Can Trichoderma-Based Biostimulants Optimize N Use Efficiency and Stimulate Growth of Leafy Vegetables in Greenhouse Intensive Cropping Systems?
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Setup and Management
2.2. Soil Plant Analysis Development (SPAD) Index, Plants Sampling, and Analysis
2.3. Leaf Colorimetry, Antioxidant Activity, and Total Phenols Analysis
2.4. Soil Analyses
2.5. Data Elaboration and Statistical Analysis
3. Results
3.1. Effect of N Fertilization Levels and Microbial-Based Biostimulants on Lettuce and Rocket Productive Performance
3.2. Effect of N Fertilization Levels and Microbial-Based Biostimulants on Lettuce and Rocket N Uptake and Balance and Nitrogen Use Efficiency
3.3. SPAD Index, Leaf Colorimetry, Antioxidant Activity, and Total Phenols Content
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kant, S. Understanding nitrate uptake, signaling and remobilisation for improving plant nitrogen use efficiency. Semin. Cell Dev. Biol. 2018, 74, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Awaad, M.S.; Badr, R.A.; Badr, M.A.; Abd-elrahman, A.H. Effects of different nitrogen and potassium sources on lettuce (Lactuca sativa L.) yield in a sandy soil. Eurasian J. Soil Sci. 2016, 5, 299–306. [Google Scholar] [CrossRef][Green Version]
- Weightman, R.M.; Hudson, E.M. Noxious or nutritious? Progress in controlling nitrate as a contaminant in leafy crop species. Food Energy Secur. 2013, 2, 141–156. [Google Scholar] [CrossRef]
- Nurzyńska-Wierdak, R. Growth and yield of garden rocket (Eruca sativa Mill.) affected by nitrogen and potassium fertilization. Acta Sci. Pol. Hortic. 2009, 8, 23–33. [Google Scholar]
- Bozkurt, S.M.; Mansuroğlu, G.S.; Kara, M.; Önder, S. Responses of lettuce to irrigation levels and nitrogen forms. Afr. J. Agric. Res. 2009, 4, 1171–1178. [Google Scholar]
- Gülser, F.; Sönmez, F.; Boysan, S. Effect of calcium nitrate and humic acid applications on growth and yield criteria of pepper seedling under salt stress. J. Environ. Biol. 2010, 31, 873–876. [Google Scholar]
- European Council. Directive of the Council of December 12, 1991 Concerning the Protection of Waters against Pollution Caused by Nitrates from Agricultural Sources (1991/676/EEC); European Commission: Brussels, Belgium, 1991. [Google Scholar]
- Davidson, E.A.; David, M.B.; Galloway, J.N.; Goodale, C.L.; Haeuber, R.; Harrison, J.A.; Howarth, R.W.; Jaynes, D.B.; Lowrance, R.R.; Nolan, B.T.; et al. Excess Nitrogen in the U.S. Environment: Trends, Risks, and Solutions; Issues in Ecology; Ecological Society of America: Washington, DC, USA, 2012. [Google Scholar]
