Performance and Kinetics Study of Self-Repairing Hydroxyl-Terminated Polybutadiene Binders Based on the Diels–Alder Reaction
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Characterization
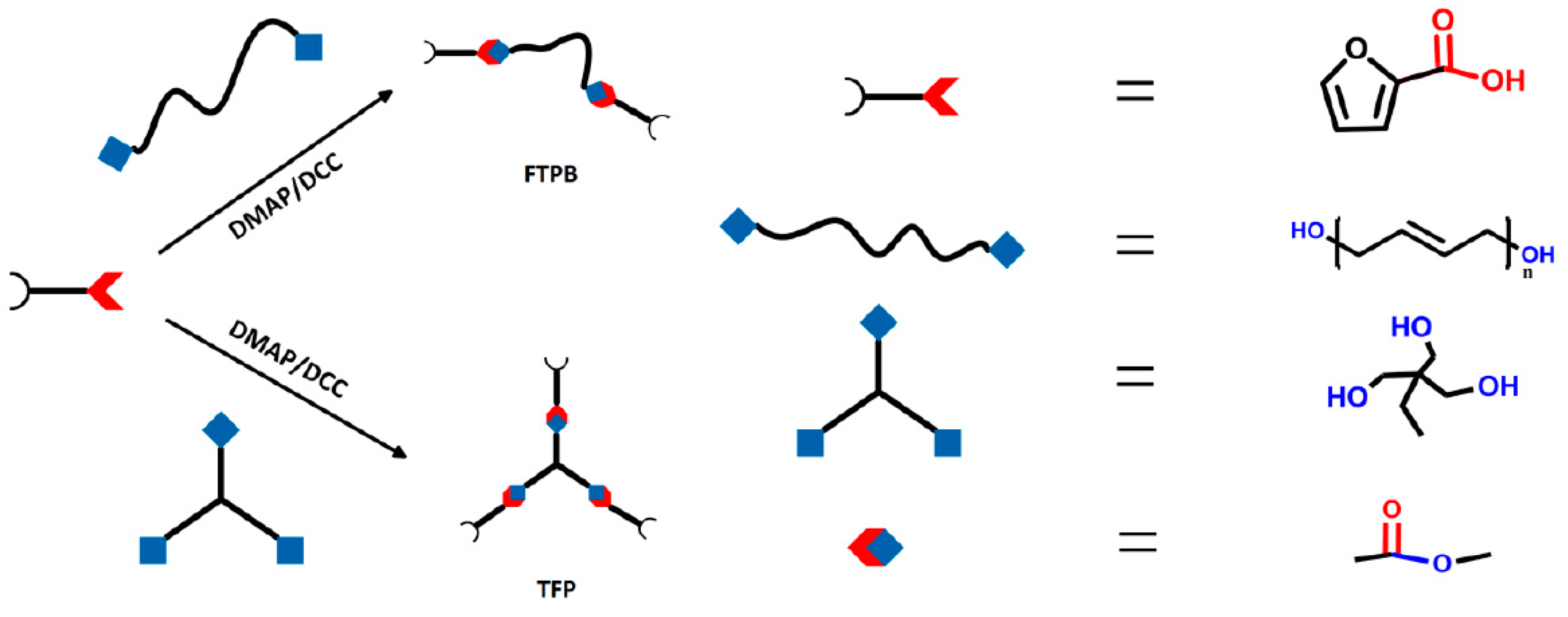
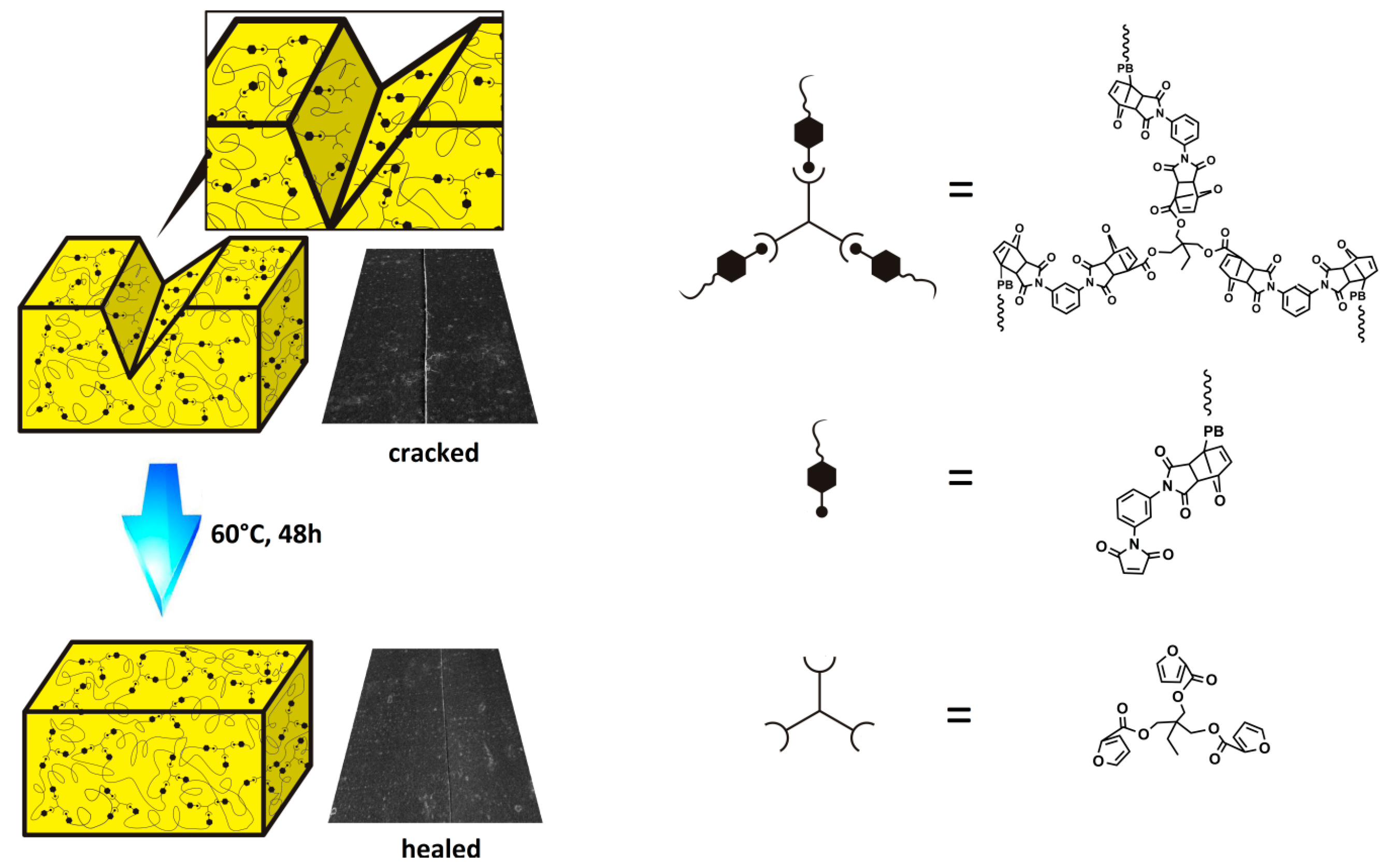
2.3. Synthesis of FTPB
2.4. Synthesis of TFP
2.5. Preparation of Self-Repairing Binder System
3. Results and Discussion
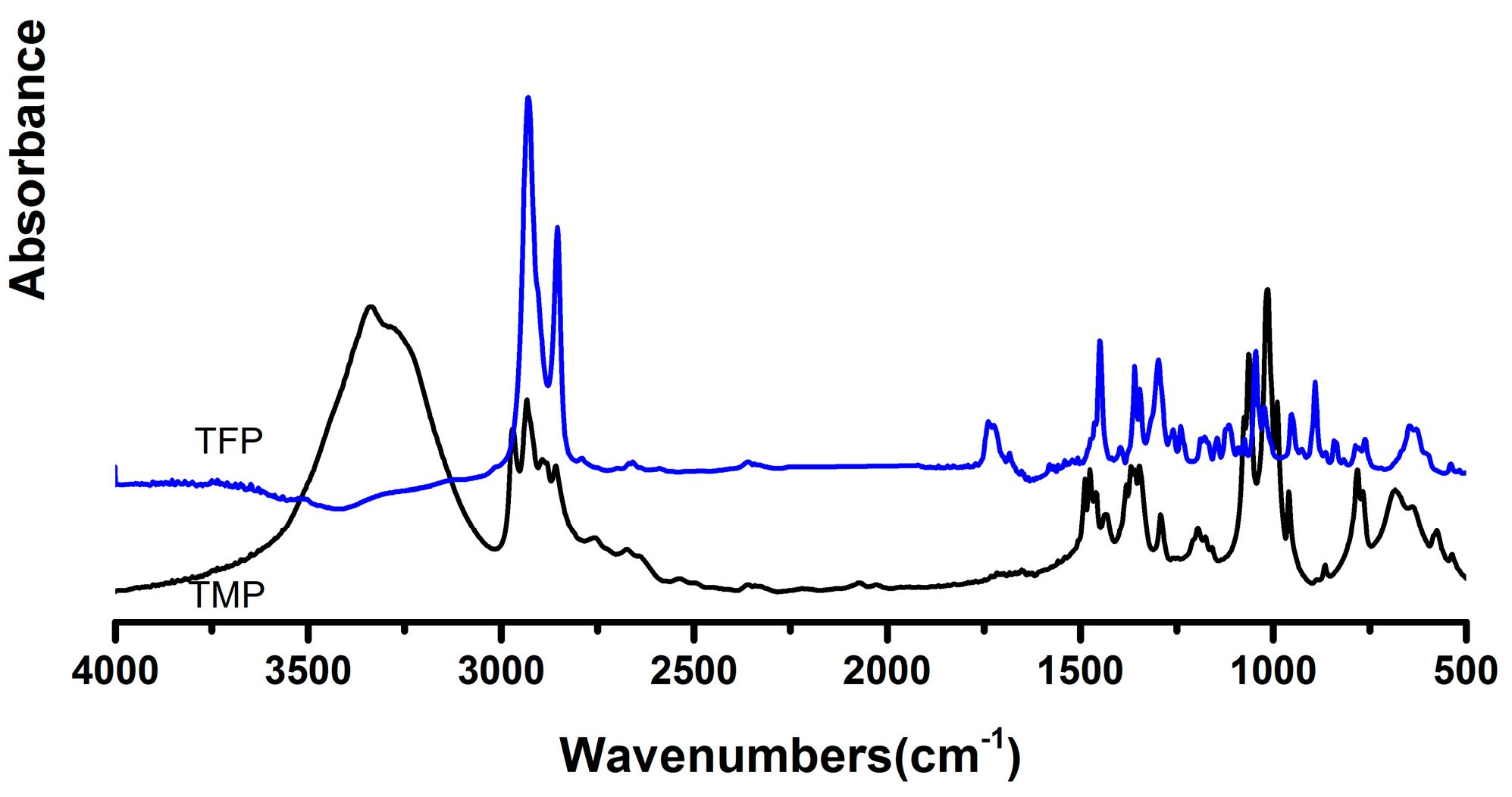
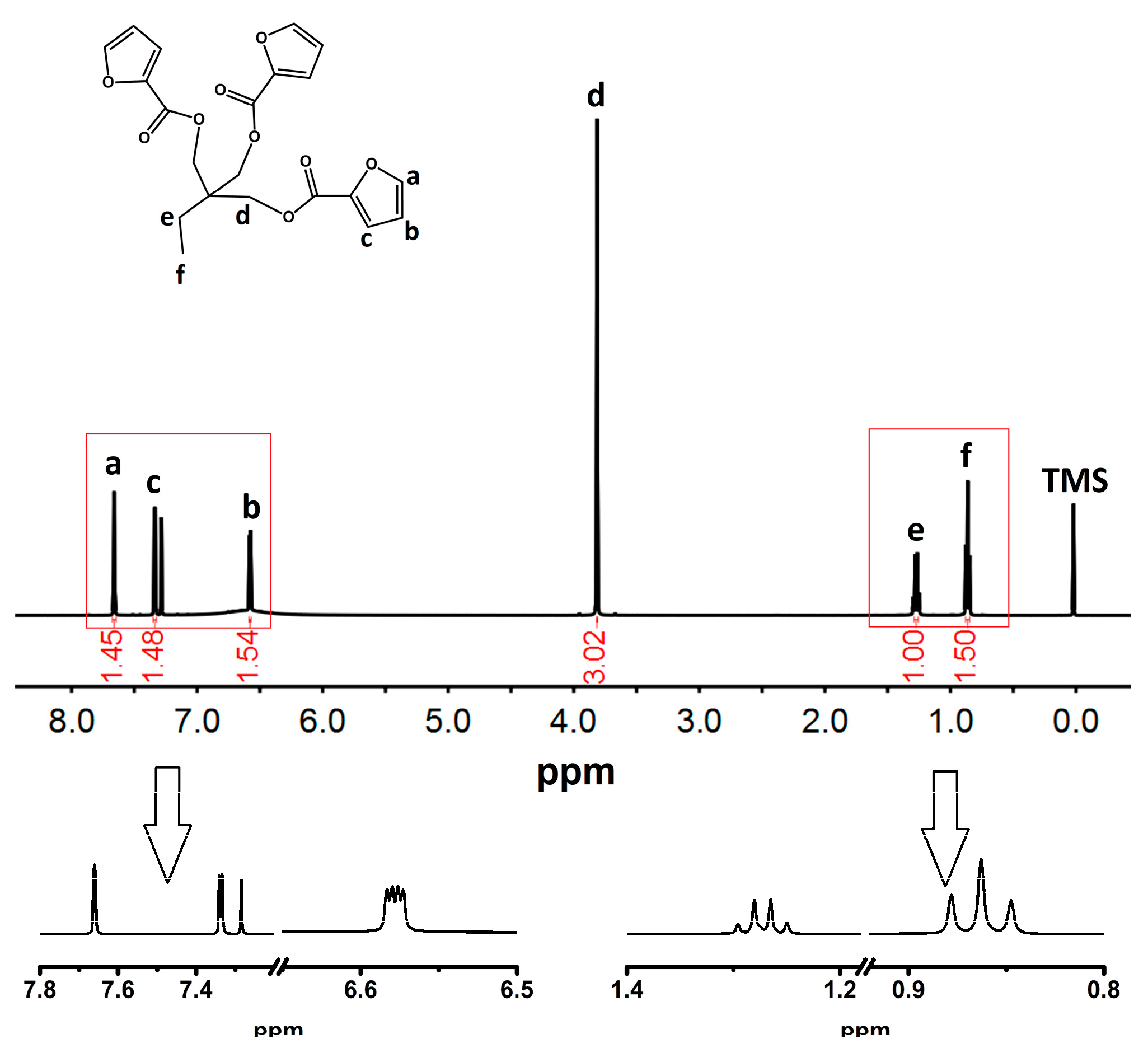
3.1. Structure Characterization of FTPB and TFP
3.2. Mechanial Property of FTPB/PDMI/TFP (FPT) Film
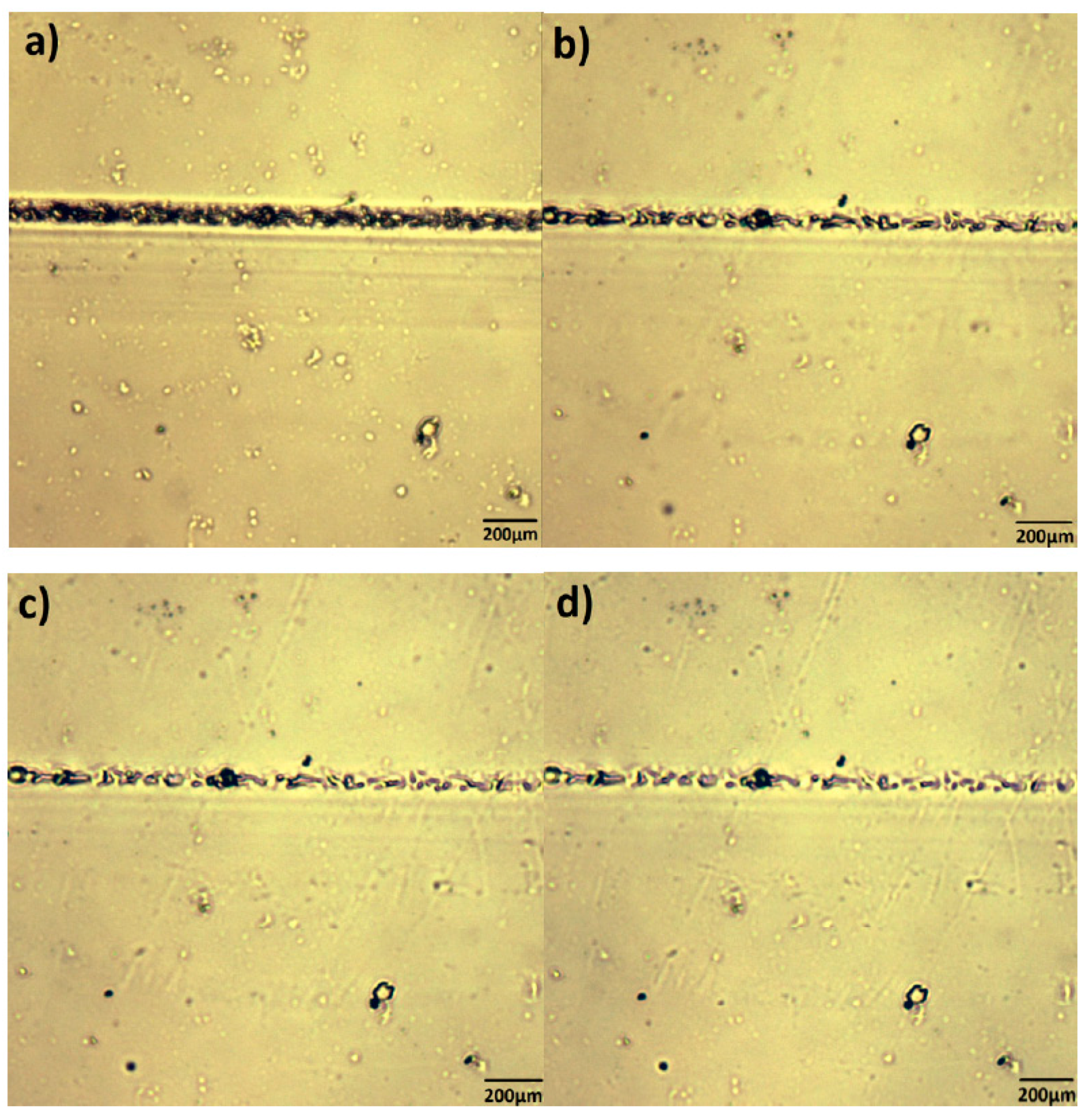
3.3. HSPM on FTPB-PDMI Film Crack Self-Repairing Progress
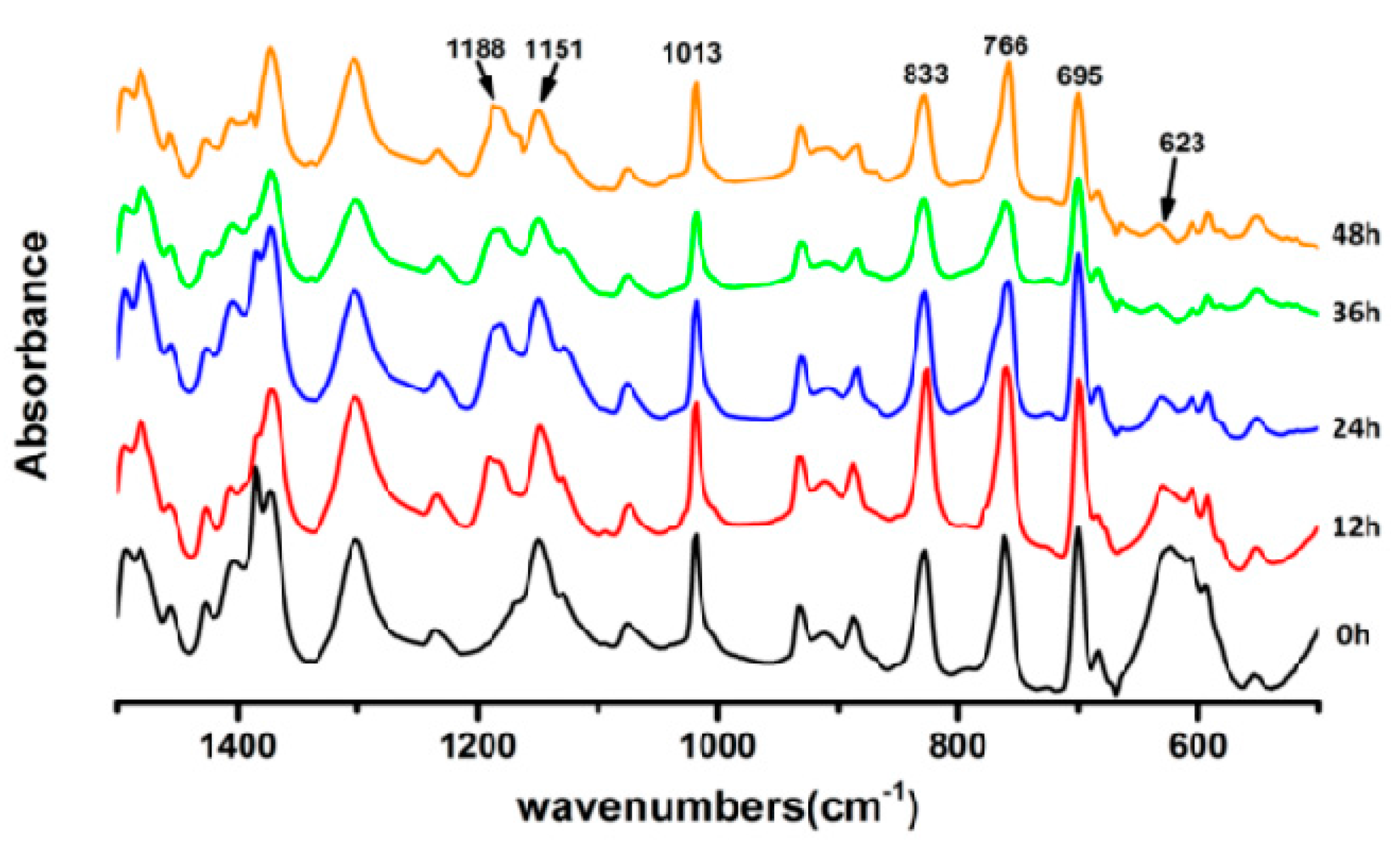
3.4. UV-Vis on FTPB-PDMI Film Crack Self-Repairing Progress
