Preparation and Characterization of Eco-Friendly Mg(OH)2/Lignin Hybrid Material and Its Use as a Functional Filler for Poly(Vinyl Chloride)
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of Mg(OH)2/Lignin Hybrid Filler
2.3. Physicochemical and Dispersive–Morphological Characteristics of Inorganic–Organic Hybrid Filler and Used Components
2.3.1. Scanning Electron Microscopy
2.3.2. Particle Size Distribution and Dispersive Parameters
2.3.3. Thermogravimetric Analysis
2.3.4. Porous Structure Characteristics
2.4. Preparation of Mg(OH)2/Lignin-PVC Composites
2.5. Composite Characteristics
2.5.1. Processing Properties
2.5.2. Mechanical Properties
2.5.3. Thermal Properties
2.5.4. Microscopic Observation
3. Results
3.1. Physicochemical and Dispersive–Morphological Characterization of Mg(OH)2/Lignin Hybrid Filler
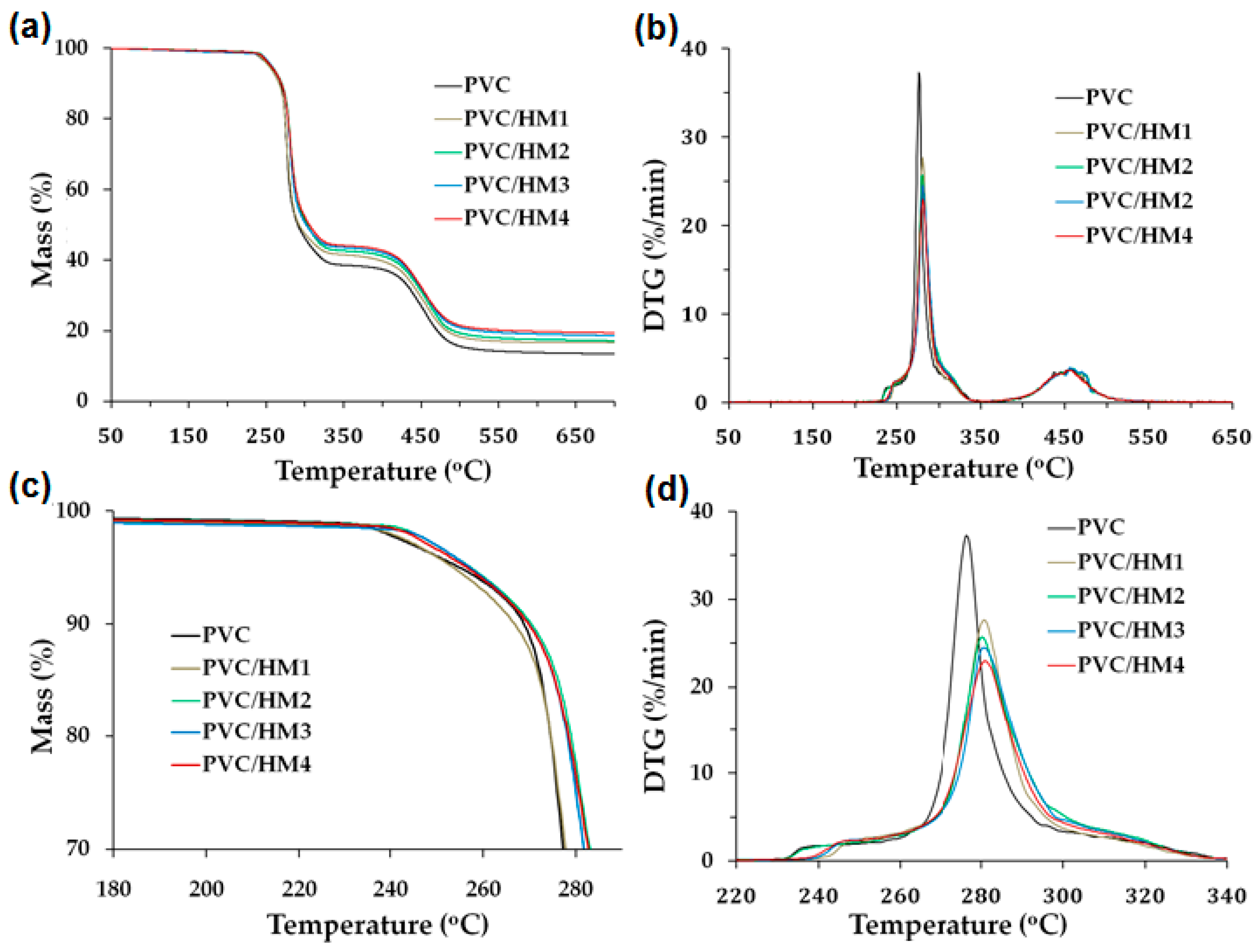
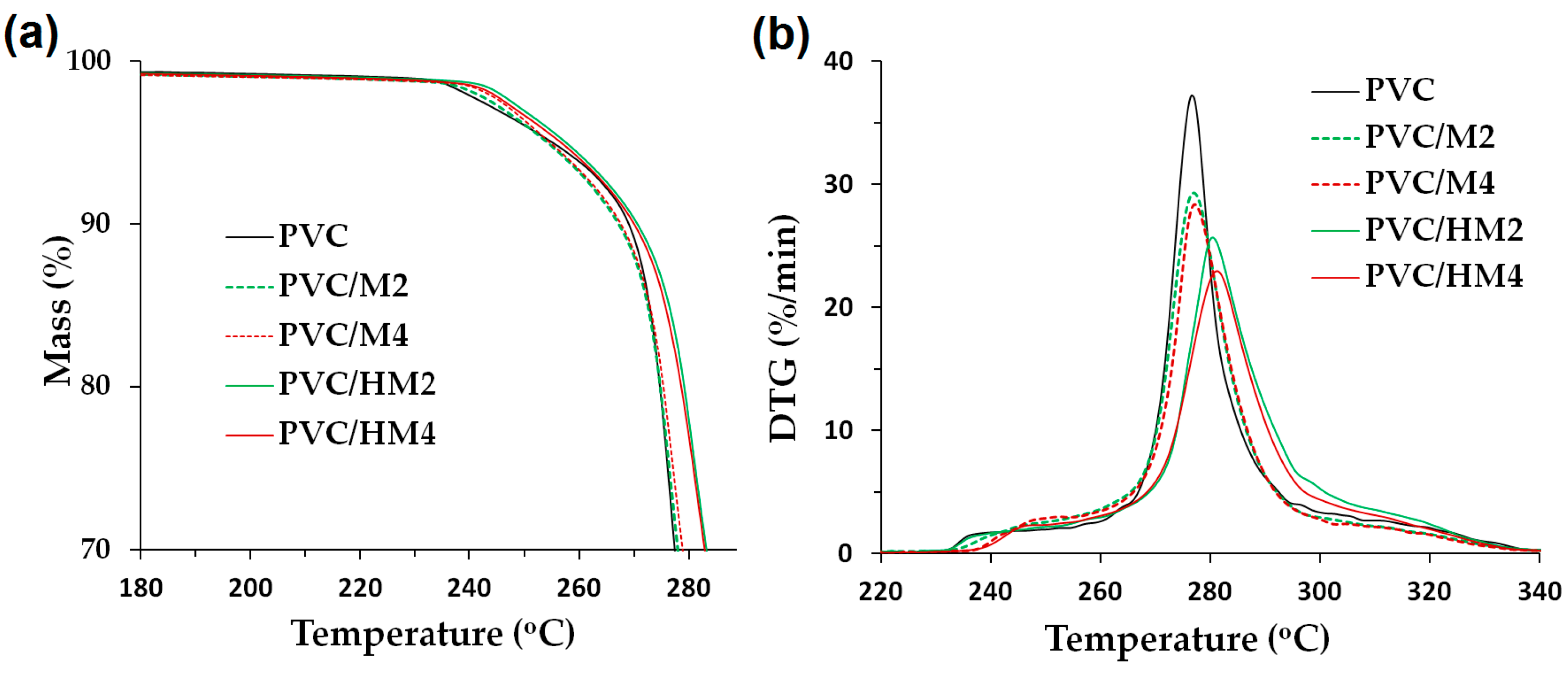
3.2. Magnesium Hydroxide/Lignin-PVC Composite Characterization
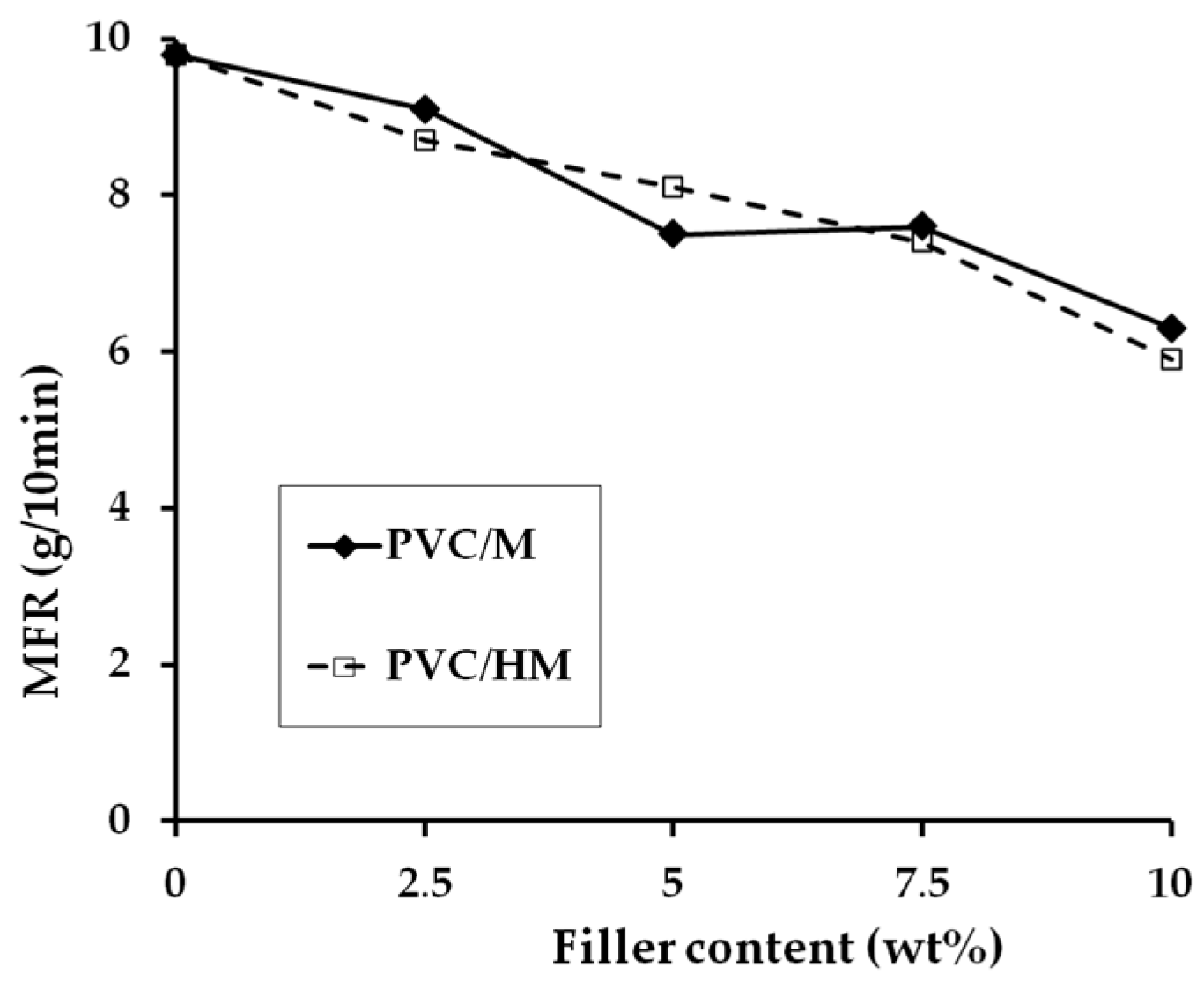
Processing Properties
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Thakur, V.K.; Thakur, M.K.; Raghavan, P.; Kessler, M.R. Progress in green polymer composites from lignin for multifunctional applications: A review. ACS Sustain. Chem. Eng. 2014, 2, 1072–1092. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K.; Gupta, R.K. Review: Raw natural fiber-based polymer composites. Int. J. Polym. Anal. Charact. 2014, 19, 256–271. [Google Scholar] [CrossRef]
- Sterzyński, T.; Tomaszewska, J.; Andrzejewski, J.; Skórczewska, K. Evaluation of glass transition temperature of PVC/POSS nanocomposites. Compos. Sci. Technol. 2015, 117, 398–403. [Google Scholar] [CrossRef]
- Klapiszewski, Ł.; Pawlak, F.; Tomaszewska, J.; Jesionowski, T. Preparation and characterization of novel PVC/silica-lignin composites. Polymers 2015, 7, 1767–1788. [Google Scholar] [CrossRef]
- Bula, K.; Klapiszewski, Ł.; Jesionowski, T. A novel functional silica/lignin hybrid material as a potential bio-based polypropylene filler. Polym. Compos. 2015, 36, 913–922. [Google Scholar] [CrossRef]
- Grząbka-Zasadzińska, A.; Klapiszewski, Ł.; Bula, K.; Jesionowski, T.; Borysiak, S. Supermolecular structure and nucleation ability of polylactide-based composites with silica/lignin hybrid fillers. J. Therm. Anal. Calorim. 2016, 126, 263–275. [Google Scholar] [CrossRef]
