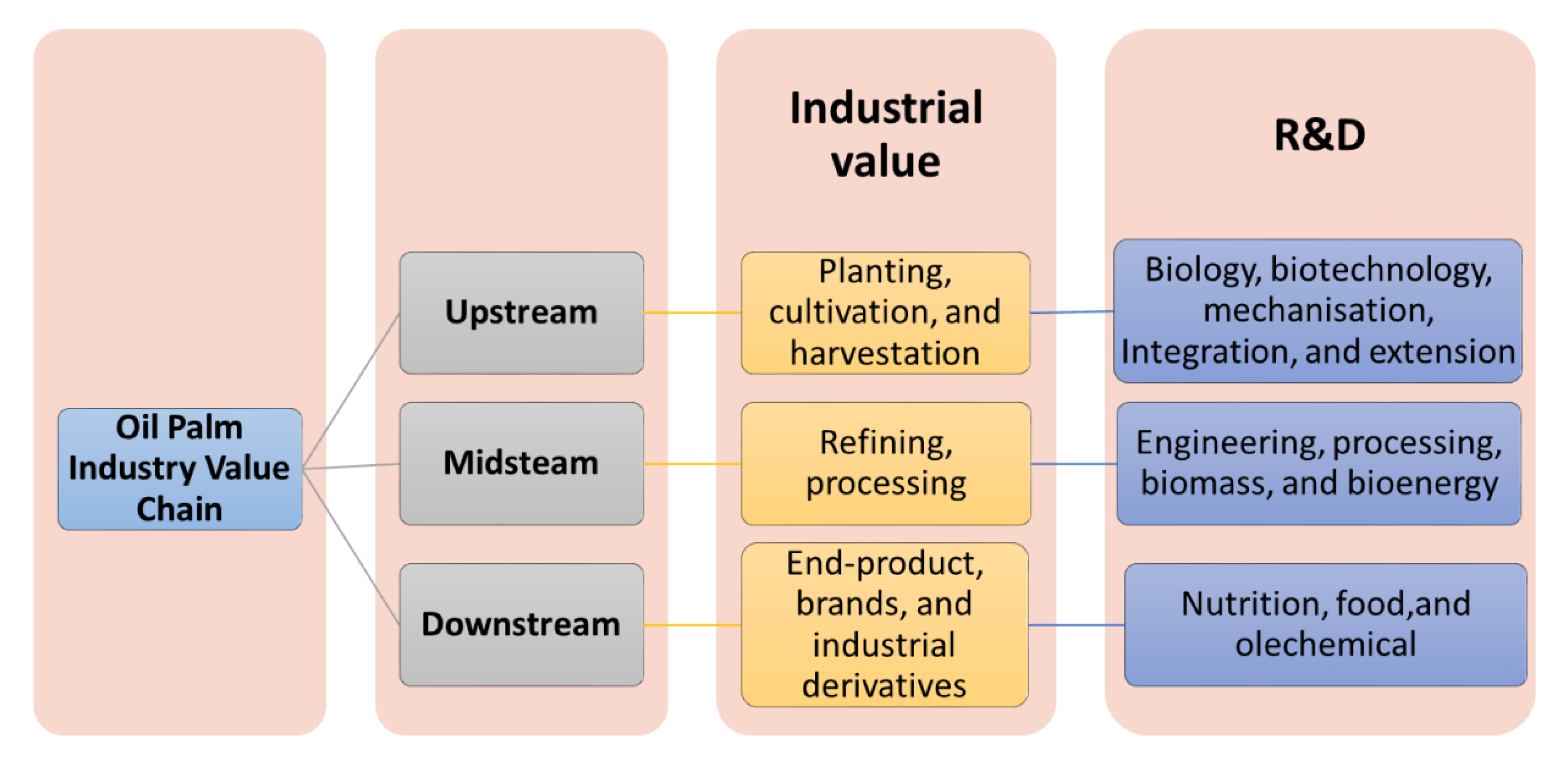
2.6. Oil Palm Industry Value Chain
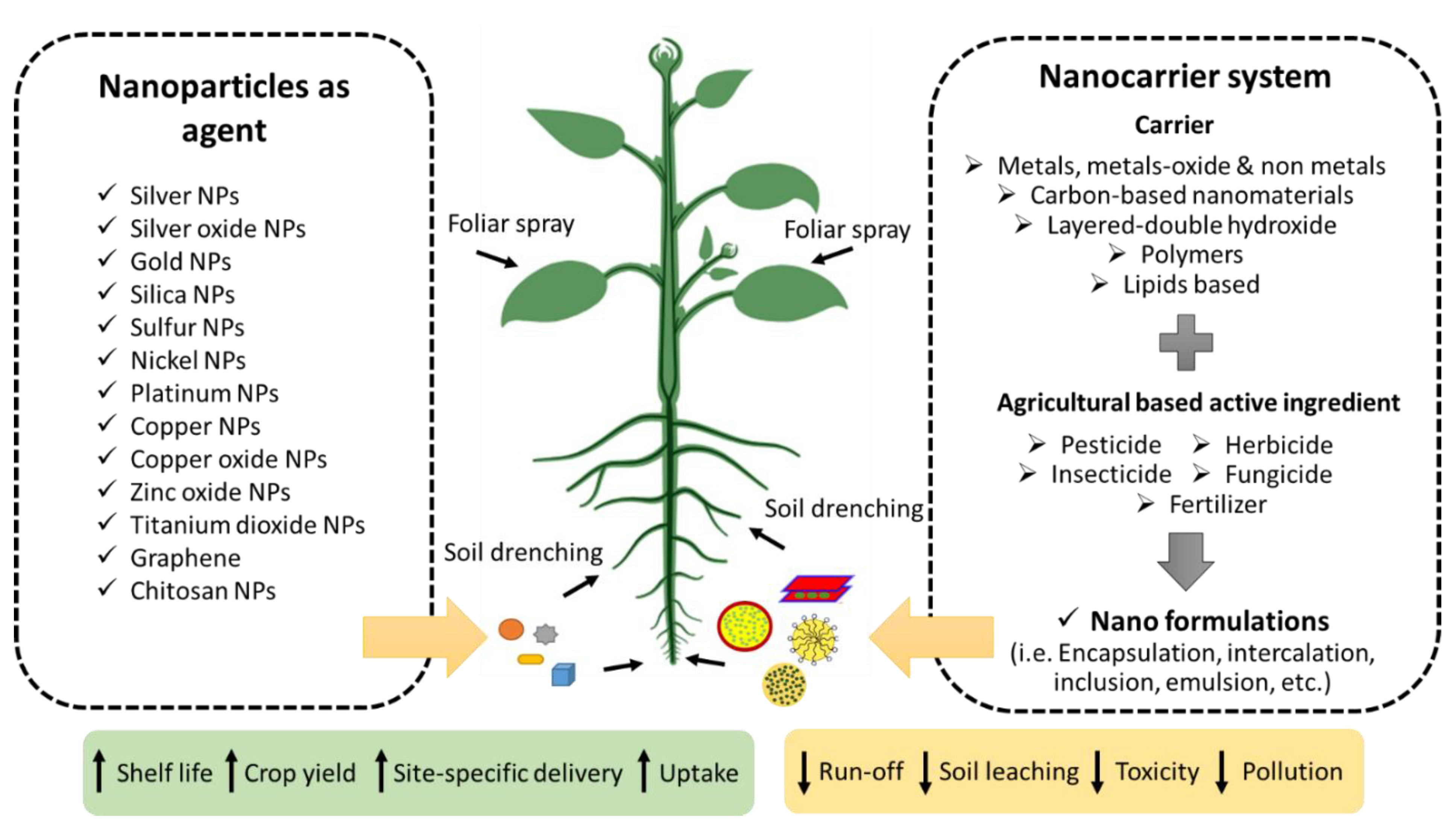
3. Nanotechnology Advancements in Agriculture
4. Recent Nanotechnology Approaches in the Oil Palm Industry
4.1. Applications of Nanotechnology in the Oil Palm Industry
4.1.1. Palm Oil Lipid-Based Nanoformulation as a Carrier System
4.1.2. Oil Palm-Based Materials in the Biosynthesis of Nanoparticles
4.1.3. Other Applications
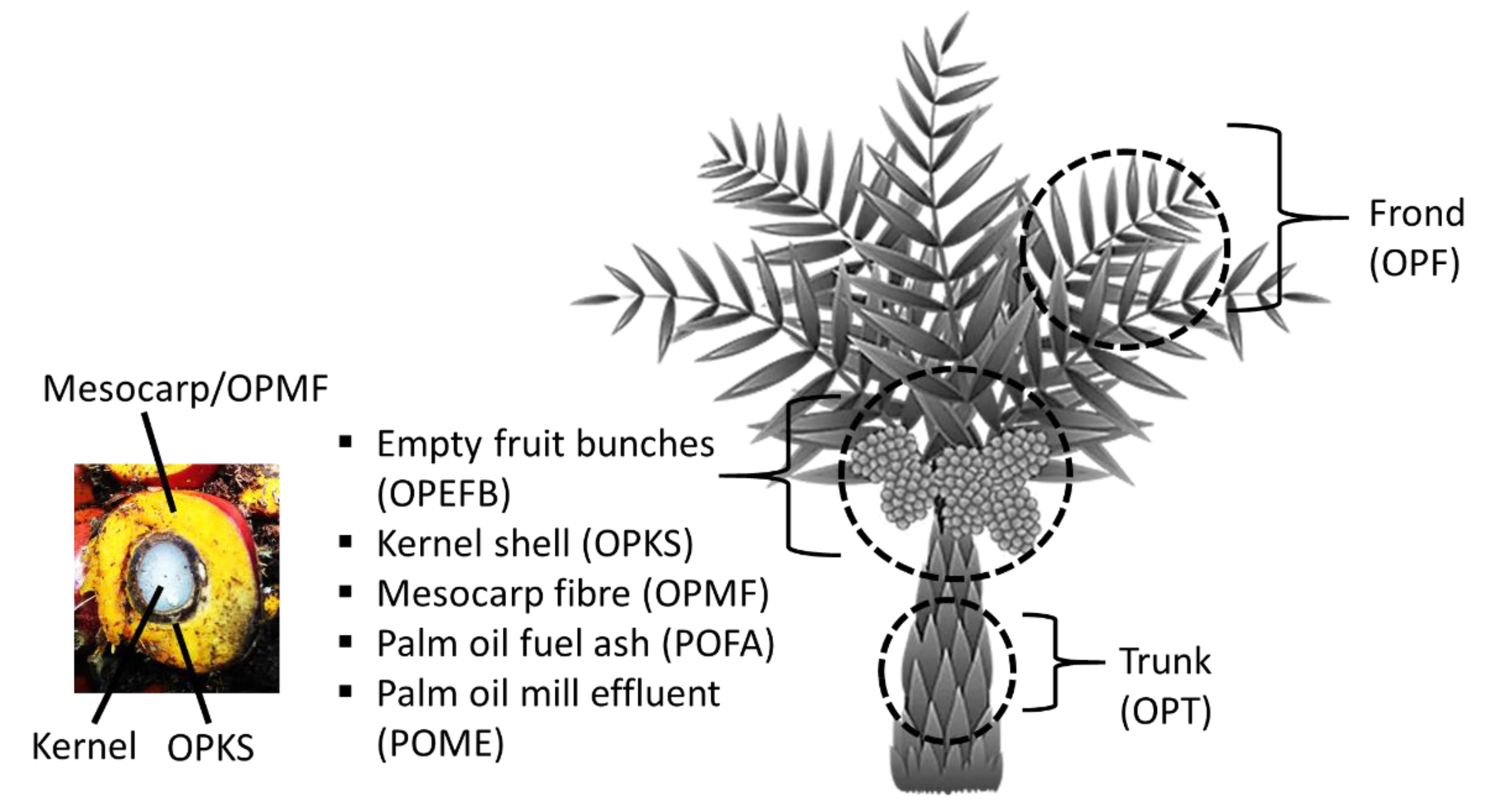
4.2. Generation of Nanomaterials from the Oil Palm Industry
4.2.1. Nanocellulose
4.2.2. Carbon-Based Nanomaterials
4.2.3. Other Nanomaterials
4.3. Nanosensor and Detection in Oil Palm Plantation
4.4. Agronanochemicals in Oil Palm Cultivation
5. Future Research Directions
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Sigit, W.; Demetria, D.H.; Yanuar, A.; Widya, K.Z.; Eko, P.; Asep, U.; Susilo, N.D.; Erni, M. Tree Crop Estate Statistic of Indonesis 2015–2017: Oil Palm; Directorate General of Estate Crops: Jakarta, Indonesia, 2016. [Google Scholar]
- Oil Palm Planted Area 2018. Available online: http://bepi.mpob.gov.my/index.php/en/statistics/area/189-area-2018/857-oil-palm-planted-area-as-at-dec-2018.html (accessed on 1 December 2018).
- Kushairi, A.; Loh, S.K.; Azman, I.; Hishamuddin, E.; Ong-Abdullah, M.; Izuddin, Z.; Razmah, G.; Sundram, S.; Parveez, G.K.A. Oil palm economic performance in Malaysia and R&D progress in 2017. J. Oil Palm Res. 2018, 30, 163–195. [Google Scholar]
- Darby, S. Palm oil facts and figures. Sime Darby Plantation: Profile and Fact Sheets; Sime Darby: Kuala Lumpur, Malaysia, 2014; pp. 1–8. [Google Scholar]
- Barcelos, E.; Rios, S.d.A.; Cunha, R.N.; Lopes, R.; Motoike, S.Y.; Babiychuk, E.; Skirycz, A.; Kushnir, S. Oil palm natural diversity and the potential for yield improvement. Front. Plant. Sci. 2015, 6, 190. [Google Scholar] [CrossRef] [PubMed]
- Wahid, M.B.; Abdullah, S.N.A.; IE, H. Oil Palm. Plant. Prod. Sci. 2005, 8, 288–297. [Google Scholar] [CrossRef]
- Nagendran, B.; Unnithan, U.; Choo, Y.; Sundram, K. Characteristics of red palm oil, a carotene-and vitamin E–rich refined oil for food uses. Food Nutr. Bull. 2000, 21, 189–194. [Google Scholar] [CrossRef]
- Kalidas, P. Pest problems of oil palm and management strategies for sustainability. Agrotech. 2012, 1, 11. [Google Scholar]
- Wood, B.; Corley, R.; Goh, K. Studies on the effect of pest damage on oil palm yield. In Proceedings of the International Oil Palm Conference, Kuala Lumpur, Malaysia, 16–18 November 1973; pp. 360–377. [Google Scholar]
- Desmier De Chenon, R.; Hasibuan, H.; Ps, S.; Purba, R.Y. Importance of food plants for parasitoids in the control of nettle caterpillars and bagworms in oil palm plantations. In Proceedings of the International Oil Palm Conference, Bali, Indonesia, 12 July 2002; pp. 520–528. [Google Scholar]
- Kalidas, P. Stress management in insect pests on oil palm, Elaeis guineensis Jacq. J. Oilseeds Res. 2004, 21, 220–223. [Google Scholar]
- Mazmira, M.; Ramlah, S.; Najib, M.; Norman, K.; Kushairi, A.; Wahid, M.B. Pest management of bagworm in southern perak by aerial spraying with Bacillus thuringiensis. Oil Palm Bulletin 2011, 63, 24–33. [Google Scholar]
- Mohamad, S.A.; Masijan, Z.; Moslim, R.; Rosman, M.; Ali, A.; Ahmad, S.N. Biological agents and insecticides to control bunch moth, Tirathaba rufivena in oil palm estates in Sarawak, Malaysia. J. Oil Palm Res. 2017, 29, 323–332. [Google Scholar] [CrossRef][Green Version]
- Harjotedjo, S.; Sahari, B.; Palgoenadi, B. Management of oil palm pests and diseases in Indonesia: learning from the past, adapting for the future. In Proceedings of the 5th MPOB-IOPRI International seminar, Kuala Lumpur, Malaysia, 2–23 November 2013; p. A21. [Google Scholar]
- Abdullah, N.; Sulaiman, F. The oil palm wastes in Malaysia. In Biomass Now-Sustainable Growth and Use; IntechOpen: Rijeka, Croatia, 2013. [Google Scholar]
- Loehr, R. Agricultural waste management: problems, processes, and approaches; Elsevier: Ithaca, NY, USA, 2012. [Google Scholar]
- Idris, A.; Kushairi, A.; Ismail, S.; Ariffin, D. Selection for partial resistance in oil palm progenies to Ganoderma basal stem rot. J. Oil Palm Res. 2004, 16, 12–18. [Google Scholar]
- Lim, T.; Chung, G.; Ko, W. Basal stem rot of oil palm caused by Ganoderma boninense. J. Plant. Pathol. 1992, 1, 147–152. [Google Scholar]
- Rao, V.; Lim, C.; Chia, C.; Teo, K. Studies on Ganoderma spread and control. Planter 2003, 79, 367–383. [Google Scholar]
- Subagio, A.; Foster, H. Implications of Ganoderma disease on loss in stand and yield production of oil palm in North Sumatra. In Proceedings of the MAPPS Conference, Kuala Lumpur, Malaysia, 18–20 August 2003. [Google Scholar]
- Durand-Gasselin, T.; Asmady, H.; Flori, A.; Jacquemard, J.C.; Hayun, Z.; Breton, F.; De Franqueville, H. Possible sources of genetic resistance in oil palm (Elaeis guineensis Jacq.) to basal stem rot caused by Ganoderma boninense–prospects for future breeding. Mycopathologia 2005, 159, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Sahebi, M.; Hanafi, M.M.; Wong, M.-Y.; Idris, A.; Azizi, P.; Jahromi, M.F.; Shokryazdan, P.; Abiri, R.; Mohidin, H. Towards immunity of oil palm against Ganoderma fungus infection. Acta Physiol. Plant. 2015, 37, 195. [Google Scholar] [CrossRef]
