Adhesives for Achieving Durable Bonds with Acetylated Wood
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bond Strength Study
2.1.1. Experimental Design
2.1.2. Acetylation
2.1.3. Bonding
2.1.4. Adhesive Testing
- Submerged specimens in tap water at room temperature in a pressure vessel.
- Maintained a vacuum of 635 ± 85 kPa for 30 min.
- Maintained a pressure of 448 ± 35 kPa for 30 min.
- Remained submerged in water until tested.
2.1.5. Shear Data Analysis
2.2. Analysis of Hydroxyl Groups on Wood Surface
2.2.1. Treatment with Trifluoroacetic Anhydride
2.2.2. X-ray Photoelectron Analysis
2.2.3. Curve Fitting
3. Results
3.1. Surface Treatment and Analysis
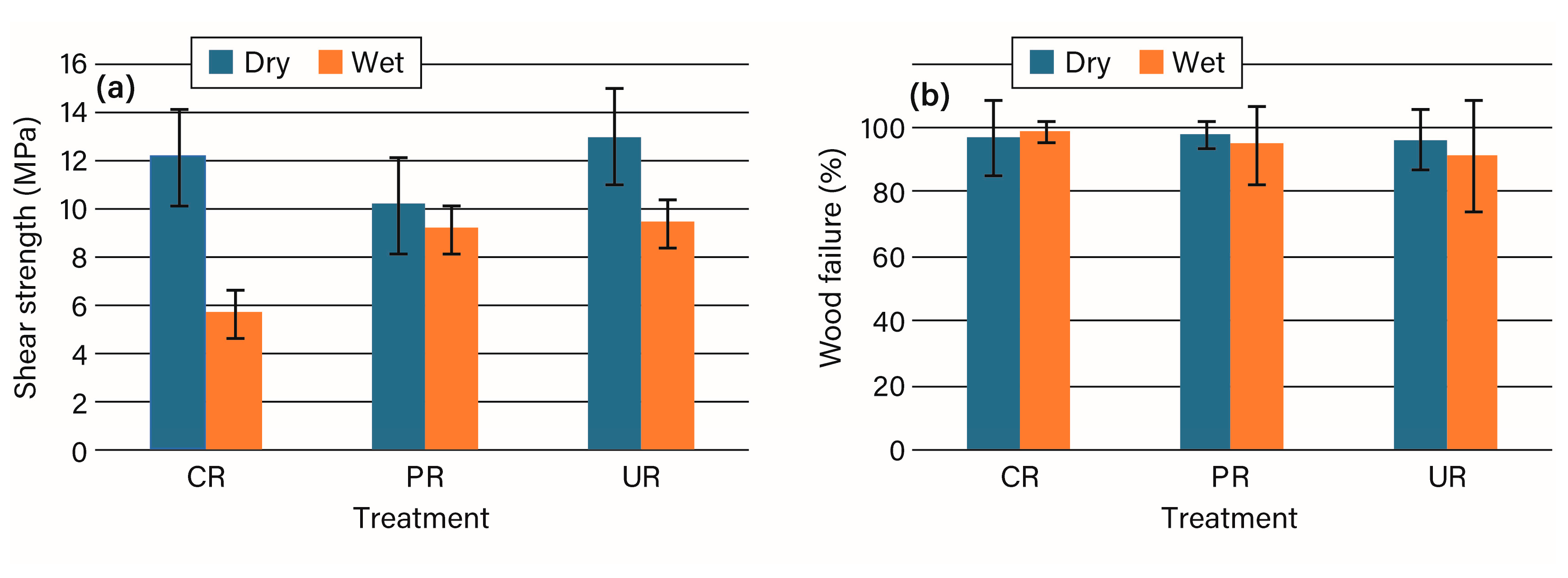
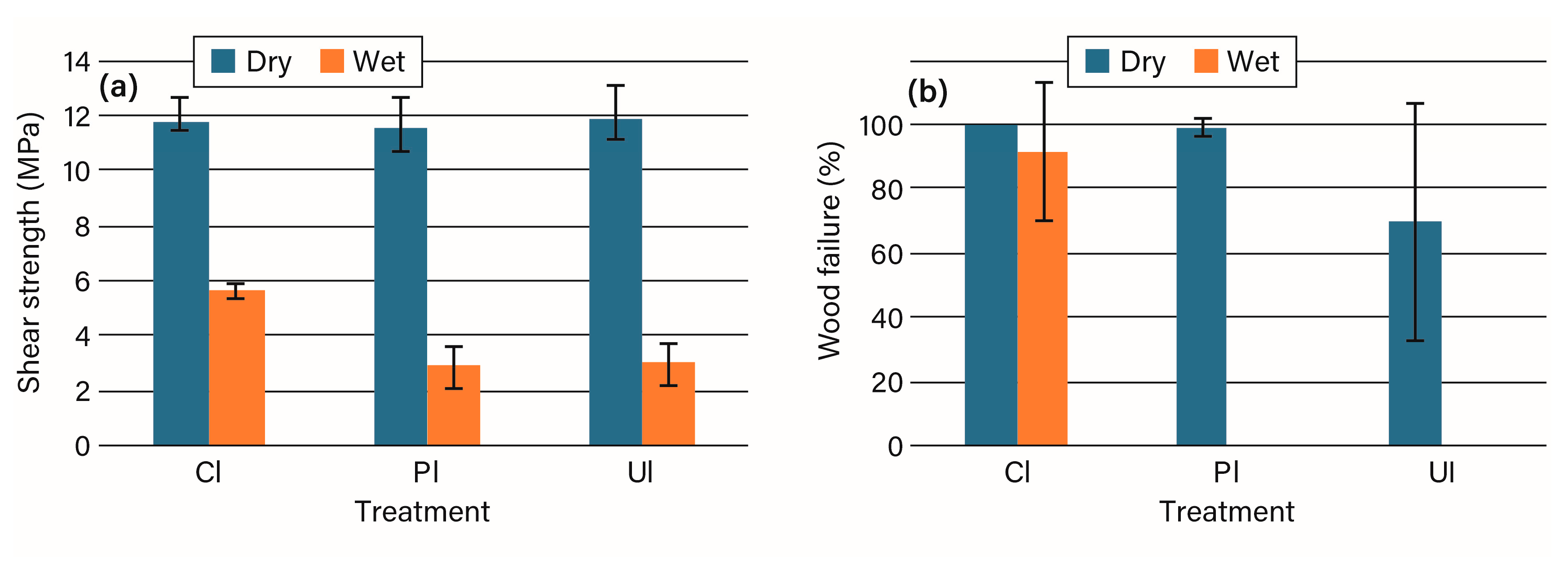
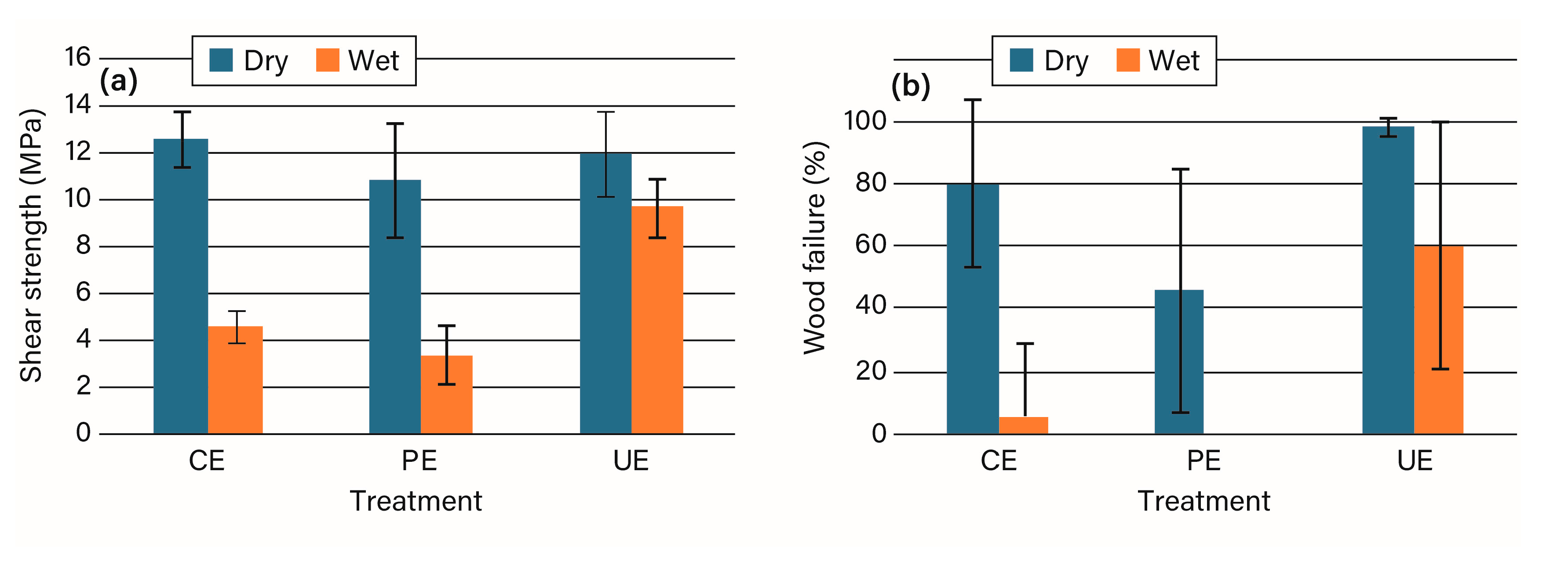
3.2. Bonding Studies
4. Discussion
- As the degree of acetylation increases, the wood absorbs less water.