- Qiu, L.; Xie, R.; Ding, P.; Qu, B. Preparation and characterization of Mg(OH)2 nanoparticles and flame-retardant property of its nanocomposites with EVA. Compos. Struct. 2003, 62, 391–395. [Google Scholar] [CrossRef]
- Chen, X.; Yu, J.; Guo, S.; Lu, S.; Luo, Z.; He, M. Surface modification of magnesium hydroxide and its application in flame retardant polypropylene composites. J. Mater. Sci. 2009, 44, 1324–1332. [Google Scholar] [CrossRef]
- Gul, R.; Islam, A.; Yasin, T.; Mir, S. Flame-retardant synergism of sepiolite and magnesium hydroxide in a linear low-density polyethylene composite. J. Appl. Polym. Sci. 2011, 121, 2772–2777. [Google Scholar] [CrossRef]
- Suihkonen, R.; Nevalainen, K.; Orell, O.; Honkanen, M.; Tang, L.; Zhang, H.; Zhang, Z.; Vuorinen, J. Performance of epoxy filled with nano- and micro-sized magnesium hydroxide. J. Mater. Sci. 2012, 47, 1480–1488. [Google Scholar] [CrossRef]
- Wang, Q. Preparation and surface modification of Mg(OH)2/siloxane nanocomposite flame retardant. J. Polym. Eng. 2015, 35, 113–117. [Google Scholar] [CrossRef]
- Ma, H.; Chen, Z.; Mao, Z. Controlled growth of magnesium hydroxide crystals and its effect on the high-temperature properties of cotton/magnesium hydroxide composites. Vacuum 2013, 95, 1–5. [Google Scholar] [CrossRef]
- Wypych, G. PVC Formulary, 2nd ed.; ChemTec Publishing: Ontario, ON, Canada, 2015. [Google Scholar]
- Schiller, M. PVC Additives: Performance, Chemistry, Developments, and Sustainability; Carl Hanser Verlag GmbH & Co.: München, Germany, 2015. [Google Scholar]
- Saad, A.L.G.; Sayed, W.M.; Ahmed, M.G.M.; Hassan, A.M. Preparation and properties of some filled poly(vinyl chloride) compositions. J. Appl. Polym. Sci. 1999, 73, 2657–2670. [Google Scholar] [CrossRef]
- Moulay, S. Chemical modification of poly(vinyl chloride)—Still on the run. Prog. Polym. Sci. 2010, 35, 303–331. [Google Scholar] [CrossRef]
- Innes, J.D. Interaction of Smoke Suppressants with Other FR Polymer System Components. In Proceedings of the Addcon ‘96 Conference, Brussels, Belgium, 21–22 May 1996. [Google Scholar]
- Wilkes, C.E.; Summers, J.W.; Daniels, C.A.; Berard, M.T. PVC Handbook; Carl Hanser Verlag: München, Germany, 2005. [Google Scholar]
