Non-Chemical Weed Management in Vegetables by Using Cover Crops: A Review
Abstract
1. Introduction
2. Yield Losses Caused by Weeds in Vegetable Crops
3. Need for Non-Chemical Weed Control in the Vegetable Crops
4. Types of Cover Crops
5. How Cover Crops Suppress the Weeds?
6. Disadvantages of Using Cover Crops for Weed Control in Vegetable Production Systems
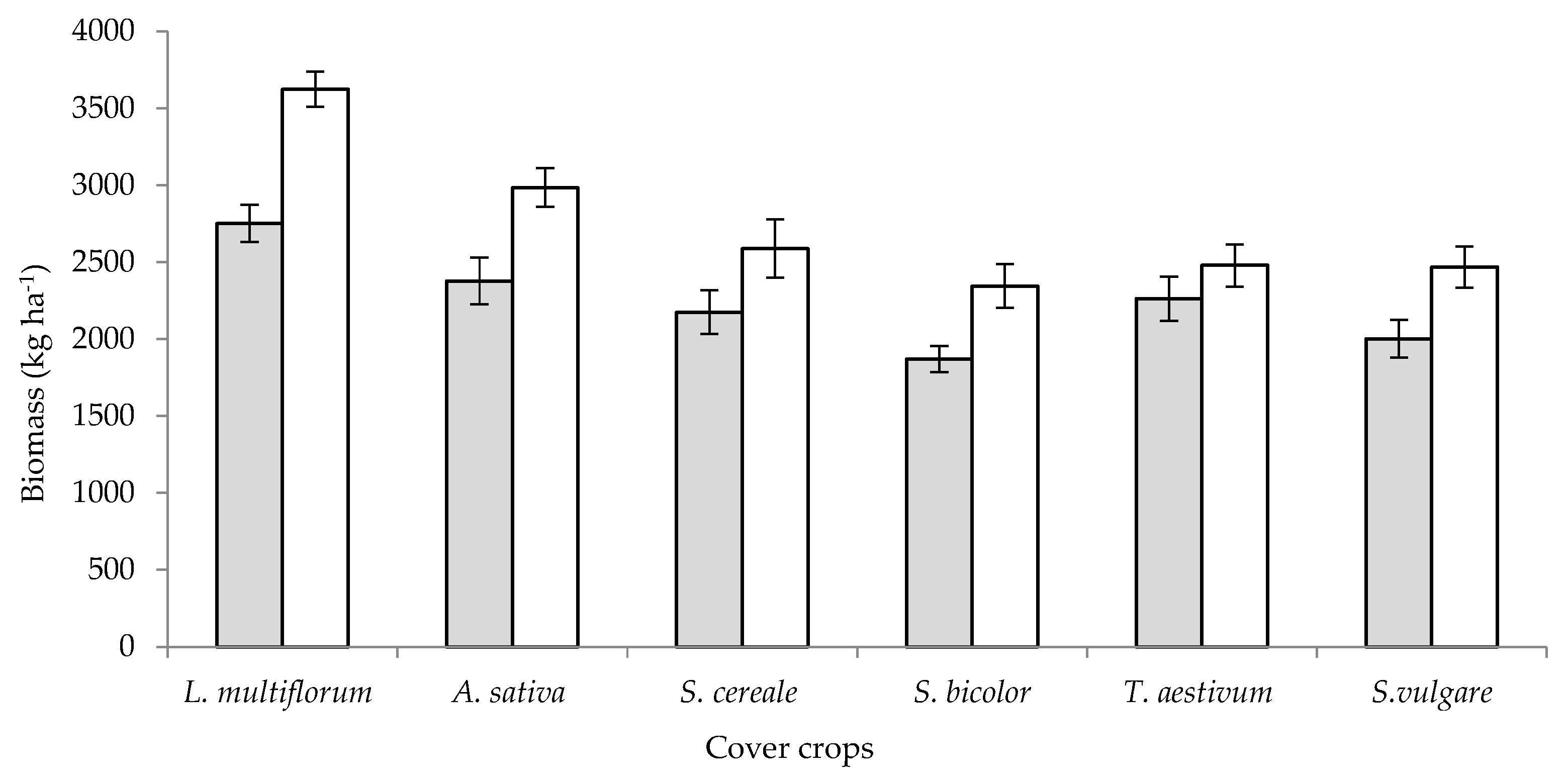
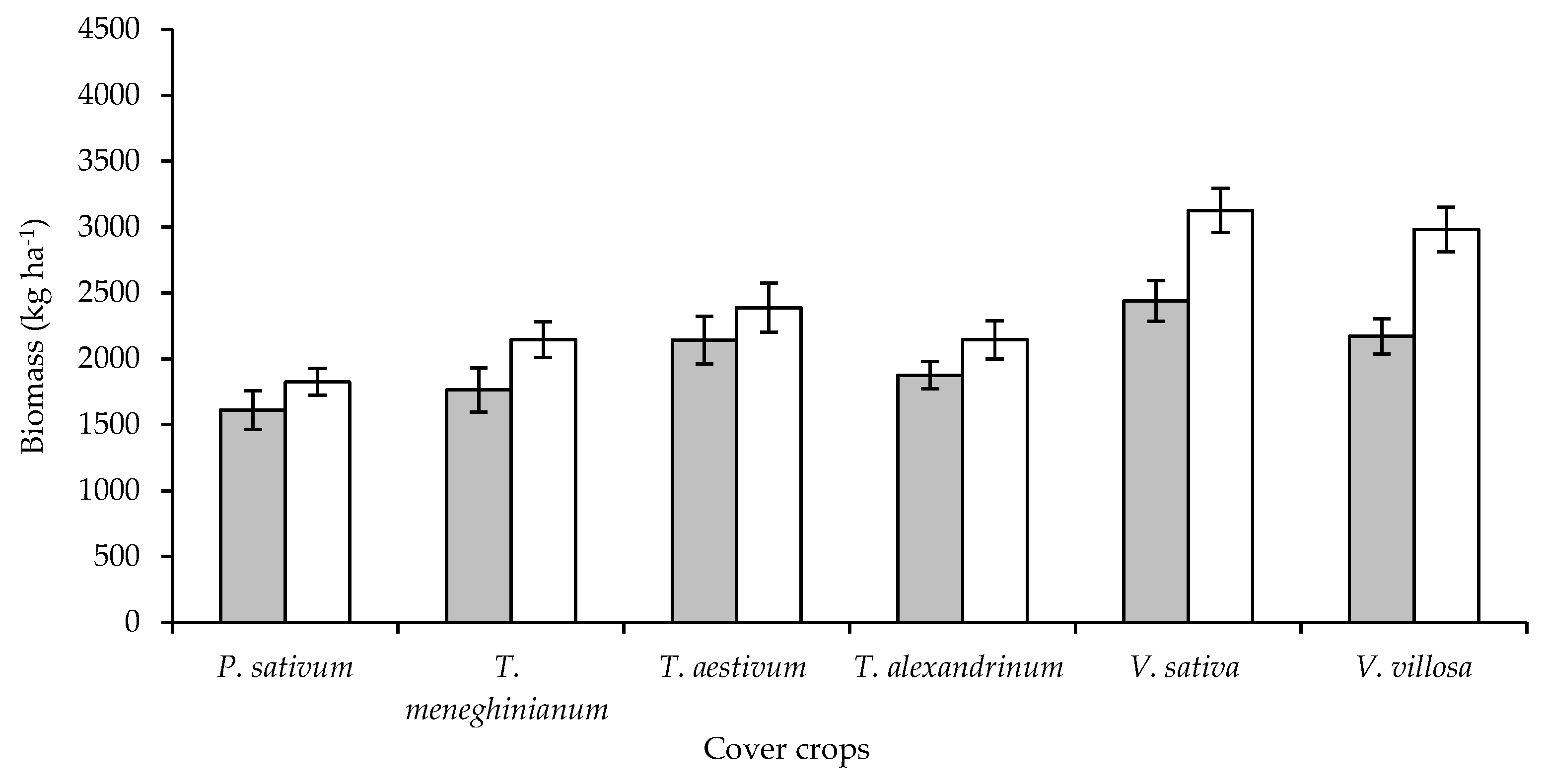
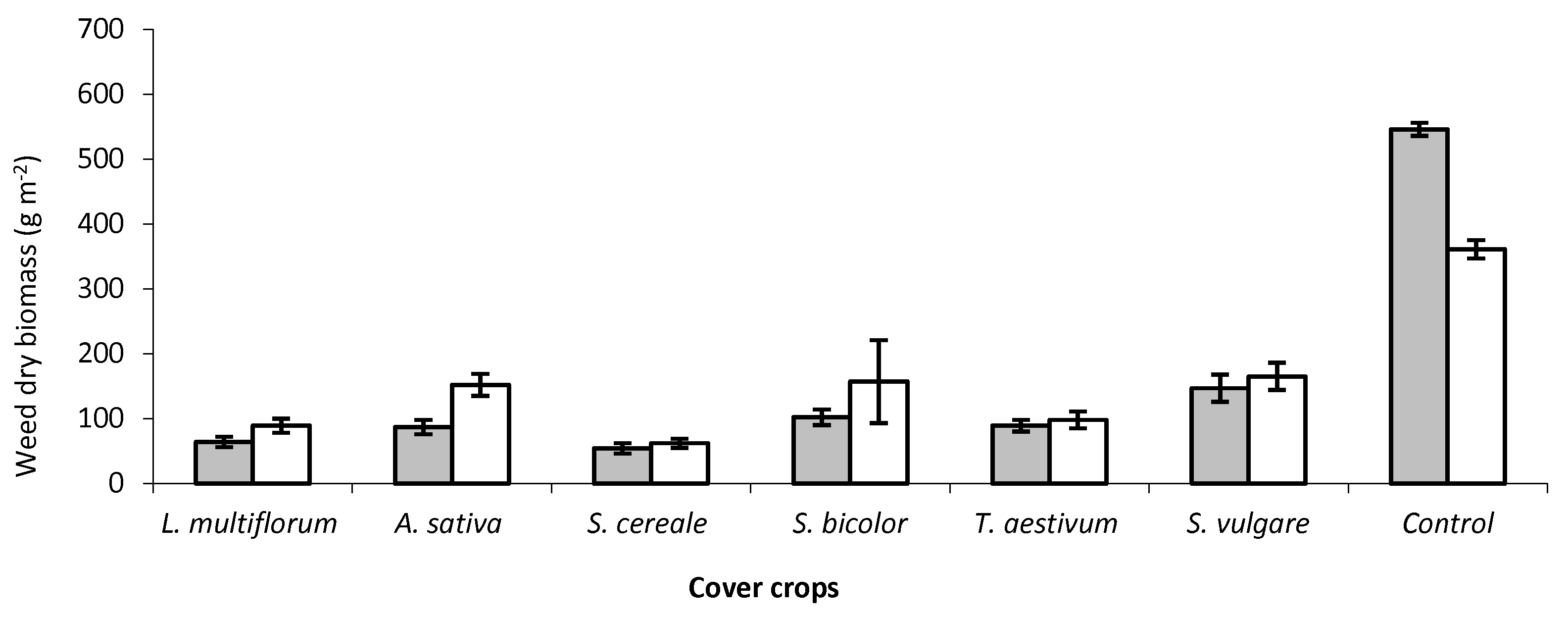
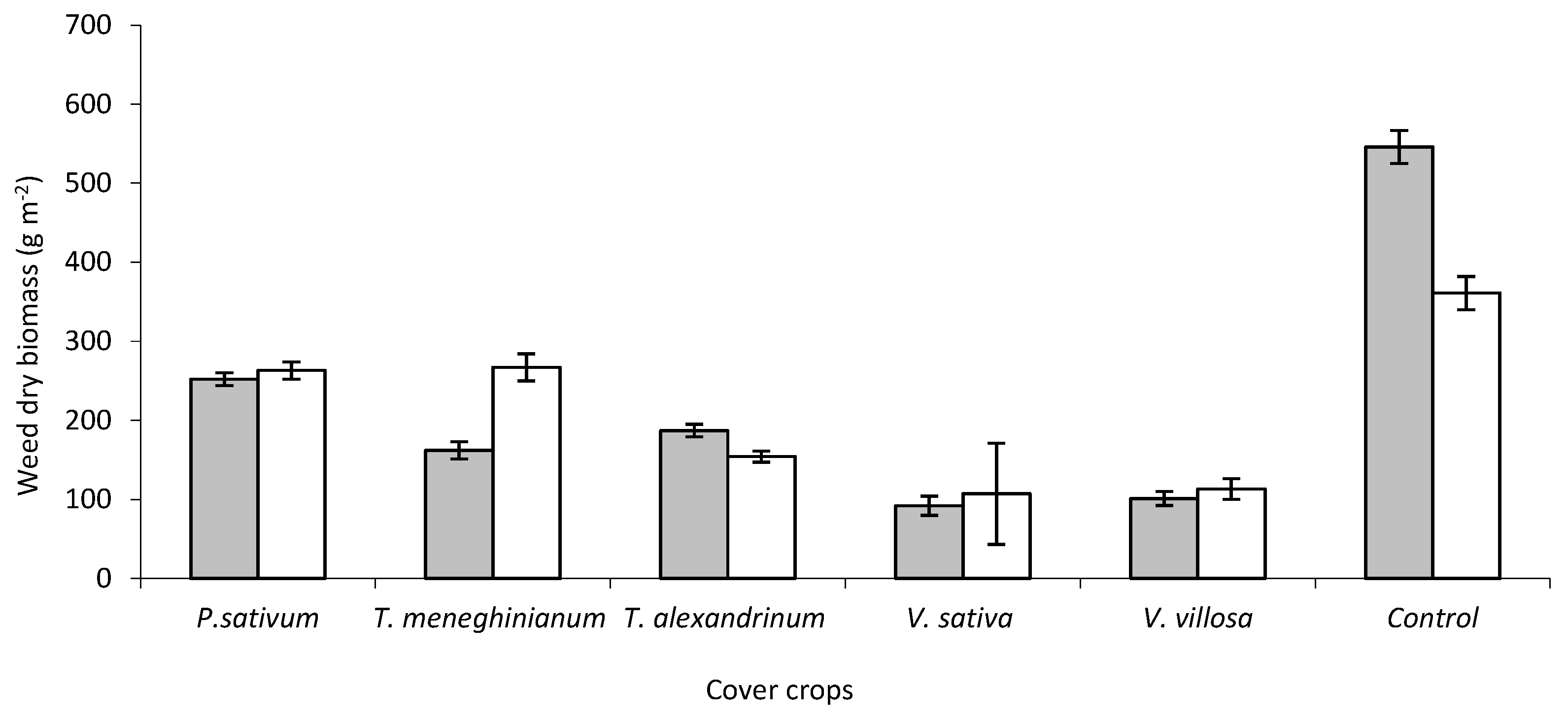
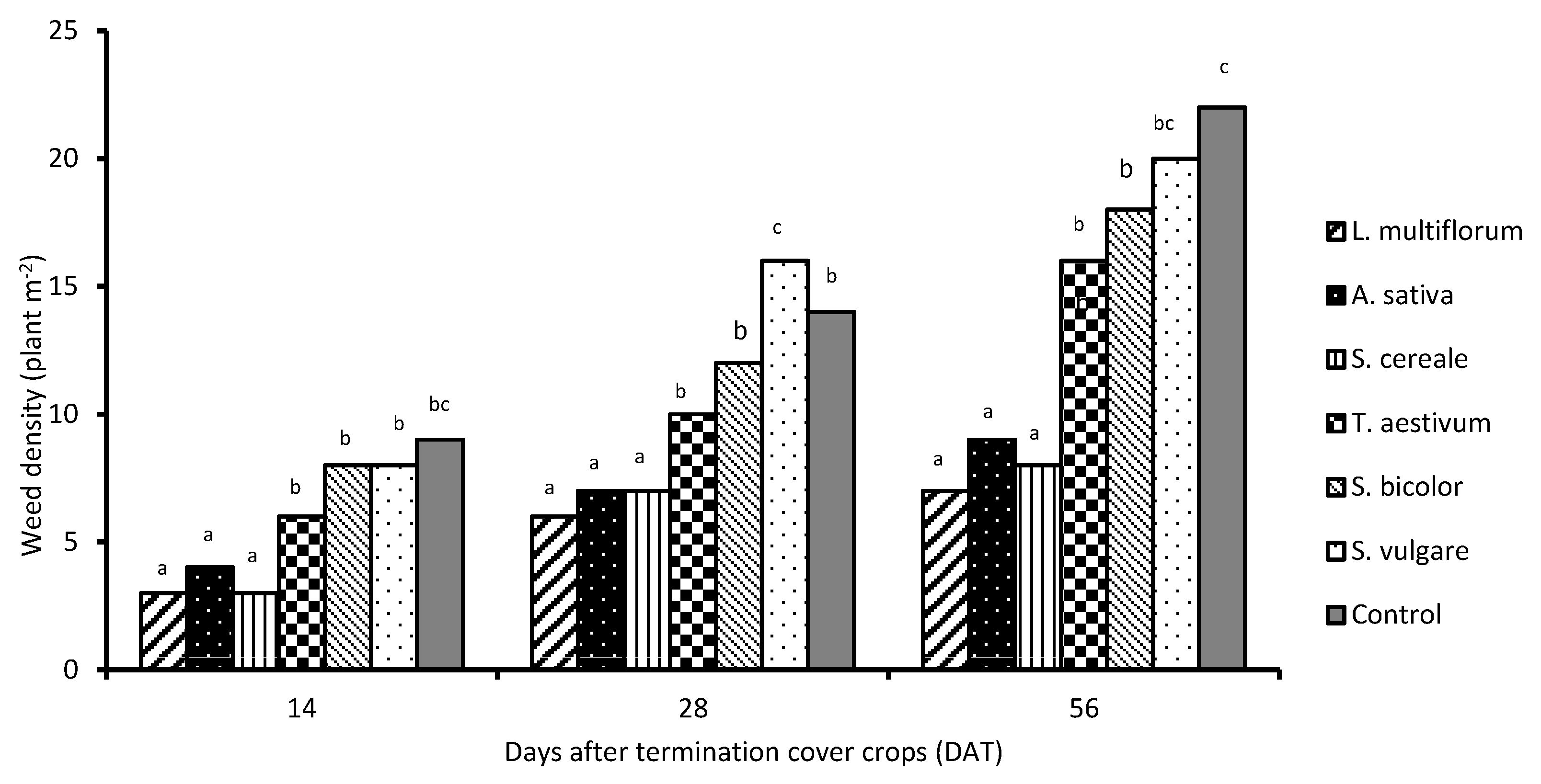
7. Cover Crops for Weed Control in Vegetable Crops
8. Integration of Cover Crops with Vegetable Crops
9. Conclusions
Funding
Conflicts of Interest
References
- Oerke, E.C. Crop losses to pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Ampong-Nyarko, K.; De Data, S.K. A Handbook for Weed Control in Rice; International Rice Research Institute: Manila, Philipines, 1991. [Google Scholar]
- Pimentel, D.; McLaughlin, L.; Zepp, A.; Lakitan, B.; Kraus, T.; Kleinman, P.; Vancini, F.; Roach, W.J.; Graap, E.; Keeton, W.S.; et al. Environmental and economic impacts of reducing U.S. agricultural pesticide use. In Handbook of Pest Management in Agriculture; Pimentel, D., Ed.; CRC Press: Boca Raton, FL, USA, 1991; pp. 679–720. [Google Scholar]
- Brown, B.; Hoshide, A.K.; Gallandt, E.R. An economic comparison of weed management systems used in small-scale organic vegetable production. Org. Agric. 2019, 9, 53–63. [Google Scholar] [CrossRef]
- Schroeder, D.; Müller-Schärer, H.; Stintson, C.S.A. A European weed survey in 10 major crop systems to identify targets for biological control. Weed Res. 1993, 33, 449–458. [Google Scholar] [CrossRef]
- Kropff, M.J.; Walter, H. EWRS and the Challenges for Weed Research at the Start of A New Millennium. Weed Res. 2000, 40, 7–10. [Google Scholar] [CrossRef]
- Jabran, K.; Chauhan, B.S. Overview and significance of non-chemical weed control. In Non-Chemical Weed Control; Elsevier: Cambridge, MA, USA, 2018; pp. 1–8. [Google Scholar]
- Dabney, S.M.; Murphree, C.E.; Triplett, G.B.; Grissinger, E.H.; Meyer, L.D.; Reinschmiedt, L.R.; Rhoton, F.E. Conservation production systems for silty uplands. In Proceedings of the 1993 Southern Conservation Tillage Conference for Sustainable Agriculture, Monroe, LA, USA, 17 June 1993; pp. 43–48. [Google Scholar]
- Sainju, U.M.; Singh, B.P. Nitrogen storage with cover crops and nitrogen fertilization in tilled and non-tilled soils. Agron. J. 2008, 100, 619–627. [Google Scholar] [CrossRef]
- Ngouajio, M.; Foko, J.; Fouejio, D. The critical period of weed control in common bean (Phaseolus vulgaris L.) in Cameroon. Crop Prot. 1997, 16, 127–133. [Google Scholar] [CrossRef]
- Chandler, J.M.; Cooke, F.T. Economics of cotton losses caused by weeds. In Weeds of Cotton; McWhorter, C.G., Abernathy, J.R., Eds.; The Cotton Foundation: Memphis, TN, USA, 1992; pp. 85–116. [Google Scholar]
- Vargas, R.; Fischer, W.B.; Kempen, H.M.; Wright, S.D. Cotton weed management. In Cotton Production; Johnson, M.S., Kerby, T.A., Hake, K.D., Eds.; UC DANR Pub: Oakland, CA, USA, 1996; pp. 187–202. [Google Scholar]
- Teasdale, J.R. Contribution of cover crops to weed management in sustainable agricultural systems. J. Prod. Agric. 1996, 9, 475–479. [Google Scholar] [CrossRef]
- Ngouajio, M.; Mennan, H. Weed populations and pickling cucumber (Cucumis sativus) yield under summer and winter cover crop systems. Crop Prot. 2005, 24, 521–526. [Google Scholar] [CrossRef]
- Mennan, H.; Ngouajio, M.; Isik, D.; Kaya, E. Effect of alternative management systems on weed populations in hazelnut (Corylus avellana L.). Crop Prot. 2006, 25, 835–841. [Google Scholar] [CrossRef]
