Farm-Scale Estimation of Total Nitrogen Content in Surface Paddy Soils by Extraction with Commercially Available Hydrogen Peroxide
Abstract
1. Introduction
2. Materials and Methods
2.1. Soil Samples
2.2. Measurement of EC (H2O2) by the Oxydol Method
2.3. Other Analyses
2.4. Statistical Analysis
3. Results
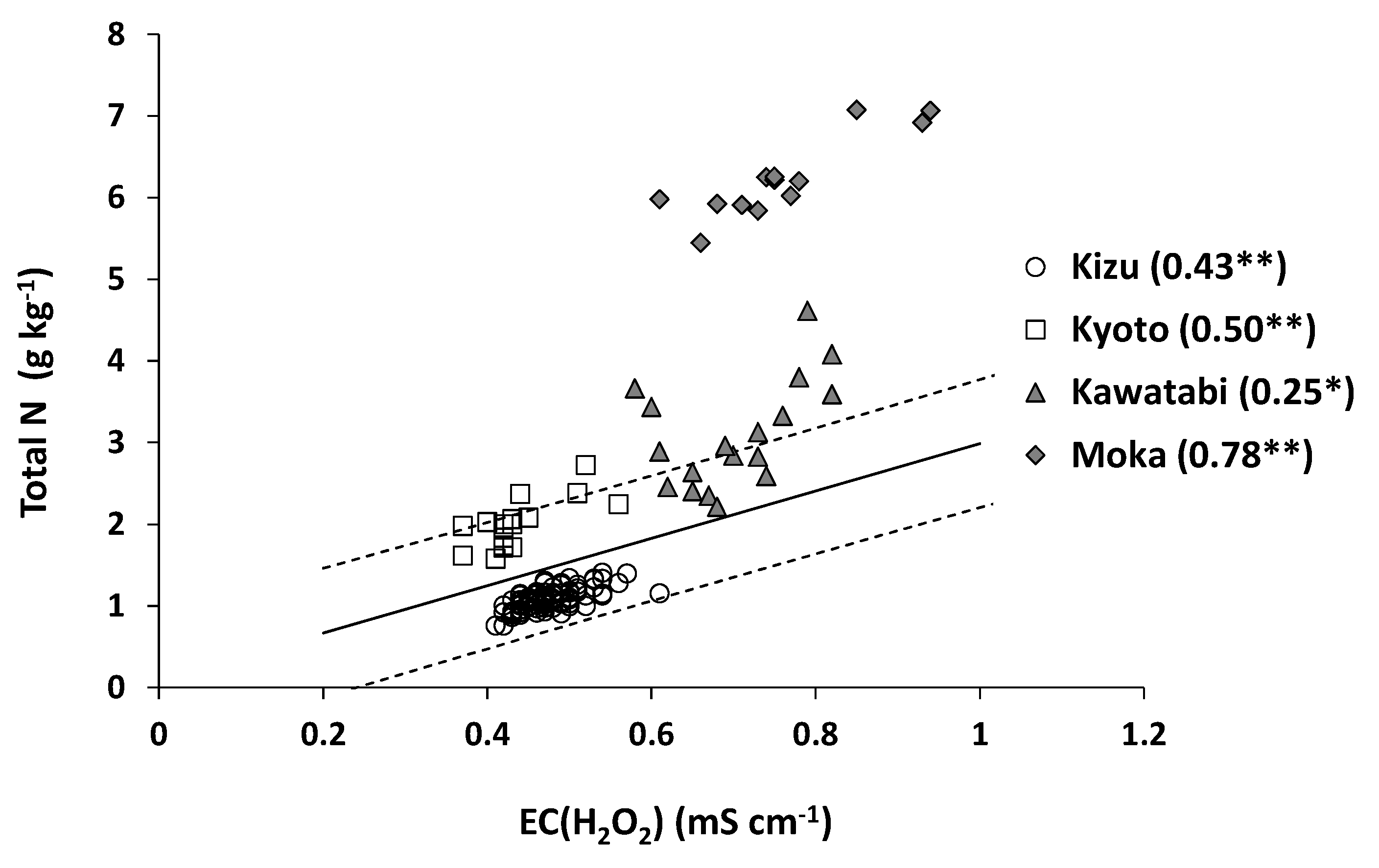
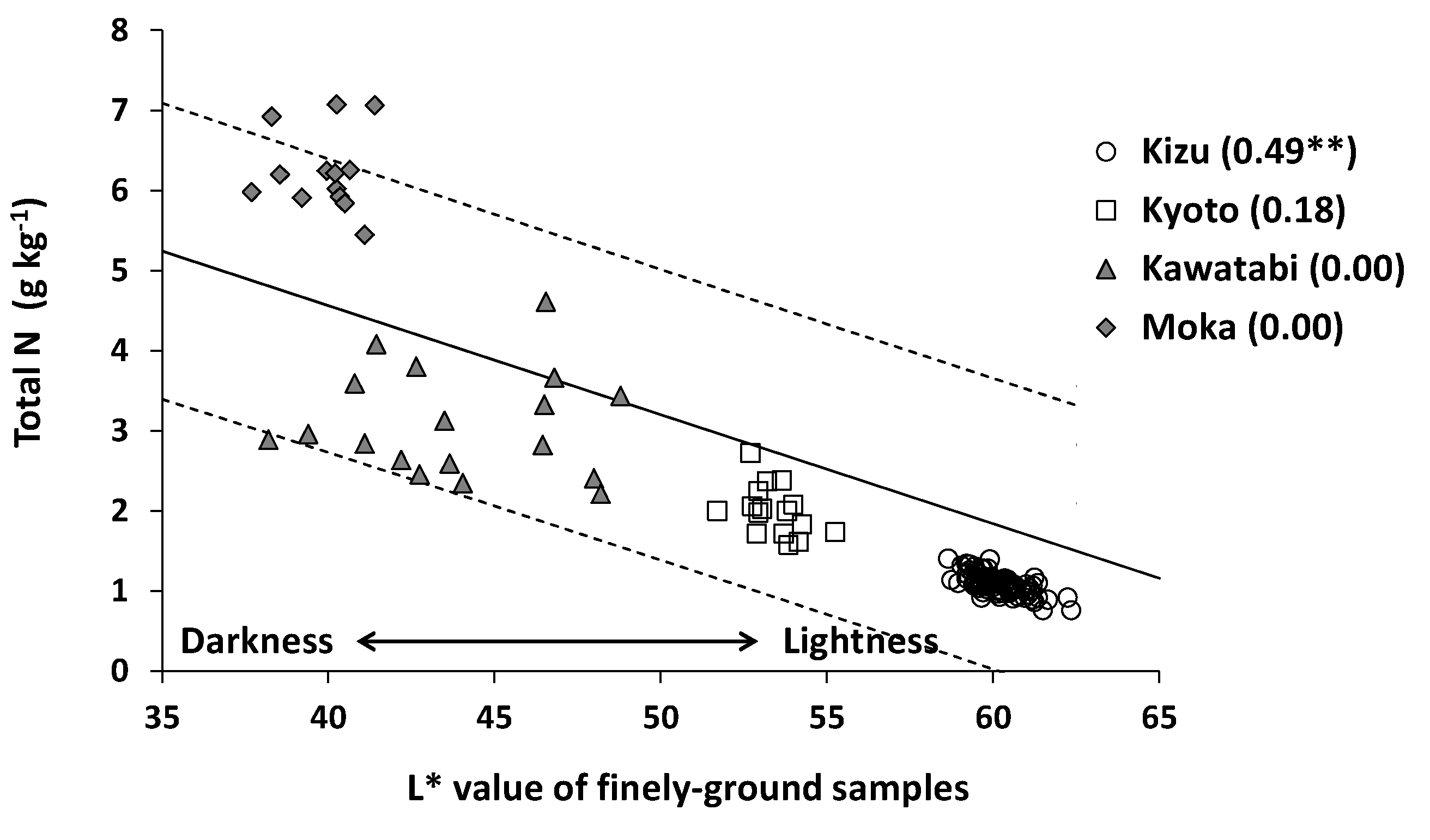
3.1. Farm-Scale Variations in Total N in Surface Paddy Soils
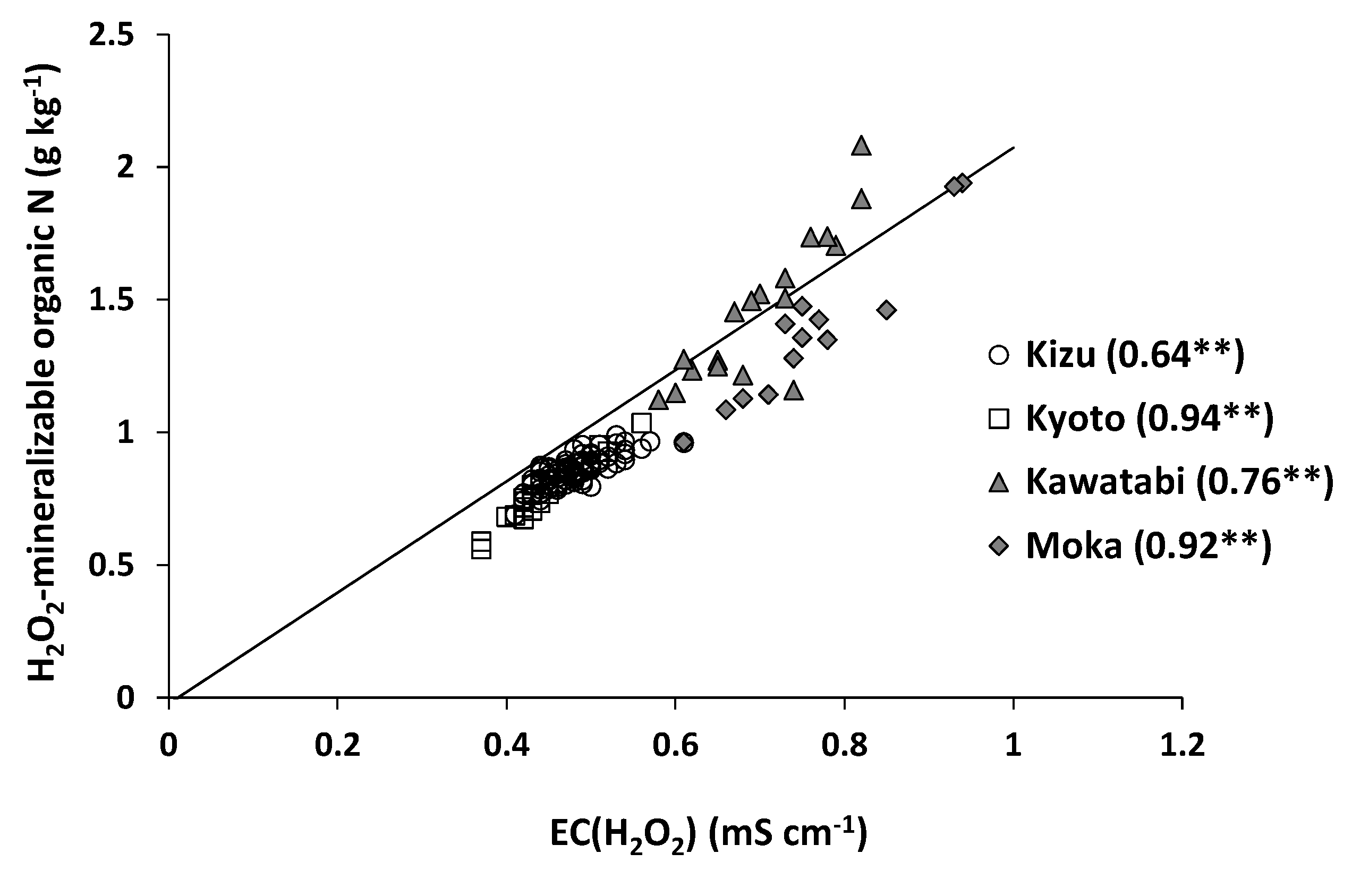
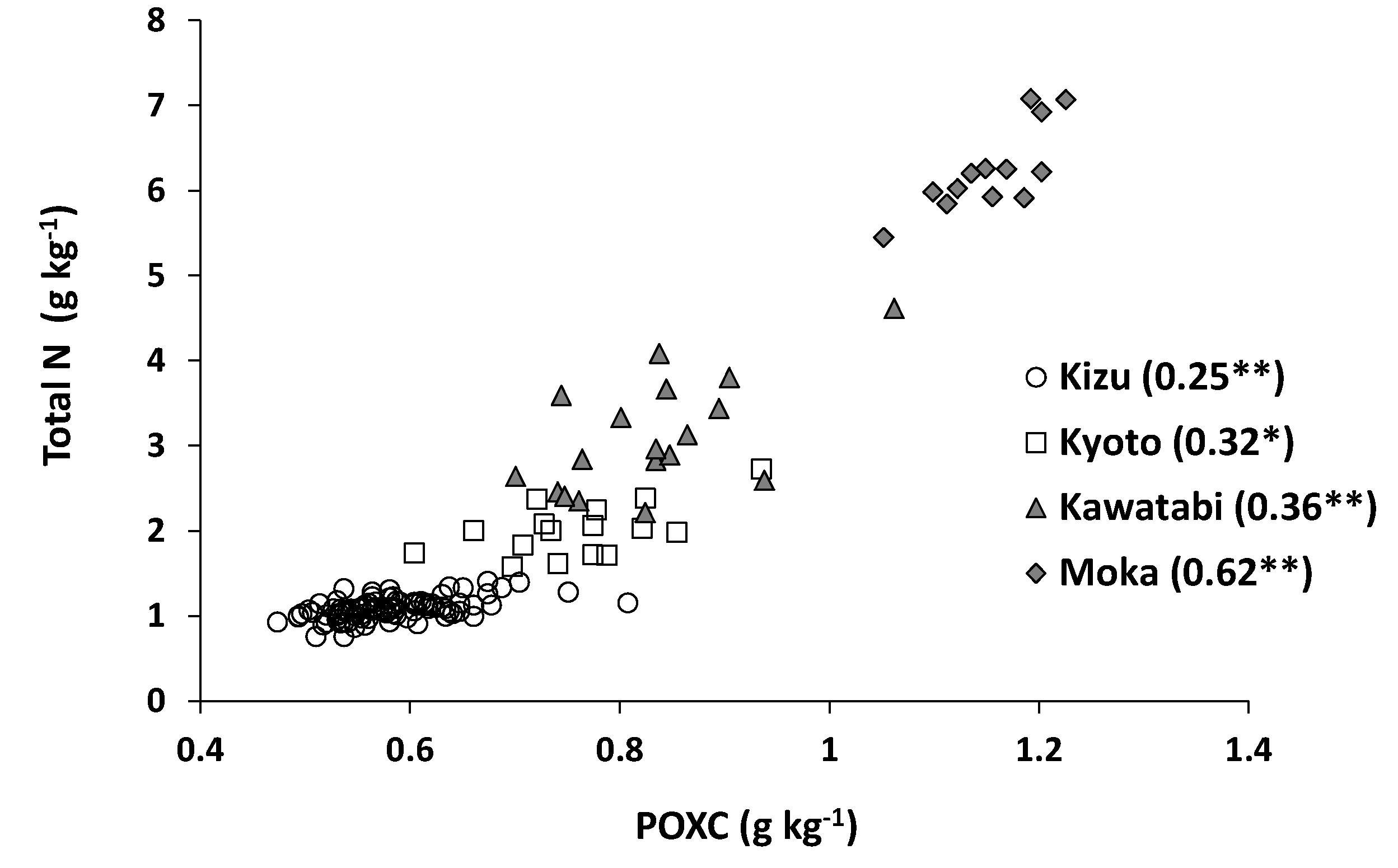
3.2. Accuracy of Estimation of Total N Content
3.3. Comparability of Commercially Available H2O2 Solutions
3.4. Comparing the Estimation Accuracy between the Oxydol Method and Other Methods
4. Discussion
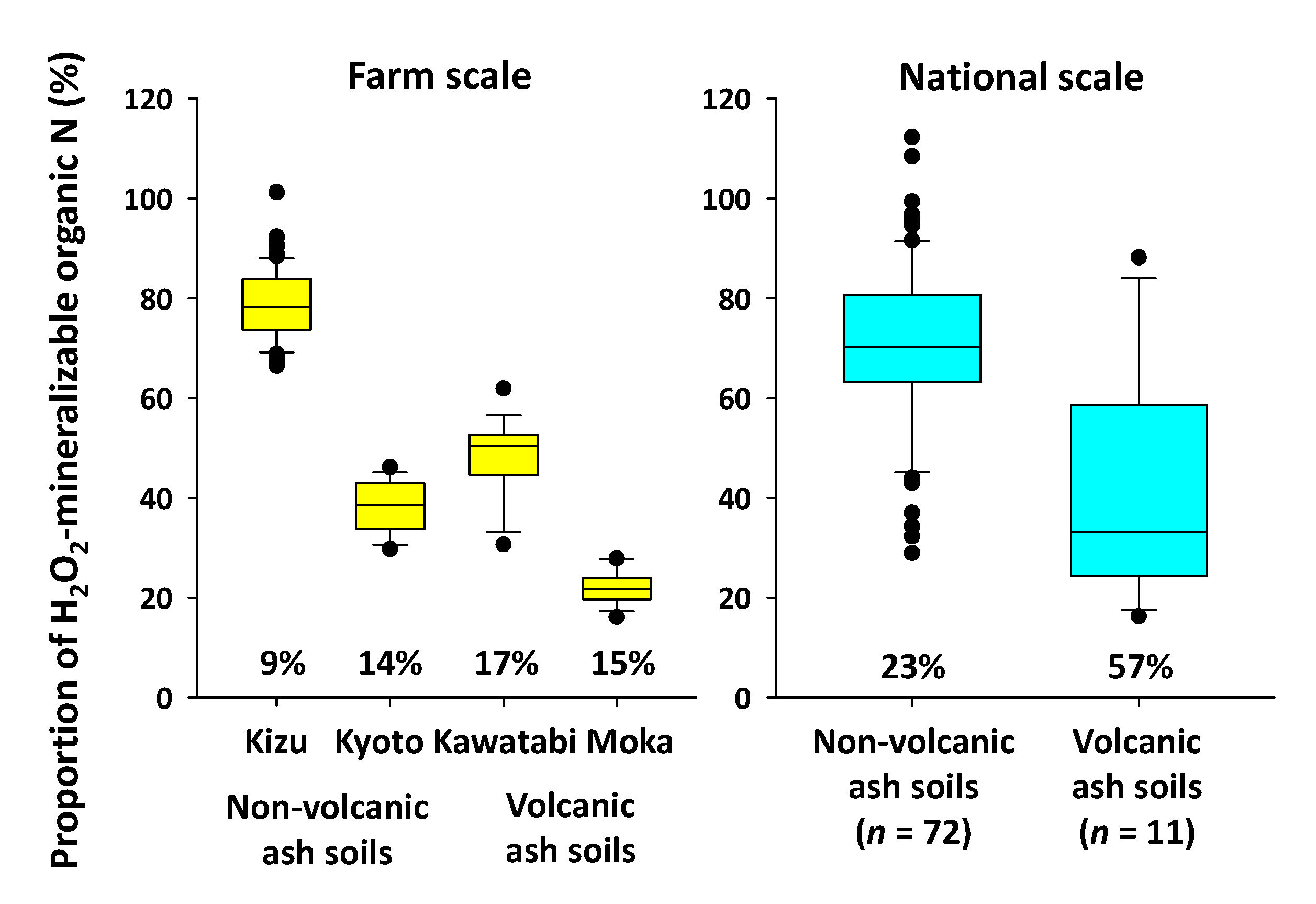
4.1. Merits and Limitations of the Oxydol Method
4.2. Applicability of the Method to Farm-Scale Samples
4.3. Possibility of Using Different Types of H2O2 Solutions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Yoshida, S. Fundamentals of Rice Crop Science; The International Rice Research Institute: Los Baños, Philippines, 1981. [Google Scholar]