- Weil, E.D.; Levchik, S.; Moy, P. Flame and smoke retardants in vinyl chloride polymers—Commercial usage and current developments. J. Fire Sci. 2006, 24, 211–236. [Google Scholar] [CrossRef]
- Fedorov, A.; Chekryshkin, Y.; Gorbunov, A. Studies of recycling of poly(vinyl chloride) in molten Na, Ca ‖ NO3, OH systems. Int. Sch. Res. Not. 2012, 768134, 1–6. [Google Scholar] [CrossRef]
- Fomin, D.L.; Deberdeev, R.Y. Effect of aluminum and magnesium hydroxides on the properties of plasticised polyvinyl chloride. Int. Polym. Sci. Technol. 2014, 41, 45–48. [Google Scholar]
- Folarin, O.M.; Sadiku, E.R. Thermal stabilizers for poly(vinyl chloride): A review. Int. J. Phys. Sci. 2011, 18, 4323–4330. [Google Scholar]
- Taghizadeh, M.T.; Fakhimi, F. Kinetics study of degradation and stabilizing effect of organic thermal stabilizers (EDTA, 1,2-propanediol, benzoic acid and phenol) for rigid poly(vinyl chloride). Iran. Polym. J. 2005, 14, 685–692. [Google Scholar]
- Starnes, W.H., Jr. Structural and mechanistic aspects of the thermal degradation of poly(vinyl chloride). Prog. Polym. Sci. 2002, 27, 2133–2170. [Google Scholar] [CrossRef]
- Liu, P.; Zhao, M.; Guo, J. Thermal stabilizers of poly(vinyl chloride)/calcium carbonate (PVC/CaCO3) composites. J. Macromol. Sci. B Phys. 2006, 45, 1135–1140. [Google Scholar] [CrossRef]
- Xue, X.; Zhang, H.; Zhang, S. Preparation of MgAl LDHs intercalated with amines and effect on thermal behavior for poly(vinyl chloride). Adv. Mater. Phys. Chem. 2014, 4, 258–266. [Google Scholar] [CrossRef]
- Yan, H.B.; Li, M.; Liu, Q.F. Hydrotalcite/Ca-Zn composite heat stabilizer for PVC heat stability. Polym. Mater. Sci. Eng. 2009, 25, 75–77. [Google Scholar]
- Gregorova, A.; Cibulkova, Z.; Kosikova, B.; Simon, P. Stabilization effect of lignin in polypropylene and recycled polypropylene. Polym. Degrad. Stab. 2005, 89, 553–558. [Google Scholar] [CrossRef]
- Kadla, J.F.; Kubo, S. Miscibility and hydrogen bonding in blends of poly(ethylene oxide) and kraft lignin. Macromolecules 2003, 36, 7803–8005. [Google Scholar] [CrossRef]
- Brebu, M.; Vasile, C. Thermal degradation of lignin—Review. Cellul. Chem. Technol. 2010, 44, 353–363. [Google Scholar]