- European Commission. Commission Regulation (EC) No 1881/2006 of 19 December 2006 Setting Maximum Levels for Certain Contaminants in Foodstuffs; European Commission: Brussels, Belgium, 2006. [Google Scholar]
- Lyu, X.; Wang, T.; Ma, Z.M.; Zhao, C.Y.; Siddiquee, K.H.M.; Ju, X.T. Enhanced efficiency nitrogen fertilizers maintain yields and mitigate global warming potential in an intensified spring wheat system. Field Crops Res. 2019, 244, 107624. [Google Scholar] [CrossRef]
- Walsh, O.; Raun, W.; Klatt, A.; Solie, J. Effect of delayed nitrogen fertilization on maize (Zea mays L.) grain yields and nitrogen use efficiency. J. Plant Nutr. 2012, 35, 538–555. [Google Scholar] [CrossRef]
- Mi, W.H.; Gao, Q.; Xia, S.Q.; Zhao, H.T.; Wu, L.H.; Mao, W.; Hu, Z.P.; Liu, Y.L. Medium-term effects of different types of N fertilizer on yield, apparent N recovery, and soil chemical properties of a double rice cropping system. Field Crops Res. 2019, 234, 87–94. [Google Scholar] [CrossRef]
- Mi, G.; Chen, F.; Yuan, L.; Zhang, F. Ideotype root system architecture for maize to achieve high yield and resource use efficiency in intensive cropping systems. Adv. Agron. 2016, 139, 73–97. [Google Scholar]
- Rouphael, Y.; Colla, G. Synergistic biostimulatory action: Designing the next generation of plant biostimulants for sustainable agriculture. Front. Plant Sci. 2018, 9, 1655. [Google Scholar] [CrossRef] [PubMed]
- Battacharyya, D.; Babgohari, M.Z.; Rathor, P.; Prithiviraj, B. Seaweed extracts as biostimulants in horticulture. Sci. Hortic. 2015, 196, 39–48. [Google Scholar] [CrossRef]
- Canellas, L.P.; Olivares, F.L.; Aguiar, N.O.; Jones, D.L.; Nebbioso, A.; Mazzei, P.; Piccolo, A. Humic and fulvic acids as biostimulants in horticulture. Sci. Hortic. 2015, 196, 15–27. [Google Scholar] [CrossRef]
- Colla, G.; Nardi, S.; Cardarelli, M.; Ertani, A.; Lucini, L.; Canaguier, R.; Youssef, R. Protein hydrolysates as biostimulants in horticulture. Sci. Hortic. 2015, 196, 28–38. [Google Scholar] [CrossRef]
- Colla, G.; Hoagland, L.; Ruzzi, M.; Cardarelli, M.; Bonini, P.; Canaguier, R.; Rouphael, Y. Biostimulant action of protein hydrolysates: Unraveling their effects on plant physiology and microbiome. Front. Plant Sci. 2017, 8, 2202. [Google Scholar] [CrossRef]