- Griffin, T.S. Nitrogen Availability. In Nitrogen in Agricultural Systems, Agronomy Monograph; no. 49; Schepers, J.S., Raun, W.R., Eds.; ASA-CSSA-SSSA: Madison, WI, USA, 2008; pp. 613–646. [Google Scholar]
- Gianello, C.; Bremner, J.M. Comparison of chemical methods of assessing potentially available organic nitrogen in soil. Commun. Soil Sci. Plant Anal. 1986, 17, 215–236. [Google Scholar] [CrossRef]
- Sharifi, M.; Zebarth, B.J.; Burton, D.L.; Grant, C.A.; Hajabbasi, M.A.; Abbassi-Kalo, G. Sodium hydroxide direct distillation: A method for estimating total nitrogen in soil. Commun. Soil Sci. Plant Anal. 2009, 40, 2505–2520. [Google Scholar] [CrossRef]
- Sano, S.; Yanai, J.; Kosaki, T. Relationships between labile organic matter and nitrogen mineralization in Japanese agricultural soils with reference to land use and soil type. Soil Sci. Plant Nutr. 2006, 52, 49–60. [Google Scholar] [CrossRef]
- Sahrawat, K.L. Nitrogen mineralization in lowland rice soils: The role of organic matter quantity and quality. Arch. Agron. Soil Sci. 2010, 56, 337–353. [Google Scholar] [CrossRef]
- Bremner, J.M. Nitrogen-Total (Chapter 37). In Methods of Soil Analysis, Part 3, Chemical Methods; SSSA Book Series no. 5; Sparks, D.L., Ed.; SSSA-ASA: Madison, WI, USA, 1996; pp. 1085–1121. [Google Scholar]
- Christianson, C.B.; Holt, L.S. Rapid digestion procedure for the determination of total nitrogen and nitrogen-15 content of soils. Soil Sci. Soc. Am. J. 1989, 53, 1917–1919. [Google Scholar] [CrossRef]
- Moritsuka, N.; Matsuoka, K.; Katsura, K.; Sano, S.; Yanai, J. Estimation of total nitrogen content in surface paddy soils by measuring their electrical conductivity after hydrogen peroxide treatment. Jpn. J. Soil Sci. Plant Nutr. 2017, 88, 327–335. (In Japanese) [Google Scholar]
- Robinson, G.W. Note on the mechanical analysis of humus soils. J. Agric. Sci. 1922, 12, 287–291. [Google Scholar] [CrossRef]