- Costes, L.; Laotid, F.; Aguedo, M.; Richel, A.; Brohez, S.; Delvosalle, C.; Dubois, P. Phosphorous and nitrogen derivatization as efficient route for improvement of lignin flame retardant acrion in PLA. Eur. Polym. J. 2016, 84, 652–667. [Google Scholar] [CrossRef]
- Maldhure, A.V.; Ekhe, J.D.; Deenadayalan, E. Mechanical properties of polypropylene blended with esterified and alkylated lignin. J. Appl. Polym. Sci. 2012, 125, 1701–1712. [Google Scholar] [CrossRef]
- Bertini, F.; Canetti, M.; Cacciamani, A.; Elegir, G.; Orlandi, M.; Zoia, L. Effect of lingo-derivatives on thermal properties and degradation behavior of poly(3-hydroxybutyrate)-based biocomposites. Polym. Degrad. Stab. 2012, 97, 1979–1987. [Google Scholar] [CrossRef]
- Sadeghifar, H.; Cui, C.; Argyropoulos, D.S. Toward thermoplastic lignin polymers. Part 1. Selective masking of phenolic hydroxyl groups in kraft lignins via methylation and oxypropylation chemistries. Ind. Eng. Chem. Res. 2012, 51, 16713–16720. [Google Scholar] [CrossRef]
- Hu, L.H.; Pan, H.; Zhou, Y.H.; Zhang, M. Methods to improve lignin’s reactivity as a phenol substitute and as replacement for other phenolic compounds: A brief review. Bioresources 2011, 6, 3515–3525. [Google Scholar]
- Saito, T.; Brown, R.H.; Hunt, M.A.; Pickel, D.L.; Pickel, J.M.; Messman, J.M.; Baker, F.S.; Keller, M.; Naskar, A.K. Turning renewable resources into recyclable polymer: Development of lignin-based thermoplastic. Green Chem. 2012, 14, 3295–3303. [Google Scholar] [CrossRef]
- Kajiwara, Y.; Chujo, Y. Microwave-enhanced hybridizations of biopolymers with silica: Effective method for rapid preparation and homogeneous dispersion. Polym. Bull. 2011, 66, 1039–1050. [Google Scholar] [CrossRef]
- Klapiszewski, Ł.; Nowacka, M.; Milczarek, G.; Jesionowski, T. Physicochemical and electrokinetic properties of silica/lignin biocomposites. Carbohydr. Polym. 2013, 94, 345–355. [Google Scholar] [CrossRef] [PubMed]
- Klapiszewski, Ł.; Bula, K.; Sobczak, M.; Jesionowski, T. Influence of processing conditions on the thermal stability and mechanical properties of PP/silica-lignin composites. Int. J. Polym. Sci. 2016, 1627258, 1–9. [Google Scholar] [CrossRef]
- Tomaszewska, J.; Klapiszewski, Ł.; Skórczewska, K.; Szalaty, T.J.; Jesionowski, T. Advanced organic-inorganic hybrid fillers as functional additives for poly(vinyl chloride). Polimery 2017, 62, 19–26. [Google Scholar] [CrossRef]