- Cooper, R.M.; Flood, J.; Rees, R. Ganoderma boninense in oil palm plantations: current thinking on epidemiology, resistance and pathology. Planter 2011, 87, 515–526. [Google Scholar]
- Othman, N.Q.; Paravamsivam, P.; Tan, J.S.; Lee, Y.P.; Kwan, Y.; Alwee, S.S.R.S. Validation of differential gene expression of transcriptome assembly via Nanostring® technologies analysis platform. J. Oil Palm Res. 2018, 30, 36–46. [Google Scholar] [CrossRef]
- Hushiarian, R.; Yusof, N.A.; Dutse, S.W. Detection and control of Ganoderma boninense: strategies and perspectives. SpringerPlus 2013, 2, 555. [Google Scholar] [CrossRef]
- Sanderson, F. An insight into spore dispersal of Ganoderma boninense on oil palm. Mycopathologia 2005, 159, 139–141. [Google Scholar] [CrossRef]
- Rees, R.; Flood, J.; Hasan, Y.; Potter, U.; Cooper, R.M. Basal stem rot of oil palm (Elaeis guineensis); mode of root infection and lower stem invasion by Ganoderma boninense. Plant. pathology 2009, 58, 982–989. [Google Scholar] [CrossRef]
- Rees, R.; Flood, J.; Hasan, Y.; Wills, M.A.; Cooper, R.M. Ganoderma boninense basidiospores in oil palm plantations: evaluation of their possible role in stem rots of Elaeis guineensis. Plant. Pathol. 2012, 61, 567–578. [Google Scholar] [CrossRef]
- Rees, R.W. Ganoderma stem rot of oil palm (Elaeis guineensis): mode of infection, epidemiology and biological control; University of Bath: Bath, UK, 2006. [Google Scholar]
- Ariffin, D.; Idris, A.; Singh, G. Status of Ganoderma in oil palm. Ganoderma diseases of perennial crops 2000, 49–68. [Google Scholar]
- Chong, K.P.; Dayou, J.; Alexander, A. Detection and control of Ganoderma boninense in oil palm crop; Springer: Berlin, Germany, 2017. [Google Scholar]
- Ho, C.; Khairuddin, H. Usefulness of soil mounding treatments in prolonging productivity of prime-aged Ganoderma infected palms. Planter 1997, 73, 239–244. [Google Scholar]
- Hashim, A. Ganoderma versus Mycorrhiza. Oil Palm Bulletin 2003, 6–14. [Google Scholar]
- Flood, J.; Bridge, P.; Holderness, M. Ganoderma diseases of perennial crops; CABI: Wallingford, UK, 2000. [Google Scholar]
- Lim, K.; Udin, W. Management of Ganoderma in peat soil in Indonesia. In Proceedings of the Second International Seminar on Oil Palm Diseases: Advances in Ganoderma Research and Management, Yogyakarta, Indonesia, 31 May 2010. [Google Scholar]
- Petit, A.-N.; Fontaine, F.; Vatsa, P.; Clément, C.; Vaillant-Gaveau, N. Fungicide impacts on photosynthesis in crop plants. Photosynth. Res. 2012, 111, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Langsdorf, G. Handbook of Agriculture; Yuste, M.P., Gostincar, J., Eds.; MarcelDekker, Inc.: New York, NY, USA, 1999; p. 768. [Google Scholar]
- Ramasamy, S. Cross-infectivity and decay ability of Ganoderma species parasitic to rubber, oil palm and tea. Ganoderma diseases of perennial crops; CABI Publishing: Oxon, UK, 2000. [Google Scholar]
- Idris, A.; Arifurrahman, R.; Kushairi, A. Hexaconale as a preventive treatment for managing Ganoderma in oil palm. MPOB TS Info. Ser. 2010, 75. [Google Scholar]
- Said, N.; Omar, D.; Nasehi, A.; Wong, M.Y. Pyraclostrobin suppressed Ganoderma basal stem rot (BSR), promoted plant growth and induced early expression of beta-1, 3-glucanase in oil palm (Elaeis guineensis). J. Oil Palm Res. 2019, 31, 248–261. [Google Scholar]
- Chung, G. Preliminary results on trunk injection of fungicides against Ganoderma basal stem rot in oil palm. In Proceedings of the Ganoderma workshop, Bangi, Selangor, Malaysia, 11 September 1990; pp. 81–97. [Google Scholar]
- Lim, T.; Hamm, R.; Mohamad, R. Persistency and volatile behaviour of selected chemicals in treated soil against three basidiomycetous root disease pathogens. Int. J. Pest. Manag. 1990, 36, 23–26. [Google Scholar] [CrossRef]
- Idris, A.; Ahmad, H.; Ariffin, D.; Ismail, S. Prolonging the productive life of Ganoderma-infected palms with hexaconazole. MPOB TS Info. Ser. 2004. [Google Scholar]
- Idris, A.; Arifurrahman, R.; Kushairi, D. An evaluation of hexaconazole for controlling Ganoderma basal stem rot of oil palm in the field as a preventive treatment. In Proceedings of the PIPOC (Agriculture, Biotechnology and Sustainability), Kuala Lumpur, Malaysia, 24–25 July 2009. [Google Scholar]
- Idris, A.; Maizatul, S. Stump treatment with dazomet for controlling Ganoderma disease in oil palm. MPOB TS Info. Ser. 2012, 107. [Google Scholar]
- Buana, R.F.N.; Wahyudi, A.T.; Mathius, N. Control activity of potential antifungal-producing Burkholderia sp. in suppressing Ganoderma boninense growth in oil palm. Asian J. Agric. Res. 2014, 8, 259–268. [Google Scholar] [CrossRef]
- Ramli, N.R.; Mohamed, M.S.; Seman, I.; Zairun, M.A.; Mohamad, M. The potential of endophytic bacteria as a biological control agent for Ganoderma disease in oil palm. Sains Malays. 2016, 45, 401–409. [Google Scholar]