- Robinson, W.O. The determination of organic matter in soils by means of hydrogen peroxide. J. Agric. Res. 1927, 34, 339–356. [Google Scholar]
- Harada, Y.; Inoko, A. The oxidation products formed from soil organic matter by hydrogen peroxide treatment. Soil Sci. Plant Nutr. 1977, 23, 513–521. [Google Scholar] [CrossRef]
- Guan, G.; Marumoto, T.; Nishiyama, M. Influence of iron and aluminium oxide on the amount of microbial biomass and decomposition of rice straw in H2O2-treated soil. Jpn. J. Soil Sci. Plant Nutr. 1999, 70, 171–176. (In Japanese) [Google Scholar]
- Eith, C.; Kolb, M.; Seubert, A.; Viehweger, K.H. Practical Ion Chromatography. An Introduction; Metrohm Monograph, Metrohm Ltd.: Herisau, Switzerland, 2001. [Google Scholar]
- Schmucker-Castner, J.; Desai, D. Rheology modification of hydrogen peroxide-based applications using cross-linked polyacrylic acid polymers. Int. J. Cosmet. Sci. 1999, 21, 313–325. [Google Scholar] [CrossRef] [PubMed]
- NARO. Digital Soil Map of Japan. 2019. Available online: https://soil-inventory.dc.affrc.go.jp/ (accessed on 15 November 2019). (In Japanese)
- Bundy, L.G.; Meisinger, J.J. Nitrogen Availability Indices (Chapter 41). In Methods of Soil Analysis, Part 2, Microbiological and Biochemical Properties; SSSA Book Series no. 5; Weaver, R.W., Ed.; SSSA: Madison, WI, USA, 1994; pp. 951–984. [Google Scholar]
- Moritsuka, N.; Izawa, G.; Katsura, K.; Matsui, N. Simple method for measuring soil sand content by nylon mesh sieving. Soil Sci. Plant Nutr. 2015, 61, 501–505. [Google Scholar] [CrossRef][Green Version]
- Mulvaney, R.L. Nitrogen—Inorganic forms (Chapter 38). In Methods of Soil Analysis, Part 3, Chemical Methods; SSSA Book Series no. 5; Sparks, D.L., Ed.; SSSA-ASA: Madison, WI, USA, 1996; pp. 1123–1184. [Google Scholar]
- Moritsuka, N.; Matsuoka, K.; Katsura, K.; Sano, S.; Yanai, J. Soil color analysis for statistically estimating—Total carbon, total nitrogen and active iron contents in Japanese agricultural soils. Soil Sci. Plant Nutr. 2014, 60, 475–485. [Google Scholar] [CrossRef]