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Trask, R.S.; Williams, H.R.; Bond, I.P. Self-healing polymer composites: mimicking nature to enhance performance. Bioinspir. Biomim. 2007, 2, P1. [Google Scholar] [CrossRef] [PubMed]
- White, S.R.; Sottos, N.R.; Geubelle, P.H.; Moore, J.S.; Kessler, M.R.; Sriram, S.R.; Brown, E.N.; Viswanathan, S. Autonomic healing of polymer composites. Nature 2001, 409, 794–817. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.N.; White, S.R.; Sottos, N.R. Microcapsule induced toughening in a self-healing polymer composite. J. Mater. Sci. 2004, 39, 1703–1710. [Google Scholar] [CrossRef]
- Dry, C. Procedures developed for self-repair of polymer matrix composite materials. Compos. Struct. 1996, 35, 263–269. [Google Scholar] [CrossRef]
- Li, V.C.; Lim, Y.M.; Chan, Y.W. Feasibility study of a passive smart self-healing cementitious composite. Compos. Part B 1998, 29, 819–827. [Google Scholar] [CrossRef]
- Turkenburg, D.H.; Fischer, H.R. Diels–Alder based, thermo-reversible cross-linked epoxies for use in self-healing composites. Polymer 2015, 79, 187–194. [Google Scholar] [CrossRef]
- Lai, J.; Mei, J.; Jia, X.; Li, C.; You, X.; Bao, Z. A stiff and healable polymer based on dynamic-covalent boroxine bonds. Adv. Mater. 2016, 28, 8277–8282. [Google Scholar] [CrossRef] [PubMed]
- García, J.M.; Jones, G.O.; Virwani, K.; McCloskey, B.D.; Boday, D.J.; Huurne, G.M.T.; Horn, H.W.; Coady, D.J.; Bintaleb, A.M.; Alabdulrahman, A.M.S.; et al. Recyclable, strong thermosets and organogels via paraformaldehyde condensation with diamines. Science 2014, 344, 732–735. [Google Scholar] [CrossRef] [PubMed]
- Casuso, P.; Odriozola, I.; Vicente, A.P.; Loinaz, I.; Cabañero, G.; Grande, H.; Dupin, D. Injectable and self-healing dynamic hydrogels based on metal (I)-thiolate/disulfide exchange as biomaterials with tunable mechanical properties. Biomacromolecules 2015, 16, 3552–3561. [Google Scholar] [CrossRef] [PubMed]