- Pilarska, A.; Wysokowski, M.; Markiewicz, E.; Jesionowski, T. Synthesis of magnesium hydroxide and its calcinates by a precipitation method with the use of magnesium sulfate and poly(ethylene glycols). Powder Technol. 2013, 235, 148–157. [Google Scholar] [CrossRef]
- Noh, J.; Kang, I.; Choi, J.; Fatima, H.; Yoo, P.J.; Oh, K.W.; Park, J. Surface modification of magnesium hydroxide nanoparticles with hexylphosphoric acid to improve thermal stabilities of polyethylene composites. Polym. Bull. 2016, 73, 2855–2866. [Google Scholar] [CrossRef]
- Pilarska, A.; Bula, K.; Myszka, K.; Rozmanowski, T.; Szwarc-Rzepka, K.; Pilarski, K.; Chrzanowski, Ł.; Czaczyk, K.; Jesionowski, T. Functional polypropylene composites filled with ultra-fine magnesium hydroxide. Open Chem. 2015, 13, 161–171. [Google Scholar] [CrossRef]
- Rodríguez-Mirasol, J.; Cordero, T.; Rodriguez, J. CO2-reactivity of eucalyptus kraft lignin chars. Carbon 1993, 31, 53–61. [Google Scholar] [CrossRef]
- Kijima, M.; Hirukawa, T.; Hanawa, F.; Hata, T. Thermal conversion of alkaline lignin and its structured derivatives to porous carbonized materials. Bioresour. Technol. 2011, 102, 6279–6285. [Google Scholar] [CrossRef] [PubMed]
- Blanco, I.; Abate, L.; Bottino, F.A.; Cicala, G.; Latteri, A. Dumbbell-shaped polyhedral oligomeric silsesquioxanes/polystyrene nanocomposites: The influence of the bridge rigidity on the resistance to thermal degradation. J. Compos. Mater. 2015, 49, 2509–2517. [Google Scholar] [CrossRef]
- Tomaszewska, J.; Sterzyński, T.; Piszczek, K. Rigid poly(vinyl chloride) (PVC) gelation in the Brabender measuring mixer. III. Transformation in the Torque Maximum. J. Appl. Polym. Sci. 2007, 106, 3158–3164. [Google Scholar]
- Shubbar, S.D.A. The effect of shear on the properties of rigid PVC. J. Eng. 2011, 17, 1567–1575. [Google Scholar]
- Liu, F.; Xu, K.; Chen, M.; Cao, D. The roles of polyacrylate in poly(vinyl chloride)-lignin composites. Polym. Compos. 2011, 32, 1399–1407. [Google Scholar] [CrossRef]
- Liu, F.; Xu, K.; Chen, M.; Cao, D. The rheological and mechanical properties of PVC-lignin blends. Int. Polym. Process. 2012, 27, 121–127. [Google Scholar] [CrossRef]