- Rouphael, Y.; Franken, P.; Schneider, C.; Schwarz, D.; Giovannetti, M.; Agnolucci, M.; De Pascale, S.; Bonini, P.; Colla, G. Arbuscular mycorrhizal fungi act as biostimulants in horticultural crops. Sci. Hortic. 2015, 196, 91–108. [Google Scholar] [CrossRef]
- Rouphael, Y.; Kyriacou, M.C.; Petropoulos, S.A.; De Pascale, S.; Colla, G. Improving vegetable quality in controlled environments. Sci. Hortic. 2018, 234, 275–289. [Google Scholar] [CrossRef]
- Rouphael, Y.; Spíchal, L.; Panzarová, K.; Casa, R.; Colla, G. High-throughput plant phenotyping for developing novel biostimulants: From lab to field or from field to lab? Front. Plant Sci. 2018, 9, 1197. [Google Scholar] [CrossRef]
- Saia, S.; Colla, G.; Raimondi, G.; Di Stasio, E.; Cardarelli, M.; Bonini, P.; Vitaglione, P.; De Pascale, S.; Rouphael, Y. An endophytic fungi-based biostimulant modulated lettuce yield, physiological and functional quality responses to both moderate and severe water limitation. Sci. Hortic. 2019, 256, 108595. [Google Scholar] [CrossRef]
- López-Bucio, J.; Pelagio-Flores, R.; Herrera-Estrella, A. Trichoderma as biostimulant: Exploiting the multi level properties of a plant beneficial fungus. Sci. Hortic. 2015, 196, 109–123. [Google Scholar] [CrossRef]
- Ruzzi, M.; Aroca, R. Plant growth-promoting rhizobacteria act as biostimulants in horticulture. Sci. Hortic. 2015, 196, 124–134. [Google Scholar] [CrossRef]
- Woo, S.L.; Ruocco, M.; Vinale, F.; Nigro, M.; Marra, R.; Lombardi, N.; Pascale, A.; Lanzuise, S.; Manganiello, G.; Lorito, M. Trichoderma-based products and their widespread use in agriculture. Open Mycol. J. 2014, 8, 71–126. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y. Biostimulants in Horticulture. Sci. Hortic. 2015, 196, 1–2. [Google Scholar] [CrossRef]
- Lorito, M.; Woo, S.L. Trichoderma: A Multi-Purpose Tool for Integrated Pest Management. In Principles of Plant-Microbe Interactions; Lugtenberg, B., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 345–353. [Google Scholar]
- Rouphael, Y.; Cardarelli, M.; Bonini, P.; Colla, G. Synergistic action of a microbial-based biostimulant and a plant derived-protein hydrolysate enhances lettuce tolerance to alkalinity and salinity. Front. Plant Sci. 2017, 8, 131. [Google Scholar] [CrossRef] [PubMed]
- Marra, R.; Lombardi, N.; D’Errico, G.; Troisi, J.; Scala, G.; Vinale, F.; Woo, S.L.; Bonanomi, G.; Lorito, M. Application of Trichoderma strains and metabolites enhances soybean productivity and nutrient content. J. Agric. Food Chem. 2019, 67, 1814–1822. [Google Scholar] [CrossRef]