- Japanese Society of Pedology. A Handbook for Soil Survey. Revised Edition; Japanese Society of Pedology: Hakuyusha, Tokyo, 1997; p. 169. (In Japanese) [Google Scholar]
- van Leeuwen, M.M.W.J.; Heuvelink, G.B.M.; Wallinga, J.; de Boer, I.J.M.; van Dam, J.C.; van Essen, E.A.; Moolenaar, S.W.; Verhoeven, F.P.M.; Stoorvogel, J.J.; Stoof, C.R. Visual soil evaluation: Reproducibility and correlation with standard measurements. Soil Tillage Res. 2018, 178, 167–178. [Google Scholar] [CrossRef]
- Weil, R.R.; Islam, K.R.; Stine, M.A.; Gruver, J.B.; Samson-Liebig, S.E. Estimating active carbon for soil quality assessment: A simplified method for laboratory and field use. Am. J. Altern. Agric. 2003, 18, 3–17. [Google Scholar] [CrossRef]
- Culman, S.W.; Freeman, M.; Snapp, S. Procedure for the Determination of Permanganate Oxidizable Carbon; Kellogg Biological Station, Michigan State University: Hickory Corners, MI, USA, 2014; Available online: https://lter.kbs.msu.edu/protocols/133 (accessed on 5 June 2019).