- Norsworthy, J.K.; Malik, M.S.; Jha, P.; Riley, M.B. Suppression of Digitaria sanguinalis and Amaranthus palmeri using autumn-sown glucosinolate-producing cover crops in organically grown bell pepper. Weed Res. 2007, 47, 425–432. [Google Scholar] [CrossRef]
- Ngouajio, M.; McGiffen, M.E., Jr.; Hutchinson, C.M. Effect of cover crop and management system on weed populations in lettuce. Crop Prot. 2003, 22, 57–64. [Google Scholar] [CrossRef]
- Samarajeewa, K.B.D.P.; Horiuchi, T.; Oba, S. Finger millet (Eleucine corocana L. Gaertn.) as a cover crop on weed control, growth and yield of soybean under different tillage systems. Soil Till. Res. 2006, 90, 93–99. [Google Scholar] [CrossRef]
- Teasdale, J.R.; Brandsaeter, L.O.; Calegari, A.; Neto, F.S.; Upadhyaya, M.K.; Blackshaw, R.E. Cover crops and weed management. In Non-chemical Weed Management: Principles, Concepts and Technology; Upadhyaya, M.K., Blackshaw, R.E., Eds.; CAB International: Cambridge, MA, USA, 2007; pp. 49–64. [Google Scholar]
- Fisk, J.W.; Hesterman, O.B.; Shrestha, A.; Kells, J.J.; Harwood, R.R.; Squire, J.M.; Sheaffer, C.C. Weed suppression by annual legume cover crops in no-tillage corn. Agron. J. 2001, 93, 319–325. [Google Scholar] [CrossRef]
- Reddy, K.N. Impact of rye cover crop and herbicides on weeds, yield, and net return in narrow-row transgenic and conventional soybean (Glycine max). Weed Technol. 2003, 17, 28–35. [Google Scholar] [CrossRef]
- Teasdale, J.R.; Daughtry, C.S. Weed suppression by live and desiccated hairy vetch (Vicia villosa). Weed Sci. 1993, 41, 207–212. [Google Scholar] [CrossRef]
- Altieri, M.A.; Lana, M.A.; Bittencourt, H.V.; Kieling, A.S.; Comin, J.J.; Lovato, P.E. Enhancing crop productivity via weed suppression in organic no-till cropping systems in Santa Catarina, Brazil. J. Sustain. Agric. 2011, 35, 855–869. [Google Scholar] [CrossRef]
- Hoffman, M.L.; Weston, L.A.; Snyder, J.C.; Regnier, E.E. Allelopathic influence of germinating seeds and seedlings of cover crops on weed species. Weed Sci. 1996, 44, 579–584. [Google Scholar] [CrossRef]
- Weiner, J. A new neighborhood model for annual-plant interference. Ecology 1982, 63, 1237–1241. [Google Scholar] [CrossRef]
- Pike, D.R.; Stoller, E.W.; Wax, L.M. Modelling soybean growth and canopy apportionment in weed-soybean (Glycine max) competition. Weed Sci. 1990, 38, 522–527. [Google Scholar] [CrossRef]
- Obopile, M.; Munthali, D.C.; Matilo, B. Farmers’ knowledge, perceptions and management of vegetable pests and diseases in Botswana. Crop Prot. 2008, 27, 1220–1224. [Google Scholar] [CrossRef]
- Akemo, M.C.; Regnier, E.E.; Bennett, M.A. Weed suppression in spring-sown rye (Secale cereale)–pea (Pisum sativum) cover crop mixes. Weed Technol. 2000, 14, 545–549. [Google Scholar] [CrossRef]
- Roberts, H.A.; Hewson, R.T.; Ricketts, M.A. Weed competition in drilled summer lettuce. Hortic. Res. 1997, 17, 39–45. [Google Scholar]
- Lanini, W.T.; Le Strange, M. Low-input management of weeds in vegetable fields. Calif. Agric. 1991, 45, 11–13. [Google Scholar]
- Dusky, J.A.; Stall, W.M. Weed management practices for lettuce production using imazethapyr. In Proceedings of the 108th Florida State Horticultural Society, Lake Alfred, FL, USA, 22–24 October 1995; pp. 204–207. [Google Scholar]
- Shrefler, J.W.; Dusky, J.A.; Shilling, D.G.; Brecke B, J.; Sanchez, C.A. Effects of phosphorus fertility on competition between lettuce (Lactuca sativa L.) and spiny amaranth (Amaranthus spinosus L.). Weed Sci. 1995, 42, 556–560. [Google Scholar] [CrossRef]