- Sundram, S.; Meon, S.; Seman, I.A.; Othman, R. Application of Arbuscular mycorrhizal fungi with Pseudomonas aeruginosa UPMP3 reduces the development of Ganoderma basal stem rot disease in oil palm seedlings. Mycorrhiza 2015, 25, 387–397. [Google Scholar] [CrossRef] [PubMed]
- Azizah, S.N.; Mubarik, N.R.; Sudirman, L.I. Potential of chitinolytic Bacillus amyloliquefaciens SAHA 12.07 and Serratia marcescens KAHN 15.12 as biocontrol agents of Ganoderma boninense. Res. J. Microbiol. 2015, 10, 452–465. [Google Scholar]
- Alexander, A.; Phin, C.K. Combination of biological agents in suppressing colonization of Ganoderma boninense of basal stem rot. Am.-Eurasian J. Sustain. Agric. 2014, 8, 1–7. [Google Scholar]
- Sundram, S.; Abdullah, F.; Ahmad, Z.A.M.; Yusuf, U.K. Efficacy of single and mixed treatments of Trichoderma harzianum as biocontrol agents of Ganoderma basal stem rot in oil palm. J. Oil Palm Res. 2008, 20, 470–483. [Google Scholar]
- Najihah, N.I.; Hanafi, M.M.; Idris, A.S.; Hakim, M.A. Silicon treatment in oil palms confers resistance to basal stem rot disease caused by Ganoderma boninense. Crop. Prot. 2015, 67, 151–159. [Google Scholar] [CrossRef]
- Bivi, M.S.H.R.; Paiko, A.S.; Khairulmazmi, A.; Akhtar, M.; Idris, A.S. Control of basal stem rot disease in oil palm by supplementation of calcium, copper, and salicylic acid. Plant. Pathol. J. 2016, 32, 396. [Google Scholar] [CrossRef] [PubMed]
- Fabien, F.T.; Hanafi, M.M.; Idris, A.; Kadir, J.; Jamaludin, N.; Mohidin, H.; Syed, R.S.O. Effect of micronutrients-enriched fertilizers on basal stem rot disease incidence and severity on oil palm (Elaeis guineensis Jacq.) seedlings. Am. J. Appl. Sci. 2014, 11, 1841–1859. [Google Scholar]
- Nur Sabrina, A.; Sariah, M.; Zaharah, A. Suppression of basal stem rot disease progress in oil palm (Elaeis guineensis) after copper and calcium supplementation. Pertanika J. Trop. 2012, 35. [Google Scholar]
- Singh, I.; Nair, R.S.; Gan, S.; Cheong, V.; Morris, A. An evaluation of crude palm oil (CPO) and tocotrienol rich fraction (TRF) of palm oil as percutaneous permeation enhancers using full-thickness human skin. Pharm. Dev. Technol. 2018, 1–7. [Google Scholar] [CrossRef]
- Mekhilef, S.; Siga, S.; Saidur, R. A review on palm oil biodiesel as a source of renewable fuel. Renew. Sust. Energy Rev. 2011, 15, 1937–1949. [Google Scholar] [CrossRef]
- Roco, M.C. Nanoparticles and nanotechnology research. J. Nanopart. Res. 1999, 1, 1–6. [Google Scholar] [CrossRef]
- Worrall, E.; Hamid, A.; Mody, K.; Mitter, N.; Pappu, H. Nanotechnology for plant disease management. Agronomy 2018, 8, 285. [Google Scholar] [CrossRef]
- Singh, N.; Amist, N.; Yadav, K.; Singh, D.; Pandey, J.; Singh, S. Zinc oxide nanoparticles as fertilizer for the germination, growth and metabolism of vegetable crops. J. Nanoeng. Nanomanuf. 2013, 3, 353–364. [Google Scholar] [CrossRef]
- Elmer, W.; White, J.C. The future of nanotechnology in plant pathology. Annu. Rev. Phytopathol. 2018, 56, 111–133. [Google Scholar] [CrossRef] [PubMed]
- Jo, Y.K.; Kim, B.H.; Jung, G. Antifungal activity of silver ions and nanoparticles on phytopathogenic fungi. Plant. Dis. 2009, 93, 1037–1043. [Google Scholar] [CrossRef] [PubMed]
- Ouda, S.M. Antifungal activity of silver and copper nanoparticles on two plant pathogens, Alternaria alternata and Botrytis cinerea. Res. J. Microbiol. 2014, 9, 34–42. [Google Scholar] [CrossRef]
- Mohammadinejad, R.; Karimi, S.; Iravani, S.; Varma, R.S. Plant-derived nanostructures: types and applications. Green Chem. 2016, 18, 20–52. [Google Scholar] [CrossRef]
- Singh, P.; Kim, Y.J.; Zhang, D.; Yang, D.C. Biological synthesis of nanoparticles from plants and microorganisms. Trends Biotechnol. 2016, 34, 588–599. [Google Scholar] [CrossRef]
- Deepa, B.; Abraham, E.; Cordeiro, N.; Mozetic, M.; Mathew, A.P.; Oksman, K.; Faria, M.; Thomas, S.; Pothan, L.A. Utilization of various lignocellulosic biomass for the production of nanocellulose: a comparative study. Cellulose 2015, 22, 1075–1090. [Google Scholar] [CrossRef]
- Momodu, D.; Okafor, C.; Manyala, N.; Bello, A.; ZebazeKana, M.G.; Ntsoenzok, E. Transformation of plant biomass waste into resourceful activated carbon nanostructures for mixed-assembly type electrochemical capacitors. Waste Biomass Valori. 2017, 1–13. [Google Scholar] [CrossRef]
- Shams, S.S.; Zhang, L.S.; Hu, R.; Zhang, R.; Zhu, J. Synthesis of graphene from biomass: a green chemistry approach. Mater. Lett. 2015, 161, 476–479. [Google Scholar] [CrossRef]
- Chen, F.; Yang, J.; Bai, T.; Long, B.; Zhou, X. Facile synthesis of few-layer graphene from biomass waste and its application in lithium ion batteries. J. Electroanal. Chem. 2016, 768, 18–26. [Google Scholar] [CrossRef]
- Sun, Z.; Zheng, M.; Hu, H.; Dong, H.; Liang, Y.; Xiao, Y.; Lei, B.; Liu, Y. From biomass wastes to vertically aligned graphene nanosheet arrays: A catalyst-free synthetic strategy towards high-quality graphene for electrochemical energy storage. Chem. Eng. J. 2018, 336, 550–561. [Google Scholar] [CrossRef]