- Yoon, J.A.; Kamada, J.; Koynov, K.; Mohin, J.; Nicolaÿ, R.; Zhang, Y.; Balazs, A.C.; Kowalewski, T.; Matyjaszewski, K. Self-healing polymer films based on thiol-disulfide exchange reactions and self-healing kinetics measured using atomic force microscopy. Macromolecules 2011, 45, 142–149. [Google Scholar] [CrossRef]
- Li, C.; Wang, C.; Keplinger, C.; Zuo, J.; Jin, L.; Sun, Y.; Zheng, P.; Cao, Y.; Lissel, F.; Linder, C.; et al. A highly stretchable autonomous self-healing elastomer. Nat. Chem. 2016, 8, 618. [Google Scholar] [CrossRef] [PubMed]
- Ahn, B.K.; Lee, D.W.; Israelachvili, J.N.; Waite, J.H. Surface-initiated self-healing of polymers in aqueous media. Nat. Mater. 2014, 13, 867–872. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Liu, Y.F.; Yao, W.S. Effect of components oncombustion and mechanical properties of HTPB propellantwith high energy. Chin. J. Explos. Propellants 2007, 30, 62–65. [Google Scholar]
- Stewart, S.A.; Backholm, M.; Burke, N.A.D.; Stöver, H.D.H. Cross-linked hydrogels formed through Diels–Alder coupling of furan-and maleimide-modified poly (methyl vinyl ether-alt-maleic acid). Langmuir 2016, 32, 1863–1870. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, L.T.T.; Nguyen, H.T.; Truong, T.T. Thermally mendable material based on a furyl-telechelic semicrystalline polymer and a maleimide crosslinker. J. Polym. Res. 2015, 22, 1–9. [Google Scholar] [CrossRef]
- Dolci, E.; Froidevaux, V.; Michaud, G.; Caillol, S. Thermoresponsive crosslinked isocyanate-free polyurethanes by Diels–Alder polymerization. J. Appl. Polym. Sci. 2017, 134. [Google Scholar] [CrossRef]
- Chen, X.; Dam, M.A.; Ono, K.; Mal, A.; Shen, H.; Nutt, S.R.; Sheran, K.; Wudl, F. A thermally re-mendable cross-linked polymeric material. Science 2002, 295, 1698–1702. [Google Scholar] [CrossRef] [PubMed]