- Green-Tracewicz, E.; Page, E.R.; Swanton, C.J. Light quality and the critical period for weed control in soybean. Weed Sci. 2012, 60, 86–91. [Google Scholar] [CrossRef]
- Morales-Payan, J.P.; Santos, B.M.; Stall, W.M.; Bewick, T.A. Effects of purple nutsedge (Cyperus rotundus) on tomato (Lycopersicon esculentum) and bell pepper (Capsicum annuum) vegetative growth and fruit yield. Weed Technol. 1997, 11, 672–676. [Google Scholar] [CrossRef]
- Amador-Ramirez, M.D. Critical period of weed control in transplanted chilli pepper. Weed Res. 2002, 42, 203–209. [Google Scholar] [CrossRef]
- Monaco, T.J.; Grayson, A.S.; Sanders, D.C. Influence of four weed species on the growth and quality of direct-seeded tomatoes (Lycopersicon esculentum). Weed Sci. 1981, 29, 394–397. [Google Scholar] [CrossRef]
- Price, A.J.; Norsworthy, J.K. Cover crops for weed management in southern reduced-tillage vegetable cropping systems. Weed Technol. 2013, 27, 212–217. [Google Scholar] [CrossRef]
- Dettmann, R.L.; Dimitri, C. Who’s buying organic vegetables? Demographic characteristics of US consumers. J. Food Prod. Mark. 2009, 16, 79–91. [Google Scholar] [CrossRef]
- Heap, I. Herbicide resistant weeds. In Integrated Pest Management; Springer: Dordrecht, Switzerland, 2014; pp. 281–301. [Google Scholar]
- Pala, F.; Mennan, H. Socio-economic and environmental effects of glyphosate consumption. In Proceedings of the 3rd Anatolian International Congress of Applied Sciences, Diyarbakir, Turkey, 28–29 December 2019. [Google Scholar]
- Coombs, C.; Lauzon, J.D.; Deen, B.; Van Eerd, L.L. Legume cover crop management on nitrogen dynamics and yield in grain corn systems. Field Crops Res. 2017, 201, 75–85. [Google Scholar] [CrossRef]
- Sturm, D.J.; Peteinatos, G.; Gerhards, R. Contribution of allelopathic effects to the overall weed suppression by different cover crops. Weed Res. 2018, 58, 331–337. [Google Scholar] [CrossRef]
- Akbari, P.; Herbert, S.J.; Hashemi, M.; Barker, A.V.; Zandvakili, O.R. Role of Cover crops and planting dates for improved weed suppression and nitrogen recovery in no-till systems. Commun. Soil Sci. Plant Anal. 2019, 50, 1722–1731. [Google Scholar] [CrossRef]
- Snapp, S.S.; Swinton, S.M.; Labarta, R.; Mutch, D.; Black, J.R.; Leep, R.; Nyiraneza, J.; O’Neil, K. Review and Interpretation: Evaluating Cover Crops for Benefits, Costs, and Performance within Cropping System Niches. Agron. J. 2005, 97, 322–332. [Google Scholar]
- Couëdel, A.; Kirkegaard, J.; Alletto, L.; Justes, E. Crucifer-legume cover crop mixtures for biocontrol: Toward a new multi-service paradigm. Adv. Agron. 2019, 157, 55–139. [Google Scholar]
- Creamer, N.G.; Bennett, M.A.; Stinner, B.R.; Cardina, J.; Regnier, E.E. Mechanisms of weed suppression in cover crop-based production systems. Am. Soc. Hortic. Sci. 1996, 31, 410–413. [Google Scholar] [CrossRef]
- Reberg-Horton, S.C.; Grossman, J.M.; Kornecki, T.S.; Meijer, A.D.; Price, A.J.; Place, G.T.; Webster, T.M. Utilizing cover crop mulches to reduce tillage in organic systems in the southeastern USA. Renew. Agric. Food Syst. 2012, 27, 41–48. [Google Scholar] [CrossRef]
- Mirsky, S.B.; Ryan, M.R.; Teasdale, J.R.; Curran, W.S.; Reberg-Horton, C.S.; Spargo, J.T.; Wells, M.S.; Keene, C.L.; Moyer, J.W. Overcoming weed management challenges in cover crop–based organic rotational no-till soybean production in the eastern United States. Weed Technol. 2013, 27, 193–203. [Google Scholar] [CrossRef]
- Mennan, H.; Ngouajio, M.; Isık, D.; Kaya, E. Effects of alternative winter cover cropping systems on weed suppression in organically grown tomato (Solanum lycopersicum). Phytoparasitica 2009, 37, 385–396. [Google Scholar] [CrossRef]