- Mahmoudian, L.; Rashidi, A.; Dehghani, H.; Rahighi, R. Single-step scalable synthesis of three-dimensional highly porous graphene with favorable methane adsorption. Chem. Eng. J. 2016, 304, 784–792. [Google Scholar] [CrossRef]
- Kaushal, M.; Wani, S.P. Nanosensors: frontiers in precision agriculture. In Nanotechnology; Springer: Singapore, 2017; pp. 279–291. [Google Scholar]
- Wang, Z.; Wei, F.; Liu, S.Y.; Xu, Q.; Huang, J.Y.; Dong, X.Y.; Yu, J.H.; Yang, Q.; Zhao, Y.D.; Chen, H. Electrocatalytic oxidation of phytohormone salicylic acid at copper nanoparticles-modified gold electrode and its detection in oilseed rape infected with fungal pathogen Sclerotinia sclerotiorum. Talanta 2010, 80, 1277–1281. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tong, Z.; Prud’homme, R.K. Stabilized polymeric nanoparticles for controlled and efficient release of bifenthrin. Pest. Manag. Sci. 2008, 64, 808–812. [Google Scholar] [CrossRef]
- Efendy Goon, D.; Kadir, S.A.; Hamimah, S.; Latip, N.A.; Mazlan, M. Palm oil in lipid-based formulations and drug delivery systems. Biomolecules 2019, 9, 64. [Google Scholar] [CrossRef]
- Brownlow, B.; Nagaraj, V.J.; Nayel, A.; Joshi, M.; Elbayoumi, T. Development and in vitro evaluation of vitamin E-enriched nanoemulsion vehicles loaded with genistein for chemoprevention against UVB-induced skin damage. J. Pharm. Sci. 2015, 104, 3510–3523. [Google Scholar] [CrossRef]
- Ng, S.H.; Woi, P.M.; Basri, M.; Ismail, Z. Characterization of structural stability of palm oil esters-based nanocosmeceuticals loaded with tocotrienol. J. Nanobiotechnology 2013, 11, 27. [Google Scholar]
- Abdullah, G.Z.; Abdulkarim, M.F.; Salman, I.M.; Ameer, O.Z.; Yam, M.F.; Mutee, A.F.; Chitneni, M.; Mahdi, E.S.; Basri, M.; Sattar, M.A. In vitro permeation and in vivo anti-inflammatory and analgesic properties of nanoscaled emulsions containing ibuprofen for topical delivery. Int. J. Nanomedicine 2011, 6, 387. [Google Scholar] [CrossRef]
- Salim, N.; Basri, M.; Rahman, M.B.; Abdullah, D.K.; Basri, H. Modification of palm kernel oil esters nanoemulsions with hydrocolloid gum for enhanced topical delivery of ibuprofen. Int. J. Nanomedicine 2012, 7, 4739. [Google Scholar] [PubMed]
- Abu-Fayyad, A.; Kamal, M.M.; Carroll, J.L.; Dragoi, A.-M.; Cody, R.; Cardelli, J.; Nazzal, S. Development and in-vitro characterization of nanoemulsions loaded with paclitaxel/γ-tocotrienol lipid conjugates. Int. J. Pharm. 2018, 536, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Ramli, S.; Norhman, N.; Zainuddin, N.; Ja’afar, S.M.; Rahman, I.A. Nanoemulsion based palm olein as vitamin E carrier. Malays. J. Analytical Sci. 2017, 21, 1399–1408. [Google Scholar]
- Sakeena, M.H.F. Anti-inflammatory and analgesic effects of ketoprofen in palm oil esters nanoemulsion. J. Oleo Sci. 2010, 59, 667–671. [Google Scholar]
- Asmawi, A.A.; Salim, N.; Ngan, C.L.; Ahmad, H.; Abdulmalek, E.; Masarudin, M.J.; Abdul Rahman, M.B. Excipient selection and aerodynamic characterization of nebulized lipid-based nanoemulsion loaded with docetaxel for lung cancer treatment. Drug Deliv. Transl. Res. 2018, 1–12. [Google Scholar] [CrossRef]
- Raviadaran, R.; Chandran, D.; Shin, L.H.; Manickam, S. Optimization of palm oil in water nano-emulsion with curcumin using microfluidizer and response surface methodology. LWT-Food Sci. Technol. 2018, 96, 58–65. [Google Scholar] [CrossRef]
- Zainol, S.; Basri, M.; Basri, H.B.; Shamsuddin, A.F.; Abdul-Gani, S.S.; Karjiban, R.A.; Abdul-Malek, E. Formulation optimization of a palm-based nanoemulsion system containing levodopa. Int. J. Mol. Sci. 2012, 13, 13049–13064. [Google Scholar] [CrossRef]
- Masoumi, H.R.F.; Basri, M.; Samiun, W.S.; Izadiyan, Z.; Lim, C.J. Enhancement of encapsulation efficiency of nanoemulsion-containing aripiprazole for the treatment of schizophrenia using mixture experimental design. Int. J. Nanomedicine 2015, 10, 6469. [Google Scholar] [CrossRef]
- Musa, S.H.; Basri, M.; Masoumi, H.R.F.; Karjiban, R.A.; Malek, E.A.; Basri, H.; Shamsuddin, A.F. Formulation optimization of palm kernel oil esters nanoemulsion-loaded with chloramphenicol suitable for meningitis treatment. Colloids Surf. B Biointerfaces 2013, 112, 113–119. [Google Scholar] [CrossRef]
- Abedi Karjiban, R.; Basri, M.; Abdul Rahman, M.B.; Salleh, A.B. Molecular dynamics simulation of palmitate ester self-assembly with diclofenac. Int. J. Mol. Sci. 2012, 13, 9572–9583. [Google Scholar] [CrossRef]
- Irfan, M.; Ahmad, T.; Moniruzzaman, M.; Abdullah, B. Effect of pH on ionic liquid mediated synthesis of gold nanoparticle using Elaise guineensis (palm oil) kernel extract. IOP Conf. Ser. Mater. Sci. Eng. 2017, 204, 012002. [Google Scholar] [CrossRef]
- Ong, H.R.; Khan, M.M.R.; Ramli, R.; Yunus, R.M.; Rahman, M.W. Glycerolysis of palm oil using copper oxide nanoparticles combined with homogeneous base catalyst. New J. Chem. 2016, 40, 8704–8709. [Google Scholar] [CrossRef]