- Sakakura, A.; Kawajiri, K.; Ohkubo, T.; Kosugi, Y.; Ishihara, K. Widely useful dmap-catalyzed esterification under auxiliary base- and solvent-free conditions. J. Am. Chem. Soc. 2007, 129, 14775–14779. [Google Scholar] [CrossRef] [PubMed]
- Paul, R.; Kende, A.S. A Mechanism for the N,N′-dicyclohexylcarbodiimide-caused dehydration of asparagineand maleamic acid derivatives. J. Am. Chem. Soc. 1964, 86, 4162–4166. [Google Scholar] [CrossRef]
- Ball, D. A microscale synthesis of mauve. J. Chem. Educ. 1998, 75, 917. [Google Scholar] [CrossRef]
- Sahu, H.; Mohanty, K. Pseudo-first order reaction kinetics and thermodynamic properties study of neem oil esterification using MgO grafted natural hydroxyapatite. RSC Adv. 2016, 6, 8892–8901. [Google Scholar] [CrossRef]

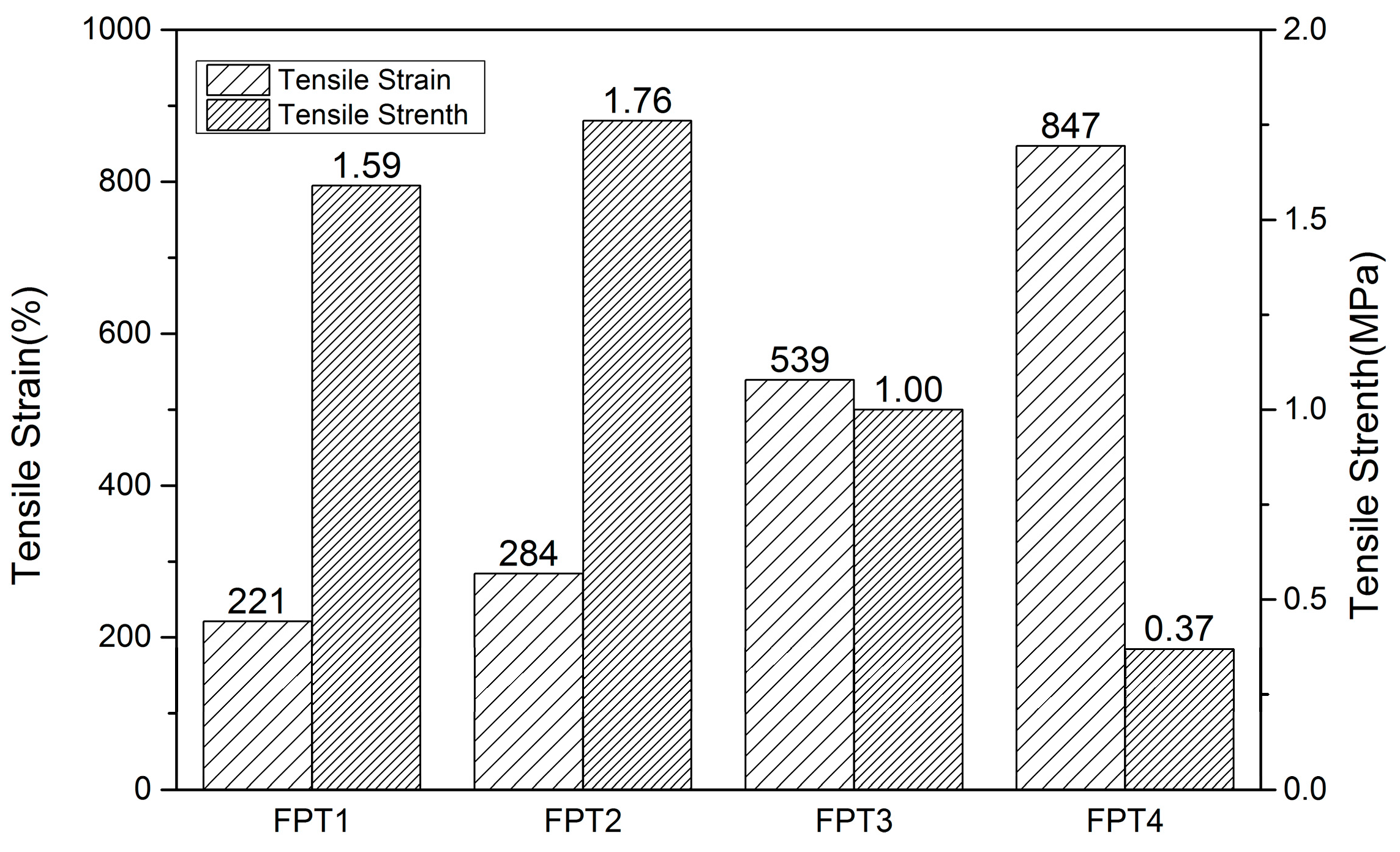