- Mennan, H.; Ngouajio, M.; Kaya, E.; Isık, D. Weed management in organically grown kale using alternative cover cropping systems. Weed Technol. 2009, 23, 81–88. [Google Scholar] [CrossRef]
- Hayden, Z.D.; Brainard, D.C.; Henshaw, B.; Ngouajio, M. Winter annual weed suppression in rye–vetch cover crop mixtures. Weed Technol. 2012, 26, 818–825. [Google Scholar] [CrossRef]
- Lawley, Y.E.; Teasdale, J.R.; Weil, R.R. The mechanism for weed suppression by a forage radish cover crop. Agron. J. 2012, 104, 205–214. [Google Scholar] [CrossRef]
- Brennan, E.B.; Smith, R.F. Winter cover crop growth and weed suppression on the central coast of California. Weed Technol. 2005, 19, 1017–1024. [Google Scholar] [CrossRef]
- Boyd, N.S.; Brennan, E.B.; Smith, R.F.; Yokota, R. Effect of seeding rate and planting arrangement on rye cover crop and weed growth. Agron. J. 2009, 101, 47–51. [Google Scholar] [CrossRef]
- Cressman, S.T.; Page, E.R.; Swanton, C.J. Weeds and the red to far-red ratio of reflected light: Characterizing the influence of herbicide selection, dose, and weed species. Weed Sci. 2011, 59, 424–430. [Google Scholar] [CrossRef]
- Rajcan, I.; Chandler, K.J.; Swanton, C.J. Red–far-red ratio of reflected light: A hypothesis of why early-season weed control is important in corn. Weed Sci. 2004, 52, 774–778. [Google Scholar] [CrossRef]
- Mirsky, S.B.; Gallandt, E.R.; Mortensen, D.A.; Curran, W.S.; Shumway, D.L. Reducing the germinable weed seedbank with soil disturbance and cover crops. Weed Res. 2010, 50, 341–352. [Google Scholar] [CrossRef]
- Moonen, A.C.; Barberi, P. Size and composition of the weed seedbank after 7 years of different cover-crop-maize management systems. Weed Res. 2004, 44, 163–177. [Google Scholar] [CrossRef]
- Mirsky, S.B.; Curran, W.S.; Mortenseny, D.M.; Ryany, M.R.; Shumway, D.L. Timing of cover-crop management effects on weed suppression in no-till planted soybean using a roller-crimper. Weed Sci. 2011, 59, 380–389. [Google Scholar] [CrossRef]
- Brennan, E.B.; Boyd, N.S.; Smith, R.F.; Foster, P. Seeding rate and planting arrangement effects on growth and weed suppression of a legume-oat cover crop for organic vegetable systems. Agron. J. 2009, 101, 979–988. [Google Scholar] [CrossRef]
- Jabran, K.; Mahajan, G.; Sardana, V.; Chauhan, B.S. Allelopathy for weed control in agricultural systems. Crop Prot. 2015, 72, 57–65. [Google Scholar] [CrossRef]
- Jabran, K. Manipulation of Allelopathic Crops for Weed Control, 1st ed.; Springer Nature International Publishing: Cham, Switzerland, 2017. [Google Scholar]
- Jabran, K. Rye allelopathy for weed control. In Manipulation of Allelopathic Crops for Weed Control; Springer: Cham, Switzerland, 2017; pp. 49–56. [Google Scholar]
- Tabaglio, V.; Marocco, A.; Schulz, M. Allelopathic cover crop of rye for integrated weed control in sustainable agroecosystems. Ital. J. Agron. 2013, 8, e5. [Google Scholar] [CrossRef]
- Chase, W.R.; Nair, M.G.; Putnam, A.R. 2, 2′-oxo-1, 1′-azobenzene: Selective toxicity of rye (Secale cereale L.) allelochemicals to weed and crop species: II. J. Chem. Ecol. 1991, 17, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Yenish, J.P.; Worsham, A.D.; Chilton, W.S. Disappearance of DIBOA-glucoside, DIBOA, and BOA from rye (Secale cereale L.) cover crop residue. Weed Sci. 1995, 43, 18–20. [Google Scholar] [CrossRef]
- Haramoto, E.R.; Gallandt, E.R. Brassica cover cropping: I. Effects on weed and crop establishment. Weed Sci. 2005, 53, 695–701. [Google Scholar] [CrossRef]
- Petersen, J.; Belz, R.; Walker, F.; Hurle, K. Weed suppression by release of isothiocyanates from turnip-rape mulch. Agron. J. 2001, 93, 37–43. [Google Scholar] [CrossRef]