- Ramimoghadam, D.; Hussein, M.Z.B.; Taufiq-Yap, Y.H. Synthesis and characterization of ZnO nanostructures using palm olein as biotemplate. Chem. Cent. J. 2013, 7, 71. [Google Scholar] [CrossRef] [PubMed]
- Zamiri, R.; Zakaria, A.; Ahangar, H.A.; Sadrolhosseini, A.R.; Mahdi, M.A. Fabrication of silver nanoparticles dispersed in palm oil using laser ablation. Int. J. Mol. Sci. 2010, 11, 4764–4770. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, T.; Irfan, M.; Bhattacharjee, S. Parametric study on gold nanoparticle synthesis using aqueous Elaise guineensis (oil palm) leaf extract: effect of precursor concentration. Procedia. Engineering 2016, 148, 1396–1401. [Google Scholar] [CrossRef]
- Ahmad, T.; Bustam, M.; Irfan, M.; Moniruzzaman, M.; Asghar, H.; Bhattacharjee, S. Effect of volume of gold chloroauric acid on size, shape and stability of biosynthesized AuNPs using aqueous Elaeis guineensis (oil palm) leaves extract. Int. J. Automot. Mech. Eng. 2018, 15. [Google Scholar] [CrossRef]
- Gan, P.P.; Ng, S.H.; Huang, Y.; Li, S.F.Y. Green synthesis of gold nanoparticles using palm oil mill effluent (POME): a low-cost and eco-friendly viable approach. Bioresour. Technol. 2012, 113, 132–135. [Google Scholar] [CrossRef]
- Usman, A.I.; Aziz, A.A.; Noqta, O.A. Bio-synthesis of triangular and hexagonal gold nanoparticles using palm oil fronds’ extracts at room temperature. Mater. Res. Express 2018, 5, 015042. [Google Scholar] [CrossRef]
- Ramli, N.A.; Jai, J.; Mohd Yusof, N.; Zamanhuri, N.A. Green synthesis of silver nanoparticles using Elaeis guineensis from palm leaves: influence of pH in reaction kinetic. Adv. Mat. Res. 2015, 1113, 560–565. [Google Scholar] [CrossRef]
- Ganesan, S.; Munuswamy, D.; Appavu, P.; Arunkumar, T.; Devaraja, Y. Effect of EGR & nanoparticles on performance and emission characteristics of a diesel engine fuelled with palm biodiesel and diesel blends. J. Oil Palm Res. 2019, 31, 130–137. [Google Scholar]
- Hasan, Z.A.A.; Idris, Z.; Gani, S.; Basri, M. In vitro safety evaluation of palm tocotrienol-rich fraction nanoemulsions for topical application. J. Oil Palm Res. 2018, 30, 150–162. [Google Scholar]
- Dungani, R.; Aditiawati, P.; Aprilia, S.; Yuniarti, K.; Karliati, T.; Suwandhi, I.; Sumardi, I. Biomaterial from Oil Palm Waste: Properties, Characterization and Applications. In Palm Oil; IntechOpen: London, UK, 2018. [Google Scholar]
- Lani, N.; Ngadi, N.; Johari, A.; Jusoh, M. Isolation, characterization, and application of nanocellulose from oil palm empty fruit bunch fiber as nanocomposites. J. Nanomater. 2014, 2014, 13. [Google Scholar] [CrossRef]
- Gea, S.; Panindia, N.; Piliang, A.; Sembiring, A.; Hutapea, Y. All-cellulose composite isolated from oil palm empty fruit bunch. J. Phys. Conf. Ser. 2018, 1116, 042013. [Google Scholar] [CrossRef]
- Okahisa, Y.; Furukawa, Y.; Ishimoto, K.; Narita, C.; Intharapichai, K.; Ohara, H. Comparison of cellulose nanofiber properties produced from different parts of the oil palm tree. Carbohyd. Polym. 2018, 198, 313–319. [Google Scholar] [CrossRef]
- Nazir, M.S.; Wahjoedi, B.A.; Yussof, A.W.; Abdullah, M.A. Eco-friendly extraction and characterization of cellulose from oil palm empty fruit bunches. BioResources 2013, 8, 2161–2172. [Google Scholar] [CrossRef]
- Al-Dulaimi, A.A.; Wanrosli, W. Isolation and characterization of nanocrystalline cellulose from totally chlorine free oil palm empty fruit bunch pulp. J. Polym. Environ. 2017, 25, 192–202. [Google Scholar] [CrossRef]
- Wibowo, A.; Madani, H.; Judawisastra, H.; Restiawaty, E.; Lazarus, C.; Budhi, Y.W. An eco-friendly preparation of cellulose nano crystals from oil palm empty fruit bunches. IOP Conf. Ser. Earth Environ. Sci. 2018, 105, 012059. [Google Scholar] [CrossRef]
- Saba, N.; Paridah, T.M.; Abdan, K.; Ibrahim, N.A. Preparation and characterization of fire retardant nano-filler from oil palm empty fruit bunch fibers. BioResources 2015, 10, 4530–4543. [Google Scholar] [CrossRef]
- Saba, N.; Tahir, P.M.; Abdan, K.; Ibrahim, N.A. Fabrication of epoxy nanocomposites from oil palm nano filler: mechanical and morphological properties. BioResources 2016, 11, 7721–7736. [Google Scholar] [CrossRef]
- Saba, N.; Jawaid, M.; Alothman, O.Y.; Almutairi, Z. Evaluation of dynamic properties of nano oil palm empty fruit bunch filler/epoxy composites. J. Mater. Res. Technol. 2018, 8, 1470–1475. [Google Scholar] [CrossRef]
- Husin, H.; Asnawi, T.; Firdaus, A.; Husaini, H.; Ibrahim, I.; Hasfita, F. Solid catalyst nanoparticles derived from oil-palm empty fruit bunches (OP-EFB) as a renewable catalyst for biodiesel production. IOP Conf. Ser. Mater. Sci. Eng. 2018, 1, 012008. [Google Scholar] [CrossRef]
- Hendriansyah, R.; Devianto, H.; Prakoso, T.; Widiatmoko, P.; Nurdin, I.; Srimurti, S.; Kusuma, K.R. Nano carbon materials from palm oil wastes for supercapacitor applications. In Proceedings of the 2017 4th International Conference on Electric Vehicular Technology (ICEVT), Bali, Indonesia, 2–5 October 2017; pp. 13–18. [Google Scholar]