- Harman, G.R. Myths and dogmas of biocontrol. Changes in perceptions derived from research on Trichoderma harzianum T-22. Plant Dis. 2000, 84, 377–393. [Google Scholar] [CrossRef]
- Harman, G.E. Overview of mechanisms and uses of Trichoderma spp. Phytopathology 2006, 96, 190–194. [Google Scholar] [CrossRef]
- Colla, G.; Rouphael, Y.; Di Mattia, E.; El-Nakhel, C.; Cardarelli, M. Co-inoculation of Glomus intraradices and Trichoderma atroviride acts as a biostimulant to promote growth, yield and nutrient uptake of vegetable crops. J. Sci. Food Agric. 2015, 95, 1706–1715. [Google Scholar] [CrossRef]
- Woo, S.L.; Pepe, O. Microbial consortia: Promising probiotics as plant biostimulants for sustainable agriculture. Front. Plant Sci. 2018, 9, 1801. [Google Scholar] [CrossRef]
- Liu, J.; Liu, H.; Huang, S.; Yang, X.; Wang, B.; Li, X.; Ma, Y. Nitrogen efficiency in long-term wheat–maize cropping systems under diverse field sites in China. Field Crops Res. 2010, 118, 145–151. [Google Scholar] [CrossRef]
- Singh, B.N.; Dwivedi, P.; Sarma, B.K.; Singh, G.S.; Singh, H.B. Trichoderma asperellum T42 reprograms tobacco for enhanced nitrogen utilization efficiency and plant growth when fed with N nutrients. Front. Plant Sci. 2018, 9, 163. [Google Scholar] [CrossRef] [PubMed]
- Fiorentino, N.; Ventorino, V.; Woo, S.L.; Pepe, O.; De Rosa, A.; Gioia, L.; Romano, I.; Lombardi, N.; Napolitano, M.; Colla, G.; et al. Trichoderma-based biostimulants modulate rhizosphere microbial populations and improve N uptake efficiency, yield, and nutritional quality of leafy vegetables. Front. Plant Sci. 2018, 9, 743. [Google Scholar] [CrossRef] [PubMed]
- Bremner, J.M. Total nitrogen. In Methods of Soil Analysis; Black, C.A., Evans, D.D., White, I.L., Ensminger, L.E., Clark, F.E., Eds.; American Society of Agronomy: Madison, WI, USA, 1965; pp. 1149–1178. [Google Scholar]
- Rouphael, Y.; Colla, G.; Giordano, M.; El-Nakhel, C.; Kyriacou, M.C.; De Pascale, S. Foliar applications of a legume-derived protein hydrolysate elicit dose dependent increases of growth, leaf mineral composition, yield and fruit quality in two greenhouse tomato cultivars. Sci. Hortic. 2017, 226, 353–360. [Google Scholar] [CrossRef]
- Fogliano, V.; Verde, V.; Randazzo, G.; Ritieni, A. Method for measuring antioxidant activity and its application to monitoring the antioxidant capacity of wines. J. Agric. Food Chem. 1999, 47, 1035–1040. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Meth. Enzymol. 1999, 299, 152–178. [Google Scholar]