- Kunz, C.; Sturm, D.J.; Varnholt, D.; Walker, F.; Gerhards, R. Allelopathic effects and weed suppressive ability of cover crops. Plant Soil Environ. 2016, 62, 60–66. [Google Scholar]
- Williams, M.M.; Mortensen, D.A.; Doran, J.W. Assessment of weed and crop fitness in cover crop residues for integrated weed management. Weed Sci. 1998, 46, 595–603. [Google Scholar] [CrossRef]
- Krogh, S.S.; Mensz, S.J.; Nielsen, S.T.; Mortensen, A.G.; Christophersen, C.; Fomsgaard, I.S. Fate of benzoxazinone allelochemicals in soil after incorporation of wheat and rye sprouts. J. Agric. Food Chem. 2006, 54, 1064–1074. [Google Scholar] [CrossRef]
- Lovett, J.; Hoult, A. Allelopathy and Self-Defense in Barley. ACS Publ. 1995, 170–183. [Google Scholar] [CrossRef]
- Oueslati, O.; Ben-Hammoudam, H.; Ghorbel, M.; El Gazzeh, M.; Kremer, R. Role of phenolic acids in expression of barley (Hordeum vulgare) autotoxicity. Allelopath. J. 2009, 23, 157–166. [Google Scholar]
- Uddin, M.R.; Park, S.U.; Dayan, F.E.; Pyon, J.Y. Herbicidal activity of formulated sorgoleone, a natural product of sorghum root exudate. Pest Manag. Sci. 2014, 70, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Belz, R.G.; Hurle, K. Differential exudation of two benzoxazinoids one of the determining factors for seedling allelopathy of Triticeae species. J. Agric. Food Chem. 2005, 53, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.; Pratley, J.; Lemerle, D.; Haig, T. Evaluation of seedling allelopathy in 453 wheat (Triticum aestivum) accessions against annual ryegrass (Lolium rigidum) by the equal compartment-agar method. Crop Pasture Sci. 2000, 51, 937–944. [Google Scholar] [CrossRef]
- Brown, P.; Morra, M. Hydrolysis products of glucosinolates in Brassica napus tissues as inhibitors of seed germination. Plant Soil 1996, 181, 307–316. [Google Scholar] [CrossRef]
- Vaughn, S.F.; Boydston, R.A. Volatile allelochemicals released by crucifer green manures. J. Chem. Ecol. 1997, 23, 2107–2116. [Google Scholar] [CrossRef]
- Inderjit. Plant phenolics in allelopathy. Bot. Rev. 1996, 62, 186–202. [Google Scholar] [CrossRef]
- Xuan, T.D.; Hong, N.H.; Khanh, T.D.; Eiji, T.; Tawata, S.; Fukuta, M. Utilization of plant allelopathy for biological control of weeds and plant pathogens in rice. In Proceedings of the Fourth World Congress on Allelopathy, Wagga Wagga, Australia, 21–26 August 2005. [Google Scholar]
- Xuan, T.D.; Chikara, J.; Ogushi, Y.; Tsuzuki, E.; Terao, H.; Khanh, T.D.; Matsuo, M. Application of kava (Piper methysticum L.) root as potential herbicide and fungicide. Crop Prot. 2003, 22, 873–881. [Google Scholar] [CrossRef]
- Norsworthy, J.K.; Riar, D.; Jha, P.; Scott, R.C. Confirmation, control, and physiology of glyphosate-resistant giant ragweed (Ambrosia trifida) in Arkansas. Weed Technol. 2011, 25, 430–435. [Google Scholar] [CrossRef]
- Bernstein, E.R.; Stoltenberg, D.E.; Posner, J.L.; Hedtcke, J.L. Weed community dynamics and suppression in tilled and no-tillage transitional organic winter rye-soybean systems. Weed Sci. 2014, 62, 125–137. [Google Scholar] [CrossRef]
- Dube, E.; Chiduza, C.; Muchaonyerwa, P. Conservation agriculture effects on soil organic matter on a Haplic Cambisol after four years of maize–oat and maize–grazing vetch rotations in South Africa. Soil Till. Res. 2012, 123, 21–28. [Google Scholar] [CrossRef]
- Finney, D.M.; Creamer, N.G.; Schultheis, J.R.; Wagger, M.G.; Brownie, C. Sorghum sudangrass as a summer cover and hay crop for organic fall cabbage production. Renew. Agric. Food Syst. 2009, 24, 225–233. [Google Scholar] [CrossRef]