- Gea, S.; Muis, Y.; Novita, T.; Piliang, A.F. Synthesis of carbon nanodots from cellulose nanocrystals oil palm empty fruit by pyrolysis method. J. Phys. Conf. 2018, 1120, 012071. [Google Scholar]
- Wafti, N.; Lau, H.L.N.; Loh, S.K.; Aziz, A.A.; Ab Rahman, Z.; May, C.Y. Activated carbon from oil palm biomass as potential adsorbent for palm oil mill effluent treatment. J. Oil Palm Res. 2017, 29, 278–290. [Google Scholar] [CrossRef][Green Version]
- Tahir, N.A.M.; Abdollah, M.F.B.; Tamaldin, N.; Amiruddin, H.; Tokoroyama, T.; Umehara, N. Potential of growing graphene from solid waste products. In Proceedings of the SAKURA Symposium on Mechanical Science and Engineering, Centre for Advanced Research on Energy, Nagoya, Japan, 8–10 November 2017; pp. 26–28. [Google Scholar]
- Nasir, S.; Hussein, M.; Yusof, N.; Zainal, Z. Oil palm waste-based precursors as a renewable and economical carbon sources for the preparation of reduced graphene oxide from graphene oxide. Nanomaterials 2017, 7, 182. [Google Scholar] [CrossRef]
- Nicholas, A.; Hussein, M.; Zainal, Z.; Khadiran, T. Palm Kernel Shell Activated Carbon as an Inorganic Framework for Shape-Stabilized Phase Change Material. Nanomaterials 2018, 8, 689. [Google Scholar] [CrossRef]
- Al Mamun, A.; Ahmed, Y.M.; Ma’an Fahmi, R.A.; Jameel, A.T.; AlSaadi, M.A. The optimum condition for the synthesis of carbon nanofibers on activated carbon to remove lead from aqueous solution. Int. J. Nanoelectron. Mater. 2019, 12, 135–144. [Google Scholar]
- Al Mamun, A.; AlSaadi, M.A.; Alam, M.Z.; Sopyan, I. Carbon nanotubes grown on oil palm shell powdered activated carbon as less hazardous and cheap substrate. Appl. Nanosci. 2018, 8, 1767–1779. [Google Scholar] [CrossRef]
- Dungani, R.; Fizree, H. Production and characterization of the defatted oil palm shell nanoparticles. Sains Malays. 2016, 45, 833–839. [Google Scholar]
- Chieng, B.; Lee, S.; Ibrahim, N.; Then, Y.; Loo, Y. Isolation and characterization of cellulose nanocrystals from oil palm mesocarp fiber. Polymers 2017, 9, 355. [Google Scholar] [CrossRef]
- Razib, M.F.M.; Wahab, R.A. Statistical optimization of Candida rugosa lipase supported on chitosan/nanocellulose composite for efficient synthesis of butyl butyrate. eProceed. Chem. 2018, 3. [Google Scholar]
- Nordin, N.A.; Sulaiman, O.; Hashim, R.; Kassim, M.H.M. Oil palm frond waste for the production of cellulose nanocrystals. J. Phys. Sci. 2017, 28, 115–126. [Google Scholar] [CrossRef]
- Mazlita, Y.; Lee, H.; Hamid, S. Preparation of cellulose nanocrystals bio-polymer from agro-industrial wastes: Separation and characterization. Polym. Polym. Compos. 2016, 24, 719–728. [Google Scholar] [CrossRef]
- Bhat, A.H.; Khalil, H.A. Exploring “nano filler” based on oil palm ash in polypropylene composites. BioResources 2011, 6, 1288–1297. [Google Scholar]
- Khalil, H.A.; Mahayuni, A.R.; Rudi, D.; Almulali, M.; Abdullah, C. Characterization of various organic waste nanofillers obtained from oil palm ash. BioResources 2012, 7, 5771–5780. [Google Scholar] [CrossRef]
- Khan, M.I.; Azizli, K.; Sufian, S.; Man, Z.; Khan, A.S. Simultaneous preparation of nano silica and iron oxide from palm oil fuel ash and thermokinetics of template removal. Rsc Adv. 2015, 5, 20788–20799. [Google Scholar] [CrossRef]
- Lim, N.H.A.S.; Samadi, M.; Hussin, M.W.; Sam, A.R.M.; Ariffin, N.F.; Ismail, M.A.; Lee, H.S.; Ariffin, M.A.M. Influence of oil palm biomass waste on compressive strength and chloride penetration of mortar. MATEC Web Conf. 2017, 138, 01008. [Google Scholar] [CrossRef][Green Version]
- Hamada, H.M.; Jokhio, G.A.; Yahaya, F.M.; Humada, A.M. Applications of Nano palm oil fuel ash and Nano fly ash in concrete. IOP Conf. Ser. Mater. Sci. Eng. 2018, 342, 012068. [Google Scholar] [CrossRef]
- Tang, W.; Lee, H.-S.; Vimonsatit, V.; Htut, T.; Singh, J.; Wan Hassan, W.; Ismail, M.; Seikh, A.; Alharthi, N. Optimization of micro and nano palm oil fuel ash to determine the carbonation resistance of the concrete in accelerated condition. Materials 2019, 12, 130. [Google Scholar] [CrossRef]
- Robaiah, M.; Rusop, M.; Abdullah, S.; Khusaimi, Z.; Azhan, H.; Asli, N.A. Synthesis graphene layer at different waste cooking palm oil temperatures. AIP Conf. Proc. 2017, 1877, 030008. [Google Scholar]
- Arie, A.A.; Hadisaputra, L.; Susanti, R.F.; Devianto, H.; Halim, M.; Enggar, R.; Lee, J. Synthesis of carbon nano materials originated from waste cooking oil using a nebulized spray pyrolysis. J. Phys. Conf. 2017, 877, 012020. [Google Scholar] [CrossRef]
- Astimar, A.A.; Kumudeswar, D.; Mohamad, H.; Anis, M. Effects of physical and chemical pre-treatments on xylose and glucose production from oil palm press fibre. J. Oil Palm Res. 2002, 14, 10–17. [Google Scholar]
- Yasim-Anuar, T.A.T.; Ariffin, H.; Norrrahim, M.N.F.; Hassan, M.A. Factors affecting spinnability of oil palm mesocarp fiber cellulose solution for the production of microfiber. BioResources 2016, 12, 715–734. [Google Scholar] [CrossRef][Green Version]
- Wang, Z.; Shen, D.; Wu, C.; Gu, S. State-of-the-art on the production and application of carbon nanomaterials from biomass. Green Chem. 2018, 20, 5031–5057. [Google Scholar] [CrossRef]