- Fagnano, M.; Adamo, P.; Zampella, M.V.; Fiorentino, N. Environmental and agronomic impact of fertilization with composted organic fraction from municipal solid waste. Agric. Ecosyst. Environ. 2011, 141, 100–107. [Google Scholar] [CrossRef]
- Ladha, J.K.; Pathak, H.J.; Krupnik, T.; Six, J.; van Kessel, C. Efficiency of fertilizer nitrogen in cereal production: Retrospects and prospects. Adv. Agron. 2005, 87, 85–156. [Google Scholar]
- Kyriacou, M.C.; Rouphael, Y.; Colla, G.; Zrenner, R.M.; Schwarz, D. Vegetable grafting: The implications of a growing agronomic imperative for vegetable fruit quality and nutritive value. Front. Plant Sci. 2017, 8, 741. [Google Scholar] [CrossRef]
- Gázquez, J.C.; Meca, D.E.; López, J.C.; Baeza, E.J.; Pérez-Parra, J.J. Comparison of different strategies for fruit set in greenhouse zucchini (Cucurbita pepo L.). Acta Hortic. 2012, 927, 187–194. [Google Scholar] [CrossRef]
- Pohl, A.; Grabowska, A.; Kalisz, A.; Sękara, A. The eggplant yield and fruit composition as affected by genetic factor and biostimulant application. Not. Bot. Horti Agrobot. Cluj Napoca 2019, 47, 929–938. [Google Scholar] [CrossRef]
- Nayyer, M.A.; Tripathi, V.K.; Kumar, S.; Lal, D.; Tiwari, B. Influence of integrated nutrient management, growth, yield, and quality of tissue cultured banana (Musa x paradisiaca) cv grand naine. Indian J. Agric. Sci. 2014, 84, 680–683. [Google Scholar]
- Clemente, R.; Walker, D.J.; Bernal, M.P. Uptake of heavy metals and As by Brassica juncea grown in a contaminated soil in Aznalcollar (Spain): The effect of soil amendments. Environ. Pollut. 2005, 138, 46–58. [Google Scholar] [CrossRef]
- Fiorentino, N.; Mori, M.; Cenvinzo, V.; Duri, L.G.; Gioia, L.; Visconti, D.; Fagnano, M. Assisted phytoremediation for restoring soil fertility in contaminated and degraded land. Ital. J. Agron. 2018, 13, 34–44. [Google Scholar]
- Justes, E.; Beaudoin, N.; Bertuzzi, P.; Charles, R.; Constantin, J.; Dürr, C.; Hermon, C.; Joannon, A.; Le Bas, C.; Mary, B.; et al. The use of cover crops to reduce nitrate leaching: Effect on the water and nitrogen balance and other ecosystem services. In Proceedings-International Fertiliser Society; International Fertiliser Society: Cambrige, UK, 2012; pp. 4–43. [Google Scholar]