- ○
- This slows the setting of the waterborne adhesives, leading to overpenetration or disruption of polymer formation of those that cure by condensation (water formation).
- ○
- This also slows the setting of adhesives that need water for curing, such as isocyanates, which is normally provided by adsorbed water for unmodified wood for curing.
- Acetylation bulks up the wood, allowing for less water absorption to occur upon soaking.
- ○
- This reduces the swelling and shrinking of wood as the humidity changes, putting less internal force on the bondline.
- ○
- Upon water soaking, the acetylated wood is not plasticized like the unmodified wood; thus, for wet tests, more of the applied load is transferred to the bondline.
- ○
- This reduces the free volume for the adhesive to enter into the cell wall.
- ○
- This may allow different adhesive components to infiltrate the cell wall compared to the unmodified wood.
- Not all acetylated wood surfaces are the same.
- ○
- Method of acetylation may influence the distribution of acetyl groups, and post-treatment steps, such as washing to remove unreacted acetic anhydride and acetic acid, may extract some wood components.
- ○
- As shown in this study, planing of the surface alters the surface by increasing the surface hydroxyl content.
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Clausen, C.A. Biodeterioration of Wood. In Wood Handbook—Wood as an Engineering Material; General Technical Report FPL-GTR-190; Ross, R.J., Ed.; U.S. Department of Agriculture, Forest Service, Forest Products Laboratory: Madison, WI, USA, 2010; p. 16. [Google Scholar]
- Hill, C.A.S. Wood Modification: Chemcial, Thermal and Other Processes; John Wiley & Sons Ltd.: Chichester, UK, 2006; p. 239. [Google Scholar]
- Rowell, R.M. Chemical modification of wood. In Handbook of Wood Chemistry and Wood Composites; Rowell, R.M., Ed.; CRC Press: Boca Raton, FL, USA, 2013; pp. 537–597. [Google Scholar]
- Vick, C.B.; Rowell, R.M. Adhesive bonding of acetylated wood. Int. J. Adhes. Adhes. 1990, 10, 263–272. [Google Scholar] [CrossRef]
- Bongers, F.; Meijerink, T.; Lütkemeier, B.; Lankveld, C.; Alexander, J.; Militz, H.; Lehringer, C. Bonding of acetylated wood. Int. Wood Prod. J. 2016, 7, 102–106. [Google Scholar] [CrossRef]
- Rowell, R.M.; Youngquist, J.A.; Sachs, I.B. Adhesive bonding of acetylated aspen flakes 1. Surface changes, hydrophobicity, adhesive penetration and strength. Int. J. Adhes. Adhes. 1987, 7, 183–188. [Google Scholar] [CrossRef]