- Gozdecki, C.; Zajchowski, S.; Kociszewski, M.; Wilczyński, A.; Mirowski, J. Effect of wood particle size on mechanical properties of industrialwood particle-polyethylene composites. Polimery 2011, 56, 375–380. [Google Scholar]
- Kociszewski, M.; Gozdecki, C.; Wilczyński, A.; Zajchowski, S.; Mirowski, J. Effect of industrial wood particle size on mechanical properties of wood-polyvinyl chloride composites. Eur. J. Wood Wood Prod. 2012, 70, 113–118. [Google Scholar] [CrossRef]
- Ambrożewicz, D.; Jakubowska, P.; Kloziński, A.; Nowacka, M.; Dudziec, B.; Andrzejewska, E.; Marciniec, B.; Jesionowski, T. A novel functional MgO∙SiO2/polyhedral oligomeric silsesquioxane hybrids as an active filler of polypropylene. Pol. J. Chem. Technol. 2013, 15, 42–48. [Google Scholar] [CrossRef]
- Elakesh, E.O.; Hull, T.R.; Price, D.; Carty, P. Thermal decomposition of chlorinated poly(vinyl chloride) (CPVC). J. Vin. Add. Technol. 2003, 9, 116–126. [Google Scholar] [CrossRef]
- Munro, S. New developments in flame retarded PVC wire and cable formulations. Proceedings of Flame Retardant Polymerics: Electrical and Electronic Applications, Newport, RI, USA, 4–7 October 1998. [Google Scholar]
- Liu, J.; Chen, G.; Yang, J. Preparation and characterization of poly(vinyl chloride)/layered double hydroxide nanocomposites with enhanced thermal stability. Polymer 2008, 49, 3923–3927. [Google Scholar] [CrossRef]
- Van der Ven, L.; van Gemert, M.L.M.; Batenburg, L.F.; Keern, J.J.; Gielgens, L.H.; Koster, T.P.M. On the action of hydrotalcite-like clay materials as stabilizers in poly(vinyl chloride). Appl. Clay Sci. 2000, 17, 25–34. [Google Scholar] [CrossRef]
- Wang, X.D.; Zhang, Q. Effect of hydrotalcite on the thermal stability, mechanical properties, rheology and flame retardance of poly(vinyl chloride). Polym. Int. 2004, 53, 698–707. [Google Scholar] [CrossRef]
- Lin, Y.J.; Li, D.; Evans, D.G.; Duan, X. Modulating effect of Mg-Al-CO3 layered double hydroxides on the thermal stability of PVC resin. Polym. Degrad. Stab. 2005, 88, 286–293. [Google Scholar] [CrossRef]
- Shen, Y.; Yu, S.; Ge, S.; Chen, X.; Ge, X.; Chen, M. Hydrothermal carbonization of medical wastes and lignocellulosic biomass for solid fuel production from lab-scale to pilot-scale. Energy 2017, 118, 312–323. [Google Scholar] [CrossRef]