- Moran, P.; Greenberg, S. Winter cover crops and vinegar for early-season weed control in sustainable cotton. J. Sustain. Agric. 2008, 32, 483–506. [Google Scholar] [CrossRef][Green Version]
- Silva, E.M. Screening five fall-sown cover crops for use in organic no-till crop production in the upper midwest. Agroecol. Sustain. Food Syst. 2014, 38, 748–763. [Google Scholar] [CrossRef]
- Alcántara, C.; Pujadas, A.; Saavedra, M. Management of Sinapis alba subsp. mairei winter cover crop residues for summer weed control in southern Spain. Crop Prot. 2011, 30, 1239–1244. [Google Scholar]
- Campiglia, E.; Caporali, F.; Radicetti, E.; Mancinelli, R. Hairy vetch (Vicia villosa Roth.) cover crop residue management for improving weed control and yield in no-tillage tomato (Lycopersicon esculentum Mill.) production. Eur. J. Agron. 2010, 33, 94–102. [Google Scholar] [CrossRef]
- Campiglia, E.; Mancinelli, R.; Radicetti, E.; Caporali, F. Effect of cover crops and mulches on weed control and nitrogen fertilization in tomato (Lycopersicon esculentum Mill.). Crop Prot. 2010, 29, 354–363. [Google Scholar] [CrossRef]
- Grünwald, N.; Hu, S.; Van Bruggen, A. Short-term cover crop decomposition in organic and conventional soils: Characterization of soil C, N, microbial and plant pathogen dynamics. Eur. J. Plant Pathol. 2000, 106, 37–50. [Google Scholar] [CrossRef]
- Masiunas, J.B. Production of vegetables using cover crop and living mulches—A review. J. Veg. Crop Prod. 1998, 4, 11–31. [Google Scholar]
- Mulvaney, M.J.; Price, A.J.; Wood, C.W. Cover crop residue and organic mulches provide weed control during limited-input no-till collard production. J. Sustain. Agric. 2011, 35, 312–328. [Google Scholar] [CrossRef]
- Kruidhof, H.M.; Bastiaans, L.; Kropff, M.J. Cover crop residue management for optimizing weed control. Plant Soil 2009, 318, 169–184. [Google Scholar] [CrossRef]
- Baumgartner, K.; Steenwerth, K.L.; Veilleux, L. Cover-crop systems affect weed communities in a California vineyard. Weed Sci. 2008, 56, 596–605. [Google Scholar] [CrossRef]
- Steinmaus, S.; Elmore, C.L.; Smith, R.J.; Donaldson, D.; Weber, E.A.; Roncoroni, J.A.; Miller, P.R.M. Mulched cover crops as an alternative to conventional weed management systems in vineyards. Weed Res. 2008, 48, 273–281. [Google Scholar] [CrossRef]
- Brainard, D.C.; Bellinder, R.R.; Kumar, V. Grass–legume mixtures and soil fertility affect cover crop performance and weed seed production. Weed Technol. 2011, 25, 473–479. [Google Scholar] [CrossRef]
- Mirsky, S.B.; Ryan, M.R.; Curran, W.S.; Teasdale, J.R.; Maul, J.; Spargo, J.T.; Moyer, J.; Grantham, A.M.; Weber, D.; Way, T.R.; et al. Conservation tillage issues: Cover crop-based organic rotational no-till grain production in the mid-Atlantic region, USA. Renew. Agric. Food Syst. 2012, 27, 31–40. [Google Scholar] [CrossRef]
- Stivers-Young, L. Growth, nitrogen accumulation, and weed suppression by fall cover crops following early harvest of vegetables. HortScience 1998, 33, 60–63. [Google Scholar]
- Masiunas, J.B.; Weston, L.A.; Weller, S.C. The impact of rye cover crops on weed populations in a tomato cropping system. Weed Sci. 1995, 43, 318–323. [Google Scholar] [CrossRef]
- Caamal-Maldonado, J.A.; Jiménez-Osornio, J.J.; Torres-Barragán, A.; Anaya, A.L. The use of allelopathic legume cover and mulch species for weed control in cropping systems. Agron. J. 2001, 93, 27–36. [Google Scholar] [CrossRef]
- Teasdale, J.R.; Abdul-Baki, A.A. Comparison of mixtures vs. monocultures of cover crops for fresh-market tomato production with and without herbicide. HortScience 1998, 33, 1163–1166. [Google Scholar] [CrossRef]