- Youngquist, J.A.; Sachs, I.B.; Rowell, R.M. Adhesive bonding of acetylated aspen flakes 2. Effects of emulsifiers on phenolic resin bonding. Int. J. Adhes. Adhes. 1988, 8, 197–200. [Google Scholar] [CrossRef]
- Vick, C.B.; Larsson, P.C.; Mahlberg, R.L.; Simonson, R.; Rowell, R.M. Structural bonding of acetylated Scandinavian softwoods for exterior lumber laminates. Int. J. Adhes. Adhes. 1993, 13, 139–149. [Google Scholar] [CrossRef]
- Frihart, C.R. Adhesive groups and how they relate to the durability of bonded wood. J. Adhes. Sci. Technol. 2009, 23, 601–617. [Google Scholar] [CrossRef]
- Norimoto, M. Chemical Modification of Wood. In Wood and Cellulose Chemistry; Hon, D.N.S., Shiraishi, N., Eds.; Marcel Dekker: New York, NY, USA, 2001. [Google Scholar]
- ASTM International. D 905-98 Standard Test Method for Strength Properties of Adhesives Bonds in Shear by Compression Loading; ASTM International: West Conshohocken, PA, USA, 1998. [Google Scholar]
- ASTM International. D 2559-12AE1 Standard Specification for Adhesives for Structural Laminated Wood Products for Use under Exterior Exposure Conditions; ASTM International: West Conshohocken, PA, USA, 2011. [Google Scholar]
- ASTM International. Standard Practice for Estimating the Percent Wood Failure in Adhesive Joints; ASTM International: West Conshohocken, PA, USA, 1999. [Google Scholar]
- Beecher, J.F.; Frihart, C.R. X-ray Photoelectron Spectroscopy for Characterization of Wood Surfaces in Adhesion Studies. In Wood Adhesives 2005; Forest Product Society: San Diego, CA, USA, 2006. [Google Scholar]
- Wheeler, D.R.; Pepper, S.V. X-ray photoelectron and mass spectroscopic study of electron irradiation and thermal stability of polytetrafluoroethylene. J. Vac. Sci. Technol. A 1990, 6, 4046–4056. [Google Scholar] [CrossRef]
- Qin, Z.; Gao, Q.; Zhang, S.; Li, J. Surface free energy and dynamic wettability of differently machined poplar woods. BioResources 2014, 9, 3088–3103. [Google Scholar] [CrossRef]
- Larsson, P.; Mahlberg, R.; Vick, C.B.; Simonson, R.; Rowell, R.M. Adhesive bonding of acetylated pine and spruce. In Proceedings of the Pacific Rim Bio-based Composites Symposium; Chemical Modification of Lignocellulosics, Rotorua, New Zealand, 7–8 November 1992; Plackett, D.V., Dunningham, E.A., Eds.; pp. 178–182. [Google Scholar]