- Czegeny, Z.; Jakab, E.; Bozi, J.; Blazso, M. Pyrolysis of wood–PVC mixtures. Formation of chloromethane from lignocellulosic materials in the presence of PVC. J. Anal. Appl. Pyrol. 2015, 113, 123–132. [Google Scholar] [CrossRef]
- Walker, J.K.; Luke, E.D.; Chen, T.; Isarov, A. Synergies of Metal Hydroxides and Metal Molybdates in Low-Smoke Flexible PVC. In Proceedings of the 57th IWCS Conference, Providence, RI, USA, 9–12 November 2008. [Google Scholar]
- Blanco, I.; Cicala, G.; Latter, A.; Saccullo, G.; El-Sabbagh, A.M.M.; Ziegmann, G. Thermal characterization of a series of lignin-based polypropylene blends. J. Therm. Anal. Calorim. 2017, 127, 147–153. [Google Scholar] [CrossRef]



| Sample Name | Dispersive Properties | ||||
|---|---|---|---|---|---|
| Particle Size Distribution from Zetasizer Nano ZS (nm) | Particle Diameter from Mastersizer 2000 (μm) | ||||
| d(0.1) * | d(0.5) ** | d(0.9) *** | D(4.3) **** | ||
| Magnesium hydroxide | 91–513 | 1.0 | 2.6 | 4.8 | 2.4 |
| Kraft lignin | 1110–5560 | 6.9 | 20.3 | 38.9 | 19.8 |
| Mg(OH)2/lignin hybrid material | 342–615; 955–5560 | 2.3 | 5.1 | 9.7 | 4.9 |
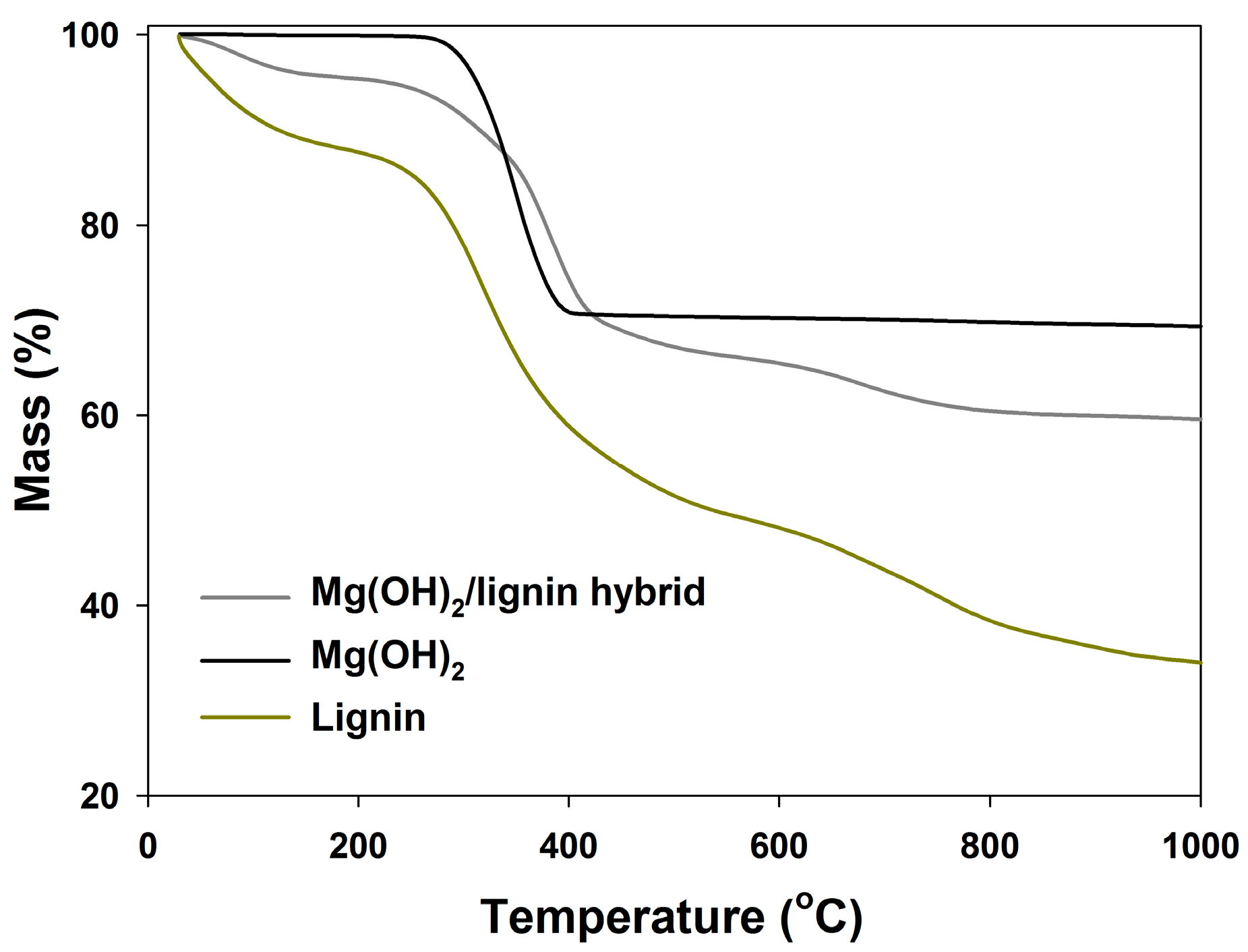
| Sample Name | TGA Results | |
|---|---|---|
| T5 * (°C) | T50 ** (°C) | |
| Mg(OH)2/lignin hybrid | 156 | 378 |
| Magnesium hydroxide | 287 | 343 |
| Lignin | 46 | 344 |
| Sample Name | Parameters of Porous Structure | ||
|---|---|---|---|
| BET Surface Area (m2/g) | Total Volume of Pores (cm3/g) | Mean Size of Pores (nm) | |
| Magnesium hydroxide | 108 | 0.10 | 2.6 |
| Kraft lignin | 1 | 0.01 | 11.4 |
| Mg(OH)2/lignin hybrid material | 73 | 0.06 | 4.2 |
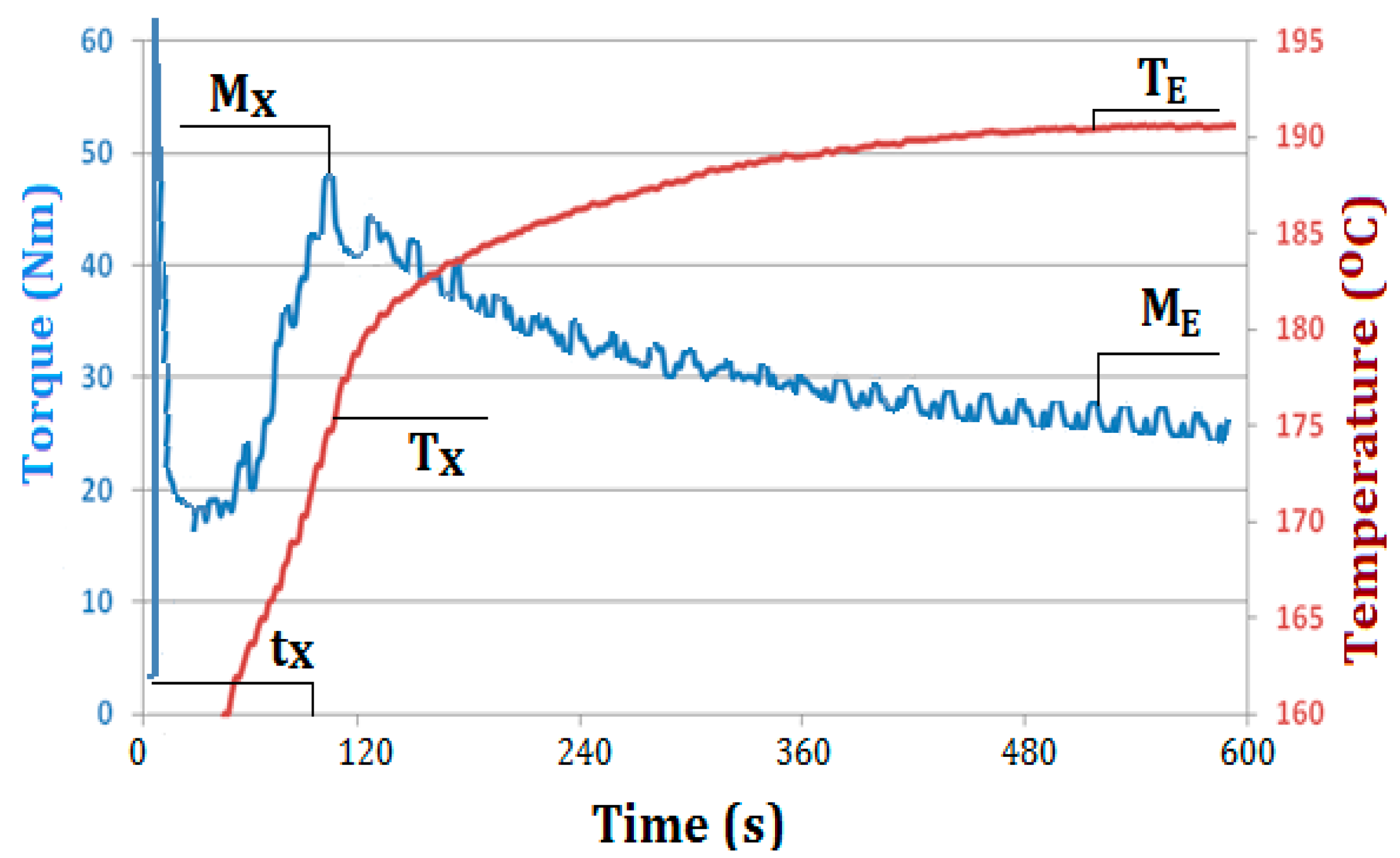
| Sample Name | MX (Nm) | TX (°C) | tX (s) | ME (Nm) | TE (°C) |
|---|---|---|---|---|---|
| PVC | 45 | 178 | 160 | 26 | 190 |
| PVC/M1 | 45 | 175 | 135 | 25 | 191 |
| PVC/M2 | 51 | 178 | 130 | 26 | 191 |
| PVC/M3 | 51 | 178 | 130 | 27 | 191 |
| PVC/M4 | 53 | 177 | 120 | 26 | 192 |
| PVC/HM1 | 46 | 175 | 105 | 27 | 191 |
| PVC/HM2 | 48 | 174 | 95 | 27 | 191 |
| PVC/HM3 | 51 | 174 | 75 | 28 | 191 |
| PVC/HM4 | 50 | 176 | 70 | 26 | 191 |
| Sample Name | Et (MPa) | σY (MPa) | σM (MPa) | εB (%) |
|---|---|---|---|---|
| PVC | 1590 ± 18.6 | 53.7 ± 0.9 | 53.7 ± 0.9 | 41.3 ± 11.7 |
| PVC/M1 | 1620 ± 27.8 | 48.3 ± 2.5 | 48.3 ± 2.5 | 19.9 ± 11.9 |
| PVC/M2 | 1770 ± 64.4 | 51.5 ± 1.5 | 51.5 ± 1.5 | 7.5 ± 2.9 |