- Isik, D.; Kaya, E.; Ngouajio, M.; Mennan, H. Weed suppression in organic pepper (Capsicum annuum L.) with winter cover crops. Crop Prot. 2009, 28, 356–363. [Google Scholar] [CrossRef]
- Hutchinson, C.M.; McGiffen, M.E. Cowpea cover crop mulch for weed control in desert pepper production. HortScience 2000, 35, 196–198. [Google Scholar] [CrossRef]
- Galloway, B.A.; Weston, L.A. Influence of cover crop and herbicide treatment on weed control and yield in no-till sweet corn (Zea mays L.) and pumpkin (Cucurbita maxima Duch.). Weed Technol. 1996, 10, 341–346. [Google Scholar] [CrossRef]
- Dhima, K.V.; Vasilakoglou, I.B.; Eleftherohorinos, I.G.; Lithourgidis, A.S. Allelopathic potential of winter cereal cover crop mulches on grass weed suppression and sugarbeet development. Crop Sci. 2006, 46, 1682–1691. [Google Scholar] [CrossRef]
- Carrera, L.M.; Abdul-Baki, A.A.; Teasdale, J.R. Cover crop management and weed suppression in no-tillage sweet corn production. HortScience 2004, 39, 1262–1266. [Google Scholar] [CrossRef]
- Burgos, N.R.; Talbert, R.E. Weed control and sweet corn (Zea mays var. rugosa) response in a no-till system with cover crops. Weed Sci. 1996, 44, 355–361. [Google Scholar] [CrossRef]
- Ranaldo, M.; Carlesi, S.; Costanzo, A.; Bàrberi, P. Can weed management in vegetable systems be improved by cover crop species mixtures? Step 2: Field implementation. In Proceedings of the 7th International Weed Science Congress, Prague (CZ), Czech, 19–25 June 2016. [Google Scholar]
- Creamer, N.G.; Bennett, M.A.; Stinner, B.R. Evaluation of cover crop mixtures for use in vegetable production systems. HortScience 1997, 32, 866–870. [Google Scholar] [CrossRef]
- Peachey, R.E.; William, R.D.; Mallory-Smith, C. Effect of no-till or conventional planting and cover crops residues on weed emergence in vegetable row crop. Weed Technol. 2004, 18, 1023–1030. [Google Scholar] [CrossRef]
- Héraux, F.M.; Hallett, S.G.; Weller, S.C. Combining Trichoderma virens-inoculated compost and a rye cover crop for weed control in transplanted vegetables. Biol. Control 2005, 34, 21–26. [Google Scholar] [CrossRef]
- Kruidhof, H.M.; Bastiaans, L.; Kropff, M.J. Ecological weed management by cover cropping: Effects on weed growth in autumn and weed establishment in spring. Weed Res. 2008, 48, 492–502. [Google Scholar] [CrossRef]
- Creamer, N.G.; Plassman, B.; Bennett, M.A.; Wood, R.K.; Stinner, B.R.; Cardina, J. A method for mechanically killing cover crops to optimize weed suppression. Am. J. Altern. Agric. 1995, 10, 157–162. [Google Scholar] [CrossRef]
- Wortman, S.E.; Francis, C.A.; Bernards, M.A.; Blankenship, E.E.; Lindquist, J.L. Mechanical termination of diverse cover crop mixtures for improved weed suppression in organic cropping systems. Weed Sci. 2013, 61, 162–170. [Google Scholar] [CrossRef]
- Tursun, N.; Işık, D.; Demir, Z.; Jabran, K. Use of living, mowed, and soil-incorporated cover crops for weed control in apricot orchards. Agronomy 2018, 8, 150. [Google Scholar] [CrossRef]
| Cover Crop Type | Name of Cover Crop | Reference |
|---|---|---|
| Cereals | Bristle oat (Avena strigosa) | [42] |
| Winter rye (Secale cereale) | [43] | |
| Oat (Avena sativa) | [43] | |
| Sudangrass (Sorghum × sudanense) | [44] | |
| Wheat (Triticum aestivum) | [44] | |
| Legumes | Pea (Pisum sativum) | [45] |
| Cowpea (Vigna unguiculata) | [44] | |
| Subterranean clover (Trifolium subterraneum) | [45] | |
| Crimson clover (Trifolium incarnatum) | [45] | |
| Egyptian clover (Trifolium alexandrinum ) | [45] | |
| Red clover (Trifolium pratense) | [45] | |
| Sunn hemp (Crotalaria juncea) | [45] | |
| Velvet bean (Mucuna pruriens) | [45] | |
| Soybean (Glycine max) | [45] | |