- Saba, N.; Tahir, P.; Jawaid, M. A review on potentiality of nano filler/natural fiber filled polymer hybrid composites. Polymers 2014, 6, 2247–2273. [Google Scholar] [CrossRef]
- Fowotade, S.A.; Yusof, N.A.; Abdullah, J.; Sulaiman, Y.; Rahman, S.F.A. Enhanced electrochemical sensing of secondary metabolites in oil palms for early detection of Ganoderma boninense based on novel nanoparticle-chitosan functionalized multi-walled carbon nanotube platform. Sens. Biosensing Res. 2019, 100274. [Google Scholar] [CrossRef]
- Akanbi, F.; Yusof, N.; Abdullah, J.; Sulaiman, Y.; Hushiarian, R. Detection of quinoline in G. boninense-infected plants using functionalized multi-walled carbon nanotubes: A field study. Sensors 2017, 17, 1538. [Google Scholar] [CrossRef]
- Hushiarian, R.; Yusof, N.; Abdullah, A.; Ahmad, S.; Dutse, S. A novel DNA nanosensor based on CdSe/ZnS quantum dots and synthesized Fe3O4 magnetic nanoparticles. Molecules 2014, 19, 4355–4368. [Google Scholar] [CrossRef]
- Hushiarian, R.; Yusof, N.A.; Abdullah, A.H.; Ahmad, S.A.A.; Dutse, S.W. Facilitating the indirect detection of genomic DNA in an electrochemical DNA biosensor using magnetic nanoparticles and DNA ligase. Anal. Chem. Res. 2015, 6, 17–25. [Google Scholar] [CrossRef]
- Dutse, S.W.; Yusof, N.A.; Ahmad, H.; Hussein, M.Z.; Hushiarian, R. DNA-based biosensor for detection of Ganoderma boninense, an oil palm pathogen utilizing newly synthesized ruthenium complex [Ru (phen) 2 (qtpy)] 2 based on a PEDOT-PSS/Ag nanoparticles modified electrode. Int. J. Electrochem. Sci. 2013, 8, 11048–11057. [Google Scholar]
- Dutse, S.W.; Yusof, N.A.; Ahmad, H.; Hussein, M.Z.; Zainal, Z. An electrochemical DNA biosensor for Ganoderma boninense pathogen of the oil palm utilizing a New ruthenium complex,[Ru (dppz) 2 (qtpy)] Cl2. Int. J. Electrochem. Sci. 2012, 7, 8105–8115. [Google Scholar]
- Mustafa, I.F.; Hussein, M.Z.; Saifullah, B.; Idris, A.S.; Hilmi, N.H.Z.; Fakurazi, S. Synthesis of (hexaconazole-zinc/aluminum-layered double hydroxide nanocomposite) fungicide nanodelivery system for controlling Ganoderma disease in oil palm. J. Agric. Food Chem. 2018, 66, 806–813. [Google Scholar] [CrossRef] [PubMed]
- Mustafa, I.F.; Hussein, M.Z.; Seman, I.A.; Hilmi, N.H.Z.; Fakurazi, S. Synthesis of dazomet-zinc/aluminum-layered double hydroxide nanocomposite and its phytotoxicity effect on oil palm seed growth. ACS Sustain. Chem. Eng. 2018, 6, 16064–16072. [Google Scholar] [CrossRef]
- Ariffin, D.; Idris, S. Investigation on the control of Ganoderma with dazomet. In Proceedings of the PORIM International Palm Oil Conference. Progress, Prospects Challenges Towards the 21st Century.(Agriculture), Kuala Lumpur, Malaysia, 20–25 September 1993. L-0218. [Google Scholar]
- Maluin, F.N.; Hussein, M.Z.; Yusof, N.A.; Fakurazi, S.; Idris, A.S.; Hilmi, Z.; Hailini, N.; Jeffery Daim, L.D. Preparation of chitosan–hexaconazole nanoparticles as fungicide nanodelivery system for combating Ganoderma disease in oil palm. Molecules 2019, 24, 2498. [Google Scholar] [CrossRef] [PubMed]
- Maluin, F.N.; Hussein, M.Z.; Yusof, N.A.; Fakurazi, S.; Idris, A.S.; Hilmi, N.H.Z.; Jeffery Daim, L.D. A potent antifungal agent for basal stem rot disease treatment in oil palms based on chitosan-dazomet nanoparticles. Int. J. Mol. Sci. 2019, 20, 2247. [Google Scholar] [CrossRef] [PubMed]
- Maluin, F.N.; Hussein, M.Z.; Yusof, N.A.; Fakurazi, S.; Abu Seman, I.; Zainol Hilmi, N.H.; Jeffery Daim, L.D. Enhanced fungicidal efficacy on Ganoderma boninense by simultaneous co-delivery of hexaconazole and dazomet from their chitosan nanoparticles. RSC Adv. 2019, 9, 27083–27095. [Google Scholar] [CrossRef]
- Azhari, M.A.; Putri, I.W.; Pratama, A.I.; Hidayah, R.E.; Ambarsari, L. Development of Trichodermin nanoemulsion based on medium chain triglycerides as antifungal of Ganoderma boninense in vitro. Curr. Biochem. 2019, 4, 2. [Google Scholar]
- Lee, K.W.; bin Omar, D.; bt Abdan, K.; Wong, M.Y. physiochemical characterization of nanoemulsion formulation of phenazine and their antifungal efficacy against Ganoderma boninense PER71 in vitro. Res. J. Pharm. Biol. Chem. Sci. 2016, 7, 3056–3066. [Google Scholar]
| Precursors/Starting Materials | Nanoformulation of Drug/Compound | Emulsion Size and Its Type | Zeta Potential and Stability | Surfactants | Applications | Ref. |
|---|---|---|---|---|---|---|
| Crude palm oil (CPO) | Tocomin®-genistein nanoemulsion | 173–209 nm; O/W | –27 to –30 mV; Stable up to 3 months at 4–8 °C | A mixture of solutol and vitamin E | Pharmaceutical and skin photoprotection | [76] |
| CPO ester | CPO-tocotrienol-xanthan gum | O/W | Stable sedimentation rates at earth gravity of 5.2, 3.0 and 2.6 mm/month | - | Cosmeceuticals | [77] |
| CPO olein ester | CPO-ibuprofen | 21 nm; O/W | - | Carbopol 940 | Anti-inflammatory and analgesic effect | [78] |
| Crude palm kernel oil (CPKO) ester | CPKO-hydrocolloid gum- ibuprofen | 16–20 nm; W/O | –14 to –42 mV | Tween 80 | Anti-inflammatory and analgesic effect | [79] |