| PVC/M3 | 1780 ± 69.8 | 53.3 ±.2.1 | 53.3 ±.2.1 | 10.2 ± 0.9 |
| PVC/M4 | 1810 ± 73.1 | 52.1 ±.2.2 | 52.1 ±.2.2 | 5.2 ± 0.8 |
| PVC/HM1 | 1760 ± 38.2 | 50.4 ± 2.7 | 50.4 ± 2.7 | 5.3 ± 1.2 |
| PVC/HM2 | 1750 ± 87.1 | 46.5 ± 3.2 | 46.5 ± 3.2 | 4.5 ± 0.9 |
| PVC/HM3 | 1740 ± 75.4 | 43.0 ±.2.1 | 43.0 ± 2.1 | 3.6 ± 0.9 |
| PVC/HM4 | 1780 ± 67.7 | 42.5 ±.3.8 | 42.5 ± 3.8 | 4.7 ± 0.8 |
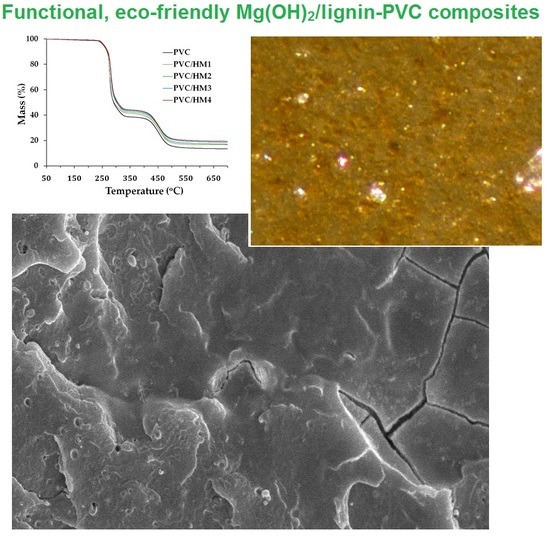
| Sample Name | TGA Results | Thermal Stability (min) | VST (°C) | LOI (%) | ||
|---|---|---|---|---|---|---|
| T5 * (°C) | T50 ** (°C) | TDTG (°C) | ||||
| PVC | 255.5 | 292.0 | 276.2 | 22 | 81.4 | 45.1 |
| PVC/M1 | 274.3 | 293.0 | 274.3 | 21 | 80.1 | 47.8 |
| PVC/M2 | 277.0 | 291.1 | 277.0 | 30 | 82.4 | 52.2 |
| PVC/M3 | 279.6 | 301.8 | 278.6 | 36 | 80.7 | 53.0 |
| PVC/M4 | 277.2 | 300.4 | 276.6 | 39 | 79.3 | 55.2 |
| PVC/HM1 | 255.1 | 297.4 | 280.9 | 24 | 86.2 | 45.8 |
| PVC/HM2 | 256.3 | 299.4 | 280.8 | 35 | 88.9 | 47.2 |
| PVC/HM3 | 258.1 | 303.8 | 281.5 | 51 | 89.1 | 48.4 |
| PVC/HM4 | 260.1 | 306.2 | 284.4 | 63 | 90.0 | 49.6 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klapiszewski, Ł.; Tomaszewska, J.; Skórczewska, K.; Jesionowski, T. Preparation and Characterization of Eco-Friendly Mg(OH)2/Lignin Hybrid Material and Its Use as a Functional Filler for Poly(Vinyl Chloride). Polymers 2017, 9, 258. https://doi.org/10.3390/polym9070258
Klapiszewski Ł, Tomaszewska J, Skórczewska K, Jesionowski T. Preparation and Characterization of Eco-Friendly Mg(OH)2/Lignin Hybrid Material and Its Use as a Functional Filler for Poly(Vinyl Chloride). Polymers. 2017; 9(7):258. https://doi.org/10.3390/polym9070258
Chicago/Turabian StyleKlapiszewski, Łukasz, Jolanta Tomaszewska, Katarzyna Skórczewska, and Teofil Jesionowski. 2017. "Preparation and Characterization of Eco-Friendly Mg(OH)2/Lignin Hybrid Material and Its Use as a Functional Filler for Poly(Vinyl Chloride)" Polymers 9, no. 7: 258. https://doi.org/10.3390/polym9070258
APA StyleKlapiszewski, Ł., Tomaszewska, J., Skórczewska, K., & Jesionowski, T. (2017). Preparation and Characterization of Eco-Friendly Mg(OH)2/Lignin Hybrid Material and Its Use as a Functional Filler for Poly(Vinyl Chloride). Polymers, 9(7), 258. https://doi.org/10.3390/polym9070258