| Faba bean (Vicia faba) | [45] | |
| Hairy vetch (Vicia villosa) | [43] | |
| Common vetch (Vicia sativa) | [46] | |
| Brassicaceae plant | Forage radish (Raphanus sativus) | [42] |
| Rapeseed, canola (Brassica napus) | [45] | |
| White mustard (Sinapis alba) | [45] | |
| Non-legumes | Buckwheat (Fagopyrum esculentum) | [42] |
| Flax (Linum usitatissimum) | [42] | |
| Niger (Guizotia abyssinica) | [42] |
| Cover Crop | Allelochemicals | Reference |
|---|---|---|
| Rye (Secale cereale) | MBOA, BOA, HMBOA, DIBOA | [71] |
| Barley (Hordeum vulgare) | Gramine, hordenine, p-hydroxybenzoic acid, vanillic acid, p-coumaric acid, syringic acid, ferulic acid | [72,73] |
| Sorghum (Sorghum bicolor) | Sorgoleone, m-coumaric acid, caffeic acid, chlorogenic acid | [74] |
| Wheat (Triticum aestivum) | DIMBOA, 2,4-Dihydroxy-1,4-benzoxazine-3-one (DIBOA) | [75] |
| DIMBOA, syringic acid, vanillic acid, p-hydroxybenzoic acid, cis-ferulic acid, trans-ferulic acid, trans-p-coumaric acid, cis-p-coumaric acid | [76] | |
| Field mustard (Brassica rapa) | 2-Phenylethyl-isothiocyanate | [67] |
| Rapeseed (Brassica napus) | Glucosinolates, 2-Phenylethyl-isothiocyanate, benzyl isothiocyanate, allyl isothiocyanate, 3-butenyl isothiocyanate, | [77,78] |
| Field mustard (Brassica campestris) | Benzyl isothiocyanate, allyl isothiocyanate, 3-butenyl isothiocyanate | [78] |
| Cover Crop | Weeds Suppressed | References |
|---|---|---|
| Wheat (Triticum aestivum) | Ipomoea lacunose, Eleusine indica, Amaranthus palmeri | [82] |
| Rye (Secale cereale) | Eleusine indica, Amaranthus palmeri, Ipomoea lacunosa | [82] |
| Rye (Secale cereale) | Chenopodium album, Abutilon theophrasti | [83] |
| Annual ryegrass (Lolium multiflorum), rye (Secale cereale), bristle oat (Avena strigosa), common vetch (Vicia sativa), radish | Brachiaria plantaginea, Bidens pilosa, Euphorbia heterophylla | [23] |
| Hairy vetch (Vicia villosa), oat (Avena sativa) | Digitaria sanguinalis, Eleusine indica, Amaranthus retroflexus, Datura stramonium | [84] |
| Sorghum sudangrass (Sorghum bicolor × Sorghum sudanense) | Broad leaved weeds | [85] |
| Bristle oat (Avena strigosa), hairy vetch (Vicia villosa) | Amaranthus palmeri, Portulaca oleracea, Helianthus annuus | [86] |
| Rye (Secale cereale), hairy vetch (Vicia villosa), barley (H. vulgare) × triticale, Austrian winter pea (Pisum sativum) | Chenopodium album, Amaranthus hybridus, Thlaspi arvense, Taraxacum officinale, Stellaria media, Elymus repens, Panicum crus-galli, Setaria glauca | [87] |
| White mustard (Sinapis alba) | Amaranthus blitoides, Chenopodium album | [88] |
| Hairy vetch (Vicia villosa), subterranean clover (Trifolium subterraneum), oat (Avena sativa)/hairy vetch (Vicia villosa) | Amaranthus retroflexus, Chenopodium album | [89,90] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mennan, H.; Jabran, K.; Zandstra, B.H.; Pala, F. Non-Chemical Weed Management in Vegetables by Using Cover Crops: A Review. Agronomy 2020, 10, 257. https://doi.org/10.3390/agronomy10020257
Mennan H, Jabran K, Zandstra BH, Pala F. Non-Chemical Weed Management in Vegetables by Using Cover Crops: A Review. Agronomy. 2020; 10(2):257. https://doi.org/10.3390/agronomy10020257
Chicago/Turabian StyleMennan, Husrev, Khawar Jabran, Bernard H. Zandstra, and Firat Pala. 2020. "Non-Chemical Weed Management in Vegetables by Using Cover Crops: A Review" Agronomy 10, no. 2: 257. https://doi.org/10.3390/agronomy10020257
APA StyleMennan, H., Jabran, K., Zandstra, B. H., & Pala, F. (2020). Non-Chemical Weed Management in Vegetables by Using Cover Crops: A Review. Agronomy, 10(2), 257. https://doi.org/10.3390/agronomy10020257