- MAFF Agricultural Production Bureau. Summary on the National Soil Survey Programs in Japanese Agricultural Land from 1979 to 2003; Agricultural Production Bureau, Ministry of Agriculture, Forestry and Fisheries: Tokyo, Japan, 2008; p. 485. (In Japanese)
- Moritsuka, N.; Matsuoka, K.; Katsura, K.; Yanai, J. Farm-scale variations in soil color as influenced by organic matter and iron oxides in Japanese paddy fields. Soil Sci. Plant Nutr. 2019, 65, 166–175. [Google Scholar] [CrossRef]
- Mikutta, R.; Kleber, M.; Kaiser, K.; Jahn, R. Organic matter removal from soils using hydrogen peroxide, sodium hypochlorite, and disodium peroxodisulfate. Soil Sci. Soc. Am. J. 2005, 69, 120–135. [Google Scholar] [CrossRef]
- Leifeld, J.; Kögel-Knabner, I. Organic carbon and nitrogen in fine soil fractions after treatment with hydrogen peroxide. Soil Biol. Biochem. 2001, 33, 2155–2158. [Google Scholar] [CrossRef]
- Yageta, Y.; Osbahr, H.; Morimoto, Y.; Clark, J. Comparing farmers’ qualitative evaluation of soil fertility with quantitative soil fertility indicators in Kitui County, Kenya. Geoderma 2019, 344, 153–163. [Google Scholar] [CrossRef]
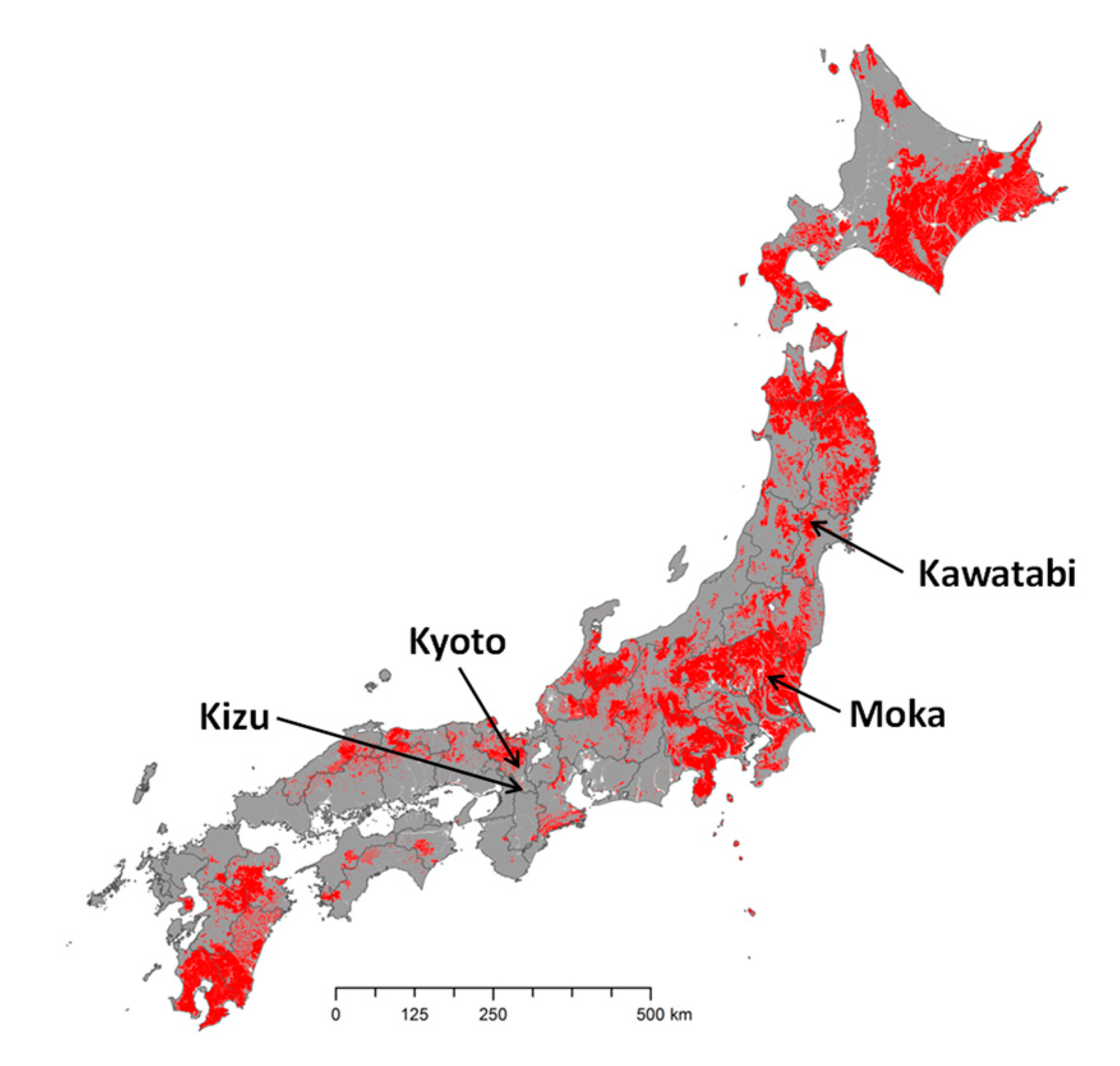


| Site (No. of Samples) | EC (1:5 w/v) (mS cm−1) | Total C (g kg−1) | C/N Ratio | Mineralized N (mg kg−1) | Sand (g kg−1) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Ave. | CV | Ave. | CV | Ave. | CV | Ave. | CV | Ave. | CV | |
| Kizu (89) | 0.17 | 30.3 | 16.1 | 18.1 | 14.7 | 10.6 | 68.7 | 13.2 | 674 | 1.9 |
| Kyoto (16) | 0.05 | 30.0 | 23.6 | 15.5 | 11.8 | 4.0 | 74.7 | 27.5 | 682 | 3.7 |
| Kawatabi (18) | 0.06 | 20.9 | 43.2 | 18.1 | 14.1 | 8.8 | 147.2 | 17.7 | 618 | 11.0 |
| Moka (13) | 0.08 | 31.6 | 89.2 | 5.7 | 14.3 | 4.3 | 111.2 | 32.6 | 441 | 2.4 |
| Type of H2O2 | Country | Type of Stabilizers |
|---|---|---|
| Wako (analytical grade, 30%) | Japan | none |
| Showa | Japan | phenacetin |
| Kenei | Japan | phenacetin, phosphoric acid |
| Kozakai | Japan | ethanol, phenacetin |
| Swan | USA | unknown |
| Essential Oxygen | USA | unknown |
| AGA | Portugal | sodium benzoate |
| Site (No. of Samples) | Average | Maximum | Minimum | CV (%) |
|---|---|---|---|---|
| Kizu (89) | 1.09 | 1.41 | 0.76 | 11.8 |