- Lucini, L.; Colla, G.; Miras Morebo, M.B.; Bernardo, L.; Cardarelli, M.; Terzi, V.; Bonini, P.; Rouphael, Y. Inoculation of Rhizoglomus irregulare or Trichoderma atroviride differentially modulates metabolite profiling of wheat root exudates. Phytochemistry 2019, 157, 158–167. [Google Scholar] [CrossRef]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Möller, K.; Stinner, W.; Deuker, A.; Leithold, G. Effects of different manuring systems with and without biogas digestion on nitrogen cycle and crop yield in mixed organic dairy farming systems. Nutr. Cycl. Agroecosyst. 2008, 82, 209–232. [Google Scholar] [CrossRef]
- Remy, J.C.; Marin-Lafleche, A. L’analyse de terre: Realisation d’un programme d’interpretation automatique. Ann. Agron. 1974, 25, 607–632. [Google Scholar]
- Lei, L.; Mc Donald, L.M. Soil moisture and temperature effects on nitrogen mineralization in a high tunnel farming system. Commun. Soil Sci. Plant Anal. 2019, 50, 2140–2150. [Google Scholar] [CrossRef]
- Fiorentino, N.; Ventorino, V.; Bertora, B.; Pepe, O.; Moschetti, G.; Grignani, C.; Fagnano, M. Changes in soil mineral N content and abundances of bacterial communities involved in N reactions under laboratory conditions as predictors of soil N availability to maize under field conditions. Biol. Fertil. Soils 2016, 52, 523–537. [Google Scholar] [CrossRef]
- Rice-Evans, C.A.; Miller, J.; Paganga, G. Antioxidant properties of phenolic compounds. Trends Plant Sci. 1997, 2, 152–159. [Google Scholar] [CrossRef]
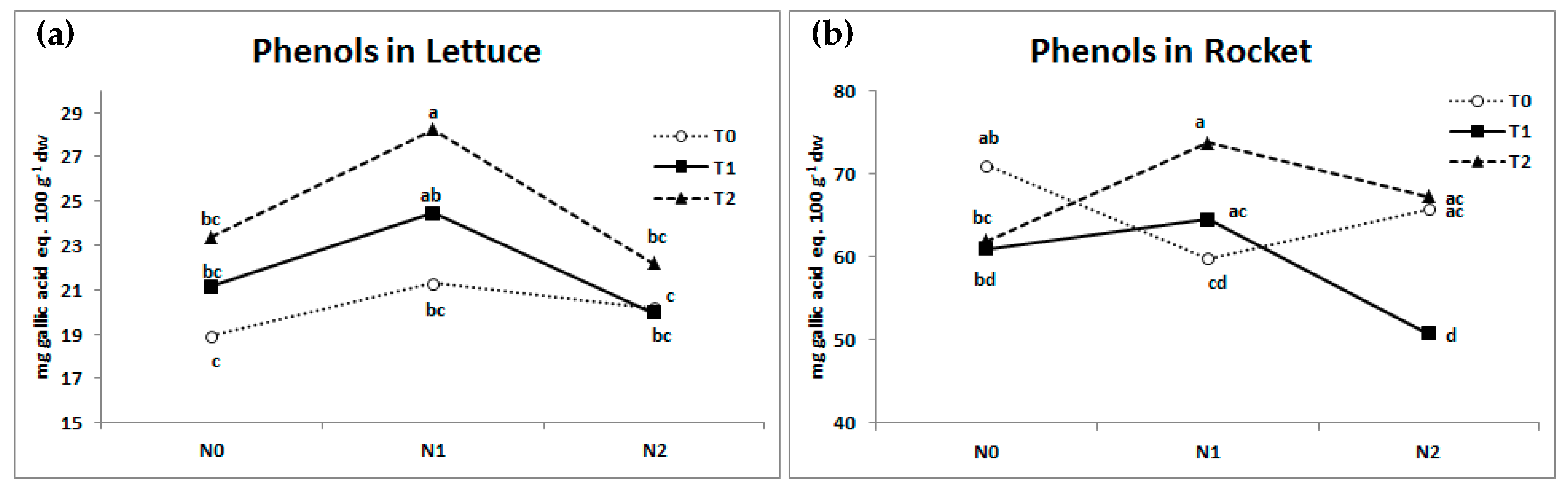
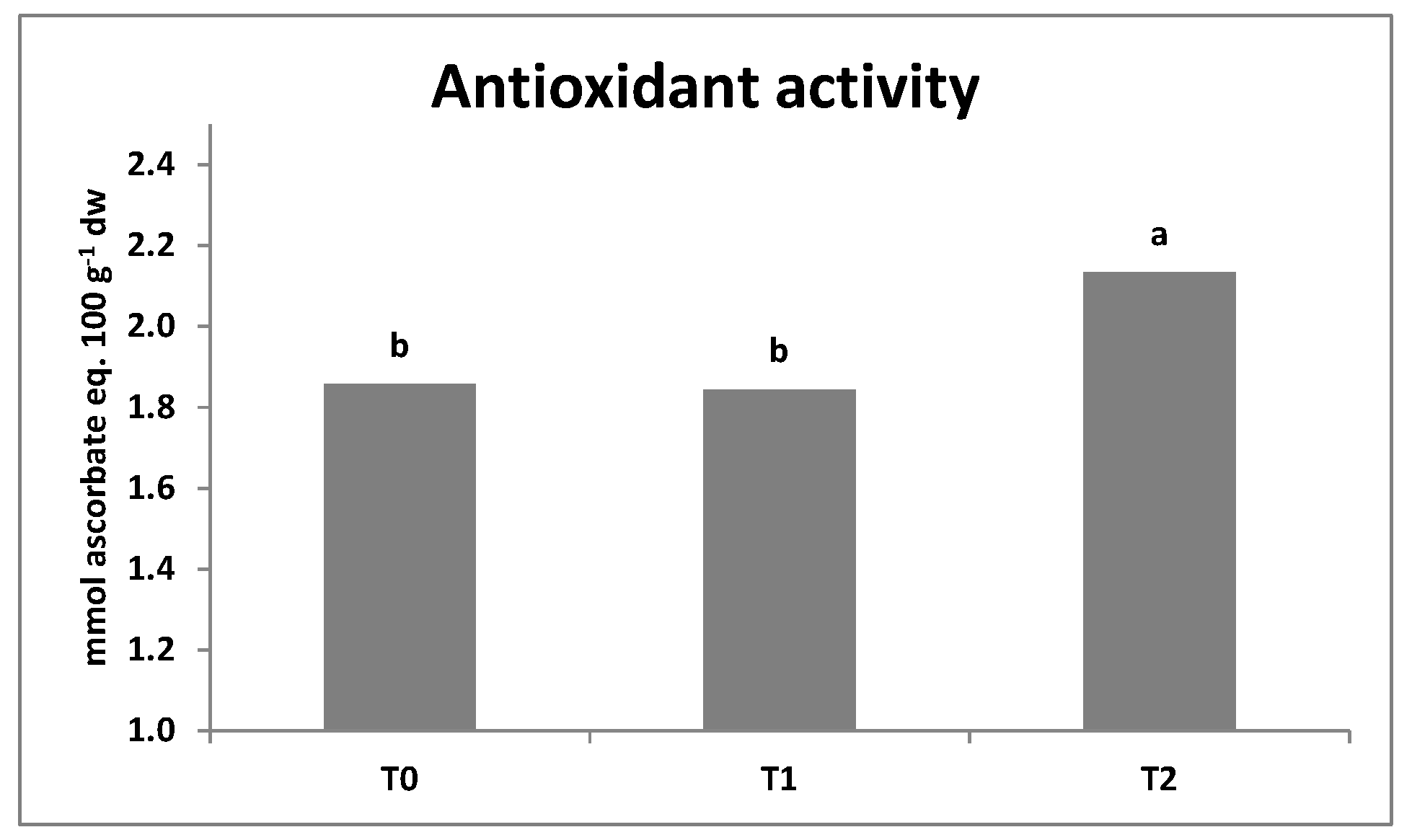
| Lettuce | Rocket | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Treatment | Level | Marketable Yield | Unmarketable Yield | Leaf Dry Matter Content | Dry Biomass Production | Marketable Yield | Unmarketable Yield | Leaf Dry Matter Content | Dry Biomass Production |
| (Mg fw ha−1) | (%) | (%) | (Mg dw ha−1) | (Mg fw ha−1) | (%) | (%) | (Mg dw ha−1) | ||
| Fertilization (N) | N2 | 45.6 a | 44.0 | 3.7 | 3.0 a | 43.2 a | 5.2 | 4.8 | 2.2 |
| N1 | 49.9 a | 43.2 | 3.5 | 3.1 a | 42.8 a | 3.8 | 5.1 | 2.3 | |
| N0 | 33.2 b | 38.4 | 3.8 | 1.9 b | 36.6 b | 4.4 | 5.4 | 2.6 | |
| Trichoderma (T) | T0 | 38.2 b | 43.4 | 3.7 | 2.5 b | 41.0 | 4.1 | 5.1 | 2.2 |
| T1 | 44.1 a | 41.3 | 3.5 | 2.6 b | 40.3 | 4.2 | 4.9 | 2.3 | |
| T2 | 46.5 a | 40.9 | 3.7 | 2.9 a | 41.2 | 5.1 | 5.3 | 2.3 | |
| N × T | N2-T0 | 45.0 ce | 42.4 | 3.8 | 3.0 b | 47.1 a | 4.8 | 4.7 | 2.3 ab |
| N2-T1 | 45.5 bd | 44.9 | 3.6 | 3.0 b | 44.5 ab | 4.6 | 4.5 | 2.1 ac | |
| N2-T2 | 46.4 bc | 44.8 | 3.6 | 2.9 b | 37.9 bd | 6.2 | 5.2 | 2.1 ac | |
| N1-T0 | 40.8 bc | 48.7 | 3.6 | 2.9 b | 43.4 ab | 2.7 | 5.3 | 2.4 ab | |