- Everhart, D.S.; Reilley, C.N. Chemical derivatization in electron spectroscopy for chemical analysis of surface functional groups introduced on low-density polyethylene film. Anal. Chem. 1981, 33, 665–676. [Google Scholar] [CrossRef]
- Ibach, R.E.; Rowell, R.M.; Lee, B.G. Decay protection based on moisture exclusion resulting from chemical modification of wood. In Proceedings of the 5th Pacific Rim Bio-Based Composites Symposium, Canberra, Australia, 10–13 December 2000. [Google Scholar]
- Obataya, E.; Shibutani, S. Swelling of acetylated wood in organic solvents. J. Mater. Sci. 2005, 40, 4113–4115. [Google Scholar] [CrossRef]
- Frihart, C.R. Are Epoxy-Wood Bonds Durable Enough? In Wood Adhesives 2005; Forest Product Society: San Diego, CA, USA, 2006. [Google Scholar]
- Obataya, E.; Gril, J. Swelling of acetylated wood I. Swelling in organic liquids. J. Wood Sci. 2005, 51, 124–129. [Google Scholar] [CrossRef]
- Hill, C.A.S. Wood modification: An update. BioResources 2011, 6, 918–919. [Google Scholar]
- Yelle, D.J.; Ralph, J. Characterizing phenol—Formaldehyde adhesive cure chemistry within the wood cell wall. Int. J. Adhes. Adhes. 2016, 70, 26–36. [Google Scholar] [CrossRef]
- Frihart, C.R.; Yelle, D.J.; Wiedenhoeft, A.C. What Does Moisture-Related Durability of Wood Bonds Mean? In Proceedings of the COST E34 Workshop on Enhancing Bobdline Performance, Sopron, Hungary, 6–7 May 2008; pp. 89–101. [Google Scholar]
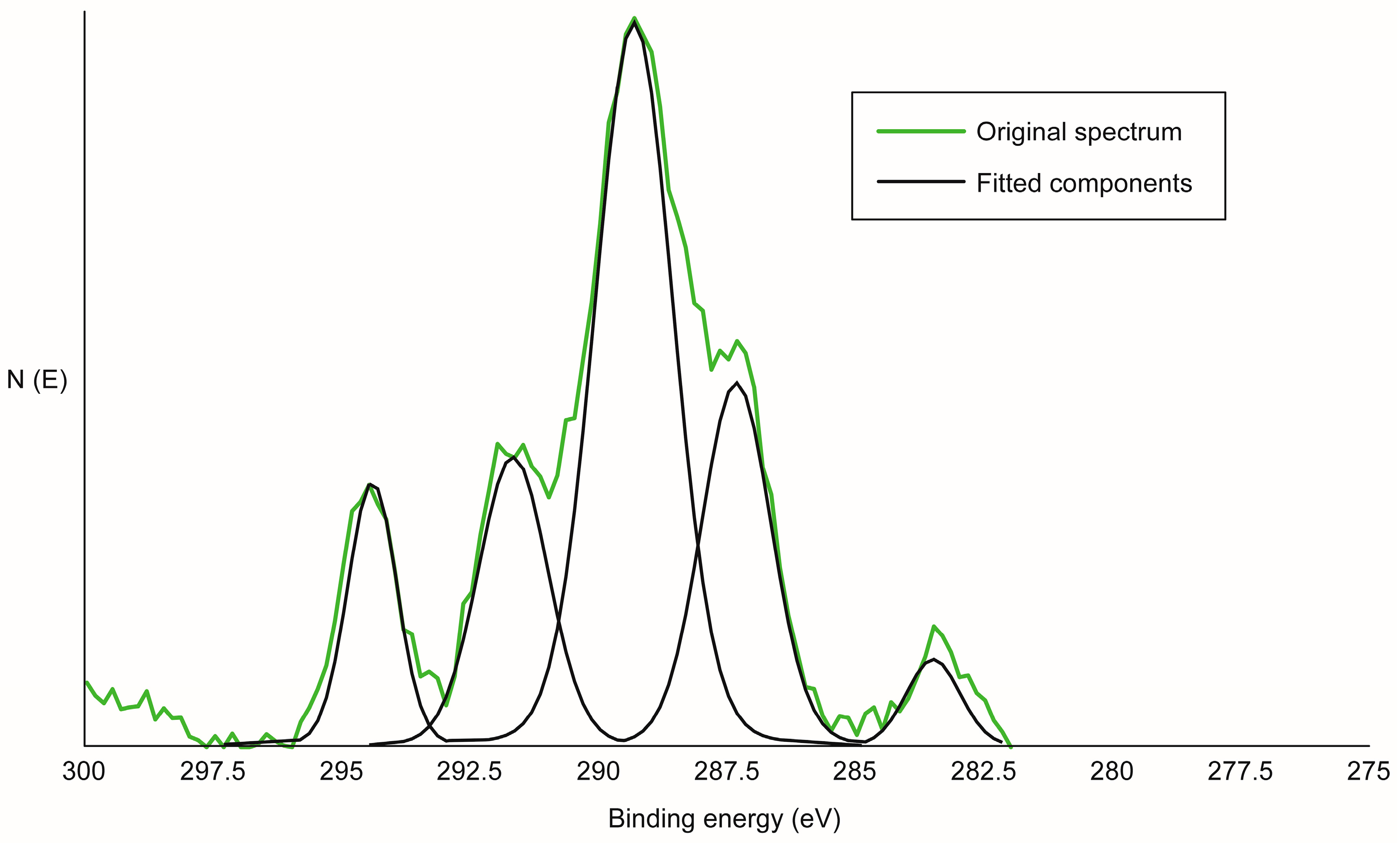
| Specimen | [CF3]/[ΣCarbon] | Free OH Relative to Control |
|---|---|---|
| Yellow-poplar | 0.13 | 1.00 |
| Acetylated poplar | 0.011 | 0.09 |
| Acetylated poplar with planing | 0.054 | 0.42 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Frihart, C.R.; Brandon, R.; Beecher, J.F.; Ibach, R.E. Adhesives for Achieving Durable Bonds with Acetylated Wood. Polymers 2017, 9, 731. https://doi.org/10.3390/polym9120731
Frihart CR, Brandon R, Beecher JF, Ibach RE. Adhesives for Achieving Durable Bonds with Acetylated Wood. Polymers. 2017; 9(12):731. https://doi.org/10.3390/polym9120731
Chicago/Turabian StyleFrihart, Charles R., Rishawn Brandon, James F. Beecher, and Rebecca E. Ibach. 2017. "Adhesives for Achieving Durable Bonds with Acetylated Wood" Polymers 9, no. 12: 731. https://doi.org/10.3390/polym9120731
APA StyleFrihart, C. R., Brandon, R., Beecher, J. F., & Ibach, R. E. (2017). Adhesives for Achieving Durable Bonds with Acetylated Wood. Polymers, 9(12), 731. https://doi.org/10.3390/polym9120731