| Kyoto (16) | 2.00 | 2.72 | 1.58 | 15.5 |
| Kawatabi (18) | 3.10 | 4.61 | 2.21 | 21.3 |
| Moka (13) | 6.24 | 7.08 | 5.45 | 8.0 |
| Nationwide (83) a | 2.24 | 6.32 | 0.75 | 49.0 |
| Nationwide (2750) b | 2.39 | no data | no data | 47.3 |
| Site | Slope | y-Intercept |
|---|---|---|
| Kizu | 2.28 | 0.00 ** |
| Kyoto | 4.29 | 0.13 ** |
| Kawatabi | 4.43 | −0.01 ** |
| Moka | 4.51 | 2.80 ** |
| Japan (nationwide) | 2.90 | 0.09 |
| Type of H2O2 | EC (mS cm−1) | Na (mg L−1) |
|---|---|---|
| Wako (30%) | 0.010 | 0.10 |
| Showa | 0.029 | 0.51 |
| Kenei | 0.050 | 0.29 |
| Kozakai | 0.026 | 0.54 |
| Swan | 0.19 | 33.2 |
| Essential Oxygen | 1.40 | 321.6 |
| AGA | 0.55 | 165.5 |
| Type of H2O2 | Average EC (H2O2) (mS cm−1) | R with Showa | Comparability with Showa |
|---|---|---|---|
| Showa | 0.53 | - | - |
| Kenei | 0.50 * | 0.99 | high |
| Kozakai | 0.55 | 0.92 | very high |
| Swan | 0.53 | 0.95 | very high |
| Essential Oxygen | 1.20 ** | −0.73 | low |
| AGA | 0.98 ** | 0.95 | medium |
| Wako (3%) | 0.49 * | 0.94 | high |
| Wako (6%) | 0.51 | 0.90 | very high |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moritsuka, N.; Saito, H.; Tajima, R.; Takahashi, Y.; Hirai, H. Farm-Scale Estimation of Total Nitrogen Content in Surface Paddy Soils by Extraction with Commercially Available Hydrogen Peroxide. Agronomy 2020, 10, 40. https://doi.org/10.3390/agronomy10010040
Moritsuka N, Saito H, Tajima R, Takahashi Y, Hirai H. Farm-Scale Estimation of Total Nitrogen Content in Surface Paddy Soils by Extraction with Commercially Available Hydrogen Peroxide. Agronomy. 2020; 10(1):40. https://doi.org/10.3390/agronomy10010040
Chicago/Turabian StyleMoritsuka, Naoki, Hiroki Saito, Ryosuke Tajima, Yukitsugu Takahashi, and Hideaki Hirai. 2020. "Farm-Scale Estimation of Total Nitrogen Content in Surface Paddy Soils by Extraction with Commercially Available Hydrogen Peroxide" Agronomy 10, no. 1: 40. https://doi.org/10.3390/agronomy10010040
APA StyleMoritsuka, N., Saito, H., Tajima, R., Takahashi, Y., & Hirai, H. (2020). Farm-Scale Estimation of Total Nitrogen Content in Surface Paddy Soils by Extraction with Commercially Available Hydrogen Peroxide. Agronomy, 10(1), 40. https://doi.org/10.3390/agronomy10010040