| N1-T1 | 53.0 ab | 41.4 | 3.2 | 2.9 b | 42.4 ac | 3.8 | 5.1 | 2.2 ac | |
| N1-T2 | 56.0 a | 39.4 | 3.8 | 3.6 a | 42.5 ac | 4.9 | 4.8 | 2.1 ac | |
| N0-T0 | 28.2 f | 39.1 | 3.8 | 1.5 d | 32.5 d | 4.8 | 5.3 | 1.8 bc | |
| N0-T1 | 33.1 ef | 37.5 | 3.8 | 2.0 c | 34.1 cd | 4.1 | 4.9 | 1.7 bc | |
| N0-T2 | 38.4 df | 38.6 | 3.9 | 2.1 c | 43.3 ab | 4.3 | 5.9 | 2.6 a | |
| Significance | N | ** | n.s. | n.s. | ** | * | n.s. | n.s. | n.s. |
| T | * | n.s. | n.s. | ** | n.s. | n.s. | n.s. | n.s. | |
| N × T | * | n.s. | n.s. | * | * | n.s. | n.s. | * | |
| Lettuce | Rocket | Crop Sequence | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Treatment | Level | N Uptake | N Balance | ANR | NUE-T | NUE-C | N Uptake | N Balance | ANR | NUE-T | NUE-C | N Balance |
| kg N ha−1 | % | kg N ha−1 | % | kg N ha−1 | ||||||||
| Fertilization (N) | N2 | 143 a | 37 | 80 | 31 | 42 | 123 | −3 | 102 | 1 | 17 | 34 |
| N1 | 150 a | −60 | 167 | 70 | 91 | 134 | −74 | 224 | 22 | 55 | −135 | |
| N0 | 87 b | 121 | ||||||||||
| Trichoderma (T) | T0 | 114 b | −2 | 112 | 56 | 56 | 125 | −48 | 177 | 50 a | 50 | −50 |
| T1 | 128 ab | −12 | 123 | 47 | 66 | 116 | −34 | 159 | 31 a | 32 | −46 | |
| T2 | 139 a | −21 | 135 | 49 | 78 | 136 | −34 | 153 | −47 b | 26 | −55 | |
| N × T | N2-T0 | 142 b | 38 a | 79 c | 41 | 41 d | 126 ac | −6 | 105 | 20 bc | 20 | 32 |
| N2-T1 | 147 ab | 33 a | 82 c | 32 | 44 cd | 115 bc | 5 | 96 | 11 cd | 11 | 37 | |
| N2-T2 | 140 b | 40 a | 78 c | 20 | 40 d | 127 ac | −7 | 106 | −28 de | 21 | 33 | |
| N1-T0 | 131 bc | −41 b | 146 b | 70 | 70 bc | 150 ab | −90 | 250 | 80 a | 80 | −131 | |
| N1-T1 | 147 ab | −57 bc | 163 b | 62 | 87 b | 133 ac | −73 | 221 | 52 ab | 52 | −130 | |
| N1-T2 | 173 a | −83 c | 192 a | 77 | 116 a | 121 ac | −61 | 201 | −66 e | 32 | −143 | |
| N0-T0 | 68 e | 101 c | ||||||||||
| N0-T1 | 91 de | 102 c | ||||||||||
| N0-T2 | 103 cd | 161 a | ||||||||||
| Significance | N | ** | ** | ** | ** | ** | n.s. | ** | ** | n.s. | ** | ** |
| T | ** | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | ** | n.s. | n.s. | |
| N × T | * | * | * | n.s. | ** | * | n.s. | n.s. | ** | n.s. | n.s. | |
| Lettuce | Rocket | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Treatment | Level | SPAD Index | L* | a* | b* | SPAD Index | L* | a* | b* |
| Fertilization (N) | N2 | 36.9 a | 48.0 | −14.1 | 20.5 | 33.5 a | 43.8 a | −17.6 b | 26.9 a |
| N1 | 36.5 ab | 47.7 | −14.2 | 20.4 | 32.1 b | 42.7 b | −16.3 a | 25.1 b | |