| Name | Tensile Strain (%) | Post-Repair Tensile Strain (%) | Tensile Strain Repair Efficiency (%) | Tensile Strength (MPa) | Post-Repair Tensile Strength (MPa) | Tensile Strength Repair Efficiency (%) |
|---|---|---|---|---|---|---|
| FPT1 | 221 | 203 | 91.8 | 1.59 | 1.32 | 83.0 |
| FPT2 | 284 | 251 | 88.4 | 1.76 | 1.55 | 88.0 |
| FPT3 | 539 | 471 | 87.3 | 0.78 | 1.00 | 78.0 |
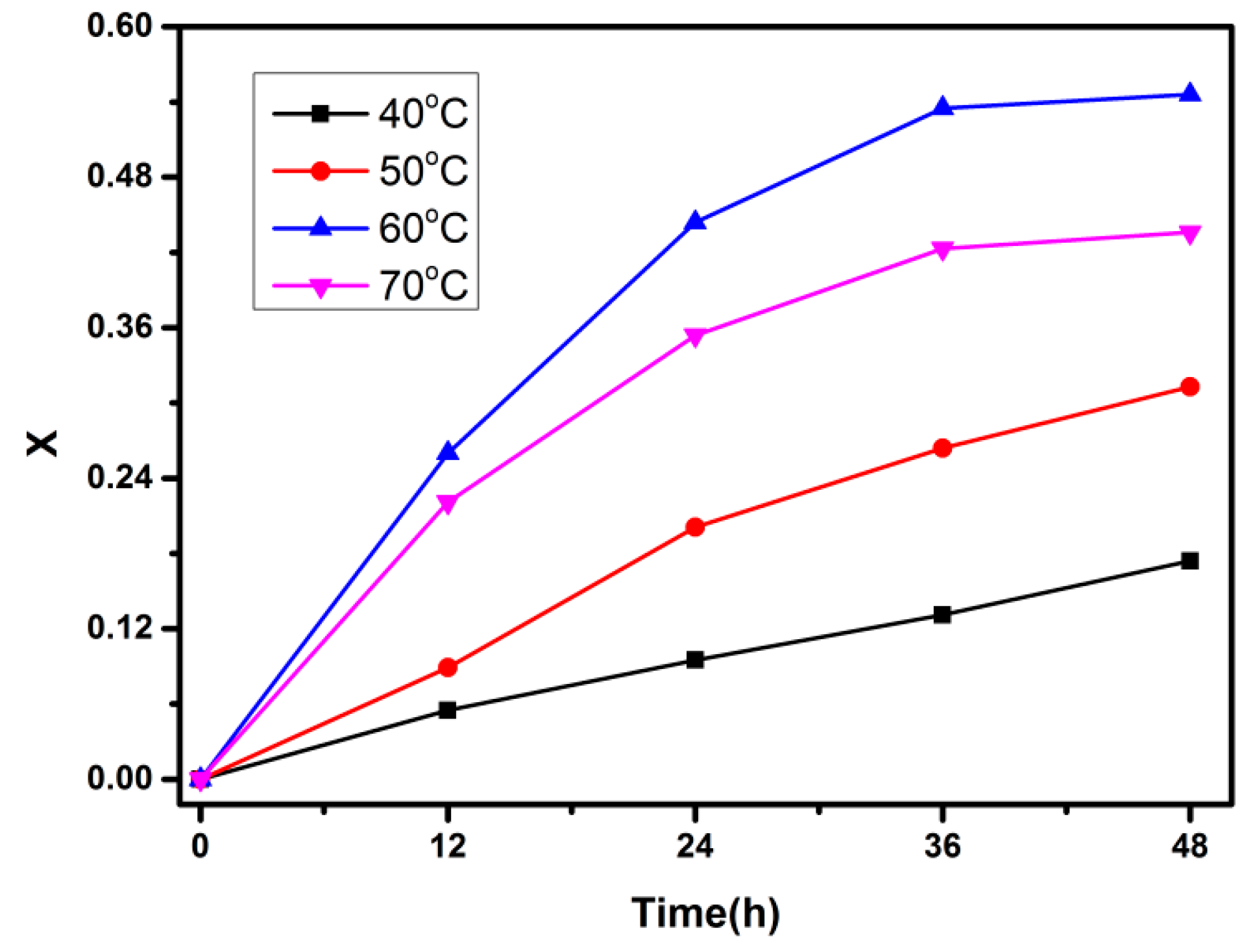
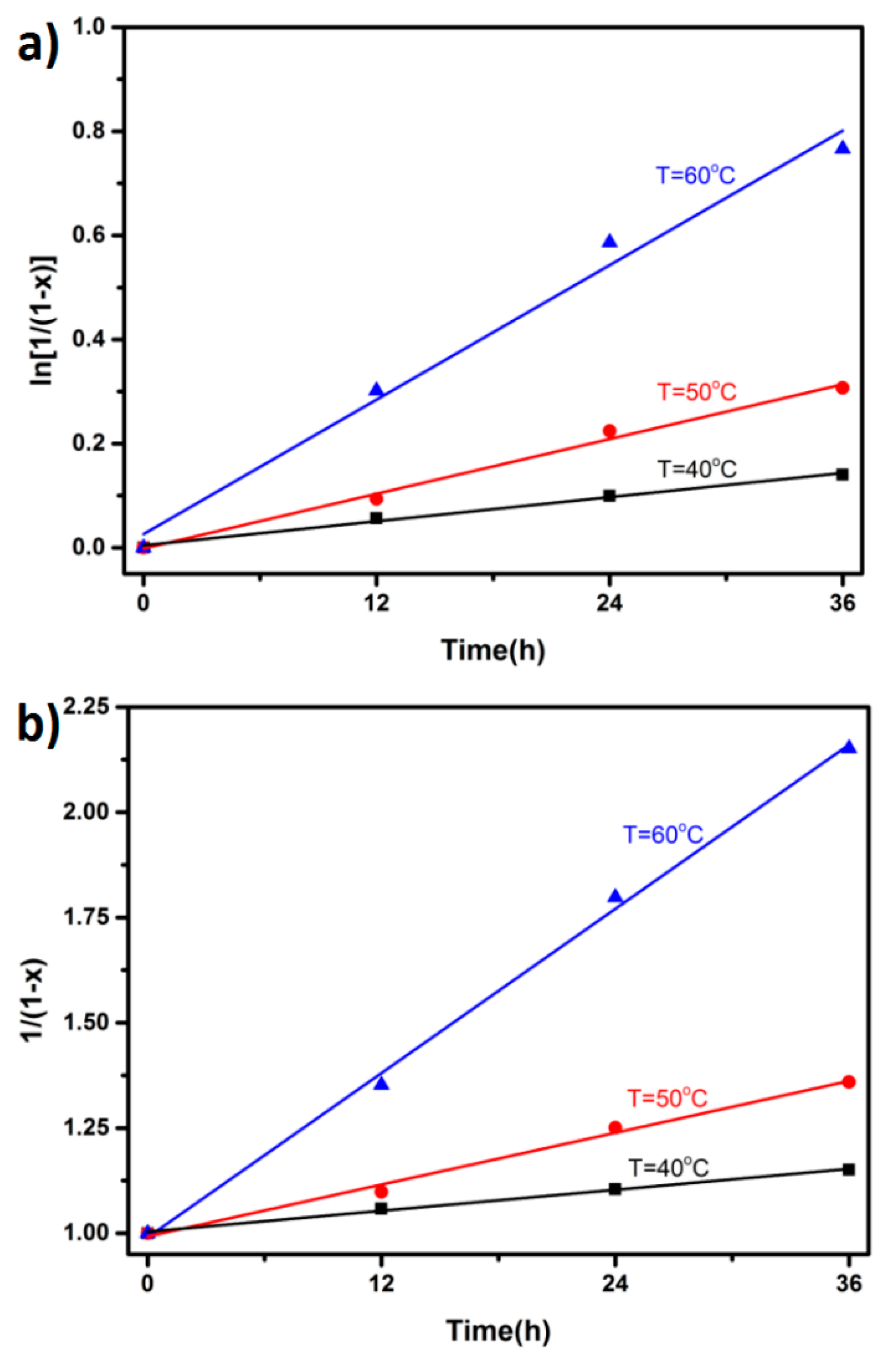
| Kinetic Order | Temperature (°C) | 40 | 50 | 60 |
|---|---|---|---|---|
| n = 1 | k1/min−1 | 0.00396 | 0.00824 | 0.02247 |
| Adj. R-square | 0.99891 | 0.99489 | 0.99337 | |
| n = 2 | k2/(min−1·mol−1·L) | 0.00437 | 0.00973 | 0.03217 |
| Adj. R-square | 0.99999 | 0.99986 | 0.99977 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liang, C.; Li, J.; Xia, M.; Li, G.; Luo, Y. Performance and Kinetics Study of Self-Repairing Hydroxyl-Terminated Polybutadiene Binders Based on the Diels–Alder Reaction. Polymers 2017, 9, 200. https://doi.org/10.3390/polym9060200
Liang C, Li J, Xia M, Li G, Luo Y. Performance and Kinetics Study of Self-Repairing Hydroxyl-Terminated Polybutadiene Binders Based on the Diels–Alder Reaction. Polymers. 2017; 9(6):200. https://doi.org/10.3390/polym9060200
Chicago/Turabian StyleLiang, Chuyao, Jie Li, Min Xia, Guoping Li, and Yunjun Luo. 2017. "Performance and Kinetics Study of Self-Repairing Hydroxyl-Terminated Polybutadiene Binders Based on the Diels–Alder Reaction" Polymers 9, no. 6: 200. https://doi.org/10.3390/polym9060200
APA StyleLiang, C., Li, J., Xia, M., Li, G., & Luo, Y. (2017). Performance and Kinetics Study of Self-Repairing Hydroxyl-Terminated Polybutadiene Binders Based on the Diels–Alder Reaction. Polymers, 9(6), 200. https://doi.org/10.3390/polym9060200