| CPO | CPO-hyaluronan-paclitaxel | 263–283 nm; W/O | –23 to –34 mV; stable up to 2 months at 4 °C and 2 weeks at 25 °C. | Tween 80 | Ovarian cancer treatment | [80] |
| CPO olein | CPO-vitamin E | 20–500 nm; O/S/W | Stable up to 1 month at 4, 25 and 40 °C | Brij 30 | Anti-aging agent | [81] |
| CPO ester | CPO-ketoprofen | 230 nm; W/O | - | Tween 80 | Anti-inflammatory and analgesic effect | [82] |
| CPKO ester | CPKO-docetaxel | 90–110 nm; O/W | –30 to –40 mV; stable up to 3 months at 5, 28 and 45 ± 1 °C | Tween 80 | Lung cancer treatment | [83] |
| CPO | CPO-curcumin | 275 nm; O/W | –36 mV; stable up to 1 month at 25 °C | Tween 80 | Anti-inflammatory and anti-cancer | [84] |
| CPO | CPO-lecithin-levodopa | 110 nm; O/O | –31 mV; stable up to 6 months at 4 °C | Cremaphor EL | Parkinson treatment | [85] |
| CPKO ester | CPKO-lecithin-glycerol-aripiprazole | 62 nm; O/O | –32 mV; stable up to 9 months at ± 5 °C | Tween 80 | Schizophrenia treatment | [86] |
| CPO ester | CPO-safflower seed oil-chloramphenicol | 95 nm; O/O | –37 mV | Tween 80 | Meningitis treatment | [87] |
| CPKO ester | CPKO-diclofenac acid | 6 nm; O/W | - | Tween 80 | Anti-inflammatory and analgesic effect | [88] |
| Precursors/Starting Materials | Nanoparticles (NPs) | Applications | Ref. |
|---|---|---|---|
| CPO | Ag NPs | Adsorption and capping agent | [92] |
| Palm oil mill effluent (POME) | Au NPs | Surfactant, template and capping agent | [95] |
| CPO | ZnO nanostructures | Biotemplates | [91] |
| CPO | CuO NPs | Glycerolysis of palm oil | [90] |
| Oil palm frond (OPF) | Ag NPs | Reducing and stabilizing agent | [93,97] |
| CPKO | Au NPs | Reducing and stabilizing agent | [89] |
| OPF | Au NPs | Reducing and stabilizing agent | [94,96] |
| Precursors/Starting Materials | Generation of Nanomaterials | Applications | Ref. |
|---|---|---|---|
| Oil palm empty fruit bunches (OPEFB) | Nanocellulose | - | [101,102,103] |
| Cellulose nanocrystal | - | [104,105,106] | |
| Nano-OPEFB |
| [107,108,109,110] | |
| Carbon nanotubes |
| [111] | |
| Carbon nanodots | - | [112] | |
| Activated carbon |
| [111,113] | |
| Graphene |
| [111,114] | |
| Graphene oxide; reduced graphene oxide | - | [115] | |
| Oil palm kernel shell (OPKS) | Nanocellulose | - | [103] |
| Activated carbon |
| [113,116,117] | |
| Graphene oxide; reduced graphene oxide | - | [115] | |
| Carbon nanotubes | - | [118] | |
| Defatted OPS nanoparticles |
| [119] | |
| Oil palm mesocarp fiber (OPMF) | Nanocellulose | - | [103] |
| Cellulose nanocrystal | - | [120] | |
| Activated carbon |
| [113] | |
| Oil palm frond (OPF) | Nanocellulose |
| [121] |
| Cellulose nanocrystal | - | [122] | |
| Graphene oxide; reduced graphene oxide | - | [115] | |
| Palm oil mill effluent (POME) | Carbon nanotubes |
| [111] |
| Oil palm trunk (OPT) | Nanocellulose | - | [103] |
| Cellulose nanocrystal | - | [123] | |
| Palm oil fuel ash (POFA) | Nano filler |
| [124,125] |
| Nano silica | - | [126] | |
| Nano-POFA |
| [127,128,129] | |
| Waste cooking palm oil | Graphene | - | [130] |
| Carbon nanosphere | [131] |
| Oil palm Waste | Cellulose (%) | Holocellulose (%) | Hemicellulose (%) | Lignin (%) |
|---|---|---|---|---|
| Oil palm empty fruit bunches (OPEFB) | 43–65 | 68–86 | 17–33 | 13–37 |
| Oil palm kernel shell (OPKS) | 27–35 | 40–47 | 15–19 | 48–55 |
| Oil palm mesocarp fiber (OPMF) | 43–44 | 70–71 | 33–35 | 22–24 |
| Oil palm fond (OPF) | 40–50 | 80–83 | 34–38 | 20–21 |
| Oil palm trunk (OPT) | 29–37 | 42–45 | 12–17 | 18–23 |
| Type of Nanosensor | Analyte | Modified Electrode | LOD | Ref. |
|---|---|---|---|---|
| Electrochemical | Secondary metabolite (quinoline) | AUNPs-aMWCNTs/CTSNPs-aMWCNTs/SPCE | 3.75 × 10−9 M | [137] |
| Electrochemical | Secondary metabolites | Bilayer of [AUNPs-aMWCNTs/CTSNPs-aMWCNTs]/SPCE | 0.01657 - 0.007793 μg/mL | [136] |
| Electrochemical | DNA | AuE/PEDOT-PSS/AuNps | 1.59 × 10−17 M | [139] |
| Electrochemical | DNA | PEDOT-PSS/AgNPs/AuE | 5.00 – 6.20 × 10−16 M | [140,141] |
| Optical | DNA | CdSe/ZnS quantum dots | - | [138] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maluin, F.N.; Hussein, M.Z.; Idris, A.S. An Overview of the Oil Palm Industry: Challenges and Some Emerging Opportunities for Nanotechnology Development. Agronomy 2020, 10, 356. https://doi.org/10.3390/agronomy10030356
Maluin FN, Hussein MZ, Idris AS. An Overview of the Oil Palm Industry: Challenges and Some Emerging Opportunities for Nanotechnology Development. Agronomy. 2020; 10(3):356. https://doi.org/10.3390/agronomy10030356
Chicago/Turabian StyleMaluin, Farhatun Najat, Mohd Zobir Hussein, and Abu Seman Idris. 2020. "An Overview of the Oil Palm Industry: Challenges and Some Emerging Opportunities for Nanotechnology Development" Agronomy 10, no. 3: 356. https://doi.org/10.3390/agronomy10030356
APA StyleMaluin, F. N., Hussein, M. Z., & Idris, A. S. (2020). An Overview of the Oil Palm Industry: Challenges and Some Emerging Opportunities for Nanotechnology Development. Agronomy, 10(3), 356. https://doi.org/10.3390/agronomy10030356