| N0 | 35.3 b | 45.1 | −15.5 | 23.5 | 31.1 b | 42.2 b | −16.3 a | 26.0 ab | |
| Trichoderma (T) | T0 | 36.0 | 48.0 | −14.2 | 20.6 | 32.1 | 43.0 | −17.0 | 26.6 |
| T1 | 36.1 | 46.6 | −14.7 | 21.1 | 31.7 | 42.9 | −16.6 | 25.8 | |
| T2 | 36.5 | 46.3 | −14.9 | 22.8 | 32.9 | 42.6 | −16.4 | 25.5 | |
| N × T | N2-T0 | 37.2 | 48.2 | −14.1 | 20.3 | 33.8 | 44.3 a | −18.6 e | 28.6 a |
| N2-T1 | 36.8 | 47.8 | −14.1 | 20.2 | 32.2 | 42.9 ac | −16.6 bd | 25.5 bc | |
| N2-T2 | 36.6 | 48.1 | −14.2 | 21.0 | 34.4 | 41.9 a | −15.9 de | 25.7 ab | |
| N1-T0 | 35.3 | 47.9 | −13.9 | 20.1 | 32.4 | 43.0 ad | −16.5 bd | 25.2 bc | |
| N1-T1 | 36.9 | 47.6 | −14.5 | 20.6 | 32.4 | 43.8 ab | −17.3 ce | 26.5 ab | |
| N1-T2 | 37.2 | 47.7 | −14.2 | 20.6 | 31.7 | 42.3 d | −16.3 a | 26.4 c | |
| N0-T0 | 35.6 | 48.0 | −14.5 | 21.2 | 30.2 | 44.2 cd | −17.8 ab | 26.9 b | |
| N0-T1 | 34.7 | 44.3 | −15.6 | 22.5 | 30.5 | 41.4 bd | −15.0 ac | 23.2 ab | |
| N0-T2 | 35.6 | 43.2 | −16.4 | 26.9 | 32.5 | 42.5 bd | −16.7 bd | 25.9 b | |
| Significance | F | * | n.s. | n.s. | n.s. | ** | ** | ** | * |
| T | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | |
| N × T | n.s. | n.s. | n.s. | n.s. | n.s. | * | ** | * | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Visconti, D.; Fiorentino, N.; Cozzolino, E.; Woo, S.L.; Fagnano, M.; Rouphael, Y. Can Trichoderma-Based Biostimulants Optimize N Use Efficiency and Stimulate Growth of Leafy Vegetables in Greenhouse Intensive Cropping Systems? Agronomy 2020, 10, 121. https://doi.org/10.3390/agronomy10010121
Visconti D, Fiorentino N, Cozzolino E, Woo SL, Fagnano M, Rouphael Y. Can Trichoderma-Based Biostimulants Optimize N Use Efficiency and Stimulate Growth of Leafy Vegetables in Greenhouse Intensive Cropping Systems? Agronomy. 2020; 10(1):121. https://doi.org/10.3390/agronomy10010121
Chicago/Turabian StyleVisconti, Donato, Nunzio Fiorentino, Eugenio Cozzolino, Sheridan Lois Woo, Massimo Fagnano, and Youssef Rouphael. 2020. "Can Trichoderma-Based Biostimulants Optimize N Use Efficiency and Stimulate Growth of Leafy Vegetables in Greenhouse Intensive Cropping Systems?" Agronomy 10, no. 1: 121. https://doi.org/10.3390/agronomy10010121
APA StyleVisconti, D., Fiorentino, N., Cozzolino, E., Woo, S. L., Fagnano, M., & Rouphael, Y. (2020). Can Trichoderma-Based Biostimulants Optimize N Use Efficiency and Stimulate Growth of Leafy Vegetables in Greenhouse Intensive Cropping Systems? Agronomy, 10(1), 121. https://doi.org/10.3390/agronomy10010121









